The Roles of microRNA miR-185 in Digestive Tract Cancers
Abstract
1. Introduction
2. Oral and Pharyngeal Cancers
2.1. Biology
2.2. Regulation
3. Esophageal Cancer
3.1. Biology
3.2. Regulation
4. Gastric Cancer
4.1. Biology
4.2. Regulation
5. Liver Cancer
5.1. Biology
5.2. Regulation
6. Pancreatic Cancer
Biology and Regulation
7. Colorectal Cancer
7.1. Biology
7.2. Regulation
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Orditura, M.; Galizia, G.; Sforza, V.; Gambardella, V.; Fabozzi, A.; Laterza, M.M.; Andreozzi, F.; Ventriglia, J.; Savastano, B.; Mabilia, A.; et al. Treatment of gastric cancer. World J. Gastroenterol. 2014, 20, 1635–1649. [Google Scholar] [CrossRef]
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting cancer incidence and deaths to 2030: The unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014, 74, 2913–2921. [Google Scholar] [CrossRef] [PubMed]
- Stenzinger, A.; Weichert, W. Genetic profiling of cancers of the digestive system: Biological insights and clinical implications. Pathobiology 2017, 84, 306–322. [Google Scholar] [CrossRef] [PubMed]
- Vedeld, H.M.; Goel, A.; Lind, G.E. Epigenetic biomarkers in gastrointestinal cancers: The current state and clinical perspectives. Semin. Cancer Biol. 2018, 51, 36–49. [Google Scholar] [CrossRef]
- Tazawa, H.; Nagasaka, T.; Kagawa, S.; Fujiwara, T. MicroRNA as a molecular target for gastrointestinal cancers. Transl. Gastrointest. Cancer 2015, 4, 219–235. [Google Scholar]
- Stang, A.; Weilert, H.; Lipp, M.J.; Oldhafer, K.J.; Hoheisel, J.D.; Zhang, C.; Bauer, A.S. MicroRNAs in blood act as biomarkers of colorectal cancer and indicate potential therapeutic targets. Mol. Oncol. 2021, 15, 2480–2490. [Google Scholar] [CrossRef] [PubMed]
- Botla, S.K.; Savant, S.; Jandaghi, P.; Bauer, A.S.; Mücke, O.; Moskalev, E.A.; Neoptolemos, J.P.; Costello, E.; Greenhalf, W.; Scarpa, A.; et al. Early epigenetic down-regulation of microRNA-192 expression promotes pancreatic cancer progression. Cancer Res. 2016, 76, 4149–4159. [Google Scholar] [CrossRef] [PubMed]
- Dror, S.; Sander, L.; Schwartz, H.; Sheinboim, D.; Barzilai, A.; Dishon, Y.; Apcher, S.; Golan, T.; Greenberger, S.; Barshack, I.; et al. Melanoma miRNA trafficking controls tumour primary niche formation. Nat. Cell Biol. 2016, 18, 1006–1017. [Google Scholar] [CrossRef]
- Keller, A.; Leidinger, P.; Vogel, B.; Backes, C.; ElSharawy, A.; Galata, V.; Mueller, S.C.; Marquart, S.; Schrauder, M.G.; Strick, R.; et al. miRNAs can be generally associated with human pathologies as exemplified for miR-144. BMC Med. 2014, 12, 224. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ha, M.; Kim, V.N. Regulation of microRNA biogenesis. Nat. Rev. Mol. Cell Biol. 2014, 15, 509–524. [Google Scholar] [CrossRef] [PubMed]
- Winter, J.; Jung, S.; Keller, S.; Gregory, R.I.; Diederichs, S. Many roads to maturity: Microrna biogenesis pathways and their regulation. Nat. Cell Biol. 2009, 11, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Croce, C.M. The role of MicroRNAs in human cancer. Signal Transduct. Target. Ther. 2016, 1, 15004. [Google Scholar] [CrossRef]
- Galvão-Lima, L.J.; Morais, A.H.F.; Valentim, R.A.M.; Barreto, E.J.S.S. miRNAs as biomarkers for early cancer detection and their application in the development of new diagnostic tools. Biomed. Eng. Online 2021, 20, 21. [Google Scholar] [CrossRef]
- Li, G.; Wang, Y.; Liu, Y.; Su, Z.; Liu, C.; Ren, S.; Deng, T.; Huang, D.; Tian, Y.; Qiu, Y. miR-185-3p regulates nasopharyngeal carcinoma radioresistance by targeting WNT2B in vitro. Cancer Sci. 2014, 105, 1560–1568. [Google Scholar] [CrossRef]
- Xu, J.; Ai, Q.; Cao, H.; Liu, Q. MiR-185-3p and miR-324-3p predict radiosensitivity of nasopharyngeal carcinoma and modulate cancer cell growth and apoptosis by targeting SMAD7. Med. Sci. Monit. 2015, 21, 2828–2836. [Google Scholar] [CrossRef]
- Liu, C.; Li, G.; Ren, S.; Su, Z.; Wang, Y.; Tian, Y.; Liu, Y.; Qiu, Y. miR-185-3p regulates the invasion and metastasis of nasopharyngeal carcinoma by targeting WNT2B in vitro. Oncol. Lett. 2017, 13, 2631–2636. [Google Scholar] [CrossRef]
- de Ceuninck van Capelle, C.; Spit, M.; Ten Dijke, P. Current perspectives on inhibitory SMAD7 in health and disease. Crit. Rev. Biochem. Mol. Biol. 2020, 55, 691–715. [Google Scholar] [CrossRef]
- Cheng, J.-Z.; Chen, J.-J.; Wang, Z.-G.; Yu, D. MicroRNA-185 inhibits cell proliferation while promoting apoptosis and autophagy through negative regulation of TGF-β1/mTOR axis and HOXC6 in nasopharyngeal carcinoma. Cancer Biomark. 2018, 23, 107–123. [Google Scholar] [CrossRef]
- Wang, L.; Yin, P.; Wang, J.; Wang, Y.; Sun, Z.; Zhou, Y.; Guan, X. Delivery of mesenchymal stem cells-derived extracellular vesicles with enriched miR-185 inhibits progression of OPMD. Artif. Cells Nanomed. Biotechnol. 2019, 47, 2481–2491. [Google Scholar] [CrossRef] [PubMed]
- Bersani, C.; Mints, M.; Tertipis, N.; Haeggblom, L.; Näsman, A.; Romanitan, M.; Dalianis, T.; Ramqvist, T. MicroRNA-155, -185 and -193b as biomarkers in human papillomavirus positive and negative tonsillar and base of tongue squamous cell carcinoma. Oral Oncol. 2018, 82, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Ramdas, L.; Giri, U.; Ashorn, C.L.; Coombes, K.R.; El-Naggar, A.; Ang, K.K.; Story, M.D. miRNA expression profiles in head and neck squamous cell carcinoma and adjacent normal tissue. Head Neck 2009, 31, 642–654. [Google Scholar] [CrossRef]
- Ratovitski, E.A. Phospho-ΔNp63α regulates AQP3, ALOX12B, CASP14 and CLDN1 expression through transcription and microRNA modulation. FEBS Lett. 2013, 587, 3581–3586. [Google Scholar] [CrossRef] [PubMed]
- Tomasik, B.; Papis-Ubych, A.; Stawiski, K.; Fijuth, J.; Kędzierawski, P.; Sadowski, J.; Stando, R.; Bibik, R.; Graczyk, Ł.; Latusek, T.; et al. Serum microRNAs as xerostomia biomarkers in patients with oropharyngeal cancer undergoing radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 2021, 111, 1237–1249. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Pan, J.; Luo, Z.; Duan, Q.; Wang, D. Long non-coding RNA FOXD3-AS1 silencing exerts tmor suppressive effects in nasopharyngeal carcinoma by downregulating FOXD3 expression via microRNA-185-3p upregulation. Cancer Gene Ther. 2021, 28, 602–618. [Google Scholar] [CrossRef]
- Li, C.; Li, W.; Cao, S.; Xu, J.; Qian, Y.; Pan, X.; Lei, D.; Wei, D. Circ_0058106 promotes proliferation, metastasis and EMT process by regulating Wnt2b/β-catenin/c-Myc pathway through miR-185-3p in hypopharyngeal squamous cell carcinoma. Cell Death Dis. 2021, 12, 1063. [Google Scholar] [CrossRef]
- Wang, Z.; Zhu, X.; Dong, P.; Cai, J. Long noncoding RNA LINC00958 promotes the oral squamous cell carcinoma by sponging miR-185-5p/YWHAZ. Life Sci. 2020, 242, 116782. [Google Scholar] [CrossRef]
- Gan, Y.; Ye, F.; He, X.X. The role of YWHAZ in cancer: A maze of opportunities and challenges. J. Cancer 2020, 11, 2252–2264. [Google Scholar] [CrossRef]
- Sun, C.-C.; Zhang, L.; Li, G.; Li, S.J.; Chen, Z.L.; Fu, Y.F.; Gong, F.Y.; Bai, T.; Zhang, D.Y.; Wu, Q.M.; et al. The lncRNA PDIA3P interacts with miR-185-5p to modulate oral squamous cell carcinoma progression by targeting cyclin D2. Mol. Ther. Nucleic Acid. 2017, 9, 100–110. [Google Scholar] [CrossRef]
- Wang, X.; Feng, X.; Wang, H. LncRNA LSINCT5 drives proliferation and migration of oral squamous cell carcinoma through the miRNA-185-5p/ZNF703 axis. J. Balk. Union Oncol. 2021, 26, 124–131. [Google Scholar]
- Bao, Q.; Liao, X.; Li, R.; Ding, N. KCNQ1OT1 promotes migration and inhibits apoptosis by modulating miR-185-5p/Rab14 axis in oral squamous cell carcinoma. Dev. Growth Differ. 2019, 61, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Li, B.X.; Yu, Q.; Shi, Z.L.; Li, P.; Fu, S. Circulating microRNAs in esophageal squamous cell carcinoma: Association with locoregional staging and survival. Int. J. Clin. Exp. Med. 2015, 8, 7241–7250. [Google Scholar] [PubMed]
- Li, C.Y.; Zhang, W.W.; Xiang, J.L.; Wang, X.H.; Li, J.; Wang, J.L. Identification of microRNAs as novel biomarkers for esophageal squamous cell carcinoma: A study based on the Cancer Genome Atlas (TCGA) and bioinformatics. Chin. Med. J. 2019, 132, 2213–2222. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Li, Y.; Luo, L.; Xiong, Q.; Zhong, X.; Xie, F.; Feng, P. Identification of miRNAs and genes for predicting Barrett’s esophagus progressing to esophageal adenocarcinoma using miRNA-mRNA integrated analysis. PLoS ONE 2021, 16, e0260353. [Google Scholar] [CrossRef]
- Wang, B.; Hao, X.; Li, X.; Liang, Y.; Li, F.; Yang, K.; Chen, H.; Lv, F.; Gao, Y. Long noncoding RNA HEIH depletion depresses esophageal carcinoma cell progression by upregulating microRNA-185 and downregulating KLK5. Cell Death Dis. 2020, 11, 1002. [Google Scholar] [CrossRef]
- Jing, R.; Chen, W.; Wang, H.; Ju, S.; Cong, H.; Sun, B.; Jin, Q.; Chu, S.; Xu, L.; Cui, M. Plasma miR-185 is decreased in patients with esophageal squamous cell carcinoma and might suppress tumor migration and invasion by targeting RAGE. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 309, G719–G729. [Google Scholar] [CrossRef]
- Zhao, Z.T.; Zhou, W.; Liu, L.Y.; Lan, T.; Zhan, Q.M.; Song, Y.M. Molecular mechanism and effect of microRNA185 on proliferation, migration and invasion of esophageal squamous cell carcinoma. Natl. Med. J. China 2013, 93, 1426–1431. [Google Scholar]
- Palanissami, G.; Paul, S.F.D. RAGE and Its Ligands: Molecular Interplay between Glycation, Inflammation, and Hallmarks of Cancer—A Review. Horm. Cancer 2018, 9, 295–325. [Google Scholar] [CrossRef]
- Wu, W.; Ren, Z.; Li, P.; Yu, D.; Chen, J.; Huang, R.; Liu, H. Six1: A critical transcription factor in tumorigenesis. Int. J. Cancer 2015, 136, 1245–1253. [Google Scholar] [CrossRef]
- Su, H.; Jin, X.; Zhang, X.; Xue, S.; Deng, X.; Shen, L.; Fang, Y.; Xie, C. Identification of microRNAs involved in the radioresistance of esophageal cancer cells. Cell Biol. Int. 2014, 38, 318–325. [Google Scholar] [CrossRef]
- Zheng, Z.F.; Su, H.F.; Zou, Y.; Peng, Z.; Wu, S.X. Expression profiles of microRNAs in radioresistant esophageal cell line. Natl. Med. J. China 2011, 91, 639–642. [Google Scholar]
- Martin, T.E.; Powell, C.T.; Wang, Z.; Bhattacharyya, S.; Walsh-Reitz, M.M.; Agarwal, K.; Toback, F.G. A novel mitogenic protein that is highly expressed in cells of the gastric antrum mucosa. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 285, G332–G343. [Google Scholar] [CrossRef][Green Version]
- Liu, J.-Q.; Deng, M.; Xue, N.-N.; Li, T.X.; Guo, Y.X.; Gao, L.; Zhao, D.; Fan, R.T. LncRNA KLF3-AS1 suppresses cell migration and invasion in ESCC by impairing miR-185-5p-targeted KLF3 inhibition. Mol. Ther. Nucleic Acid. 2020, 20, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Bibi, F.; Pushparaj, P.N.; Naseer, M.I.; Yasir, M.; Azhar, E.I. Decrypting the microRNA signatures in gastric cancer using high-throughput miRNA array coupled with systems biological approaches for precision medicine. Genet. Mol. Res. 2017, 16, 16039845. [Google Scholar] [CrossRef]
- Xing, R.; Li, W.; Cui, J.; Zhang, J.; Kang, B.; Wang, Y.; Wang, Z.; Liu, S.; Lu, Y. Gastrokine 1 induces senescence through p16/Rb pathway activation in gastric cancer cells. Gut 2012, 61, 43–52. [Google Scholar] [CrossRef]
- Alarcón-Millán, J.; Martínez-Carrillo, D.N.; Peralta-Zaragoza, O.; Fernández-Tilapa, G. Regulation of GKN1 expression in gastric carcinogenesis: A problem to resolve (Review). Int. J. Oncol. 2019, 55, 555–569. [Google Scholar] [PubMed]
- Choi, B.J.; Yoon, J.H.; Choi, W.S.; Kim, O.; Nam, S.W.; Lee, J.Y.; Park, W.S. GKN1 and miR-185 are associated with CpG island methylator phenotype in gastric cancers. Mol. Cell. Toxicol. 2013, 9, 227–233. [Google Scholar] [CrossRef]
- Yoon, J.H.; Choi, Y.J.; Choi, W.S.; Ashktorab, H.; Smoot, D.T.; Nam, S.W.; Lee, J.Y.; Park, W.S. GKN1-miR-185-DNMT1 axis suppresses gastric carcinogenesis through regulation of epigenetic alteration and cell cycle. Clin. Cancer Res. 2013, 19, 4599–4610. [Google Scholar] [CrossRef]
- Yoon, J.H.; Choi, Y.J.; Choi, W.S.; Nam, S.W.; Lee, J.Y.; Park, W.S. Functional analysis of the NH2-terminal hydrophobic region and BRICHOS domain of GKN1. Biochem. Biophys. Res. Commun. 2013, 440, 689–695. [Google Scholar] [CrossRef]
- Yoon, J.H.; Choi, W.S.; Kim, O.; Choi, B.J.; Nam, S.W.; Lee, J.Y.; Park, W.S. Gastrokine 1 inhibits gastric cancer cell migration and invasion by downregulating RhoA expression. Gastric Cancer 2017, 20, 274–285. [Google Scholar] [CrossRef]
- Hao, T.; Jiang, J.; Wu, W.; Tanaka, A.; Katoh, H.; Aburatani, H.; Lauwers, G.Y.; Fukayama, M. RhoA mutations in diffuse-type gastric cancer. Dig. Med. Res. 2020, 3, 4. [Google Scholar] [CrossRef]
- Li, Y.; Tan, B.B.; Zhao, Q.; Fan, L.Q.; Wang, D.; Liu, Y.; Liu, Q.W. Effect and mechanism of miR-185 inhibition to drug resistance of human gastric cancer cell line SGC7901. Chin. J. Cancer Prev. Treat. 2015, 22, 1347–1351. [Google Scholar]
- Li, Q.; Wang, J.X.; He, Y.Q.; Feng, C.; Zhang, X.J.; Sheng, J.Q.; Li, P.F. MicroRNA-185 regulates chemotherapeutic sensitivity in gastric cancer by targeting apoptosis repressor with caspase recruitment domain. Cell Death Dis. 2014, 5, e1197. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.; Li, Y.; Zhao, Q.; Fan, L.; Wang, D. ZNF139 increases multidrug resistance in gastric cancer cells by inhibiting miR-185. Biosci. Rep. 2018, 38, BSR20181023. [Google Scholar] [CrossRef]
- Tan, B.; Li, Y.; Di, Y.; Fan, L.; Zhao, Q.; Liu, Q.; Wang, D.; Jia, N. Clinical value of peripheral blood microRNA detection in evaluation of SOX regimen as neoadjuvant chemotherapy for gastric cancer. J. Clin. Lab. Anal. 2018, 32, e22363. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.; Jiang, H.; Wu, Y.; Xie, L.; Dai, W.; Tang, H.; Tang, S. MiR-185 is an independent prognosis factor and suppresses tumor metastasis in gastric cancer. Mol. Cell. Biochem. 2014, 386, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Tan, B.; Li, Y.; Zhao, Q.; Yuan, H.; Liu, Y.; Wang, D.; Zhang, Z. Upregulation of miR-185 promotes apoptosis of the human gastric cancer cell line MGC803. Mol. Med. Rep. 2018, 17, 3115–3122. [Google Scholar] [CrossRef] [PubMed]
- Qiu, F.; Xiong, J.-P.; Deng, J.; Xiang, X.-J. TRIM29 functions as an oncogene in gastric cancer and is regulated by miR-185. Int. J. Clin. Exp. Pathol. 2015, 8, 5053–5061. [Google Scholar] [PubMed]
- Huang, C.; Wu, Y.-J.; He, W.-F.; Zhao, S.L.; Ouyang, Y.Y.; Ai, X.H.; Liu, Z.Q.; Tang, S.Y. MiR-185-3p regulates epithelial mesenchymal transition via PI3K/Akt signaling pathway by targeting cathepsin D in gastric cancer cells. Transl. Cancer Res. 2020, 9, 6988–7000. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Suo, A.L.; Li, Z.F.; Liu, L.Y.; Tian, T.; Ni, L.; Zhang, W.G.; Nan, K.J.; Song, T.S.; Huang, C. MicroRNA profiling of human gastric cancer. Mol. Med. Rep. 2009, 2, 963–970. [Google Scholar] [PubMed]
- Treece, A.L.; Duncan, D.L.; Tang, W.; Elmore, S.; Morgan, D.R.; Dominguez, R.L.; Speck, O.; Meyers, M.O.; Gulley, M.L. Gastric adenocarcinoma microRNA profiles in fixed tissue and in plasma reveal cancer-associated and Epstein-Barr virus-related expression patterns. Lab. Investig. 2016, 96, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Xu, W.; Liu, J.; Liu, X.; Huo, B.; Li, B.; Wang, Z. Optimizing miRNA-module diagnostic biomarkers of gastric carcinoma via integrated network analysis. PLoS ONE 2018, 13, e0198445. [Google Scholar] [CrossRef]
- Zhou, X.; Zhu, W.; Li, H.; Wen, W.; Cheng, W.; Wang, F.; Wu, Y.; Qi, L.; Fan, Y.; Chen, Y.; et al. Diagnostic value of a plasma microRNA signature in gastric cancer: A microRNA expression analysis. Sci. Rep. 2015, 5, 11251. [Google Scholar] [CrossRef]
- Huang, Z.; Zhu, D.; Wu, L.; He, M.; Zhou, X.; Zhang, L.; Zhang, H.; Wang, W.; Zhu, J.; Cheng, W.; et al. Six serum-based miRNAs as potential diagnostic biomarkers for gastric cancer. Cancer Epidemiol. Biomark. Prev. 2017, 26, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.; Kim, H.K.; Rettig, R.L.; Kim, J.; Lee, E.T.; Aprelikova, O.; Choi, I.J.; Munroe, D.J.; Green, J.E. MiRNA signature associated with outcome of gastric cancer patients following chemotherapy. BMC Med. Genom. 2011, 4, 79. [Google Scholar] [CrossRef]
- Zhang, Q.; Chen, B.; Liu, P.; Yang, J. XIST promotes gastric cancer (GC) progression through TGF-β1 via targeting miR-185. J. Cell. Biochem. 2018, 119, 2787–2796. [Google Scholar] [CrossRef]
- Wu, X.; Shan, Y.; Cao, F.; Zhang, B.; Wang, H.; Liu, H.; Xu, Y.; Yu, H. Long noncoding RNA FOXD2-AS1 incuced gastric cancer cell MGC-803 to apatinib resistance by regulating miR-185-5p/CCND2 modulating axis. Chin. J. Cancer Biother. 2018, 25, 1104–1112. [Google Scholar]
- Arias-Romero, L.E.; Chernoff, J. Targeting Cdc42 in cancer. Expert Opin. Ther. Target. 2013, 17, 1263–1273. [Google Scholar] [CrossRef]
- Ma, W.; Sze, K.M.F.; Chan, L.K.; Lee, J.M.; Wei, L.L.; Wong, C.M.; Lee, T.K.; Wong, C.C.; Ng, I.O. RhoE/ROCK2 regulates chemoresistance through NF-κB/IL-6/STAT3 signaling in hepatocellular carcinoma. Oncotarget 2016, 7, 41445–41459. [Google Scholar] [CrossRef]
- Zhang, Q.; Chen, Y.; Liu, K. miR-185 inhibits cell migration and invasion of hepatocellular carcinoma through CDC42. Oncol. Lett. 2018, 16, 3101–3107. [Google Scholar] [CrossRef]
- Niu, Y.; Tang, G. miR-185-5p targets ROCK2 and inhibits cell migration and invasion of hepatocellular carcinoma. Oncol. Lett. 2019, 17, 5087–5093. [Google Scholar] [CrossRef]
- Lin, Z.; He, R.; Luo, H.; Lu, C.; Ning, Z.; Wu, Y.; Han, C.; Tan, G.; Wang, Z. Integrin-β5, a MIR-185-targeted gene, promotes hepatocellular carcinoma tumorigenesis by regulating β-catenin stability. J. Exp. Clin. Cancer Res. 2018, 37, 17. [Google Scholar] [CrossRef]
- Zhu, S.-M.; Chen, C.-M.; Jiang, Z.-Y.; Yuan, B.; Ji, M.; Wu, F.H.; Jin, J. MicroRNA-185 inhibits cell proliferation and epithelial-mesenchymal transition in hepatocellular carcinoma by targeting Six2. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 1712–1719. [Google Scholar] [PubMed]
- Qadir, X.V.; Han, C.; Lu, D.; Zhang, J.; Wu, T. MiR-185 inhibits hepatocellular carcinoma growth by targeting the DNMT1/PTEN/Akt pathway. Am. J. Pathol. 2014, 184, 2355–2364. [Google Scholar] [CrossRef]
- Qadir, X.V.; Han, C.; Wu, T. Epigenetic regulation of PTEN by miR-185 and miR-200b in liver cancer. Am. Assoc. Cancer Res. 2012, 78, 3143. [Google Scholar] [CrossRef]
- Zhou, L.; Liu, S.; Han, M.; Feng, S.; Liang, J.; Li, Z.; Li, Y.; Lu, H.; Liu, T.; Ma, Y.; et al. MicroRNA-185 induces potent autophagy via AKT signaling in hepatocellular carcinoma. Tumor Biol. 2017, 39, 1010428317694313. [Google Scholar] [CrossRef]
- Zhi, Q.; Zhu, J.; Guo, X.; He, S.; Xue, X.; Zhou, J.; Hu, B.; Li, H.; Chen, S.; Zhao, H.; et al. Metastasis-related miR-185 is a potential prognostic biomarker for hepatocellular carcinoma in early stage. Biomed. Pharmacother. 2013, 67, 393–398. [Google Scholar] [CrossRef]
- Kim, M.; Kogan, N.; Slack, F.J. Cis-acting elements in its 3′ UTR mediate post-transcriptional regulation of KRAS. Oncotarget 2016, 7, 11770–11784. [Google Scholar] [CrossRef]
- Li, J.; Jin, B.; Wang, T.; Li, W.; Wang, Z.; Zhang, H.; Song, Y.; Li, N. Serum microRNA expression profiling identifies serum biomarkers for HCV-related hepatocellular carcinoma. Cancer Biomark. 2019, 26, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Han, J.; Chen, J.; Dong, J.; Xia, Y.; Liu, J.; Jiang, Y.; Dai, J.; Lu, J.; Jin, G.; et al. Plasma miRNAs as early biomarkers for detecting hepatocellular carcinoma. Int. J. Cancer 2015, 137, 1679–1690. [Google Scholar] [CrossRef] [PubMed]
- Budhu, A.; Jia, H.L.; Forgues, M.; Liu, C.G.; Goldstein, D.; Lam, A.; Zanetti, K.A.; Ye, Q.H.; Qin, L.X.; Croce, C.M.; et al. Identification of metastasis-related microRNAs in hepatocellular carcinoma. Hepatology 2008, 47, 897–907. [Google Scholar] [CrossRef]
- Zou, L.; Chai, J.; Gao, Y.; Guan, J.; Liu, Q.; Du, J.J. Down-regulated PLAC8 promotes hepatocellular carcinoma cell proliferation by enhancing PI3K/Akt/GSK3β/Wnt/β-catenin signaling. Biomed. Pharmacother. 2016, 84, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhang, Z.; Zhao, D.; Feng, W.; Meng, F.; Han, S.; Lin, B.; Shi, X. Long Noncoding RNA (lncRNA) FOXD2-AS1 promotes cell proliferation and metastasis in hepatocellular carcinoma by regulating MiR-185/AKT Axis. Med. Sci. Monit. 2019, 25, 9618–9629. [Google Scholar] [CrossRef] [PubMed]
- Tran, D.D.H.; Kessler, C.; Niehus, S.E.; Mahnkopf, M.; Koch, A.; Tamura, T. Myc target gene, long intergenic noncoding RNA, Linc00176 in hepatocellular carcinoma regulates cell cycle and cell survival by titrating tumor suppressor microRNAs. Oncogene 2018, 37, 75–85. [Google Scholar] [CrossRef]
- Braconi, C.; Kogure, T.; Valeri, N.; Huang, N.; Nuovo, G.; Costinean, S.; Negrini, M.; Miotto, E.; Croce, C.M.; Patel, T. MicroRNA-29 can regulate expression of the long non-coding RNA gene MEG3 in hepatocellular cancer. Oncogene 2011, 30, 4750–4756. [Google Scholar] [CrossRef]
- Zamani, M.; Sadeghizadeh, M.; Behmanesh, M.; Najafi, F. Dendrosomal curcumin increases expression of the long non-coding RNA gene MEG3 via up-regulation of epi-miRs in hepatocellular cancer. Phytomedicine 2015, 22, 961–967. [Google Scholar] [CrossRef]
- Otsuka, M.; Takata, A.; Yoshikawa, T.; Kojima, K.; Kishikawa, T.; Shibata, C.; Takekawa, M.; Yoshida, H.; Omata, M.; Koike, K. Receptor for activated protein kinase C: Requirement for efficient microRNA function and reduced expression in hepatocellular carcinoma. PLoS ONE 2011, 6, e24359. [Google Scholar] [CrossRef]
- Fan, H.X.; Feng, Y.J.; Zhao, X.P.; He, Y.Z.; Tang, H. MiR-185-5p suppresses HBV gene expression by targeting ELK1 in hepatoma carcinoma cells. Life Sci. 2018, 213, 9–17. [Google Scholar] [CrossRef]
- Wang, Y.-P.; Huang, Y.; Hou, T.; Lu, M. LncRNA XIST acts as a ceRNA sponging miR-185-5p to modulate pancreatic cancer cell proliferation via targeting CCND2. Transl. Cancer Res. 2020, 9, 1427–1438. [Google Scholar] [CrossRef]
- Wang, W.; Li, X.; Guan, C.; Hu, Z.; Zhao, Y.; Li, W.; Jiang, X. LncRNA PCAT6 promotes the proliferation, migration and invasion of pancreatic ductal adenocarcinoma via regulating miR-185-5p/CBX2 axis. Pathol. Res. Pract. 2020, 216, 153074. [Google Scholar] [CrossRef] [PubMed]
- Morvaridi, S.; Dhall, D.; Greene, M.I.; Pandol, S.J.; Wang, Q. Role of YAP and TAZ in pancreatic ductal adenocarcinoma and in stellate cells associated with cancer and chronic pancreatitis. Sci. Rep. 2015, 5, 16759. [Google Scholar] [CrossRef]
- Xia, D.; Li, X.; Niu, Q.; Liu, X.; Xu, W.; Ma, C.; Gu, H.; Liu, Z.; Shi, L.; Tian, X.; et al. MicroRNA-185 suppresses pancreatic cell proliferation by targeting transcriptional coactivator with PDZ-binding motif in pancreatic cancer. Exp. Ther. Med. 2018, 15, 657–666. [Google Scholar] [CrossRef]
- Gao, W.; Wang, J.; Wang, M.; Wang, C. Potential gene and microRNA biomarkers for pancreatic cancer with gene expression analysis. Int. J. Clin. Exp. Pathol. 2016, 9, 1917–1924. [Google Scholar]
- Liu, R.; Chen, X.; Du, Y.; Yao, W.; Shen, L.; Wang, C.; Hu, Z.; Zhuang, R.; Ning, G.; Zhang, C.; et al. Serum microRNA expression profile as a biomarker in the diagnosis and prognosis of pancreatic cancer. Clin. Chem. 2012, 58, 610–618. [Google Scholar] [CrossRef]
- Al-Shaheri, F.N.; Alhamdani, M.S.S.; Bauer, A.S.; Giese, N.; Büchler, M.W.; Hackert, T.; Hoheisel, J.D. Blood biomarkers for differential diagnosis and early detection of pancreatic cancer. Cancer Treat. Rev. 2021, 96, 102193. [Google Scholar] [CrossRef]
- Sawicki, T.; Ruszkowska, M.; Danielewicz, A.; Niedźwiedzka, E.; Arłukowicz, T.; Przybyłowicz, K.E. A review of colorectal cancer in terms of epidemiology, risk factors, development, symptoms and diagnosis. Cancers 2021, 13, 2025. [Google Scholar] [CrossRef]
- Zhang, W.; Sun, Z.; Su, L.; Wang, F.; Jiang, Y.; Yu, D.; Zhang, F.; Sun, Z.; Liang, W. miRNA-185 serves as a prognostic factor and suppresses migration and invasion through Wnt1 in colon cancer. Eur. J. Pharmacol. 2018, 825, 75–84. [Google Scholar] [CrossRef]
- Dong-Xu, W.; Jia, L.; Su-Juan, Z. MicroRNA-185 is a novel tumor suppressor by negatively modulating the Wnt/β-catenin pathway in human colorectal cancer. Indian J. Cancer 2015, 52, e182–e185. [Google Scholar] [CrossRef]
- Liu, C.; Ji, L.; Song, X. Long non coding RNA UCA1 contributes to the autophagy and survival of colorectal cancer cells via sponging miR-185-5p to up-regulate the WISP2/β-catenin pathway. RSC Adv. 2019, 9, 14160–14166. [Google Scholar] [CrossRef]
- Liu, M.; Lang, N.; Chen, X.; Tang, Q.; Liu, S.; Huang, J.; Zheng, Y.; Bi, F. MiR-185 targets RhoA and Cdc42 expression and inhibits the proliferation potential of human colorectal cells. Cancer Lett. 2011, 301, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.M.; Lu, H. Autoregulatory suppression of c-Myc by miR-185-3p. J. Biol. Chem. 2011, 286, 33901–33909. [Google Scholar] [CrossRef]
- Zhou, C.; Kong, W.; Ju, T.; Xie, Q.; Zhai, L. MiR-185-3p mimic promotes the chemosensitivity of CRC cells via AQP5. Cancer Biol. Ther. 2020, 21, 790–798. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liu, X.; Feng, B.; Liu, N.; Wu, Q.; Han, Y.; Nie, Y.; Wu, K.; Shi, Y.; Fan, D. STIM1, a direct target of microRNA-185, promotes tumor metastasis and is associated with poor prognosis in colorectal cancer. Oncogene 2015, 34, 4808–4820. [Google Scholar] [CrossRef] [PubMed]
- Afshar, S.; Najafi, R.; Sedighi Pashaki, A.; Sharifi, M.; Nikzad, S.; Gholami, M.H.; Khoshghadam, A.; Amini, R.; Karimi, J.; Saidijam, M. MiR-185 enhances radiosensitivity of colorectal cancer cells by targeting IGF1R and IGF2. Biomed. Pharmacother. 2018, 106, 763–769. [Google Scholar] [CrossRef]
- Lu, Z.J.; Lu, L.G.; Tao, K.Z.; Chen, D.F.; Xia, Q.; Weng, J.J.; Zhu, F.; Wang, X.P.; Zheng, P. MicroRNA-185 suppresses growth and invasion of colon cancer cells through inhibition of the hypoxiainducible factor-2α pathway in vitro and in vivo. Mol. Med. Rep. 2014, 10, 2401–2408. [Google Scholar] [CrossRef]
- Yuan, M.; Zhang, X.; Zhang, J.; Wang, K.; Zhang, Y.; Shang, W.; Zhang, Y.; Cui, J.; Shi, X.; Na, H.; et al. DC-SIGN–LEF1/TCF1–miR-185 feedback loop promotes colorectal cancer invasion and metastasis. Cell Death Differ. 2020, 27, 379–395. [Google Scholar] [CrossRef]
- Li, J.; Liu, H.; Zou, L.; Ke, J.; Zhang, Y.; Zhu, Y.; Yang, Y.; Gong, Y.; Tian, J.; Zou, D.; et al. A functional variant in GREM1 confers risk for colorectal cancer by disrupting a hsa-miR-185-3p binding site. Oncotarget 2017, 8, 61318–61326. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Meng, X.; Peng, X.; Yao, Q.; Zhu, F.; Ding, Z.; Sun, H.; Liu, X.; Li, D.; Lu, Y.; et al. Impaired AGO2/miR-185-3p/NRP1 axis promotes colorectal cancer metastasis. Cell Death Dis. 2021, 12, 390. [Google Scholar] [CrossRef]
- Vivekanandhan, S.; Mukhopadhyay, D. Genetic status of KRAS influences transforming growth factor-beta (TGF-β) signaling: An insight into neuropilin-1 (NRP1) mediated tumorigenesis. Semin. Cancer Biol. 2019, 54, 72–79. [Google Scholar] [CrossRef]
- Tang, H.I.; Deng, M.; Liao, Q.J. Expression and clinical significance of mir-185 in tissues of colon carcinoma. J. Med. Sci. Cent. S. China 2011, 5, 1. [Google Scholar]
- Zhang, N.; Hu, X.; Du, Y.; Du, J. The role of miRNAs in colorectal cancer progression and chemoradiotherapy. Biomed. Pharmacother. 2021, 134, 111099. [Google Scholar] [CrossRef]
- Bobowicz, M.; Skrzypski, M.; Czapiewski, P.; Marczyk, M.; Maciejewska, A.; Jankowski, M.; Szulgo-Paczkowska, A.; Zegarski, W.; Pawłowski, R.; Polańska, J.; et al. Prognostic value of 5-microRNA based signature in T2-T3N0 colon cancer. Clin. Exp. Metastasis 2016, 33, 765–773. [Google Scholar] [CrossRef]
- Matsumura, T.; Sugimachi, K.; Iinuma, H.; Takahashi, Y.; Kurashige, J.; Sawada, G.; Ueda, M.; Uchi, R.; Ueo, H.; Takano, Y.; et al. Exosomal microRNA in serum is a novel biomarker of recurrence in human colorectal cancer. Br. J. Cancer 2015, 113, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.; Leong, S.M.; Kee, Z.; Caramat, P.V.; Teo, J.; Blanco, M.; Koay, E.; Cheong, W.K.; Soh, T.I.; Yong, W.P.; et al. Longitudinal monitoring reveals dynamic changes in circulating tumor cells (CTCs) and CTC-associated miRNAs in response to chemotherapy in metastatic colorectal cancer patients. Cancer Lett. 2018, 423, 1–8. [Google Scholar] [CrossRef]
- Sur, D.; Balacescu, L.; Cainap, S.S.; Visan, S.; Pop, L.; Burz, C.; Havasi, A.; Buiga, R.; Cainap, C.; Irimie, A.; et al. Predictive efficacy of MiR-125b-5p, MiR-17-5p, and MiR-185-5p in liver metastasis and chemotherapy response among advanced stage colorectal cancer patients. Front. Oncol. 2021, 11, 651380. [Google Scholar] [CrossRef]
- Maciel-Dominguez, A.; Swan, D.; Ford, D.; Hesketh, J. Selenium alters miRNA profile in an intestinal cell line: Evidence that miR-185 regulates expression of GPX2 and SEPSH2. Mol. Nutr. Food Res. 2013, 57, 2195–2205. [Google Scholar] [CrossRef]
- Akçakaya, P.; Ekelund, S.; Kolosenko, I.; Caramuta, S.; Ozata, D.M.; Xie, H.; Lindforss, U.; Olivecrona, H.; Lui, W.O. miR-185 and miR-133b deregulation is associated with overall survival and metastasis in colorectal cancer. Int. J. Oncol. 2011, 39, 311–318. [Google Scholar] [PubMed]
- Baldi, S.; Khamgan, H.; Qian, Y.; Wu, H.; Zhang, Z.; Zhang, M.; Gao, Y.; Safi, M.; Al-Radhi, M.; Zuo, Y.F. Downregulated ARID1A by miR-185 is associated with poor prognosis and adverse outcomes in colon adenocarcinoma. Front. Oncol. 2021, 11, 679334. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, W.; Nan, F.; Ren, F.; Wang, H.; Xu, Y.; Zhang, F. MicroRNA expression profile of colon cancer stem-like cells in HT29 adenocarcinoma cell line. Biochem. Biophys. Res. Commun. 2011, 404, 273–278. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, X.; Wang, Y. Altered gene expression and miRNA expression associated with cancerous IEC-6 cell transformed by MNNG. J. Exp. Clin. Cancer Res. 2009, 28, 56. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.; Zhang, J.; Yang, C.; Xiang, L.; Liu, W. Silencing of long noncoding RNA UCA1 inhibits colon cancer invasion, migration and epithelial-mesenchymal transition and tumour formation by upregulating miR-185-5p in vitro and in vivo. Cell Biochem. Funct. 2020, 38, 176–184. [Google Scholar] [CrossRef]
- Wang, M.; Zheng, Q.; Zhao, Z.; Deng, H.; Zhang, Q.; Yao, H. HES5 activates long noncoding RNA UCA1 to induce colorectal cancer progression by modulating miR-185/NOTCH3 signaling. Gastroenterol. Res. Pract. 2021, 2021, 7249818. [Google Scholar] [CrossRef]
- Ou, C.; Sun, Z.; He, X.; Li, X.; Fan, S.; Zheng, X.; Peng, Q.; Li, G.; Li, X.; Ma, J. Targeting YAP1/LINC00152/FSCN1 signaling axis prevents the progression of colorectal cancer. Adv. Sci. 2020, 7, 1901380. [Google Scholar] [CrossRef]
- Zhuang, S.-T.; Cai, Y.-J.; Liu, H.-P.; Qin, Y.; Wen, J.F. LncRNA NEAT1/miR-185-5p/IGF2 axis regulates the invasion and migration of colon cancer. Mol. Genet. Genom. Med. 2020, 8, e1125. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Qiao, L.; Zhou, Y.; Ma, N.; Wang, C.; Zhou, J. Long non-coding RNA FOXD2-AS1 contributes to colorectal cancer proliferation through its interaction with microRNA-185-5p. Cancer Sci. 2018, 109, 2235–2242. [Google Scholar] [CrossRef]
- Sun, W.; Nie, W.; Wang, Z.; Zhang, H.; Li, Y.; Fang, X. Lnc haglr promotes colon cancer progression through sponging mir-185-5p and activating cdk4 and cdk6 in vitro and in vivo. Onco Target. Ther. 2020, 13, 5913–5925. [Google Scholar] [CrossRef]
- Zhang, L.; Dong, X.; Yan, B.; Yu, W.; Shan, L. CircAGFG1 drives metastasis and stemness in colorectal cancer by modulating YY1/CTNNB1. Cell Death Dis. 2020, 11, 542. [Google Scholar] [CrossRef]
- Lu, W.; Huang, Z.; Wang, J.; Liu, H. Long non-coding RNA DANCR accelerates colorectal cancer progression via regulating the miR-185-5p/HMGA2 axis. J. Biochem. 2022, 171, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Shi, C.; Xu, D.; Lin, X.; Ji, T.; Shi, Z.; Zhuge, X.; Zhuo, S.; Yang, Q. LncRNA ASB16-AS1 drives proliferation, migration, and invasion of colorectal cancer cells through regulating miR-185-5p/TEAD1 axis. Cell Cycle 2021, 21, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.; Tian, Q.; Guan, L.; Niu, S. The dual role of miR-186 in cancers: Oncomir battling with tumor suppressor miRNA. Front. Oncol. 2020, 10, 233. [Google Scholar] [CrossRef] [PubMed]
- Dagogo-Jack, I.; Shaw, A.T. Tumour heterogeneity and resistance to cancer therapies. Nat. Rev. Clin. Oncol. 2017, 15, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Erhard, F.; Haas, J.; Lieber, D.; Malterer, G.; Jaskiewicz, L.; Zavolan, M.; Dölken, L.; Zimmer, R. Widespread context dependency of microRNA-mediated regulation. Genome Res. 2014, 24, 906–919. [Google Scholar] [CrossRef] [PubMed]

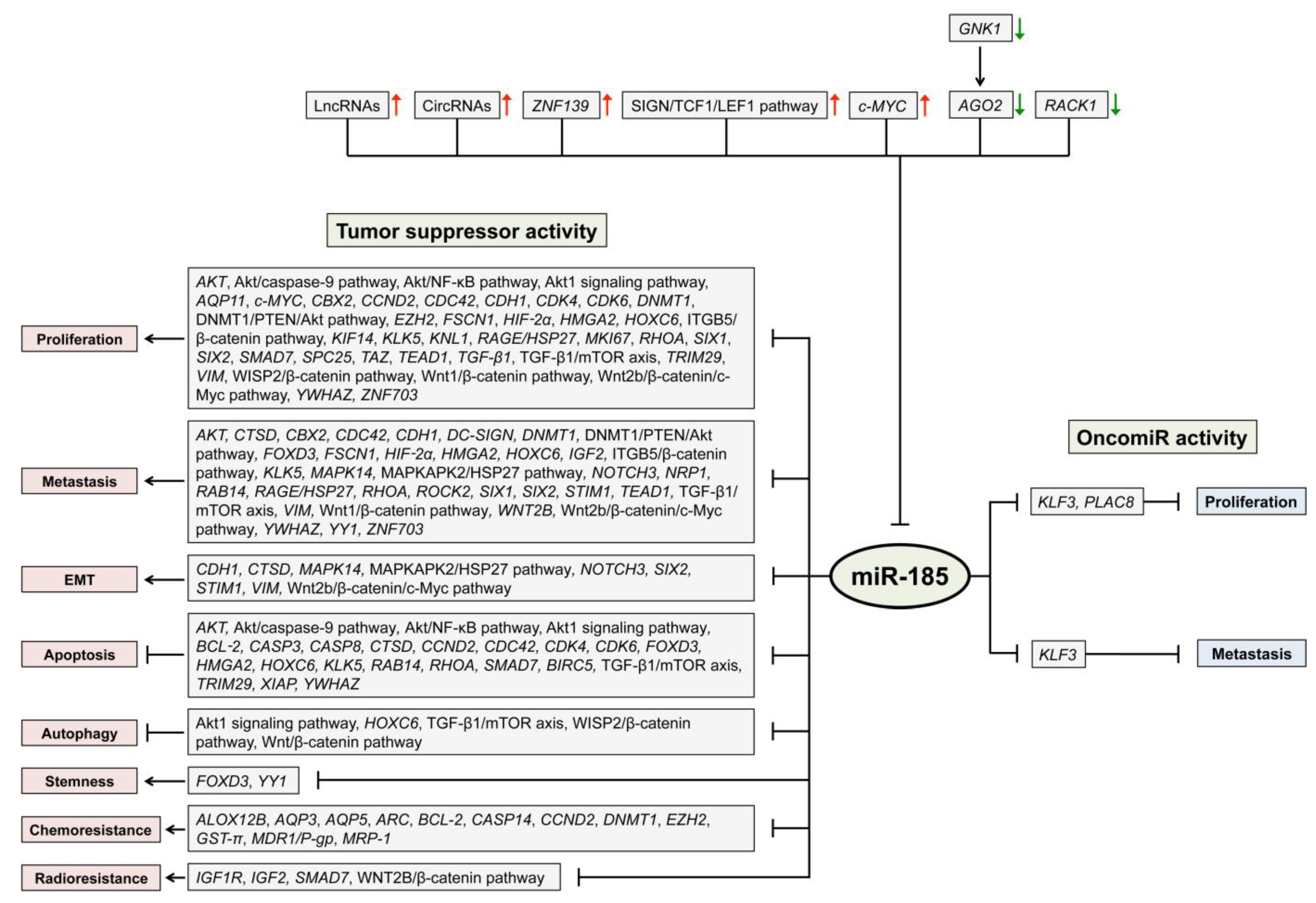
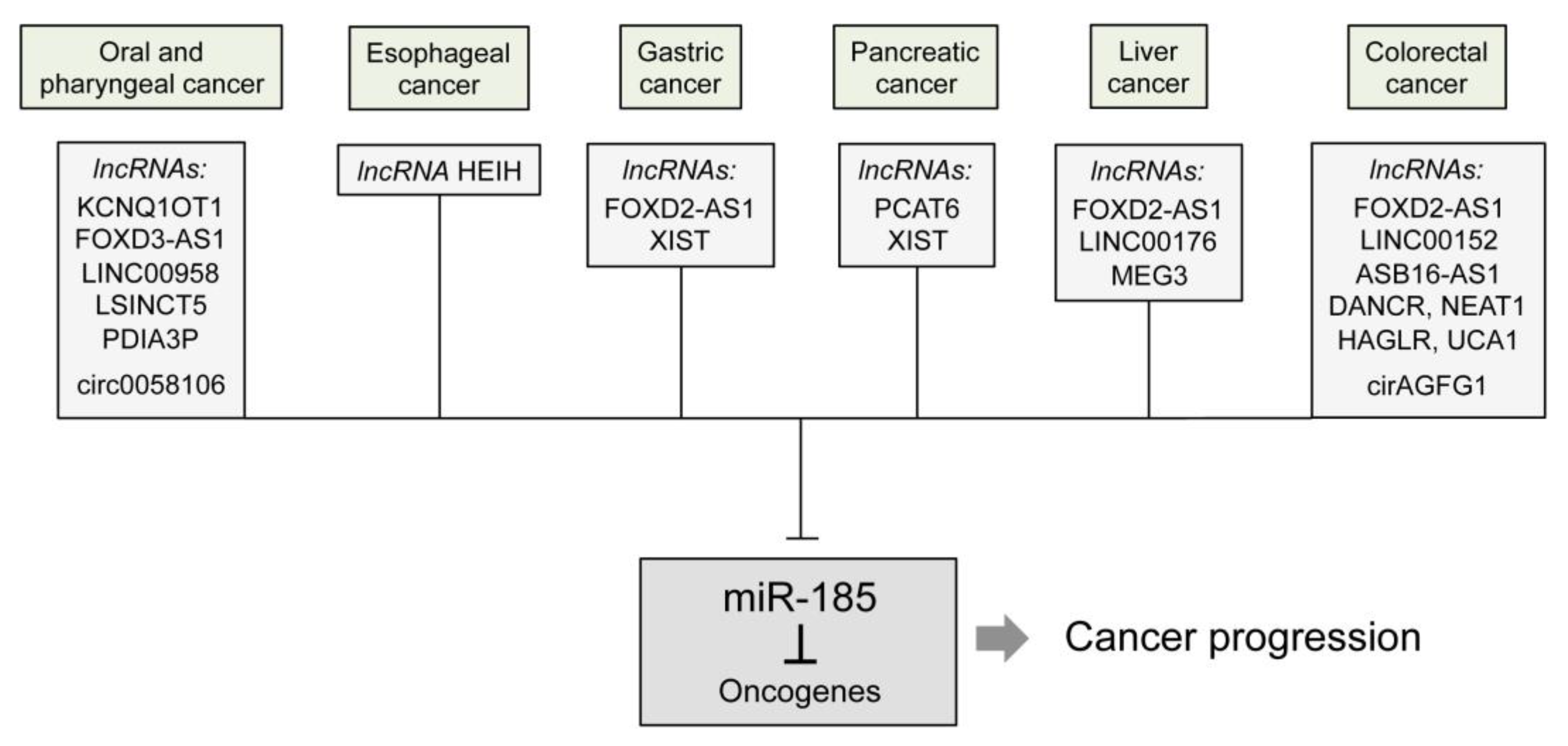
| Cancer Tissue | Identified by In Vitro Experiments | Predicted from Data | Verified by In Vivo Experiments |
|---|---|---|---|
| Mouth and pharynx | ALOX12B, AQP3,CASP-14,CCND2, FOXD3, HOXC6, RAB14,SMAD7, TGF-β1/mTOR axis, WNT2B, YWHAZ, ZNF703, Akt/CASP-9 pathway, Akt/NF-κB pathway, Wnt2b/β-catenin/c-Myc pathway | CCND2, FOXD3, HOXC6, YWHAZ, ZNF703, Akt/CASP-9 pathway, Akt/NF-κB pathway | |
| Esophagus | KLF3, KLK5,RAGE, SIX1 | KLK5,RAGE | |
| Stomach | ARC, BCL-2, CASP-3, CASP-8, CCND2, CTSD,DNMT1, EZH2,GST-π, MDR1/P-gp, MRP-1,RHOA, TGF-β1, TRIM29, XIAP, Wnt/β-catenin pathway | AQP5, ESRRA, RAC3, RGS14, ZNFN1A4 | ARC,CTSD, DNMT1, EZH2, RHOA |
| Liver | AQP11, AKT, CDC42, CDH1, DNMT1, DNMT3A, DNMT3B, ELK1, KIF14,KNL1, MEG3,MKI67, PLAC8,ROCK2, SIX2, SPC25, VIM, Akt1 signaling pathway, DNMT1/PTEN/Akt pathway, ITGB5/β-catenin- and PLAC8/Wnt/β-catenin pathway | CDC42, DNMT1/PTEN/Akt pathway, ITGB5/β-catenin pathway | |
| Pancreas | CBX2, CCND2, TAFAZZIN | CORO2B, NTRK3 | CBX2, CCND2, TAFAZZIN |
| Colon or rectum | AQP5, ARID1A,c-MYC, CDC42, CDK4, CDK6, DC-SIGN, FSCN1, GREM1, HIF-2α, HMGA2, IGF1R, IGF2, MAPK14, NOTCH3, NRP1, RHOA, STIM1, TEAD1, YY1, MAPKAPK2/HSP27 pathway, WISP2/β-catenin pathway, Wnt/β-catenin pathway | AQP5, CDC42, CDK4, CDK6, DC-SIGN, FSCN1, HIF-2α, HMGA2, IGF2, MAPK14, NRP1, STIM1, TEAD1,YY1, MAPKAPK2/HSP27 pathway, WISP2/β-catenin pathway |
| Upstream Regulator | Process | Affected Gene or Pathway/Function | Ref. |
|---|---|---|---|
| FOXD3-AS1 | Reduction of miR-185 level | FOXD3 | [26] |
| irc0058106 | Wnt2b/β-catenin/c-Myc pathway | [27] | |
| LINC00958 | YWHAZ | [28] | |
| PDIA3P | CCND2 | [30] | |
| LSINCT5 | ZNF703 | [31] | |
| KCNQ1OT1 | Increased tumorigenesis and reduced apoptosis | [32] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Babaeenezhad, E.; Naghibalhossaini, F.; Rajabibazl, M.; Jangravi, Z.; Hadipour Moradi, F.; Fattahi, M.D.; Hoheisel, J.D.; Sarabi, M.M.; Shahryarhesami, S. The Roles of microRNA miR-185 in Digestive Tract Cancers. Non-Coding RNA 2022, 8, 67. https://doi.org/10.3390/ncrna8050067
Babaeenezhad E, Naghibalhossaini F, Rajabibazl M, Jangravi Z, Hadipour Moradi F, Fattahi MD, Hoheisel JD, Sarabi MM, Shahryarhesami S. The Roles of microRNA miR-185 in Digestive Tract Cancers. Non-Coding RNA. 2022; 8(5):67. https://doi.org/10.3390/ncrna8050067
Chicago/Turabian StyleBabaeenezhad, Esmaeel, Fakhraddin Naghibalhossaini, Masoumeh Rajabibazl, Zohreh Jangravi, Forouzan Hadipour Moradi, Mohammad Davood Fattahi, Jörg D. Hoheisel, Mostafa Moradi Sarabi, and Soroosh Shahryarhesami. 2022. "The Roles of microRNA miR-185 in Digestive Tract Cancers" Non-Coding RNA 8, no. 5: 67. https://doi.org/10.3390/ncrna8050067
APA StyleBabaeenezhad, E., Naghibalhossaini, F., Rajabibazl, M., Jangravi, Z., Hadipour Moradi, F., Fattahi, M. D., Hoheisel, J. D., Sarabi, M. M., & Shahryarhesami, S. (2022). The Roles of microRNA miR-185 in Digestive Tract Cancers. Non-Coding RNA, 8(5), 67. https://doi.org/10.3390/ncrna8050067






