Selenite Removal from Aqueous Solution Using Silica–Iron Oxide Nanocomposite Adsorbents
Abstract
1. Introduction
2. Results and Discussion
2.1. Scanning Electron Microscopy, SEM
2.2. Selenium Adsorption Studies
2.2.1. Kinetic and Thermodynamic Studies
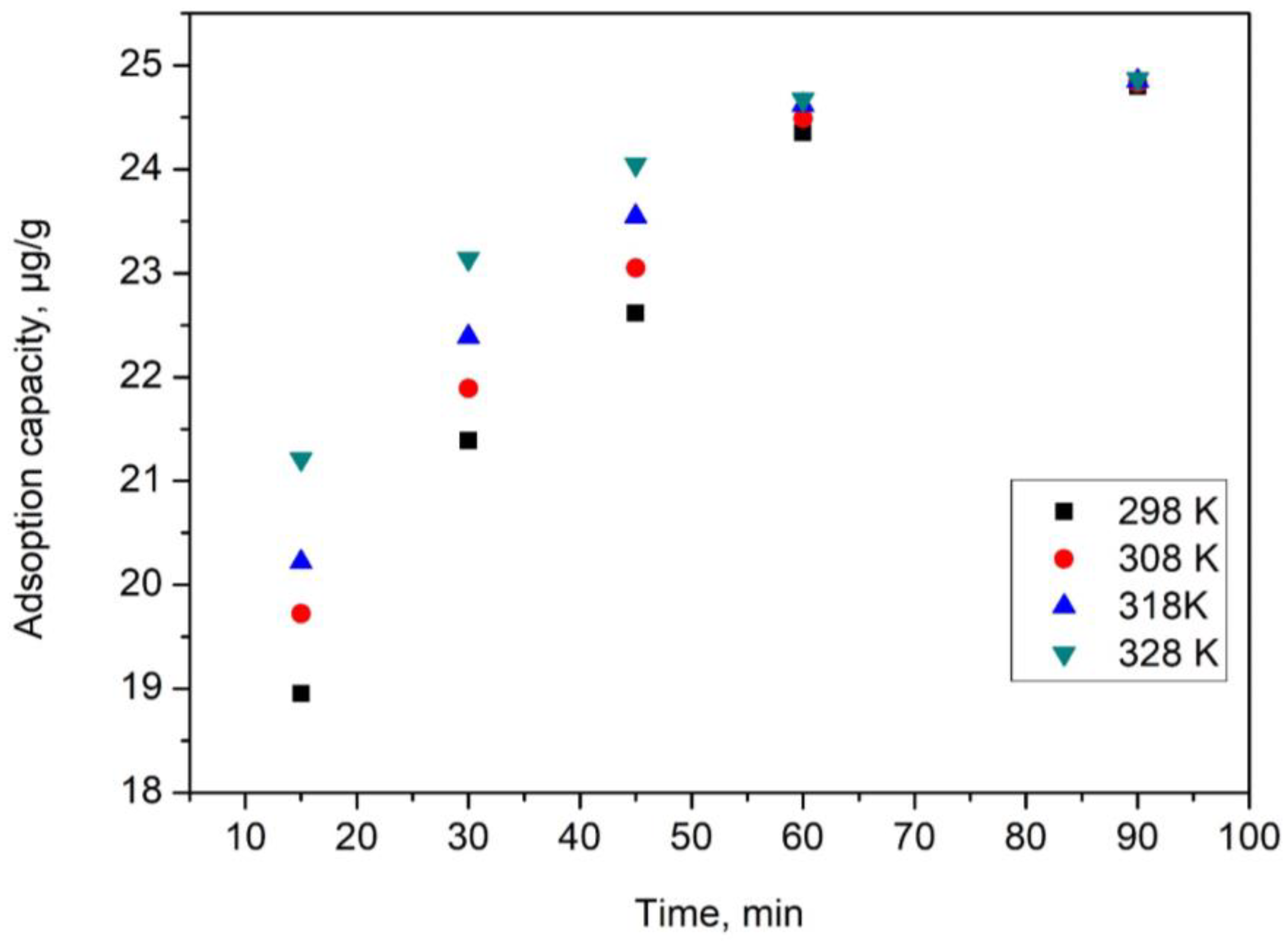
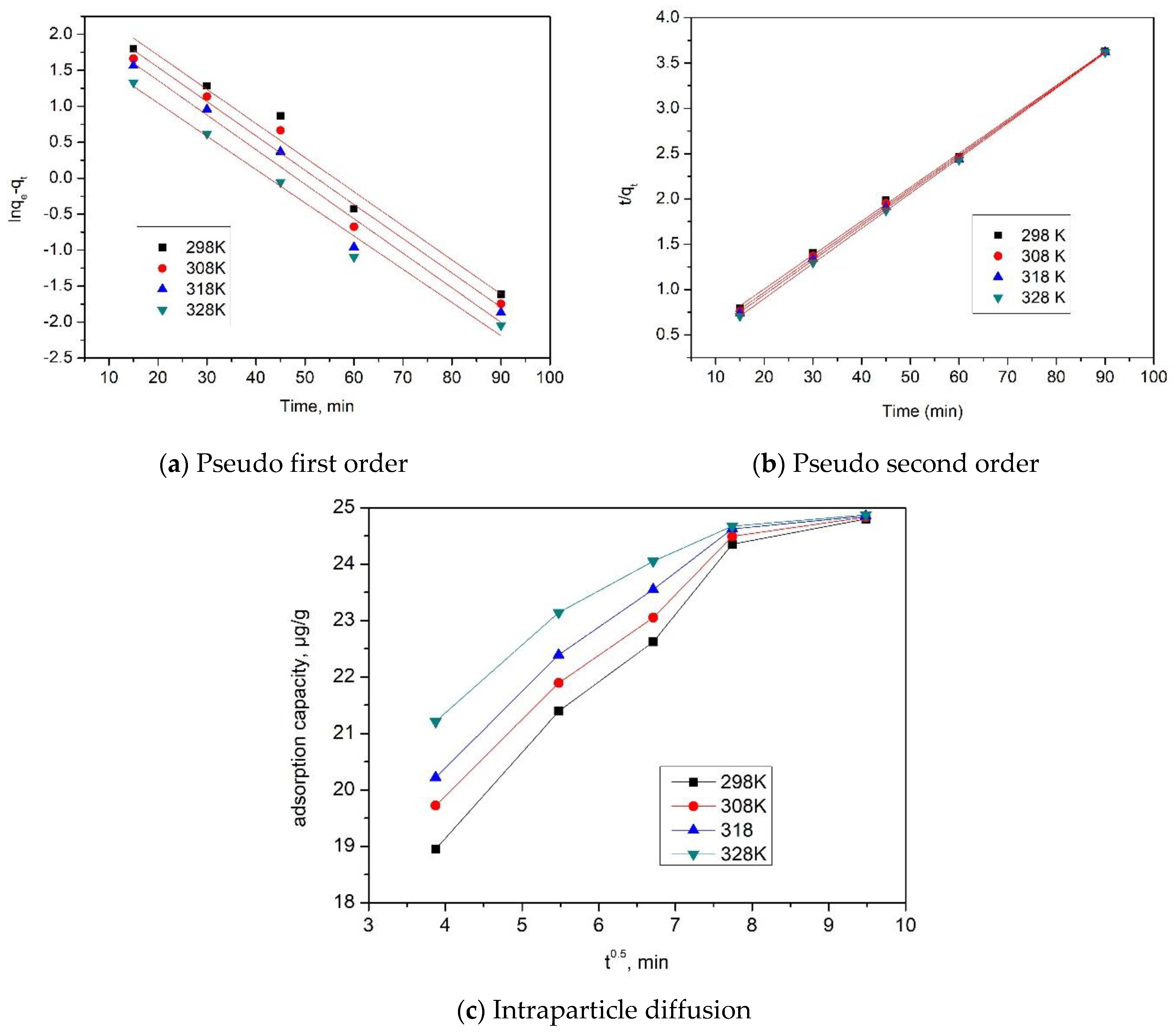
2.2.2. Kinetic Studies
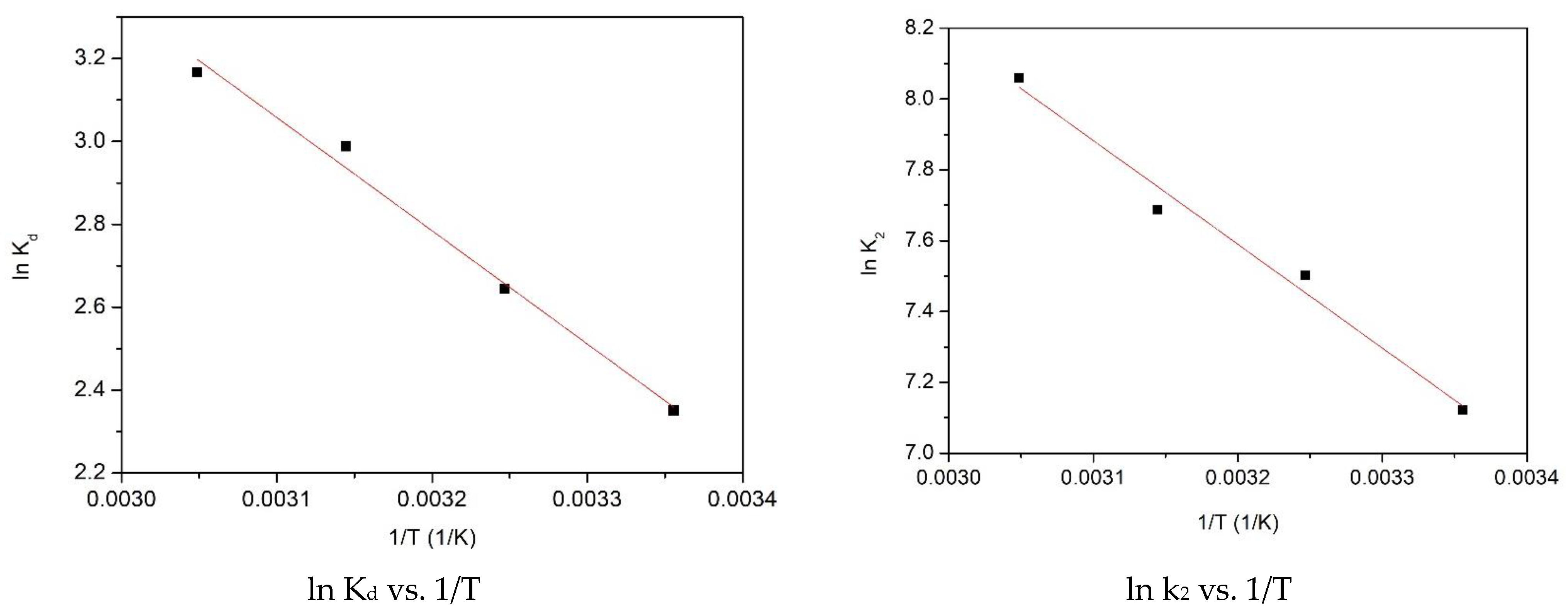
2.2.3. Thermodynamic Studies
2.2.4. Equilibrium Studies
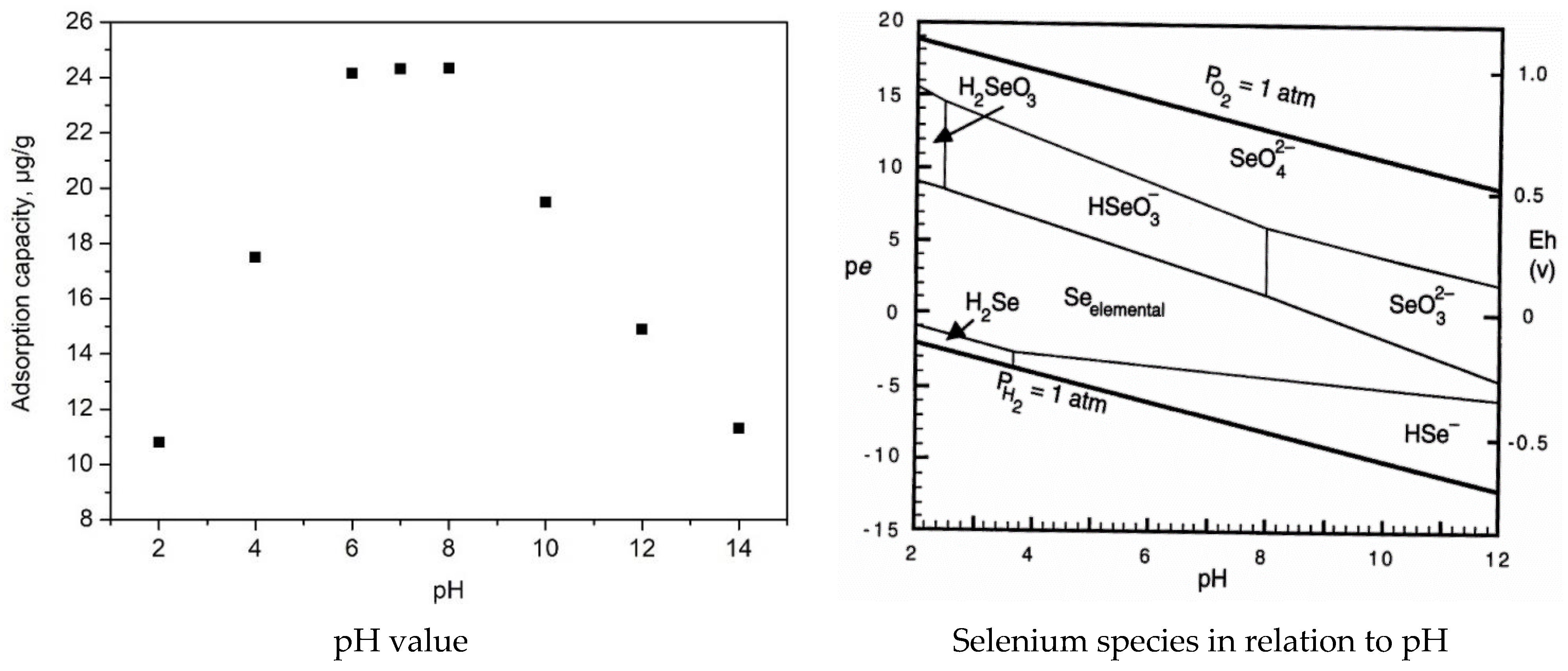
2.2.5. pH Effect
3. Conclusions
4. Materials and Methods
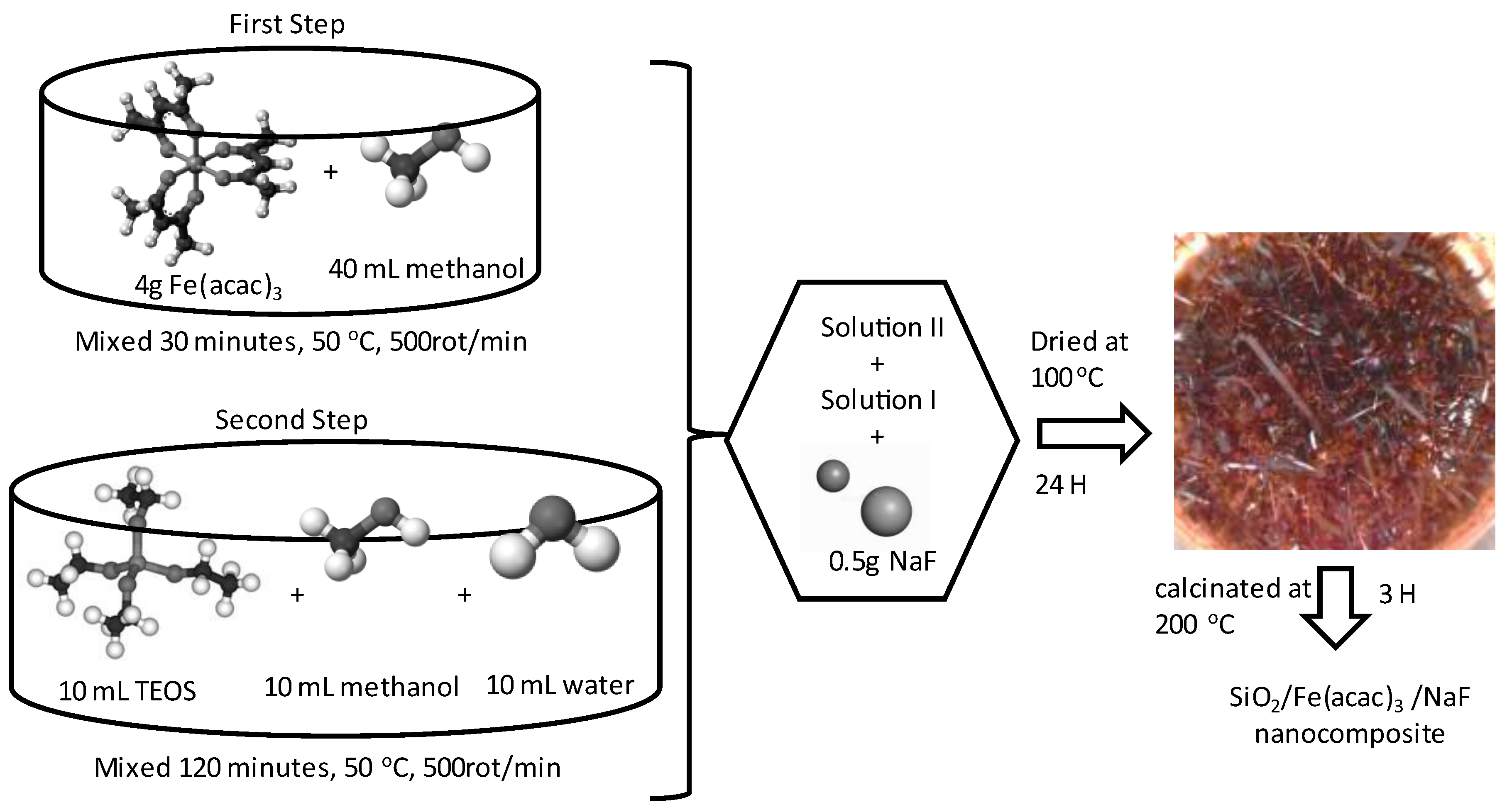
4.1. Synthesis and Characterization of SiO2/Fe(acac)3/NaF Material
4.2. Selenium Adsorption Studies
4.2.1. Kinetic and Thermodynamic Studies
4.2.2. Kinetic Studies
- where qe—equilibrium adsorption capacity, µg g−1;
- qt—adsorption capacity at t time, µg g−1;
- k1—speed constant for pseudo-first order equation, min−1;
- t—contact time, min.
- where qe—equilibrium adsoprtion capacity, µg g−1;
- qt—adsorption capacity at t time, µg g−1;
- k2—speed constant for pseudo-second-order equation, g µg−1 min−1;
- t—contact time, min.
- where qt—adsorption capacity at t time, µg g−1;
- kdiff—speed constant for intraparticle diffusion, µg g−1 min−0.5;
- C—constant correlated with the thickness of the liquid film surrounding the adsorbent particles.
4.2.3. Thermodynamic Studies
- where ΔG0—standard Gibbs free energy variation, J mol−1;
- ΔH0—standard enthalpy variation, J mol−1;
- ΔS0—standard entropy variation, J mol−1 K−1;
- T—absolute temperature, K.
- where Kd—equilibrium constant;
- ΔS0—standard entropy variation, J mol−1 K−1;
- ΔH0—standard enthalpy variation, J mol−1;
- R—the ideal gas constant, 8.314 J mol−1 K−1.
- where k2—speed constant, g min−1 mg−1;
- A—Arrhenius constant, g min mg−1;
- Ea—activation energy, kJ mol−1;
- T—absolute temperature, K;
- R—the ideal gas constant, 8.314 J mol−1 K−1.
4.2.4. Equilibrium Studies
- where qL—Langmuir maximum adsorption capacity, µg g−1;
- KL—Langmuir constant.
- where KS—constant related to the adsorption capacity of the adsorbent;
- nS—heterogeneity factor.
4.2.5. pH Effect
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Zhang, N. Selenite removal using GAC based iron-coated adsorbents. In Civil and Environmental Engineering; West Virginia University: Morgantown, WV, USA, 2008. [Google Scholar]
- Lakin, H.W. Selenium in Our Environment. In Trace Elements in the Environment; American Chemical Society: Washington, DC, USA, 1973; pp. 96–111. [Google Scholar]
- He, Y.; Xiang, Y.; Zhou, Y.; Yang, Y.; Zhang, J.; Huang, H.; Shang, C.; Luo, L.; Gao, J.; Tang, L. Selenium contamination, consequences and remediation techniques in water and soils: A review. Environ. Res. 2018, 164, 288–301. [Google Scholar] [CrossRef] [PubMed]
- Kalaitzidou, K.; Nikoletopoulos, A.A.; Bakouros, L.; Zouboulis, A.; Mitrakas, M. Selenite Removal from Water. Environ. Sci. Proc. 2020, 2, 23. [Google Scholar]
- Pettine, M.; McDonald, T.J.; Sohn, M.; Anquandah, G.A.; Zboril, R.; Sharma, V.K. A critical review of selenium analysis in natural water samples. Trends Environ. Anal. Chem. 2015, 5, 1–7. [Google Scholar] [CrossRef]
- Tabelin, C.B.; Igarashi, T.; Villacorte-Tabelin, M.; Park, I.; Opiso, E.M.; Ito, M.; Hiroyoshi, N. Arsenic, selenium, boron, lead, cadmium, copper, and zinc in naturally contaminated rocks: A review of their sources, modes of enrichment, mechanisms of release, and mitigation strategies. Sci. Total Environ. 2018, 645, 1522–1553. [Google Scholar] [CrossRef] [PubMed]
- Etteieb, S.; Magdouli, S.; Zolfaghari, M.; Brar, S. Monitoring and analysis of selenium as an emerging contaminant in mining industry: A critical review. Sci. Total Environ. 2020, 698, 134339. [Google Scholar] [CrossRef] [PubMed]
- Ali, I.; Shrivastava, V. Recent advances in technologies for removal and recovery of selenium from (waste) water: A systematic review. J. Environ. Manag. 2021, 294, 112926. [Google Scholar] [CrossRef]
- He, Y.; Liu, J.; Han, G.; Chung, T.-S. Novel thin-film composite nanofiltration membranes consisting of a zwitterionic co-polymer for selenium and arsenic removal. J. Membr. Sci. 2018, 555, 299–306. [Google Scholar] [CrossRef]
- Yoon, S.; Cho, K.-H.; Kim, M.; Park, S.-J.; Lee, C.-G.; Choi, N.-C. Selenium Removal from Aqueous Solution Using a Low-Cost Functional Ceramic Membrane Derived from Waste Cast Iron. Water 2023, 15, 312. [Google Scholar] [CrossRef]
- Opiso, E.M.; Sato, T.; Yoneda, T. Immobilization of selenium by Mg-bearing minerals and its implications for selenium removal from contaminated water and wastewater. Appl. Clay Sci. 2016, 123, 121–128. [Google Scholar] [CrossRef]
- Staicu, L.C.; Morin-Crini, N.; Crini, G. Desulfurization: Critical step towards enhanced selenium removal from industrial effluents. Chemosphere 2017, 172, 111–119. [Google Scholar] [CrossRef]
- Kazeem, T.; Labaran, B.; Ahmed, H.-U.-R.; Mohammed, T.; Essa, M.; Al-Suwaiyan, M.; Vohra, M. Treatment of Aqueous Selenocyanate Anions Using Electrocoagulation. Int. J. Electrochem. Sci. 2019, 14, 10538–10564. [Google Scholar] [CrossRef]
- Baek, K.; Kasem, N.; Ciblak, A.; Vesper, D.; Padilla, I.; Alshawabkeh, A.N. Electrochemical removal of selenate from aqueous solutions. Chem. Eng. J. 2013, 215–216, 678–684. [Google Scholar] [CrossRef]
- Vohra, M.S.; Labaran, B.A. Photocatalytic treatment of mixed selenocyanate and phenol streams: Process modeling, optimization, and kinetics. Environ. Prog. Sustain. Energy 2020, 39, e13401. [Google Scholar] [CrossRef]
- Botlaguduru, V.S.V.; Batchelor, B.; Abdel-Wahab, A. Application of UV–sulfite advanced reduction process to bromate removal. Remov. J. Water Process Eng. 2015, 5, 76–82. [Google Scholar] [CrossRef]
- Vellanki, B.P.; Batchelor, B. Perchlorate reduction by the sulfite/ultraviolet light advanced reduction process. J. Hazard. Mater. 2013, 262, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Vellanki, B.P.; Batchelor, B.; Abdel-Wahab, A. Degradation of 1,2-dichloroethane with advanced reduction processes (ARPs): Effects of process variables and mechanisms. Chem. Eng. J. 2014, 237, 300–307. [Google Scholar] [CrossRef]
- Yoon, S.; Han, D.S.; Liu, X.; Batchelor, B.; Abdel-Wahab, A. Degradation of 1,2-dichloroethane using advanced reduction processes. J. Environ. Chem. Eng. 2014, 2, 731–737. [Google Scholar] [CrossRef]
- Zou, R.; Zhang, H.; Luo, G.; Fang, C.; Shi, M.; Hu, H.; Li, X.; Yao, H. Selenium migration behaviors in wet flue gas desulfurization slurry and an in-situ treatment approach. Chem. Eng. J. 2020, 385, 123891. [Google Scholar] [CrossRef]
- Paul, T.; Saha, N.C. Environmental Arsenic and Selenium Contamination and Approaches towards Its Bioremediation through the Exploration of Microbial Adaptations: A Review. Pedosphere 2019, 29, 554–568. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Raza, A.; Hawrylak-Nowak, B.; Matraszek-Gawron, R.; Nahar, K.; Fujita, M. Selenium Toxicity in Plants and Environment: Biogeochemistry and Remediation Possibilities. Plants 2020, 9, 1711. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Wang, J.; Liu, Q.; Zeng, H. Water-dispersible magnetic nanoparticle–graphene oxide composites for selenium removal. Carbon 2014, 77, 710–721. [Google Scholar] [CrossRef]
- Johansson, C.L.; Paul, N.A.; de Nys, R.; Roberts, D.A. The complexity of biosorption treatments for oxyanions in a multi-element mine effluent. J. Environ. Manag. 2015, 151, 386–392. [Google Scholar] [CrossRef]
- Gurunathan, P.; Hari, S.; Suseela, S.B.; Sankararajan, R.; Mukannan, A. Production, characterization and effectiveness of cellulose acetate functionalized ZnO nanocomposite adsorbent for the removal of Se (VI) ions from aqueous media. Environ. Sci. Pollut. Res. 2019, 26, 528–543. [Google Scholar] [CrossRef]
- Zhao, Q.; Huang, J.-C.; He, S.; Zhou, W. Enhancement of a constructed wetland water treatment system for selenium removal. Sci. Total Environ. 2020, 714, 136741. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zhang, A.; Zhang, J.; Wang, Q.; Huang, X.; Wu, Y.; Tang, C. Enhanced Selenate Removal in Aqueous Phase by Copper-Coated Activated Carbon. Materials 2020, 13, 468. [Google Scholar] [CrossRef]
- Kalaitzidou, K.; Nikoletopoulos, A.-A.; Tsiftsakis, N.; Pinakidou, F.; Mitrakas, M. Adsorption of Se(IV) and Se(VI) species by iron oxy-hydroxides: Effect of positive surface charge density. Sci. Total Environ. 2019, 687, 1197–1206. [Google Scholar] [CrossRef]
- Vilardi, G.; Mpouras, T.; Dermatas, D.; Verdone, N.; Polydera, A.; Di Palma, L. Nanomaterials application for heavy metals recovery from polluted water: The combination of nano zero-valent iron and carbon nanotubes. Competitive adsorption non-linear modeling. Chemosphere 2018, 201, 716–729. [Google Scholar] [CrossRef]
- Ji, Y.; Li, L.; Wang, Y.-T. Selenium removal by activated alumina in batch and continuous-flow reactors. Water Environ. Res. 2020, 92, 51–59. [Google Scholar] [CrossRef]
- Toyos-Rodríguez, C.; Calleja-García, J.; Torres-Sánchez, L.; López, A.; Abu-Dief, A.M.; Costa, A.; Elbaile, L.; Crespo, R.D.; Garitaonandia, J.S.; Lastra, E.; et al. A Simple and Reliable Synthesis of Superparamagnetic Magnetite Nanoparticles by Thermal Decomposition of Fe(acac)3. J. Nanomater. 2019, 2019, 2464010. [Google Scholar] [CrossRef]
- Mladin, G.; Ciopec, M.; Negrea, A.; Duteanu, N.; Negrea, P.; Ianasi, P.; Ianasi, C. Silica- Iron Oxide Nanocomposite Enhanced with Porogen Agent Used for Arsenic Removal. Materials 2022, 15, 5366. [Google Scholar] [CrossRef] [PubMed]
- Shafiq, I.; Hussain, M.; Rashid, R.; Shafique, S.; Akhter, P.; Yang, W.; Ahmed, A.; Nawaz, Z.; Park, Y.-K. Development of hierarchically porous LaVO4 for efficient visible-light-driven photocatalytic desulfurization of diesel. Chem. Eng. J. 2021, 420, 130529. [Google Scholar] [CrossRef]
- Zhang, S.; Ning, S.; Liu, H.; Zhou, J.; Wang, S.; Zhang, W.; Wang, X.; Wei, Y. Highly-efficient separation and recovery of ruthenium from electroplating wastewater by a mesoporous silica-polymer based adsorbent. Microporous Mesoporous Mater. 2020, 303, 110293. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, F.; Cheng, W.; Wang, J.; Ma, J. Adsorption Equilibrium and Kinetics of the Removal of Ammoniacal Nitrogen by Zeolite X/Activated Carbon Composite Synthesized from Elutrilithe. J. Chem. 2017, 2017, 1936829. [Google Scholar] [CrossRef]
- Bandara, P.C.; Perez, J.V.D.; Nadres, E.T.; Nannapaneni, R.G.; Krakowiak, K.J.; Rodrigues, D.F. Graphene Oxide Nanocomposite Hydrogel Beads for Removal of Selenium in Contaminated Water. ACS Appl. Polym. Mater. 2019, 1, 2668–2679. [Google Scholar] [CrossRef]
- Jegadeesan, G.B.; Mondal, K.; Lalvani, S.B. Adsorption of Se(IV) and Se (VI) Using Copper-Impregnated Activated Carbon and Fly Ash-Extracted Char Carbon. Water Air Soil Pollut. 2015, 226, 234. [Google Scholar] [CrossRef]
- Yamani, J.S.; Lounsbury, A.W.; Zimmerman, J.B. Adsorption of selenite and selenate by nanocrystalline aluminum oxide, neat and impregnated in chitosan beads. Water Res. 2014, 50, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Meher, A.K.; Jadhav, A.; Labhsetwar, N.; Bansiwal, A. Simultaneous removal of selenite and selenate from drinking water using mesoporous activated alumina. Appl. Water Sci. 2019, 10, 10. [Google Scholar] [CrossRef]
- Rovira, M.; Giménez, J.; Martínez, M.; Martínez-Lladó, X.; de Pablo, J.; Martí, V.; Duro, L. Sorption of selenium(IV) and selenium(VI) onto natural iron oxides: Goethite and hematite. J. Hazard. Mater. 2008, 150, 279–284. [Google Scholar] [CrossRef]
- Martínez, M.; Giménez, J.; de Pablo, J.; Rovira, M.; and Duro, L. Sorption of selenium(IV) and selenium(VI) onto magnetit. Appl. Surf. Sci. 2006, 252, 3767–3773. [Google Scholar] [CrossRef]
- Zhang, N.; Gang, D.; Lin, L.-S. Adsorptive Removal of Parts per Million Level Selenate Using Iron-Coated GAC Adsorbents. J. Environ. Eng. 2010, 136, 1089–1095. [Google Scholar] [CrossRef]
- Peak, D. Adsorption mechanisms of selenium oxyanions at the aluminum oxide/water interface. J. Colloid Interface Sci. 2006, 303, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, C.M.; Hernandez, J.; Parsons, J.G.; Gardea-Torresdey, J.L. A study of the removal of selenite and selenate from aqueous solutions using a magnetic iron/manganese oxide nanomaterial and ICP-MS. Microchemical Journal 2010, 96, 324–329. [Google Scholar] [CrossRef]
- Kuan, W.-H.; Lo, S.-L.; Wang, M.K.; and Lin, C.-F. Removal of Se(IV) and Se(VI) from water by aluminum-oxide-coated sand. Water Res. 1998, 32, 915–923. [Google Scholar] [CrossRef]
- Chan, Y.T.; Kuan, W.H.; Chen, T.Y.; Wang, M.K. Adsorption mechanism of selenate and selenite on the binary oxide systems. Water Res. 2009, 43, 4412–4420. [Google Scholar] [CrossRef]
- Das, S.; Jim, M.H.; Essilfie-Dughan, J. Adsorption of selenate onto ferrihydrite, goethite, and lepidocrocite under neutral pH conditions. Appl. Geochem. 2013, 28, 185–193. [Google Scholar] [CrossRef]
- Khamkhash, A.; Srivastava, V.; Ghosh, T.; Akdogan, G.; Ganguli, R.; Aggarwal, S. Mining-Related Selenium Contamination in Alaska, and the State of Current Knowledge. Minerals 2017, 7, 46. [Google Scholar] [CrossRef]
- Lagergren, S. About the theory of so-called adsorption of soluble substabces. Sven. Vetenskapsakad. Handingarl 1898, 24, 1–39. [Google Scholar]
- Weber, W.J.; Morris, J.C. Equilibria and Capacities for Adsorption on Carbon. J. Sanit. Eng. Div. 1964, 90, 79–108. [Google Scholar] [CrossRef]
- Atkins, P.; de Paula, J. Atkins’ Physical Chemistry; Oxford University Press: Oxford, UK, 2005; p. 1008. [Google Scholar]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H.M.F. Over the adsorption in solution. J. Phys. Chem. 1906, 57, 385–470. [Google Scholar]
- Sips, R. On the Structure of a Catalyst Surface. J. Chem. Phys. 1948, 16, 490–495. [Google Scholar] [CrossRef]
- Erhayem, M.; Al-Tohami, F.; Mohamed, R.; Ahmida, K. Isotherm, Kinetic and Thermodynamic Studies for the Sorption of Mercury (II) onto Activated Carbon from Rosmarinus officinalis Leaves. Am. J. Anal. Chem. 2015, 6, 10. [Google Scholar] [CrossRef]
- Ayawei, N.; Ebelegi, A.N.; Wankasi, D. Modelling and Interpretation of Adsorption Isotherms. J. Chem. 2017, 2017, 3039817. [Google Scholar] [CrossRef]
- Inyinbor, A.A.; Adekola, F.A.; Olatunji, G.A. Kinetics, isotherms and thermodynamic modeling of liquid phase adsorption of Rhodamine B dye onto Raphia hookerie fruit epicarp. Water Resour. Ind. 2016, 15, 14–27. [Google Scholar] [CrossRef]



| Elements | Wt,% | At,% |
|---|---|---|
| C | 23.73 | 37.70 |
| O | 36.72 | 43.80 |
| Fe | 26.48 | 9.05 |
| Na | 2.31 | 1.89 |
| F | 1.43 | 1.21 |
| Si | 9.34 | 6.35 |
| TOTAL | 100 | 100 |
| Pseudo first order | ||||
| Temperature (K) | qe,exp (µg/g) | k1 (min−1) | qe,calc (µg/g) | R2 |
| 298 | 24.35 | 0.0383 | 10.07 | 0.9906 |
| 308 | 24.49 | 0.0386 | 14.78 | 0.9905 |
| 318 | 24.62 | 0.0388 | 17.64 | 0.9900 |
| 328 | 24.67 | 0.0371 | 20.61 | 0.9920 |
| Pseudo second order | ||||
| Temperature (K) | qe,exp (µg g−1) | k2 (g µg−1∙min−1) | qe,calc (µg g−1) | R2 |
| 298 | 24.35 | 1238.8 | 26.31 | 0.9997 |
| 308 | 24.49 | 1811.1 | 26.88 | 0.9996 |
| 318 | 24.62 | 2183.0 | 27.24 | 0.9997 |
| 328 | 24.67 | 3164.2 | 27.70 | 0.9996 |
| Intraparticle diffusion model | ||||
| Temperature (K) | Kdiff (mg·g−1 min−1/2) | C | R2 | |
| 298 | 2.62 | 10.2 | 0.8792 | |
| 308 | 2.88 | 10.9 | 0.8173 | |
| 318 | 2.94 | 11.3 | 0.8019 | |
| 328 | 2.98 | 12.01 | 0.8945 | |
| ΔH° (J mol−1) | ΔS° (J mol−1 K−1) | ΔG° (J mol−1) | R2 | |||
|---|---|---|---|---|---|---|
| 22.71 | 95.84 | 298 K | 308 K | 318 K | 328 K | 0.9899 |
| −28.53 | −29.49 | −30.45 | −31.41 | |||
| Langmuir Isotherm | |||
| qm,exp (µg g−1) | KL (L µg−1) | qL (µg g−1) | R2 |
| 6180 | 6.2·10−4 | 7086 | 0.9810 |
| Freundlich isotherm | |||
| KF (µg g−1) | 1/nF | R2 | |
| 208.2 | 0.35 | 0.8391 | |
| Sips isotherm | |||
| KS | qS (µg g−1) | 1/nS | R2 |
| 1.62 | 6718 | 0.21 | 0.9916 |
| Temkin isotherm | |||
| AT [L/g] | bT | R2 | |
| 0.52 | 144.07 | 0.2788 | |
| Dubinin–Radushkevich isotherm | |||
| qm [mg/g] | ε | R2 | |
| 1.01 | 0.366 | 0.5649 | |
| Materials | q, mg g−1 | References |
|---|---|---|
| Graphene oxide nanocomposite hydrogel beads | 1.62 | [36] |
| Fly ash extracted char carbon | 0.68 | [37] |
| Chitosan beads | 2.00 | [38] |
| Iron-Oxy hydroxides | 0.001 | [28] |
| Mesoporous activated alumina | 0.0054 | [39] |
| Fly ash extracted char carbon | 0.44 | [37] |
| Hematite | 0.24 | [40] |
| Magnetite | 0.25 | [41] |
| Iron-coated granular activated carbon | 0.39 | [42] |
| Corundum | 0.59 | [43] |
| Nano-Jacobsite | 0.77 | [44] |
| Aluminum-oxide-coated sand | 0.92 | [45] |
| Binary oxide [Fe (III)/SiO2] | 1.33 | [46] |
| Ferrihydrite | 2.00 | [47] |
| Chitosan beads | 2.00 | [38] |
| SiO2/Fe(acac)3/NaF | 6.00 | This paper |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mladin, G.; Ciopec, M.; Negrea, A.; Duteanu, N.; Negrea, P.; Svera, P.; Ianăşi, C. Selenite Removal from Aqueous Solution Using Silica–Iron Oxide Nanocomposite Adsorbents. Gels 2023, 9, 497. https://doi.org/10.3390/gels9060497
Mladin G, Ciopec M, Negrea A, Duteanu N, Negrea P, Svera P, Ianăşi C. Selenite Removal from Aqueous Solution Using Silica–Iron Oxide Nanocomposite Adsorbents. Gels. 2023; 9(6):497. https://doi.org/10.3390/gels9060497
Chicago/Turabian StyleMladin, Georgiana, Mihaela Ciopec, Adina Negrea, Narcis Duteanu, Petru Negrea, Paula Svera (m. Ianăşi), and Cătălin Ianăşi. 2023. "Selenite Removal from Aqueous Solution Using Silica–Iron Oxide Nanocomposite Adsorbents" Gels 9, no. 6: 497. https://doi.org/10.3390/gels9060497
APA StyleMladin, G., Ciopec, M., Negrea, A., Duteanu, N., Negrea, P., Svera, P., & Ianăşi, C. (2023). Selenite Removal from Aqueous Solution Using Silica–Iron Oxide Nanocomposite Adsorbents. Gels, 9(6), 497. https://doi.org/10.3390/gels9060497








