Non-Coding RNAs: Foes or Friends for Targeting Tumor Microenvironment
Abstract
1. Introduction
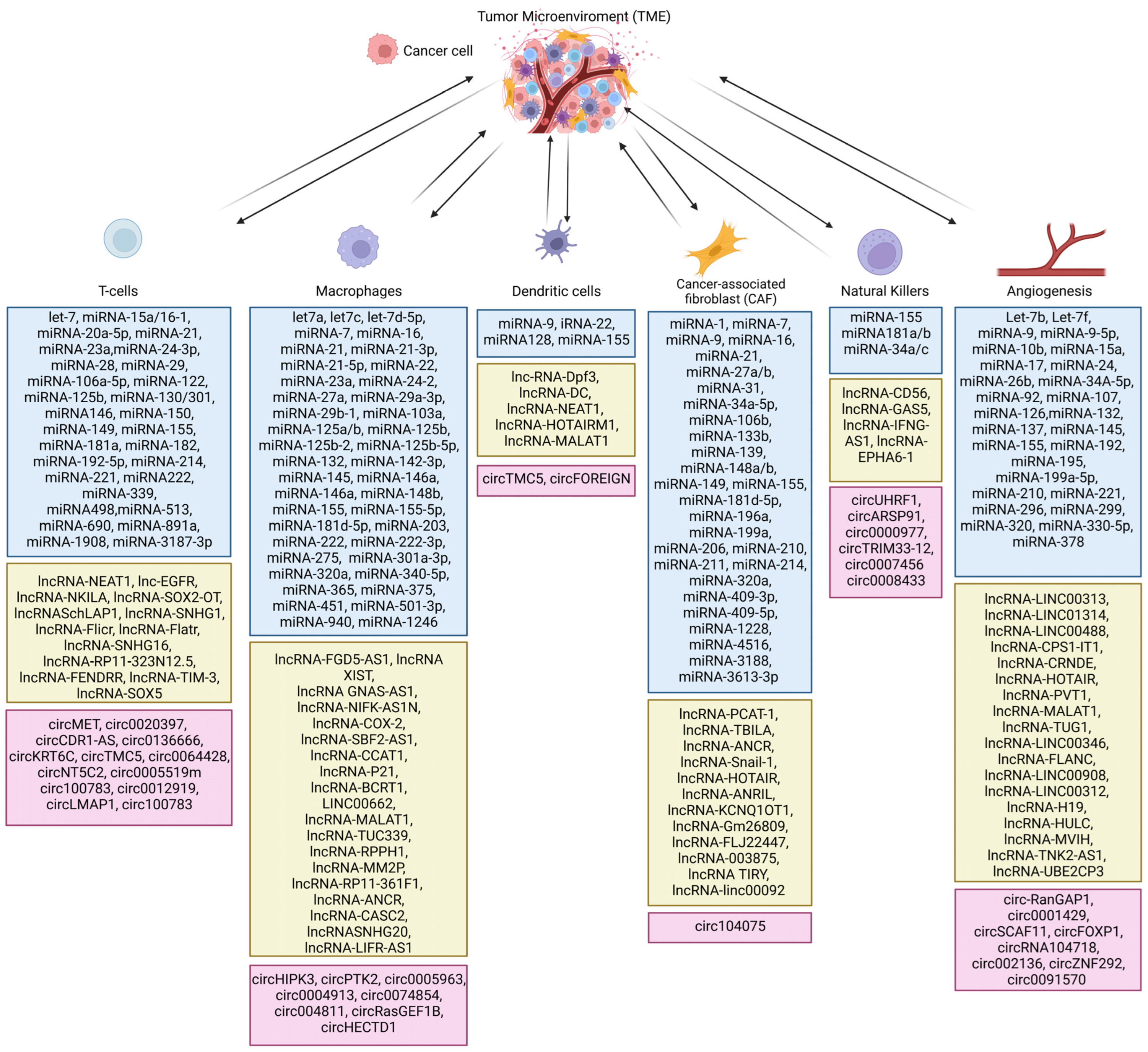
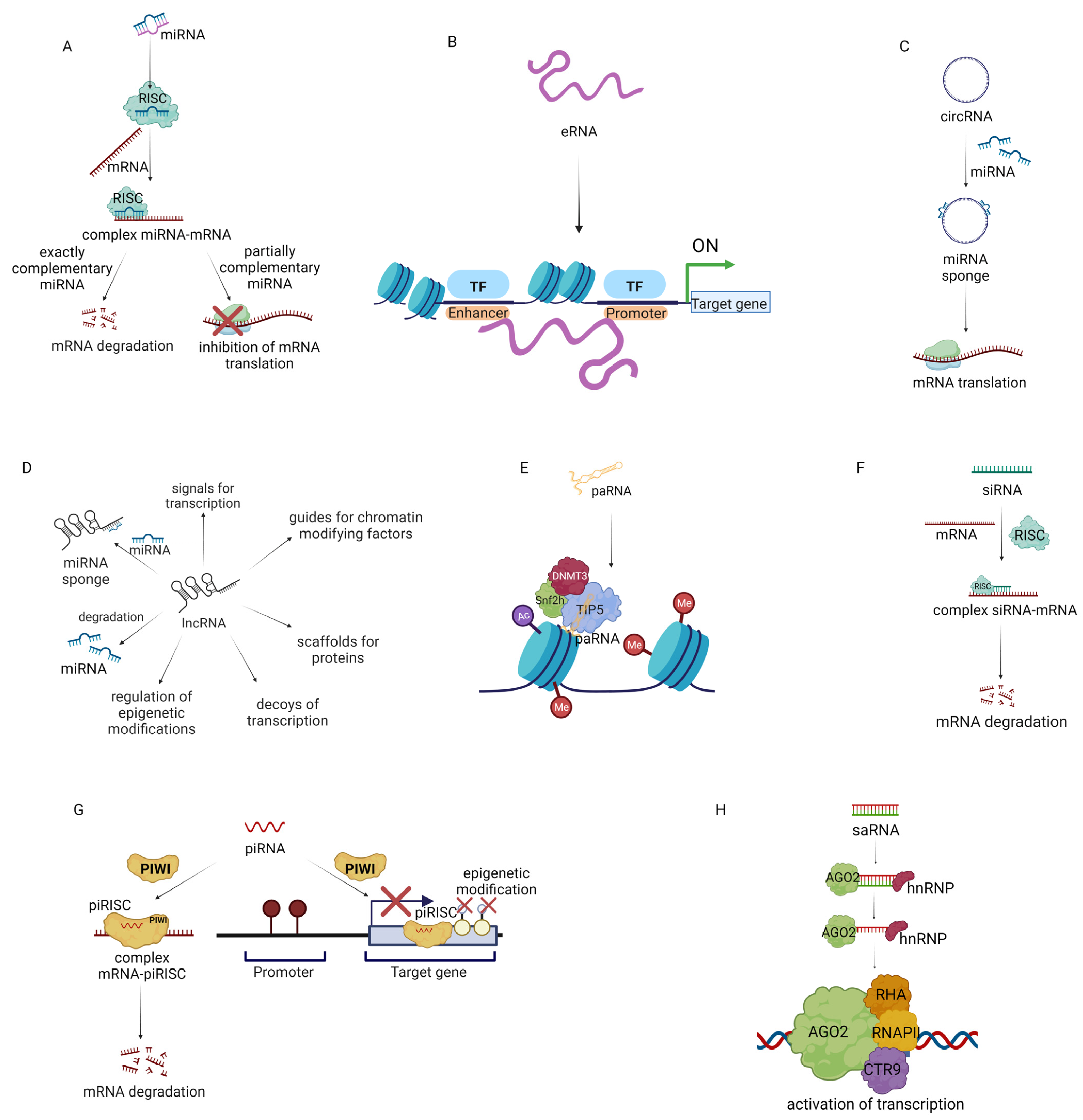
2. Role of Non-Coding RNAs in TME
2.1. Cytokines and Non-Coding RNA Intertwined in TME
2.2. Exosomes as External and Internal Carriers of Non-Coding RNAs in TME
2.3. Role of Non-Coding RNA in Cancer-Associated Fibroblasts (CAFs) in TME
2.4. Non-Coding RNA in the Regulation of Response of Macrophages in TME
2.5. Non-Coding RNAs in the Regulation of T-Cell Activity in the TME
2.6. Non-Coding RNAs in the Regulation of B Cells in TME
2.7. Non-Coding RNA in the Regulation of EMT in TME
3. RNA Sequencing to Characterize ncRNAs in the TME
3.1. RNA Sequencing for Breast Cancer
3.2. RNA Sequencing for Lung Cancer
3.3. RNA Sequencing of Colorectal Cancer
3.4. RNA Sequencing of Ovarian Cancer
3.5. RNA Sequencing of Prostate Cancer
3.6. RNA Sequencing of Gastric Cancer
3.7. RNA Sequencing of Pediatric Cancers
3.7.1. RNA Sequencing of Pediatric Leukemias
3.7.2. RNA Sequencing of Pediatric Brain Tumors
4. ncRNA Promising Drugs and Drug Targets in Cancer Treatment
4.1. ncRNA-Based Therapeutics in Cancer Treatment
4.1.1. MiRNA-Based Therapeutics for Cancer Treatment
4.1.2. eRNA-Based Therapeutics for Cancer Treatment
4.1.3. CircRNA-Based Therapeutics for Cancer Treatment
4.1.4. lncRNA-Based Therapeutics for Cancer Treatment
4.1.5. siRNA-Based Therapeutics for Cancer Treatment
4.1.6. piRNA-Based Therapeutics for Cancer Treatment
4.1.7. saRNA Small Activating RNA-Based Therapeutics for Cancer Treatment
4.2. Therapeutics Targeting ncRNAs in Cancer Treatment
5. Achievements and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AI | Artificial Intelligence |
| ALL | Acute Lymphoblastic Leukemia |
| AML | Acute Myeloid Leukemia |
| Bru | 5′-Bromouridine |
| Bulk RNA-seq | Bulk RNA Sequencing |
| CAF | Cancer-Associated Fibroblasts |
| Cas9n | Cas9 nickase |
| CHIP | Chromatin Immunoprecipitation |
| circRNA | Circular RNA |
| CML | Chronic Myeloid Leukemia |
| CTR9 | Component of PAF complex |
| DC | Dendritic Cells |
| DLBC | Diffuse Large B Cell Lymphoma |
| EMT | Epithelial-Mesenchymal Transition |
| ERG-TMPRSS2 | Ets-Related Gene—Transmembrane Protease Serine 2 |
| eRNA | Enhancer RNA |
| ESCA | Esophageal Carcinoma |
| EU | Ethynyluridine |
| GD-PEG-ECO | arginine/glycine/aspartate peptide—polyethylene glycol-1-aminoethyliminobis[N-oleicylcysteinyl-1-aminoethylpropionamide |
| HBOC | Hereditary Breast And Ovarian Cancer Predisposition Syndrome |
| hnRNP | Heterogeneous Nuclear Ribonucleoprotein |
| HNSCC | Head And Neck Squamous Cell Carcinoma |
| IL-1Ra | Interleukin-1 Receptor Antagonist |
| KICH | Kidney Chromophobe |
| LGG | Brain Lower Grade Glioma |
| lncRNA | Long Non-Coding RNA |
| LUAD | Lung Adenocarcinoma |
| LUSC | Lung Squamous Cell Carcinoma |
| LUSC | Lung Squamous Cell Carcinoma |
| miRNA | Micro-RNA |
| ncRNA | Non-Coding RNA |
| NK | Natural Killers |
| NSCLC | Non-Small Cell Lung Cancer |
| PAMAM | Poly(Amidoamine) |
| paRNA | Promoter-Associated RNA |
| pBs | Peripheral Blood Cells |
| pDCs | Plasmacytoid Dendritic Cells |
| piRNA | PIWI-Interacting RNA |
| RHA | RNA helicase A |
| RNAP II | RNA polymerase II |
| rRNA | Ribosomal RNA |
| RT-qPCR | Reverse Transcription–Quantitative Polymerase Chain Reaction |
| s4U | 4-Thiouridine |
| saRNA | Small activating RNA |
| SCLC | Small Lung Cell Cancer |
| sc-RNA-seq | Single-Cell RNA Sequencing |
| siRNA | Small Interfering RNA |
| SKCM | Skin Cutaneous Melanoma |
| snoRNA | Small Nucleolar RNA |
| snRNA | Small Nuclear RNA |
| sp-RNA-seq | Spatial RNA Sequencing |
| TERC | Telomerase RNA |
| THCA | Thyroid Carcinoma |
| TIMER | Tumor Immune Estimation Resource |
| TME | Tumor Microenvironment |
| TNBC | Triple-Negative Breast Cancer |
| TNF | Tumor Necrosis Factors |
| tRF | TRNA Derived Fragments |
| tRNA | Transfer RNA |
| UVM | Uveal Melanoma |
| XAI | Explainable Artificial Intelligence |
References
- Kikutake, C.; Yoshihara, M.; Suyama, M. Pan-cancer analysis of non-coding recurrent mutations and their possible involvement in cancer pathogenesis. NAR Cancer 2021, 3, zcab008. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Wu, W.; Chen, Q.; Chen, M. Non-Coding RNAs and their Integrated Networks. J. Integr. Bioinform. 2019, 16, 20190027. [Google Scholar] [CrossRef]
- Conesa, A.; Madrigal, P.; Tarazona, S.; Gomez-Cabrero, D.; Cervera, A.; McPherson, A.; Szcześniak, M.W.; Gaffney, D.J.; Elo, L.L.; Zhang, X.; et al. A survey of best practices for RNA-seq data analysis. Genome Biol. 2016, 17, 13. [Google Scholar] [CrossRef] [PubMed]
- Kaikkonen, M.U.; Lam, M.T.Y.; Glass, C.K. Non-coding RNAs as regulators of gene expression and epigenetics. Cardiovasc. Res. 2011, 90, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Yan, P.; Liang, Y.; Sun, Y.; Shen, J.; Zhou, S.; Lin, H.; Liang, X.; Cai, X. Circular RNA expression is suppressed by androgen receptor (AR)-regulated adenosine deaminase that acts on RNA (ADAR1) in human hepatocellular carcinoma. Cell Death Dis. 2017, 8, e3171. [Google Scholar] [CrossRef]
- Sun, J.G.; Li, X.B.; Yin, R.H.; Li, X.F. lncRNA VIM-AS1 promotes cell proliferation, metastasis and epithelial-mesenchymal transition by activating the Wnt/β-catenin pathway in gastric cancer. Mol. Med. Rep. 2020, 22, 4567–4578. [Google Scholar] [CrossRef]
- Mai, D.; Ding, P.; Tan, L.; Zhang, J.; Pan, Z.; Bai, R.; Li, C.; Li, M.; Zhou, Y.; Tan, W.; et al. PIWI-interacting RNA-54265 is oncogenic and a potential therapeutic target in colorectal adenocarcinoma. Theranostics 2018, 8, 5213–5230. [Google Scholar] [CrossRef]
- Alhasan, L. MiR-126 Modulates Angiogenesis in Breast Cancer by Targeting VEGF-A -mRNA. Asian Pac. J. Cancer Prev. 2019, 20, 193–197. [Google Scholar] [CrossRef]
- Smith, C.J.; Osborn, A.M. Advantages and limitations of quantitative PCR (Q-PCR)-based approaches in microbial ecology. FEMS Microbiol. Ecol. 2009, 67, 6–20. [Google Scholar] [CrossRef]
- Jaluria, P.; Konstantopoulos, K.; Betenbaugh, M.; Shiloach, J. A perspective on microarrays: Current applications, pitfalls, and potential uses. Microb. Cell Factories 2007, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Ning, B.; Shi, T. Single-Cell RNA-Seq Technologies and Related Computational Data Analysis. Front. Genet. 2019, 10, 317. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.-t.; Li, X.; Qin, P.-z.; Zhu, Y.; Xu, S.-n.; Chen, J.-p. Technical Advances in Single-Cell RNA Sequencing and Applications in Normal and Malignant Hematopoiesis. Front. Oncol. 2018, 8, 582. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, C.-Y. From bulk, single-cell to spatial RNA sequencing. Int. J. Oral Sci. 2021, 13, 36. [Google Scholar] [CrossRef]
- Duffy, E.E.; Schofield, J.A.; Simon, M.D. Gaining insight into transcriptome-wide RNA population dynamics through the chemistry of 4-thiouridine. Wiley Interdiscip. Rev. RNA 2019, 10, e1513. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, D.F.; Costa, V.M.; Silvestre, R.; Bastos, M.L.; Carvalho, F. Methods for the analysis of transcriptome dynamics. Toxicol. Res. 2019, 8, 597–612. [Google Scholar] [CrossRef]
- Park, P.J. ChIP-seq: Advantages and challenges of a maturing technology. Nat. Rev. Genet. 2009, 10, 669–680. [Google Scholar] [CrossRef]
- Labani-Motlagh, A.; Ashja-Mahdavi, M.; Loskog, A. The Tumor Microenvironment: A Milieu Hindering and Obstructing Antitumor Immune Responses. Front. Immunol. 2020, 11, 940. [Google Scholar] [CrossRef]
- Zhang, M.; Gao, D.; Shi, Y.; Wang, Y.; Joshi, R.; Yu, Q.; Liu, D.; Alotaibi, F.; Zhang, Y.; Wang, H.; et al. miR-149-3p reverses CD8(+) T-cell exhaustion by reducing inhibitory receptors and promoting cytokine secretion in breast cancer cells. Open Biol. 2019, 9, 190061. [Google Scholar] [CrossRef]
- Yim, G.W.; Lee, D.W.; Kim, J.I.; Kim, Y.T. Long Non-coding RNA LOC285194 Promotes Epithelial Ovarian Cancer Progression via the Apoptosis Signaling Pathway. In Vivo 2022, 36, 121–131. [Google Scholar] [CrossRef]
- Ren, Y.; Zhou, X.; Mei, M.; Yuan, X.-B.; Han, L.; Wang, G.-X.; Jia, Z.-F.; Xu, P.; Pu, P.-Y.; Kang, C.-S. MicroRNA-21 inhibitor sensitizes human glioblastoma cells U251 (PTEN-mutant) and LN229 (PTEN-wild type) to taxol. BMC Cancer 2010, 10, 27. [Google Scholar] [CrossRef]
- Hattab, D.; Gazzali, A.M.; Bakhtiar, A. Clinical Advances of siRNA-Based Nanotherapeutics for Cancer Treatment. Pharmaceutics 2021, 13, 1009. [Google Scholar] [CrossRef]
- Benedetti, A.; Turco, C.; Fontemaggi, G.; Fazi, F. Non-Coding RNAs in the Crosstalk between Breast Cancer Cells and Tumor-Associated Macrophages. Noncoding RNA 2022, 8, 16. [Google Scholar] [CrossRef]
- Wang, W.-T.; Han, C.; Sun, Y.-M.; Chen, T.-Q.; Chen, Y.-Q. Noncoding RNAs in cancer therapy resistance and targeted drug development. J. Hematol. Oncol. 2019, 12, 55. [Google Scholar] [CrossRef]
- Taheri, M.; Barth, D.A.; Kargl, J.; Rezaei, O.; Ghafouri-Fard, S.; Pichler, M. Emerging Role of Non-Coding RNAs in Regulation of T-Lymphocyte Function. Front. Immunol. 2021, 12, 756042. [Google Scholar] [CrossRef] [PubMed]
- Carlos-Reyes, Á.; Romero-Garcia, S.; Contreras-Sanzón, E.; Ruiz, V.; Prado-Garcia, H. Role of Circular RNAs in the Regulation of Immune Cells in Response to Cancer Therapies. Front. Genet. 2022, 13, 823238. [Google Scholar] [CrossRef]
- Nguyen, M.-H.T.; Luo, Y.-H.; Li, A.-L.; Tsai, J.-C.; Wu, K.-L.; Chung, P.-J.; Ma, N. miRNA as a Modulator of Immunotherapy and Immune Response in Melanoma. Biomolecules 2021, 11, 1648. [Google Scholar] [CrossRef]
- Yang, F.; Ning, Z.; Ma, L.; Liu, W.; Shao, C.; Shu, Y.; Shen, H. Exosomal miRNAs and miRNA dysregulation in cancer-associated fibroblasts. Mol. Cancer 2017, 16, 148. [Google Scholar] [CrossRef] [PubMed]
- Annese, T.; Tamma, R.; De Giorgis, M.; Ribatti, D. microRNAs Biogenesis, Functions and Role in Tumor Angiogenesis. Front. Oncol. 2020, 10, 581007. [Google Scholar] [CrossRef] [PubMed]
- Pi, Y.N.; Qi, W.C.; Xia, B.R.; Lou, G.; Jin, W.L. Long Non-Coding RNAs in the Tumor Immune Microenvironment: Biological Properties and Therapeutic Potential. Front. Immunol. 2021, 12, 697083. [Google Scholar] [CrossRef]
- Jin, K.-T.; Yao, J.-Y.; Fang, X.-L.; Di, H.; Ma, Y.-Y. Roles of lncRNAs in cancer: Focusing on angiogenesis. Life Sci. 2020, 252, 117647. [Google Scholar] [CrossRef] [PubMed]
- Mabeta, P.; Hull, R.; Dlamini, Z. LncRNAs and the Angiogenic Switch in Cancer: Clinical Significance and Therapeutic Opportunities. Genes 2022, 13, 152. [Google Scholar] [CrossRef] [PubMed]
- Ti, W.; Wang, J.; Cheng, Y. The Interaction between Long Non-Coding RNAs and Cancer-Associated Fibroblasts in Lung Cancer. Front. Cell Dev. Biol. 2021, 9, 714125. [Google Scholar] [CrossRef] [PubMed]
- Fang, Z.; Xu, J.; Zhang, B.; Wang, W.; Liu, J.; Liang, C.; Hua, J.; Meng, Q.; Yu, X.; Shi, S. The promising role of noncoding RNAs in cancer-associated fibroblasts: An overview of current status and future perspectives. J. Hematol. Oncol. 2020, 13, 154. [Google Scholar] [CrossRef]
- Chen, C.; Xia, C.; Tang, H.; Jiang, Y.; Wang, S.; Zhang, X.; Huang, T.; Yuan, X.; Wang, J.; Peng, L. Circular RNAs Involve in Immunity of Digestive Cancers from Bench to Bedside: A Review. Front. Immunol. 2022, 13, 833058. [Google Scholar] [CrossRef]
- Song, H.; Liu, Q.; Liao, Q. Circular RNA and tumor microenvironment. Cancer Cell Int. 2020, 20, 211. [Google Scholar] [CrossRef]
- Meng, D.; Jia, R.; Yuan, S.; Wei, M.; Bao, X.; Zhu, C.; Wang, W.; Li, Z. Research progress on the circRNA-mediated regulation of tumor angiogenesis through ceRNA mechanisms (Review). Oncol. Rep. 2023, 49, 12. [Google Scholar] [CrossRef]
- Li, Z.; Wang, H.; Zhang, Z.; Meng, X.; Liu, D.; Tang, Y. Germline and somatic mutation profile in Cancer patients revealed by a medium-sized pan-Cancer panel. Genomics 2021, 113, 1930–1939. [Google Scholar] [CrossRef]
- Pessôa-Pereira, D.; Evangelista, A.F.; Causin, R.L.; da Costa Vieira, R.A.; Abrahão-Machado, L.F.; Santana, I.V.V.; da Silva, V.D.; de Souza, K.C.B.; de Oliveira-Silva, R.J.; Fernandes, G.C.; et al. miRNA expression profiling of hereditary breast tumors from BRCA1- and BRCA2-germline mutation carriers in Brazil. BMC Cancer 2020, 20, 143. [Google Scholar] [CrossRef]
- Urbanek-Trzeciak, M.O.; Galka-Marciniak, P.; Nawrocka, P.M.; Kowal, E.; Szwec, S.; Giefing, M.; Kozlowski, P. Pan-cancer analysis of somatic mutations in miRNA genes. EBioMedicine 2020, 61, 103051. [Google Scholar] [CrossRef] [PubMed]
- Calin, G.A.; Croce, C.M. MicroRNA-cancer connection: The beginning of a new tale. Cancer Res. 2006, 66, 7390–7394. [Google Scholar] [CrossRef]
- Ziebarth, J.D.; Bhattacharya, A.; Cui, Y. Integrative Analysis of Somatic Mutations Altering MicroRNA Targeting in Cancer Genomes. PLoS ONE 2012, 7, e47137. [Google Scholar] [CrossRef] [PubMed]
- Baghban, R.; Roshangar, L.; Jahanban-Esfahlan, R.; Seidi, K.; Ebrahimi-Kalan, A.; Jaymand, M.; Kolahian, S.; Javaheri, T.; Zare, P. Tumor microenvironment complexity and therapeutic implications at a glance. Cell Commun. Signal. 2020, 18, 59. [Google Scholar] [CrossRef] [PubMed]
- Roche, J. The Epithelial-to-Mesenchymal Transition in Cancer. Cancers 2018, 10, 52. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, J.V.; Cobucci, R.N.O.; Jatobá, C.A.N.; de Medeiros Fernandes, T.A.A.; de Azevedo, J.W.V.; de Araújo, J.M.G. The Role of the Mediators of Inflammation in Cancer Development. Pathol. Oncol. Res. 2015, 21, 527–534. [Google Scholar] [CrossRef]
- Dinarello, C.A. Historical insights into cytokines. Eur. J. Immunol. 2007, 37, S34–S45. [Google Scholar] [CrossRef]
- Hughes, C.E.; Nibbs, R.J.B. A guide to chemokines and their receptors. FEBS J. 2018, 285, 2944–2971. [Google Scholar] [CrossRef]
- Briukhovetska, D.; Dörr, J.; Endres, S.; Libby, P.; Dinarello, C.A.; Kobold, S. Interleukins in cancer: From biology to therapy. Nat. Rev. Cancer 2021, 21, 481–499. [Google Scholar] [CrossRef]
- Abdolvahab, M.H.; Darvishi, B.; Zarei, M.; Majidzadeh, A.K.; Farahmand, L. Interferons: Role in cancer therapy. Immunotherapy 2020, 12, 833–855. [Google Scholar] [CrossRef]
- Mercogliano, M.F.; Bruni, S.; Elizalde, P.V.; Schillaci, R. Tumor Necrosis Factor α Blockade: An Opportunity to Tackle Breast Cancer. Front. Oncol. 2020, 10, 584. [Google Scholar] [CrossRef]
- Chow, M.T.; Luster, A.D. Chemokines in cancer. Cancer Immunol. Res. 2014, 2, 1125–1131. [Google Scholar] [CrossRef]
- Aldinucci, D.; Borghese, C.; Casagrande, N. The CCL5/CCR5 Axis in Cancer Progression. Cancers 2020, 12, 1765. [Google Scholar] [CrossRef]
- Qiu, X.; Li, C.; Chen, H. Long Noncoding RNA ZFAS1 Promotes Progression of Oral Squamous Cell Carcinoma through Targeting miR-6499-3p/CCL5 Axis. In Vivo 2021, 35, 3211–3220. [Google Scholar] [CrossRef] [PubMed]
- Dutcher, J.P.; Schwartzentruber, D.J.; Kaufman, H.L.; Agarwala, S.S.; Tarhini, A.A.; Lowder, J.N.; Atkins, M.B. High dose interleukin-2 (Aldesleukin)—Expert consensus on best management practices-2014. J. ImmunoTher. Cancer 2014, 2, 26. [Google Scholar] [CrossRef] [PubMed]
- Clinical Trials Associated with Interleukins and Cancer. Available online: https://clinicaltrials.gov/ct2/results?term=interleukin-+cancer (accessed on 21 March 2023).
- Kumari, N.; Dwarakanath, B.S.; Das, A.; Bhatt, A.N. Role of interleukin-6 in cancer progression and therapeutic resistance. Tumour Biol. 2016, 37, 11553–11572. [Google Scholar] [CrossRef]
- Masuda, K.; Ripley, B.; Nishimura, R.; Mino, T.; Takeuchi, O.; Shioi, G.; Kiyonari, H.; Kishimoto, T. Arid5a controls IL-6 mRNA stability, which contributes to elevation of IL-6 level in vivo. Proc. Natl. Acad. Sci. USA 2013, 110, 9409–9414. [Google Scholar] [CrossRef] [PubMed]
- Nyati, K.K.; Hashimoto, S.; Singh, S.K.; Tekguc, M.; Metwally, H.; Liu, Y.C.; Okuzaki, D.; Gemechu, Y.; Kang, S.; Kishimoto, T. The novel long noncoding RNA AU021063, induced by IL-6/Arid5a signaling, exacerbates breast cancer invasion and metastasis by stabilizing Trib3 and activating the Mek/Erk pathway. Cancer Lett. 2021, 520, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Fenton, S.E.; Saleiro, D.; Platanias, L.C. Type I and II Interferons in the Anti-Tumor Immune Response. Cancers 2021, 13, 1037. [Google Scholar] [CrossRef]
- Jorgovanovic, D.; Song, M.; Wang, L.; Zhang, Y. Roles of IFN-γ in tumor progression and regression: A review. Biomark. Res. 2020, 8, 49. [Google Scholar] [CrossRef]
- Mineo, M.; Lyons, S.M.; Zdioruk, M.; von Spreckelsen, N.; Ferrer-Luna, R.; Ito, H.; Alayo, Q.A.; Kharel, P.; Larsen, A.G.; Fan, W.Y.; et al. Tumor Interferon Signaling Is Regulated by a lncRNA INCR1 Transcribed from the PD-L1 Locus. Mol. Cell 2020, 78, 1207–1223.e1208. [Google Scholar] [CrossRef]
- Laha, D.; Grant, R.; Mishra, P.; Nilubol, N. The Role of Tumor Necrosis Factor in Manipulating the Immunological Response of Tumor Microenvironment. Front. Immunol. 2021, 12, 656908. [Google Scholar] [CrossRef]
- Zheng, M.; Wu, Z.; Wu, A.; Huang, Z.; He, N.; Xie, X. MiR-145 promotes TNF-α-induced apoptosis by facilitating the formation of RIP1-FADDcaspase-8 complex in triple-negative breast cancer. Tumour Biol. 2016, 37, 8599–8607. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wu, H.; Li, P.; Zhao, Y.; Liu, M.; Tang, H. NF-κB-modulated miR-130a targets TNF-α in cervical cancer cells. J. Transl. Med. 2014, 12, 155. [Google Scholar] [CrossRef] [PubMed]
- Patel, H.; Nilendu, P.; Jahagirdar, D.; Pal, J.K.; Sharma, N.K. Modulating secreted components of tumor microenvironment: A masterstroke in tumor therapeutics. Cancer Biol. Ther. 2018, 19, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Wang, L.; Zeng, X.; Schwarz, H.; Nanda, H.S.; Peng, X.; Zhou, Y. Exosomes, a New Star for Targeted Delivery. Front. Cell Dev. Biol. 2021, 9, 751079. [Google Scholar] [CrossRef]
- Tao, H.; Xu, H.; Zuo, L.; Li, C.; Qiao, G.; Guo, M.; Zheng, L.; Leitgeb, M.; Lin, X. Exosomes-coated bcl-2 siRNA inhibits the growth of digestive system tumors both in vitro and in vivo. Int. J. Biol. Macromol. 2020, 161, 470–480. [Google Scholar] [CrossRef]
- Tai, Y.L.; Chen, K.C.; Hsieh, J.T.; Shen, T.L. Exosomes in cancer development and clinical applications. Cancer Sci. 2018, 109, 2364–2374. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Li, Z.; Yu, Y.; Zeng, Q.; Cheng, Y.; Ji, W.; Xia, W.; Lu, S. Exosomal miR-499a-5p promotes cell proliferation, migration and EMT via mTOR signaling pathway in lung adenocarcinoma. Exp. Cell Res. 2019, 379, 203–213. [Google Scholar] [CrossRef]
- Li, C.; Ni, Y.-Q.; Xu, H.; Xiang, Q.-Y.; Zhao, Y.; Zhan, J.-K.; He, J.-Y.; Li, S.; Liu, Y.-S. Roles and mechanisms of exosomal non-coding RNAs in human health and diseases. Signal Transduct. Target. Ther. 2021, 6, 383. [Google Scholar] [CrossRef]
- Li, C.; Zhou, T.; Chen, J.; Li, R.; Chen, H.; Luo, S.; Chen, D.; Cai, C.; Li, W. The role of Exosomal miRNAs in cancer. J. Transl. Med. 2022, 20, 6. [Google Scholar] [CrossRef]
- Yousefi, H.; Maheronnaghsh, M.; Molaei, F.; Mashouri, L.; Reza Aref, A.; Momeny, M.; Alahari, S.K. Long noncoding RNAs and exosomal lncRNAs: Classification, and mechanisms in breast cancer metastasis and drug resistance. Oncogene 2020, 39, 953–974. [Google Scholar] [CrossRef]
- Pathania, A.S.; Challagundla, K.B. Exosomal Long Non-coding RNAs: Emerging Players in the Tumor Microenvironment. Mol. Ther. -Nucleic Acids 2021, 23, 1371–1383. [Google Scholar] [CrossRef]
- Zeng, R.; Song, X.J.; Liu, C.W.; Ye, W. LncRNA ANRIL promotes angiogenesis and thrombosis by modulating microRNA-99a and microRNA-449a in the autophagy pathway. Am. J. Transl. Res. 2019, 11, 7441–7448. [Google Scholar] [PubMed]
- Kanlikilicer, P.; Rashed, M.H.; Bayraktar, R.; Mitra, R.; Ivan, C.; Aslan, B.; Zhang, X.; Filant, J.; Silva, A.M.; Rodriguez-Aguayo, C.; et al. Ubiquitous Release of Exosomal Tumor Suppressor miR-6126 from Ovarian Cancer Cells. Cancer Res. 2016, 76, 7194–7207. [Google Scholar] [CrossRef]
- Kanlikilicer, P.; Bayraktar, R.; Denizli, M.; Rashed, M.H.; Ivan, C.; Aslan, B.; Mitra, R.; Karagoz, K.; Bayraktar, E.; Zhang, X.; et al. Exosomal miRNA confers chemo resistance via targeting Cav1/p-gp/M2-type macrophage axis in ovarian cancer. eBioMedicine 2018, 38, 100–112. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Zhang, W.; Zhang, C.; Wang, L.; Chen, H.; Xu, J. Exosomal non-coding RNAs have a significant effect on tumor metastasis. Mol. Ther. Nucleic Acids 2022, 29, 16–35. [Google Scholar] [CrossRef]
- Xie, Y.; Dang, W.; Zhang, S.; Yue, W.; Yang, L.; Zhai, X.; Yan, Q.; Lu, J. The role of exosomal noncoding RNAs in cancer. Mol. Cancer 2019, 18, 37. [Google Scholar] [CrossRef]
- Jiang, S.; Fu, R.; Shi, J.; Wu, H.; Mai, J.; Hua, X.; Chen, H.; Liu, J.; Lu, M.; Li, N. CircRNA-Mediated Regulation of Angiogenesis: A New Chapter in Cancer Biology. Front. Oncol. 2021, 11, 553706. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R. The biology and function of fibroblasts in cancer. Nat. Rev. Cancer 2016, 16, 582–598. [Google Scholar] [CrossRef]
- Nurmik, M.; Ullmann, P.; Rodriguez, F.; Haan, S.; Letellier, E. In search of definitions: Cancer-associated fibroblasts and their markers. Int. J. Cancer 2020, 146, 895–905. [Google Scholar] [CrossRef]
- Mitra, A.K.; Zillhardt, M.; Hua, Y.; Tiwari, P.; Murmann, A.E.; Peter, M.E.; Lengyel, E. MicroRNAs reprogram normal fibroblasts into cancer-associated fibroblasts in ovarian cancer. Cancer Discov. 2012, 2, 1100–1108. [Google Scholar] [CrossRef]
- Aprelikova, O.; Yu, X.; Palla, J.; Wei, B.R.; John, S.; Yi, M.; Stephens, R.; Simpson, R.M.; Risinger, J.I.; Jazaeri, A.; et al. The role of miR-31 and its target gene SATB2 in cancer-associated fibroblasts. Cell Cycle 2010, 9, 4387–4398. [Google Scholar] [CrossRef]
- Bożyk, A.; Wojas-Krawczyk, K.; Krawczyk, P.; Milanowski, J. Tumor Microenvironment-A Short Review of Cellular and Interaction Diversity. Biology 2022, 11, 929. [Google Scholar] [CrossRef] [PubMed]
- Sahai, E.; Astsaturov, I.; Cukierman, E.; DeNardo, D.G.; Egeblad, M.; Evans, R.M.; Fearon, D.; Greten, F.R.; Hingorani, S.R.; Hunter, T.; et al. A framework for advancing our understanding of cancer-associated fibroblasts. Nat. Rev. Cancer 2020, 20, 174–186. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Ji, G.; Le, X.; Wang, C.; Xu, L.; Feng, M.; Zhang, Y.; Yang, H.; Xuan, Y.; Yang, Y.; et al. Long Noncoding RNA LINC00092 Acts in Cancer-Associated Fibroblasts to Drive Glycolysis and Progression of Ovarian Cancer. Cancer Res. 2017, 77, 1369–1382. [Google Scholar] [CrossRef] [PubMed]
- Murdoch, C.; Muthana, M.; Coffelt, S.B.; Lewis, C.E. The role of myeloid cells in the promotion of tumour angiogenesis. Nat. Rev. Cancer 2008, 8, 618–631. [Google Scholar] [CrossRef]
- Lin, Y.; Xu, J.; Lan, H. Tumor-associated macrophages in tumor metastasis: Biological roles and clinical therapeutic applications. J. Hematol. Oncol. 2019, 12, 76. [Google Scholar] [CrossRef] [PubMed]
- Lewis, C.E.; Pollard, J.W. Distinct Role of Macrophages in Different Tumor Microenvironments. Cancer Res. 2006, 66, 605–612. [Google Scholar] [CrossRef]
- Aharinejad, S.; Paulus, P.; Sioud, M.; Hofmann, M.; Zins, K.; Schäfer, R.; Stanley, E.R.; Abraham, D. Colony-stimulating factor-1 blockade by antisense oligonucleotides and small interfering RNAs suppresses growth of human mammary tumor xenografts in mice. Cancer Res. 2004, 64, 5378–5384. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-González, I.; Bobien, A.; Molnar, C.; Schmid, S.; Strotbek, M.; Boerries, M.; Busch, H.; Olayioye, M.A. miR-149 Suppresses Breast Cancer Metastasis by Blocking Paracrine Interactions with Macrophages. Cancer Res. 2020, 80, 1330–1341. [Google Scholar] [CrossRef]
- Anfray, C.; Ummarino, A.; Andón, F.T.; Allavena, P. Current Strategies to Target Tumor-Associated-Macrophages to Improve Anti-Tumor Immune Responses. Cells 2019, 9, 46. [Google Scholar] [CrossRef]
- Song, Y.; Tang, C.; Yin, C. Combination antitumor immunotherapy with VEGF and PIGF siRNA via systemic delivery of multi-functionalized nanoparticles to tumor-associated macrophages and breast cancer cells. Biomaterials 2018, 185, 117–132. [Google Scholar] [CrossRef]
- Wayne, E.C.; Long, C.; Haney, M.J.; Batrakova, E.V.; Leisner, T.M.; Parise, L.V.; Kabanov, A.V. Targeted Delivery of siRNA Lipoplexes to Cancer Cells Using Macrophage Transient Horizontal Gene Transfer. Adv. Sci. 2019, 6, 1900582. [Google Scholar] [CrossRef]
- Saravia, J.; Chapman, N.M.; Chi, H. Helper T cell differentiation. Cell. Mol. Immunol. 2019, 16, 634–643. [Google Scholar] [CrossRef]
- Caminero, F.; Iqbal, Z.; Tadi, P. Histology, Cytotoxic T Cells. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Workman, C.J.; Szymczak-Workman, A.L.; Collison, L.W.; Pillai, M.R.; Vignali, D.A. The development and function of regulatory T cells. Cell Mol. Life Sci. 2009, 66, 2603–2622. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Li, Y.; Zhu, B. T-cell exhaustion in the tumor microenvironment. Cell Death Dis. 2015, 6, e1792. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Gu, W.; Li, J.; Chen, C.; Xu, Z.P. Silencing PD-1 and PD-L1 with nanoparticle-delivered small interfering RNA increases cytotoxicity of tumor-infiltrating lymphocytes. Nanomedicine 2019, 14, 955–967. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, Y.; Yu, X.; Teng, X.-L.; Ding, R.; Hu, Z.; Wang, A.; Wang, Z.; Ye, Y.; Zou, Q. ZFP91 disturbs metabolic fitness and antitumor activity of tumor-infiltrating T cells. J. Clin. Investig. 2021, 131, e144318. [Google Scholar] [CrossRef]
- Chakraborty, D.; Ivan, C.; Amero, P.; Khan, M.; Rodriguez-Aguayo, C.; Başağaoğlu, H.; Lopez-Berestein, G. Explainable Artificial Intelligence Reveals Novel Insight into Tumor Microenvironment Conditions Linked with Better Prognosis in Patients with Breast Cancer. Cancers 2021, 13, 3450. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.H.; Liu, S.Z.; Wang, G.Z.; Zhou, G.B. CXCL13 in Cancer and Other Diseases: Biological Functions, Clinical Significance, and Therapeutic Opportunities. Life 2021, 11, 1282. [Google Scholar] [CrossRef]
- Yang, M.; Lu, J.; Zhang, G.; Wang, Y.; He, M.; Xu, Q.; Xu, C.; Liu, H. CXCL13 shapes immunoactive tumor microenvironment and enhances the efficacy of PD-1 checkpoint blockade in high-grade serous ovarian cancer. J. ImmunoTher. Cancer 2021, 9, e001136. [Google Scholar] [CrossRef]
- Xia, J.; Xie, Z.; Niu, G.; Lu, Z.; Wang, Z.; Xing, Y.; Ren, J.; Hu, Z.; Hong, R.; Cao, Z.; et al. Single-cell landscape and clinical outcomes of infiltrating B cells in colorectal cancer. Immunology 2023, 168, 135–151. [Google Scholar] [CrossRef]
- Gao, Y.; Zhang, W.; Liu, C.; Li, G. miR-200 affects tamoxifen resistance in breast cancer cells through regulation of MYB. Sci. Rep. 2019, 9, 18844. [Google Scholar] [CrossRef] [PubMed]
- Kiesslich, T.; Pichler, M.; Neureiter, D. Epigenetic control of epithelial-mesenchymal-transition in human cancer. Mol. Clin. Oncol. 2013, 1, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.; Yan, S.; Lu, L.; Yao, H.; He, G.; Chen, S.; Li, Y.; Peng, X.; Cheng, Z.; Wu, M.; et al. Lovastatin Inhibits EMT and Metastasis of Triple-Negative Breast Cancer Stem Cells through Dysregulation of Cytoskeleton-Associated Proteins. Front. Oncol. 2021, 11, 656687. [Google Scholar] [CrossRef]
- Pal, B.; Chen, Y.; Vaillant, F.; Capaldo, B.D.; Joyce, R.; Song, X.; Bryant, V.L.; Penington, J.S.; Di Stefano, L.; Ribera, N.T.; et al. A single-cell RNA expression atlas of normal, preneoplastic and tumorigenic states in the human breast. EMBO J. 2021, 40, e107333. [Google Scholar] [CrossRef] [PubMed]
- Floris, M.; Olla, S.; Schlessinger, D.; Cucca, F. Genetic-Driven Druggable Target Identification and Validation. Trends Genet. 2018, 34, 558–570. [Google Scholar] [CrossRef]
- Vegliante, R.; Pastushenko, I.; Blanpain, C. Deciphering functional tumor states at single-cell resolution. EMBO J. 2022, 41, e109221. [Google Scholar] [CrossRef]
- Im, Y.; Kim, Y. A Comprehensive Overview of RNA Deconvolution Methods and Their Application. Mol. Cells 2023, 46, 99–105. [Google Scholar] [CrossRef]
- Miller, D.T.; Lee, K.; Chung, W.K.; Gordon, A.S.; Herman, G.E.; Klein, T.E.; Stewart, D.R.; Amendola, L.M.; Adelman, K.; Bale, S.J.; et al. ACMG SF v3.0 list for reporting of secondary findings in clinical exome and genome sequencing: A policy statement of the American College of Medical Genetics and Genomics (ACMG). Genet. Med. 2021, 23, 1381–1390. [Google Scholar] [CrossRef]
- Forrest, I.S.; Chaudhary, K.; Vy, H.M.T.; Petrazzini, B.O.; Bafna, S.; Jordan, D.M.; Rocheleau, G.; Loos, R.J.F.; Nadkarni, G.N.; Cho, J.H.; et al. Population-Based Penetrance of Deleterious Clinical Variants. JAMA 2022, 327, 350–359. [Google Scholar] [CrossRef]
- Kingdom, R.; Wright, C.F. Incomplete Penetrance and Variable Expressivity: From Clinical Studies to Population Cohorts. Front. Genet. 2022, 13, 920390. [Google Scholar] [CrossRef] [PubMed]
- Cook, D.; Brown, D.; Alexander, R.; March, R.; Morgan, P.; Satterthwaite, G.; Pangalos, M.N. Lessons learned from the fate of AstraZeneca’s drug pipeline: A five-dimensional framework. Nat. Rev. Drug Discov. 2014, 13, 419–431. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Yersal, O.; Barutca, S. Biological subtypes of breast cancer: Prognostic and therapeutic implications. World J. Clin. Oncol. 2014, 5, 412–424. [Google Scholar] [CrossRef]
- Jia, X.; Lei, H.; Jiang, X.; Yi, Y.; Luo, X.; Li, J.; Chen, Y.; Liu, S.; Yang, C. Identification of Crucial lncRNAs for Luminal A Breast Cancer through RNA Sequencing. Int. J. Endocrinol. 2022, 2022, 6577942. [Google Scholar] [CrossRef] [PubMed]
- Goff, S.L.; Danforth, D.N. The Role of Immune Cells in Breast Tissue and Immunotherapy for the Treatment of Breast Cancer. Clin. Breast Cancer 2021, 21, e63–e73. [Google Scholar] [CrossRef]
- Zhao, T.; Khadka, V.S.; Deng, Y. Identification of lncRNA biomarkers for lung cancer through integrative cross-platform data analyses. Aging 2020, 12, 14506–14527. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Fan, J.; He, Y.; Xiong, A.; Yu, J.; Li, Y.; Zhang, Y.; Zhao, W.; Zhou, F.; Li, W.; et al. Single-cell profiling of tumor heterogeneity and the microenvironment in advanced non-small cell lung cancer. Nat. Commun. 2021, 12, 2540. [Google Scholar] [CrossRef]
- Peng, Z.; Ye, M.; Ding, H.; Feng, Z.; Hu, K. Spatial transcriptomics atlas reveals the crosstalk between cancer-associated fibroblasts and tumor microenvironment components in colorectal cancer. J. Transl. Med. 2022, 20, 302. [Google Scholar] [CrossRef]
- Yamada, A.; Yu, P.; Lin, W.; Okugawa, Y.; Boland, C.R.; Goel, A. A RNA-Sequencing approach for the identification of novel long non-coding RNA biomarkers in colorectal cancer. Sci. Rep. 2018, 8, 575. [Google Scholar] [CrossRef]
- Brouwer, J.; Kluiver, J.; Almeida, R.C.d.; Modderman, R.; Terpstra, M.M.; Kok, K.; Withoff, S.; Hollema, H.; Reitsma, W.; Bock, G.H.d.; et al. Small RNA sequencing reveals a comprehensive miRNA signature of BRCA1-associated high-grade serous ovarian cancer. J. Clin. Pathol. 2016, 69, 979–985. [Google Scholar] [CrossRef]
- Stur, E.; Corvigno, S.; Xu, M.; Chen, K.; Tan, Y.; Lee, S.; Liu, J.; Ricco, E.; Kraushaar, D.; Castro, P.; et al. Spatially resolved transcriptomics of high-grade serous ovarian carcinoma. iScience 2022, 25, 103923. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, Y.; Zhang, J.; Hu, Q.; Zhi, F.; Zhang, S.; Mao, D.; Zhang, Y.; Liang, H. Significance of the TMPRSS2:ERG gene fusion in prostate cancer. Mol. Med. Rep. 2017, 16, 5450–5458. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Weinstein, H.N.W.; Allegakoen, P.; Wadsworth, M.H.; Xie, J.; Yang, H.; Castro, E.A.; Lu, K.L.; Stohr, B.A.; Feng, F.Y.; et al. Single-cell analysis of human primary prostate cancer reveals the heterogeneity of tumor-associated epithelial cell states. Nat. Commun. 2022, 13, 141. [Google Scholar] [CrossRef]
- Prensner, J.R.; Iyer, M.K.; Balbin, O.A.; Dhanasekaran, S.M.; Cao, Q.; Brenner, J.C.; Laxman, B.; Asangani, I.A.; Grasso, C.S.; Kominsky, H.D.; et al. Transcriptome sequencing across a prostate cancer cohort identifies PCAT-1, an unannotated lincRNA implicated in disease progression. Nat. Biotechnol. 2011, 29, 742–749. [Google Scholar] [CrossRef] [PubMed]
- Orimo, A.; Gupta, P.B.; Sgroi, D.C.; Arenzana-Seisdedos, F.; Delaunay, T.; Naeem, R.; Carey, V.J.; Richardson, A.L.; Weinberg, R.A. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 2005, 121, 335–348. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Chen, G.; Yan, J.; Wang, X.; Zhu, Y.; Zhu, L. Single-cell sequencing analysis reveals gastric cancer microenvironment cells respond vastly different to oxidative stress. J. Transl. Med. 2022, 20, 250. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Ramnarayanan, K.; Sundar, R.; Padmanabhan, N.; Srivastava, S.; Koiwa, M.; Yasuda, T.; Koh, V.; Huang, K.K.; Tay, S.T.; et al. Single-Cell Atlas of Lineage States, Tumor Microenvironment, and Subtype-Specific Expression Programs in Gastric Cancer. Cancer Discov. 2022, 12, 670–691. [Google Scholar] [CrossRef]
- Fattahi, S.; Nikbakhsh, N.; Taheri, H.; Ranaee, M.; Akhavan-Niaki, H. RNA Sequencing of Early-Stage Gastric Adenocarcinoma Reveals Multiple Activated Pathways and Novel Long Non-Coding RNAs in Patient Tissue Samples. Rep. Biochem. Mol. Biol. 2021, 9, 478–489. [Google Scholar] [CrossRef]
- Ferlay, J.; Ervik, M.; Lam, F.; Colombet, M.; Mery, L.; Piñeros, M.; Znaor, A.; Soerjomataram, I.; Bray, F. Global cancer observatory: Cancer today. Int. Agency Res. Cancer 2018, 3, 2019. [Google Scholar]
- Pfister, S.M.; Reyes-Múgica, M.; Chan, J.K.C.; Hasle, H.; Lazar, A.J.; Rossi, S.; Ferrari, A.; Jarzembowski, J.A.; Pritchard-Jones, K.; Hill, D.A.; et al. A Summary of the Inaugural WHO Classification of Pediatric Tumors: Transitioning from the Optical into the Molecular Era. Cancer Discov. 2022, 12, 331–355. [Google Scholar] [CrossRef]
- Caron, M.; St-Onge, P.; Sontag, T.; Wang, Y.C.; Richer, C.; Ragoussis, I.; Sinnett, D.; Bourque, G. Single-cell analysis of childhood leukemia reveals a link between developmental states and ribosomal protein expression as a source of intra-individual heterogeneity. Sci. Rep. 2020, 10, 8079. [Google Scholar] [CrossRef]
- Vanhooren, J.; Van Camp, L.; Depreter, B.; de Jong, M.; Uyttebroeck, A.; Van Damme, A.; Dedeken, L.; Dresse, M.F.; van der Werff Ten Bosch, J.; Hofmans, M.; et al. Deciphering the Non-Coding RNA Landscape of Pediatric Acute Myeloid Leukemia. Cancers 2022, 14, 2098. [Google Scholar] [CrossRef]
- Xia, J.; Wang, M.; Zhu, Y.; Bu, C.; Li, T. Differential mRNA and long noncoding RNA expression profiles in pediatric B-cell acute lymphoblastic leukemia patients. BMC Pediatr. 2022, 22, 10. [Google Scholar] [CrossRef]
- Rickert, C.H.; Paulus, W. Epidemiology of central nervous system tumors in childhood and adolescence based on the new WHO classification. Childs Nerv. Syst. 2001, 17, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Fang, F.Y.; Rosenblum, J.S.; Ho, W.S.; Heiss, J.D. New Developments in the Pathogenesis, Therapeutic Targeting, and Treatment of Pediatric Medulloblastoma. Cancers 2022, 14, 2285. [Google Scholar] [CrossRef] [PubMed]
- Joshi, P.; Jallo, G.; Perera, R.J. In silico analysis of long non-coding RNAs in medulloblastoma and its subgroups. Neurobiol. Dis. 2020, 141, 104873. [Google Scholar] [CrossRef]
- Deshpande, D.; Chhugani, K.; Chang, Y.; Karlsberg, A.; Loeffler, C.; Zhang, J.; Muszyńska, A.; Munteanu, V.; Yang, H.; Rotman, J.; et al. RNA-seq data science: From raw data to effective interpretation. Front. Genet. 2023, 14, 997383. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Dragomir, M.P.; Yang, C.; Li, Q.; Horst, D.; Calin, G.A. Targeting non-coding RNAs to overcome cancer therapy resistance. Signal Transduct. Target. Ther. 2022, 7, 121. [Google Scholar] [CrossRef]
- Wang, C.; Liu, H. Factors influencing degradation kinetics of mRNAs and half-lives of microRNAs, circRNAs, lncRNAs in blood in vitro using quantitative PCR. Sci. Rep. 2022, 12, 7259. [Google Scholar] [CrossRef]
- Chen, X.; Mangala, L.S.; Rodriguez-Aguayo, C.; Kong, X.; Lopez-Berestein, G.; Sood, A.K. RNA interference-based therapy and its delivery systems. Cancer Metastasis Rev. 2018, 37, 107–124. [Google Scholar] [CrossRef] [PubMed]
- Torsin, L.I.; Petrescu, G.E.D.; Sabo, A.A.; Chen, B.; Brehar, F.M.; Dragomir, M.P.; Calin, G.A. Editing and Chemical Modifications on Non-Coding RNAs in Cancer: A New Tale with Clinical Significance. Int. J. Mol. Sci. 2021, 22, 581. [Google Scholar] [CrossRef]
- Gaglione, M.; Messere, A. Recent Progress in Chemically Modified siRNAs. Mini-Rev. Med. Chem. 2010, 10, 578–595. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, B.; Goody, P.R.; Hosen, M.R.; Nehl, D.; Mohammadi, N.; Zietzer, A.; Düsing, P.; Pfeifer, A.; Nickenig, G.; Jansen, F. NcRNAs in Vascular and Valvular Intercellular Communication. Front. Mol. Biosci. 2021, 8, 749681. [Google Scholar] [CrossRef] [PubMed]
- Winkle, M.; El-Daly, S.M.; Fabbri, M.; Calin, G.A. Noncoding RNA therapeutics—Challenges and potential solutions. Nat. Rev. Drug Discov. 2021, 20, 629–651. [Google Scholar] [CrossRef]
- Kim, K.W. PIWI Proteins and piRNAs in the Nervous System. Mol. Cells 2019, 42, 828–835. [Google Scholar] [CrossRef]
- Huang, S.; Yoshitake, K.; Asakawa, S. A Review of Discovery Profiling of PIWI-Interacting RNAs and Their Diverse Functions in Metazoans. Int. J. Mol. Sci. 2021, 22, 11166. [Google Scholar] [CrossRef]
- Alshaer, W.; Zureigat, H.; Al Karaki, A.; Al-Kadash, A.; Gharaibeh, L.; Hatmal, M.m.M.; Aljabali, A.A.A.; Awidi, A. siRNA: Mechanism of action, challenges, and therapeutic approaches. Eur. J. Pharmacol. 2021, 905, 174178. [Google Scholar] [CrossRef]
- López-Urrutia, E.; Montes, L.P.B.; Ladrón de Guevara Cervantes, D.; Pérez-Plasencia, C.; Campos-Parra, A.D. Crosstalk between Long Non-coding RNAs, Micro-RNAs and mRNAs: Deciphering Molecular Mechanisms of Master Regulators in Cancer. Front. Oncol. 2019, 9, 669. [Google Scholar] [CrossRef]
- Yan, Q.; Zhu, C.; Guang, S.; Feng, X. The Functions of Non-coding RNAs in rRNA Regulation. Front. Genet. 2019, 10, 290. [Google Scholar] [CrossRef]
- Ji, W.; Sun, B.; Su, C. Targeting MicroRNAs in Cancer Gene Therapy. Genes 2017, 8, 21. [Google Scholar] [CrossRef]
- Shu, J.; Silva, B.V.R.e.; Gao, T.; Xu, Z.; Cui, J. Dynamic and Modularized MicroRNA Regulation and Its Implication in Human Cancers. Sci. Rep. 2017, 7, 13356. [Google Scholar] [CrossRef]
- Kara, G.; Arun, B.; Calin, G.A.; Ozpolat, B. miRacle of microRNA-Driven Cancer Nanotherapeutics. Cancers 2022, 14, 3818. [Google Scholar] [CrossRef]
- Chatterjee, A.; Jana, S.; Chatterjee, S.; Wastall, L.M.; Mandal, G.; Nargis, N.; Roy, H.; Hughes, T.A.; Bhattacharyya, A. MicroRNA-222 reprogrammed cancer-associated fibroblasts enhance growth and metastasis of breast cancer. Br. J. Cancer 2019, 121, 679–689. [Google Scholar] [CrossRef]
- Xing, Y.; Ruan, G.; Ni, H.; Qin, H.; Chen, S.; Gu, X.; Shang, J.; Zhou, Y.; Tao, X.; Zheng, L. Tumor Immune Microenvironment and Its Related miRNAs in Tumor Progression. Front. Immunol. 2021, 12, 624725. [Google Scholar] [CrossRef]
- Wang, W.; Liu, Y.; Guo, J.; He, H.; Mi, X.; Chen, C.; Xie, J.; Wang, S.; Wu, P.; Cao, F.; et al. miR-100 maintains phenotype of tumor-associated macrophages by targeting mTOR to promote tumor metastasis via Stat5a/IL-1ra pathway in mouse breast cancer. Oncogenesis 2018, 7, 97. [Google Scholar] [CrossRef]
- Xiao, M.; Li, J.; Li, W.; Wang, Y.; Wu, F.; Xi, Y.; Zhang, L.; Ding, C.; Luo, H.; Li, Y.; et al. MicroRNAs activate gene transcription epigenetically as an enhancer trigger. RNA Biol. 2017, 14, 1326–1334. [Google Scholar] [CrossRef] [PubMed]
- Harrison, L.J.; Bose, D. Enhancer RNAs step forward: New insights into enhancer function. Development 2022, 149, dev200398. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Lin, Z.; Liu, Q.; Chen, S.; Gao, X.; Guo, W.; Gong, F.; Wei, J.; Lin, H. Enhancer RNA SLIT2 Inhibits Bone Metastasis of Breast Cancer through Regulating P38 MAPK/c-Fos Signaling Pathway. Front. Oncol. 2021, 11, 743840. [Google Scholar] [CrossRef]
- Mo, J.; Zhang, L.; Li, H.; Duan, H.; Wang, D.; Zhao, X.; Xie, Y. The enhancer RNA ADCY10P1 is associated with the progression of ovarian cancer. J. Ovarian Res. 2022, 15, 61. [Google Scholar] [CrossRef] [PubMed]
- Su, M.; Xiao, Y.; Ma, J.; Tang, Y.; Tian, B.; Zhang, Y.; Li, X.; Wu, Z.; Yang, D.; Zhou, Y.; et al. Circular RNAs in Cancer: Emerging functions in hallmarks, stemness, resistance and roles as potential biomarkers. Mol. Cancer 2019, 18, 90. [Google Scholar] [CrossRef]
- Meng, L.; Chang, S.; Sang, Y.; Ding, P.; Wang, L.; Nan, X.; Xu, R.; Liu, F.; Gu, L.; Zheng, Y.; et al. Circular RNA circCCDC85A inhibits breast cancer progression via acting as a miR-550a-5p sponge to enhance MOB1A expression. Breast Cancer Res. 2022, 24, 1. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Lv, H.; Yang, S.; Yang, X. CircRNA PLOD2 enhances ovarian cancer propagation by controlling miR-378. Saudi J. Biol. Sci. 2021, 28, 6260–6265. [Google Scholar] [CrossRef] [PubMed]
- Elsayed, A.M.; Amero, P.; Salama, S.A.; Abdelaziz, A.H.; Lopez-Berestein, G.; Rodriguez-Aguayo, C. Back to the Future: Rethinking the Great Potential of lncRNA(S) for Optimizing Chemotherapeutic Response in Ovarian Cancer. Cancers 2020, 12, 2406. [Google Scholar] [CrossRef]
- Jiang, M.C.; Ni, J.J.; Cui, W.Y.; Wang, B.Y.; Zhuo, W. Emerging roles of lncRNA in cancer and therapeutic opportunities. Am. J. Cancer Res. 2019, 9, 1354–1366. [Google Scholar]
- Huang, D.; Chen, J.; Yang, L.; Ouyang, Q.; Li, J.; Lao, L.; Zhao, J.; Liu, J.; Lu, Y.; Xing, Y.; et al. NKILA lncRNA promotes tumor immune evasion by sensitizing T cells to activation-induced cell death. Nat. Immunol. 2018, 19, 1112–1125. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.J.; Lin, X.J.; Tang, X.Y.; Zheng, T.T.; Zhang, X.Y.; Hua, K.Q. Long noncoding RNA TC0101441 induces epithelial-mesenchymal transition in epithelial ovarian cancer metastasis by downregulating KiSS1. Int. J. Cancer 2020, 146, 2588–2598. [Google Scholar] [CrossRef]
- Oncul, S.; Amero, P.; Rodriguez-Aguayo, C.; Calin, G.A.; Sood, A.K.; Lopez-Berestein, G. Long non-coding RNAs in ovarian cancer: Expression profile and functional spectrum. RNA Biol. 2020, 17, 1523–1534. [Google Scholar] [CrossRef]
- Zhang, Z.; Yan, C.; Li, K.; Bao, S.; Li, L.; Chen, L.; Zhao, J.; Sun, J.; Zhou, M. Pan-cancer characterization of lncRNA modifiers of immune microenvironment reveals clinically distinct de novo tumor subtypes. NPJ Genom. Med. 2021, 6, 52. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, L.; Dong, X.; Yang, X.; Zhang, X.; Liu, Z.; Zhao, X.; Wen, T. Overexpression of NNT-AS1 Activates TGF-β Signaling to Decrease Tumor CD4 Lymphocyte Infiltration in Hepatocellular Carcinoma. Biomed. Res. Int. 2020, 2020, 8216541. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Z.; Chen, M.; Chen, H.; Zhong, Q.; Liang, L.; Li, B. lncRNA TCL6 correlates with immune cell infiltration and indicates worse survival in breast cancer. Breast Cancer 2020, 27, 573–585. [Google Scholar] [CrossRef]
- Mercatelli, N.; Palombo, R.; Paronetto, M.P. Emerging Contribution of PancRNAs in Cancer. Cancers 2020, 12, 2035. [Google Scholar] [CrossRef] [PubMed]
- Chellini, L.; Frezza, V.; Paronetto, M.P. Dissecting the transcriptional regulatory networks of promoter-associated noncoding RNAs in development and cancer. J. Exp. Clin. Cancer Res. 2020, 39, 51. [Google Scholar] [CrossRef]
- Palombo, R.; Frisone, P.; Fidaleo, M.; Mercatelli, N.; Sette, C.; Paronetto, M.P. The Promoter-Associated Noncoding RNA pncCCND1_B Assembles a Protein–RNA Complex to Regulate Cyclin D1 Transcription in Ewing Sarcoma. Cancer Res. 2019, 79, 3570–3582. [Google Scholar] [CrossRef]
- Chen, Y.; Huang, Y.; Gao, X.; Li, Y.; Lin, J.; Chen, L.; Chang, L.; Chen, G.; Guan, Y.; Pan, L.K.; et al. CCND1 Amplification Contributes to Immunosuppression and Is Associated with a Poor Prognosis to Immune Checkpoint Inhibitors in Solid Tumors. Front. Immunol. 2020, 11, 1620. [Google Scholar] [CrossRef]
- Chalbatani, G.M.; Dana, H.; Gharagouzloo, E.; Grijalvo, S.; Eritja, R.; Logsdon, C.D.; Memari, F.; Miri, S.R.; Rad, M.R.; Marmari, V. Small interfering RNAs (siRNAs) in cancer therapy: A nano-based approach. Int. J. Nanomed. 2019, 14, 3111–3128. [Google Scholar] [CrossRef]
- Satpathy, M.; Mezencev, R.; Wang, L.; McDonald, J.F. Targeted in vivo delivery of EGFR siRNA inhibits ovarian cancer growth and enhances drug sensitivity. Sci. Rep. 2016, 6, 36518. [Google Scholar] [CrossRef]
- Blackburn, W.H.; Dickerson, E.B.; Smith, M.H.; McDonald, J.F.; Lyon, L.A. Peptide-functionalized nanogels for targeted siRNA delivery. Bioconjugate Chem. 2009, 20, 960–968. [Google Scholar] [CrossRef]
- Eljack, S.; Allard-Vannier, E.; Misericordia, Y.; Hervé-Aubert, K.; Aubrey, N.; Chourpa, I.; Faggad, A.; David, S. Combination of Nanovectorized siRNA Directed against Survivin with Doxorubicin for Efficient Anti-Cancer Activity in HER2+ Breast Cancer Cells. Pharmaceutics 2022, 14, 2537. [Google Scholar] [CrossRef]
- Cai, A.; Hu, Y.; Zhou, Z.; Qi, Q.; Wu, Y.; Dong, P.; Chen, L.; Wang, F. PIWI-Interacting RNAs (piRNAs): Promising Applications as Emerging Biomarkers for Digestive System Cancer. Front. Mol. Biosci. 2022, 9, 848105. [Google Scholar] [CrossRef]
- Liu, Y.; Dou, M.; Song, X.; Dong, Y.; Liu, S.; Liu, H.; Tao, J.; Li, W.; Yin, X.; Xu, W. The emerging role of the piRNA/piwi complex in cancer. Mol. Cancer 2019, 18, 123. [Google Scholar] [CrossRef]
- Tan, L.; Mai, D.; Zhang, B.; Jiang, X.; Zhang, J.; Bai, R.; Ye, Y.; Li, M.; Pan, L.; Su, J.; et al. PIWI-interacting RNA-36712 restrains breast cancer progression and chemoresistance by interaction with SEPW1 pseudogene SEPW1P RNA. Mol. Cancer 2019, 18, 9. [Google Scholar] [CrossRef]
- Kwok, A.; Raulf, N.; Habib, N. Developing small activating RNA as a therapeutic: Current challenges and promises. Ther. Deliv. 2019, 10, 151–164. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.P.; Sinigaglia, L.; Gomez, V.; Nicholls, J.; Habib, N.A. RNA Activation—A Novel Approach to Therapeutically Upregulate Gene Transcription. Molecules 2021, 26, 6530. [Google Scholar] [CrossRef]
- Xiong, Y.; Ke, R.; Zhang, Q.; Lan, W.; Yuan, W.; Chan, K.N.I.; Roussel, T.; Jiang, Y.; Wu, J.; Liu, S.; et al. Small Activating RNA Modulation of the G Protein-Coupled Receptor for Cancer Treatment. Adv. Sci. 2022, 9, 2200562. [Google Scholar] [CrossRef]
- Wilson, K.; Shiuan, E.; Brantley-Sieders, D.M. Oncogenic functions and therapeutic targeting of EphA2 in cancer. Oncogene 2021, 40, 2483–2495. [Google Scholar] [CrossRef]
- M.D. Anderson Cancer Center; Gateway for Cancer Research; Institutional Funding for Federally Supported Clinical Trials; National Cancer Institute. EphA2 siRNA in Treating Patients with Advanced or Recurrent Solid Tumors. 2015. Available online: https://ClinicalTrials.gov/show/NCT01591356 (accessed on 21 March 2023).
- City of Hope Medical Center; National Cancer Institute. CpG-STAT3 siRNA CAS3/SS3 and Localized Radiation Therapy for the Treatment of Relapsed/Refractory B-Cell NHL. 2023. Available online: https://classic.clinicaltrials.gov/show/NCT04995536 (accessed on 21 March 2023).
- M.D. Anderson Cancer Center. iExosomes in Treating Participants with Metastatic Pancreas Cancer with KrasG12D Mutation. 2021. Available online: https://ClinicalTrials.gov/show/NCT03608631 (accessed on 21 March 2023).
- Crunkhorn, S. RNA-based approaches target KRAS. Nat. Rev. Drug Discov. 2017, 16, 529. [Google Scholar] [CrossRef]
- Silenseed, Ltd. A Phase 2 Study of siG12D LODER in Combination with Chemotherapy in Patients with Locally Advanced Pancreatic Cancer. 2018. Available online: https://ClinicalTrials.gov/show/NCT01676259 (accessed on 21 March 2023).
- Nitto BioPharma, Inc. A Study of NBF-006 in Non-Small Cell Lung, Pancreatic, or Colorectal Cancer. 2019. Available online: https://ClinicalTrials.gov/show/NCT03819387 (accessed on 21 March 2023).
- Otsuka, K.; Satoyoshi, R.; Nanjo, H.; Miyazawa, H.; Abe, Y.; Tanaka, M.; Yamamoto, Y.; Shibata, H. Acquired/intratumoral mutation of KRAS during metastatic progression of colorectal carcinogenesis. Oncol. Lett. 2012, 3, 649–653. [Google Scholar] [CrossRef]
- Sirnaomics. A Study to Evaluate Safety, Efficacy of Intralesional Injection of STP705 in Patients with isSCC. 2021. Available online: https://ClinicalTrials.gov/show/NCT04844983 (accessed on 21 March 2023).
- Molyneaux, M.; Xu, J.; Evans, D.; Lu, P. Effect on tumor growth by TGF-β1/COX-2 siRNA combination product (STP705) in a human cholangiocarcinoma (HuCCT-1) xenograft tumor model in nude mice. J. Clin. Oncol. 2019, 37, e14652. [Google Scholar] [CrossRef]
- Dicerna Pharmaceuticals, Inc. A Novo Nordisk Company, Phase Ib/2, Multicenter, Dose Escalation Study of DCR-MYC in Patients with Hepatocellular Carcinoma. 2015. Available online: https://classic.clinicaltrials.gov/show/NCT02314052 (accessed on 21 March 2023).
- Dicerna Pharmaceuticals, Inc. A Novo Nordisk Company, Phase I, Multicenter, Dose Escalation Study of DCR-MYC in Patients with Solid Tumors, Multiple Myeloma, or Lymphoma. 2014. Available online: https://classic.clinicaltrials.gov/show/NCT02110563 (accessed on 21 March 2023).
- University Health Network, Toronto; Takara Bio Inc. Study of TBI-1301 (NY-ESO-1 Specific TCR Gene Transduced Autologous T Lymphocytes) in Patients with Solid Tumors. 2016. Available online: https://classic.clinicaltrials.gov/show/NCT02869217 (accessed on 21 March 2023).
- Takara Bio Inc. Study of TBI-1301 (NY-ESO-1 T Cell Receptor Gene Transduced Autologous T Lymphocytes) in Patients with Synovial Sarcoma. 2017. Available online: https://classic.clinicaltrials.gov/show/NCT03250325 (accessed on 21 March 2023).
- Mie University; Takara Bio Inc.; Shionogi; Fiverings Co., Ltd.; Statcom Co. Ltd. Investigator Initiated Phase 1 Study of TBI-1301. 2015. Available online: https://classic.clinicaltrials.gov/show/NCT02366546 (accessed on 21 March 2023).
- Arbutus Biopharma Corporation. Safety, Pharmacokinetics and Preliminary Anti-Tumor Activity of Intravenous TKM-080301 in Subjects with Advanced Hepatocellular Carcinoma. 2014. Available online: https://classic.clinicaltrials.gov/show/NCT02191878 (accessed on 21 March 2023).
- Arbutus Biopharma Corporation. A Dose Finding Study of TKM-080301 Infusion in Neuroendocrine Tumors (NET) and Adrenocortical Carcinoma (ACC) Patients. 2010. Available online: https://classic.clinicaltrials.gov/show/NCT01262235 (accessed on 21 March 2023).
- National Cancer Institute; National Institutes of Health Clinical Center. TKM 080301 for Primary or Secondary Liver Cancer. 2011. Available online: https://classic.clinicaltrials.gov/show/NCT01437007 (accessed on 21 March 2023).
- InteRNA. First-in-Human Study of INT-1B3 in Patients with Advanced Solid Tumors. 2020. Available online: https://classic.clinicaltrials.gov/show/NCT04675996 (accessed on 21 March 2023).
- Mirna Therapeutics, Inc. Pharmacodynamics Study of MRX34, MicroRNA Liposomal Injection in Melanoma Patients with Biopsy Accessible Lesions. 2016. Available online: https://classic.clinicaltrials.gov/show/NCT02862145 (accessed on 21 March 2023).
- Mirna Therapeutics, Inc.; Cancer Prevention Research Institute of Texas. A Multicenter Phase I Study of MRX34, MicroRNA miR-RX34 Liposomal Injection. 2013. Available online: https://classic.clinicaltrials.gov/show/NCT01829971 (accessed on 21 March 2023).
- Asbestos Diseases Research Foundation; EnGeneIC Limited. MesomiR 1: A Phase I Study of TargomiRs as 2nd or 3rd Line Treatment for Patients with Recurrent MPM and NSCLC. 2014. Available online: https://classic.clinicaltrials.gov/show/NCT02369198 (accessed on 21 March 2023).
- Mina Alpha Limited. Radomised Phase II Study of MTL-CEBPA Plus Sorafenib or Sorafenib Alone. 2022. Available online: https://classic.clinicaltrials.gov/show/NCT04710641 (accessed on 21 March 2023).
- Setten, R.L.; Lightfoot, H.L.; Habib, N.A.; Rossi, J.J. Development of MTL-CEBPA: Small Activating RNA Drug for Hepatocellular Carcinoma. Curr. Pharm. Biotechnol. 2018, 19, 611–621. [Google Scholar] [CrossRef]
- Mina Alpha Limited. A Study of MTL-CEBPA in Combination with a PD-1 Inhibitor in Patients with Advanced Solid Tumours (TIMEPOINT). 2019. Available online: https://classic.clinicaltrials.gov/show/NCT04105335 (accessed on 21 March 2023).
- Mina Alpha Limited. First-in-Human Safety, Tolerability and Antitumour Activity Study of MTL-CEBPA in Patients with Advanced Liver Cancer. 2016. Available online: https://classic.clinicaltrials.gov/show/NCT02716012 (accessed on 21 March 2023).
- National University Hospital, Singapore. Phase I Study of RNA Oligonucleotide, MTL-CEBPA, Atezolizumab and Bevacizumab in Patients with Advanced Hepatocellular Carcinoma. 2021. Available online: https://classic.clinicaltrials.gov/show/NCT05097911 (accessed on 21 March 2023).
- Atri, C.; Guerfali, F.Z.; Laouini, D. Chapter 6—MicroRNAs in diagnosis and therapeutics. In AGO-Driven Non-Coding RNAs; Mallick, B., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 137–177. [Google Scholar]
- Zhao, R.; Fu, J.; Zhu, L.; Chen, Y.; Liu, B. Designing strategies of small-molecule compounds for modulating non-coding RNAs in cancer therapy. J. Hematol. Oncol. 2022, 15, 14. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Challa, S.; Sang, P.; She, F.; Li, C.; Gray, G.M.; Nimmagadda, A.; Teng, P.; Odom, T.; Wang, Y.; et al. One-Bead–Two-Compound Thioether Bridged Macrocyclic γ-AApeptide Screening Library against EphA2. J. Med. Chem. 2017, 60, 9290–9298. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, A.M.; Sun, Z.; Ayat, N.; Schilb, A.; Liu, X.; Jiang, H.; Sun, D.; Scheidt, J.; Qian, V.; He, S.; et al. Systemic Delivery of Tumor-Targeting siRNA Nanoparticles against an Oncogenic LncRNA Facilitates Effective Triple-Negative Breast Cancer Therapy. Bioconjugate Chem. 2019, 30, 907–919. [Google Scholar] [CrossRef] [PubMed]
- Maranon, D.G.; Wilusz, J. Mind the Gapmer: Implications of Co-transcriptional Cleavage by Antisense Oligonucleotides. Mol. Cell 2020, 77, 932–933. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Guan, J.; Gao, Y.; Zhu, Y.; Li, H.; Guo, H.; He, Q.; Guan, Z.; Yang, Z. Modified ASO conjugates encapsulated with cytidinyl/cationic lipids exhibit more potent and longer-lasting anti-HCC effects. Mol. Ther. Nucleic Acids 2023, 32, 807–821. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.; Klümper, N.; Schmidt, D.; Ritter, M.; Ellinger, J.; Hauser, S. Depletion of the m(6)A demethylases FTO and ALKBH5 impairs growth and metastatic capacity through EMT phenotype change in clear cell renal cell carcinoma. Am. J. Transl. Res. 2023, 15, 1744–1755. [Google Scholar]
- Yang, K.; Zhang, W.; Zhong, L.; Xiao, Y.; Sahoo, S.; Fassan, M.; Zeng, K.; Magee, P.; Garofalo, M.; Shi, L. Long non-coding RNA HIF1A-As2 and MYC form a double-positive feedback loop to promote cell proliferation and metastasis in KRAS-driven non-small cell lung cancer. Cell Death Differ. 2023, 30, 1533–1549. [Google Scholar] [CrossRef]
- Yoshida, M.; Oda, C.; Mishima, K.; Tsuji, I.; Obika, S.; Shimojo, M. An antisense amido-bridged nucleic acid gapmer oligonucleotide targeting SRRM4 alters REST splicing and exhibits anti-tumor effects in small cell lung cancer and prostate cancer cells. Cancer Cell Int. 2023, 23, 8. [Google Scholar] [CrossRef]
- Dabiri, A.; Sharifi, M.; Sarmadi, A. Knockdown of SOX12 Expression Induced Apoptotic Factors is Associated with TWIST1 and CTNNB1 Expression in Human Acute Myeloid Leukemia Cells. Int. J. Mol. Cell Med. 2021, 10, 249–258. [Google Scholar] [CrossRef]
- Chen, H.Y.; Chan, S.J.; Liu, X.; Wei, A.C.; Jian, R.I.; Huang, K.W.; Lang, Y.D.; Shih, J.H.; Liao, C.C.; Luan, C.L.; et al. Long noncoding RNA Smyca coactivates TGF-β/Smad and Myc pathways to drive tumor progression. J. Hematol. Oncol. 2022, 15, 85. [Google Scholar] [CrossRef]
- Ii, H.; Kasahara, Y.; Yamaguma, H.; Kageyama, S.; Kawauchi, A.; Obika, S.; Nakata, S. Administration of Gapmer-type Antisense Oligonucleotides Targeting γ-Glutamylcyclotransferase Suppresses the Growth of A549 Lung Cancer Xenografts. Anticancer Res. 2022, 42, 1221–1227. [Google Scholar] [CrossRef]
- García-Garrido, E.; Cordani, M.; Somoza, Á. Modified Gold Nanoparticles to Overcome the Chemoresistance to Gemcitabine in Mutant p53 Cancer Cells. Pharmaceutics 2021, 13, 2067. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, P.; Sharifi, M.; Ghadiri, A. Downregulation of MIR100HG Induces Apoptosis in Human Megakaryoblastic Leukemia Cells. Indian J. Hematol. Blood Transfus. 2021, 37, 232–239. [Google Scholar] [CrossRef]
- Ma, Y.; Zhao, W.; Li, Y.; Pan, Y.; Wang, S.; Zhu, Y.; Kong, L.; Guan, Z.; Wang, J.; Zhang, L.; et al. Structural optimization and additional targets identification of antisense oligonucleotide G3139 encapsulated in a neutral cytidinyl-lipid combined with a cationic lipid in vitro and in vivo. Biomaterials 2019, 197, 182–193. [Google Scholar] [CrossRef] [PubMed]
- Shimojo, M.; Kasahara, Y.; Inoue, M.; Tsunoda, S.I.; Shudo, Y.; Kurata, T.; Obika, S. A gapmer antisense oligonucleotide targeting SRRM4 is a novel therapeutic medicine for lung cancer. Sci. Rep. 2019, 9, 7618. [Google Scholar] [CrossRef] [PubMed]
- Taiana, E.; Favasuli, V.; Ronchetti, D.; Todoerti, K.; Pelizzoni, F.; Manzoni, M.; Barbieri, M.; Fabris, S.; Silvestris, I.; Cantafio, M.E.G.; et al. Long non-coding RNA NEAT1 targeting impairs the DNA repair machinery and triggers anti-tumor activity in multiple myeloma. Leukemia 2020, 34, 234–244. [Google Scholar] [CrossRef]
- Papachristodoulou, A.; Silginer, M.; Weller, M.; Schneider, H.; Hasenbach, K.; Janicot, M.; Roth, P. Therapeutic Targeting of TGFβ Ligands in Glioblastoma Using Novel Antisense Oligonucleotides Reduces the Growth of Experimental Gliomas. Clin. Cancer Res. 2019, 25, 7189–7201. [Google Scholar] [CrossRef]
- Rapozzi, V.; Cogoi, S.; Xodo, L.E. Antisense locked nucleic acids efficiently suppress BCR/ABL and induce cell growth decline and apoptosis in leukemic cells. Mol. Cancer Ther. 2006, 5, 1683–1692. [Google Scholar] [CrossRef]
- Hansen, J.B.; Fisker, N.; Westergaard, M.; Kjaerulff, L.S.; Hansen, H.F.; Thrue, C.A.; Rosenbohm, C.; Wissenbach, M.; Orum, H.; Koch, T. SPC3042: A proapoptotic survivin inhibitor. Mol. Cancer Ther. 2008, 7, 2736–2745. [Google Scholar] [CrossRef]
- Emmrich, S.; Wang, W.; John, K.; Li, W.; Pützer, B.M. Antisense gapmers selectively suppress individual oncogenic p73 splice isoforms and inhibit tumor growth in vivo. Mol. Cancer 2009, 8, 61. [Google Scholar] [CrossRef]
- Manier, S.; Powers, J.T.; Sacco, A.; Glavey, S.V.; Huynh, D.; Reagan, M.R.; Salem, K.Z.; Moschetta, M.; Shi, J.; Mishima, Y.; et al. The LIN28B/let-7 axis is a novel therapeutic pathway in multiple myeloma. Leukemia 2017, 31, 853–860. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Liu, Q.; Li, H.; Kang, C.; Liu, Y.; Guo, T.; Shang, K.; Yan, C.; Cheng, G.; Lee, R.J. Lipid Nanoparticles Loaded with an Antisense Oligonucleotide Gapmer against Bcl-2 for Treatment of Lung Cancer. Pharm. Res. 2017, 34, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Seystahl, K.; Papachristodoulou, A.; Burghardt, I.; Schneider, H.; Hasenbach, K.; Janicot, M.; Roth, P.; Weller, M. Biological Role and Therapeutic Targeting of TGF-β(3) in Glioblastoma. Mol. Cancer Ther. 2017, 16, 1177–1186. [Google Scholar] [CrossRef]
- Booy, E.P.; McRae, E.K.; Koul, A.; Lin, F.; McKenna, S.A. The long non-coding RNA BC200 (BCYRN1) is critical for cancer cell survival and proliferation. Mol. Cancer 2017, 16, 109. [Google Scholar] [CrossRef] [PubMed]
- De Clara, E.; Gourvest, M.; Ma, H.; Vergez, F.; Tosolini, M.; Dejean, S.; Demur, C.; Delabesse, E.; Recher, C.; Touriol, C.; et al. Long non-coding RNA expression profile in cytogenetically normal acute myeloid leukemia identifies a distinct signature and a new biomarker in NPM1-mutated patients. Haematologica 2017, 102, 1718–1726. [Google Scholar] [CrossRef]
- Amodio, N.; Stamato, M.A.; Juli, G.; Morelli, E.; Fulciniti, M.; Manzoni, M.; Taiana, E.; Agnelli, L.; Cantafio, M.E.G.; Romeo, E.; et al. Drugging the lncRNA MALAT1 via LNA gapmeR ASO inhibits gene expression of proteasome subunits and triggers anti-multiple myeloma activity. Leukemia 2018, 32, 1948–1957. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Lin, J.; Fang, H.; Fang, J.; Li, C.; Chen, W.; Liu, S.; Ondrejka, S.; Gong, Z.; Reu, F.; et al. Targeting the MALAT1/PARP1/LIG3 complex induces DNA damage and apoptosis in multiple myeloma. Leukemia 2018, 32, 2250–2262. [Google Scholar] [CrossRef]
- Gutschner, T.; Hämmerle, M.; Eißmann, M.; Hsu, J.; Kim, Y.; Hung, G.; Revenko, A.; Arun, G.; Stentrup, M.; Groß, M.; et al. The Noncoding RNA MALAT1 Is a Critical Regulator of the Metastasis Phenotype of Lung Cancer Cells. Cancer Res. 2013, 73, 1180–1189. [Google Scholar] [CrossRef] [PubMed]
- Morelli, E.; Biamonte, L.; Federico, C.; Amodio, N.; Di Martino, M.T.; Cantafio, M.E.G.; Manzoni, M.; Scionti, F.; Samur, M.K.; Gullà, A.; et al. Therapeutic vulnerability of multiple myeloma to MIR17PTi, a first-in-class inhibitor of pri-miR-17-92. Blood 2018, 132, 1050–1063. [Google Scholar] [CrossRef]
- Salehi, M.; Sharifi, M. Induction of apoptosis and necrosis in human acute erythroleukemia cells by inhibition of long non-coding RNA PVT1. Mol. Biol. Res. Commun. 2018, 7, 89–96. [Google Scholar] [CrossRef]
- Salehi, M.; Sharifi, M.; Bagheri, M. Knockdown of Long Noncoding RNA Plasmacytoma Variant Translocation 1 with Antisense Locked Nucleic Acid GapmeRs Exerts Tumor-Suppressive Functions in Human Acute Erythroleukemia Cells through Downregulation of C-MYC Expression. Cancer Biother. Radiopharm. 2019, 34, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Leech, S.H.; Olie, R.A.; Gautschi, O.; Simões-Wüst, A.P.; Tschopp, S.; Häner, R.; Hall, J.; Stahel, R.A.; Zangemeister-Wittke, U. Induction of apoptosis in lung-cancer cells following bcl-xL anti-sense treatment. Int. J. Cancer 2000, 86, 570–576. [Google Scholar] [CrossRef]
- Jepsen, J.S.; Pfundheller, H.M.; Lykkesfeldt, A.E. Downregulation of p21(WAF1/CIP1) and estrogen receptor alpha in MCF-7 cells by antisense oligonucleotides containing locked nucleic acid (LNA). Oligonucleotides 2004, 14, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Simões-Wüst, A.P.; Hopkins-Donaldson, S.; Sigrist, B.; Belyanskaya, L.; Stahel, R.A.; Zangemeister-Wittke, U. A functionally improved locked nucleic acid antisense oligonucleotide inhibits Bcl-2 and Bcl-xL expression and facilitates tumor cell apoptosis. Oligonucleotides 2004, 14, 199–209. [Google Scholar] [CrossRef]
- Biroccio, A.; D’Angelo, C.; Jansen, B.; Gleave, M.E.; Zupi, G. Antisense clusterin oligodeoxynucleotides increase the response of HER-2 gene amplified breast cancer cells to Trastuzumab. J. Cell Physiol. 2005, 204, 463–469. [Google Scholar] [CrossRef]
- Leucci, E.; Vendramin, R.; Spinazzi, M.; Laurette, P.; Fiers, M.; Wouters, J.; Radaelli, E.; Eyckerman, S.; Leonelli, C.; Vanderheyden, K.; et al. Melanoma addiction to the long non-coding RNA SAMMSON. Nature 2016, 531, 518–522. [Google Scholar] [CrossRef]
- Lin, J.; Hu, Y.; Zhao, J.J. Repression of Multiple Myeloma Cell Growth In Vivo by Single-wall Carbon Nanotube (SWCNT)-delivered MALAT1 Antisense Oligos. J. Vis. Exp. 2018, 142, e58598. [Google Scholar] [CrossRef]
- Larcher, L.M.; Wang, T.; Veedu, R.N. Development of Novel antimiRzymes for Targeted Inhibition of miR-21 Expression in Solid Cancer Cells. Molecules 2019, 24, 2489. [Google Scholar] [CrossRef]
- Santa-Inez, D.C.; Fuziwara, C.S.; Saito, K.C.; Kimura, E.T. Targeting the Highly Expressed microRNA miR-146b with CRISPR/Cas9n Gene Editing System in Thyroid Cancer. Int. J. Mol. Sci. 2021, 22, 7992. [Google Scholar] [CrossRef]
- Dartmouth-Hitchcock Medical Center. Evaluating the Expression Levels of MicroRNA-10b in Patients with Gliomas. 2013. Available online: https://classic.clinicaltrials.gov/show/NCT01849952 (accessed on 8 August 2023).
- miRagen Therapeutics, Inc. PRISM: Efficacy and Safety of Cobomarsen (MRG-106) in Subjects with Mycosis Fungoides Who Have Completed the SOLAR Study. 2019. Available online: https://classic.clinicaltrials.gov/show/NCT03837457 (accessed on 8 August 2023).
- miRagen Therapeutics, Inc. SOLAR: Efficacy and Safety of Cobomarsen (MRG-106) vs. Active Comparator in Subjects with Mycosis Fungoides. 2019. Available online: https://classic.clinicaltrials.gov/show/NCT03713320 (accessed on 8 August 2023).
- miRagen Therapeutics, Inc. Safety, Tolerability and Pharmacokinetics of MRG-106 in Patients with Mycosis Fungoides (MF), CLL, DLBCL or ATLL. 2016. Available online: https://classic.clinicaltrials.gov/show/NCT02580552 (accessed on 8 August 2023).
- NCIC Clinical Trials Group; Canadian Cancer Trials Group. Hormone Therapy and OGX-011 before Radical Prostatectomy in Treating Patients with Prostate Cancer. 2002. Available online: https://classic.clinicaltrials.gov/show/NCT00054106 (accessed on 8 August 2023).
- NCIC Clinical Trials Group; Canadian Cancer Trials Group. OGX-011 and Docetaxel in Treating Patients with Metastatic or Locally Recurrent Solid Tumors. 2003. Available online: https://classic.clinicaltrials.gov/show/NCT00471432 (accessed on 8 August 2023).
- NCIC Clinical Trials Group; Canadian Cancer Trials Group. OGX-011 and Docetaxel in Treating Women with Locally Advanced or Metastatic Breast Cancer. 2005. Available online: https://classic.clinicaltrials.gov/show/NCT00258375 (accessed on 8 August 2023).
- Chi, K.N.; Yu, E.Y.; Jacobs, C.; Bazov, J.; Kollmannsberger, C.; Higano, C.S.; Mukherjee, S.D.; Gleave, M.E.; Stewart, P.S.; Hotte, S.J. A phase I dose-escalation study of apatorsen (OGX-427), an antisense inhibitor targeting heat shock protein 27 (Hsp27), in patients with castration-resistant prostate cancer and other advanced cancers. Ann. Oncol. 2016, 27, 1116–1122. [Google Scholar] [CrossRef]
- Achieve Life Sciences. Safety Study of an Antisense Product in Prostate, Ovarian, NSCL, Breast or Bladder Cancer. 2007. Available online: https://classic.clinicaltrials.gov/show/NCT00487786 (accessed on 8 August 2023).
- British Columbia Cancer Agency; Achieve Life Sciences. OGX-427 in Castration Resistant Prostate Cancer Patients. 2010. Available online: https://classic.clinicaltrials.gov/show/NCT01120470 (accessed on 8 August 2023).
- Noah Hahn, M.D.; Achieve Life Sciences; Hoosier Cancer Research Network. Phase 2 Study of Docetaxel +/- OGX-427 in Patients with Relapsed or Refractory Metastatic Bladder Cancer. 2013. Available online: https://classic.clinicaltrials.gov/show/NCT01780545 (accessed on 8 August 2023).
- Flamingo Therapeutics NV. Activity and Safety of Danvatirsen and Pembrolizumab in HNSCC. 2023. Available online: https://classic.clinicaltrials.gov/show/NCT05814666 (accessed on 8 August 2023).
- M.D. Anderson Cancer Center; National Cancer Institute (NCI). AstraZeneca, Danvatirsen and Durvalumab in Treating Patients with Advanced and Refractory Pancreatic, Non-Small Cell Lung Cancer, and Mismatch Repair Deficient Colorectal Cancer. 2017. Available online: https://classic.clinicaltrials.gov/show/NCT02983578 (accessed on 8 August 2023).
- MedImmune LLC. Neoadjuvant Durvalumab Alone or in Combination with Novel Agents in Resectable Non-Small Cell Lung Cancer. 2019. Available online: https://classic.clinicaltrials.gov/show/NCT03794544 (accessed on 8 August 2023).
- AstraZeneca. A Study of Novel Anti-Cancer Agents in Patients with Previously Untreated NSCLC. 2018. Available online: https://classic.clinicaltrials.gov/show/NCT03819465 (accessed on 8 August 2023).
- Weber, C.E.; Kuo, P.C. The tumor microenvironment. Surg. Oncol. 2012, 21, 172–177. [Google Scholar] [CrossRef]
- Balkwill, F.; Mantovani, A. Inflammation and cancer: Back to Virchow? Lancet 2001, 357, 539–545. [Google Scholar] [CrossRef]
- Koh, D.-M.; Papanikolaou, N.; Bick, U.; Illing, R.; Kahn, C.E.; Kalpathi-Cramer, J.; Matos, C.; Martí-Bonmatí, L.; Miles, A.; Mun, S.K.; et al. Artificial intelligence and machine learning in cancer imaging. Commun. Med. 2022, 2, 133. [Google Scholar] [CrossRef]
| Method | Brief Description | Advantages | Disadvantages | References |
|---|---|---|---|---|
| RT-qPCR | Technique based on PCR | High sensitivity | Time-consuming; requires primers; Allows for the evaluation of the expression of known transcripts | [9] |
| Microarray | Technique based on the ability of complementary nucleic acid molecules to form double-stranded structures | Well-defined protocols for hybridization | Stringent criteria for sample collection requires a high quantity of RNA; expensive | [10] |
| Bulk RNA-seq | Technique to evaluate mean gene expression of thousands of cells | Costs less than single-cell and spatial RNA-seq; requires less time compared to single-cell RNA-seq and spatial RNA-seq | Less detailed information on individual cells provide averaged gene expression in whole cells | [11] |
| Single-cell RNA-seq | Sequencing technique to evaluate mRNA in a single cell | Enables analysis of the whole transcriptome including non-coding sequences; low background noise | Requires adequate preparation of tissue/cells; expensive; time-consuming; does not provide spatial information on transcriptome | [12] |
| Spatial RNA-seq | Sequencing technique for evaluation of mRNA in the tissue area | Analyzes the whole transcriptome evaluates the interactions between cancer cells and tumor microenvironment | Requires adequate preparation of tissue/cells; cannot analyze a single cell; expensive; time-consuming | [13] |
| Metabolic labeling | Technique in which RNA is labeled with uracil analogs (s4U, EU, Bru) in cell culture | Efficient; well-validated; high resolution | Uracil analogs are cytotoxic | [14] |
| Nuclear run-on | Technique in which RNA in the isolated nucleus is labeled | Measurement of transcription in the primary state; allows distinct transcription and post-transcription changes in gene expression | Requires the ice-cold temperature to isolate nuclei; requires a large number of cells; results are dependent on the efficient induction of transcription outside the cell | [15] |
| Chromatin immunoprecipitation (ChIP) | Technology based on antibodies to selectively isolate DNA-binding proteins and their DNA targets | Allows monitoring of changes in a single promoter in a time-dependent manner; may be used to follow transcription factors in the whole human genome | Low resolution; expensive; risk of protein rearrangement during analysis | [16] |
| Type of ncRNA | Clinical Trial | Objectives of Study | Cancer | Reference |
|---|---|---|---|---|
| EphA-2 siRNA (siRNA) | Phase 1 (recruiting) | Silencing EphA-2 | Solid tumors | [188,189] |
| CpG-STAT3 siRNA CAS3/SS3 (CpG oligonucleotide and siRNA) | Phase 1 (recruiting) | Downregulation of STAT3 | Relapsed B-cell non-Hodgkin lymphomas | [190] |
| KRAS G12- LODER (siRNA) | Phase 1 (recruiting) | Silencing KRAS G12D | Pancreatic cancer | [191,192] |
| Phase 2 (recruiting) | Silencing KRAS G12D to increase the anticancer activity of gemcitabine in combination with nab-paclitaxel or FOLFIRINOX chemotherapy | Pancreatic cancer | [193] | |
| GSTP siRNA/NBF-006(siRNA) | Phase 1 (recruiting) | Silencing GSTP to decrease KRAS signaling pathway | NSCLC Pancreatic cancer Colorectal cancer | [194,195] |
| Two siRNAs: TGF-β1 COX-2 /STP705 (siRNA) | Phase 2 (not recruiting) | Silencing expression of TGF-β1 and COX-2 to inhibit cell survival and induce tumor cell apoptosis | Cutaneous squamous cell carcinoma skin cancer | [196,197] |
| DCR-MYC (siRNA) | Phase 1/2 (terminated) | Inhibition of MYC expression | Hepatocellular carcinoma | [198] |
| Phase 1 (terminated) | Inhibition of MYC expression | Solid tumors Multiple Myeloma Non-Hodgkins Lymphoma Pancreatic Neuroendocrine Tumors Primary Central Nervous system tumors (PNET) N | [199] | |
| TBI-1301 (siRNA) | Phase 1 (active) | Silencing endogenous TCR on T cells in combination with cyclophosphamide and fludarabine | Synovial Sarcoma Melanoma Esophageal Cancer Ovarian Cancer Lung Cancer Bladder Cancer Liver Cancer | [200] |
| Phase 1/2 (active) | Combination of TBI-1301 and cyclophosphamide | Synovial Sarcoma | [201] | |
| Phase 1 (unknown) | Combination of TBI-1301, cyclophosphamide and fludarabine | Solid tumors | [202] | |
| TKM-080301 (siRNA) | Phase 1/2 (completed) | Inhibition of PLK1 expression | Hepatocellular carcinoma Hepatoma Liver cancer | [203] |
| Phase 1/2 (completed) | Neuroendocrine Tumors Adrenocortical Carcinoma | [204] | ||
| Phase 1 (completed) | Solid cancers with hepatic metastases | [205] | ||
| INT1-B3 (mimic miRNA) | Phase 1 (recruiting) | Mimic miRNA-193a-3p | Solid tumors | [206] |
| MRX34 (mimic miRNA) | Phase 1/2 (withdrawn) | Combination of MRX34 and dexamethasone | Melanoma | [207] |
| Phase 1 (terminated) | Mimic miRNA-34 | Primary Liver Cancer SCLC Lymphoma Melanoma Multiple Myeloma Renal Cell Carcinoma NSCLC | [208] | |
| Mesomir-1 (mimic miRNA) | Phase 1 (completed) | Mimic miRNA-16 | Malignant Pleural Mesothelioma NSCLC | [209] |
| MTL-CEBPA (saRNA) | Phase 2 (recruiting) | Increase C/EBP-α expression | Hepatocellular carcinoma | [210,211] |
| Phase 1 (active) | Combination therapy of MTL-CEBPA and pembrolizumab | Breast cancer Lung Cancer Ovarian Cancer Pancreatic cancer Gall bladder cancer Hepatocellular cancer Neuroendocrine cancer Cholangiocarcinoma | [212] | |
| Phase 1 (active) | Combination therapy MTL-CEBPA and sorafenib | Hepatocellular carcinoma | [213] | |
| Phase 1 (recruiting) | Combination therapy MTL-CEBPA and atezolizumab and bevacizumab | Hepatocellular carcinoma | [214] |
| Gapmer | Cancer | Function | Type of Research | Reference |
|---|---|---|---|---|
| CT102 gapmer for IGF1R mRNA | HCC | Inhibition of PI3K/AKT pathway, Induction of apoptosis in tumor cells by interaction with GAS2, POLA2, LGALS2 | In vitro, in vivo | [220] |
| Gapmers for ALKBH5 or FTO | Clear Renal Cell Carcinoma | Inhibition of migration, proliferation of tumor cells, downregulation of Vimentin and PCNA | In vitro | [221] |
| Gapmer for HIF1A-As2 | NSCLC | Increase sensitivity of NSCLC tumors to MYC inhibitor (10058-F4) and cisplatin treatment Inhibition of colony formation, spheroid formation in vitro Inhibition of tumor growth in PDX (patient-derived xenograft) model | In vitro, in vivo | [222] |
| Gapmer for SRRM4 | NSCLC, Prostate cancer | Reduction in cell growth | In vitro | [223] |
| Gapmer for SOX12 | Human Acute Myeloid Leukemia Cells | Inhibition of expression of SOX12 Activation of apoptosis by increased activity of caspase 3 and 9 | In vitro | [224] |
| Gapmer for Smyca | Breast cancer | Inhibition of TGF-β/Smad and c-Myc pathways Inhibition of tumor growth | In vitro In vivo | [225] |
| Gapmer for GGCT | Lung cancer | Inhibition of expression of GGCT to decrease the viability of tumor cells Activation of apoptosis via caspase 3 and 8 Activation of AMPK Inhibition of tumor growth | In vitro, in vivo | [226] |
| Gapmer for p53 mutant protein | Breast and pancreas cancers | Inhibition of cell viability and proliferation Decrease expression of proapoptotic protein Bcl-2 | In vitro | [227] |
| Gapmer for lncRNA MIR100HG | Acute Megakaryocyte Leukemia | Inhibition of lncRNA MIR100HG Induction of apoptosis Increased level of TGF-B expression | In vitro | [228] |
| G3139 for Bcl-2 mRNA | Breast cancer | Reduction in cell viability of breast cancer cells Induction of apoptosis Inhibition of tumor growth Decreasing expression of Bcl-2 | In vitro, in vivo | [229] |
| Gapmer for SRRM4 | SCLC | Inhibition of miRNA-4516 expression Inhibition of cell growth | In vitro, in vivo | [230] |
| Gapmer for NEAT1 | Multiple Myeloma | Inhibition of cell Activation of caspase 3 Inhibition of cell proliferation Inhibition of tumor growth | In vitro, in vivo | [231] |
| Gapmer ISTH0047 and ISTH2047 | Glioblastoma | Decrease expression of TGFB1/2 Inhibition of migration and invasion Increased survival of rodent glioma models in vivo | In vitro, in vivo | [232] |
| Gapmer for b2a2 and b3a2 BCR/ABL | Leukemia | Inhibit viability of cells Activation of executive caspases 3/7 | In vitro | [233] |
| SPC3042 | Prostate cancer | Increase expression of caspase 3/7 activity Decrease expression of antiapoptotic Bcl-2 mRNA Decrease tumor weight in combination with taxol | In vitro, in vivo | [234] |
| Gapmer for DNp73 | Lung cancer, Melanoma | Induction of apoptosis Inhibition of tumor growth | In vitro, in vivo | [235] |
| Gapmer for let-7 miRNA | Multiple myeloma | Decrease expression of MYC, KRAAS, CCND1, E2F6, DICER1, HMGA1 Inhibition of tumor growth | In vitro, in vivo | [236] |
| Gapmer for Bcl-2 | Lung cancer | Induction of apoptosis Decrease expression of Bcl-2 mRNA Inhibition of tumor growth | In vitro, in vivo | [237] |
| Gapmer targeting TGF-B3 | Glioblastoma | Decrease expression of pSMAD2, Inhibition of invasion, Inhibition of tumor growth | In vitro, in vivo | [238] |
| Gapmer for BC200 | Breast cancer, Hepatocellular cancer, Lung cancer, Ovarian Cancer | Induction of apoptosis | In vitro | [239] |
| GapmeR for XLOC_109948 | Acute Myeloid Leukemia | Induction of apoptosis | In vitro | [240] |
| Gapmer for lncRNA MALAT1 | Multiple Myeloma | Decrease expression of MALAT1, inhibition of colony formation, inhibition of tumor growth | In vitro, in vivo | [241] |
| Gapmer for MALAT1 | Multiple Myeloma | Increased activation of PAR signaling Induction of caspase-3 activity Activation of apoptosis | In vitro, in vivo | [242] |
| Gapmer for MALAT1 | Lung cancer | Decreased cell migration, reduced number of nodules | In vitro, in vivo | [243] |
| Gapmer for MALAT1 | Multiple Myeloma | Induction of apoptosis Inhibition of tumor growth | In vivo | [252] |
| Gapmer for miR-17–92s | Multiple Myeloma | Reduced viability of cells, induction of apoptosis | In vitro, in vivo | [244] |
| Gapmer for lncRNA PVT1 | Acute Erythroleukemia | Induction of apoptosis Inhibition of c-MYC expression | In vitro | [245,246] |
| Gapmers for Bcl-XL | Lung cancer | Inhibition of Bcl-2 expression Induction of apoptosis by activation of caspase 3 | In vitro | [247] |
| Gapmer for p21 | Breast cancer | Inhibition of ERα expression | In vitro | [248] |
| Gapmer for Bcl-2 | Breast cancer | Inhibition of cell viability Activation of caspase 3 | In vitro | [249] |
| Gapmer for Clusterin | Breast cancer | Induction of apoptosis in combination with trastuzumab | In vitro | [250] |
| Gapmer for SAMMSON | Melanoma | Inhibition of lncRNA SAMMSON | In vitro, in vivo | [251] |
| Name of Drug (Type of Drug) | Clinical Trial Phase | Objectives of Study | Cancer | Reference |
|---|---|---|---|---|
| Anti-mir-10b (antagomir) | Diagnostic (Recruiting) | Testing in vitro sensitivity of individual primary tumors | Glioblastomas | [255] |
| Cobomarsen/MRG-106 (antagomir) | Phase 2 (Terminated) | Inhibition of miRNA-155 | Cutaneous T-Cell Lymphoma | [256] |
| Phase 2 (Terminated) | Combination of cobomarsen with vorinostat | Cutaneous T-Cell Lymphoma | [257] | |
| Phase 1 (Completed) | Inhibition of miRNA-155 | Cutaneous T-cell Lymphoma Chronic Lymphocytic Leukemia Diffuse Large B-Cell Lymphoma Adult T-cell Leukemia | [258] | |
| OGX-011 | Phase 1 (Completed) | Increase anticancer activity of flutamide and buserelin | Prostate cancer | [259] |
| Phase 1 (Completed) | Combination therapy with docetaxel | Solid tumors | [260] | |
| Phase 2 (Completed) | Combination therapy with docetaxel | Breast cancer | [261] | |
| OGX427 (Apatorsen) | Phase 1 (Unknown) | Inhibition of Hsp27 to treat patients with advanced cancers | Advanced cancer | [262] |
| Phase 1 (Completed) | Safety of OGX427 in the treatment of prostate cancer, ovarian cancer, NSCLC, breast cancer, bladder cancer | Cancers | [263] | |
| Phase 2 (Completed) | Inhibition of Hsp27 to slow the progression of prostate cancer | Prostate cancer | [264] | |
| Phase 2 (Completed) | Combination therapy of OGX-427 and docetaxel | Bladder carcinoma | [265] | |
| Danvatirsen (AZD9150) | Phase 2 (Recruiting) | Effectiveness of combination therapy of danvatirsen and pembrolizumab | HNSCC | [266] |
| Phase 2 (Active, not recruiting) | Effectiveness of combination therapy davatirsen and durvalumab | Advanced and refractory pancreatic, non-small cell lung cancer, colorectal cancer | [267] | |
| Phase 2 (Completed) | Effectiveness of combination therapy of duralumab with oleclumab or monalizumab or danvatirsen | Lung cancer | [268] | |
| Phase 1 (Active, not recruiting) | Effectiveness of durvalumab with davatirsen/oleclumab/carboplatin/gemcitabine/cisplatin/Nab-paclitaxel | NSCLC | [269] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szymanowska, A.; Rodriguez-Aguayo, C.; Lopez-Berestein, G.; Amero, P. Non-Coding RNAs: Foes or Friends for Targeting Tumor Microenvironment. Non-Coding RNA 2023, 9, 52. https://doi.org/10.3390/ncrna9050052
Szymanowska A, Rodriguez-Aguayo C, Lopez-Berestein G, Amero P. Non-Coding RNAs: Foes or Friends for Targeting Tumor Microenvironment. Non-Coding RNA. 2023; 9(5):52. https://doi.org/10.3390/ncrna9050052
Chicago/Turabian StyleSzymanowska, Anna, Cristian Rodriguez-Aguayo, Gabriel Lopez-Berestein, and Paola Amero. 2023. "Non-Coding RNAs: Foes or Friends for Targeting Tumor Microenvironment" Non-Coding RNA 9, no. 5: 52. https://doi.org/10.3390/ncrna9050052
APA StyleSzymanowska, A., Rodriguez-Aguayo, C., Lopez-Berestein, G., & Amero, P. (2023). Non-Coding RNAs: Foes or Friends for Targeting Tumor Microenvironment. Non-Coding RNA, 9(5), 52. https://doi.org/10.3390/ncrna9050052