Antibacterial Activity of PVA Hydrogels Embedding Oxide Nanostructures Sensitized by Noble Metals and Ruthenium Dye
Abstract
1. Introduction
2. Results and Discussion
2.1. Electron Microscopy Characterization
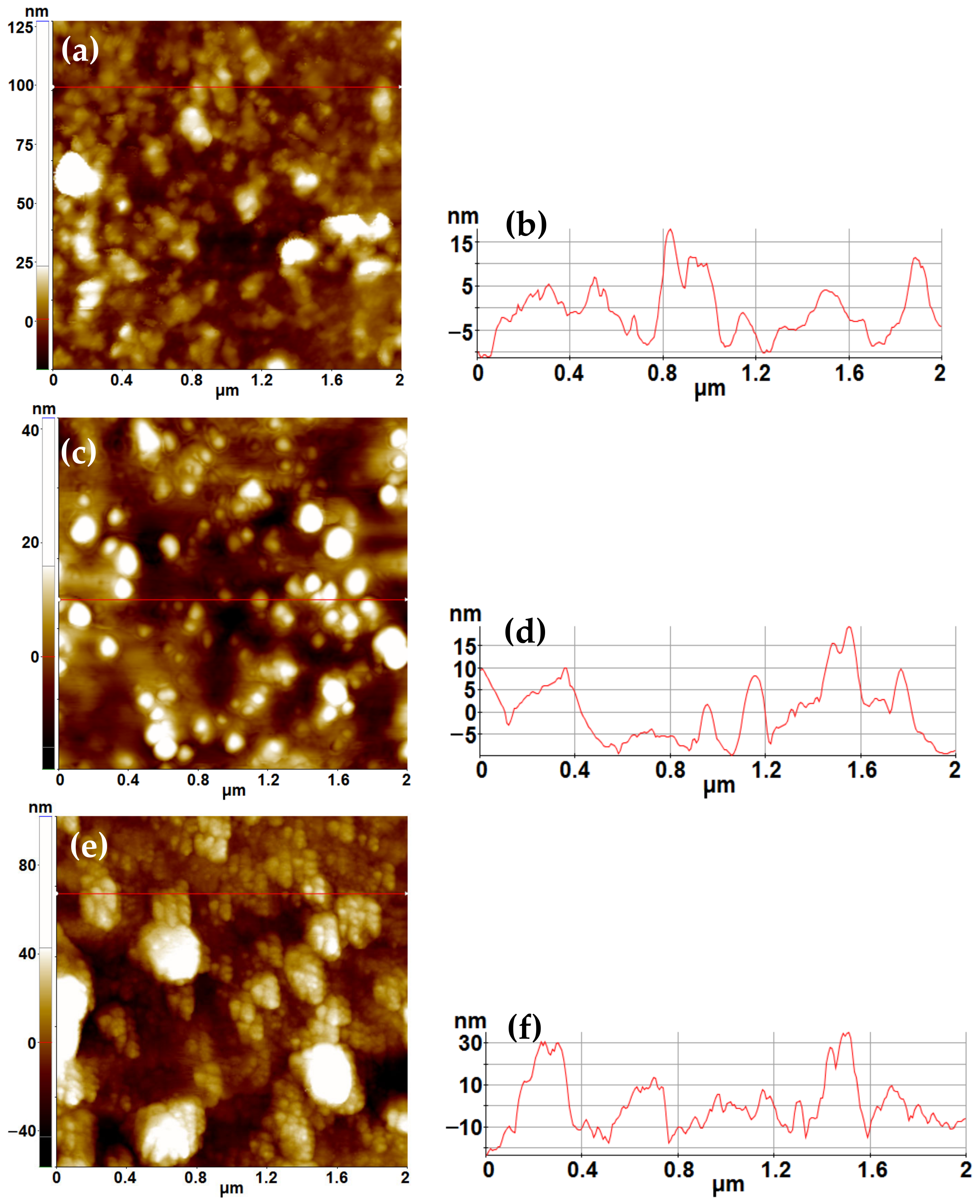
2.2. AFM Characterization
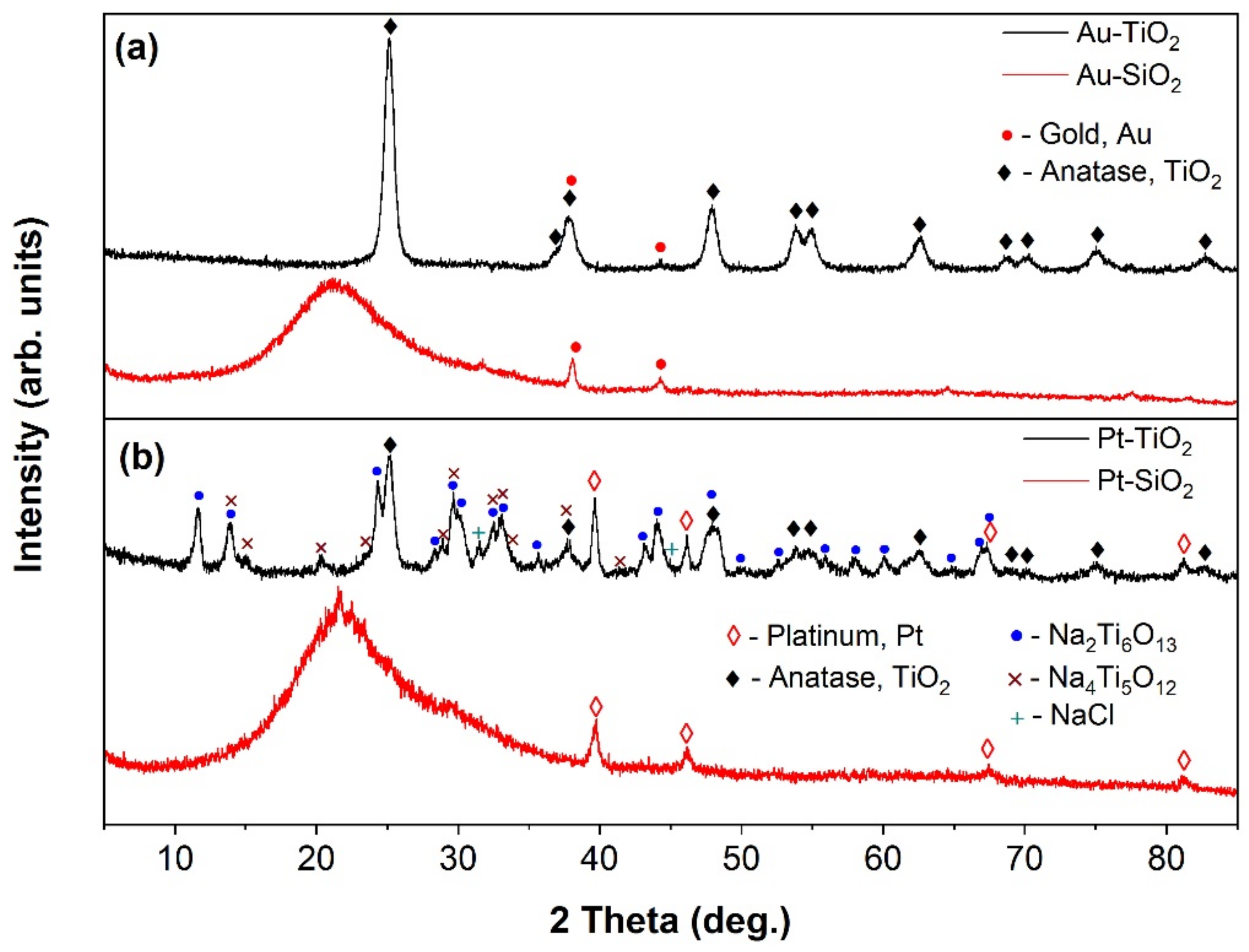
2.3. XRD and XRF Characterization
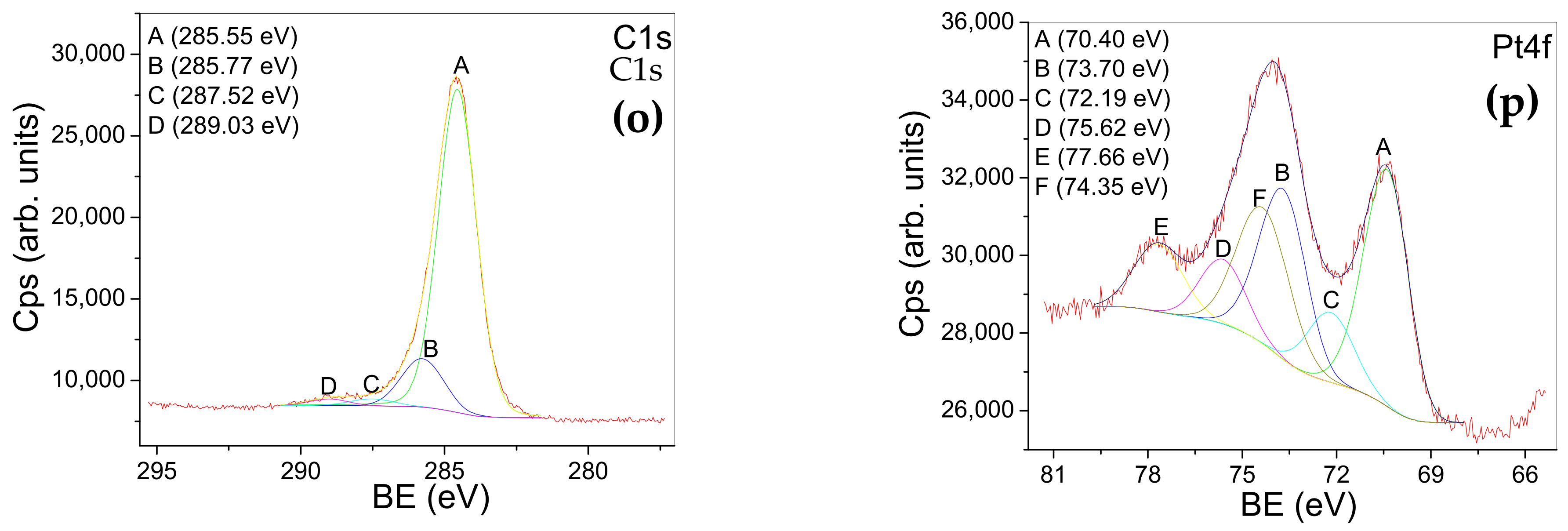
2.4. X-ray Photoelectron Spectroscopy
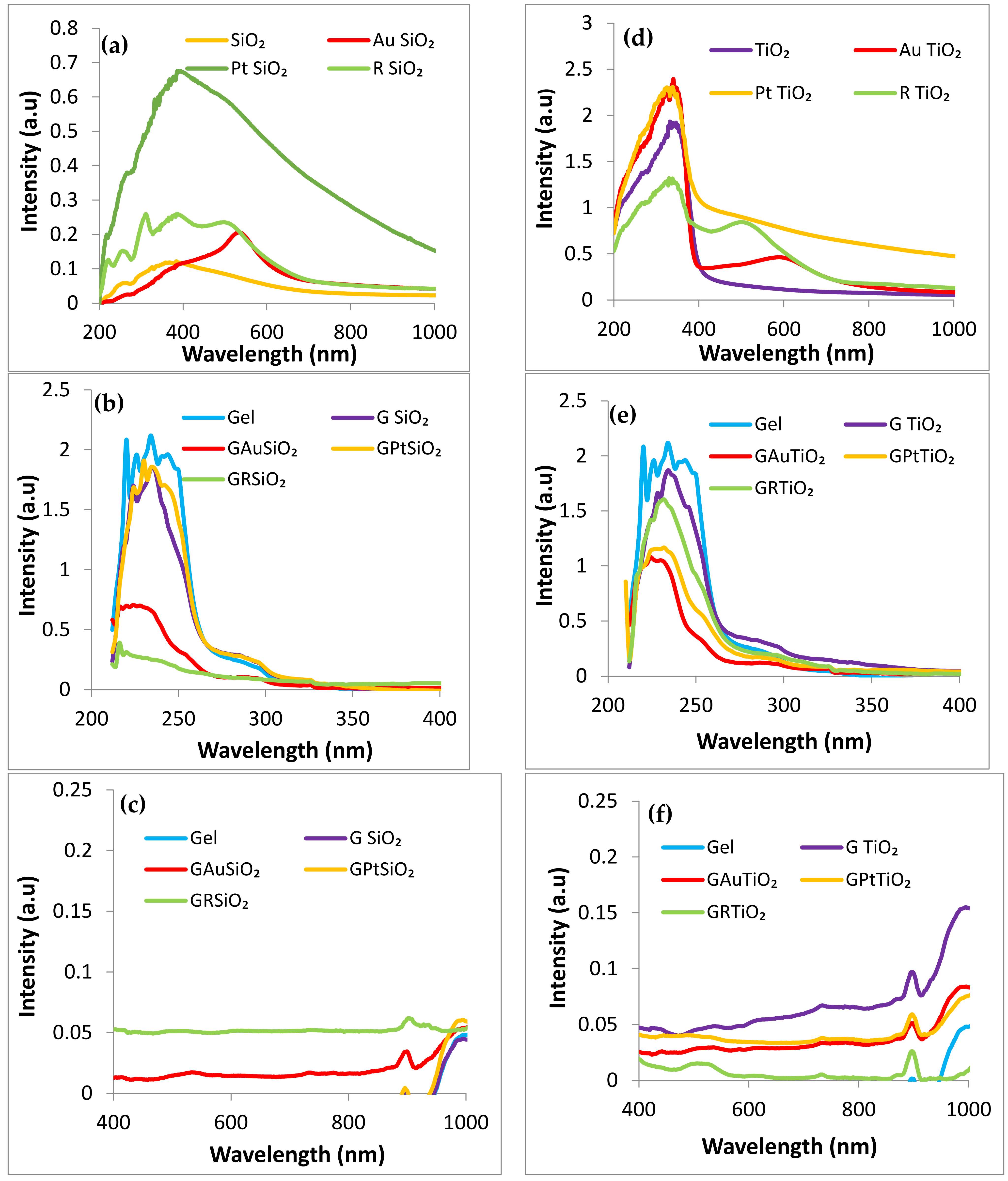
2.5. UV–Vis Characterization
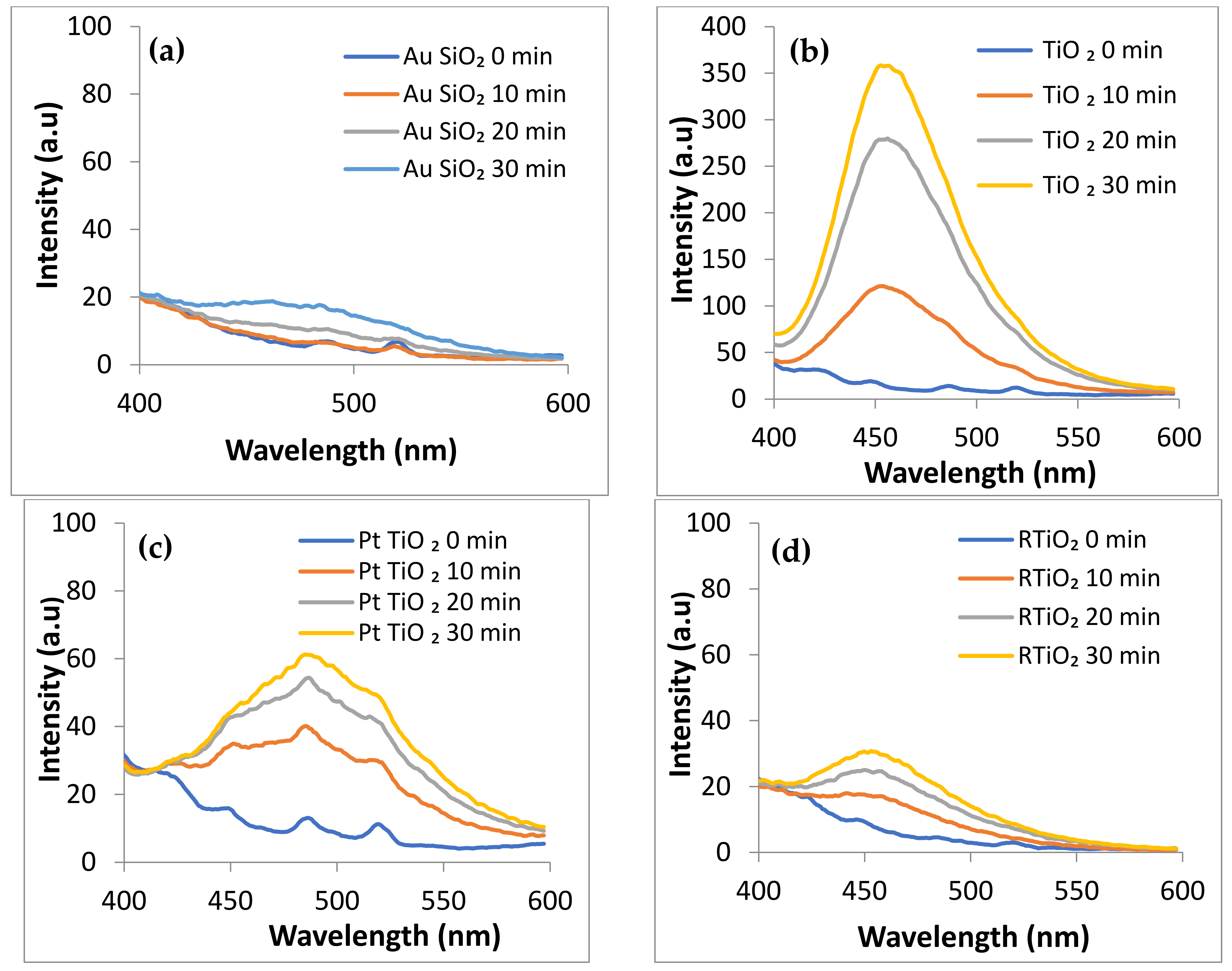
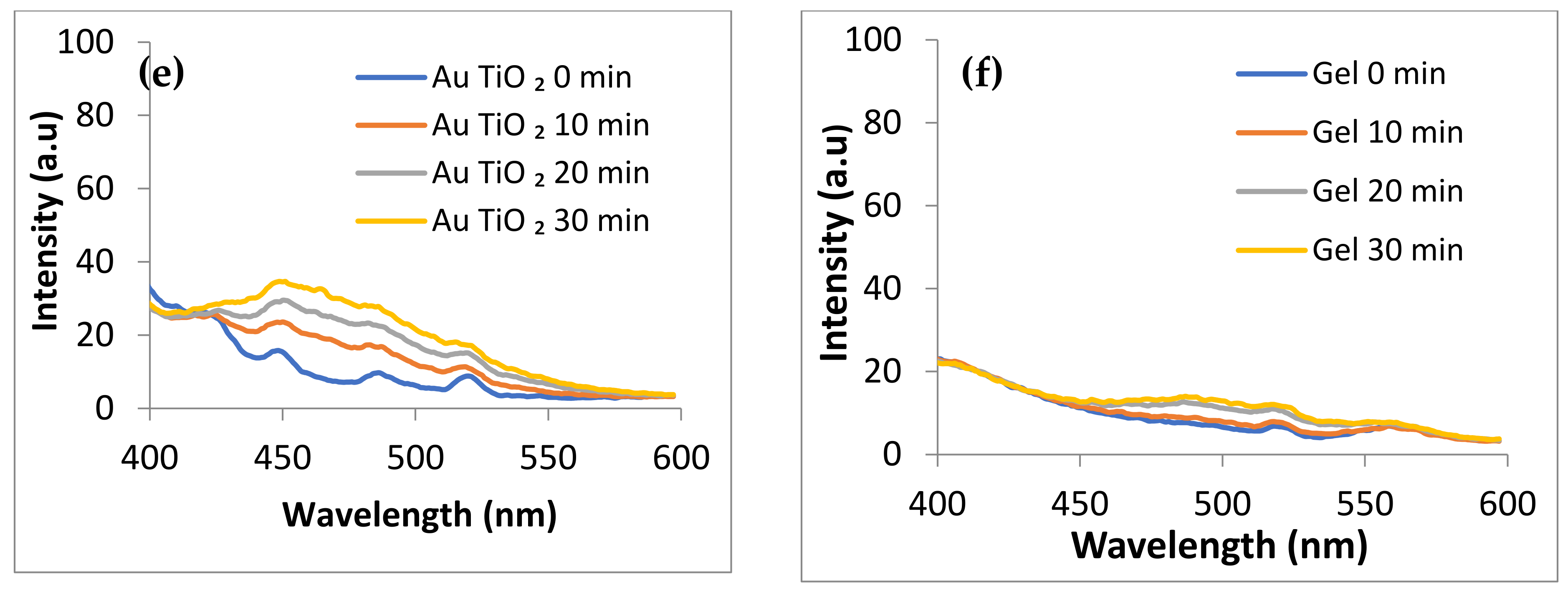
2.6. ROS Photogeneration
- i.
- Generation of hydroxyl radicals (•OH) under light irradiation
- ii.
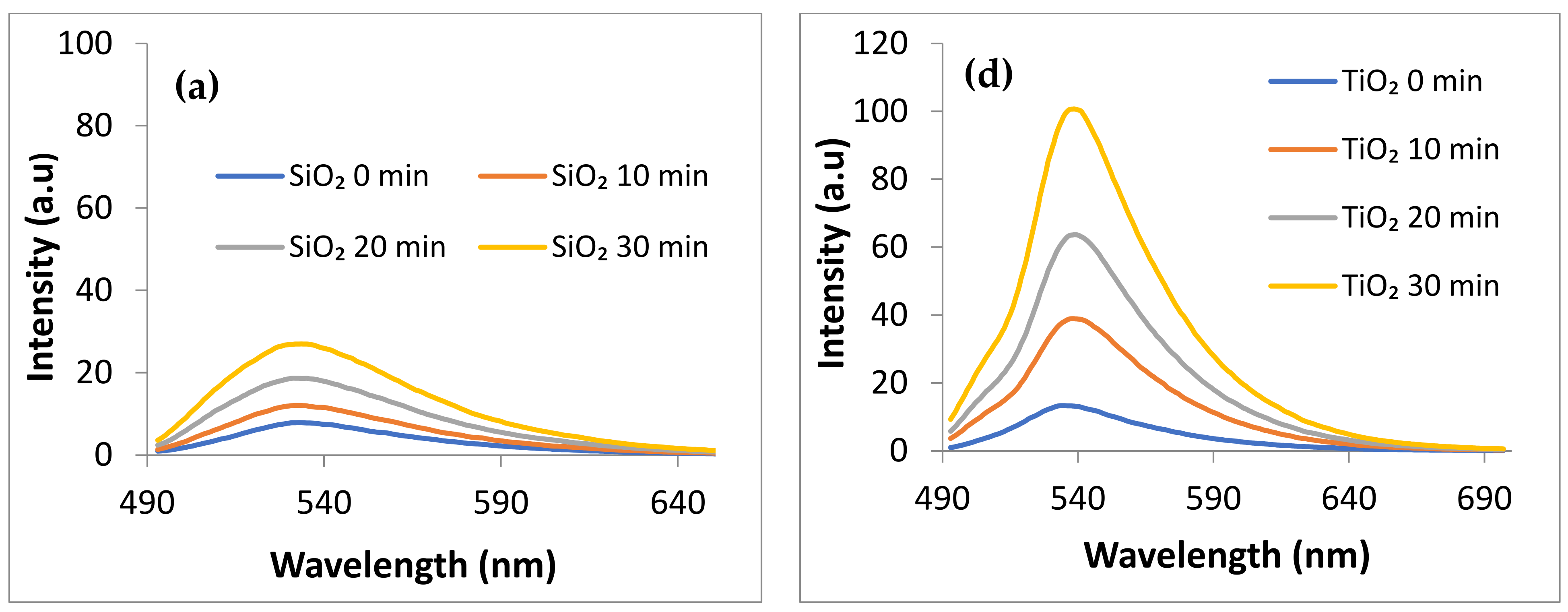
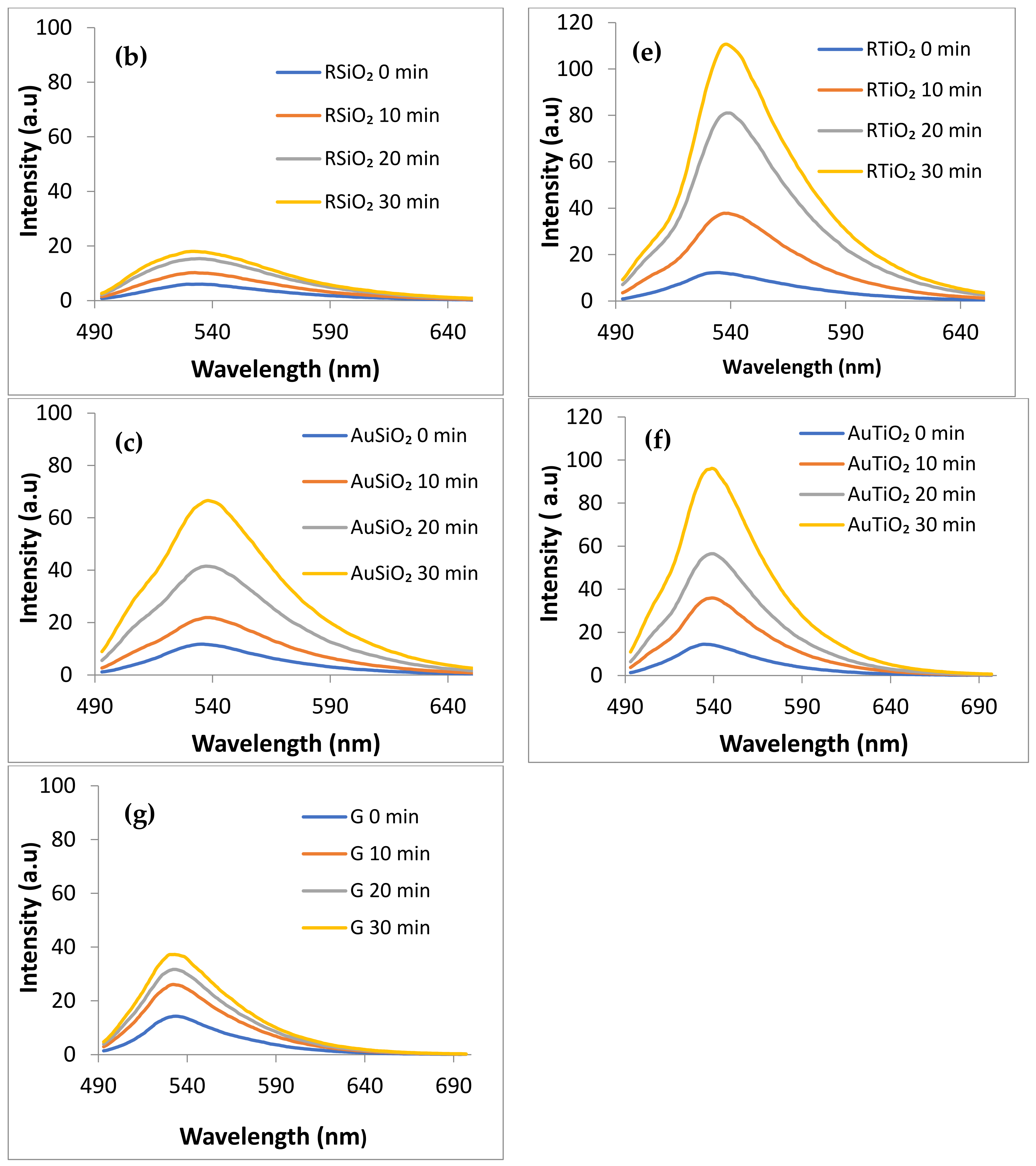
- Generation of Singlet Oxygen (1O2) under visible irradiation (λ > 420 nm) monitored by using Singlet Oxygen Sensor (SOSG) λexc = 480 nm
2.7. Electrokinetic Potential Measurements
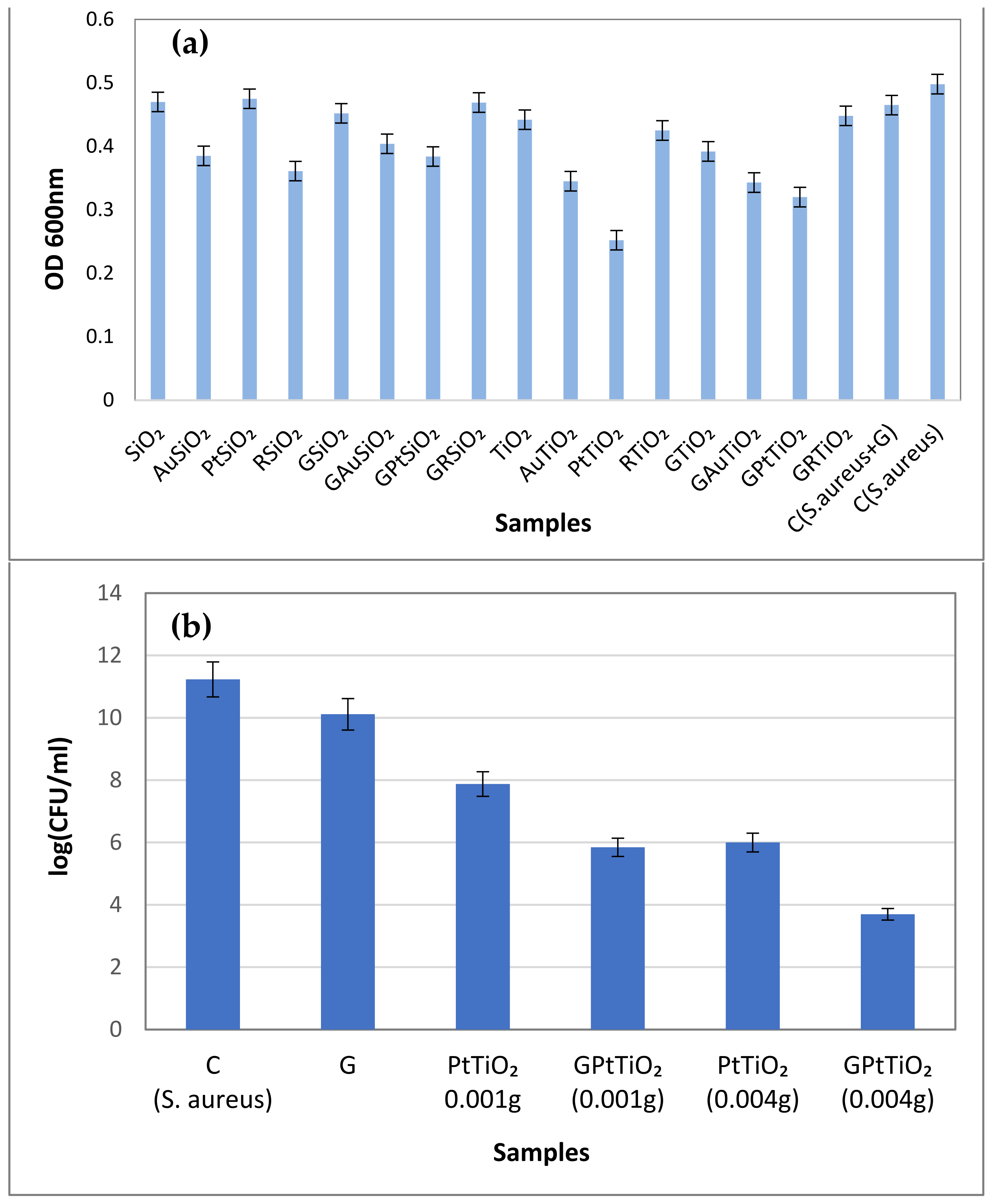
2.8. Antibacterial Activity Assays of the Investigated Samples against S. aureus
2.8.1. Antibacterial Activity Assays in Dark
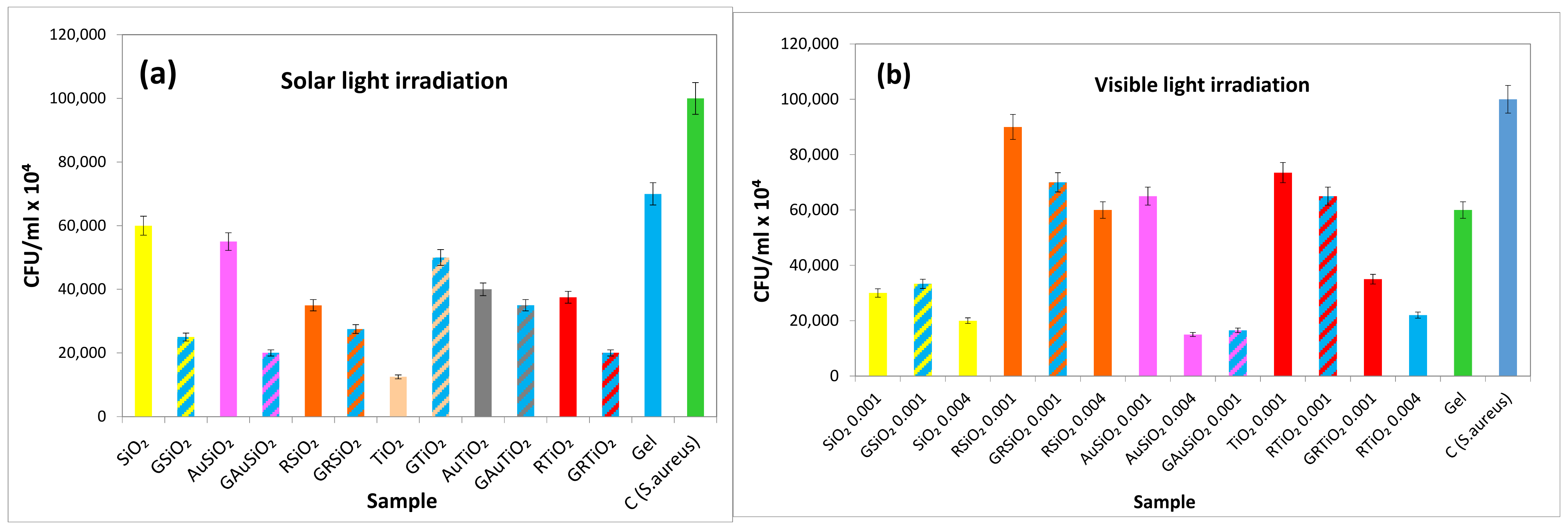
2.8.2. Antibacterial Activity Assays under Light Exposure
3. Conclusions
4. Materials and Methods
4.1. Synthesis of Materials
4.1.1. Synthesis of SiO2 and TiO2 Matrices
4.1.2. Glutathione Capped Gold Nanoparticles
4.1.3. Glutathione Capped Platinum Nanoparticles
4.1.4. Modification of SiO2 and TiO2 with Ruthenium Dye
4.1.5. PVA Hydrogel Synthesis
4.2. Characterization of Materials
4.2.1. Electron Microscopy Characterization
4.2.2. AFM Characterization
4.2.3. XRD and XRF Measurements
4.2.4. XPS Characterization
4.2.5. Diffuse Reflectance UV–Vis Characterization
4.2.6. Monitoring of ROS Photogeneration
- (i)
- Singlet oxygen (1O2) formation under visible light exposure
- (ii)
- Hydroxyl radicals (•OH) generation under simulated solar light
4.2.7. Electrokinetic Potential Measurements
4.2.8. Antibacterial Activity
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rao, L.; Meng, Q.-F.; Huang, Q.; Liu, P.; Bu, L.-L.; Kondamareddy, K.K.; Guo, S.-S.; Liu, W.; Zhao, X.-Z. Photocatalytic Degradation of Cell Membrane Coatings for Controlled Drug Release. Adv. Healthc. Mater. 2016, 5, 1420–1427. [Google Scholar] [CrossRef]
- Kumar, A.; Park, G.D.; Patel, S.K.S.; Kondaveeti, S.; Otari, S.; Anwar, M.Z.; Kalia, V.C.; Singh, Y.; Kim, S.C.; Cho, B.-K.; et al. SiO2 Microparticles with Carbon Nanotube-Derived Mesopores as an Efficient Support for Enzyme Immobilization. Chem. Eng. J. 2019, 359, 1252–1264. [Google Scholar] [CrossRef]
- Lele, B.S.; Russell, A.J. Enhancing Enzyme Stability Against TiO2-UV Induced Inactivation. Biomacromolecules 2005, 6, 475–482. [Google Scholar] [CrossRef]
- Ma, J.; Zhang, M.; Su, W.; Wu, B.; Yang, Z.; Wang, X.; Qiao, B.; Pei, H.; Tu, J.; Chen, D.; et al. Photoelectrochemical Enzyme Biosensor Based on TiO2 Nanorod/TiO2 Quantum Dot/Polydopamine/Glucose Oxidase Composites with Strong Visible-Light Response. Langmuir 2022, 38, 751–761. [Google Scholar] [CrossRef]
- Wang, Z.; Yang, H.; Wang, M.; Petti, L.; Jiang, T.; Jia, Z.; Xie, S.; Zhou, J. SERS-Based Multiplex Immunoassay of Tumor Markers Using Double SiO2@Ag Immune Probes and Gold-Film Hemisphere Array Immune Substrate. Colloids Surf. Physicochem. Eng. Asp. 2018, 546, 48–58. [Google Scholar] [CrossRef]
- Zhang, L.; Ning, C.; Zhou, T.; Liu, X.; Yeung, K.W.K.; Zhang, T.; Xu, Z.; Wang, X.; Wu, S.; Chu, P.K. Polymeric Nanoarchitectures on Ti-Based Implants for Antibacterial Applications. ACS Appl. Mater. Interfaces 2014, 6, 17323–17345. [Google Scholar] [CrossRef]
- Vandamme, K.; Thevissen, K.; Mesquita, M.F.; Coropciuc, R.-G.; Agbaje, J.; Thevissen, P.; da Silva, W.J.; Vleugels, J.; De Cremer, K.; Gerits, E.; et al. Implant Functionalization with Mesoporous Silica: A Promising Antibacterial Strategy, but Does Such an Implant Osseointegrate? Clin. Exp. Dent. Res. 2021, 7, 502–511. [Google Scholar] [CrossRef] [PubMed]
- Alexandre, N.; Ribeiro, J.; Gärtner, A.; Pereira, T.; Amorim, I.; Fragoso, J.; Lopes, A.; Fernandes, J.; Costa, E.; Santos-Silva, A.; et al. Biocompatibility and Hemocompatibility of Polyvinyl Alcohol Hydrogel Used for Vascular Grafting—In Vitro and in Vivo Studies. J. Biomed. Mater. Res. A 2014, 102, 4262–4275. [Google Scholar] [CrossRef]
- Rodríguez-Rodríguez, R.; Espinosa-Andrews, H.; Velasquillo-Martínez, C.; García-Carvajal, Z.Y. Composite Hydrogels Based on Gelatin, Chitosan and Polyvinyl Alcohol to Biomedical Applications: A Review. Int. J. Polym. Mater. Polym. Biomater. 2020, 69, 1–20. [Google Scholar] [CrossRef]
- Kobayashi, M.; Hyu, H.S. Development and Evaluation of Polyvinyl Alcohol-Hydrogels as an Artificial Atrticular Cartilage for Orthopedic Implants. Materials 2010, 3, 2753–2771. [Google Scholar] [CrossRef]
- Hou, Y.; Chen, C.; Liu, K.; Tu, Y.; Zhang, L.; Li, Y. Preparation of PVA Hydrogel with High-Transparence and Investigations of Its Transparent Mechanism. RSC Adv. 2015, 5, 24023–24030. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, W.; Niu, J.; Chen, Y. Mechanism of Photogenerated Reactive Oxygen Species and Correlation with the Antibacterial Properties of Engineered Metal-Oxide Nanoparticles. ACS Nano 2012, 6, 5164–5173. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.F.; Li, J.P.; Qian, K.; Xu, W.P.; Lu, Y.; Huang, W.X.; Yu, S.H. Large Scale Photochemical Synthesis of M@TiO2 Nanocomposites (M = Ag, Pd, Au, Pt) and Their Optical Properties, CO Oxidation Performance, and Antibacterial Effect. Nano Res. 2010, 3, 244–255. [Google Scholar] [CrossRef]
- My-Thao Nguyen, T.; Anh-Thu Nguyen, T.; Tuong-Van Pham, N.; Ly, Q.-V.; Thuy-Quynh Tran, T.; Thach, T.-D.; Nguyen, C.-L.; Banh, K.-S.; Le, V.-D.; Nguyen, L.-P.; et al. Biosynthesis of metallic nanoparticles from waste Passiflora edulis peels for their antibacterial effect and catalytic activity. Arab. J. Chem. 2021, 14, 103096. [Google Scholar] [CrossRef]
- Behzad, F.; Naghib, S.M.; Kouhbanani, M.A.J.; Tabatabaei, S.N.; Zare, Y.; Rhee, K.Y. An overview of the plant-mediated green synthesis of noble metal nanoparticles for antibacterial applications. J. Ind. Eng. Chem. 2021, 94, 92–104. [Google Scholar] [CrossRef]
- Jeyaraj, M.; Gurunathan, S.; Qasim, M.; Kan, M.-H.; Kim, J.-H. A Comprehensive review on the synthesis, characterization, and biomedical application of platinum nanoparticles. Nanomaterials 2019, 9, 1719. [Google Scholar] [CrossRef]
- Ahmed, K.B.A.; Raman, T.; Anbazhagan, V. Platinum nanoparticles inhibit bacteria proliferation and rescue zebrafish from bacterial infection. RSC Adv. 2016, 6, 44415. [Google Scholar] [CrossRef]
- Aygun, A.; Gülbagca, F.; Ozer, L.Y.; Ustaoglu, B.; Altunoglu, Y.C.; Baloglu, M.C.; Atalar, M.N.; Alma, M.H.; Sen, F. Biological synthesis of silver nanoparticles using Rheum ribes and evaluation of their anticarcinogenic and antimicrobial potential: A novel approach in phytonanotechnology. J. Pharm. Biomed. Anal. 2020, 179, 112961. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, S.; Nazam, N.; Mohd, S.; Rizvi, D.; Ahmad, K.; Baig, M.H.; Lee, E.J.; Choi, I. Mechanistic insights into the antimicrobial actions of metallic nanoparticles and their implications for multidrug resistance. Int. J. Mol. Sci. 2019, 20, 2468. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Tatsuma, T. Mechanisms and Applications of Plasmon-Induced Charge Separation at TiO2 Films Loaded with Gold Nanoparticles. J. Am. Chem. Soc. 2005, 127, 7632–7637. [Google Scholar] [CrossRef] [PubMed]
- Buso, D.; Pacifico, J.; Martucci, A.; Mulvaney, P. Gold-Nanoparticle-Doped TiO2 Semiconductor Thin Films: Optical Characterization. Adv. Funct. Mater. 2007, 17, 347–354. [Google Scholar] [CrossRef]
- Vankayala, R.; Sagadevan, A.; Vijayaraghavan, P.; Kuo, C.-L.; Hwang, K.C. Metal Nanoparticles Sensitize the Formation of Singlet Oxygen. Angew. Chem. Int. Ed. 2011, 50, 10640–10644. [Google Scholar] [CrossRef]
- Farooq, S.; de Araujo, R.E. Identifying High Performance Gold Nanoshells for Singlet Oxygen Generation Enhancement. Photodiagnosis Photodyn. Ther. 2021, 35, 102466. [Google Scholar] [CrossRef]
- Hajipour, M.J.; Fromm, K.M.; Akbar Ashkarran, A.; Jimenez de Aberasturi, D.; de Larramendi, I.R.; Rojo, T.; Serpooshan, V.; Parak, W.J.; Mahmoudi, M. Antibacterial Properties of Nanoparticles. Trends Biotechnol. 2012, 30, 499–511. [Google Scholar] [CrossRef]
- Joe, A.; Park, S.-H.; Shim, K.-D.; Kim, D.-J.; Jhee, K.-H.; Lee, H.-W.; Heo, C.-H.; Kim, H.-M.; Jang, E.-S. Antibacterial Mechanism of ZnO Nanoparticles under Dark Conditions. J. Ind. Eng. Chem. 2017, 45, 430–439. [Google Scholar] [CrossRef]
- Chwalibog, A.; Sawosz, E.; Hotowy, A.; Szeliga, J.; Mitura, S.; Mitura, K.; Grodzik, M.; Orlowski, P.; Sokolowska, A. Visualization of Interaction between Inorganic Nanoparticles and Bacteria or Fungi. Int. J. Nanomedicine 2010, 5, 1085–1094. [Google Scholar] [CrossRef] [PubMed]
- Maisch, T.; Baier, J.; Franz, B.; Maier, M.; Landthaler, M.; Szeimies, R.-M.; Bäumler, W. The Role of Singlet Oxygen and Oxygen Concentration in Photodynamic Inactivation of Bacteria. Proc. Natl. Acad. Sci. USA 2007, 104, 7223–7228. [Google Scholar] [CrossRef] [PubMed]
- Socoteanu, R.; Boscencu, R.; Manda, G.; Anastasescu, M.; Neagoe, I.V.; Machado, I.F.; Dinca, O.; Bucur, A.; Ferreira, L.F.V. Morphologic evaluation of some promising A3B porphyrinic type compounds designed for theranostic applications in cancer. Chem. Phys. 2021, 544, 111115. [Google Scholar] [CrossRef]
- De Baróid, Á.T.; McCoy, C.P.; Craig, R.A.; Carson, L.; Andrews, G.P.; Jones, D.S.; Gorman, S.P. Optimization of Singlet Oxygen Production from Photosensitizer-Incorporated, Medically Relevant Hydrogels. J. Biomed. Mater. Res. B Appl. Biomater. 2017, 105, 320–326. [Google Scholar] [CrossRef]
- Vázquez-Ortega, F.; Lagunes, I.; Trigos, Á. Cosmetic Dyes as Potential Photosensitizers of Singlet Oxygen Generation. Dyes Pigments 2020, 176, 108248. [Google Scholar] [CrossRef]
- Bokare, A.D.; Choi, W. Singlet-Oxygen Generation in Alkaline Periodate Solution. Environ. Sci. Technol. 2015, 49, 14392–14400. [Google Scholar] [CrossRef]
- Wang, C.; Makvandi, P.; Zare, E.N.; Tay, F.R.; Niu, L. Advances in Antimicrobial Organic and Inorganic Nanocompounds in Biomedicine. Adv. Ther. 2020, 3, 2000024. [Google Scholar] [CrossRef]
- Anastasescu, C.; Zaharescu, M.; Balint, I. Unexpected Photocatalytic Activity of Simple and Platinum Modified Tubular SiO2 for the Oxidation of Oxalic Acid to CO2. Catal. Lett. 2009, 132, 81–86. [Google Scholar] [CrossRef]
- Anastasescu, C.; Zaharescu, M.; Angelescu, D.; Munteanu, C.; Bratan, V.; Spataru, T.; Negrila, C.; Spataru, N.; Balint, I. Defect-Related Light Absorption, Photoluminiscence and Photocatalytic Activity of SiO2 with Tubular Morphology. Sol. Energy Mater. Sol. Cells 2017, 159, 325–335. [Google Scholar] [CrossRef]
- Anastasescu, C.; Negrila, C.; Angelescu, D.G.; Atkinson, I.; Anastasescu, M.; Spataru, N.; Zaharescu, M.; Balint, I. Particularities of Photocatalysis and Formation of Reactive Oxygen Species on Insulators and Semiconductors: Cases of SiO2, TiO2 and Their Composite SiO2–TiO2. Catal. Sci. Technol. 2018, 8, 5657–5668. [Google Scholar] [CrossRef]
- Du, J.; Gebicki, J.M. Proteins are Major Initial Cell Targets of Hydroxyl Free Radicals. Int. J. Biochem. Cell Biol. 2004, 36, 2334–2343. [Google Scholar] [CrossRef] [PubMed]
- Chifor, E.; Bordeianu, I.; Anastasescu, C.; Calderon-Moreno, J.M.; Bratan, V.; Eftemie, D.-I.; Anastasescu, M.; Preda, S.; Plavan, G.; Pelinescu, D.; et al. Bioactive coatings based on nanostructured TiO2 modified with noble metal nanoparticles and lysozyme for Ti dental implants. Nanomaterials 2022, 12, 3186. [Google Scholar] [CrossRef]
- Rajendran, S.; Santhana Prabha, S.; Joseph Rathish, R.; Singh, G.; Al-Hashem, A. Antibacterial Activity of Platinum Nanoparticles. In Nanotoxicity: Prevention and Antibacterial Applications of Nanomaterials; Elsevier: Amsterdam, The Netherlands, 2020; pp. 275–281. ISBN 978-0-12-819943-5. [Google Scholar]
- Vimalraj, S.; Ashokkumar, T.; Saravanan, S. Biogenic gold nanoparticles synthesis mediated by Mangifera indica seed aqueous extracts exhibits antibacterial, anticancer and anti-angiogenic properties. Biomed. Pharmacother. 2018, 105, 440–448. [Google Scholar] [CrossRef]
- Loiseau, A.; Asila, V.; Boitel-Aullen, G.; Lam, M.; Salmain, M.; Boujday, S. Silver-based plasmonic nanoparticles for and their use in biosensing. Biosensors 2019, 9, 78. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Umar, K.; Mohamad Ibrahim, M.N. Silver nanoparticles: Various methods of synthesis, size affecting factors and their potential applications—A review. Appl. Nanosci. 2020, 10, 1369–1378. [Google Scholar] [CrossRef]
- Zhang, L.; Mazouzi, Y.; Salmain, M.; Liedberg, B.; Boujday, S. Antibody-gold nanoparticle bioconjugates for biosensors: Synthesis, characterization and selected applications. Biosens. Bioelectr. 2020, 165, 112370. [Google Scholar] [CrossRef] [PubMed]
- Saravanakumar, K.; Anand Mariadoss, A.V.; Sathiyaseelan, A.; Wang, M.-H. Synthesis and characterization of nano-chitosan capped gold nanoparticles with multifunctional bioactive properties. Int. J. Biol. Macromolec. 2020, 165, 747–757. [Google Scholar] [CrossRef]
- Tian, B.; Liu, Y. Antibacterial applications and safety issues of silica-based materials: A review. Int. J. Appl. Ceram. Technol. 2021, 18, 289–301. [Google Scholar] [CrossRef]
- Fonseca, S.; Cayer, M.-P.; Tanvir Ahmmed, K.M.; Khadem-Mohtaram, N.; Charette, S.J.; Brouard, D. Characterization of the antibacterial activity of an SiO2 nanoparticular coating to prevent bacterial contamination in blood products. Antibiotics 2022, 11, 107. [Google Scholar] [CrossRef] [PubMed]
- Sapkota, K.; Chaudhary, P.; Han, S.S. Environmentally sustainable route to SiO2@Au–Ag nanocomposites for biomedical and catalytic applications. RSC Adv. 2018, 8, 31311. [Google Scholar] [CrossRef] [PubMed]
- Gankhuyag, S.; Bae, D.S.; Lee, K.; Lee, S. One-pot synthesis of SiO2@Ag mesoporous nanoparticle coating for inhibition of Escherichia coli bacteria on various surfaces. Nanomaterials 2021, 11, 549. [Google Scholar] [CrossRef]
- Kumar, S.; Jain, A.; Panwar, S.; Sharma, I.; Jeon, H.C.; Kang, T.W.; Choubey, R.K. Effect of silica on the ZnS nanoparticles for stable and sustainable antibacterial application. Int. J. Appl. Ceram. Technol. 2019, 16, 531–540. [Google Scholar] [CrossRef]
- Hou, Y.-X.; Abdullah, H.; Kuo, D.-H.; Leu, S.-J.; Gultom, N.S.; Su, C.-H. A comparison study of SiO2/nano metal oxide composite sphere for antibacterial application. Compos. B Eng. 2018, 133, 166–176. [Google Scholar] [CrossRef]
- Anastasescu, C.; Spataru, N.; Culita, D.; Atkinson, I.; Spataru, T.; Bratan, V.; Munteanu, C.; Anastasescu, M.; Negrila, C.; Balint, I. Chemically assembled light harvesting CuOx-TiO2 p–n heterostructures. Chem. Eng. J. 2015, 281, 303–311. [Google Scholar] [CrossRef]
- Wu, Z.; Chen, J.; Jin, R. One-pot synthesis of Au 25 (SG) 18 2- and 4-nm gold nanoparticles and comparison of their size-dependent properties. Adv. Funct. Mater. 2011, 21, 177–183. [Google Scholar] [CrossRef]
- Eklund, S.E.; Cliffel, D.E. Synthesis and catalytic properties of soluble platinum nanoparticles protected by a thiol monolayer. Langmuir 2004, 20, 6012–6018. [Google Scholar] [CrossRef] [PubMed]
- Pibiri, I.; Buscemi, S.; Piccionello, A.P.; Pace, A. Photochemically produced singlet oxygen: Applications and perspectives. ChemPhotoChem 2018, 2, 535–547. [Google Scholar] [CrossRef]
- Ràgas, X.; Jiménez-Garcia, A.; Batlori, X.; Nonell, S. Singlet oxygen photosensitisation by the fluorescent probe Singlet Oxygen Sensor Green. Chem. Commun. 2009, 20, 2920–2922. [Google Scholar] [CrossRef] [PubMed]
- Hirano, K.; Kobayashi, T. Coumarin fluorometry to quantitatively detectable OH radicals in ultrasound aqueous medium. Ultrason. Sonochem. 2016, 30, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Leandri, V.; Gardner, J.M.; Jonsson, M. Coumarin as a quantitative probe for hydroxyl radical formation in heterogeneous photocatalysis. Phys. Chem. C 2019, 123, 6667–6674. [Google Scholar] [CrossRef]




| Sample | Si2p/Ti2p | O1s | C1s | Au4f/Pt4f |
|---|---|---|---|---|
| AuSiO2 | Peak A 103 eV-assigned to Si4+state (SiO2) (Figure 5a) | Peak A-531.60 eV -assigned to: bonds with carbon, adsorbed oxygen, and sodium carbonates Peak B-533.04 eV -assigned to oxygen in SiO2 Peak C-535.46 eV -assigned to Na Auger (Figure 5b) | Peak A-284.77 eV -assigned to: C-C and C-H bonds Peak B-286.26 eV -assigned to C-O bonds PeakC-287.52 eV -assigned to: C=O and O-C-O bonds Peak D 288.99 eV -assigned to: O-C=O (COOH, COOR) bonds (Figure 5c) | Au4f -assigned to a small amount of Au on the surface of the sample (Figure 5d) |
| PtSiO2 | Peak A 103 eV -assigned to Si4+state (SiO2) (Figure 5e) | Peak A 531.49 eV -assigned to: metal hydroxide (NaOH), O=C bonds, molecularly adsorbed oxygen PeakB 532.98 eV -assigned to oxygen in SiO2 bonds with carbon Peak C 536.40 eV -assigned to Na Auger (Figure 5f) | Peak A 285.00 eV -assigned to: C-C and C-H bonds PeakC- 286.27 eV PeakC- 288.69 eV -assigned to: C-O and O-C=O bonds (Figure 5g) | A-B doublet (71.3;74.61eV) -assigned to metallic Pt PeakC- 75.12 eV -assigned to the loss from Na 2s (Figure 5h) |
| AuTiO2 | Peak A 464.35 eV Peak A 458.59 eV (2p3/2;2p1/2) -assigned to Ti in TiO2 (Figure 5i) | Peak A-529.90 eV -assigned to oxygen chemical states bonded in TiO2 Peak B-531.53 eV -assigned to: O=C (adsorbed) bonds, the presence of metal hydroxide (NaOH) Peak C-532.64 eV -assigned to O-C bonds Peak D-535.20 eV -assigned to Na Auger transition (Figure 5j) | Peak A 284.84 eV -assigned to: C-C and C-H bonds Peak B 286.16 eV -assigned to C-O bonds Peak C 288.42 eV -assigned to O-C=O (COOH and COOR) bonds (Figure 5k) | Au4f Peak A 83.03 eV Peak B 88.66 eV (4f7/2; 4f5/2) -the binding energy is lower than that of ordinary metal, suggesting that Au receives electrons. (Figure 5l) |
| PtTiO2 | Peak A 464.46 eV Peak A 458.69 eV (2p3/2;2p1/2) -assigned to Ti in TiO2 (Figure 5m) | Peak A 530.17 eV -assigned to oxygen chemical states bonded in TiO2 Peak B 531.17 eV -assigned to: O=C bonds; metal hydroxide (NaOH) Peak C 532.31 eV -assigned to O-C bonds Peak D 534.51 eV -assigned to Na Auger transition (Figure 5n) | Peak A 285 eV -assigned to: C-C and C-H bonds Peak B 285.77 eV -assigned to C-O bonds Peak C 287.52 eV Peak D 289.03 eV -assigned to: O-C-O, O-C=O (COOH and COOR) bonds (Figure 5o) | A-B doublet (70.4; 73.7 eV) -assigned to metallic Pt (suggesting an induced negativity) C-D doublet (72.1, 75.6 eV) -assigned to Pt(OH)2 or a Pt sub-oxide F-E doublet (74.3; 77.6 eV) -assigned to PtO and PtO2. (Figure 5p) |
| Powder | SiO2 | AuSiO2 | PtSiO2 | * RSiO2 | TiO2 | AuTiO2 | PtTiO2 | * RTiO2 |
|---|---|---|---|---|---|---|---|---|
| PVA gel containing oxide powders | GSiO2 | GAuSiO2 | GPtSiO2 | GRSiO2 | GTiO2 | GAuTiO2 | GPtTiO2 | GRTiO2 |
| PVA gel | Gel (G) | |||||||
| Powder Samples | Electrokinetic Potential (mV) | Gel Embedding Powder Samples | Electrokinetic Potential (mV) |
|---|---|---|---|
| Gel | −1.10 | ||
| SiO2 | −14.60 | GSiO2 | −6.00 |
| AuSiO2 | −17.73 | GAuSiO2 | −4.81 |
| PtSiO2 | −60.43 | GPtSiO2 | −18.00 |
| RSiO2 | −17.40 | GRSiO2 | −7.58 |
| TiO2 | −30.00 | GRTiO2 | –0.99 |
| AuTiO2 | −17.63 | GAuTiO2 | −2.07 |
| PtTiO2 | −25.60 | GPtTiO2 | −4.61 |
| RTiO2 | −15.33 | GRTiO2 | −0.99 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pelinescu, D.; Anastasescu, M.; Bratan, V.; Maraloiu, V.-A.; Negrila, C.; Mitrea, D.; Calderon-Moreno, J.; Preda, S.; Gîfu, I.C.; Stan, A.; et al. Antibacterial Activity of PVA Hydrogels Embedding Oxide Nanostructures Sensitized by Noble Metals and Ruthenium Dye. Gels 2023, 9, 650. https://doi.org/10.3390/gels9080650
Pelinescu D, Anastasescu M, Bratan V, Maraloiu V-A, Negrila C, Mitrea D, Calderon-Moreno J, Preda S, Gîfu IC, Stan A, et al. Antibacterial Activity of PVA Hydrogels Embedding Oxide Nanostructures Sensitized by Noble Metals and Ruthenium Dye. Gels. 2023; 9(8):650. https://doi.org/10.3390/gels9080650
Chicago/Turabian StylePelinescu, Diana, Mihai Anastasescu, Veronica Bratan, Valentin-Adrian Maraloiu, Catalin Negrila, Daiana Mitrea, Jose Calderon-Moreno, Silviu Preda, Ioana Catalina Gîfu, Adrian Stan, and et al. 2023. "Antibacterial Activity of PVA Hydrogels Embedding Oxide Nanostructures Sensitized by Noble Metals and Ruthenium Dye" Gels 9, no. 8: 650. https://doi.org/10.3390/gels9080650
APA StylePelinescu, D., Anastasescu, M., Bratan, V., Maraloiu, V.-A., Negrila, C., Mitrea, D., Calderon-Moreno, J., Preda, S., Gîfu, I. C., Stan, A., Ionescu, R., Stoica, I., Anastasescu, C., Zaharescu, M., & Balint, I. (2023). Antibacterial Activity of PVA Hydrogels Embedding Oxide Nanostructures Sensitized by Noble Metals and Ruthenium Dye. Gels, 9(8), 650. https://doi.org/10.3390/gels9080650










