Recent Advances in Seeded and Seed-Layer-Free Atomic Layer Deposition of High-K Dielectrics on Graphene for Electronics
Abstract
1. Introduction
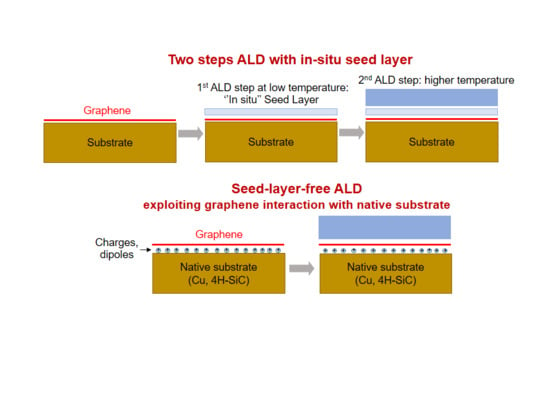
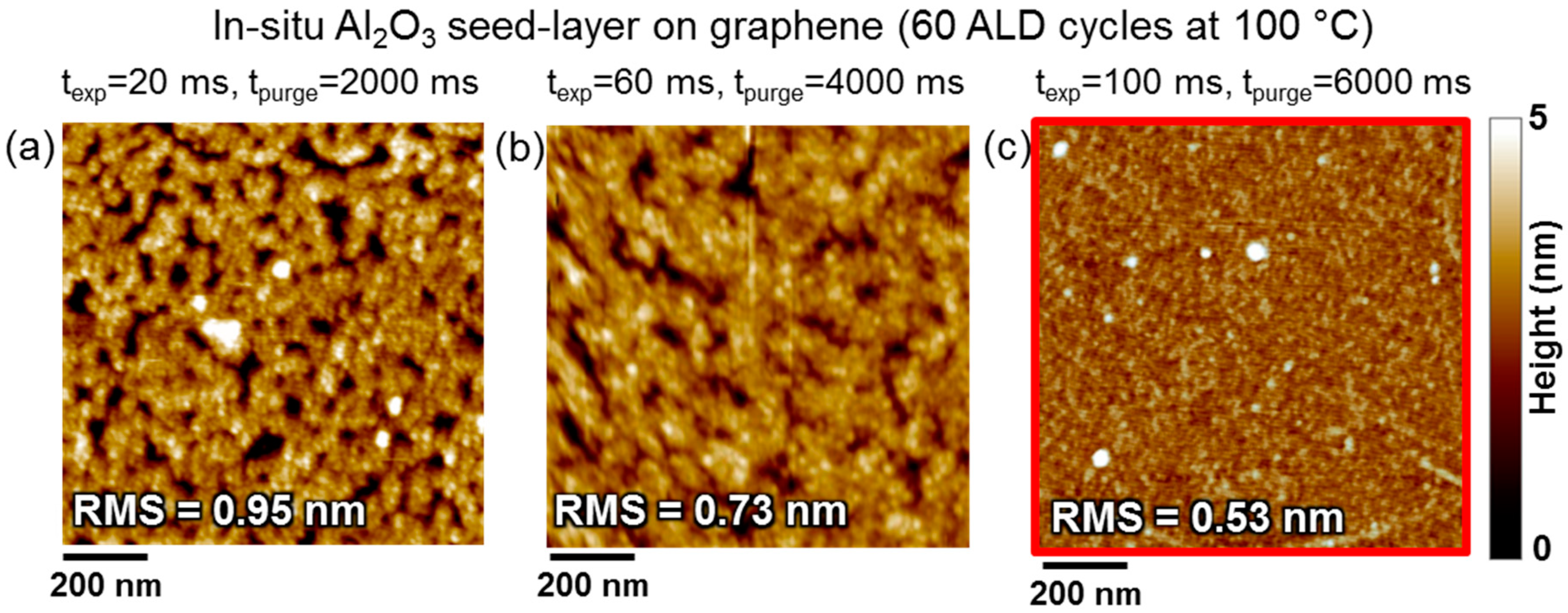
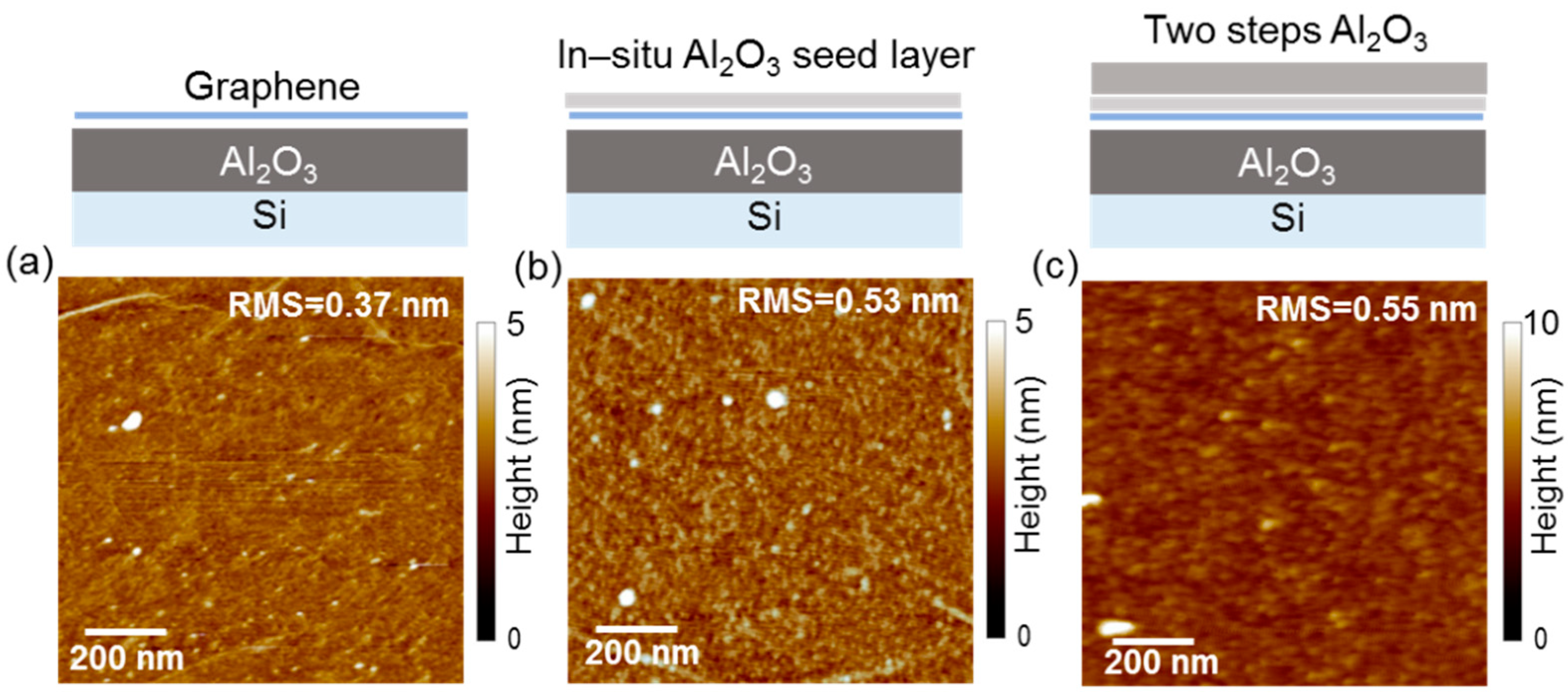
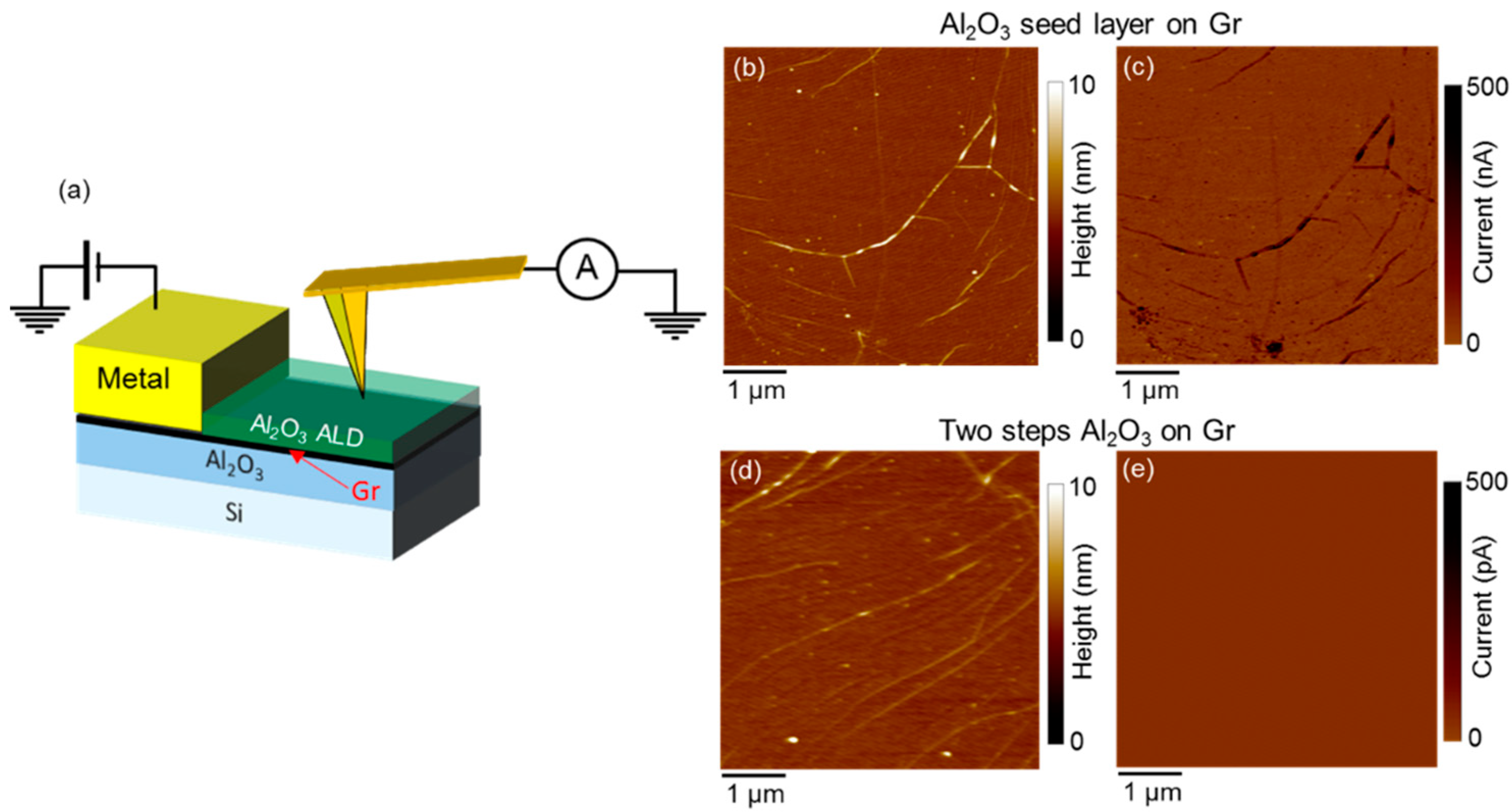
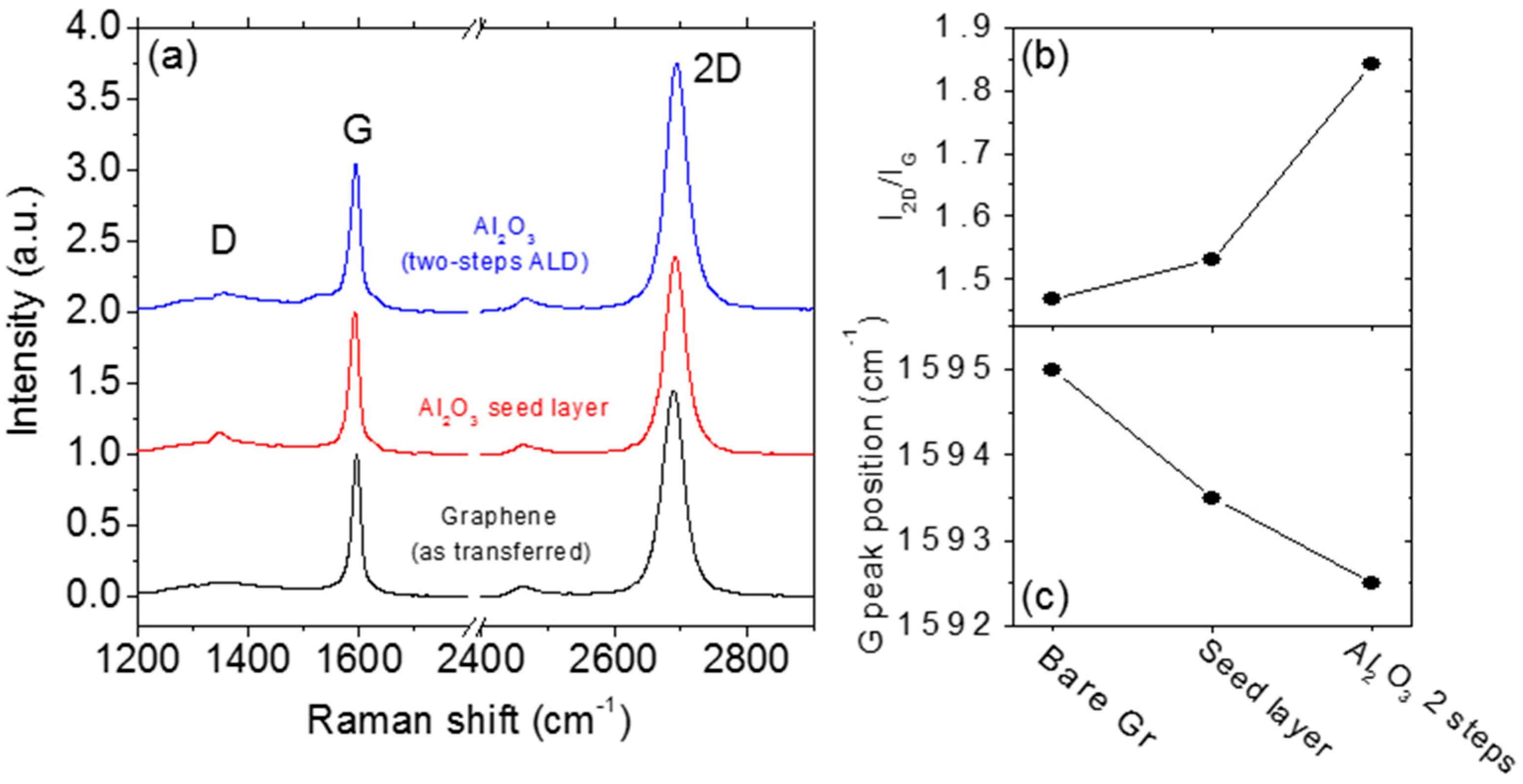
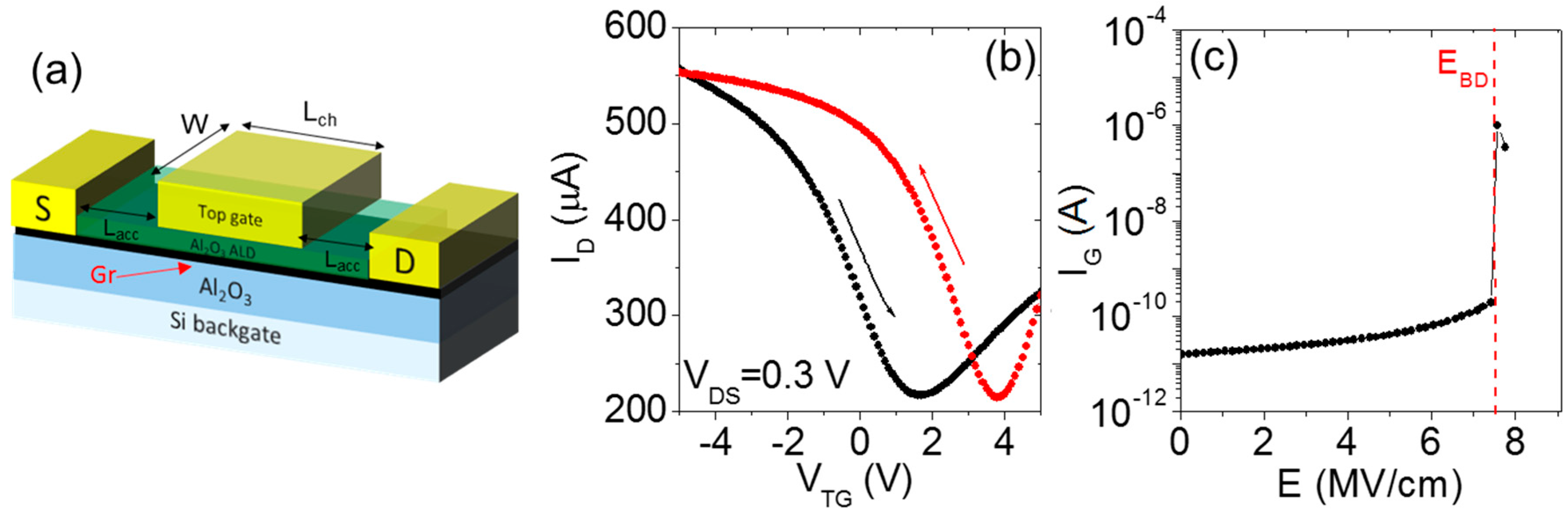
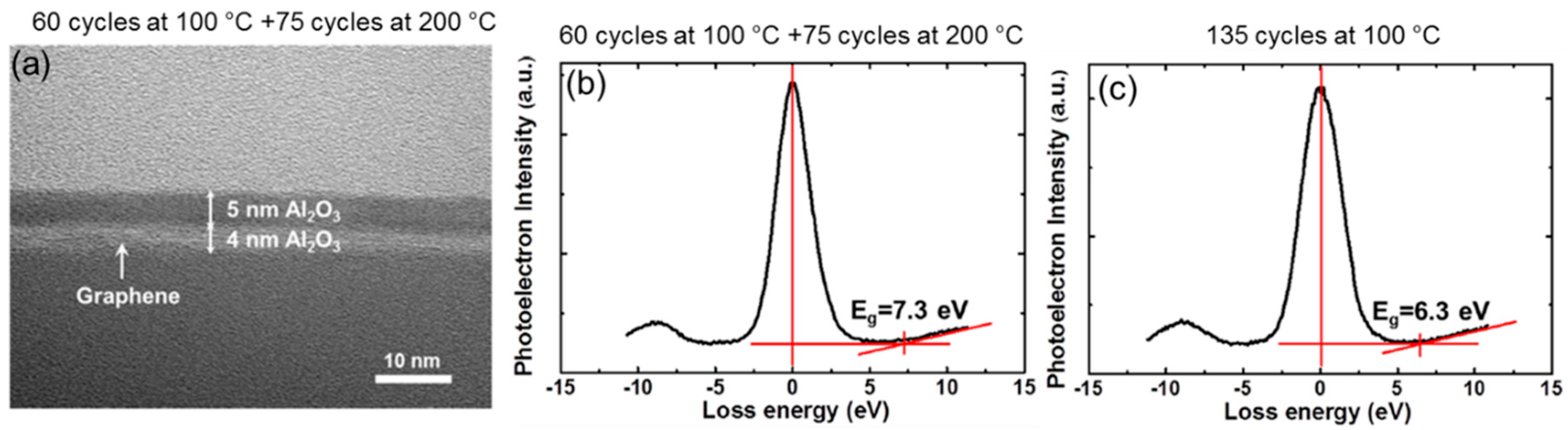
2. Two-Step ALD Growth on Graphene with an “In-Situ” Seed Layer
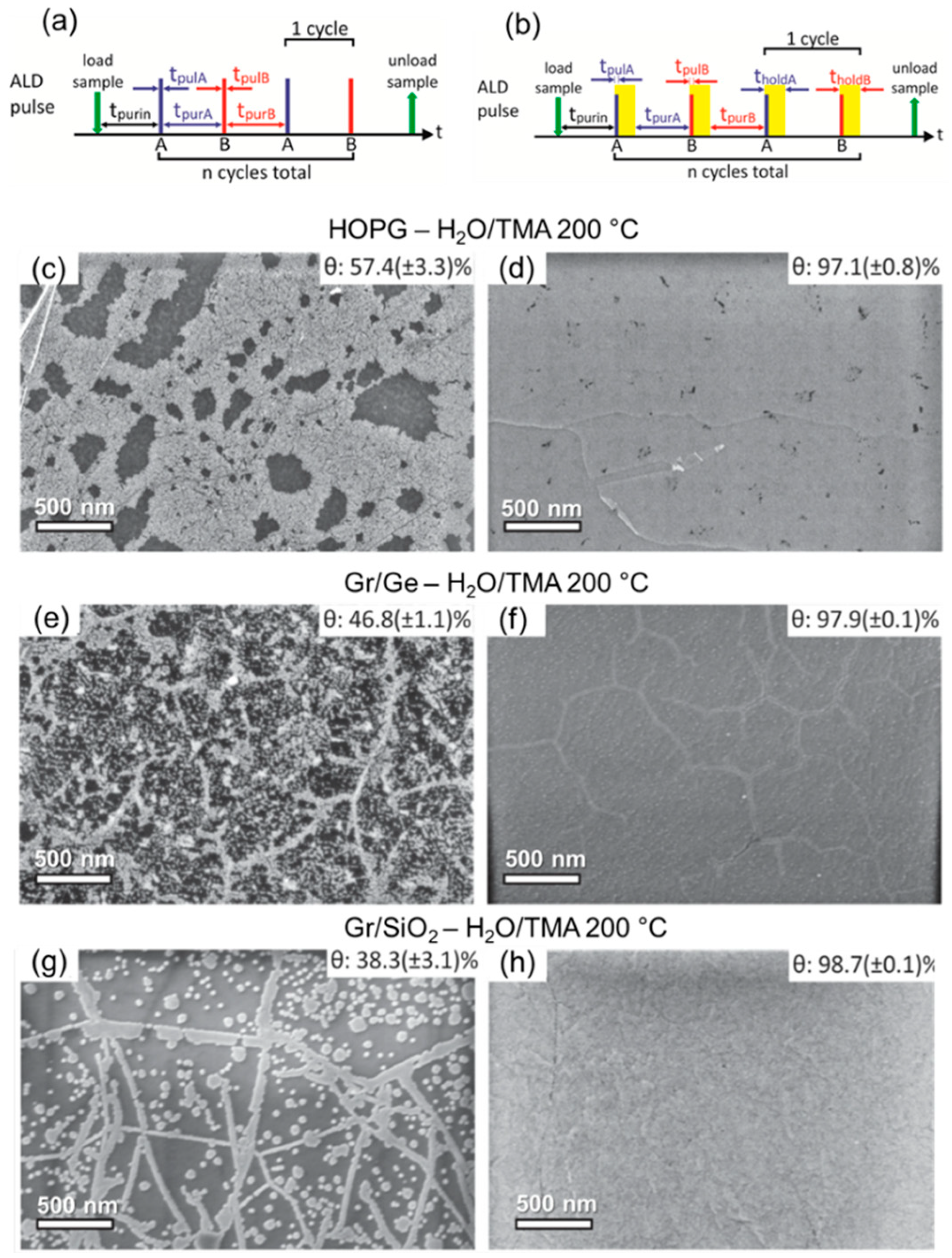
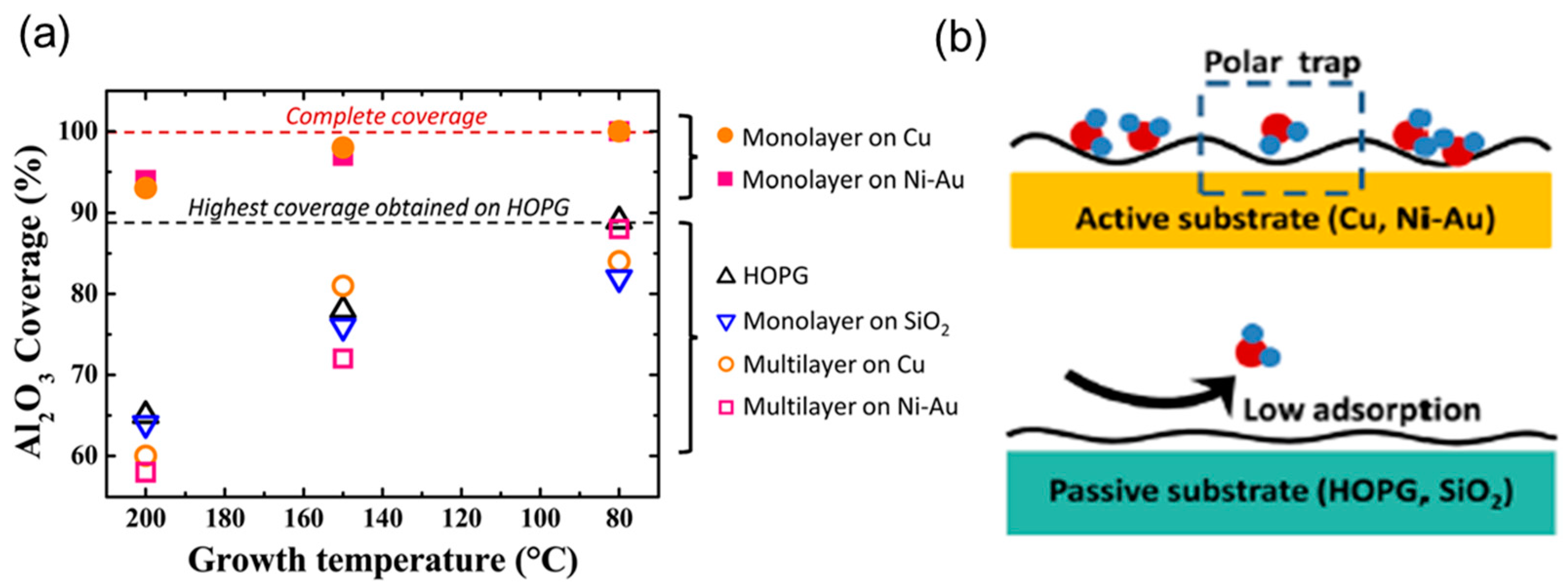
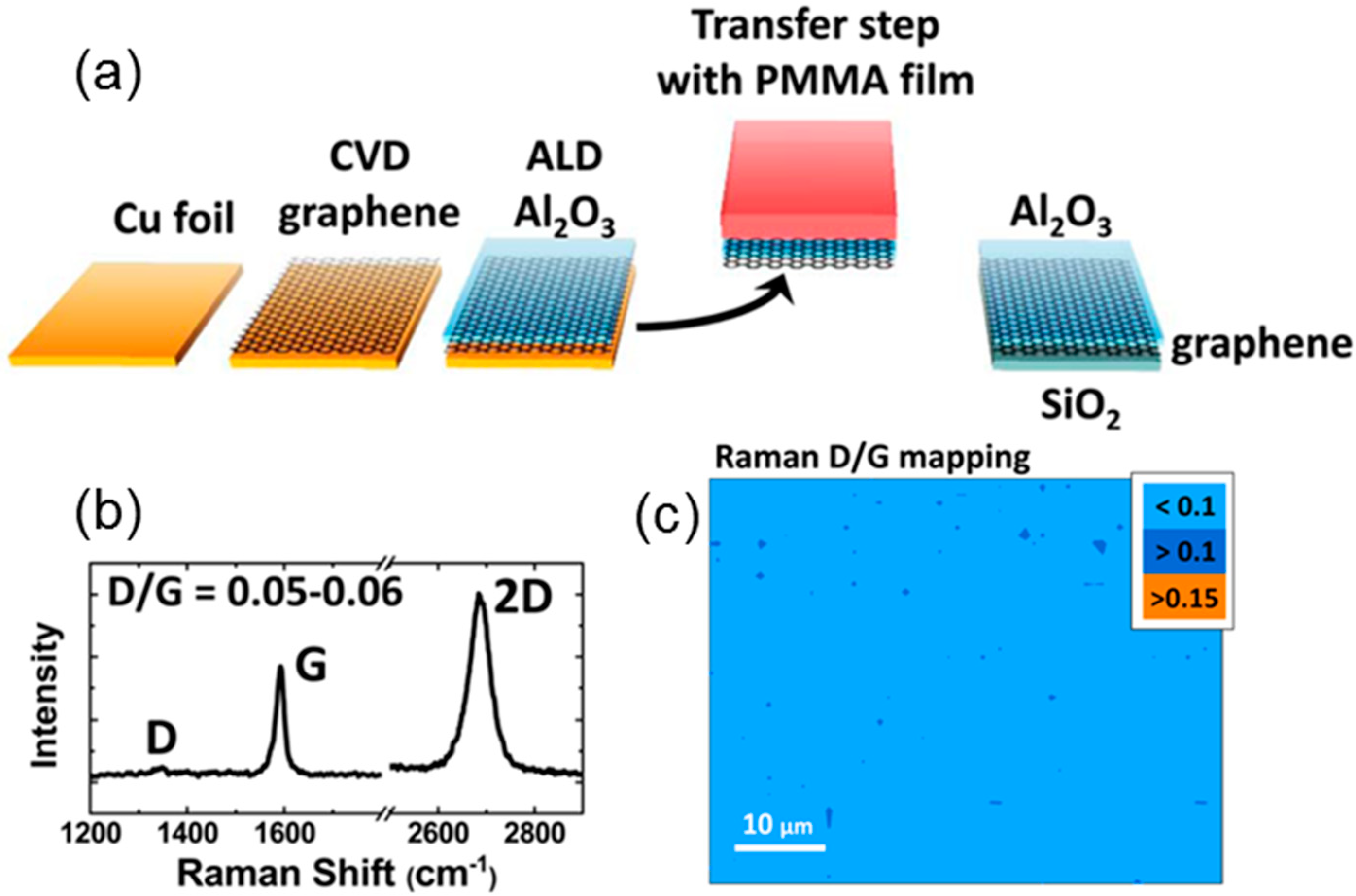
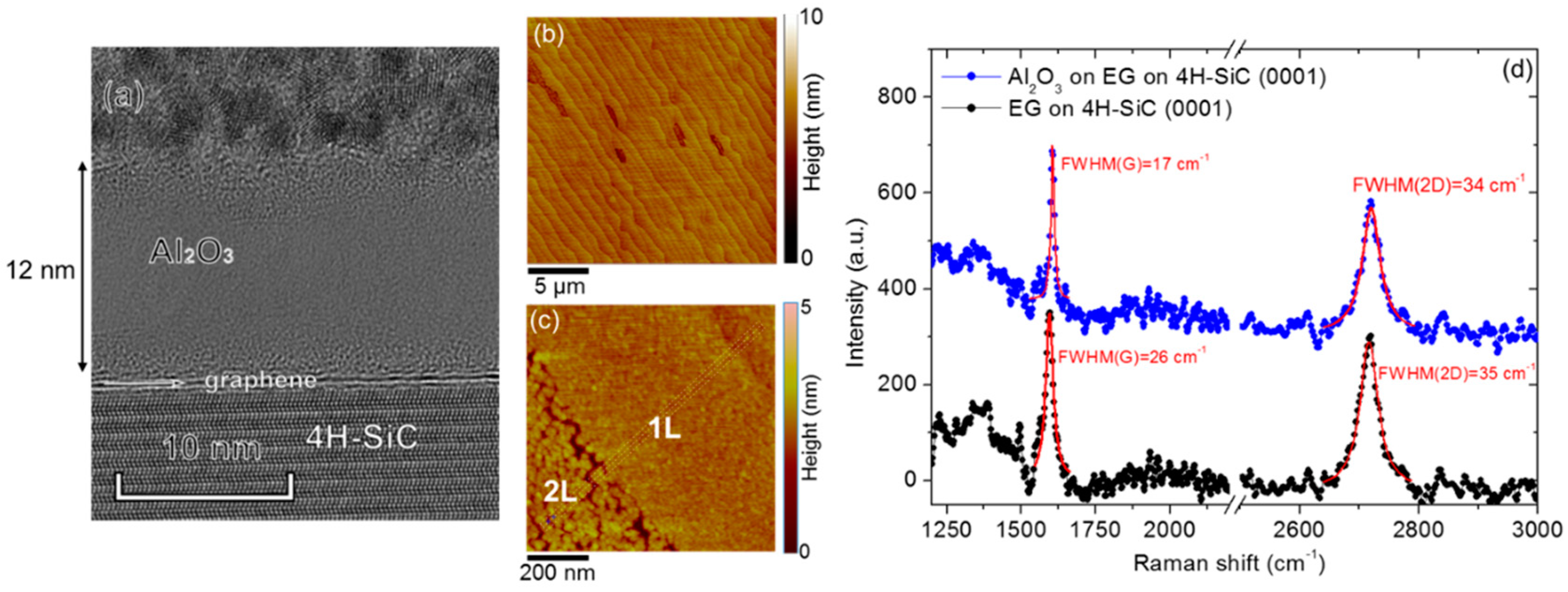
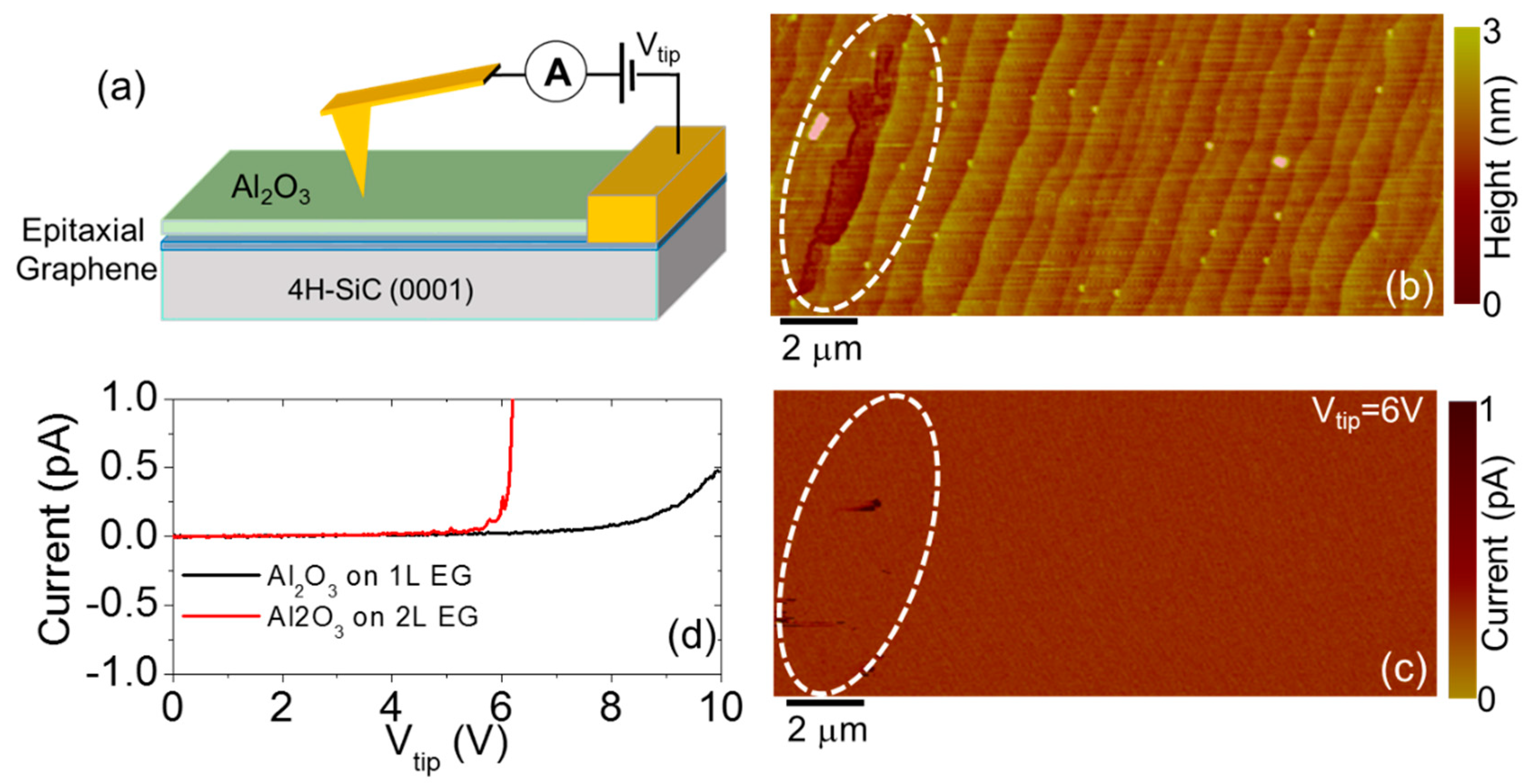
3. Recent Approaches to “Seed-Layer Free” ALD on Graphene
4. Conclusions and Outlooks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric Field Effect in Atomically Thin Carbon Films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Giannazzo, F.; Raineri, V. Graphene: Synthesis and nanoscale characterization of electronic properties. Riv. Del Nuovo Cim. 2012, 35, 267–304. [Google Scholar]
- Dean, C.R.; Young, A.F.; Meric, I.; Lee, C.; Wang, L.; Sorgenfrei, S.; Watanabe, K.; Taniguchi, T.; Kim, P.; Shepard, K.L.; et al. Boron nitride substrates for high-quality graphene electronics. Nat. Nanotechnol. 2010, 5, 722–726. [Google Scholar] [CrossRef] [PubMed]
- Mayorov, A.S.; Gorbachev, R.V.; Morozov, S.V.; Britnell, L.; Jalil, R.; Ponomarenko, L.A.; Blake, P.; Novoselov, K.S.; Watanabe, K.; Taniguchi, T. Micrometer-scale ballistic transport in encapsulated graphene at room temperature. Nano Lett. 2011, 11, 2396–2399. [Google Scholar] [CrossRef] [PubMed]
- Giannazzo, F.; Sonde, S.; Lo Nigro, R.; Rimini, E.; Raineri, V. Mapping the Density of Scattering Centers Limiting the Electron Mean Free Path in Graphene. Nano Lett. 2011, 11, 4612–4618. [Google Scholar] [CrossRef] [PubMed]
- Nair, R.R.; Blake, P.; Grigorenko, A.N.; Novoselov, K.S.; Booth, T.J.; Stauber, T.; Peres, N.M.; Geim, A.K. Fine Structure Constant Defines Visual Transparency of Graphene. Science 2008, 320, 1308. [Google Scholar] [CrossRef] [PubMed]
- Balandin, A.; Ghosh, S.; Bao, W.; Calizo, I.; Teweldebrahn, D.; Miao, F.; Lau, C.N. Superior Thermal Conductivity of Single-Layer Graphene. Nano Lett. 2008, 8, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 2008, 321, 385–388. [Google Scholar] [CrossRef]
- Lin, Y.-M.; Dimitrakopoulos, C.; Jenkins, K.A.; Farmer, D.B.; Chiu, H.-Y.; Grill, A.; Avouris, P. 100-GHz Transistors from Wafer-Scale Epitaxial Graphene. Science 2010, 327, 662. [Google Scholar] [CrossRef]
- Wu, Y.; Lin, Y.; Bol, A.A.; Jenkins, K.A.; Xia, F.; Farmer, D.B.; Zhu, Y.; Avouris, P. High-Frequency, Scaled Graphene Transistors on Diamond-Like Carbon. Nature 2011, 472, 74–78. [Google Scholar] [CrossRef]
- Fisichella, G.; Lo Verso, S.; Di Marco, S.; Vinciguerra, V.; Schilirò, E.; Di Franco, S.; Lo Nigro, R.; Roccaforte, F.; Zurutuza, A.; Centeno, A.; et al. Advances in the fabrication of graphene transistors on flexible substrates. Beilstein J. Nanotechnol. 2017, 8, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Giannazzo, F.; Greco, G.; Roccaforte, F.; Sonde, S.S. Vertical Transistors Based on 2D Materials: Status and Prospects. Crystals 2018, 8, 70. [Google Scholar] [CrossRef]
- Mehr, W.; Dabrowski, J.; Scheytt, J.C.; Lippert, G.; Xie, Y.H.; Lemme, M.C.; Ostling, M.; Lupina, G. Vertical Graphene Base Transistor. IEEE Electron Device Lett. 2012, 33, 691–693. [Google Scholar] [CrossRef]
- Giannazzo, F.; Fisichella, G.; Greco, G.; Schilirò, E.; Deretzis, I.; Lo Nigro, R.; La Magna, A.; Roccaforte, F.; Iucolano, F.; Lo Verso, S.; et al. Fabrication and Characterization of Graphene Heterostructures with Nitride Semiconductors for High Frequency Vertical Transistors. Phys. Status Solidi 2017, 215, 1700653. [Google Scholar] [CrossRef]
- Cabrero-Vilatela, A.; Alexander-Webber, J.A.; Sagade, A.A.; Aria, A.I.; Braeuninger-Weimer, P.; Martin, M.-B.; Weatherup, R.S.; Hofmann, S. Atomic layer deposited oxide films as protective interface layers for integrated graphene transfer. Nanotechnology 2017, 28, 485201. [Google Scholar] [CrossRef] [PubMed]
- Bae, T.-E.; Kim, H.; Jung, J.; Cho, W.-J. Fabrication of High-Performance Graphene Field-Effect Transistor with Solution-Processed Al2O3 Sensing Membrane. Appl. Phys. Lett. 2014, 104, 153506. [Google Scholar] [CrossRef]
- George, S.M. Atomic Layer Deposition: An overview. Chem. Rev. 2010, 110, 111–131. [Google Scholar] [CrossRef] [PubMed]
- Niinistö, L.; Päiväsaari, J.; Niinistö, J.; Putkonen, M.; Nieminen, M. Advanced electronic and optoelectronic materials by Atomic Layer Deposition: An overview with special emphasis on recent progress in processing of high-k dielectrics and other oxide materials. Phys. Status Solidi 2004, 201, 1443–1452. [Google Scholar] [CrossRef]
- Van Delft, J.A.; Garcia-Alosno, D.; Kessels, W.M.M. Atomic layer deposition for photovoltaics: Applications and prospects for solar cell manufacturing. Semicond. Sci. Technol. 2012, 27, 074002. [Google Scholar] [CrossRef]
- Muñoz-Rojas, D.; MacManus-Driscoll, J. Spatial atmospheric atomic layer deposition: A new laboratory and industrial tool for low-cost photovoltaics. Mater. Horiz. 2014, 1, 314–320. [Google Scholar] [CrossRef]
- Graniel, O.; Weber, M.; Balme, S.; Miele, P.; Bechelany, M. Atomic layer deposition for biosensing applications. Biosens. Bioelectron. 2018, 122, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Weber, M.; Iatsunskyi, I.; Coy, E.; Miele, P.; Cornu, D.; Bechelany, M. Novel and Facile Route for the Synthesis of Tunable Boron Nitride Nanotubes Combining Atomic Layer Deposition and Annealing Processes for Water Purification. Adv. Mater. Interfaces 2018, 5, 1800056. [Google Scholar] [CrossRef]
- Tan, L.K.; Liu, B.; Teng, J.H.; Guo, S.; Low, H.Y.; Ping Loh, K. Atomic layer deposition of a MoS2 film. Nanoscale 2014, 6, 10584–10588. [Google Scholar] [CrossRef] [PubMed]
- Ni, Z.H.; Wang, H.M.; Ma, Y.; Kasim, J.; Wu, Y.H.; Shen, Z.X. Tunable Stress and Controlled Thickness Modification in Graphene by Annealing. ACS Nano 2008, 2, 1033–1039. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Tabakman, S.M.; Dai, H. Atomic Layer Deposition of Metal Oxides on Pristine and Functionalized Graphene. J. Am. Chem. Soc. 2008, 130, 8152. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Fu, Q.; Cui, Y.; Mu, R.; Jin, L.; Bao, X. Enhanced reactivity of graphene wrinkles and their function as nanosized gas inlets for reactions under graphene. Phys. Chem. Chem. Phys. 2013, 15, 19042. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Lee, H.-B.-R.; Johnson, R.W.; Tanskanen, J.T.; Liu, N.; Kim, M.-G.; Pang, C.; Ahn, C.; Bent, S.F.; Bao, Z. Selective metal deposition at graphene line defects by atomic layer deposition. Nat. Commun. 2014, 5, 4781. [Google Scholar] [CrossRef] [PubMed]
- Vervuurt, R.H.J.; Kessels, W.M.M.; Bol, A.A. Atomic Layer Deposition for Graphene Device Integration. Adv. Mater. Interfaces 2017, 4, 1700232. [Google Scholar] [CrossRef]
- Lee, B.; Park, S.Y.; Kim, H.C.; Cho, K.; Vogel, E.M.; Kim, M.J.; Wallace, R.M.; Kim, J. Conformal Al2O3 dielectric layer deposited by atomic layer deposition for graphene-based nanoelectronics. Appl. Phys. Lett. 2008, 92, 203102. [Google Scholar] [CrossRef]
- Robinson, Z.R.; Jernigan, G.G.; Wheeler, V.D.; Hernández, S.C.; Eddy, C.R.; Mowll, T.R.; Ong, E.W.; Ventrice, C.A.; Geisler, H.; Pletikosic, I.; et al. Growth and characterization of Al2O3 films on fluorine functionalized epitaxial graphene. J. Appl. Phys. 2016, 120, 075302. [Google Scholar] [CrossRef]
- Lim, T.; Kim, D.; Ju, S. Direct deposition of aluminum oxide gate dielectric on graphene channel using nitrogen plasma treatment. Appl. Phys. Lett. 2013, 113, 013107. [Google Scholar] [CrossRef]
- Vervuurt, R.J.; Karasulu, B.; Verheijen, M.A.; Kessels, W.M.M.; Bol, A.A. Uniform Atomic Layer Deposition of Al2O3 on Graphene by Reversible Hydrogen Plasma Functionalization. Chem. Mater. 2017, 29, 2090–2100. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, V.; Garces, N.; Nyakiti, L.; Myers-Ward, R.; Jernigan, G.; Culbertson, J.; Eddy, C.; Gaskill, D.K. Fluorine functionalization of epitaxial graphene for uniform deposition of thin high-κ dielectrics. Carbon 2012, 50, 2307. [Google Scholar] [CrossRef]
- Hollander, M.J.; LaBella, M.; Hughes, Z.R.; Zhu, M.; Trumbull, K.A.; Cavalero, R.; Snyder, D.W.; Wang, X.; Hwang, E.; Datta, S.; et al. Enhanced Transport and Transistor Performance with Oxide Seeded High-k Gate Dielectrics on Wafer-Scale Epitaxial Graphene. Nano Lett. 2011, 11, 3601–3607. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Nah, J.; Jo, I.; Shahrjerdi, D.; Colombo, L.; Yao, Z.; Tutuc, E.; Banerjee, S.K. Realization of a High Mobility Dual-Gated Graphene Field-Effect Transistor with Al2O3 Dielectric. Appl. Phys. Lett. 2009, 94, 062107. [Google Scholar] [CrossRef]
- Fallahazad, B.; Lee, K.; Lian, G.; Kim, S.; Corbet, C.M.; Ferrer, D.A.; Colombo, L.; Tutuc, E. Scaling of Al2O3 Dielectric for Graphene Field-Effect Transistors. Appl. Phys. Lett. 2012, 100, 093112. [Google Scholar] [CrossRef]
- Alles, H.; Aarik, J.; Aidla, A.; Fay, A.; Kozlova, J.; Niilisk, A.; Paärs, M.; Raähn, M.; Wiesner, M.; Hakonen, P.; et al. Atomic Layer Deposition of HfO2 on Graphene from HfCl4 and H2O. Open Phys. 2011, 9, 319–324. [Google Scholar] [CrossRef]
- Zheng, L.; Cheng, X.; Cao, D.; Wang, G.; Wang, Z.; Xu, D.; Xia, C.; Shen, L.; Yu, Y.; Shen, D. Improvement of Al2O3 Films on Graphene Grown by Atomic Layer Deposition with Pre-H2O Treatment. ACS Appl. Mater. Interfaces 2014, 6, 7014–7019. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Cheng, X.; Wang, Z.; Xia, C.; Cao, D.; Shen, L.; Wang, Q.; Yu, Y.; Shen, D. Reversible n-Type Doping of Graphene by H2O Based Atomic-Layer Deposition and Its Doping Mechanism. J. Phys. Chem. C 2015, 119, 5995–6000. [Google Scholar] [CrossRef]
- Zheng, L.; Cheng, X.; Cao, D.; Zhang, D.; Wang, Z.; Xu, D.; Xia, C.; Shenab, L.; Yua, Y. Al2O3–Gd2O3 double-films grown on graphene directly by H2O-assisted atomic layer deposition. RSC Adv. 2014, 4, 44296–44301. [Google Scholar] [CrossRef]
- Fisichella, G.; Schilirò, E.; Di Franco, S.; Fiorenza, P.; Lo Nigro, R.; Roccaforte, F.; Ravesi, S.; Giannazzo, F. Interface Electrical Properties of Al2O3 Thin Films on Graphene Obtained by Atomic Layer Deposition with an in Situ Seedlike Layer. ACS Appl. Mater. Interfaces 2017, 9, 7761–7771. [Google Scholar] [CrossRef] [PubMed]
- Aria, A.I.; Nakanishi, K.; Xiao, L.; Braeuninger-Weimer, P.; Sagade, A.A.; Alexander-Webber, J.A.; Hofmann, S. Parameter Space of Atomic Layer Deposition of Ultrathin Oxides on Graphene. ACS Appl. Mater. Interfaces 2016, 8, 30564–30575. [Google Scholar] [CrossRef] [PubMed]
- Dlubak, B.; Kidambi, P.R.; Weatherup, R.S.; Hofmann, S.; Robertson, J. Substrate-assisted nucleation of ultra-thin dielectric layers on graphene by atomic layer deposition. Appl. Phys. Lett. 2012, 100, 173113. [Google Scholar] [CrossRef]
- Schilirò, E.; Lo Nigro, R.; Roccaforte, F.; Deretzis, J.; La Magna, A.; Armano, A.; Agnello, S.; Pecz, B.; Ivanov, I.G.; Giannazzo, F. Seed-Layer-Free Atomic Layer Deposition of Highly Uniform Al2O3 Thin Films onto Monolayer Epitaxial Graphene on Silicon Carbide. Adv. Mater. Interfaces 2019, 1900097, 1–11. [Google Scholar]
- Grebel, H.; Stan, L.; Sumant, A.V.; Liu, Y.; Gosztola, D.; Ocola, L.; Fisher, B. Transfer of Graphene with Protective Oxide Layers. ChemEngineering 2018, 2, 58. [Google Scholar] [CrossRef]
- Zhang, Y.; Qiu, Z.; Cheng, X.; Xie, H.; Wang, H.; Xie, X.; Yu, Y.; Liu, R. Direct Growth of High-Quality Al2O3 Dielectric on Graphene Layers by Low-Temperature H2O-Based ALD. J. Phys. D Appl. Phys. 2014, 47, 055106. [Google Scholar] [CrossRef]
- Armano, A.; Buscarino, G.; Cannas, M.; Gelardi, F.M.; Giannazzo, F.; Schiliro, E.; Lo Nigro, R.; Agnello, S. Influence of oxide substrates on monolayer graphene doping process by thermal treatments in oxygen. Carbon 2019, 149, 546–555. [Google Scholar] [CrossRef]
- Zhang, Y.; Guerra-Nunez, C.; Utke, I.; Michler, J.; Agrawal, P.; Rossell, M.D.; Erni, R. Atomic Layer Deposition of Titanium Oxide on Single-Layer Graphene: An Atomic-Scale Study Toward Understanding Nucleation and Growth. Chem. Mater. 2017, 29, 2232–2238. [Google Scholar] [CrossRef]
- Rafiee, J.; Mi, X.; Gullapalli, H.; Thomas, A.V.; Yavari, F.; Shi, Y.; Ajayan, P.M.; Koratkar, N.A. Wetting transparency of graphene. Nat. Mater. 2012, 11, 217–222. [Google Scholar] [CrossRef]
- Berger, C.; Song, Z.; Li, X.; Wu, X.; Brown, N.; Naud, C.; Mayou, D.; Li, T.; Hass, J.; Marchenkov, A.N.; et al. Electronic Confinement and Coherence in Patterned Epitaxial Graphene. Science 2006, 312, 1191–1196. [Google Scholar] [CrossRef]
- Bouhafs, C.; Zakharov, A.A.; Ivanov, I.G.; Giannazzo, F.; Eriksson, J.; Stanishev, V.; Kühne, P.; Iakimov, T.; Hofmann, T.; Schubert, M.; et al. Multi-scale investigation of interface properties, stacking order and decoupling of few layer graphene on C-face 4H-SiC. Carbon 2017, 116, 722–732. [Google Scholar] [CrossRef]
- Ostler, M.; Deretzis, I.; Mammadov, S.; Giannazzo, F.; Nicotra, G.; Spinella, C.; Seyller, T.; La Magna, A. Direct growth of quasi-free-standing epitaxial graphene on nonpolar SiC surfaces. Phys. Rev. B 2013, 88, 085408. [Google Scholar] [CrossRef]
- Tzalenchuk, A.; Lara-Avila, S.; Kalaboukhov, A.; Paolillo, S.; Syvajarvi, M.; Yakimova, R.; Kazakova, O.; Janssen, T.J.B.M.; Fal’ko, V.; Kubatkin, S. Towards a quantum resistance standard based on epitaxial graphene. Nat. Nanotechnol. 2010, 5, 186. [Google Scholar] [CrossRef] [PubMed]
- Roccaforte, F.; Fiorenza, P.; Greco, G.; Lo Nigro, R.; Giannazzo, F.; Iucolano, F.; Saggio, M. Emerging trends in wide band gap semiconductors (SiC and GaN) technology for power devices. Microelectron. Eng. 2018, 187, 66–77. [Google Scholar] [CrossRef]
- Varchon, F.; Feng, R.; Hass, J.; Li, X.; Nguyen, B.N.; Naud, C.; Mallet, P.; Veuillen, J.Y.; Berger, C.; Conrad, E.H.; et al. Electronic Structure of Epitaxial Graphene Layers on SiC: Effect of the Substrate. Phys. Rev. Lett. 2007, 99, 126805. [Google Scholar] [CrossRef] [PubMed]
- Nicotra, G.; Ramasse, Q.M.; Deretzis, I.; La Magna, A.; Spinella, C.; Giannazzo, F. Delaminated Graphene at Silicon Carbide Facets: Atomic Scale Imaging and Spectroscopy. ACS Nano 2013, 7, 3045–3052. [Google Scholar] [CrossRef] [PubMed]
- Riedl, C.; Zakharov, A.A.; Starke, U.; Riedl, C. Precise in situ thickness analysis of epitaxial graphene layers on SiC (0001) using low-energy electron diffraction and angle resolved ultraviolet photoelectron spectroscopy. Appl. Phys. Lett. 2008, 93, 033106. [Google Scholar] [CrossRef]
- Sonde, S.; Giannazzo, F.; Raineri, V.; Yakimova, R.; Huntzinger, J.-R.; Tiberj, A.; Camassel, J. Electrical properties of the graphene/4H-SiC (0001) interface probed by scanning current spectroscopy. Phys. Rev. B 2009, 80, 241406. [Google Scholar] [CrossRef]
- Emtsev, K.V.; Bostwick, A.; Horn, K.; Jobst, J.; Kellogg, G.L.; Ley, L.; McChesney, J.L.; Ohta, T.; Reshanov, S.A.; Röhrl, J.; et al. Towards wafer-size graphene layers by atmospheric pressure graphitization of silicon carbide. Nat. Mater. 2009, 8, 203–207. [Google Scholar] [CrossRef]
- Vecchio, C.; Sonde, S.; Bongiorno, C.; Rambach, M.; Yakimova, R.; Raineri, V.; Giannazzo, F. Nanoscale structural characterization of epitaxial graphene grown on off-axis 4H-SiC (0001). Nanoscale Res. Lett. 2011, 6, 269. [Google Scholar] [CrossRef] [PubMed]
- Speck, F.; Ostler, M.; Rohrl, J.; Emtsev, K.V.; Hundhausen, M.; Ley, L.; Seyller, T. Atomic layer deposited aluminum oxide films on graphite and graphene studied by XPS and AFM. Phys. Status Solidi C 2010, 7, 398. [Google Scholar] [CrossRef]
- Robinson, J.A.; LaBella, M.; Trumbull, K.A.; Weng, X.; Cavelero, R.; Daniels, T.; Hughes, Z.; Hollander, M.; Fanton, M. Epitaxial graphene materials integration: Effects of dielectric overlayers on structural and electronic properties. ACS Nano 2010, 4, 2667. [Google Scholar] [CrossRef] [PubMed]
- Bissett, M.A.; Konabe, S.; Okada, S.; Tsuji, M.; Ago, H. Enhanced Chemical Reactivity of Graphene Induced by Mechanical Strain. ACS Nano 2013, 7, 10335–10343. [Google Scholar] [CrossRef] [PubMed]
- Park, M.J.; Choi, H.-H.; Park, B.; Lee, J.Y.; Lee, C.-H.; Choi, Y.S.; Kim, Y.; Yoo, J.M.; Lee, H.; Hong, B.H. Enhanced Chemical Reactivity of Graphene by Fermi Level Modulation. Chem. Mater. 2018, 30, 5602–5609. [Google Scholar] [CrossRef]
- Giannazzo, F.; Fisichella, G.; Greco, G.; Fiorenza, P.; Roccaforte, F. Conductive Atomic Force Microscopy: Applications in Nanomaterials; Lanza, M., Ed.; Wiley-VCH Verlag: Weinheim, Germany, 2017; pp. 163–186. [Google Scholar]
- Zhu, W.; Neumayer, D.; Perebeinos, V.; Avouris, P. Silicon nitride gate dielectrics and band gap engineering in graphene layers. Nano Lett. 2010, 10, 3572–3576. [Google Scholar] [CrossRef] [PubMed]
- Giannazzo, F.; Deretzis, I.; Nicotra, G.; Fisichella, G.; Ramasse, Q.M.; Spinella, C.; Roccaforte, F.; La Magna, A. High resolution study of structural and electronic properties of epitaxial graphene grown on off-axis 4H–SiC (0001). J. Cryst. Growth 2014, 393, 150–155. [Google Scholar] [CrossRef]
- Meng, X.; Byun, Y.C.; Kim, H.; Lee, J.; Lucero, A.; Cheng, L.; Kim, J. Atomic Layer Deposition of Silicon Nitride Thin Films: A Review of Recent Progress, Challenges, and Outlooks. Materials 2016, 9, 1007. [Google Scholar] [CrossRef]
- Schilirò, E.; Giannazzo, F.; Bongiorno, C.; Di Franco, S.; Greco, G.; Roccaforte, F.; Prystawko, P.; Kruszewski, P.; Leszczyński, M.; Krysko, M.; et al. Structural and electrical properties of AlN thin films on GaN substrates grown by plasma Enhanced-Atomic Layer Deposition. Mater. Sci. Semicond. Process. 2019, 97, 35–39. [Google Scholar] [CrossRef]
- Giannazzo, F.; Fisichella, G.; Greco, G.; La Magna, A.; Roccaforte, F.; Pecz, B.; Yakimova, R.; Dagher, R.; Michon, A.; Cordier, Y. Graphene integration with nitride semiconductors for high power and high frequency electronics. Phys. Status Solidi 2017, 214, 1600460. [Google Scholar] [CrossRef]
- Li, M.-Y.; Su, S.-K.; Philip Wong, H.-S.; Li, L.-J. How 2D semiconductors could extend Moore’s law. Nature 2019, 567, 169–170. [Google Scholar] [CrossRef]
- Giannazzo, F. Engineering 2D heterojunctions with dielectrics. Nature Electronics 2019, 2, 54–55. [Google Scholar] [CrossRef]
- Radisavljevic, B.; Radenovic, A.; Brivio, J.; Giacometti, V.; Kis, A. Single-layer MoS2 transistors. Nat. Nanotechnol. 2011, 6, 147–150. [Google Scholar] [CrossRef] [PubMed]
| High-κ | Graphene Type | Substrate | Method | Seed Layer | Temperature | Thickness | Graphene Damage |
|---|---|---|---|---|---|---|---|
| Al2O3 [46] | Exfoliated from HOPG | SiO2 | One-step ALD process | - | 100 °C | 11 nm | No defects |
| HfO2 [37] | Exfoliated from HOPG | SiO2 | Two-step ALD process | H2O-based Low temperature HfO2 layer | 170 °C/300 °C | 1 nm/20–30 nm | No defects |
| Al2O3 [38] | Transferred CVD-Gr | SiO2 | Two-step ALD process | H2O-assisted low temperature Al2O3 layer | 100 °C/200–250–300 °C | 1–5 nm/8–3 nm | Defects introduction at 250 °C and 300 °C |
| Al2O3 [41] | Transferred CVD-Gr | Al2O3 | Two-step ALD process | H2O-assisted low-temperature Al2O3 layer | 100 °C/250 °C | 4 nm/18 nm | No defects |
| Al2O3, HfO2 [45] | CVD-Gr | Gr/Cu | Two-step ALD process | H2O-assisted low-temperature Al2O3/HfO2 layer | 90 °C/200 °C | 10 nm | Defects after Gr-transferring |
| Al2O3 (seed-layer)/Gd2O3 [40] | Transferred CVD-Gr | SiO2 | Two-step ALD process | H2O-assisted low-temperature Al2O3 layer | 100 °C/200 °C | 5 nm/20 nm | No defects |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schilirò, E.; Lo Nigro, R.; Roccaforte, F.; Giannazzo, F. Recent Advances in Seeded and Seed-Layer-Free Atomic Layer Deposition of High-K Dielectrics on Graphene for Electronics. C 2019, 5, 53. https://doi.org/10.3390/c5030053
Schilirò E, Lo Nigro R, Roccaforte F, Giannazzo F. Recent Advances in Seeded and Seed-Layer-Free Atomic Layer Deposition of High-K Dielectrics on Graphene for Electronics. C. 2019; 5(3):53. https://doi.org/10.3390/c5030053
Chicago/Turabian StyleSchilirò, Emanuela, Raffaella Lo Nigro, Fabrizio Roccaforte, and Filippo Giannazzo. 2019. "Recent Advances in Seeded and Seed-Layer-Free Atomic Layer Deposition of High-K Dielectrics on Graphene for Electronics" C 5, no. 3: 53. https://doi.org/10.3390/c5030053
APA StyleSchilirò, E., Lo Nigro, R., Roccaforte, F., & Giannazzo, F. (2019). Recent Advances in Seeded and Seed-Layer-Free Atomic Layer Deposition of High-K Dielectrics on Graphene for Electronics. C, 5(3), 53. https://doi.org/10.3390/c5030053