Thermosensitive Intravitreal In Situ Implant of Cefuroxime Based on Poloxamer 407 and Hyaluronic Acid
Abstract
:1. Introduction
2. Results and Discussion
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Methods
4.3. Development of In Vitro Model
4.4. IV. Chorioallantoic Membrane Test
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Durand, M.L. Bacterial and Fungal Endophthalmitis. Clin. Microbiol. Rev. 2017, 30, 597–613. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, Y.; Schimel, A.M.; Pathengay, A.; Colyer, M.H.; Flynn, H.W. Endophthalmitis following open-globe injuries. Eye 2012, 26, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Relhan, N.; Forster, R.K.; Flynn, H.W. Endophthalmitis: Then and Now. Am. J. Ophthalmol. 2018, 187, xx–xxvii. [Google Scholar] [CrossRef] [PubMed]
- Haseeb, A.A.; Elhusseiny, A.M.; Siddiqui, M.Z.; Ahmad, K.T.; Sallam, A.B. Fungal Endophthalmitis: A Comprehensive Review. J. Fungi 2021, 7, 996. [Google Scholar] [CrossRef]
- Khambati, A.; Wright, R.E.; Das, S.; Pasula, S.; Sepulveda, A.; Hernandez, F.; Kanwar, M.; Chandrasekar, P.; Kumar, A. Aspergillus Endophthalmitis: Epidemiology, Pathobiology, and Current Treatments. J. Fungi 2022, 8, 656. [Google Scholar] [CrossRef]
- Nikolaenko, V.P.; Belov, D.F. Antibiotic prophylaxis of acute postoperative endophthalmitis. Russ. J. Clin. Ophthalmol. 2021, 21, 220–226. [Google Scholar] [CrossRef]
- Azar, G.; Thiagasorupan, P.; Dureau, P.; Martin, G.; Edelson, C.; Metge, F.; Caputo, G. Pediatric endophthalmitis: Clinical profile, outcomes, and a proposed protocol. Graefes Arch. Clin. Exp. Ophthalmol. 2021, 259, 3405–3412. [Google Scholar] [CrossRef]
- Tan, C.L.; Sheorey, H.; Allen, P.J.; Dawkins, R.C.H. Endophthalmitis: Microbiology and Organism Identification Using Current and Emerging Techniques. Ocul. Immunol. Inflamm. 2023, 31, 393–401. [Google Scholar] [CrossRef]
- Gower, E.W.; Lindsley, K.; Tulenko, S.E.; Nanji, A.A.; Leyngold, I.; McDonnell, P.J. Perioperative antibiotics for prevention of acute endophthalmitis after cataract surgery. Cochrane Database Syst. Rev. 2017, 2, CD006364. [Google Scholar] [CrossRef]
- Rana, K.; Bahrami, B.; Zyl, L.; Esterman, A.; Goggin, M. Efficacy of intracameral antibiotics following manual small incision cataract surgery in reducing the rates of endophthalmitis: A meta-analysis. Clin. Exp. Ophthalmol. 2021, 49, 25–37. [Google Scholar] [CrossRef]
- Far, P.M.; Yeung, S.C.; Farimani, P.L.; Qian, J.; Zhang, A.Q.; Kertes, P.J.; You, Y.; Yan, P. Tap and inject versus pars plana vitrectomy for postprocedural endophthalmitis. Retina 2021, 41, 2009–2016. [Google Scholar] [CrossRef]
- Bowen, R.C.; Zhou, A.X.; Bondalapati, S.; Lawyer, T.W.; Snow, K.B.; Evans, P.R.; Bardsley, T.; McFarland, M.; Kliethermes, M.; Shi, D. Comparative analysis of the safety and efficacy of intracameral cefuroxime, moxifloxacin and vancomycin at the end of cataract surgery: A meta-analysis. Br. J. Ophthalmol. 2018, 102, 1268–1276. [Google Scholar] [CrossRef]
- Svetozarskiy, S.N.; Andreev, A.N. Complications of intracameral cefuroxime in cataract surgery. Vestnik Ophtalmol. 2018, 134, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Irimia, T.; Dinu-Pîrvu, C.-E.; Ghica, M.; Lupuleasa, D.; Muntean, D.-L.; Udeanu, D.; Popa, L. Chitosan-Based In Situ Gels for Ocular Delivery of Therapeutics: A State-of-the-Art Review. Mar. Drugs 2018, 16, 373. [Google Scholar] [CrossRef] [PubMed]
- Agrahari, V.; Mandal, A.; Agrahari, V.; Trinh, H.M.; Joseph, M.; Ray, A.; Hadji, H.; Mitra, R.; Pal, D.; Mitra, A.K. A comprehensive insight on ocular pharmacokinetics. Drug Deliv. Transl. Res. 2016, 6, 735–754. [Google Scholar] [CrossRef]
- Jumelle, C.; Gholizadeh, S.; Annabi, N.; Dana, R. Advances and limitations of drug delivery systems formulated as eye drops. J. Control Release 2020, 321, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Nagpal, M. Intravitreal moxifloxacin injections in acute post-cataract surgery endophthalmitis: Efficacy and safety. Indian J. Ophthalmol. 2021, 69, 326–330. [Google Scholar] [CrossRef] [PubMed]
- Tarragó, R.; Olea, J.L.; Ramírez, C.; Escudero, L. Endoftalmitis tras inyecciones intravítreas. Incidencia, manejo y pronóstico. Arch. Soc. Española Oftalmol. 2017, 92, 107–111. [Google Scholar] [CrossRef]
- Patel, D.; Patel, S.N.; Chaudhary, V.; Garg, S.J. Complications of intravitreal injections: 2022. Curr. Opin. Ophthalmol. 2022, 33, 137–146. [Google Scholar] [CrossRef]
- de la Torre, C.; Coll, C.; Ultimo, A.; Sancenón, F.; Martínez-Máñez, R.; Ruiz-Hernández, E. In Situ-Forming Gels Loaded with Stimuli-Responsive Gated Mesoporous Silica Nanoparticles for Local Sustained Drug Delivery. Pharmaceutics 2023, 15, 1071. [Google Scholar] [CrossRef]
- Logan, C.M.; Fernandes-Cunha, G.M.; Chen, F.; Le, P.; Mundy, D.; Na, K.S.; Myung, D. In Situ-forming Collagen Hydrogels Crosslinked by Multifunctional Polyethylene Glycol as a Matrix Therapy for Corneal Defects: 2-Month Follow-up In Vivo. Cornea 2023, 42, 97–104. [Google Scholar] [CrossRef]
- Bisht, R.; Jaiswal, J.K.; Chen, Y.-S.; Jin, J.; Rupenthal, I.D. Light-responsive in situ forming injectable implants for effective drug delivery to the posterior segment of the eye. Expert. Opin. Drug Deliv. 2016, 13, 953–962. [Google Scholar] [CrossRef]
- Wang, Q.; Sun, C.; Xu, B.; Tu, J.; Shen, Y. Synthesis, physicochemical properties and ocular pharmacokinetics of thermosensitive in situ hydrogels for ganciclovir in cytomegalovirus retinitis treatment. Drug Deliv. 2018, 25, 59–69. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, Z.; Xie, D.; Liu, D.; Chen, Z.; Li, K.; Li, Z.; Tichnell, B.; Liu, Z. Design and synthesis study of the thermo-sensitive poly (N-vinylpyrrolidone-b- N, N-diethylacrylamide). Des. Monomers Polym. 2018, 21, 43–54. [Google Scholar] [CrossRef] [PubMed]
- Taheri, S.L.; Rezazadeh, M.; Hassanzadeh, F.; Akbari, V.; Dehghani, A.; Talebi, A.; Mostafavi, S.A. Preparation, physicochemical, and retinal anti-angiogenic evaluation of poloxamer hydrogel containing dexamethasone/avastin-loaded chitosan-N-acetyl-L-cysteine nanoparticles. Int. J. Biol. Macromol. 2022, 220, 1605–1618. [Google Scholar] [CrossRef] [PubMed]
- Ottonelli, I.; Bighinati, A.; Adani, E.; Loll, F.; Caraffi, R.; Vandelli, M.A.; Boury, F.; Tosi, G.; Duskey, J.T.; Marigo, V. Optimization of an Injectable Hydrogel Depot System for the Controlled Release of Retinal-Targeted Hybrid Nanoparticles. Pharmaceutics 2022, 15, 25. [Google Scholar] [CrossRef]
- Yu, Y.; Lau, L.C.M.; Lo, A.C.; Chau, Y. Injectable Chemically Crosslinked Hydrogel for the Controlled Release of Bevacizumab in Vitreous: A 6-Month In Vivo Study. Trans. Vis. Sci. Tech. 2015, 4, 5. [Google Scholar] [CrossRef]
- Awwad, S.; Abubakre, A.; Angkawinitwong, U.; Khaw, P.T.; Brocchini, S. In situ antibody-loaded hydrogel for intravitreal delivery. Eur. J. Pharm. Sci. 2019, 137, 104993. [Google Scholar] [CrossRef]
- Thacker, M.; Tseng, C.-L.; Lin, F.-H. Substitutes and Colloidal System for Vitreous Replacement and Drug Delivery: Recent Progress and Future Prospective. Polymers 2020, 13, 121. [Google Scholar] [CrossRef] [PubMed]
- Barth, H.; Crafoord, S.; Andréasson, S.; Ghosh, F. A cross-linked hyaluronic acid hydrogel (Healaflow®) as a novel vitreous substitute. Graefes Arch. Clin. Exp. Ophthalmol. 2016, 254, 697–703. [Google Scholar] [CrossRef]
- Soliman, K.A.; Ullah, K.; Shah, A.; Jones, D.S.; Singh, T.R.R. Poloxamer-based in situ gelling thermoresponsive systems for ocular drug delivery applications. Drug Discov. Today 2019, 24, 1575–1586. [Google Scholar] [CrossRef]
- Adrianto, M.F.; Annuryanti, F.; Wilson, C.G.; Sheshala, R.; Thakur, R.R.S. In vitro dissolution testing models of ocular implants for posterior segment drug delivery. Drug Deliv. Transl. Res. 2022, 12, 1355–1375. [Google Scholar] [CrossRef]
- Lin, H.-R.; Sung, K.C. Carbopol/pluronic phase change solutions for ophthalmic drug delivery. J. Control Release 2000, 69, 379–388. [Google Scholar] [CrossRef]
- Shahmatov, K.S. The study of the viscosity of the vitreous body from Cadaver. Mod. Probl. Sci. Educ. Surg. 2017, 5. [Google Scholar]
- Loch, C.; Nagel, S.; Guthoff, R.; Seidlitz, A.; Weitschies, W. The Vitreous Model—A new in vitro test method simulating the vitreous body. Biomed. Eng. 2012, 57, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Ribatti, D. The chick embryo chorioallantoic membrane (CAM). A multifaceted experimental model. Mech. Dev. 2016, 141, 70–77. [Google Scholar] [CrossRef]
- Shinoda, K.; Matsumoto, S.C.; Yagura, K.; Terauchi, G.; Shoji, T.; Yoshikawa, Y.; Igawa, Y.; Mizota, A.; Miyake, Y. Intraocular Temperature Distribution in Eyes Undergoing Different Types of Surgical Procedures during Vitreous Surgery. J. Clin. Med. 2022, 11, 2053. [Google Scholar] [CrossRef] [PubMed]
- Thakur, S.S.; Shenoy, S.K.; Suk, J.S.; Hanes, J.S.; Rupenthal, I.D. Validation of hyaluronic acid-agar-based hydrogels as vitreous humor mimetics for in vitro drug and particle migration evaluations. Eur. J. Pharm. Biopharm. 2020, 148, 118–125. [Google Scholar] [CrossRef]
- Kim, H.M.; Ha, S.; Hong, H.K.; Hwang, Y.; Kim, P.; Yang, E.; Chung, J.Y.; Park, S.; Park, Y.J.; Park, K.H. Intraocular Distribution and Kinetics of Intravitreally Injected Antibodies and Nanoparticles in Rabbit Eyes. Trans. Vis. Sci. Tech. 2020, 9, 20. [Google Scholar] [CrossRef]
- Auel, T.; Scherke, L.P.; Hadlich, S.; Mouchantat, S.; Grimm, M.; Weitschies, W.; Seidlitz, A. Ex Vivo Visualization of Distribution of Intravitreal Injections in the Porcine Vitreous and Hydrogels Simulating the Vitreous. Pharmaceutics 2023, 15, 786. [Google Scholar] [CrossRef]
- Henein, C.; Awwad, S.; Ibeanu, N.; Vlatakis, S.; Brocchini, S.; Tee Khaw, P.; Bouremel, Y. Hydrodynamics of Intravitreal Injections into Liquid Vitreous Substitutes. Pharmaceutics 2019, 11, 371. [Google Scholar] [CrossRef]
- Auel, T.; Großmann, L.; Schulig, L.; Weitschies, W.; Seidlitz, A. The EyeFlowCell: Development of a 3D-Printed Dissolution Test Setup for Intravitreal Dosage Forms. Pharmaceutics 2021, 13, 1394. [Google Scholar] [CrossRef]
- Bakhrushina, E.O.; Novozhilova, E.V.; Shumkova, M.M.; Pyzhov, V.S.; Nikonenko, M.S.; Bardakov, A.I.; Demina, N.B.; Krasnyuk, I.I.; Krasnyuk, I.I. New Biopharmaceutical Characteristics of In Situ Systems Based on Poloxamer 407. Gels 2023, 9, 508. [Google Scholar] [CrossRef]
- Bakhrushina, E.O.; Pomyutkina, M.V.; Popova, A.A.; Khodenok, A.I.; Demina, N.B. Study of poloxamer 188 and polyethylene glycols influence on in situ systems thermoreversible properties. Prob. Biol. Med. Pharm. Chem. 2022, 25, 20–25. [Google Scholar] [CrossRef]
- Salem, H.F.; Kharshoum, R.M.; Abou-Taleb, H.A.; Naguib, D.M. Nanosized Transferosome-Based Intranasal In Situ Gel for Brain Targeting of Resveratrol: Formulation, Optimization, In Vitro Evaluation, and In Vivo Pharmacokinetic Study. AAPS PharmSciTech 2019, 20, 181. [Google Scholar] [CrossRef]
- Gan, S.; Zakaria, S.; Chia, C.H.; Chen, R.S.; Ellis, A.V.; Kaco, H. Highly porous regenerated cellulose hydrogel and aerogel prepared from hydrothermal synthesized cellulose carbamate. PLoS ONE 2017, 12, e0173743. [Google Scholar] [CrossRef] [PubMed]
- Stein, S.; Auel, T.; Kempin, W.; Bogdahn, M.; Weitschies, W.; Seidlitz, A. Influence of the test method on in vitro drug release from intravitreal model implants containing dexamethasone or fluorescein sodium in poly (d,l-lactide-co-glycolide) or polycaprolactone. Eur. J. Pharm. Biopharm. 2018, 127, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Stein, S.; Bogdahn, M.; Rosenbaum, C.; Weitschies, W.; Seidlitz, A. Distribution of fluorescein sodium and triamcinolone acetonide in the simulated liquefied and vitrectomized Vitreous Model with simulated eye movements. Eur. J. Pharm. Sci. 2017, 109, 233–243. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, X.; Liu, Y.; Di, X. Thermosensitive In Situ Gel Based on Solid Dispersion for Rectal Delivery of Ibuprofen. AAPS PharmSciTech 2018, 19, 338–347. [Google Scholar] [CrossRef]
- Zhang, C.; Xu, T.; Zhang, D.; He, W.; Wang, S.; Jiang, T. Disulfiram thermosensitive in-situ gel based on solid dispersion for cataract. Asian J. Pharm. 2018, 13, 527–535. [Google Scholar] [CrossRef]
- Koval’skii, I.V.; Krasnyuk, I.I.; Krasnyuk, I.I.; Nikulina, O.I.; Belyatskaya, A.V.; Kharitonov, Y.Y.; Fel’dman, N.B.; Lutsenko, S.V.; Grikh, V.V. Studies of the Solubility of Rutin from Solid Dispersions. Pharm. Chem. J. 2014, 47, 612–615. [Google Scholar] [CrossRef]



| Ingredient | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
|---|---|---|---|---|---|---|---|---|
| Cefuroxime | - | - | - | - | - | - | - | - |
| P407 | 18% | 18% | 18% | 18% | 18% | 18% | 18% | 18% |
| P188 | 0.5% | 1% | - | 2% | 1.5 | 2.5% | 3% | - |
| HA | 0.5% | 0.5% | 0.5% | 0.5% | 0.5% | 0.5% | 0.5% | 0.5% |
| PEG 1500 | - | - | - | - | - | - | - | 1.5% |
| Ascorbic acid | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 |
| Purified water | ad 20 mL | |||||||
| Composition | Critical Points of Change in the Indicator (Weeks) | Average Temperature of the PHASE transition (°C) | SD | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 18 | 19 | 20 | 21 | 22 | 39 | 41 | |||
| 1 | 31.4 | 27 | 26 | 27.8 | 27.2 | 24 * | - | - | 28.2 | 3.79 |
| 2 | 32.8 | 28.6 | 28.5 | 28.8 | 25.3 * | - | - | - | 30.7 | 2.08 |
| 3 | 29.9 | 27.6 | 25 * | - | - | - | - | - | 27.9 | 1.54 |
| 4 | 33.1 | 31.4 | 30.9 | 25 * | - | - | - | - | 33.2 | 1.71 |
| 5 | 32.7 | 28 | 27 | 24.5 * | - | - | - | - | 30.9 | 5.22 |
| 6 | 43.9 | 34 | 34 | 36 | 34 | 33 | 25 * | - | 35.0 | 5.34 |
| 7 | 44.3 | 38 | 37 | 37 | 36 | 36 | 31 | 25 * | 36.4 | 6.40 |
| 8 | 35 | 25 * | - | - | - | - | - | - | 31.7 | 3.81 |
| Composition | Type of Hyaluronic Acid | Average Temperature of the Phase Transition (°C) | SD | pH | Diffusion Volume (% of Model Volume) |
|---|---|---|---|---|---|
| C1 | Hyaluronic acid (Cube3) | 34.8 | 6.13 | 6.89 | 8.6 ± 1.65 |
| C2 | Hyaluronic acid (400–600) | 37.4 | 4.38 | 6.65 | 25.3 ± 2.4 |
| C3 | Hyaluronic acid (1400–1800) | 37.1 | 4.14 | 6.62 | 7.0 ± 1.9 |
| Composition | Type of Solid Dispersion | Average Temperature of the Phase Transition (°C) | SD | pH | Diffusion Volume (% of Model Volume) |
|---|---|---|---|---|---|
| C4 | PVP (K15) | 36.3 | 2.72 | 6.72 | 6.3 ±1.5 |
| C5 | PEG 400 | 34.6 | 2.69 | 6.69 | 8.0 ± 2.3 |
| C6 | PEG 1500 | 35.7 | 3.19 | 6.70 | 5.6 ± 2.0 |
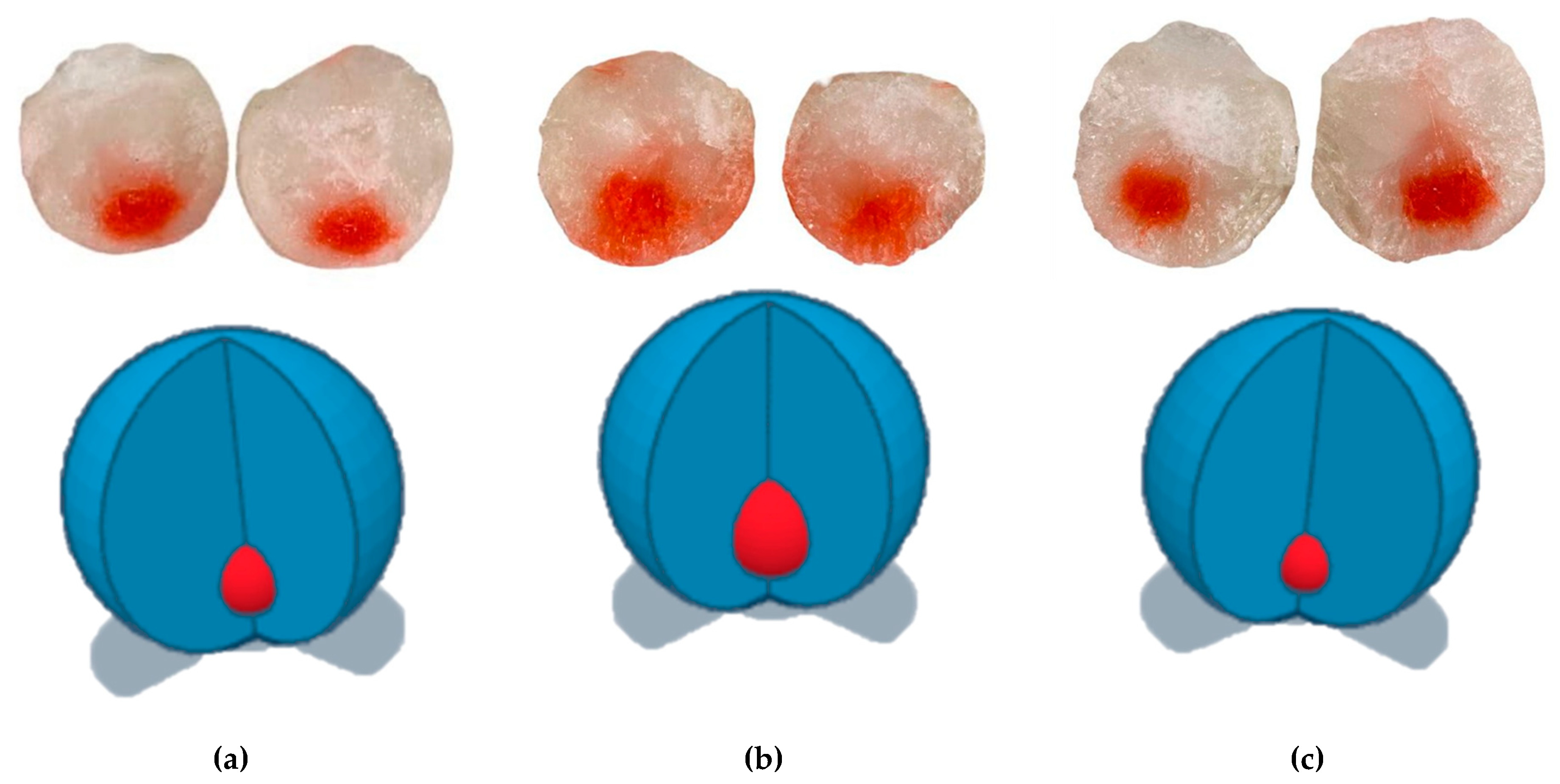
| Range | Parameter |
|---|---|
| 0 | The color of the membrane has not been changed, no hemorrhage |
| 1 | Noticeable disappearance of membrane color, no hemorrhage |
| 2 | Partial loss of membrane color, a hemorrhage |
| 3 | Complete color loss, hemorrhage over the entire surface of the film’s contact with the membrane |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bakhrushina, E.O.; Dubova, A.I.; Nikonenko, M.S.; Grikh, V.V.; Shumkova, M.M.; Korochkina, T.V.; Krasnyuk, I.I., Jr.; Krasnyuk, I.I. Thermosensitive Intravitreal In Situ Implant of Cefuroxime Based on Poloxamer 407 and Hyaluronic Acid. Gels 2023, 9, 693. https://doi.org/10.3390/gels9090693
Bakhrushina EO, Dubova AI, Nikonenko MS, Grikh VV, Shumkova MM, Korochkina TV, Krasnyuk II Jr., Krasnyuk II. Thermosensitive Intravitreal In Situ Implant of Cefuroxime Based on Poloxamer 407 and Hyaluronic Acid. Gels. 2023; 9(9):693. https://doi.org/10.3390/gels9090693
Chicago/Turabian StyleBakhrushina, Elena O., Anastasia I. Dubova, Maria S. Nikonenko, Viktoriya V. Grikh, Marina M. Shumkova, Tatyana V. Korochkina, Ivan I. Krasnyuk, Jr., and Ivan I. Krasnyuk. 2023. "Thermosensitive Intravitreal In Situ Implant of Cefuroxime Based on Poloxamer 407 and Hyaluronic Acid" Gels 9, no. 9: 693. https://doi.org/10.3390/gels9090693





