A Novel Approach for Enhancing the Terpenoid Content in Wine Using Starmerella bacillaris
Abstract
1. Introduction
2. Materials and Methods
2.1. Yeasts
2.2. Microfermentation
2.3. Microvinifications in CDMM Medium with Glycosidic Aroma Precursors (GAPs)
2.3.1. Microvinifications with Glycosidic Aroma Precursors
2.3.2. Monitoring of Fermentation
2.4. Analysis and Identification of Terpenoids
2.5. β-Glucosidase Activity
2.6. Statistical and Data Analysis
3. Results
3.1. Analysis of Terpenoid Compounds
3.2. Sensory Relevance of the Detected Terpenoid Compounds
3.3. β-Glucosidase Activity and Fermentations in the Presence of GAPs
4. Discussion
4.1. Terpenoid Compounds Generated by S. bacillaris
4.2. Sensory Relevance of the Terpenoid Compounds Generated by S. bacillaris
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Christianson, D.W. Structural and Chemical Biology of Terpenoid Cyclases. Chem. Rev. 2017, 117, 11570–11648. [Google Scholar] [CrossRef]
- Déclaire Mabou, F.; Belinda, I.; Yossa, N. Terpenes: Structural Classification and Biological Activities. IOSR J. Pharm. Biol. Sci. 2021, 16, 25–40. [Google Scholar] [CrossRef]
- Dunlevy, J.D.; Kalua, C.M.; Keyzers, R.A.; Boss, P.K. The Production of Flavour & Aroma Compounds in Grape Berries. In Grapevine Molecular Physiology and Biotechnology, 2nd ed.; Springer: Dordrecht, The Netherlands, 2009; pp. 293–340. [Google Scholar] [CrossRef]
- Chizzola, R. Regular Monoterpenes and Sesquiterpenes (Essential Oils). In Natural Products: Phytochemistry, Botany and Metabolism of Alkaloids, Phenolics and Terpenes; Springer: Berlin/Heidelberg, Germany, 2013; pp. 2973–3008. [Google Scholar] [CrossRef]
- Maicas, S.; Mateo, J.J. Hydrolysis of Terpenyl Glycosides in Grape Juice and Other Fruit Juices: A Review. Appl. Microbiol. Biotechnol. 2005, 67, 322–335. [Google Scholar] [CrossRef] [PubMed]
- Hjelmeland, A.K.; Ebeler, S.E. Glycosidically Bound Volatile Aroma Compounds in Grapes and Wine: A Review. Am. J. Enol. Vitic. 2015, 66, 1–11. [Google Scholar] [CrossRef]
- Mele, M.A.; Kang, H.M.; Lee, Y.T.; Islam, M.Z. Grape Terpenoids: Flavor Importance, Genetic Regulation, and Future Potential. Crit. Rev. Food Sci. Nutr. 2021, 61, 1429–1447. [Google Scholar] [CrossRef]
- Black, C.A.; Parker, M.; Siebert, T.E.; Capone, D.L.; Francis, I.L. Terpenoids and Their Role in Wine Flavour: Recent Advances. Aust. J. Grape Wine Res. 2015, 21, 582–600. [Google Scholar] [CrossRef]
- Carrau, F.M.; Medina, K.; Boido, E.; Farina, L.; Gaggero, C.; Dellacassa, E.; Versini, G.; Henschke, P.A. De Novo Synthesis of Monoterpenes by Saccharomyces cerevisiae Wine Yeasts. FEMS Microbiol. Lett. 2005, 243, 107–115. [Google Scholar] [CrossRef]
- Carrau, F.M.; Boido, E.; Dellacassa, E. Terpenoids in Grapes and Wines: Origin and Micrometabolism during the Vinification Process. Nat. Prod. Commun. 2008, 3, 577–592. [Google Scholar] [CrossRef]
- Drawert, F.; Barton, H. Biosynthesis of Flavor Compounds by Microorganisms. 3. Production of Monoterpenes by the Yeast Kluyveromyces Lactis. J. Agric. Food Chem. 1978, 26, 765–766. [Google Scholar] [CrossRef]
- Hock, R.; Benda, I.; Schreier, P. Formation of Terpenes by Yeasts during Alcoholic Fermentation. Z. Lebensm.-Unters.-Forsch. 1984, 179, 450–452. [Google Scholar] [CrossRef]
- Klingenberg, A.; Sprecher, E. Production of Monoterpenes in Liquid Cultures by the Yeast Ambrosiozyma monospora. Planta Med. 1985, 51, 264–265. [Google Scholar] [CrossRef]
- Morata, A.; Escott, C.; Bañuelos, M.A.; Loira, I.; Del Fresno, J.M.; González, C.; Suárez-lepe, J.A. Contribution of Non-Saccharomyces Yeasts to Wine Freshness. A Review. Biomolecules 2020, 10, 34. [Google Scholar] [CrossRef]
- Ferreira, A.M.; Clímaco, M.C.; Faia, A.M. The Role of Non-Saccharomyces Species in Releasing Glycosidic Bound Fraction of Grape Aroma Components—A Preliminary Study. J. Appl. Microbiol. 2001, 91, 67–71. [Google Scholar] [CrossRef]
- López, S.; Mateo, J.; Maicas, S. Characterisation of Hanseniaspora Isolates with Potential Aroma-Enhancing Properties in Muscat Wines. S. Afr. J. Enol. Vitic. 2014, 35, 292–303. [Google Scholar] [CrossRef][Green Version]
- Beckner Whitener, M.E.; Stanstrup, J.; Panzeri, V.; Carlin, S.; Divol, B.; Du Toit, M.; Vrhovsek, U. Untangling the Wine Metabolome by Combining Untargeted SPME–GCxGC-TOF-MS and Sensory Analysis to Profile Sauvignon Blanc Co-Fermented with Seven Different Yeasts. Metabolomics 2016, 12, 53. [Google Scholar] [CrossRef]
- Englezos, V.; Rantsiou, K.; Cravero, F.; Torchio, F.; Giacosa, S.; Ortiz-Julien, A.; Gerbi, V.; Rolle, L.; Cocolin, L. Volatile Profiles and Chromatic Characteristics of Red Wines Produced with Starmerella bacillaris and Saccharomyces cerevisiae. Food Res. Int. 2018, 109, 298–309. [Google Scholar] [CrossRef]
- Sadoudi, M.; Tourdot-Maréchal, R.; Rousseaux, S.; Steyer, D.; Gallardo-Chacón, J.; Ballester, J.; Vichi, S.; Guérin-Schneider, R.; Caixach, J.; Alexandre, H. Yeast-Yeast Interactions Revealed by Aromatic Profile Analysis of Sauvignon Blanc Wine Fermented by Single or Co-Culture of Non-Saccharomyces and Saccharomyces Yeasts. Food Microbiol. 2012, 32, 243–253. [Google Scholar] [CrossRef]
- Mills, D.A.; Johannsen, E.A.; Cocolin, L. Yeast Diversity and Persistence in Botrytis-Affected Wine Fermentations. Appl. Environ. Microbiol. 2002, 68, 4884–4893. [Google Scholar] [CrossRef] [PubMed]
- Duarte, F.L.; Pimentel, N.H.; Teixeira, A.; Fonseca, A. Saccharomyces bacillaris Is Not a Synonym of Candida stellata: Reinstatement as Starmerella bacillaris Comb. Nov. Antonie Van Leeuwenhoek Int. J. Gen. Mol. Microbiol. 2012, 102, 653–658. [Google Scholar] [CrossRef] [PubMed]
- Englezos, V.; Torchio, F.; Cravero, F.; Marengo, F.; Giacosa, S.; Gerbi, V.; Rantsiou, K.; Rolle, L.; Cocolin, L. Aroma Profile and Composition of Barbera Wines Obtained by Mixed Fermentations of Starmerella bacillaris (Synonym Candida zemplinina) and Saccharomyces cerevisiae. LWT 2016, 73, 567–575. [Google Scholar] [CrossRef]
- Tufariello, M.; Fragasso, M.; Pico, J.; Panighel, A.; Castellarin, S.D.; Flamini, R.; Grieco, F. Influence of Non-Saccharomyces on Wine Chemistry: A Focus on Aroma-Related Compounds. Molecules 2021, 26, 644. [Google Scholar] [CrossRef]
- Lemos Junior, W.J.F.; Treu, L.; Nadai, C.; da Silva Duarte, V.; Campanaro, S.; Fabrega-Prats, M.; Giacomini, A.; Corich, V. Genomic Insights into the Glutathione Metabolism of the Wine Yeast Starmerella bacillaris. Oeno One 2021, 55, 105–117. [Google Scholar] [CrossRef]
- García, M.; Esteve-Zarzoso, B.; Cabellos, J.M.; Arroyo, T. Advances in the Study of Candida stellata. Fermentation 2018, 4, 74. [Google Scholar] [CrossRef]
- Englezos, V.; Giacosa, S.; Rantsiou, K.; Rolle, L.; Cocolin, L. Starmerella bacillaris in Winemaking: Opportunities and Risks. Curr. Opin. Food Sci. 2017, 17, 30–35. [Google Scholar] [CrossRef]
- Rosa, A.L.; Miot-Sertier, C.; Laizet, Y.; Salin, F.; Sipiczki, M.; Bely, M.; Masneuf-Pomarede, I.; Albertin, W. Draft Genome Sequence of the Starmerella bacillaris (Syn., Candida zemplinina) Type Strain CBS 9494. Microbiol. Resour. Announc. 2018, 7, e00872-18. [Google Scholar] [CrossRef]
- Raymond Eder, M.L.; Rosa, A.L. Genetic, Physiological, and Industrial Aspects of the Fructophilic Non-Saccharomyces Yeast Species, Starmerella bacillaris. Fermentation 2021, 7, 87. [Google Scholar] [CrossRef]
- Tufariello, M.; Capozzi, V.; Spano, G.; Cantele, G.; Venerito, P.; Mita, G.; Grieco, F. Effect of Co-Inoculation of Candida zemplinina, Saccharomyces cerevisiae and Lactobacillus plantarum for the Industrial Production of Negroamaro Wine in Apulia (Southern Italy). Microorganisms 2020, 8, 726. [Google Scholar] [CrossRef]
- Martin, V.; Giorello, F.; Fariña, L.; Minteguiaga, M.; Salzman, V.; Boido, E.; Aguilar, P.S.; Gaggero, C.; Dellacassa, E.; Mas, A.; et al. De Novo Synthesis of Benzenoid Compounds by the Yeast Hanseniaspora vineae Increases the Flavor Diversity of Wines. J. Agric. Food Chem. 2016, 64, 4574–4583. [Google Scholar] [CrossRef] [PubMed]
- Massera, A.; Soria, A.; Catania, C.; Krieger, S.; Combina, M. Simultaneous Inoculation of Malbec (Vitis vinifera) Musts with Yeast and Bacteria: Effects on Fermentation Performance, Sensory and Sanitary Attributes of Wines. Food Technol. Biotechnol. 2009, 47, 192–201. [Google Scholar]
- Boido, E.; Lloret, L.; Medina, K.; Fariña, L.; Carrau, F.; Versini, G.; Dellacassa, E. Aroma Composition of Vitis vinifera Cv. Tannat: The Typical Red Wine from Uruguay. J. Agric. Food Chem. 2003, 51, 5408–5413. [Google Scholar] [CrossRef] [PubMed]
- Carrau, F.M.; Medina, K.; Farina, L.; Boido, E.; Henschke, P.A.; Dellacassa, E. Production of Fermentation Aroma Compounds by Saccharomyces cerevisiae Wine Yeasts: Effects of Yeast Assimilable Nitrogen on Two Model Strains. FEMS Yeast Res. 2008, 8, 1196–1207. [Google Scholar] [CrossRef]
- Pallmann, C.L.; Brown, J.A.; Olineka, T.L.; Cocolin, L.; Mills, D.A.; Bisson, L.F. Use of WL Medium to Profile Native Flora Fermentations. Am. J. Enol. Vitic. 2001, 52, 198–203. [Google Scholar] [CrossRef]
- Fariña, L.; Boido, E.; Ares, G.; Gonzalez, N.; Lado, J.; Curbelo, R.; Almeida, L.; Medina, K.; Carrau, F.; Dellacassa, E. Solid Phase Microextraction for the Characterization of Food Aroma and Particular Sensory Defects. In Flavors and Fragrances in Food Processing: Preparation and Characterization Methods; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2022; Volume 1433, pp. 299–325. ISBN 9780841297319. [Google Scholar]
- Pérez, G.; Fariña, L.; Barquet, M.; Boido, E.; Gaggero, C.; Dellacassa, E.; Carrau, F. A Quick Screening Method to Identify β-Glucosidase Activity in Native Wine Yeast Strains: Application of Esculin Glycerol Agar (EGA) Medium. World J. Microbiol. Biotechnol. 2011, 27, 47–55. [Google Scholar] [CrossRef]
- Aznar, M.; Lopez, R.; Cacho, J.; Ferreira, V. Prediction of aged red wine aroma properties from aroma chemical composition. Partial least squares regression models. J. Agric. Food Chem. 2003, 51, 2700–2707. [Google Scholar] [CrossRef]
- Ferreira, V.; López, R.; Cacho, J.F. Quantitative Determination of the Odorants of Young Red Wines from Different Grape Varieties. J. Sci. Food Agric. 2002, 80, 1659–1667. [Google Scholar] [CrossRef]
- Condurso, C.; Cincotta, F.; Tripodi, G.; Sparacio, A.; Giglio, D.M.L.; Sparla, S.; Verzera, A. Effects of Cluster Thinning on Wine Quality of Syrah Cultivar (Vitis vinifera L.). Eur. Food Res. Technol. 2016, 242, 1719–1726. [Google Scholar] [CrossRef]
- Zea, L.; Moyano, L.; Moreno, J.; Cortes, B.; Medina, M. Discrimination of the Aroma Fraction of Sherry Wines Obtained by Oxidative and Biological Ageing. Food Chem. 2001, 75, 79–84. [Google Scholar] [CrossRef]
- Englezos, V.; Rantsiou, K.; Torchio, F.; Rolle, L.; Gerbi, V.; Cocolin, L. Exploitation of the Non-Saccharomyces Yeast Starmerella bacillaris (Synonym Candida zemplinina) in Wine Fermentation: Physiological and Molecular Characterizations. Int. J. Food Microbiol. 2015, 199, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Fariña, L.; Villar, V.; Ares, G.; Carrau, F.; Dellacassa, E.; Boido, E. Volatile Composition and Aroma Profile of Uruguayan Tannat Wines. Food Res. Int. 2015, 69, 244–255. [Google Scholar] [CrossRef]
- Ninkuu, V.; Zhang, L.; Yan, J.; Fu, Z.; Yang, T.; Zeng, H. Biochemistry of Terpenes and Recent Advances in Plant Protection. Int. J. Mol. Sci. 2021, 22, 5710. [Google Scholar] [CrossRef]
- Maury, J.; Asadollahi, M.A.; Møller, K.; Clark, A.; Nielsen, J. Microbial Isoprenoid Production: An Example of Green Chemistry through Metabolic Engineering. Adv. Biochem. Eng. Biotechnol. 2005, 100, 19–51. [Google Scholar]
- Zhao, J.; Li, C.; Zhang, Y.; Shen, Y.; Hou, J.; Bao, X. Dynamic Control of ERG20 Expression Combined with Minimized Endogenous Downstream Metabolism Contributes to the Improvement of Geraniol Production in Saccharomyces cerevisiae. Microb. Cell Fact. 2017, 16, 17. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.Z.; Yao, M.D.; Wang, Y.; Zhou, L.; Song, T.Q.; Liu, H.; Xiao, W.H.; Yuan, Y.J. Manipulation of GES and ERG20 for Geraniol Overproduction in Saccharomyces cerevisiae. Metab. Eng. 2017, 41, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Bao, X.; Li, C.; Shen, Y.; Hou, J. Improving Monoterpene Geraniol Production through Geranyl Diphosphate Synthesis Regulation in Saccharomyces cerevisiae. Appl. Microbiol. Biotechnol. 2016, 100, 4561–4571. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Bai, X.; Zhou, X.; Wang, J.; Guo, N.; Deng, Y. Whole-Genome Analysis of Starmerella bacillaris CC-PT4 against MRSA, a Non-Saccharomyces Yeast Isolated from Grape. J. Fungi 2022, 8, 1255. [Google Scholar] [CrossRef]
- Lemos Junior, W.J.F.; Duarte, V.d.S.; Treu, L.; Campanaro, S.; Nadai, C.; Giacomini, A.; Corich, V. Whole Genome Comparison of Two Starmerella bacillaris Strains with Other Wine Yeasts Uncovers Genes Involved in Modulating Important Winemaking Traits. FEMS Yeast Res. 2018, 18, foy069. [Google Scholar] [CrossRef]
- Jordá, T.; Puig, S. Regulation of Ergosterol Biosynthesis in Saccharomyces cerevisiae. Genes 2020, 11, 795. [Google Scholar] [CrossRef]
- Keller, N.P. Fungal Secondary Metabolism: Regulation, Function and Drug Discovery. Nat. Rev. Microbiol. 2019, 17, 167–180. [Google Scholar] [CrossRef]
- Scariot, F.J.; Pansera, M.S.; Delamare, A.P.L.; Echeverrigaray, S. Citral and Geraniol Induce Necrotic and Apoptotic Cell Death on Saccharomyces cerevisiae. World J. Microbiol. Biotechnol. 2021, 37, 42. [Google Scholar] [CrossRef]
- Scariot, F.J.; Pansera, M.S.; Longaray Delamare, A.P.; Echeverrigaray, S. Antifungal Activity of Monoterpenes against the Model Yeast Saccharomyces cerevisiae. J. Food Process Preserv. 2021, 45, e15433. [Google Scholar] [CrossRef]
- Nickerson, K.W.; Atkin, A.L.; Hornby, J.M. Quorum Sensing in Dimorphic Fungi: Farnesol and Beyond. Appl. Environ. Microbiol. 2006, 72, 3805–3813. [Google Scholar] [CrossRef]
- Steyer, D.; Erny, C.; Claudel, P.; Riveill, G.; Karst, F.; Legras, J.L. Genetic Analysis of Geraniol Metabolism during Fermentation. Food Microbiol. 2013, 33, 228–234. [Google Scholar] [CrossRef]
- King, A.; Dickinson, J.R. Biotransformation of Monoterpene Alcohols by Saccharomyces cerevisiae, Torulaspora delbrueckii and Kluyveromyces lactis. Yeast 2000, 16, 499–506. [Google Scholar] [CrossRef]
- Steyer, D.; Ambroset, C.; Brion, C.; Claudel, P.; Delobel, P.; Sanchez, I.; Erny, C.; Blondin, B.; Karst, F.; Legras, J.-L. QTL Mapping of the Production of Wine Aroma Compounds by Yeast. BMC Genom. 2012, 13, 573. [Google Scholar] [CrossRef]
- Chauhan, N.M.; Mohan Karuppayil, S. Dual Identities for Various Alcohols in Two Different Yeasts. Mycology 2021, 12, 25–38. [Google Scholar] [CrossRef]
- Giungato, P.; Di Gilio, A.; Palmisani, J.; Marzocca, A.; Mazzone, A.; Brattoli, M.; Giua, R.; de Gennaro, G. Synergistic Approaches for Odor Active Compounds Monitoring and Identification: State of the Art, Integration, Limits and Potentialities of Analytical and Sensorial Techniques. TrAC—Trends Anal. Chem. 2018, 107, 116–129. [Google Scholar] [CrossRef]
- Ren, L.; Hu, Y.; Liang, W. A Comprehensive Review and Variability Analysis of Odor Detection Thresholds and Their Influence on Building Material Odor Assessments. Build. Environ. 2025, 280, 113122. [Google Scholar] [CrossRef]
- Santana, G.R.O.; Crepalde, L.T.; Hernandes, K.C.; Silveira, R.D.; Padilha, G.; Reitenbach, A.F.; de Souza, A.L.K.; Welke, J.E.; Caliari, V.; Burin, V.M. Impact of Skin Contact on the Volatile Composition and Sensory Properties of White Wines from Resistant Varieties. Food Chem. 2025, 489, 144967. [Google Scholar] [CrossRef]
- Chigo-Hernandez, M.M.; Dubois, A.; Tomasino, E. Aroma Perception of Rose Oxide, Linalool and α-Terpineol Combinations in Gewürztraminer Wine. Fermentation 2022, 8, 30. [Google Scholar] [CrossRef]
- Darnal, A.; Ceccon, A.; Magni, M.; Robatscher, P.; Poggesi, S.; Boselli, E.; Longo, E. Enantioselective-GCxGC Determination of α-Terpinyl Ethyl Ether in Wine: Quantitative Analysis and Identification of Main Terpene Precursors. Appl. Food Res. 2024, 4, 100538. [Google Scholar] [CrossRef]
- Chen, H.; Bai, S.; Yang, B.; Ren, R.; Tang, Z.; Zhang, Z.; Zeng, Q. Inter- and Intra-Varietal Clonal Differences Influence the Aroma Compound Profiles of Wines Analyzed by GC–MS and GC-IMS. Food Chem. X 2025, 25, 102136. [Google Scholar] [CrossRef]
- Garbay, J.; Tempere, S.; Ballet, L.; Barbe, J.-C.; Lytra, G. Investigating the Olfactory Impact of Monoterpenic Compounds on Fruity Aroma Perception in Model Red Wine Solutions. J. Agric. Food Chem. 2025, 73, 4208–4218. [Google Scholar] [CrossRef]
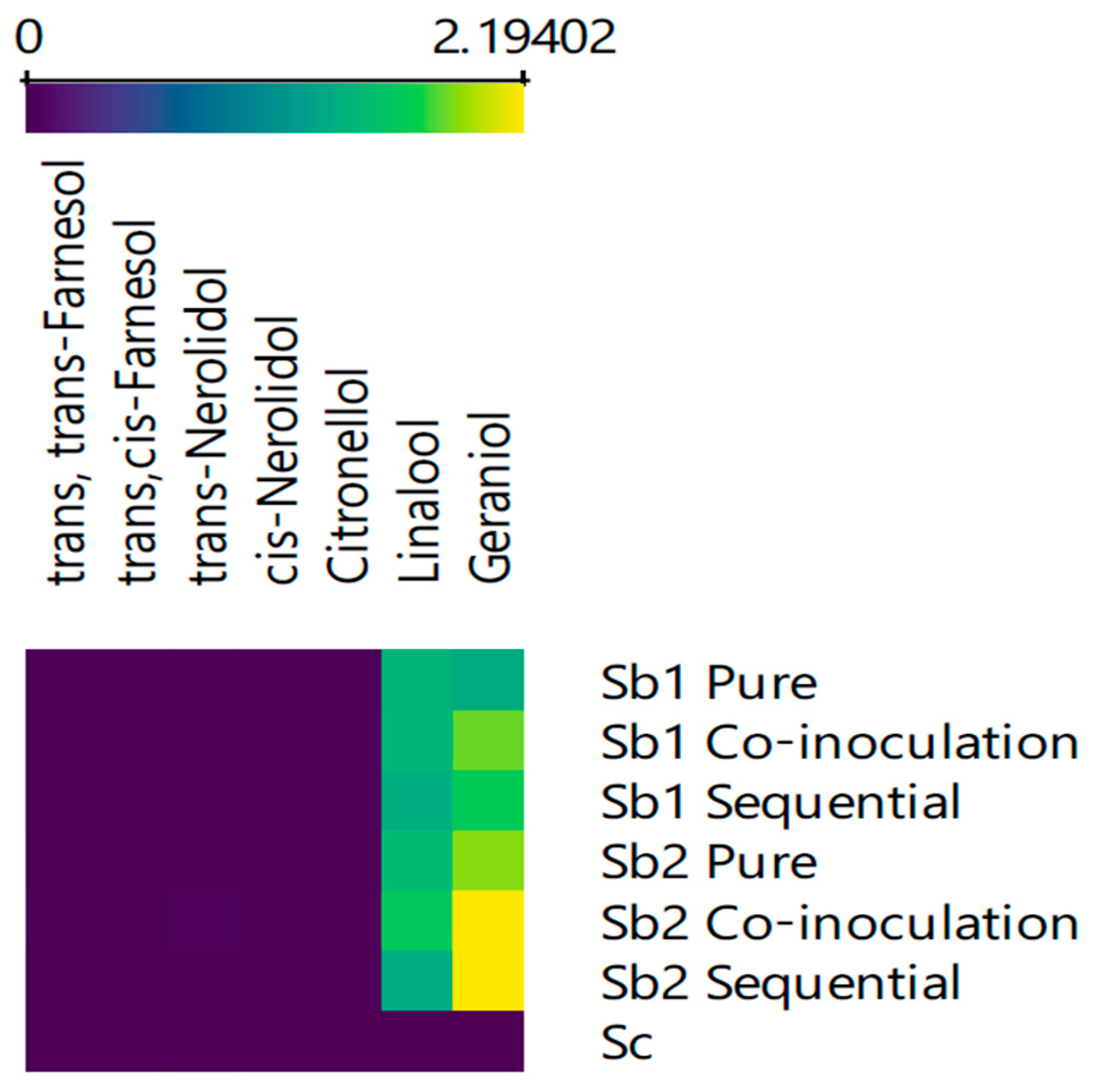
| Treatments | Type of Inoculum | S. bacillaris Inoculum Size (Cells/mL) * | S. cerevisiae Inoculum Size (Cells/mL) * |
|---|---|---|---|
| A | Pure strains (Sb1, Sb2, and Sc) | 1 × 106 | 1 × 106 |
| B | Sequential (48 h) | 1 × 106 | 1 × 106 |
| C | Co-inoculated | 1 × 106 | 1 × 105 |
| Strains and Inoculation Strategy | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| SC | Sb1 | Sb2 | ||||||||
| KI | Compounds (mg/L) | Pure | Co- Inoculation | Sequential | Pure | Co- Inoculation | Sequential | Odour Threshold (μg/L) | Odour Descriptor | |
| 1860 | Geraniol | nd | 26.7 ± 1.3 a | 37.0 ± 3.5 c | 33.3 ± 2.0 b | 37.9 ± 4.8 c | 43.9 ± 1.6 c | 43.9 ± 14.6 c | 20 [37] | Rose and geranium |
| 1558 | Linalool | nd | 35.9 ± 3.8 a,b | 36.3 ± 7.7 a,b | 34.6 ± 4.3 a,b | 37.7 ± 1.9 a,b | 41.3 ± 2.8 b | 34.3 ± 5.2 a | 25.2 [38] | Citrus, floral, lavender |
| 1765 | Citronellol | nd | nd | 4.1 ± 1.5 b | 2.6 ± 0.9 a,b | nd | 3.1 ± 0.5 a,b | 2.4 ± 0.1 a | 100 [39] | Citronella, rose, green |
| 2025 | cis- Nerolidol | nd | 6.4 ± 0.1 a | 7.6 ± 2.3 a | nd | nd | 6.6 ± 0.1 a | nd | 700 [39] | Floral, green, citrus, woody |
| 2042 | trans-Nerolidol | 12.4 ± 0.7 a | 20.6 ± 4.2 b | 22.2 ± 3.6 b | 13.8 ± 2.8 a | 20.5 ± 1.7 b | 32.8 ± 1.8 c | 12.6 ± 1.5 a | 700 [39] | Floral and woody |
| 2331 | trans,cis- Farnesol | nd | 1.7 ± 0.7 a | nd | nd | 5.9 ± 0.1 b | nd | nd | 1000 [40] | Sweet floral |
| 2350 | trans,trans-Farnesol | 3.4 ± 1.8 a | 7.9 ± 2.6 a | 8.6 ± 0.9 a | nd | 13.3 ± 4.4 b | 26.3 ± 1.6 c | nd | 1000 [40] | Sweet floral |
| Compounds (μg/L) | |||||||
|---|---|---|---|---|---|---|---|
| Strains | Linalool | Geraniol | Citronellol | cis-Nerolidol | trans-Nerolidol | trans,cis- Farnesol | trans,trans- Farnesol |
| Sc | nd | nd | nd | nd | 26.0 ± 4.8 | nd | 23.8 ± 13.0 c |
| Sb1 | 37.1 ± 3.8 a | 44.4 ± 4.9 b | nd | nd | 23.9 ± 5.5 | 1.3 ± 0.6 a | 6.3 ± 0.7 b |
| Sb2 | 40.1 ± 5.8 a | 48.2 ± 13.1 b | nd | nd | 33.4 ± 8.0 | nd | 11.8 ± 5.2 b,c |
| Control | nd | nd | nd | nd | nd | nd | nd |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Listur, M.B.; Martín, V.; Medina, K.; Carrau, F.; Boido, E.; Dellacassa, E.; Fariña, L. A Novel Approach for Enhancing the Terpenoid Content in Wine Using Starmerella bacillaris. Fermentation 2025, 11, 496. https://doi.org/10.3390/fermentation11090496
Listur MB, Martín V, Medina K, Carrau F, Boido E, Dellacassa E, Fariña L. A Novel Approach for Enhancing the Terpenoid Content in Wine Using Starmerella bacillaris. Fermentation. 2025; 11(9):496. https://doi.org/10.3390/fermentation11090496
Chicago/Turabian StyleListur, María Belén, Valentina Martín, Karina Medina, Francisco Carrau, Eduardo Boido, Eduardo Dellacassa, and Laura Fariña. 2025. "A Novel Approach for Enhancing the Terpenoid Content in Wine Using Starmerella bacillaris" Fermentation 11, no. 9: 496. https://doi.org/10.3390/fermentation11090496
APA StyleListur, M. B., Martín, V., Medina, K., Carrau, F., Boido, E., Dellacassa, E., & Fariña, L. (2025). A Novel Approach for Enhancing the Terpenoid Content in Wine Using Starmerella bacillaris. Fermentation, 11(9), 496. https://doi.org/10.3390/fermentation11090496









