Tumor Microenvironment-Responsive 6-Mercaptopurine-Releasing Injectable Hydrogel for Colon Cancer Treatment
Abstract
1. Introduction
2. Results and Discussion
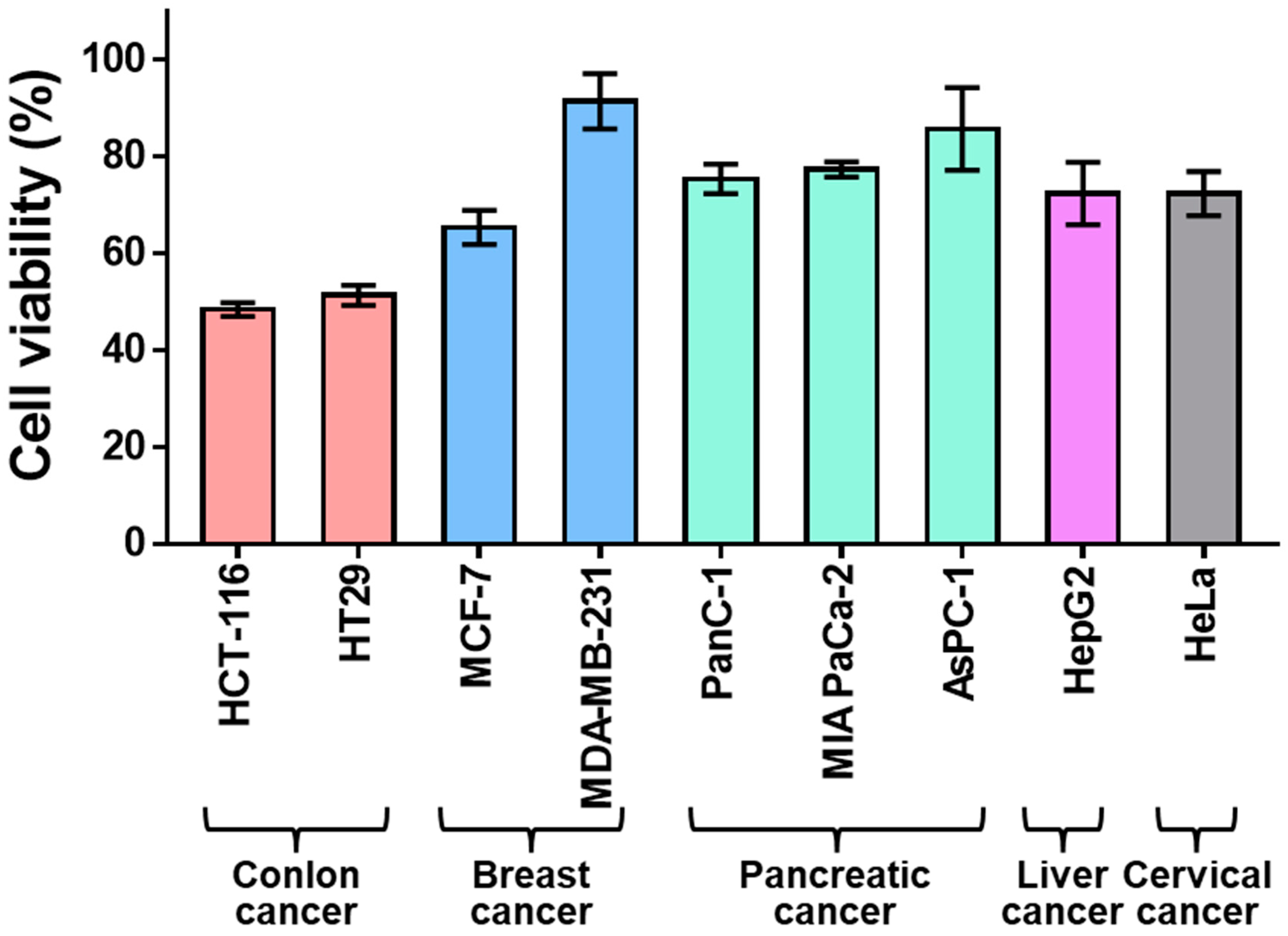
2.1. Sensitivity of Colon Cancer to 6-Mercaptopurine
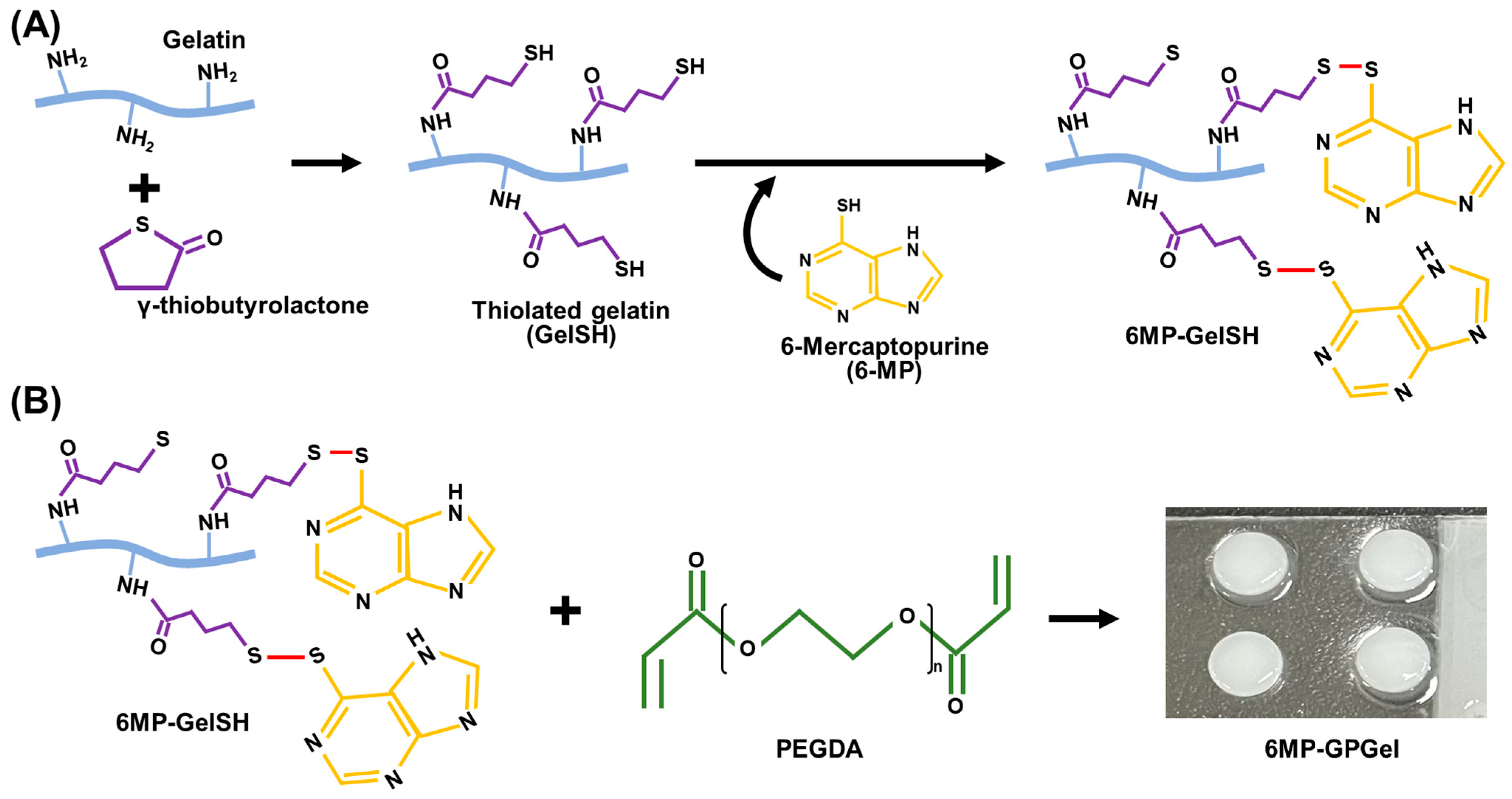
2.2. Characterization of Hydrogel
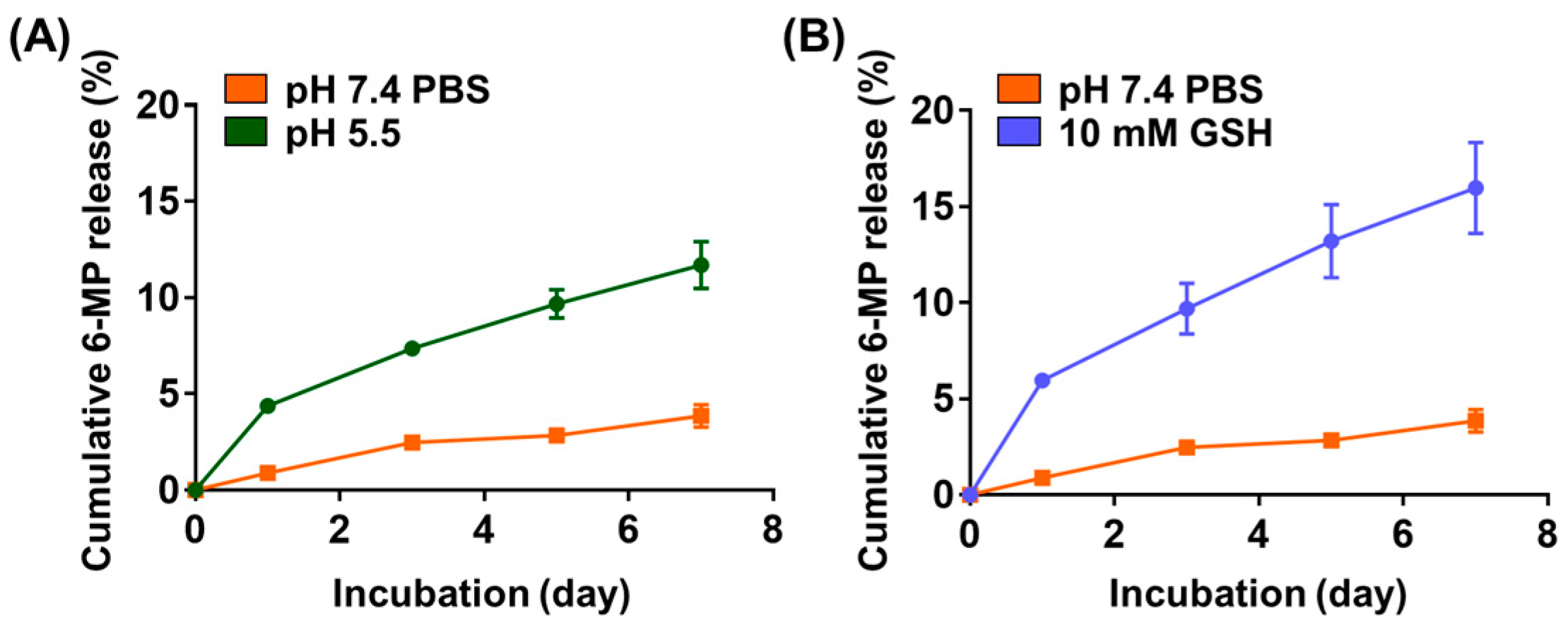
2.3. Release of 6-MP from 6MP-GPGel
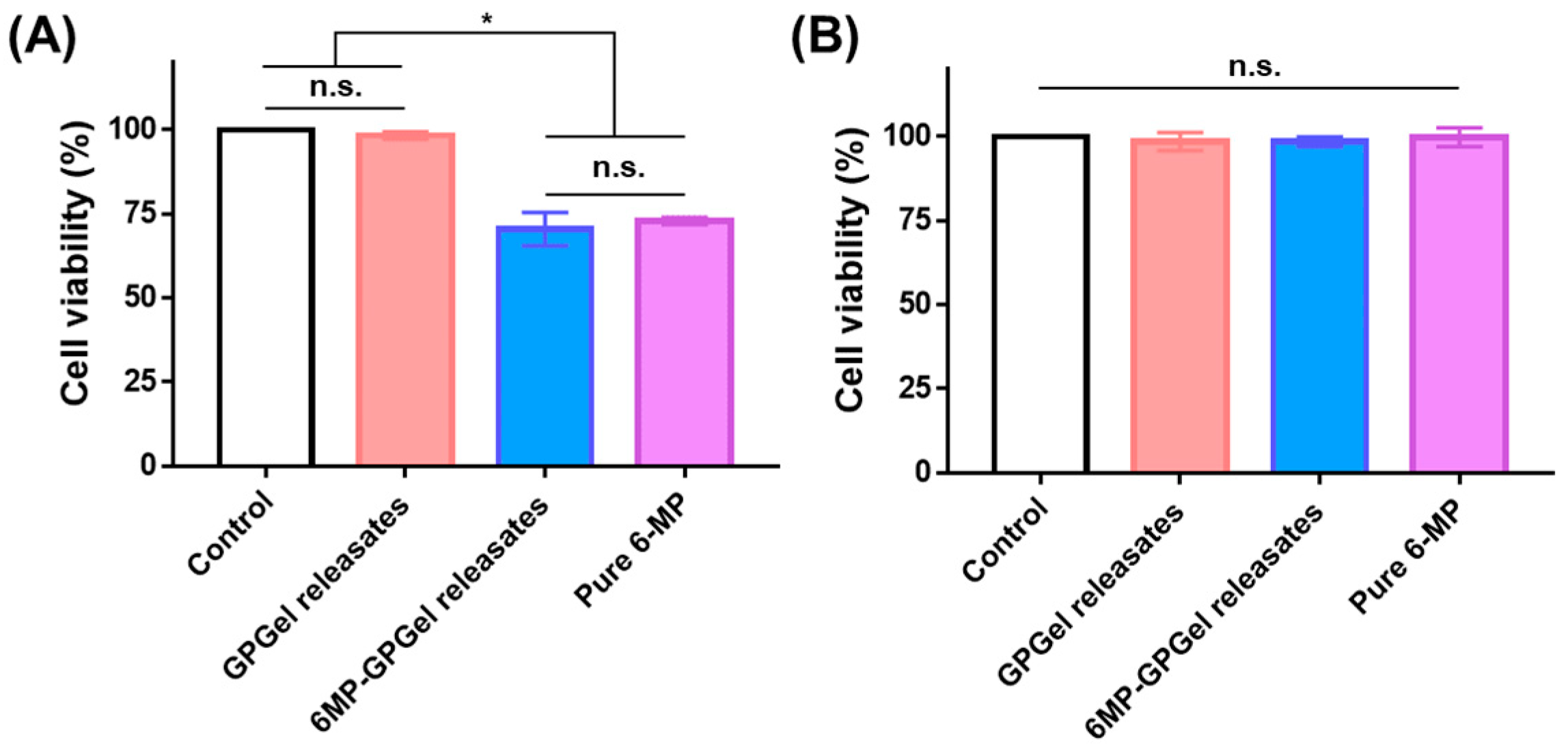
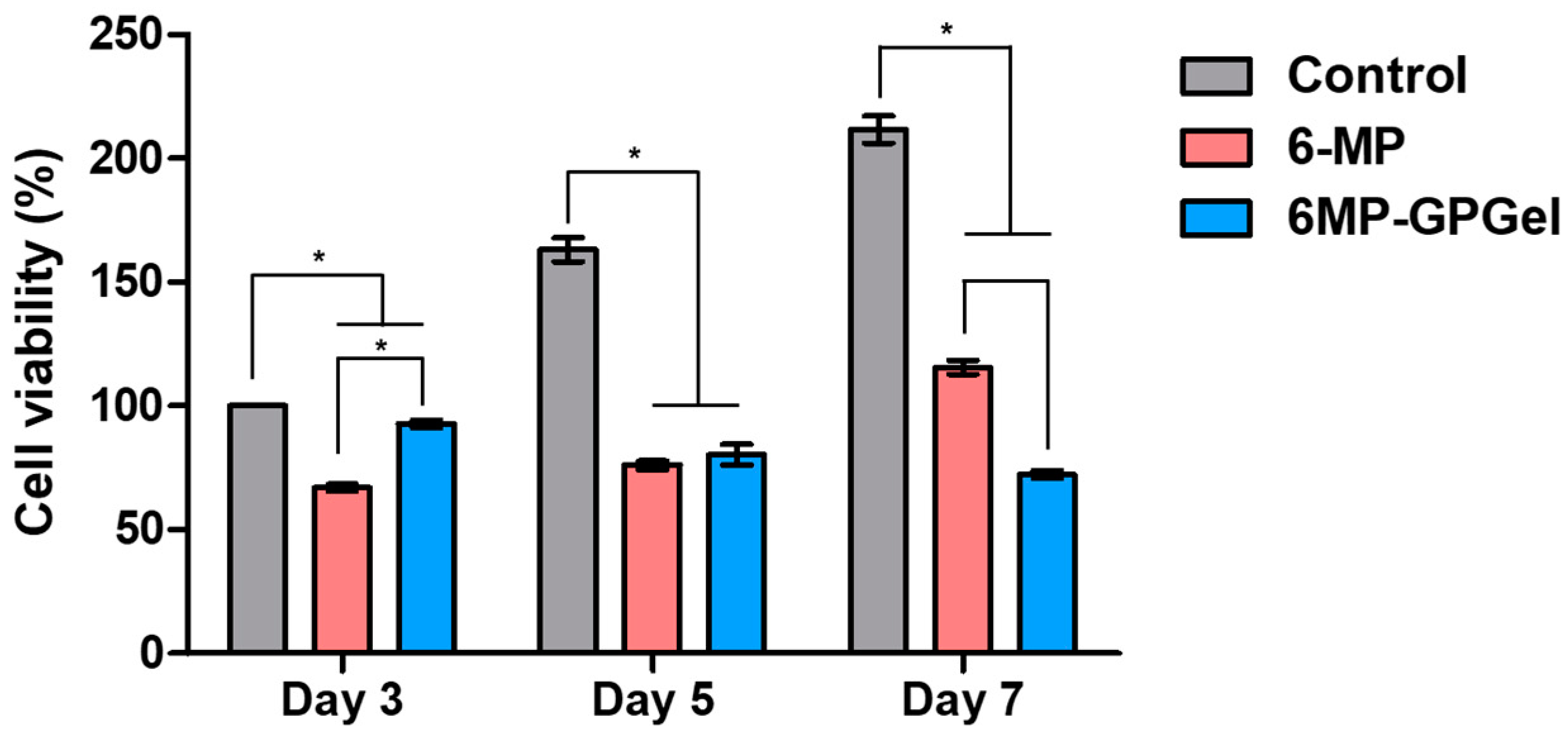
2.4. Anticancer Efficacy of 6MP-GPGel
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Anticancer Efficacy of 6-Mercaptopurine
4.3. Synthesis of Thiolated Gelatin
4.4. Conjugation of 6-Mercaptopurine to Thiolated Gelatin
4.5. Fabrication of Drug-Embedded Composite Hydrogel
4.6. Scanning Electron Microscopy
4.7. In Vitro 6-Mercaptopurine Release Profile
4.8. Anticancer Activity of Released 6-MP from Hydrogel
4.9. Anticancer Effect of Composite Hydrogel
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA-Cancer J. Clin 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Sawicki, T.; Ruszkowska, M.; Danielewicz, A.; Niedzwiedzka, E.; Arlukowicz, T.; Przybylowicz, K.E. A Review of Colorectal Cancer in Terms of Epidemiology, Risk Factors, Development, Symptoms and Diagnosis. Cancers 2021, 13, 2025. [Google Scholar] [CrossRef]
- Kim, S.; Jwa, Y.; Hong, J.; Kim, K. Inhibition of Colon Cancer Recurrence via Exogenous TRAIL Delivery Using Gel-like Coacervate Microdroplets. Gels 2022, 8, 427. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Pathak, S.; Subramanium, V.D.; Dharanivasan, G.; Murugesan, R.; Verma, R.S. Strategies for targeted drug delivery in treatment of colon cancer: Current trends and future perspectives. Drug Discov. Today 2017, 22, 1224–1232. [Google Scholar] [CrossRef]
- Khan, F.A.; Albalawi, R.; Pottoo, F.H. Trends in targeted delivery of nanomaterials in colon cancer diagnosis and treatment. Med. Res. Rev. 2022, 42, 227–258. [Google Scholar] [CrossRef]
- Kim, S.; Choi, Y.; Kim, K. Coacervate-mediated novel pancreatic cancer drug Aleuria Aurantia lectin delivery for augmented anticancer therapy. Biomater. Res. 2022, 26, 35. [Google Scholar] [CrossRef]
- Mani, G.; Kim, S.; Kim, K. Development of Folate-Thioglycolate-Gold Nanoconjugates by Using Citric Acid-PEG Branched Polymer for Inhibition of MCF-7 Cancer Cell Proliferation. Biomacromolecules 2018, 19, 3257–3267. [Google Scholar] [CrossRef]
- Lali Raveendran, R.; Valsala, M.; Sreenivasan Anirudhan, T. Development of nanosilver embedded injectable liquid crystalline hydrogel from alginate and chitosan for potent antibacterial and anticancer applications. J. Ind. Eng. Chem. 2023, 119, 261–273. [Google Scholar] [CrossRef]
- Sun, Z.Y.; Song, C.J.; Wang, C.; Hu, Y.Q.; Wu, J.H. Hydrogel-Based Controlled Drug Delivery for Cancer Treatment: A Review. Mol. Pharm. 2020, 17, 373–391. [Google Scholar] [CrossRef] [PubMed]
- Fan, D.Y.; Tian, Y.; Liu, Z.J. Injectable Hydrogels for Localized Cancer Therapy. Front. Chem. 2019, 7, 675. [Google Scholar] [CrossRef]
- Zolfagharian, S.; Zahedi, P.; Shafiee Ardestani, M.; Khatibi, A.; Jafarkhani, S. Sodium alginate/xanthan-based nanocomposite hydrogels containing 5-fluorouracil: Characterization and cancer cell death studies in presence of halloysite nanotube. J. Ind. Eng. Chem. 2023, 120, 374–386. [Google Scholar] [CrossRef]
- Guo, X.S.; Cheng, Y.; Zhao, X.T.; Luo, Y.L.; Chen, J.J.; Yuan, W.E. Advances in redox-responsive drug delivery systems of tumor microenvironment. J. Nanobiotechnol. 2018, 16, 74. [Google Scholar] [CrossRef]
- Li, M.; Yin, S.; Lin, M.; Chen, X.; Pan, Y.; Peng, Y.; Sun, J.; Kumar, A.; Liu, J. Current status and prospects of metal–organic frameworks for bone therapy and bone repair. J. Mater. Chem. B 2022, 10, 5105–5128. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.M.; Gao, X.Z.; Raza, F.; Zafar, H.; Huang, G.H.; Yang, Y.Y.; Shi, F.; Wang, D.Q.; He, X. Design of GSH-Responsive Curcumin Nanomicelles for Oesophageal Cancer Therapy. Pharmaceutics 2022, 14, 1802. [Google Scholar] [CrossRef]
- Wang, Q.; Guan, J.K.; Wan, J.L.; Li, Z.F. Disulfide based prodrugs for cancer therapy. Rsc. Adv. 2020, 10, 24397–24409. [Google Scholar] [CrossRef]
- Xu, Z.; Wu, Z.; Huang, S.; Ye, K.; Jiang, Y.; Liu, J.; Liu, J.; Lu, X.; Li, B. A metal-organic framework-based immunomodulatory nanoplatform for anti-atherosclerosis treatment. J. Control. Release 2023, 354, 615–625. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Yan, Q.; Xia, C.; Wang, X.; Kumar, A.; Wang, Y.; Liu, Y.; Pan, Y.; Liu, J. Recent advances in cell membrane coated metal–organic frameworks (MOFs) for tumor therapy. J. Mater. Chem. B 2021, 9, 4459–4474. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Choi, Y.; Lee, W.; Kim, K. Fabrication Parameter-Dependent Physico-Chemical Properties of Thiolated Gelatin/PEGDA Interpenetrating Network Hydrogels. Tissue Eng. Regen. Med. 2022, 19, 309–319. [Google Scholar] [CrossRef]
- Hathout, R.M.; Omran, M.K. Gelatin-based particulate systems in ocular drug delivery. Pharm. Dev. Technol. 2016, 21, 379–386. [Google Scholar] [CrossRef]
- Abdelhedi, O.; Salem, A.; Nasri, R.; Nasri, M.; Jridi, M. Food applications of bioactive marine gelatin films. Curr. Opin. Food Sci. 2022, 43, 206–215. [Google Scholar] [CrossRef]
- Han, L.; Xu, J.L.; Lu, X.; Gan, D.L.; Wang, Z.X.; Wang, K.F.; Zhang, H.P.; Yuan, H.P.; Weng, J. Biohybrid methacrylated gelatin/polyacrylamide hydrogels for cartilage repair. J. Mater. Chem. B 2017, 5, 731–741. [Google Scholar] [CrossRef] [PubMed]
- Gyles, D.A.; Castro, L.D.; Silva, J.O.C.; Ribeiro-Costa, R.M. A review of the designs and prominent biomedical advances of natural and synthetic hydrogel formulations. Eur. Polym. J. 2017, 88, 373–392. [Google Scholar] [CrossRef]
- Gajendiran, M.; Rhee, J.S.; Kim, K. Recent Developments in Thiolated Polymeric Hydrogels for Tissue Engineering Applications. Tissue Eng. Part B Rev. 2018, 24, 66–74. [Google Scholar] [CrossRef]
- Cho, H.; Kim, J.; Kim, S.; Jung, Y.C.; Wang, Y.; Kang, B.J.; Kim, K. Dual delivery of stem cells and insulin-like growth factor-1 in coacervate-embedded composite hydrogels for enhanced cartilage regeneration in osteochondral defects. J. Control. Release 2020, 327, 284–295. [Google Scholar] [CrossRef]
- Kim, S.; Kim, J.; Gajendiran, M.; Yoon, M.; Hwang, M.P.; Wang, Y.D.; Kang, B.J.; Kim, K. Enhanced Skull Bone Regeneration by Sustained Release of BMP-2 in Interpenetrating Composite Hydrogels. Biomacromolecules 2018, 19, 4239–4249. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Ramos, A.A.; Marchetti-Laurent, C.; Poindessous, V.; Antonio, S.; Laurent-Puig, P.; Bortoli, S.; Loriot, M.A.; Pallet, N. 6-mercaptopurine promotes energetic failure in proliferating T cells. Oncotarget 2017, 8, 43048–43060. [Google Scholar] [CrossRef]
- Zaboli, M.; Raissi, H.; Zaboli, M. Investigation of nanotubes as the smart carriers for targeted delivery of mercaptopurine anticancer drug. J. Biomol. Struct. Dyn. 2022, 40, 4579–4592. [Google Scholar] [CrossRef]
- Zhang, T.; Gan, Z.; Zhen, S.; Hu, Y.; Hu, X. Ratiometric fluorescent probe based on carbon dots and Zn-doped CdTe QDs for detection of 6-Mercaptopurine. Opt. Mater. 2022, 134, 113196. [Google Scholar] [CrossRef]
- Koshy, S.T.; Desai, R.M.; Joly, P.; Li, J.Y.; Bagrodia, R.K.; Lewin, S.A.; Joshi, N.S.; Mooney, D.J. Click-Crosslinked Injectable Gelatin Hydrogels. Adv. Healthc. Mater. 2016, 5, 541–547. [Google Scholar] [CrossRef]
- Andreazza, R.; Morales, A.; Pieniz, S.; Labidi, J. Gelatin-Based Hydrogels: Potential Biomaterials for Remediation. Polymers 2023, 15, 1026. [Google Scholar] [CrossRef]
- Zhang, J.J.; Wang, J.; Zhang, H.; Lin, J.H.; Ge, Z.G.; Zou, X.N. Macroporous interpenetrating network of polyethylene glycol (PEG) and gelatin for cartilage regeneration. Biomed. Mater. 2016, 11, 035014. [Google Scholar] [CrossRef] [PubMed]
- Raman, T.S.; Kuehnert, M.; Daikos, O.; Scherzer, T.; Krommelbein, C.; Mayr, S.G.; Abel, B.; Schulze, A. A study on the material properties of novel PEGDA/gelatin hybrid hydrogels polymerized by electron beam irradiation. Front. Chem. 2023, 10, 1094981. [Google Scholar] [CrossRef] [PubMed]
- Gong, M.; Yang, J.; Li, Y.S.; Gu, J.L. Glutathione-responsive nanoscale MOFs for effective intracellular delivery of the anticancer drug 6-mercaptopurine. Chem. Commun. 2020, 56, 6448–6451. [Google Scholar] [CrossRef]
- Kennedy, L.; Sandhu, J.K.; Harper, M.E.; Cuperlovic-Culf, M. Role of Glutathione in Cancer: From Mechanisms to Therapies. Biomolecules 2020, 10, 1429. [Google Scholar] [CrossRef]
- Li, Y.; An, L.; Lin, J.; Tian, Q.; Yang, S. Smart nanomedicine agents for cancer, triggered by pH, glutathione, H2O2, or H2S. Int. J. Nanomed. 2019, 14, 5729–5749. [Google Scholar] [CrossRef] [PubMed]
- Justus, C.; Dong, L.; Yang, L. Acidic tumor microenvironment and pH-sensing G protein-coupled receptors. Front. Physiol. 2013, 4, 354. [Google Scholar] [CrossRef]
- Deirram, N.; Zhang, C.H.; Kermaniyan, S.S.; Johnston, A.P.R.; Such, G.K. pH-Responsive Polymer Nanoparticles for Drug Delivery. Macromol. Rapid. Comm. 2019, 40, e1800917. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, S.; So, J.H.; Kim, K.; Koo, H.J. Cytotoxicity of Gallium-Indium Liquid Metal in an Aqueous Environment. ACS Appl. Mater. Inter. 2018, 10, 17448–17454. [Google Scholar] [CrossRef]
- Wang, M.O.; Etheridge, J.M.; Thompson, J.A.; Vorwald, C.E.; Dean, D.; Fisher, J.P. Evaluation of the In Vitro Cytotoxicity of Cross-Linked Biomaterials. Biomacromolecules 2013, 14, 1321–1329. [Google Scholar] [CrossRef]
- Ji, G.F.; Zhang, Y.; Si, X.H.; Yao, H.C.; Ma, S.; Xu, Y.D.; Zhao, J.Y.; Ma, C.; He, C.L.; Tang, Z.H.; et al. Biopolymer Immune Implants’ Sequential Activation of Innate and Adaptive Immunity for Colorectal Cancer Postoperative Immunotherapy. Adv. Mater. 2021, 33, e2004559. [Google Scholar] [CrossRef]
- Wang, W.J.; Fang, C.J.; Wang, X.Z.; Chen, Y.X.; Wang, Y.N.; Feng, W.; Yan, C.H.; Zhao, M.; Peng, S.Q. Modifying mesoporous silica nanoparticles to avoid the metabolic deactivation of 6-mercaptopurine and methotrexate in combinatorial chemotherapy. Nanoscale 2013, 5, 6249–6253. [Google Scholar] [CrossRef] [PubMed]
- Nezhad-Mokhtari, P.; Ghorbani, M.; Mahmoodzadeh, F. Smart co-delivery of 6-mercaptopurine and methotrexate using disulphide-based PEGylated-nanogels for effective treatment of breast cancer. New J. Chem. 2019, 43, 12159–12167. [Google Scholar] [CrossRef]
- Jakubowski, M.; Kucinska, M.; Ratajczak, M.; Pokora, M.; Murias, M.; Voelkel, A.; Sandomierski, M. Zinc forms of faujasite zeolites as a drug delivery system for 6-mercaptopurine. Microporous Mesoporous Mater. 2022, 343, 112194. [Google Scholar] [CrossRef]
- Gajendiran, M.; Kim, S.; Jo, H.; Kim, K. Fabrication of pH responsive coacervates using a polycation-b-polypropylene glycol diblock copolymer for versatile delivery platforms. J. Ind. Eng. Chem. 2020, 90, 36–46. [Google Scholar] [CrossRef]
- Kim, S.; Lee, J.; Hwang, M.P.; Wang, Y.; Kim, K. Influence of fiber architecture and growth factor formulation on osteoblastic differentiation of mesenchymal stem cells in coacervate-coated electrospun fibrous scaffolds. J. Ind. Eng. Chem. 2019, 79, 236–244. [Google Scholar] [CrossRef]
- Kim, K.; Chen, W.C.W.; Heo, Y.; Wang, Y. Polycations and their biomedical applications. Prog. Polym. Sci. 2016, 60, 18–50. [Google Scholar] [CrossRef]
- Rehman, S.U.; Yaseen, Z.; Husain, M.A.; Sarwar, T.; Ishqi, H.M.; Tabish, M. Interaction of 6 Mercaptopurine with Calf Thymus DNA–Deciphering the Binding Mode and Photoinduced DNA Damage. PLoS ONE 2014, 9, e93913. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.; Lee, W.; Park, H.; Kim, K. Tumor Microenvironment-Responsive 6-Mercaptopurine-Releasing Injectable Hydrogel for Colon Cancer Treatment. Gels 2023, 9, 319. https://doi.org/10.3390/gels9040319
Kim S, Lee W, Park H, Kim K. Tumor Microenvironment-Responsive 6-Mercaptopurine-Releasing Injectable Hydrogel for Colon Cancer Treatment. Gels. 2023; 9(4):319. https://doi.org/10.3390/gels9040319
Chicago/Turabian StyleKim, Sungjun, Wonjeong Lee, Heewon Park, and Kyobum Kim. 2023. "Tumor Microenvironment-Responsive 6-Mercaptopurine-Releasing Injectable Hydrogel for Colon Cancer Treatment" Gels 9, no. 4: 319. https://doi.org/10.3390/gels9040319
APA StyleKim, S., Lee, W., Park, H., & Kim, K. (2023). Tumor Microenvironment-Responsive 6-Mercaptopurine-Releasing Injectable Hydrogel for Colon Cancer Treatment. Gels, 9(4), 319. https://doi.org/10.3390/gels9040319






