Hybrid Carbon-Based Clathrates for Energy Storage
Abstract
:1. Introduction
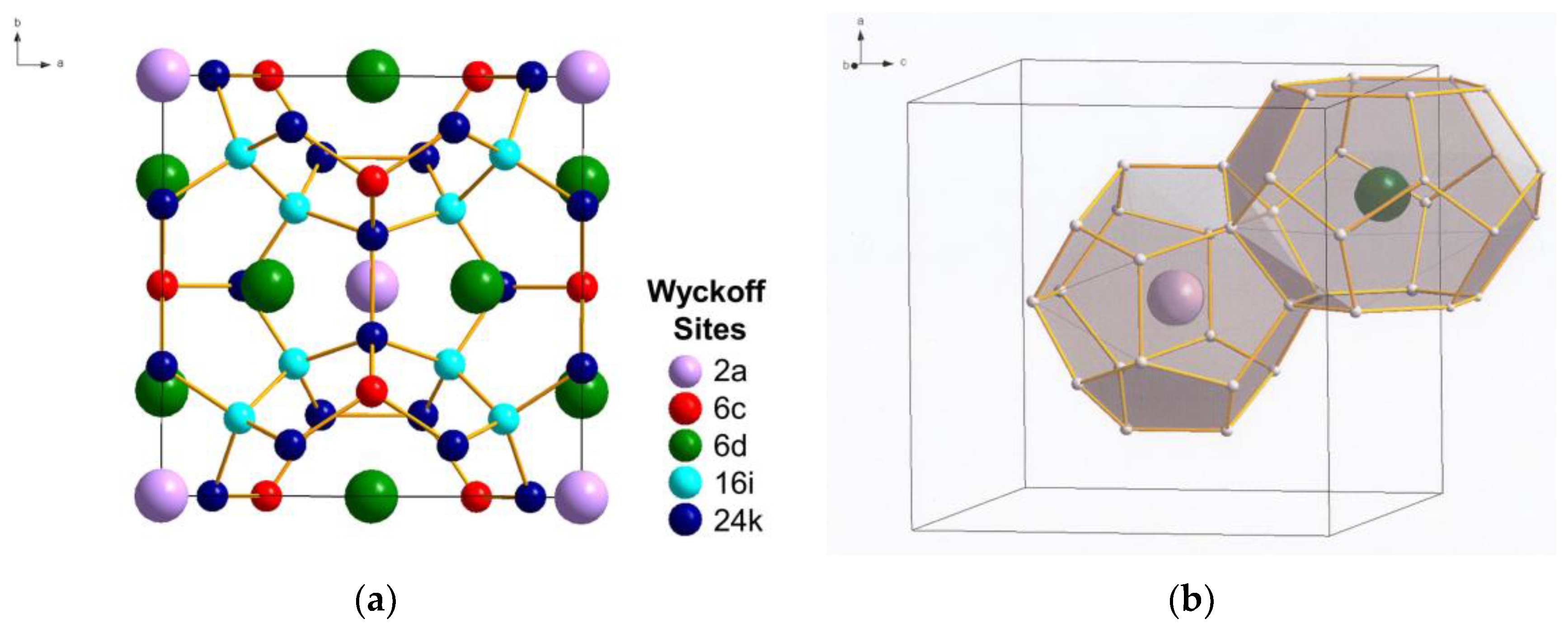
2. Carbon Clathrates
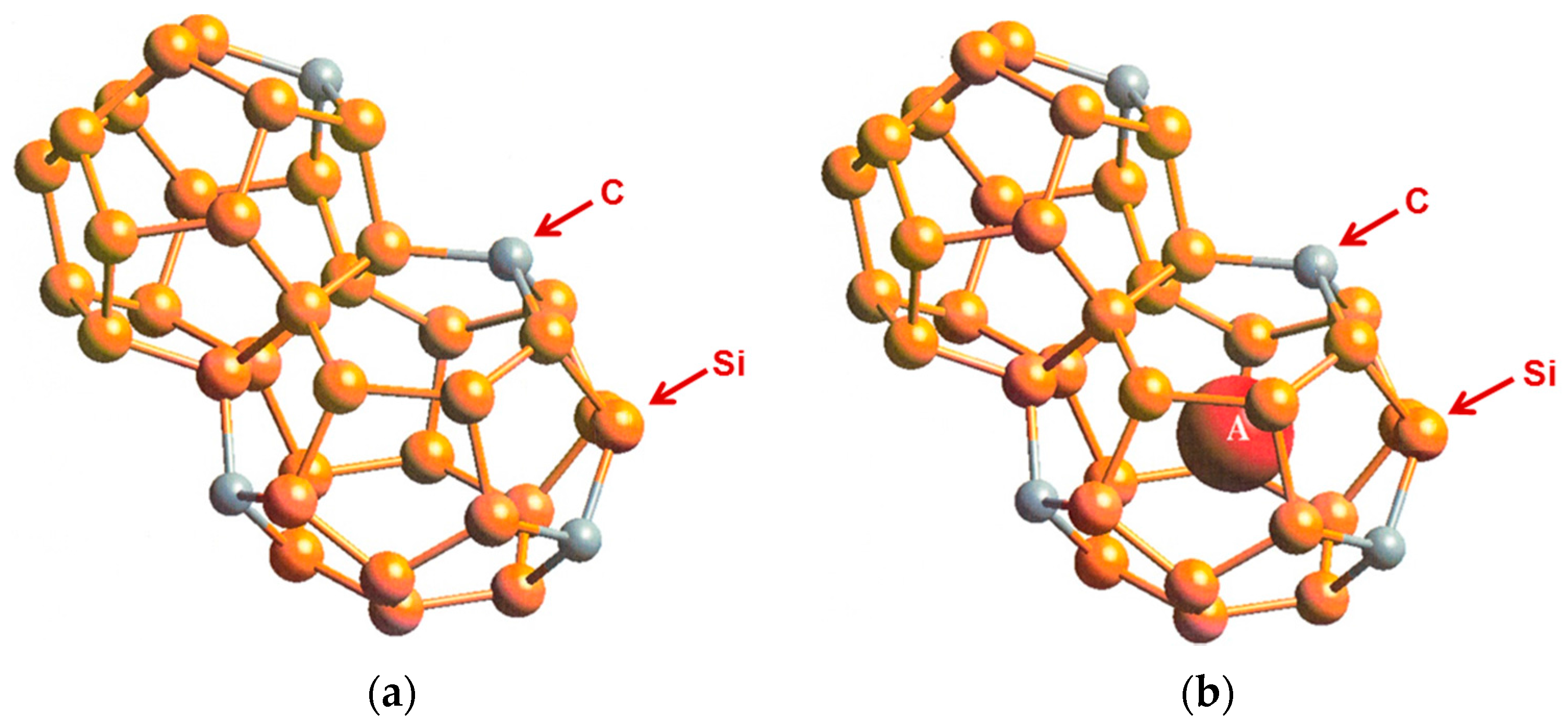
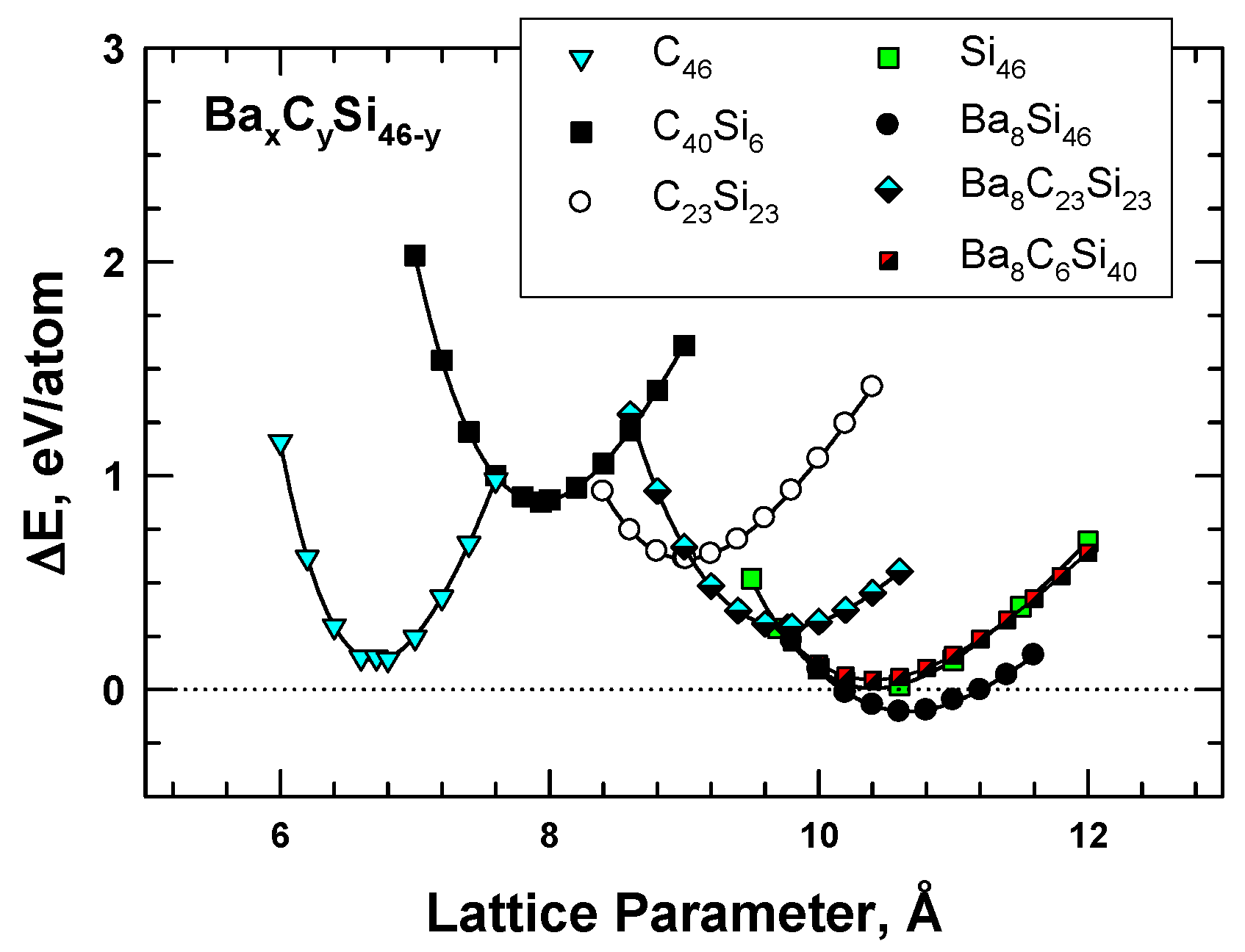
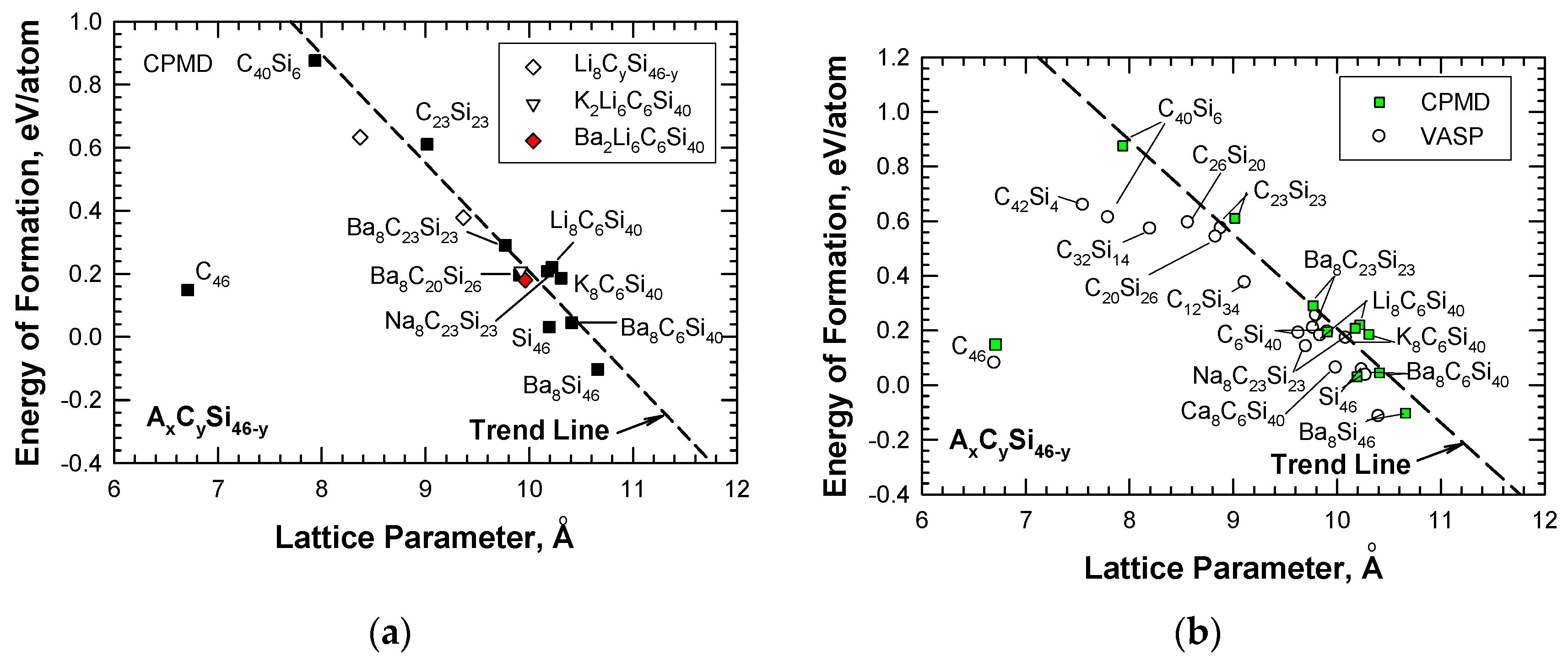
3. Hybrid Carbon–Silicon Clathrates
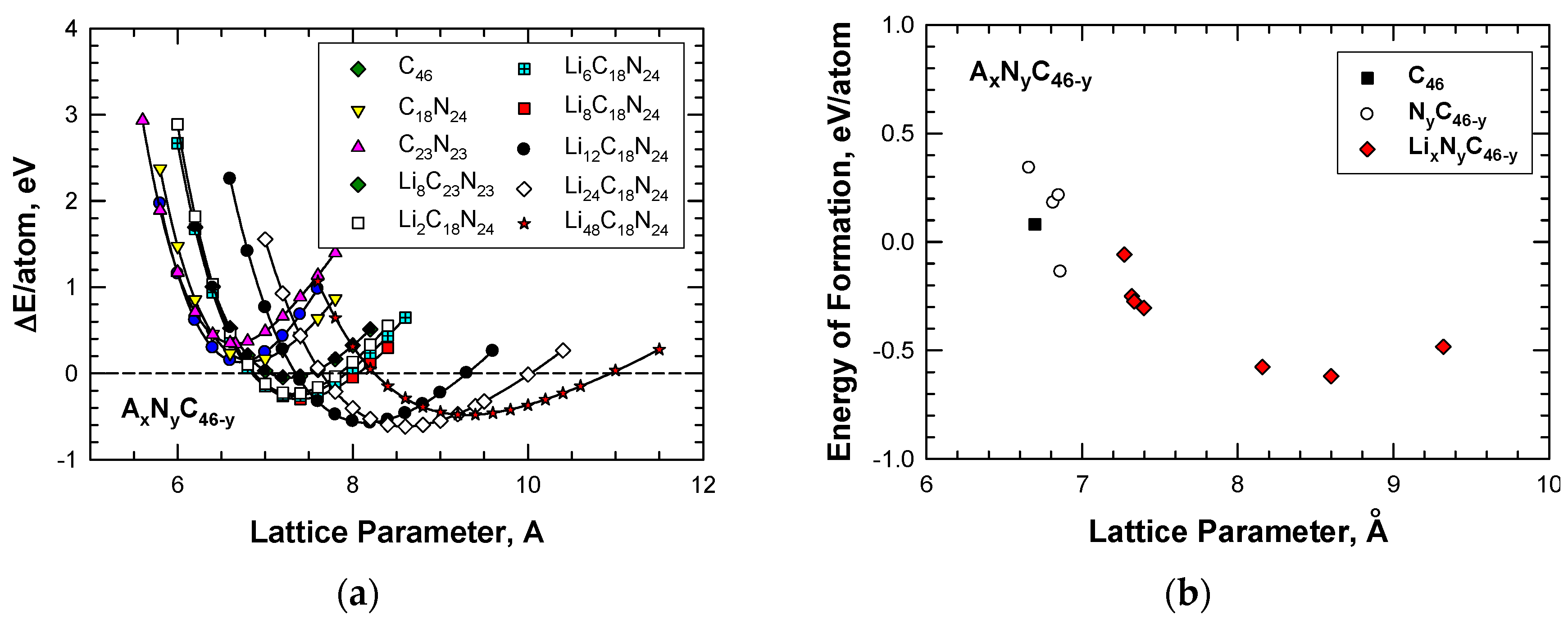
4. Hybrid Carbon–Nitrogen and Carbon–Boron Clathrates
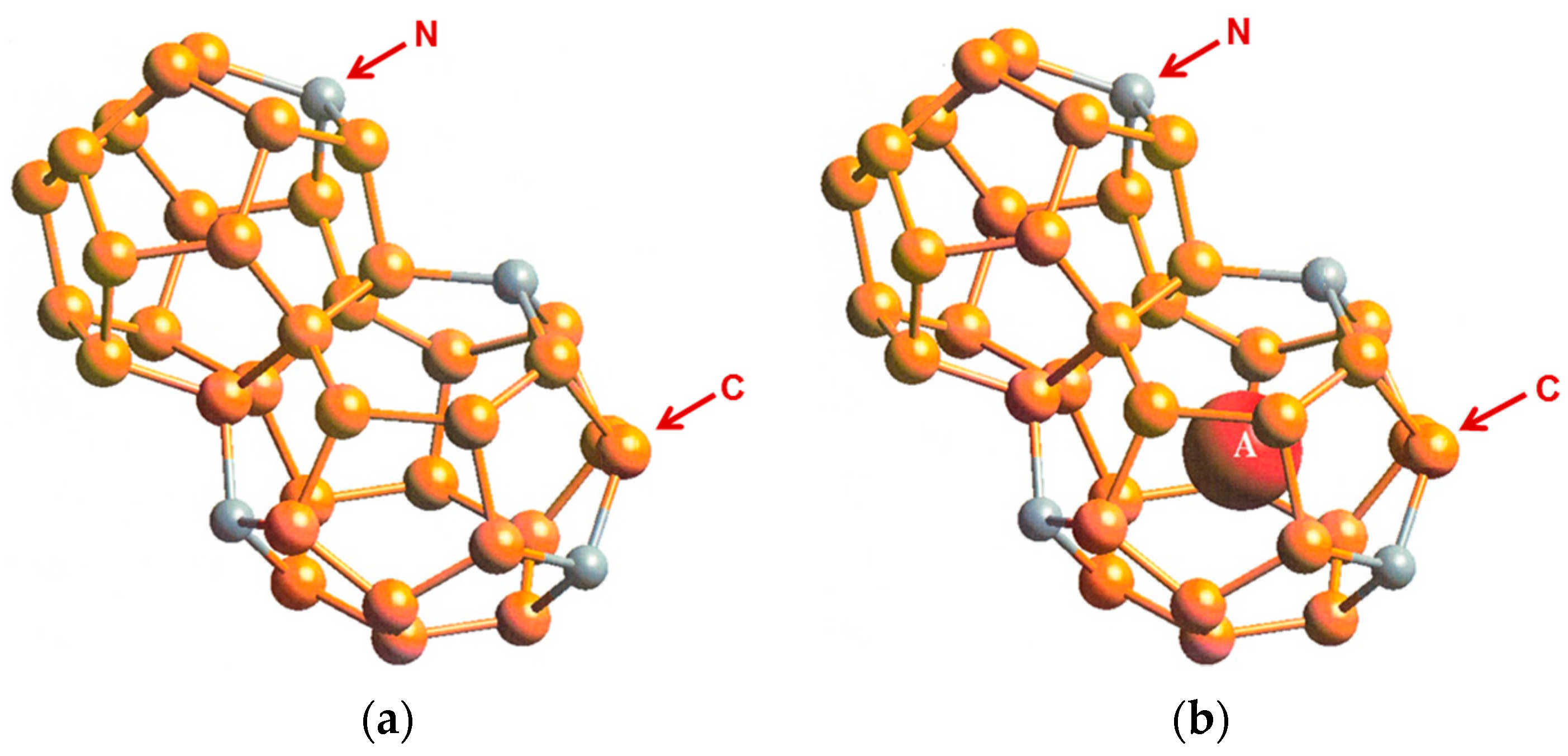
4.1 Hybrid Carbon–Nitrogen Clathrates
4.2. Hybrid Carbon–Boron Clathrates
5. Potential Applications of Hybrid Carbon Clathrates
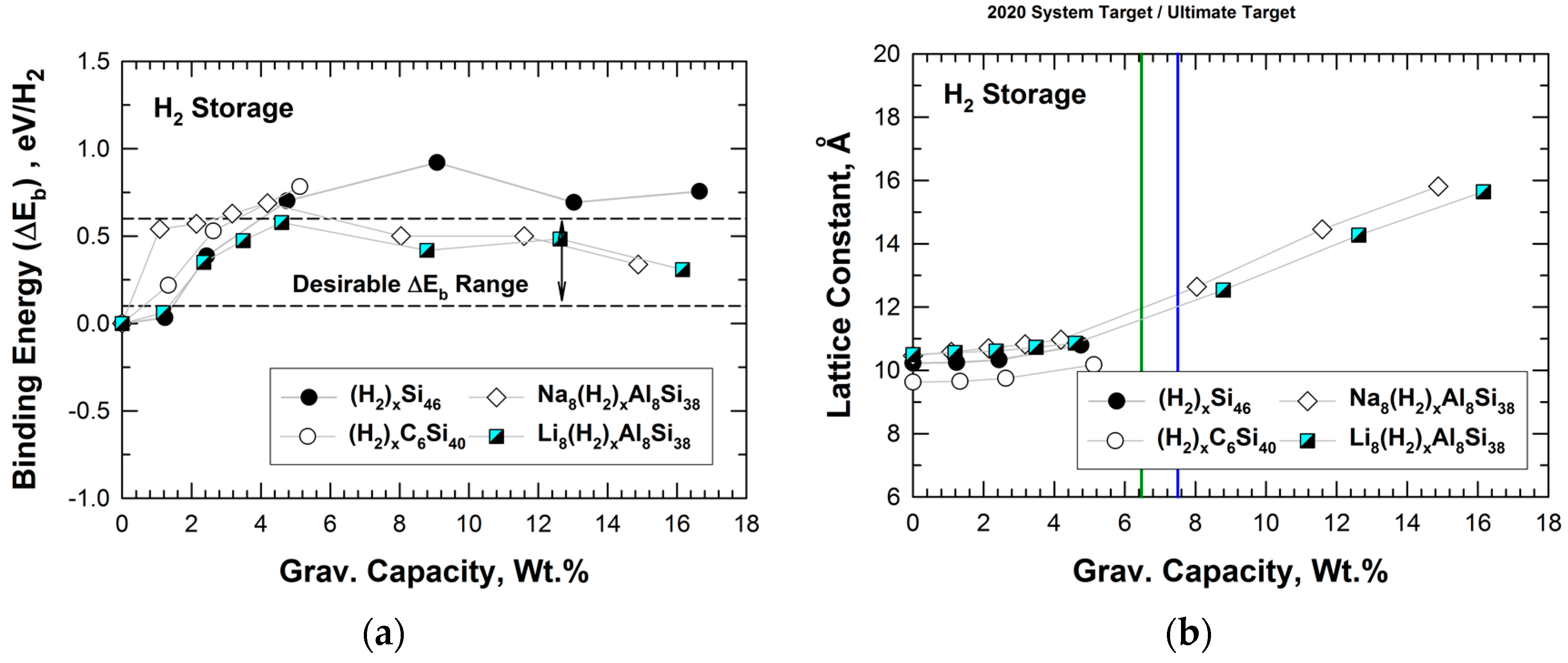
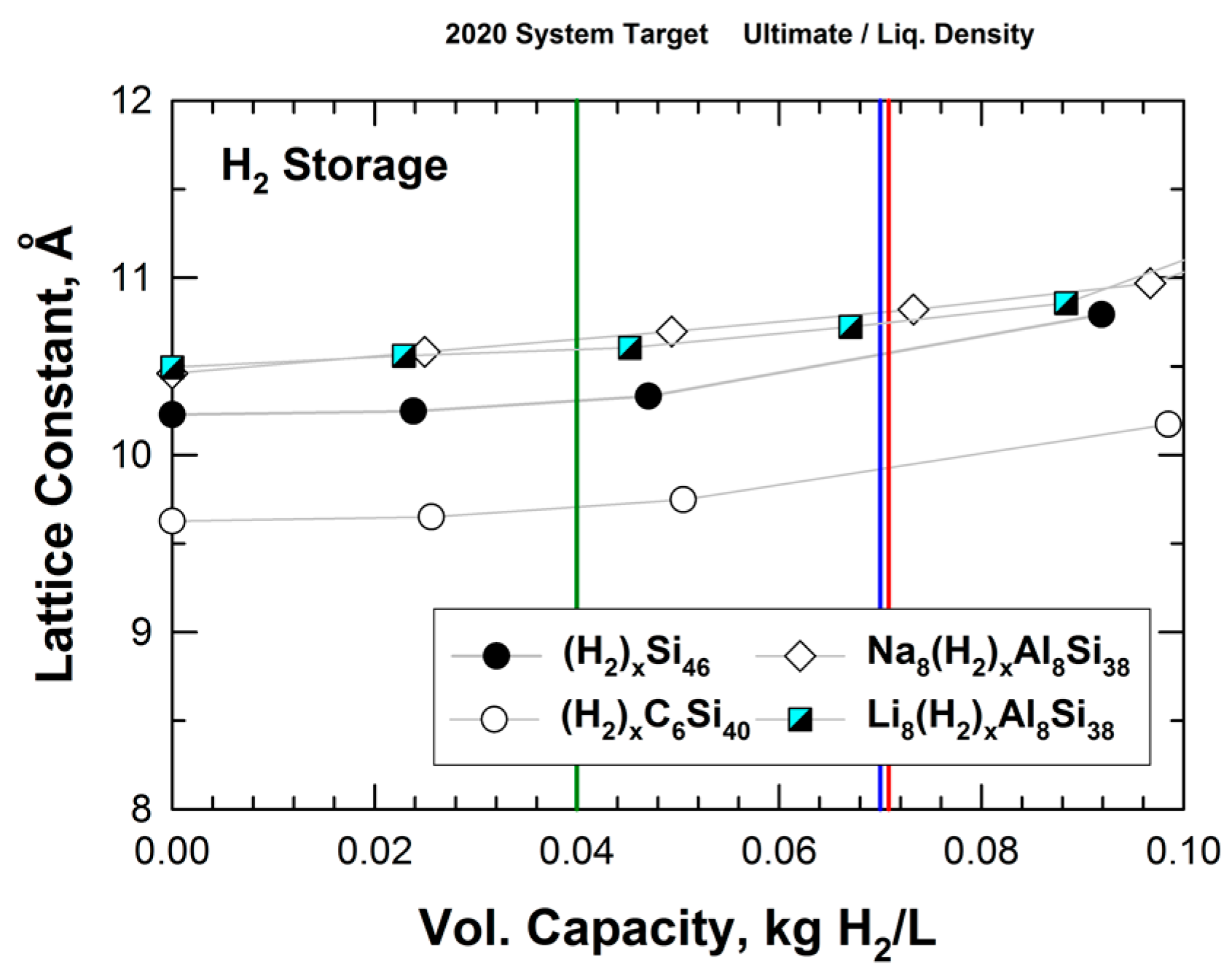
5.1. Hybrid Carbon–Silicon Clathrates as Hydrogen Storage Materials
5.2. Hybrid Carbon–Silicon Clathrates as Electronic or Electrode Materials
5.3. Hybrid Carbon–Nitrogen Clathrates as Hard Materials
6. Synthesis of Hybrid Carbon Clathrates
6.1. Hybrid Carbon–Silicon Clathrates
6.2. Hybrid Carbon–Nitrogen Clathrates
7. Summary
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rey, N.; Muñoz, A.; Rodríquez-Hernández, P.; San Miguel, A. First-principles study of lithium-doped carbon clathrates under pressure. J. Phys. Condens. Matter 2008, 20. [Google Scholar] [CrossRef]
- Wang, J.T.; Chen, C.; Wang, D.S.; Mizuseki, H.; Kawazoe, Y. Phase stability of carbon clathrates at high pressure. J. Appl. Phys. 2010, 107, 063507. [Google Scholar] [CrossRef]
- Perottoni, C.A.; da Jornada, J.A.H. The carbon analogues of type-I silicon clathrates. J. Phys. Condens. Matter 2001, 13, 5981–5998. [Google Scholar] [CrossRef]
- Rogl, P. Formation of clathrates. In Proceedings of the 24th International Conference on Thermoelectrics 2005, ICT 2005, Clemson, SC, USA, 19–23 June 2005; pp. 440–445. [Google Scholar]
- Nesper, R.; Vogel, K.; Blöchl, P.E. Hypothetical carbon modifications derived from zeolite frameworks. Angew. Chem. Int. Ed. Engl. 1993, 32, 701–703. [Google Scholar] [CrossRef]
- Benedek, G.; Galvani, E.; Sanguinetti, S.; Serra, S. Hollow diamonds: Stability and elastic properties. Chem. Phys. Lett. 1995, 244, 339–344. [Google Scholar] [CrossRef]
- Yamanaka, S.; Kubo, A.; Inumaru, K.; Komaguchi, K.; Kini, N.S.; Inoue, T.; Irifune, T. Electron conductive three-dimensional polymer of cuboidal C60. Phys. Rev. Lett. 2006, 96. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.S.; Miller, M.A.; Liang, W.; Ellis-Terrell, C.; Peng, X. First-principles computational design and synthesis of hybrid carbon-silicon clathrates. J. Mater. Sci. 2014, 49, 2723–2733. [Google Scholar] [CrossRef]
- Dovesi, R.; Saunders, V.R.; Roetti, C.; Causa, M.; Harrison, N.M. CRYSTAL95 User’s Manual; University of Torino: Torino, Italy, 1996. [Google Scholar]
- Furthmüller, J.; Hafner, J.; Kresse, G. Ab initio calculation of the structural and electronic properties of carbon and boron nitride using ultrasoft pseudopotentials. Phys. Rev. B 1994, 50, 15606–15622. [Google Scholar] [CrossRef]
- Dresselhaus, M.S.; Dresselhaus, G.; Eklund, P.C. Science of Fullerenes and Carbon Nanotubes; Academic Press: New York, NY, USA, 1996. [Google Scholar]
- Kresse, G.; Marsman, M.; Furthmüller, J. Vienna Ab-initio Simulation Package—VASP the Guide; Universität Wien: Wien, Austria, 2012. [Google Scholar]
- Car, R.; Parrinello, M. Molecular Dynamics: An ab initio Electronic Structure and Molecular Dynamics Program, Version 3.13.1. The CPMD Consortium, 27 June 2008. Available online: http://www.cpmd.org (accessed on 10 February 2016).
- Car, R.; Parrinello, M. Unified approach for molecular dynamics and density-functional theory. Phys. Rev. Lett. 1985, 55, 2471–2474. [Google Scholar] [CrossRef] [PubMed]
- Ker, A.; Todorov, E.; Rousseau, R.; Uehara, K.; Lannuzel, F.X.; Tse, J.S. Structure and phase stability of binary zintl-phase compounds: Lithium–group 13 intermetallics and metal-doped group 14 clathrate compounds. Chem. Eur. J. 2002, 8, 2787–2798. [Google Scholar] [CrossRef]
- Bernasconi, M.; Gaito, S.; Benedek, G. Clathrates as effective p-type and n-type tetrahedral carbon semiconductors. Phys. Rev. B 2000, 61, 12689–12692. [Google Scholar] [CrossRef]
- Iqbal, Z.; Zhang, Y.; Grebel, H.; Vijayalakshmi, S.; Lahamer, A.; Benedek, G.; Bernasconi, M.; Cariboni, J.; Spagnolatti, I.; Sharma, R.; et al. Evidence for a solid phase of dodecahedral C20. Eur. Phys. J. B Condens. Matter Complex Syst. 2003, 31, 509–515. [Google Scholar] [CrossRef]
- Piskoti, C.; Yarger, J.; Zettl, A. C36, a new carbon solid. Nature 1998, 393, 771–774. [Google Scholar] [CrossRef]
- Tsujii, N.; Roudebush, J.H.; Zevalkink, A.; Cox-Uvarov, C.A.; Snyder, G.J.; Kauzlarich, S.M. Phase stability and chemical composition dependence of the thermoelectric properties of the type-I clathrate Ba8A1xSi46−x (8≤x≤15). J. Solid State Chem. 2011, 184, 1293–1303. [Google Scholar] [CrossRef]
- Chan, K.S.; Miller, M.A.; Liang, W.; Ellis-Terrell, C. Computational design and synthesis of nitrogen-substituted carbon and silicon clathrates. Mater. Res. Lett. 2014, 2, 70–75. [Google Scholar] [CrossRef]
- Zeng, T.; Hoffmann, R.; Nesper, R.; Ashcroft, N.W.; Strobel, T.A.; Proserpio, D.M. Li-filled, B-substituted carbon clathrates. J. Am. Chem. Soc. 2015, 137, 12639–12652. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.S.; Miller, M.A.; Peng, X. First principles computational study of hydrogen storage in silicon clathrates. Mater. Res. Lett. 2018, 6, 72–78. [Google Scholar] [CrossRef]
- Department of Energy. US Drive Target Explanation Document: Onboard Hydrogen Storage for Light-Duty Fuel Cell Vehicles; Department of Energy: Washington, DC, USA, 2015.
- Chan, K.S.; Peng, X. First-principles study of electronic structure of type I hybrid carbon-silicon clathrates. J. Electron. Mater. 2016, 45, 4246–4256. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef] [PubMed]
- Kresse, G.; Joubert, D. From ultrasoft pseudopotentials to projector augmented-wave method. Phys. Rev. B 1999, 59, 1758–1775. [Google Scholar] [CrossRef]
- Blöchl, P.E. Projected augmented-wave method. Phys. Rev. B 1994, 50, 17953–17979. [Google Scholar] [CrossRef]
- Heyd, J.; Peralta, J.E.; Scuseria, G.E.; Martin, R.L. Energy band gaps and lattice parameters evaluated with the Heyd-Scuseria-Ernzerhof screened hydrid functional. J. Chem. Phys. 2005, 123, 174101. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Raghavan, R.; Wagner, N.A.; Davidowski, S.K.; Baggetto, L.; Zhao, R.; Cheng, Q.; Yarger, J.L.; Veith, G.M.; Ellis-Terrell, C.; et al. Type I clathrates as novel silicon anodes: An electrochemical and structural investigation. Adv. Sci. 2015, 2, 1500057. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.S.; Miller, M.A.; Ellis-Terrell, C.; Chan, C.K. Synthesis and characterization of empty silicon clathrates for anode applications in Li-ion batteries. MRS Adv. 2016, 1, 3043–3048. [Google Scholar] [CrossRef]
- Chan, K.S.; Miller, M.A.; Liang, W.; Ellis-Terrell, C.; Chan, C.K. First principles and experimental studies of empty Si46 as anode materials for Li-ion batteries. J. Mater. Res. 2016, 31, 3657–3665. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, X.G.; Zhang, H.L.; Zhao, Z.G.; Li, F.; Liu, C.; Cheng, H.M. Composite anode mateiral of silicon/graphite/carbon nanotubes for Li-ion batteries. Electrochim. Acta 2006, 51, 4994–5000. [Google Scholar] [CrossRef]
- Yoon, T.; Nguyen, C.C.; Seo, D.M.; Lucht, B.L. Capacity fading mechanisms of silicon nanoparticle negative electrodes for Lithium ion batteries. J. Electrochem. Soc. 2015, 162, A2325–A2330. [Google Scholar] [CrossRef]
- Wang, S.Q.; Ye, H.Q. Ab initio elastic constants for the lonsdaleite phases of C, Si and Ge. J. Phys. Condens. Matter 2003, 15, 5307. [Google Scholar] [CrossRef]
- He, J.; Guo, L.; Guo, X.; Liu, R.; Tian, Y.; Wang, H.; Gao, C. Predicting hardness of dense C3N4 polymorphs. Appl. Phys. Lett. 2006, 88, 101906. [Google Scholar] [CrossRef]
- Teter, D.M. Computational alchemy: The search for new superhard materials. MRS Bull. 1998, 23, 22–27. [Google Scholar] [CrossRef]
- He, H.; Sekine, T.; Kobayashi, T.; Hirosaki, H.; Suzuki, I. Shock-induced phase transition of β−Si3N4 to c−Si3N4. Phys. Rev. B 2000, 62, 11412–11417. [Google Scholar] [CrossRef]
- Jiang, J.Z.; Lindelov, H.; Gerward, L.; Ståhl, K.; Recio, J.M.; Mori-Sanchez, P.; Carlson, S.; Mezouar, M.; Dooryhee, E.; Fitch, A.; Frost, D.J. Compressibility and thermal expansion of cubic silicon nitride. Phys. Rev. B 2002, 65, 161202. [Google Scholar] [CrossRef]
- Carnahan, R.D. Elastic properties of silicon carbide. J. Am. Ceram. Soc. 1968, 51, 223–224. [Google Scholar] [CrossRef]
- Sophisticated Alloys. Available online: www.alloys.com (accessed on 9 November 2017).
- Guloy, A.M.; Ramlau, R.; Tang, Z.; Schnelle, W.; Baitinger, M.; Grin, Y. A guest-free germanium clathrate. Nature 2006, 443, 320–323. [Google Scholar] [CrossRef] [PubMed]

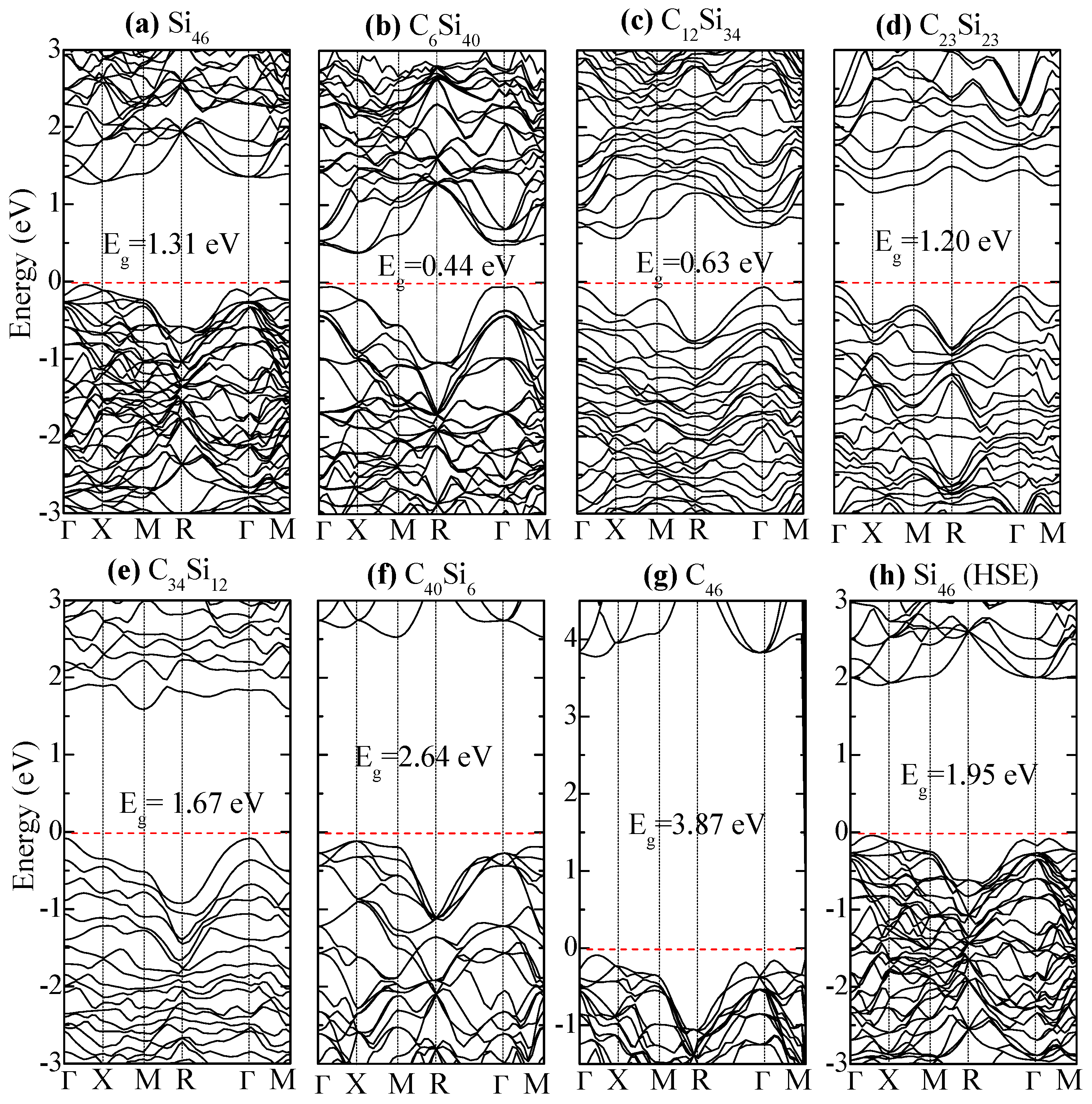
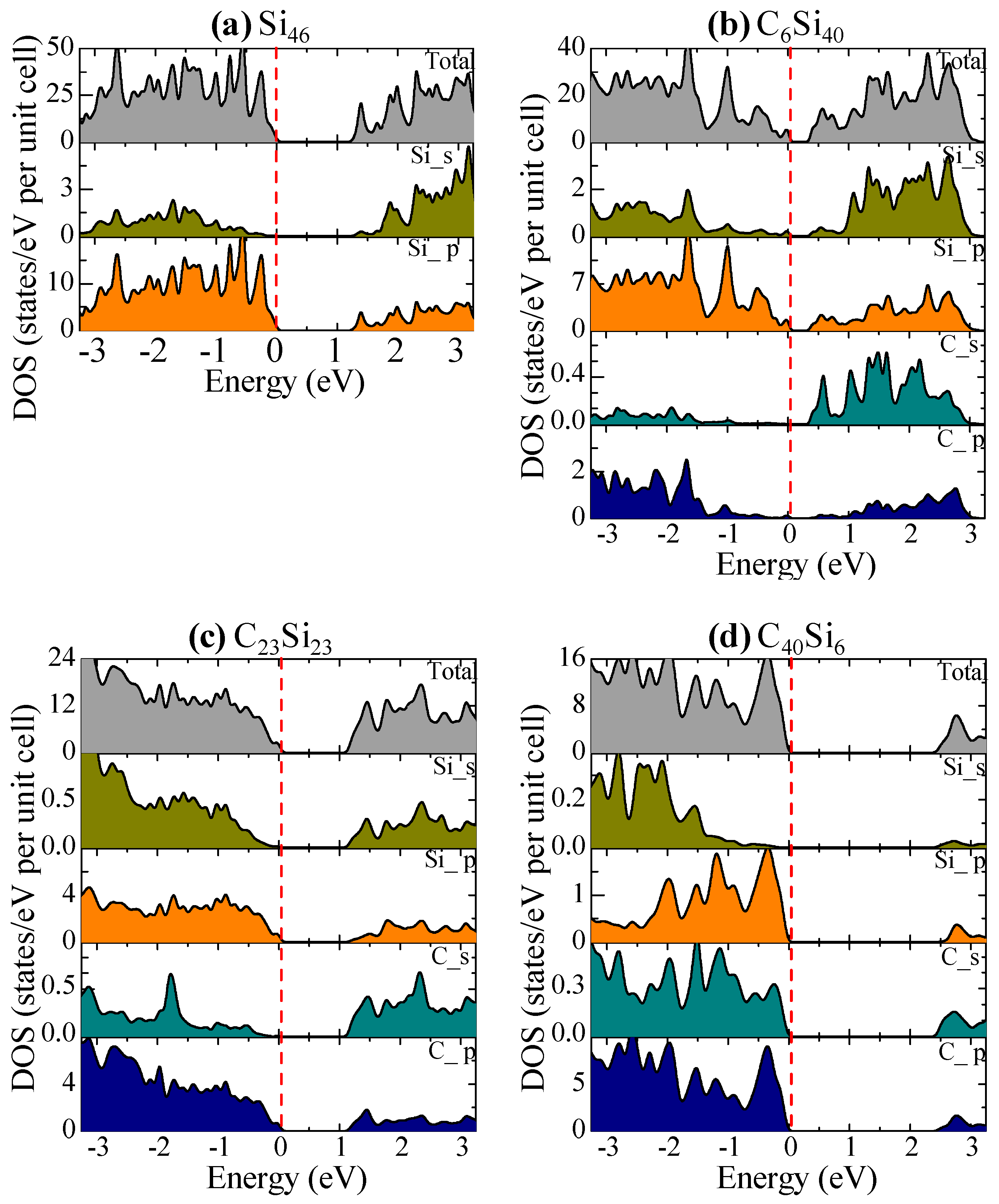
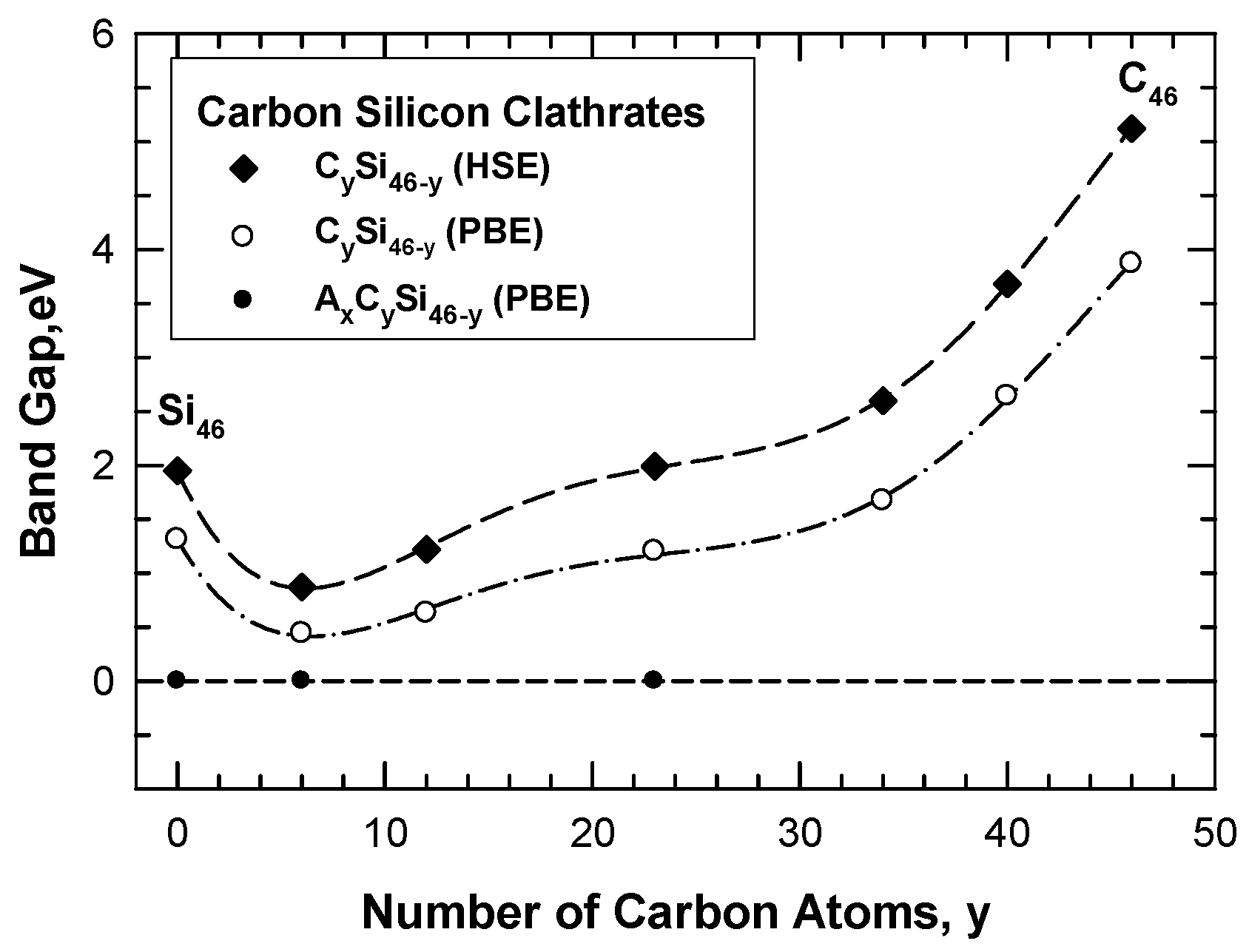
| Structure | Method | V0 (Å3atom) | a0 (Å) | c0 (Å) | Ecoh, eV/Atom | ΔEf, eV/Atom |
|---|---|---|---|---|---|---|
| fcc-C136 | VASP a [2] | 6.601 | 9.647 | −7.709 [2] | 0.074 | |
| DMOL b [6] | 6.688 | 9.689 | N.A. | −7.263 [6] | 0.087 | |
| CPMD c [5] | 0.07 | |||||
| sc-C46 | VASP [2] | 6.543 | 6.702 | N.A. | −7.677 [2] | 0.106 |
| DMOL [6] | 6.604 | 6.722 | N.A | −7.235 | 0.115 | |
| VASP [7] | 6.526 | 6.696 | N.A | 0.09 | ||
| VASP [8] | 6.6973 | 0.08 | ||||
| CPMD [8] | 6.7138 | 0.14 | ||||
| CPMD [5] | 0.09 | |||||
| CRYSTAL95 [9] d | 6.259 | 6.7029 | 0.20 [3] | |||
| hcp-C40 | VASP [2] | 6.609 | 6.695 | 6.809 | −7.656 | 0.127 |
| DMOL [6] | 6.697 | 6.725 | 6.839 | −7.213 | −0.013 | |
| Diamond | VASP [2] | 5.705 | 3.574 | N.A. | −7.783 | 0 |
| DMOL [6] | 5.787 | 3.591 | −7.350 | 0 | ||
| Expt. [10,11] | 5.673 | 3.567 | −7.20 | 0 | ||
| Graphite | VASP [2] | 8.878 | 2.470 | 6.721 | −7.916 | −0.133 |
| DMOL [6] | 8.983 | 2.479 | 6.747 | −7.516 | −0.166 | |
| Expt. [10,11] | 8.803 | 2.662 | 6.708 | −7.40 | −0.20 |
| Compound | ao, Å | ΔEf, eV/Atom |
|---|---|---|
| C46 | 6.6973 | 0.0815 |
| C18N24 | 6.8622 | −0.1377 |
| C19N27 | 6.8148 | 0.1803 |
| C20N26 | 6.8502 | 0.2144 |
| C23N23 | 6.6596 | 0.3416 |
| Li2C18N24 | 7.3192 | −0.25 |
| Li6C18N24 | 7.3336 | −0.2742 |
| Li8C18N23 | 7.3979 | −0.3051 |
| Li8C23N23 | 7.2716 | −0.0578 |
| Li12C18N24 | 8.1592 | −0.5765 |
| Li24C18N24 | 8.6003 | −0.6192 |
| Li48C18N28 | 9.3206 | −0.4832 |
| Compound | Structure | B, GPa |
|---|---|---|
| C46 | Type I Clathrate (Simple cubic) | 373.7 |
| 371 [1] | ||
| 363.7 [2] | ||
| 409 [3] | ||
| C18N24 | Type I Clathrate | 257.35 |
| C23N23 | Type I Clathrate | 311.6 |
| Li8C23N23 | Type I Clathrate | 245.18 |
| Si23C23 | Type I Clathrate | 124.03 |
| Si6C40 | Type I Clathrate | 196.8 |
| Si18N24 | Type I Clathrate | 96.08 |
| Si23N23 | Type I Clathrate | 114.13 |
| C6Si40 | Type I Clathrate | 62.9 |
| C (Diamond) | Diamond cubic | 438.8 [35] |
| 442 * [36] | ||
| α-C3N4 | Hexagonal | 378.7 [35] |
| β-C3N4 | Cubic spinel | 419.1 [36] |
| c-Si3N4 | Cubic spinel | 300 [37] |
| 300 * [38] | ||
| SiC | Cubic (Zinc blend) | 225 * [39] |
| Composition | Target Compound | Actual Compound Produced | Type I Clathrate | Yield, % |
|---|---|---|---|---|
| 1 | Na2Li6Al10C6Si30 | None (all sublimed except Si) | No | 0 |
| 2 | Li8Al10C6Si30 | Si, SiC, and AlLiSi | No | 0 |
| 3 | Ba8C6Si40 | BaSi2, Si, and graphite | No | 0 |
| 4 | Ba8C20Si26 | Ba, SiC, BaSi2, Si, and Ba8C20Si26 | Yes (Ba8C20Si26) | 39 |
| 5 | Ba8C6Si40 | Ba, SiC, BaSi2, Si, and Ba8C6Si40 | Yes (Ba8C6Si40) | 26 |
| 6 | Ba8C23Si23 | Ba, SiC, BaSi2, Si, and Ba8C23Si23 | Yes (Ba8C23Si23) | 16 |
| Composition | Target Compound | Actual Compound Produced | Type I Clathrate |
|---|---|---|---|
| 1 | Mg8N18C28 | MgO, g-C2N4, and MgCN2 | No |
| 2 | Ba8N18C24 | Ba, g-C3N4, and Ba8C18N24 | Yes (Ba8C18N24) |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chan, K.S. Hybrid Carbon-Based Clathrates for Energy Storage. C 2018, 4, 7. https://doi.org/10.3390/c4010007
Chan KS. Hybrid Carbon-Based Clathrates for Energy Storage. C. 2018; 4(1):7. https://doi.org/10.3390/c4010007
Chicago/Turabian StyleChan, Kwai S. 2018. "Hybrid Carbon-Based Clathrates for Energy Storage" C 4, no. 1: 7. https://doi.org/10.3390/c4010007
APA StyleChan, K. S. (2018). Hybrid Carbon-Based Clathrates for Energy Storage. C, 4(1), 7. https://doi.org/10.3390/c4010007




