Nanocomposite Hydrogels as Functional Extracellular Matrices
Abstract
:1. Introduction: Nanocomposite Hydrogels for Tissue Engineering
2. Nano-Enhanced Tissue Scaffolds
2.1. Nanoparticles for Mechanical Reinforcement
2.2. Nanomaterials Controlling Structural Alignment
2.3. Nanoparticles Enhancing Electrical Conductivity
3. Tunable, Remote, and Localized Cell Function Modulation
3.1. Thermal Stimulation
3.2. Electric Stimulation
3.3. Nanoscale Mechanical Properties and Mechanotransduction
4. Nanosensing Functionalities in Engineered Extracellular Matrices
4.1. Monitoring the Chemical Micro-Environment
4.1.1. Metabolic Activity Sensing
| Target | Fluorescent Nano/Micro-Probe | Ref. |
|---|---|---|
| pH †§ | Carbon QDs | [281] |
| pH * | Silicon NPs coated with dopamine and Rhodamine B isothiocyanate | [272] |
| pH † | SNARF-1 encapsulated in CaCO3 NPs | [266] |
| pH ♦ | silicon microcapsules containing molybdenum disulfide (MoS2) nanosheets coated with rhodamine B terminated DEA and BMA | [271] |
| pH † | Polyurethane NPs containing fluorescein and diphenylanthracene | [267] |
| pH | NaYF4:Yb,Er upconversion NP | [282] |
| O2 * | Pd-tetra- (4-carboxyphenyl) tetrabenzoporphyrin dendrimer and Alexa 647 in polyacrylamide NPs | [268] |
| O2 *‖ | Mesoporous silica containing NPRu(dpp)3]2+Cl2 with an upconversion nanoparticle | [283] |
| O2 † | Ru(dpp)3Cl2 (C72H48Cl2N6Ru) immobilized on a polystyrene NP | [263] |
| O2 *§ | phosphorescent Pt(II)-tetrakis(pentafluorophenyl)porphyrin and poly(9,9-diheptylfluorene-alt-9,9-di-p-tolyl-9H-fluorene) encapsulated within poly(methyl methacrylate-co-methacrylic acid)-based nanoparticles | [264] |
| O2 † | palladium-benzoporphyrin in porous poly(2-hydroxyethyl methacrylate) gell particles | [262] |
| O2 † | tris (4,7-diphenyl-1,10-phenanthroline) ruthenium (II) dichloride, or Ru(Ph2phen3)Cl2 with Nile blue chloride in PDMS | [269,278,284] |
| O2 † | PSMA encapslation of platinum(II) meso(2,3,4,5,6-pentafluoro)phenyl porphyrin and Bu3Coum | [260] |
| O2 †‖ | Micelle encapsulated palladium(II)porphyrins with QDs | [261] |
| H2O2 * | DCFDA in an Ormosil NP | [276] |
| H2O2 ‖ | peroxalate NP containing pentacene | [277] |
| HO * | carboxyfluorescein-DNA-GNP | [285] |
4.1.2. Other Signaling Biomarkers
4.2. Sensing Endogenous Physical Fields
4.2.1. Temperature
4.2.2. Mechanosensing
4.2.3. Electric Activity
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 2D | Two dimensional |
| 3D | Three dimensional |
| Ab | Antibody |
| AFM | Atomic force microscopy |
| Ag | Antigen |
| AIOP | Aligned iron oxide particles |
| AP | Action potential |
| BNDF | Brain-derived Neurotrophic Factor |
| CNT | Carbon nanotube |
| CV | Cyclic voltammogram |
| DIV | Days in vitro |
| DRG | Dorsal root ganglion |
| E-Field | Electric-field |
| ECM | Extracellular matrix |
| EIS | Electrochemical impedance spectroscopy |
| ET | Electron Transfer |
| FET | Field-effect-transistor |
| FRET | Förster Resonance Energy Transfer |
| GAG | Glycosaminoglycan |
| GelMA | Gelatin methacrylate |
| GNP | Gold nanoparticle |
| GNR | Gold nanorod |
| GNS | Gold nanoshell |
| GO | Graphene oxide |
| HEK | Human embryonic kidney cell |
| IONP | Iron oxide nanoparticle |
| LOD | Limit of detection |
| LSPR | Localized surface plasmon resonance |
| MOF | Metal-organic-framework |
| MRI | Magnetic resonance imaging |
| NIR | Near-infrared |
| NP | Nanoparticle |
| Ormosil | Organically-modified silica |
| PA | Polyacrylamide |
| PAA | Poly(acrylic acid) |
| PALM | Photoactivated localization microscopy |
| PBS | Phosphate buffered saline |
| PBT | Poly(butylene terephthalate) |
| PCL | Polycaprolactone |
| PCP | Polydimethylsiloxane coated with sodium carboxymethyl cellulose, covered with poly(3,4-ethylenedioxythiophene) |
| PDMAEMA | Poly(2-(dimethylamino)ethyl methacrylate) |
| PDMS | Poly(dimethylsiloxane) |
| PEDOT | Poly(3,4-ethylenedioxythiophene) |
| PEG | Polyethylene glycol |
| PEOT | Poly(ethylene oxide terephthalate) |
| PLGA | Poly (L-lactide-co-glycolide) |
| PLLA | Poly(L-lactide) |
| PNIPAM | Poly(N-isopropylacrylamide) |
| PNIPMAM | Poly(N-isopropylmethacrylamide) |
| PSMA | Poly-styrene-co-maleic anhydride |
| PVA | Poly(vinyl alcohol) |
| QD | Quantum dot |
| QY | Quantum yield |
| rGO | Reduced graphene oxide |
| RIOP | Randomly oriented iron oxide particles |
| ROS | Reactive oxygen species |
| SERS | Surface-enhanced Raman scattering |
| SNR | Signal-to-noise ratio |
| SPR | Surface plasmon resonance |
| STED | Stimulated emission depletion microscopy |
| TE | Tissue engineering |
| TFM | Traction force microscopy |
| TIRF | Total internal reflection |
| TRPV | Transient receptor potential vanilloid |
| UCNP | Upconversion nanoparticle |
| UV | Ultraviolet |
References
- Parks, W.C.; Mecham, R.P. (Eds.) Extracellular Matrix Degradation; Springer: Berlin/Heidelberg, Germany, 2011; Volume 1, ISBN 978-3-642-16860-4. [Google Scholar]
- Wheeldon, I.; Farhadi, A.; Bick, A.G.; Jabbari, E.; Khademhosseini, A. Nanoscale Tissue Engineering: Spatial Control over Cell-Materials Interactions. Nanotechnology 2011, 22, 212001. [Google Scholar] [CrossRef] [PubMed]
- Nicolas, J.; Magli, S.; Rabbachin, L.; Sampaolesi, S.; Nicotra, F.; Russo, L. 3D Extracellular Matrix Mimics: Fundamental Concepts and Role of Materials Chemistry to Influence Stem Cell Fate. Biomacromolecules 2020, 21, 1968–1994. [Google Scholar] [CrossRef] [PubMed]
- Cai, S.; Wu, C.; Yang, W.; Liang, W.; Yu, H.; Liu, L. Recent Advance in Surface Modification for Regulating Cell Adhesion and Behaviors. Nanotechnol. Rev. 2020, 9, 971–989. [Google Scholar] [CrossRef]
- Kshitiz; Park, J.; Kim, P.; Helen, W.; Engler, A.J.; Levchenko, A.; Kim, D.-H. Control of Stem Cell Fate and Function by Engineering Physical Microenvironments. Integr. Biol. 2012, 4, 1008–1018. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Li, F.; Zhao, X.; Ma, Y.; Li, Y.; Lin, M.; Jin, G.; Lu, T.J.; Genin, G.M.; Xu, F. Functional and Biomimetic Materials for Engineering of the Three-Dimensional Cell Microenvironment. Chem. Rev. 2017, 117, 12764–12850. [Google Scholar] [CrossRef]
- Özkale, B.; Sakar, M.S.; Mooney, D.J. Active Biomaterials for Mechanobiology. Biomaterials 2021, 267, 120497. [Google Scholar] [CrossRef]
- Paul, G.R.; Malhotra, A.; Müller, R. Mechanical Stimuli in the Local in Vivo Environment in Bone: Computational Approaches Linking Organ-Scale Loads to Cellular Signals. Curr. Osteoporos. Rep. 2018, 16, 395–403. [Google Scholar] [CrossRef]
- Li, C.; Xu, Q. Mechanical Stress-Initiated Signal Transduction in Vascular Smooth Muscle Cells in Vitro and in Vivo. Cell. Signal. 2007, 19, 881–891. [Google Scholar] [CrossRef]
- Reneman, R.S.; Arts, T.; Hoeks, A.P.G. Wall Shear Stress—An Important Determinant of Endothelial Cell Function and Structure—In the Arterial System in Vivo. J. Vasc. Res. 2006, 43, 251–269. [Google Scholar] [CrossRef]
- Zhao, M. Electrical Fields in Wound Healing—An Overriding Signal That Directs Cell Migration. Semin. Cell Dev. Biol. 2009, 20, 674–682. [Google Scholar] [CrossRef]
- Song, B.; Zhao, M.; Forrester, J.; McCaig, C. Nerve Regeneration and Wound Healing Are Stimulated and Directed by an Endogenous Electrical Field in Vivo. J. Cell Sci. 2004, 117, 4681–4690. [Google Scholar] [CrossRef] [PubMed]
- Hunter, R.H.F.; López-Gatius, F. Temperature Gradients in the Mammalian Ovary and Genital Tract: A Clinical Perspective. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 252, 382–386. [Google Scholar] [CrossRef] [PubMed]
- Arai, S.; Suzuki, M.; Park, S.-J.; Yoo, J.S.; Wang, L.; Kang, N.-Y.; Ha, H.-H.; Chang, Y.-T. Mitochondria-Targeted Fluorescent Thermometer Monitors Intracellular Temperature Gradient. Chem. Commun. 2015, 51, 8044–8047. [Google Scholar] [CrossRef] [PubMed]
- Son, B.; Kim, H.D.; Kim, M.; Kim, J.A.; Lee, J.; Shin, H.; Hwang, N.S.; Park, T.H. Physical Stimuli-Induced Chondrogenic Differentiation of Mesenchymal Stem Cells Using Magnetic Nanoparticles. Adv. Healthc. Mater. 2015, 4, 1339–1347. [Google Scholar] [CrossRef]
- Marino, A.; Arai, S.; Hou, Y.; Pellegrino, M.; Mazzolai, B.; Mattoli, V.; Suzuki, M.; Ciofani, G. Assessment of the Effects of a Wireless Neural Stimulation Mediated by Piezoelectric Nanoparticles. In Neuromethods; Humana Press: New York, NY, USA, 2018; pp. 109–120. ISBN 9781493975846. [Google Scholar]
- Navaei, A.; Moore, N.; Sullivan, R.T.; Truong, D.; Migrino, R.Q.; Nikkhah, M. Electrically Conductive Hydrogel-Based Micro-Topographies for the Development of Organized Cardiac Tissues. RSC Adv. 2017, 7, 3302–3312. [Google Scholar] [CrossRef]
- Antoni, D.; Burckel, H.; Josset, E.; Noel, G. Three-Dimensional Cell Culture: A Breakthrough in Vivo. Int. J. Mol. Sci. 2015, 16, 5517–5527. [Google Scholar] [CrossRef]
- Alhaque, S.; Themis, M.; Rashidi, H. Three-Dimensional Cell Culture: From Evolution to Revolution. Philos. Trans. R. Soc. B Biol. Sci. 2018, 373, 20170216. [Google Scholar] [CrossRef]
- Pampaloni, F.; Reynaud, E.G.; Stelzer, E.H.K. The Third Dimension Bridges the Gap between Cell Culture and Live Tissue. Nat. Rev. Mol. Cell Biol. 2007, 8, 839–845. [Google Scholar] [CrossRef]
- Fathi-Achachelouei, M.; Knopf-Marques, H.; Ribeiro da Silva, C.E.; Barthès, J.; Bat, E.; Tezcaner, A.; Vrana, N.E. Use of Nanoparticles in Tissue Engineering and Regenerative Medicine. Front. Bioeng. Biotechnol. 2019, 7, 113. [Google Scholar] [CrossRef]
- DeForest, C.A.; Anseth, K.S. Advances in Bioactive Hydrogels to Probe and Direct Cell Fate. Annu. Rev. Chem. Biomol. Eng. 2012, 3, 421–444. [Google Scholar] [CrossRef]
- Naito, H.; Yoshimura, M.; Mizuno, T.; Takasawa, S.; Tojo, T.; Taniguchi, S. The Advantages of Three-Dimensional Culture in a Collagen Hydrogel for Stem Cell Differentiation. J. Biomed. Mater. Res. A 2013, 101, 2838–2845. [Google Scholar] [CrossRef] [PubMed]
- Antman-Passig, M.; Levy, S.; Gartenberg, C.; Schori, H.; Shefi, O. Mechanically Oriented 3D Collagen Hydrogel for Directing Neurite Growth. Tissue Eng. Part A 2017, 23, 403–414. [Google Scholar] [CrossRef] [PubMed]
- Short, A.R.; Czeisler, C.; Stocker, B.; Cole, S.; Otero, J.J.; Winter, J.O. Imaging Cell-Matrix Interactions in 3D Collagen Hydrogel Culture Systems. Macromol. Biosci. 2017, 17, 1600478. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, T.A.E.; Dare, E.V.; Hincke, M. Fibrin: A Versatile Scaffold for Tissue Engineering Applications. Tissue Eng. Part B Rev. 2008, 14, 199–215. [Google Scholar] [CrossRef]
- Kim, J.-S.; Kim, T.H.; Kang, D.L.; Baek, S.Y.; Lee, Y.; Koh, Y.-G.; Kim, Y.I. Chondrogenic Differentiation of Human ASCs by Stiffness Control in 3D Fibrin Hydrogel. Biochem. Biophys. Res. Commun. 2020, 522, 213–219. [Google Scholar] [CrossRef]
- LeSavage, B.L.; Suhar, N.A.; Madl, C.M.; Heilshorn, S.C. Production of Elastin-like Protein Hydrogels for Encapsulation and Immunostaining of Cells in 3D. J. Vis. Exp. 2018, 2018, e57739. [Google Scholar] [CrossRef]
- Mbundi, L.; González-Pérez, M.; González-Pérez, F.; Juanes-Gusano, D.; Rodríguez-Cabello, J.C. Trends in the Development of Tailored Elastin-like Recombinamer–Based Porous Biomaterials for Soft and Hard Tissue Applications. Front. Mater. 2021, 7, 412. [Google Scholar] [CrossRef]
- Barros, D.; Amaral, I.F.; Pêgo, A.P. Laminin-Inspired Cell-Instructive Microenvironments for Neural Stem Cells. Biomacromolecules 2020, 21, 276–293. [Google Scholar] [CrossRef]
- Liu, N.; Ye, X.; Yao, B.; Zhao, M.; Wu, P.; Liu, G.; Zhuang, D.; Jiang, H.; Chen, X.; He, Y.; et al. Advances in 3d Bioprinting Technology for Cardiac Tissue Engineering and Regeneration. Bioact. Mater. 2021, 6, 1388–1401. [Google Scholar] [CrossRef]
- Subbiah, R.; Guldberg, R.E. Materials Science and Design Principles of Growth Factor Delivery Systems in Tissue Engineering and Regenerative Medicine. Adv. Healthc. Mater. 2019, 8, 1801000. [Google Scholar] [CrossRef]
- Unal, A.Z.; West, J.L. Synthetic ECM: Bioactive Synthetic Hydrogels for 3D Tissue Engineering. Bioconjug. Chem. 2020, 31, 2253–2271. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Kotaki, M.; Inai, R.; Ramakrishna, S. Potential of Nanofiber Matrix as Tissue-Engineering Scaffolds. Tissue Eng. 2005, 11, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Dhandayuthapani, B.; Yoshida, Y.; Maekawa, T.; Kumar, D.S. Polymeric Scaffolds in Tissue Engineering Application: A Review. Int. J. Polym. Sci. 2011, 2011, 290602. [Google Scholar] [CrossRef]
- Magin, C.M.; Alge, D.L.; Anseth, K.S. Bio-Inspired 3D Microenvironments: A New Dimension in Tissue Engineering. Biomed. Mater. 2016, 11, 22001. [Google Scholar] [CrossRef]
- Koons, G.L.; Diba, M.; Mikos, A.G. Materials Design for Bone-Tissue Engineering. Nat. Rev. Mater. 2020, 5, 584–603. [Google Scholar] [CrossRef]
- Ghasemi, F.; Hormozi-Nezhad, M.R.; Mahmoudi, M. Identification of Catecholamine Neurotransmitters Using Fluorescence Sensor Array. Anal. Chim. Acta 2016, 917, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Venkatraman, S.K.; Swamiappan, S. Review on Calcium- and Magnesium-based Silicates for Bone Tissue Engineering Applications. J. Biomed. Mater. Res. A 2020, 108, 1546–1562. [Google Scholar] [CrossRef]
- Chan, C.K.; Kumar, T.S.; Liao, S.; Murugan, R.; Ngiam, M.; Ramakrishnan, S. Biomimetic Nanocomposites for Bone Graft Applications. Nanomedicine 2006, 1, 177–188. [Google Scholar] [CrossRef]
- Gong, H.; Chen, F.; Huang, Z.; Gu, Y.; Zhang, Q.; Chen, Y.; Zhang, Y.; Zhuang, J.; Cho, Y.-K.; Fang, R.H.; et al. Biomembrane-Modified Field Effect Transistors for Sensitive and Quantitative Detection of Biological Toxins and Pathogens. ACS Nano 2019, 13, 3714–3722. [Google Scholar] [CrossRef]
- Chen, Q.; Wang, C.; Zhang, X.; Chen, G.; Hu, Q.; Li, H.; Wang, J.; Wen, D.; Zhang, Y.; Lu, Y.; et al. In Situ Sprayed Bioresponsive Immunotherapeutic Gel for Post-Surgical Cancer Treatment. Nat. Nanotechnol. 2019, 14, 89–97. [Google Scholar] [CrossRef]
- Jiang, Y.; Krishnan, N.; Heo, J.; Fang, R.H.; Zhang, L. Nanoparticle–Hydrogel Superstructures for Biomedical Applications. J. Control. Release 2020, 324, 505–521. [Google Scholar] [CrossRef] [PubMed]
- Palmese, L.L.; Thapa, R.K.; Sullivan, M.O.; Kiick, K.L. Hybrid Hydrogels for Biomedical Applications. Curr. Opin. Chem. Eng. 2019, 24, 143–157. [Google Scholar] [CrossRef] [PubMed]
- Thoniyot, P.; Tan, M.J.; Karim, A.A.; Young, D.J.; Loh, X.J. Nanoparticle–Hydrogel Composites: Concept, Design, and Applications of These Promising, Multi-Functional Materials. Adv. Sci. 2015, 2, 1400010. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Duan, L.; Zhang, Y.; Cao, J.; Zhang, K. Current Hydrogel Advances in Physicochemical and Biological Response-Driven Biomedical Application Diversity. Signal Transduct. Target. Ther. 2021, 6, 426. [Google Scholar] [CrossRef]
- Abrego, C.J.G.; Dedroog, L.; Deschaume, O.; Wellens, J.; Vananroye, A.; Lettinga, P.; Patterson, J.; Bartic, C. Multiscale Characterization of the Mechanical Properties of Fibrin and Polyethylene Glycol (PEG) Hydrogels for Tissue Engineering Applications. Macromol. Chem. Phys. 2022, 223, 2100366. [Google Scholar] [CrossRef]
- Xing, W.; Tang, Y. On Mechanical Properties of Nanocomposite Hydrogels: Searching for Superior Properties. Nano Mater. Sci. 2022, 4, 83–96. [Google Scholar] [CrossRef]
- Jaiswal, M.K.; Xavier, J.R.; Carrow, J.K.; Desai, P.; Alge, D.; Gaharwar, A.K. Mechanically Stiff Nanocomposite Hydrogels at Ultralow Nanoparticle Content. ACS Nano 2016, 10, 246–256. [Google Scholar] [CrossRef]
- Ashtari, K.; Nazari, H.; Ko, H.; Tebon, P.; Akhshik, M.; Akbari, M.; Alhosseini, S.N.; Mozafari, M.; Mehravi, B.; Soleimani, M.; et al. Electrically Conductive Nanomaterials for Cardiac Tissue Engineering. Adv. Drug Deliv. Rev. 2019, 144, 162–179. [Google Scholar] [CrossRef]
- Sherrell, P.C.; Cieślar-Pobuda, A.; Ejneby, M.S.; Sammalisto, L.; Gelmi, A.; de Muinck, E.; Brask, J.; Łos, M.J.; Rafat, M. Rational Design of a Conductive Collagen Heart Patch. Macromol. Biosci. 2017, 17, 1600446. [Google Scholar] [CrossRef]
- Wang, H.; Tang, X.; Li, W.; Chen, J.; Li, H.; Yan, J.; Yuan, X.; Wu, H.; Liu, C. Enhanced Osteogenesis of Bone Marrow Stem Cells Cultured on Hydroxyapatite/Collagen I Scaffold in the Presence of Low-Frequency Magnetic Field. J. Mater. Sci. Mater. Med. 2019, 30, 89. [Google Scholar] [CrossRef]
- Chronopoulou, L.; Cacciotti, I.; Amalfitano, A.; di Nitto, A.; D’Arienzo, V.; Nocca, G.; Palocci, C. Biosynthesis of Innovative Calcium Phosphate/Hydrogel Composites: Physicochemical and Biological Characterisation. Nanotechnology 2021, 32, 95102. [Google Scholar] [CrossRef] [PubMed]
- Xavier, J.R.; Thakur, T.; Desai, P.; Jaiswal, M.K.; Sears, N.; Cosgriff-Hernandez, E.; Kaunas, R.; Gaharwar, A.K. Bioactive Nanoengineered Hydrogels for Bone Tissue Engineering: A Growth-Factor-Free Approach. ACS Nano 2015, 9, 3109–3118. [Google Scholar] [CrossRef] [PubMed]
- Gaharwar, A.K.; Rivera, C.; Wu, C.-J.; Chan, B.K.; Schmidt, G. Photocrosslinked Nanocomposite Hydrogels from PEG and Silica Nanospheres: Structural, Mechanical and Cell Adhesion Characteristics. Mater. Sci. Eng. C 2013, 33, 1800–1807. [Google Scholar] [CrossRef] [PubMed]
- Vukajlovic, D.; Parker, J.; Bretcanu, O.; Novakovic, K. Chitosan Based Polymer/Bioglass Composites for Tissue Engineering Applications. Mater. Sci. Eng. C 2019, 96, 955–967. [Google Scholar] [CrossRef]
- Gupta, P.; Agrawal, A.; Murali, K.; Varshney, R.; Beniwal, S.; Manhas, S.; Roy, P.; Lahiri, D. Differential Neural Cell Adhesion and Neurite Outgrowth on Carbon Nanotube and Graphene Reinforced Polymeric Scaffolds. Mater. Sci. Eng. C 2019, 97, 539–551. [Google Scholar] [CrossRef]
- Shin, S.R.; Bae, H.; Cha, J.M.; Mun, J.Y.; Chen, Y.-C.; Tekin, H.; Shin, H.; Zarabi, S.; Dokmeci, M.R.; Tang, S.; et al. Carbon Nanotube Reinforced Hybrid Microgels as Scaffold Materials for Cell Encapsulation. ACS Nano 2012, 6, 362–372. [Google Scholar] [CrossRef]
- Song, J.; Zhang, P.; Cheng, L.; Liao, Y.; Xu, B.; Bao, R.; Wang, W.; Liu, W. Nano-Silver in Situ Hybridized Collagen Scaffolds for Regeneration of Infected Full-Thickness Burn Skin. J. Mater. Chem. B 2015, 3, 4231–4241. [Google Scholar] [CrossRef]
- Kim, J.-H.; Sheikh, F.A.; Ju, H.W.; Park, H.J.; Moon, B.M.; Lee, O.J.; Park, C.H. 3D Silk Fibroin Scaffold Incorporating Titanium Dioxide (TiO2) Nanoparticle (NPs) for Tissue Engineering. Int. J. Biol. Macromol. 2014, 68, 158–168. [Google Scholar] [CrossRef]
- Li, Y.; Shi, X.; Tian, L.; Sun, H.; Wu, Y.; Li, X.; Li, J.; Wei, Y.; Han, X.; Zhang, J.; et al. AuNP-Collagen Matrix with Localized Stiffness for Cardiac-Tissue Engineering: Enhancing the Assembly of Intercalated Discs by Β1-Integrin-Mediated Signaling. Adv. Mater. 2016, 28, 10230–10235. [Google Scholar] [CrossRef]
- Li, Y.; Huang, G.; Gao, B.; Li, M.; Genin, G.M.; Lu, T.J.; Xu, F. Magnetically Actuated Cell-Laden Microscale Hydrogels for Probing Strain-Induced Cell Responses in Three Dimensions. NPG Asia Mater. 2016, 8, e238. [Google Scholar] [CrossRef]
- Shin, S.R.; Jung, S.M.; Zalabany, M.; Kim, K.; Zorlutuna, P.; Kim, S.B.; Nikkhah, M.; Khabiry, M.; Azize, M.; Kong, J.; et al. Carbon-Nanotube-Embedded Hydrogel Sheets for Engineering Cardiac Constructs and Bioactuators. ACS Nano 2013, 7, 2369–2380. [Google Scholar] [CrossRef] [PubMed]
- Marcelo, G.; López-González, M.; Mendicuti, F.; Tarazona, M.P.; Valiente, M. Poly(N-Isopropylacrylamide)/Gold Hybrid Hydrogels Prepared by Catechol Redox Chemistry. Characterization and Smart Tunable Catalytic Activity. Macromolecules 2014, 47, 6028–6036. [Google Scholar] [CrossRef]
- Chen, H.; Sun, J.; Wang, Z.; Zhou, Y.; Lou, Z.; Chen, B.; Wang, P.; Guo, Z.; Tang, H.; Ma, J.; et al. Magnetic Cell-Scaffold Interface Constructed by Superparamagnetic IONP Enhanced Osteogenesis of Adipose-Derived Stem Cells. ACS Appl. Mater. Interfaces 2018, 10, 44279–44289. [Google Scholar] [CrossRef] [PubMed]
- Arno, M.C.; Inam, M.; Weems, A.C.; Li, Z.; Binch, A.L.A.; Platt, C.I.; Richardson, S.M.; Hoyland, J.A.; Dove, A.P.; O’Reilly, R.K. Exploiting the Role of Nanoparticle Shape in Enhancing Hydrogel Adhesive and Mechanical Properties. Nat. Commun. 2020, 11, 1420. [Google Scholar] [CrossRef]
- Yanagioka, M.; Toney, M.F.; Frank, C.W. Influence of Interfacial Layer between Nanoparticles and Polymeric Matrix on Viscoelastic Properties of Hydrogel Nanocomposites. Macromolecules 2009, 42, 1331–1343. [Google Scholar] [CrossRef]
- Pardo, A.; Gómez-Florit, M.; Barbosa, S.; Taboada, P.; Domingues, R.M.A.; Gomes, M.E. Magnetic Nanocomposite Hydrogels for Tissue Engineering: Design Concepts and Remote Actuation Strategies to Control Cell Fate. ACS Nano 2021, 15, 175–209. [Google Scholar] [CrossRef]
- Ladoux, B.; Mège, R.-M.; Trepat, X. Front–Rear Polarization by Mechanical Cues: From Single Cells to Tissues. Trends Cell Biol. 2016, 26, 420–433. [Google Scholar] [CrossRef]
- Wickström, S.A.; Niessen, C.M. Cell Adhesion and Mechanics as Drivers of Tissue Organization and Differentiation: Local Cues for Large Scale Organization. Curr. Opin. Cell Biol. 2018, 54, 89–97. [Google Scholar] [CrossRef]
- Shradhanjali, A.; Riehl, B.D.; Kwon, I.K.; Lim, J.Y. Cardiomyocyte Stretching for Regenerative Medicine and Hypertrophy Study. Tissue Eng. Regen. Med. 2015, 12, 398–409. [Google Scholar] [CrossRef]
- Gangatharan, G.; Schneider-Maunoury, S.; Breau, M.A. Role of Mechanical Cues in Shaping Neuronal Morphology and Connectivity. Biol. Cell 2018, 110, 125–136. [Google Scholar] [CrossRef]
- Dedroog, L.; Deschaume, O.; Abrego, C.J.G.; Koos, E.; de Coene, Y.; Vananroye, A.; Thielemans, W.; Bartic, C.; Lettinga, M.P. Stress-Controlled Shear Flow Alignment of Collagen Type I Hydrogel Systems. Acta Biomaterialia 2022, 150, 128–137. [Google Scholar] [CrossRef]
- Kwok, C.B.; Ho, F.C.; Li, C.W.; Ngan, A.H.W.; Chan, D.; Chan, B.P. Compression-Induced Alignment and Elongation of Human Mesenchymal Stem Cell (HMSC) in 3D Collagen Constructs Is Collagen Concentration Dependent. J. Biomed. Mater. Res. A 2013, 101, 1716–1725. [Google Scholar] [CrossRef]
- Shahbazi, M.-A.; Ghalkhani, M.; Maleki, H. Directional Freeze-casting: A Bioinspired Method to Assemble Multifunctional Aligned Porous Structures for Advanced Applications. Adv. Eng. Mater. 2020, 22, 2000033. [Google Scholar] [CrossRef]
- Roeder, L.; Bender, P.; Kundt, M.; Tschöpe, A.; Schmidt, A.M. Magnetic and Geometric Anisotropy in Particle-Crosslinked Ferrohydrogels. Phys. Chem. Chem. Phys. 2015, 17, 1290–1298. [Google Scholar] [CrossRef] [PubMed]
- Fuhrer, R.; Athanassiou, E.K.; Luechinger, N.A.; Stark, W.J. Crosslinking Metal Nanoparticles into the Polymer Backbone of Hydrogels Enables Preparation of Soft, Magnetic Field-Driven Actuators with Muscle-like Flexibility. Small 2009, 5, 383–388. [Google Scholar] [CrossRef]
- Kokkinis, D.; Schaffner, M.; Studart, A.R. Multimaterial Magnetically Assisted 3D Printing of Composite Materials. Nat. Commun. 2015, 6, 8643. [Google Scholar] [CrossRef] [PubMed]
- Sano, K.; Ishida, Y.; Aida, T. Synthesis of Anisotropic Hydrogels and Their Applications. Angew. Chem. Int. Ed. 2018, 57, 2532–2543. [Google Scholar] [CrossRef] [PubMed]
- Ramón-Azcón, J.; Ahadian, S.; Estili, M.; Liang, X.; Ostrovidov, S.; Kaji, H.; Shiku, H.; Ramalingam, M.; Nakajima, K.; Sakka, Y.; et al. Dielectrophoretically Aligned Carbon Nanotubes To Control Electrical and Mechanical Properties of Hydrogels to Fabricate Contractile Muscle Myofibers. Adv. Mater. 2013, 25, 4028–4034. [Google Scholar] [CrossRef] [PubMed]
- Ahadian, S.; Ramón-Azcón, J.; Estili, M.; Liang, X.; Ostrovidov, S.; Shiku, H.; Ramalingam, M.; Nakajima, K.; Sakka, Y.; Bae, H.; et al. Hybrid Hydrogels Containing Vertically Aligned Carbon Nanotubes with Anisotropic Electrical Conductivity for Muscle Myofiber Fabrication. Sci. Rep. 2015, 4, 4271. [Google Scholar] [CrossRef]
- Ahadian, S.; Yamada, S.; Ramón-Azcón, J.; Estili, M.; Liang, X.; Nakajima, K.; Shiku, H.; Khademhosseini, A.; Matsue, T. Hybrid Hydrogel-Aligned Carbon Nanotube Scaffolds to Enhance Cardiac Differentiation of Embryoid Bodies. Acta Biomater. 2016, 31, 134–143. [Google Scholar] [CrossRef]
- Inadomi, T.; Ikeda, S.; Okumura, Y.; Kikuchi, H.; Miyamoto, N. Photo-Induced Anomalous Deformation of Poly(N-Isopropylacrylamide) Gel Hybridized with an Inorganic Nanosheet Liquid Crystal Aligned by Electric Field. Macromol. Rapid Commun. 2014, 35, 1741–1746. [Google Scholar] [CrossRef] [PubMed]
- Small, W.R.; Paunov, V.N. Dielectrophoretic Fabrication of Electrically Anisotropic Hydrogels with Bio-Functionalised Silver Nanowires. J. Mater. Chem. B 2013, 1, 5798–5805. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Bai, S.; Ding, Z.; Guo, H.; Shao, Z.; Zhu, H.; Kaplan, D.L. Hydrogel Assembly with Hierarchical Alignment by Balancing Electrostatic Forces. Adv. Mater. Interfaces 2016, 3, 1500687. [Google Scholar] [CrossRef]
- Gao, L.; Zhao, X. Electrorheological Behaviors of Barium Titanate/Gelatin Composite Hydrogel Elastomers. J. Appl. Polym. Sci. 2004, 94, 2517–2521. [Google Scholar] [CrossRef]
- Zhao, Z.; Fang, R.; Rong, Q.; Liu, M. Bioinspired Nanocomposite Hydrogels with Highly Ordered Structures. Adv. Mater. 2017, 29, 1703045. [Google Scholar] [CrossRef]
- Gao, F.; Xie, W.; Miao, Y.; Wang, D.; Guo, Z.; Ghosal, A.; Li, Y.; Wei, Y.; Feng, S.S.; Zhao, L.; et al. Magnetic Hydrogel with Optimally Adaptive Functions for Breast Cancer Recurrence Prevention. Adv. Healthc. Mater. 2019, 8, 1900203. [Google Scholar] [CrossRef]
- Mitsumata, T.; Horikoshi, Y.; Negami, K. High-Power Actuators Made of Two-Phase Magnetic Gels. Jpn. J. Appl. Phys. 2008, 47, 7257–7261. [Google Scholar] [CrossRef]
- Sakai, R.; Teramoto, Y.; Nishio, Y. Producing a Magnetically Anisotropic Soft Material: Synthesis of Iron Oxide Nanoparticles in a Carrageenan/PVA Matrix and Stretching of the Hybrid Gelatinous Bulk. Polym. J. 2018, 50, 251–260. [Google Scholar] [CrossRef]
- Bender, P.; Günther, A.; Tschöpe, A.; Birringer, R. Synthesis and Characterization of Uniaxial Ferrogels with Ni Nanorods as Magnetic Phase. J. Magn. Magn. Mater. 2011, 323, 2055–2063. [Google Scholar] [CrossRef]
- Diaz-Bleis, D.; Vales-Pinzón, C.; Freile-Pelegrín, Y.; Alvarado-Gil, J.J. Thermal Characterization of Magnetically Aligned Carbonyl Iron/Agar Composites. Carbohydr. Polym. 2014, 99, 84–90. [Google Scholar] [CrossRef]
- Mitsumata, T.; Honda, A.; Kanazawa, H.; Kawai, M. Magnetically Tunable Elasticity for Magnetic Hydrogels Consisting of Carrageenan and Carbonyl Iron Particles. J. Phys. Chem. B 2012, 116, 12341–12348. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Gong, X.; Fan, Y.; Xia, H. Physically Crosslinked Poly(Vinyl Alcohol) Hydrogels with Magnetic Field Controlled Modulus. Soft Matter 2011, 7, 6205–6212. [Google Scholar] [CrossRef]
- Maggini, L.; Liu, M.; Ishida, Y.; Bonifazi, D. Anisotropically Luminescent Hydrogels Containing Magnetically-Aligned MWCNTs-Eu(III) Hybrids. Adv. Mater. 2013, 25, 2462–2467. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Ohtani, M.; Takata, M.; Saeki, A.; Seki, S.; Ishida, Y.; Aida, T. Magnetically Induced Anisotropic Orientation of Graphene Oxide Locked by in Situ Hydrogelation. ACS Nano 2014, 8, 4640–4649. [Google Scholar] [CrossRef]
- De France, K.J.; Yager, K.G.; Chan, K.J.W.; Corbett, B.; Cranston, E.D.; Hoare, T. Injectable Anisotropic Nanocomposite Hydrogels Direct in Situ Growth and Alignment of Myotubes. Nano Lett. 2017, 17, 6487–6495. [Google Scholar] [CrossRef]
- Echave, M.C.; Domingues, R.M.A.; Gómez-Florit, M.; Pedraz, J.L.; Reis, R.L.; Orive, G.; Gomes, M.E. Biphasic Hydrogels Integrating Mineralized and Anisotropic Features for Interfacial Tissue Engineering. ACS Appl. Mater. Interfaces 2019, 11, 47771–47784. [Google Scholar] [CrossRef]
- Araújo-Custódio, S.; Gomez-Florit, M.; Tomás, A.R.; Mendes, B.B.; Babo, P.S.; Mithieux, S.M.; Weiss, A.; Domingues, R.M.A.; Reis, R.L.; Gomes, M.E. Injectable and Magnetic Responsive Hydrogels with Bioinspired Ordered Structures. ACS Biomater. Sci. Eng. 2019, 5, 1392–1404. [Google Scholar] [CrossRef]
- Tomás, A.R.; Goncąlves, A.I.; Paz, E.; Freitas, P.; Domingues, R.M.A.; Gomes, M.E. Magneto-Mechanical Actuation of Magnetic Responsive Fibrous Scaffolds Boosts Tenogenesis of Human Adipose Stem Cells. Nanoscale 2019, 11, 18255–18271. [Google Scholar] [CrossRef]
- Eguchi, Y.; Ohtori, S.; Sekino, M.; Ueno, S. Effectiveness of Magnetically Aligned Collagen for Neural Regeneration in Vitro and in Vivo. Bioelectromagnetics 2015, 36, 233–243. [Google Scholar] [CrossRef]
- Dubey, N. Neuronal Contact Guidance in Magnetically Aligned Fibrin Gels: Effect of Variation in Gel Mechano-Structural Properties. Biomaterials 2001, 22, 1065–1075. [Google Scholar] [CrossRef]
- Tognato, R.; Armiento, A.R.; Bonfrate, V.; Levato, R.; Malda, J.; Alini, M.; Eglin, D.; Giancane, G.; Serra, T. A Stimuli-Responsive Nanocomposite for 3D Anisotropic Cell-Guidance and Magnetic Soft Robotics. Adv. Funct. Mater. 2019, 29, 1804647. [Google Scholar] [CrossRef]
- Betsch, M.; Cristian, C.; Lin, Y.-Y.; Blaeser, A.; Schöneberg, J.; Vogt, M.; Buhl, E.M.; Fischer, H.; Duarte Campos, D.F. Incorporating 4D into Bioprinting: Real-Time Magnetically Directed Collagen Fiber Alignment for Generating Complex Multilayered Tissues. Adv. Healthc. Mater. 2018, 7, 1800894. [Google Scholar] [CrossRef] [PubMed]
- Omidinia-Anarkoli, A.; Boesveld, S.; Tuvshindorj, U.; Rose, J.C.; Haraszti, T.; de Laporte, L. An Injectable Hybrid Hydrogel with Oriented Short Fibers Induces Unidirectional Growth of Functional Nerve Cells. Small 2017, 13, 1702207. [Google Scholar] [CrossRef] [PubMed]
- Rose, J.C.; Cámara-Torres, M.; Rahimi, K.; Köhler, J.; Möller, M.; de Laporte, L. Nerve Cells Decide to Orient inside an Injectable Hydrogel with Minimal Structural Guidance. Nano Lett. 2017, 17, 3782–3791. [Google Scholar] [CrossRef]
- Arias, S.L.; Shetty, A.; Devorkin, J.; Allain, J.-P. Magnetic Targeting of Smooth Muscle Cells in Vitro Using a Magnetic Bacterial Cellulose to Improve Cell Retention in Tissue-Engineering Vascular Grafts. Acta Biomater. 2018, 77, 172–181. [Google Scholar] [CrossRef]
- Bannerman, A.D.; Li, X.; Wan, W. A ‘Degradable’ Poly(Vinyl Alcohol) Iron Oxide Nanoparticle Hydrogel. Acta Biomater. 2017, 58, 376–385. [Google Scholar] [CrossRef]
- Kito, T.; Shibata, R.; Ishii, M.; Suzuki, H.; Himeno, T.; Kataoka, Y.; Yamamura, Y.; Yamamoto, T.; Nishio, N.; Ito, S.; et al. IPS Cell Sheets Created by a Novel Magnetite Tissue Engineering Method for Reparative Angiogenesis. Sci. Rep. 2013, 3, 1418. [Google Scholar] [CrossRef]
- Zhang, W.; Yang, G.; Wang, X.; Jiang, L.; Jiang, F.; Li, G.; Zhang, Z.; Jiang, X. Magnetically Controlled Growth-Factor-Immobilized Multilayer Cell Sheets for Complex Tissue Regeneration. Adv. Mater. 2017, 29, 1703795. [Google Scholar] [CrossRef]
- Lee, E.A.; Yim, H.; Heo, J.; Kim, H.; Jung, G.; Hwang, N.S. Application of Magnetic Nanoparticle for Controlled Tissue Assembly and Tissue Engineering. Arch. Pharm. Res. 2014, 37, 120–128. [Google Scholar] [CrossRef]
- Rödling, L.; Volz, E.M.; Raic, A.; Brändle, K.; Franzreb, M.; Lee-Thedieck, C. Magnetic Macroporous Hydrogels as a Novel Approach for Perfused Stem Cell Culture in 3D Scaffolds via Contactless Motion Control. Adv. Healthc. Mater. 2018, 7, 1701403. [Google Scholar] [CrossRef]
- Lee, J.; Abdeen, A.A.; Kilian, K.A. Rewiring Mesenchymal Stem Cell Lineage Specification by Switching the Biophysical Microenvironment. Sci. Rep. 2015, 4, 5188. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Tibbitt, M.W.; Basta, L.; Anseth, K.S. Mechanical Memory and Dosing Influence Stem Cell Fate. Nat. Mater. 2014, 13, 645–652. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.; Yu, T.; Tang, S.; Xu, X.; Guo, Z.; Qian, J.; Cheng, Y.; Zhao, Y.; Yan, S.; Zhang, H.; et al. Dual Anisotropicity Comprising 3D Printed Structures and Magnetic Nanoparticle Assemblies: Towards the Promotion of Mesenchymal Stem Cell Osteogenic Differentiation. NPG Asia Mater. 2021, 13, 19. [Google Scholar] [CrossRef]
- Abdeen, A.A.; Lee, J.; Bharadwaj, N.A.; Ewoldt, R.H.; Kilian, K.A. Temporal Modulation of Stem Cell Activity Using Magnetoactive Hydrogels. Adv. Healthc. Mater. 2016, 5, 2536–2544. [Google Scholar] [CrossRef]
- Zhang, L.; Wei, F.; Bai, Q.; Song, D.; Zheng, Z.; Wang, Y.; Liu, X.; Abdulrahman, A.-A.; Bian, Y.; Xu, X.; et al. Oscillating Magnetic Field Regulates Cell Adherence and Endothelialization Based on Magnetic Nanoparticle-Modified Bacterial Cellulose. ACS Appl. Mater. Interfaces 2020, 12, 52467–52478. [Google Scholar] [CrossRef]
- Mehrali, M.; Thakur, A.; Pennisi, C.P.; Talebian, S.; Arpanaei, A.; Nikkhah, M.; Dolatshahi-Pirouz, A. Nanoreinforced Hydrogels for Tissue Engineering: Biomaterials That Are Compatible with Load-Bearing and Electroactive Tissues. Adv. Mater. 2017, 29, 1603612. [Google Scholar] [CrossRef]
- Nazari, H.; Azadi, S.; Hatamie, S.; Zomorrod, M.S.; Ashtari, K.; Soleimani, M.; Hosseinzadeh, S. Fabrication of Graphene-Silver/Polyurethane Nanofibrous Scaffolds for Cardiac Tissue Engineering. Polym. Adv. Technol. 2019, 30, 2086–2099. [Google Scholar] [CrossRef]
- Shapira, A.; Feiner, R.; Dvir, T. Composite Biomaterial Scaffolds for Cardiac Tissue Engineering. Int. Mater. Rev. 2016, 61, 1–19. [Google Scholar] [CrossRef]
- Gorain, B.; Choudhury, H.; Pandey, M.; Kesharwani, P.; Abeer, M.M.; Tekade, R.K.; Hussain, Z. Carbon Nanotube Scaffolds as Emerging Nanoplatform for Myocardial Tissue Regeneration: A Review of Recent Developments and Therapeutic Implications. Biomed. Pharmacother. 2018, 104, 496–508. [Google Scholar] [CrossRef]
- Xu, D.; Fan, L.; Gao, L.; Xiong, Y.; Wang, Y.; Ye, Q.; Yu, A.; Dai, H.; Yin, Y.; Cai, J.; et al. Micro-Nanostructured Polyaniline Assembled in Cellulose Matrix via Interfacial Polymerization for Applications in Nerve Regeneration. ACS Appl. Mater. Interfaces 2016, 8, 17090–17097. [Google Scholar] [CrossRef]
- Sun, H.; Zhou, J.; Huang, Z.; Qu, L.; Lin, N.; Liang, C.; Dai, R.; Tang, L.; Tian, F. Carbon Nanotube-Incorporated Collagen Hydrogels Improve Cell Alignment and the Performance of Cardiac Constructs. Int. J. Nanomed. 2017, 12, 3109–3120. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Zhao, H.; Huang, C.; Du, Y. Mechanically and Electrically Enhanced CNT–Collagen Hydrogels as Potential Scaffolds for Engineered Cardiac Constructs. ACS Biomater. Sci. Eng. 2017, 3, 3017–3021. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Chen, D.; Wang, Z.; Zhang, Z.; Xia, Y.; Xue, H.; Liu, Y. Preparation of an Electrically Conductive Graphene Oxide/Chitosan Scaffold for Cardiac Tissue Engineering. Appl. Biochem. Biotechnol. 2019, 188, 952–964. [Google Scholar] [CrossRef] [PubMed]
- Nair, R.S.; Ameer, J.M.; Alison, M.R.; Anilkumar, T.V. A Gold Nanoparticle Coated Porcine Cholecyst-Derived Bioscaffold for Cardiac Tissue Engineering. Colloids Surf. B Biointerfaces 2017, 157, 130–137. [Google Scholar] [CrossRef]
- Shevach, M.; Fleischer, S.; Shapira, A.; Dvir, T. Gold Nanoparticle-Decellularized Matrix Hybrids for Cardiac Tissue Engineering. Nano Lett. 2014, 14, 5792–5796. [Google Scholar] [CrossRef]
- Sridhar, S.; Venugopal, J.R.; Sridhar, R.; Ramakrishna, S. Cardiogenic Differentiation of Mesenchymal Stem Cells with Gold Nanoparticle Loaded Functionalized Nanofibers. Colloids Surf. B Biointerfaces 2015, 134, 346–354. [Google Scholar] [CrossRef]
- Wickham, A.; Vagin, M.; Khalaf, H.; Bertazzo, S.; Hodder, P.; Dånmark, S.; Bengtsson, T.; Altimiras, J.; Aili, D. Electroactive Biomimetic Collagen-Silver Nanowire Composite Scaffolds. Nanoscale 2016, 8, 14146–14155. [Google Scholar] [CrossRef]
- Mou, Y.; Lv, S.; Xiong, F.; Han, Y.; Zhao, Y.; Li, J.; Gu, N.; Zhou, J. Effects of Different Doses of 2,3-Dimercaptosuccinic Acid-Modified Fe2O3 Nanoparticles on Intercalated Discs in Engineered Cardiac Tissues. J. Biomed. Mater. Res. B Appl. Biomater. 2018, 106, 121–130. [Google Scholar] [CrossRef]
- Kalishwaralal, K.; Jeyabharathi, S.; Sundar, K.; Selvamani, S.; Prasanna, M.; Muthukumaran, A. A Novel Biocompatible Chitosan–Selenium Nanoparticles (SeNPs) Film with Electrical Conductivity for Cardiac Tissue Engineering Application. Mater. Sci. Eng. C 2018, 92, 151–160. [Google Scholar] [CrossRef]
- Shevach, M.; Maoz, B.M.; Feiner, R.; Shapira, A.; Dvir, T. Nanoengineering Gold Particle Composite Fibers for Cardiac Tissue Engineering. J. Mater. Chem. B 2013, 1, 5210. [Google Scholar] [CrossRef]
- Lee, J.; Manoharan, V.; Cheung, L.; Lee, S.; Cha, B.-H.; Newman, P.; Farzad, R.; Mehrotra, S.; Zhang, K.; Khan, F.; et al. Nanoparticle-Based Hybrid Scaffolds for Deciphering the Role of Multimodal Cues in Cardiac Tissue Engineering. ACS Nano 2019, 13, 12525–12539. [Google Scholar] [CrossRef] [PubMed]
- Dvir, T.; Timko, B.P.; Brigham, M.D.; Naik, S.R.; Karajanagi, S.S.; Levy, O.; Jin, H.; Parker, K.K.; Langer, R.; Kohane, D.S. Nanowired Three-Dimensional Cardiac Patches. Nat. Nanotechnol. 2011, 6, 720–725. [Google Scholar] [CrossRef] [PubMed]
- Dvir, T.; Timko, B.P.; Kohane, D.S.; Langer, R. Nanotechnological Strategies for Engineering Complex Tissues. Nat. Nanotechnol. 2011, 6, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Fleischer, S.; Shevach, M.; Feiner, R.; Dvir, T. Coiled Fiber Scaffolds Embedded with Gold Nanoparticles Improve the Performance of Engineered Cardiac Tissues. Nanoscale 2014, 6, 9410–9414. [Google Scholar] [CrossRef]
- Navaei, A.; Saini, H.; Christenson, W.; Sullivan, R.T.; Ros, R.; Nikkhah, M. Gold Nanorod-Incorporated Gelatin-Based Conductive Hydrogels for Engineering Cardiac Tissue Constructs. Acta Biomater. 2016, 41, 133–146. [Google Scholar] [CrossRef]
- Shin, S.R.; Shin, C.; Memic, A.; Shadmehr, S.; Miscuglio, M.; Jung, H.Y.; Jung, S.M.; Bae, H.; Khademhosseini, A.; Tang, X.S.; et al. Aligned Carbon Nanotube-Based Flexible Gel Substrates for Engineering Biohybrid Tissue Actuators. Adv. Funct. Mater. 2015, 25, 4486–4495. [Google Scholar] [CrossRef]
- Ahadian, S.; Naito, U.; Surya, V.J.; Darvishi, S.; Estili, M.; Liang, X.; Nakajima, K.; Shiku, H.; Kawazoe, Y.; Matsue, T. Fabrication of Poly(Ethylene Glycol) Hydrogels Containing Vertically and Horizontally Aligned Graphene Using Dielectrophoresis: An Experimental and Modeling Study. Carbon 2017, 123, 460–470. [Google Scholar] [CrossRef]
- Saderi, N.; Rajabi, M.; Akbari, B.; Firouzi, M.; Hassannejad, Z. Fabrication and Characterization of Gold Nanoparticle-Doped Electrospun PCL/Chitosan Nanofibrous Scaffolds for Nerve Tissue Engineering. J. Mater. Sci. Mater. Med. 2018, 29, 134. [Google Scholar] [CrossRef]
- Liu, X.; Miller II, A.L.; Park, S.; Waletzki, B.E.; Terzic, A.; Yaszemski, M.J.; Lu, L. Covalent Crosslinking of Graphene Oxide and Carbon Nanotube into Hydrogels Enhances Nerve Cell Responses. J. Mater. Chem. B 2016, 4, 6930–6941. [Google Scholar] [CrossRef]
- Liu, X.; Miller, A.L.; Park, S.; Waletzki, B.E.; Zhou, Z.; Terzic, A.; Lu, L. Functionalized Carbon Nanotube and Graphene Oxide Embedded Electrically Conductive Hydrogel Synergistically Stimulates Nerve Cell Differentiation. ACS Appl. Mater. Interfaces 2017, 9, 14677–14690. [Google Scholar] [CrossRef]
- Baranes, K.; Shevach, M.; Shefi, O.; Dvir, T. Gold Nanoparticle-Decorated Scaffolds Promote Neuronal Differentiation and Maturation. Nano Lett. 2016, 16, 2916–2920. [Google Scholar] [CrossRef] [PubMed]
- Koppes, A.N.; Keating, K.W.; McGregor, A.L.; Koppes, R.A.; Kearns, K.R.; Ziemba, A.M.; McKay, C.A.; Zuidema, J.M.; Rivet, C.J.; Gilbert, R.J.; et al. Robust Neurite Extension Following Exogenous Electrical Stimulation within Single Walled Carbon Nanotube-Composite Hydrogels. Acta Biomater. 2016, 39, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Song, J.; Zheng, W.; Zhao, X.; Ouyang, Y.; Yuan, W.; Fan, C. 3D Manufacture of Gold Nanocomposite Channels Facilitates Neural Differentiation and Regeneration. Adv. Funct. Mater. 2018, 28, 1707077. [Google Scholar] [CrossRef]
- Ghosh, S.; Haldar, S.; Gupta, S.; Bisht, A.; Chauhan, S.; Kumar, V.; Roy, P.; Lahiri, D. Anisotropically Conductive Biodegradable Scaffold with Coaxially Aligned Carbon Nanotubes for Directional Regeneration of Peripheral Nerves. ACS Appl. Bio Mater. 2020, 3, 5796–5812. [Google Scholar] [CrossRef]
- Demir, U.S.; Shahbazi, R.; Calamak, S.; Ozturk, S.; Gultekinoglu, M.; Ulubayram, K. Gold Nano-Decorated Aligned Polyurethane Nanofibers for Enhancement of Neurite Outgrowth and Elongation. J. Biomed. Mater. Res. A 2018, 106, 1604–1613. [Google Scholar] [CrossRef]
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; Vander Elst, L.; Muller, R.N. Magnetic Iron Oxide Nanoparticles: Synthesis, Stabilization, Vectorization, Physicochemical Characterizations, and Biological Applications. Chem. Rev. 2008, 108, 2064–2110. [Google Scholar] [CrossRef]
- Chen, R.; Romero, G.; Christiansen, M.G.; Mohr, A.; Anikeeva, P. Wireless Magnetothermal Deep Brain Stimulation. Science 2015, 347, 1477–1480. [Google Scholar] [CrossRef]
- Estelrich, J.; Antònia Busquets, M. Iron Oxide Nanoparticles in Photothermal Therapy. Molecules 2018, 23, 1567. [Google Scholar] [CrossRef]
- Nakajima, Y.; Kawano, T.; Taki, Y.; Kojima, C. Visible Light-Responsive Cell Scaffolds with Bilayer Structures for Single Cell Separation. Res. Chem. Intermed. 2018, 44, 4745–4754. [Google Scholar] [CrossRef]
- Zhu, M.; Baffou, G.; Meyerbröker, N.; Polleux, J. Micropatterning Thermoplasmonic Gold Nanoarrays to Manipulate Cell Adhesion. ACS Nano 2012, 6, 7227–7233. [Google Scholar] [CrossRef]
- Huang, H.; Delikanli, S.; Zeng, H.; Ferkey, D.M.; Pralle, A. Remote Control of Ion Channels and Neurons through Magnetic-Field Heating of Nanoparticles. Nat. Nanotechnol. 2010, 5, 602–606. [Google Scholar] [CrossRef] [PubMed]
- Fragal, E.H.; Fragal, V.H.; da Silva, G.H.; Gonçalves, S.P.C.; Martinez, D.S.T.; Rubira, A.F.; Silva, R. Enhancing Near-Infrared Photothermal Efficiency of Biocompatible Flame-Synthesized Carbon Nano-Onions with Metal Dopants and Silica Coating. ACS Appl. Bio Mater. 2020, 3, 5984–5994. [Google Scholar] [CrossRef] [PubMed]
- Roper, D.K.; Ahn, W.; Hoepfner, M. Microscale Heat Transfer Transduced by Surface Plasmon Resonant Gold Nanoparticles. J. Phys. Chem. C 2007, 111, 3636–3641. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Guo, L. Nanomaterial-Enabled Neural Stimulation. Front. Neurosci. 2016, 10, 69. [Google Scholar] [CrossRef]
- Migliori, B.; di Ventra, M.; Kristan, W. Photoactivation of Neurons by Laser-Generated Local Heating. AIP Adv. 2012, 2, 32154. [Google Scholar] [CrossRef] [PubMed]
- Farah, N.; Zoubi, A.; Matar, S.; Golan, L.; Marom, A.; Butson, C.R.; Brosh, I.; Shoham, S. Holographically Patterned Activation Using Photo-Absorber Induced Neural–Thermal Stimulation. J. Neural Eng. 2013, 10, 56004. [Google Scholar] [CrossRef]
- Paściak, A.; Pilch-Wróbel, A.; Marciniak, Ł.; Schuck, P.J.; Bednarkiewicz, A. Standardization of Methodology of Light-to-Heat Conversion Efficiency Determination for Colloidal Nanoheaters. ACS Appl. Mater. Interfaces 2021, 13, 44556–44567. [Google Scholar] [CrossRef]
- Santra, T.S.; Kar, S.; Chen, T.C.; Chen, C.W.; Borana, J.; Lee, M.C.; Tseng, F.G. Near-Infrared Nanosecond-Pulsed Laser-Activated Highly Efficient Intracellular Delivery Mediated by Nano-Corrugated Mushroom-Shaped Gold-Coated Polystyrene Nanoparticles. Nanoscale 2020, 12, 12057–12067. [Google Scholar] [CrossRef]
- Pallavicini, P.; Chirico, G.; Taglietti, A. Harvesting Light to Produce Heat: Photothermal Nanoparticles for Technological Applications and Biomedical Devices. Chem. Eur. J. 2021, 27, 15361–15374. [Google Scholar] [CrossRef]
- Maestro, L.M.; Haro-González, P.; Sánchez-Iglesias, A.; Liz-Marzán, L.M.; García Solé, J.; Jaque, D. Quantum Dot Thermometry Evaluation of Geometry Dependent Heating Efficiency in Gold Nanoparticles. Langmuir 2014, 30, 1650–1658. [Google Scholar] [CrossRef]
- Ferreira-Gonçalves, T.; Ferreira, D.; Ferreira, H.A.; Reis, C.P. Nanogold-Based Materials in Medicine: From Their Origins to Their Future. Nanomedicine 2021, 16, 2695–2723. [Google Scholar] [CrossRef] [PubMed]
- Paviolo, C.; Haycock, J.W.; Yong, J.; Yu, A.; Stoddart, P.R.; McArthur, S.L. Laser Exposure of Gold Nanorods Can Increase Neuronal Cell Outgrowth. Biotechnol. Bioeng. 2013, 110, 2277–2291. [Google Scholar] [CrossRef] [PubMed]
- Paviolo, C.; Thompson, A.C.; Yong, J.; Brown, W.G.A.; Stoddart, P.R. Nanoparticle-Enhanced Infrared Neural Stimulation. J. Neural Eng. 2014, 11, 65002. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Liu, J.; Chen, C. Remote Control and Modulation of Cellular Events by Plasmonic Gold Nanoparticles: Implications and Opportunities for Biomedical Applications. ACS Nano 2017, 11, 2403–2409. [Google Scholar] [CrossRef] [PubMed]
- Marino, A.; Arai, S.; Hou, Y.; Degl’Innocenti, A.; Cappello, V.; Mazzolai, B.; Chang, Y.-T.; Mattoli, V.; Suzuki, M.; Ciofani, G. Gold Nanoshell-Mediated Remote Myotube Activation. ACS Nano 2017, 11, 2494–2508. [Google Scholar] [CrossRef]
- Carvalho-de-Souza, J.L.; Treger, J.S.; Dang, B.; Kent, S.B.H.; Pepperberg, D.R.; Bezanilla, F. Photosensitivity of Neurons Enabled by Cell-Targeted Gold Nanoparticles. Neuron 2015, 86, 207–217. [Google Scholar] [CrossRef]
- Damnjanovic, R.; Bazard, P.; Frisina, R.D.; Bhethanabotla, V.R. Hybrid Electro-Plasmonic Neural Stimulation with Visible-Light-Sensitive Gold Nanoparticles. ACS Nano 2020, 14, 10917–10928. [Google Scholar] [CrossRef]
- Yong, J.; Needham, K.; Brown, W.G.A.; Nayagam, B.A.; McArthur, S.L.; Yu, A.; Stoddart, P.R. Gold-Nanorod-Assisted near-Infrared Stimulation of Primary Auditory Neurons. Adv. Healthc. Mater. 2014, 3, 1862–1868. [Google Scholar] [CrossRef]
- Eom, K.; Kim, J.; Choi, J.M.; Kang, T.; Chang, J.W.; Byun, K.M.; Jun, S.B.; Kim, S.J. Enhanced Infrared Neural Stimulation Using Localized Surface Plasmon Resonance of Gold Nanorods. Small 2014, 10, 3853–3857. [Google Scholar] [CrossRef]
- Yoo, S.; Hong, S.; Choi, Y.; Park, J.-H.; Nam, Y. Photothermal Inhibition of Neural Activity with Near-Infrared-Sensitive Nanotransducers. ACS Nano 2014, 8, 8040–8049. [Google Scholar] [CrossRef]
- Yoo, S.; Kim, R.; Park, J.-H.; Nam, Y. Electro-Optical Neural Platform Integrated with Nanoplasmonic Inhibition Interface. ACS Nano 2016, 10, 4274–4281. [Google Scholar] [CrossRef]
- Shapiro, M.G.; Homma, K.; Villarreal, S.; Richter, C.-P.; Bezanilla, F. Infrared Light Excites Cells by Changing Their Electrical Capacitance. Nat. Commun. 2012, 3, 736. [Google Scholar] [CrossRef]
- Plaksin, M.; Kimmel, E.; Shoham, S. Correspondence: Revisiting the Theoretical Cell Membrane Thermal Capacitance Response. Nat. Commun. 2017, 8, 1431. [Google Scholar] [CrossRef]
- Albert, E.S.; Bec, J.M.; Desmadryl, G.; Chekroud, K.; Travo, C.; Gaboyard, S.; Bardin, F.; Marc, I.; Dumas, M.; Lenaers, G.; et al. TRPV4 Channels Mediate the Infrared Laser-Evoked Response in Sensory Neurons. J. Neurophysiol. 2012, 107, 3227–3234. [Google Scholar] [CrossRef]
- Ostberg, J.R.; Kabingu, E.; Repasky, E.A. Thermal Regulation of Dendritic Cell Activation and Migration from Skin Explants. Int. J. Hyperth. 2003, 19, 520–533. [Google Scholar] [CrossRef] [PubMed]
- Appenheimer, M.M.; Evans, S.S. Temperature and Adaptive Immunity. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2018; Volume 156, pp. 397–415. [Google Scholar]
- Mohamed, M.A.; Fallahi, A.; El-Sokkary, A.M.A.; Salehi, S.; Akl, M.A.; Jafari, A.; Tamayol, A.; Fenniri, H.; Khademhosseini, A.; Andreadis, S.T.; et al. Stimuli-Responsive Hydrogels for Manipulation of Cell Microenvironment: From Chemistry to Biofabrication Technology. Prog. Polym. Sci. 2019, 98, 101147. [Google Scholar] [CrossRef] [PubMed]
- Hribar, K.C.; Meggs, K.; Liu, J.; Zhu, W.; Qu, X.; Chen, S. Three-Dimensional Direct Cell Patterning in Collagen Hydrogels with near-Infrared Femtosecond Laser. Sci. Rep. 2015, 5, 17203. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Shoichet, M.S. A Photolabile Hydrogel for Guided Three-Dimensional Cell Growth and Migration. Nat. Mater. 2004, 3, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Hescham, S.A.; Chiang, P.H.; Gregurec, D.; Moon, J.; Christiansen, M.G.; Jahanshahi, A.; Liu, H.; Rosenfeld, D.; Pralle, A.; Anikeeva, P.; et al. Magnetothermal Nanoparticle Technology Alleviates Parkinsonian-like Symptoms in Mice. Nat. Commun. 2021, 12, 5569. [Google Scholar] [CrossRef] [PubMed]
- Santos-Sacchi, J.; Tan, W. Complex Nonlinear Capacitance in Outer Hair Cell Macro-Patches: Effects of Membrane Tension. Sci. Rep. 2020, 10, 6222. [Google Scholar] [CrossRef]
- Kotikova, M.R.; Chizhov, A.V. Hyperbolic Equations for Neuronal Membrane Deformation Waves Accompanying an Action Potential. Biochem. Biophys. Res. Commun. 2022, 591, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Gholami Derami, H.; Gupta, P.; Weng, K.; Seth, A.; Gupta, R.; Silva, J.R.; Raman, B.; Singamaneni, S. Reversible Photothermal Modulation of Electrical Activity of Excitable Cells Using Polydopamine Nanoparticles. Adv. Mater. 2021, 33, 2008809. [Google Scholar] [CrossRef] [PubMed]
- Pappas, T.C.; Wickramanyake, W.M.S.; Jan, E.; Motamedi, M.; Brodwick, M.; Kotov, N.A. Nanoscale Engineering of a Cellular Interface with Semiconductor Nanoparticle Films for Photoelectric Stimulation of Neurons. Nano Lett. 2007, 7, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Lugo, K.; Miao, X.; Rieke, F.; Lin, L.Y. Remote Switching of Cellular Activity and Cell Signaling Using Light in Conjunction with Quantum Dots. Biomed. Opt. Express 2012, 3, 447–454. [Google Scholar] [CrossRef]
- Bahmani Jalali, H.; Mohammadi Aria, M.; Dikbas, U.M.; Sadeghi, S.; Ganesh Kumar, B.; Sahin, M.; Kavakli, I.H.; Ow-Yang, C.W.; Nizamoglu, S. Effective Neural Photostimulation Using Indium-Based Type-II Quantum Dots. ACS Nano 2018, 12, 8104–8114. [Google Scholar] [CrossRef]
- Bareket, L.; Waiskopf, N.; Rand, D.; Lubin, G.; David-Pur, M.; Ben-Dov, J.; Roy, S.; Eleftheriou, C.; Sernagor, E.; Cheshnovsky, O.; et al. Semiconductor Nanorod–Carbon Nanotube Biomimetic Films for Wire-Free Photostimulation of Blind Retinas. Nano Lett. 2014, 14, 6685–6692. [Google Scholar] [CrossRef]
- Guduru, R.; Liang, P.; Hong, J.; Rodzinski, A.; Hadjikhani, A.; Horstmyer, J.; Levister, E.; Khizroev, S. Magnetoelectric ‘Spin’ on Stimulating the Brain. Nanomedicine 2015, 10, 2051–2061. [Google Scholar] [CrossRef]
- Marino, A.; Arai, S.; Hou, Y.; Sinibaldi, E.; Pellegrino, M.; Chang, Y.-T.; Mazzolai, B.; Mattoli, V.; Suzuki, M.; Ciofani, G. Piezoelectric Nanoparticle-Assisted Wireless Neuronal Stimulation. ACS Nano 2015, 9, 7678–7689. [Google Scholar] [CrossRef]
- McLaughlin, S. The Electrostatic Properties of Membranes. Annu. Rev. Biophys. Biophys. Chem. 1989, 18, 113–136. [Google Scholar] [CrossRef]
- Peterka, D.S.; Takahashi, H.; Yuste, R. Imaging Voltage in Neurons. Neuron 2011, 69, 9–21. [Google Scholar] [CrossRef]
- Dhandayuthapani, B.; Poulose, A.C.; Nagaoka, Y.; Hasumura, T.; Yoshida, Y.; Maekawa, T.; Kumar, D.S. Biomimetic Smart Nanocomposite: In Vitro Biological Evaluation of Zein Electrospun Fluorescent Nanofiber Encapsulated CdS Quantum Dots. Biofabrication 2012, 4, 25008. [Google Scholar] [CrossRef] [PubMed]
- Wei, G.; Keller, T.F.; Zhang, J.; Jandt, K.D. Novel 1-D Biophotonic Nanohybrids: Protein Nanofibers Meet Quantum Dots. Soft Matter 2011, 7, 2011. [Google Scholar] [CrossRef]
- Charras, G.; Sahai, E. Physical Influences of the Extracellular Environment on Cell Migration. Nat. Rev. Mol. Cell Biol. 2014, 15, 813–824. [Google Scholar] [CrossRef] [PubMed]
- Sapir, Y.; Cohen, S.; Friedman, G.; Polyak, B. The Promotion of in Vitro Vessel-like Organization of Endothelial Cells in Magnetically Responsive Alginate Scaffolds. Biomaterials 2012, 33, 4100–4109. [Google Scholar] [CrossRef]
- Beebe, D.J.; Moore, J.S.; Bauer, J.M.; Yu, Q.; Liu, R.H.; Devadoss, C.; Jo, B.H. Functional Hydrogel Structures for Autonomous Flow Control inside Microfluidic Channels. Nature 2000, 404, 588–590. [Google Scholar] [CrossRef]
- Yoshikawa, H.Y.; Rossetti, F.F.; Kaufmann, S.; Kaindl, T.; Madsen, J.; Engel, U.; Lewis, A.L.; Armes, S.P.; Tanaka, M. Quantitative Evaluation of Mechanosensing of Cells on Dynamically Tunable Hydrogels. J. Am. Chem. Soc. 2011, 133, 1367–1374. [Google Scholar] [CrossRef]
- Chandorkar, Y.; Castro Nava, A.; Schweizerhof, S.; van Dongen, M.; Haraszti, T.; Köhler, J.; Zhang, H.; Windoffer, R.; Mourran, A.; Möller, M.; et al. Cellular Responses to Beating Hydrogels to Investigate Mechanotransduction. Nat. Commun. 2019, 10, 4027. [Google Scholar] [CrossRef]
- Kloxin, A.M.; Kasko, A.M.; Salinas, C.N.; Anseth, K.S. Photodegradable Hydrogels for Dynamic Tuning of Physical and Chemical Properties. Science 2009, 324, 59–63. [Google Scholar] [CrossRef]
- Li, L.; Scheiger, J.M.; Levkin, P.A. Design and Applications of Photoresponsive Hydrogels. Adv. Mater. 2019, 31, 1807333. [Google Scholar] [CrossRef]
- Lin, D.C.; Yurke, B.; Langrana, N.A. Mechanical Properties of a Reversible, DNA-Crosslinked Polyacrylamide Hydrogel. J. Biomech. Eng. 2004, 126, 104–110. [Google Scholar] [CrossRef]
- Kabb, C.P.; O’Bryan, C.S.; Deng, C.C.; Angelini, T.E.; Sumerlin, B.S. Photoreversible Covalent Hydrogels for Soft-Matter Additive Manufacturing. ACS Appl. Mater. Interfaces 2018, 10, 16793–16801. [Google Scholar] [CrossRef]
- Wu, X.; Huang, W.; Wu, W.-H.; Xue, B.; Xiang, D.; Li, Y.; Qin, M.; Sun, F.; Wang, W.; Zhang, W.-B.; et al. Reversible Hydrogels with Tunable Mechanical Properties for Optically Controlling Cell Migration. Nano Res. 2018, 11, 5556–5565. [Google Scholar] [CrossRef]
- Kim, P.; Zarzar, L.D.; He, X.; Grinthal, A.; Aizenberg, J. Hydrogel-Actuated Integrated Responsive Systems (HAIRS): Moving towards Adaptive Materials. Curr. Opin. Solid State Mater. Sci. 2011, 15, 236–245. [Google Scholar] [CrossRef]
- Sutton, A.; Shirman, T.; Timonen, J.V.I.; England, G.T.; Kim, P.; Kolle, M.; Ferrante, T.; Zarzar, L.D.; Strong, E.; Aizenberg, J. Photothermally Triggered Actuation of Hybrid Materials as a New Platform for in Vitro Cell Manipulation. Nat. Commun. 2017, 8, 14700. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Lu, J.Q. Optothermally Responsive Nanocomposite Generating Mechanical Forces for Cells Enabled by Few-Walled Carbon Nanotubes. ACS Nano 2014, 8, 11695–11706. [Google Scholar] [CrossRef]
- Liu, Z.; Liu, Y.; Chang, Y.; Seyf, H.R.; Henry, A.; Mattheyses, A.L.; Yehl, K.; Zhang, Y.; Huang, Z.; Salaita, K. Nanoscale Optomechanical Actuators for Controlling Mechanotransduction in Living Cells. Nat. Methods 2016, 13, 143–146. [Google Scholar] [CrossRef]
- Ramey-Ward, A.N.; Su, H.; Salaita, K. Mechanical Stimulation of Adhesion Receptors Using Light-Responsive Nanoparticle Actuators Enhances Myogenesis. ACS Appl. Mater. Interfaces 2020, 12, 35903–35917. [Google Scholar] [CrossRef]
- Yu, W.; Deschaume, O.; Dedroog, L.; Garcia Abrego, C.J.; Zhang, P.; Wellens, J.; de Coene, Y.; Jooken, S.; Clays, K.; Thielemans, W.; et al. Light-addressable Nanocomposite Hydrogels Allow Plasmonic Actuation and in Situ Temperature Monitoring in 3D Cell Matrices. Adv. Funct. Mater. 2022, 32, 2108234. [Google Scholar] [CrossRef]
- Kojima, C.; Nakajima, Y.; Oeda, N.; Kawano, T.; Taki, Y. Visible Laser-Induced in Situ Cell Detachment from Gold Nanoparticle-Embedded Collagen Gel. Macromol. Biosci. 2017, 17, 1600341. [Google Scholar] [CrossRef]
- Cui, H.; Zhang, P.; Wang, W.; Li, G.; Hao, Y.; Wang, L.; Wang, S. Near-Infrared (NIR) Controlled Reversible Cell Adhesion on a Responsive Nano-Biointerface. Nano Res. 2017, 10, 1345–1355. [Google Scholar] [CrossRef]
- Tang, Y.; Wu, C.; Wu, Z.; Hu, L.; Zhang, W.; Zhao, K. Fabrication and in Vitro Biological Properties of Piezoelectric Bioceramics for Bone Regeneration. Sci. Rep. 2017, 7, 43360. [Google Scholar] [CrossRef] [PubMed]
- Amna, T.; Hassan, M.S.; Sheikh, F.A.; Lee, H.K.; Seo, K.-S.; Yoon, D.; Hwang, I.H. Zinc Oxide-Doped Poly(Urethane) Spider Web Nanofibrous Scaffold via One-Step Electrospinning: A Novel Matrix for Tissue Engineering. Appl. Microbiol. Biotechnol. 2013, 97, 1725–1734. [Google Scholar] [CrossRef] [PubMed]
- Alshehri, A.M.; Wilson, O.C.; Dahal, B.; Philip, J.; Luo, X.; Raub, C.B. Magnetic Nanoparticle-Loaded Alginate Beads for Local Micro-Actuation of in Vitro Tissue Constructs. Colloids Surf. B Biointerfaces 2017, 159, 945–955. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.J.; Singh, R.K.; Seo, S.J.; Kim, T.H.; Kim, J.H.; Lee, E.J.; Kim, H.W. Magnetic Scaffolds of Polycaprolactone with Functionalized Magnetite Nanoparticles: Physicochemical, Mechanical, and Biological Properties Effective for Bone Regeneration. RSC Adv. 2014, 4, 17325–17336. [Google Scholar] [CrossRef]
- Yuan, Z.; Memarzadeh, K.; Stephen, A.S.; Allaker, R.P.; Brown, R.A.; Huang, J. Development of a 3D Collagen Model for the in Vitro Evaluation of Magnetic-Assisted Osteogenesis. Sci. Rep. 2018, 8, 16270. [Google Scholar] [CrossRef] [PubMed]
- Bañobre-López, M.; Piñeiro-Redondo, Y.; de Santis, R.; Gloria, A.; Ambrosio, L.; Tampieri, A.; Dediu, V.; Rivas, J. Poly(Caprolactone) Based Magnetic Scaffolds for Bone Tissue Engineering. J. Appl. Phys. 2011, 109, 7B313. [Google Scholar] [CrossRef]
- Herrera-Posada, S.; Mora-Navarro, C.; Ortiz-Bermudez, P.; Torres-Lugo, M.; McElhinny, K.M.; Evans, P.G.; Calcagno, B.O.; Acevedo, A. Magneto-Responsive Liquid Crystalline Elastomer Nanocomposites as Potential Candidates for Dynamic Cell Culture Substrates. Mater. Sci. Eng. C 2016, 65, 369–378. [Google Scholar] [CrossRef]
- Chen, W.; Zhang, Y.; Kumari, J.; Engelkamp, H.; Kouwer, P.H.J. Magnetic Stiffening in 3D Cell Culture Matrices. Nano Lett. 2021, 21, 6740–6747. [Google Scholar] [CrossRef]
- Yun, H.-M.; Ahn, S.-J.; Park, K.-R.; Kim, M.-J.; Kim, J.-J.; Jin, G.-Z.; Kim, H.-W.; Kim, E.-C. Magnetic Nanocomposite Scaffolds Combined with Static Magnetic Field in the Stimulation of Osteoblastic Differentiation and Bone Formation. Biomaterials 2016, 85, 88–98. [Google Scholar] [CrossRef]
- Cezar, C.A.; Kennedy, S.M.; Mehta, M.; Weaver, J.C.; Gu, L.; Vandenburgh, H.; Mooney, D.J. Biphasic Ferrogels for Triggered Drug and Cell Delivery. Adv. Healthc. Mater. 2014, 3, 1869–1876. [Google Scholar] [CrossRef]
- Gonçalves, A.I.; Rodrigues, M.T.; Carvalho, P.P.; Bañobre-López, M.; Paz, E.; Freitas, P.; Gomes, M.E. Exploring the Potential of Starch/Polycaprolactone Aligned Magnetic Responsive Scaffolds for Tendon Regeneration. Adv. Healthc. Mater. 2016, 5, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Wong, D.S.H.; Li, J.; Yan, X.; Wang, B.; Li, R.; Zhang, L.; Bian, L. Magnetically Tuning Tether Mobility of Integrin Ligand Regulates Adhesion, Spreading, and Differentiation of Stem Cells. Nano Lett. 2017, 17, 1685–1695. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Chen, G.; Xu, X.; Abdou, P.; Jiang, Q.; Shi, D.; Gu, Z. Advances of Injectable Hydrogel-Based Scaffolds for Cartilage Regeneration. Regen. Biomater. 2019, 6, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Abdollahiyan, P.; Oroojalian, F.; Hejazi, M.; de la Guardia, M.; Mokhtarzadeh, A. Nanotechnology, and Scaffold Implantation for the Effective Repair of Injured Organs: An Overview on Hard Tissue Engineering. J. Control. Release 2021, 333, 391–417. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Cheng, Y.; Cai, J.; Zhao, X.; Ouyang, Y.; Yuan, W.-E.; Fan, C. Advances in Electrical and Magnetic Stimulation on Nerve Regeneration. Regen. Med. 2019, 14, 969–979. [Google Scholar] [CrossRef] [PubMed]
- Tay, A.; di Carlo, D. Magnetic Nanoparticle-Based Mechanical Stimulation for Restoration of Mechano-Sensitive Ion Channel Equilibrium in Neural Networks. Nano Lett. 2017, 17, 886–892. [Google Scholar] [CrossRef]
- Hughes, S.; McBain, S.; Dobson, J.; el Haj, A.J. Selective Activation of Mechanosensitive Ion Channels Using Magnetic Particles. J. R. Soc. Interface 2008, 5, 855–863. [Google Scholar] [CrossRef]
- Hughes, S.; el Haj, A.J.; Dobson, J. Magnetic Micro- and Nanoparticle Mediated Activation of Mechanosensitive Ion Channels. Med. Eng. Phys. 2005, 27, 754–762. [Google Scholar] [CrossRef]
- Tay, A.; Sohrabi, A.; Poole, K.; Seidlits, S.; di Carlo, D. A 3D Magnetic Hyaluronic Acid Hydrogel for Magnetomechanical Neuromodulation of Primary Dorsal Root Ganglion Neurons. Adv. Mater. 2018, 30, 1800927. [Google Scholar] [CrossRef]
- Kanczler, J.M.; Sura, H.S.; Magnay, J.; Green, D.; Oreffo, R.O.C.; Dobson, J.P.; el Haj, A.J. Controlled Differentiation of Human Bone Marrow Stromal Cells Using Magnetic Nanoparticle Technology. Tissue Eng. Part A 2010, 16, 3241–3250. [Google Scholar] [CrossRef]
- Chouhan, D.; Mehrotra, S.; Majumder, O.; Mandal, B.B. Magnetic Actuator Device Assisted Modulation of Cellular Behavior and Tuning of Drug Release on Silk Platform. ACS Biomater. Sci. Eng. 2019, 5, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Chung, S.E.; Choi, S.-E.; Lee, H.; Kim, J.; Kwon, S. Programming Magnetic Anisotropy in Polymeric Microactuators. Nat. Mater. 2011, 10, 747–752. [Google Scholar] [CrossRef] [PubMed]
- Lind, J.U.; Busbee, T.A.; Valentine, A.D.; Pasqualini, F.S.; Yuan, H.; Yadid, M.; Park, S.-J.; Kotikian, A.; Nesmith, A.P.; Campbell, P.H.; et al. Instrumented Cardiac Microphysiological Devices via Multimaterial Three-Dimensional Printing. Nat. Mater. 2017, 16, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Shandilya, R.; Bhargava, A.; Bunkar, N.; Tiwari, R.; Goryacheva, I.Y.; Mishra, P.K. Nanobiosensors: Point-of-Care Approaches for Cancer Diagnostics. Biosens. Bioelectron. 2019, 130, 147–165. [Google Scholar] [CrossRef]
- Xianyu, Y.; Wang, Q.; Chen, Y. Magnetic Particles-Enabled Biosensors for Point-of-Care Testing. TrAC Trends Anal. Chem. 2018, 106, 213–224. [Google Scholar] [CrossRef]
- Islam, T.; Hasan, M.M.; Awal, A.; Nurunnabi, M.; Ahammad, A.J.S. Metal Nanoparticles for Electrochemical Sensing: Progress and Challenges in the Clinical Transition of Point-of-Care Testing. Molecules 2020, 25, 5787. [Google Scholar] [CrossRef]
- Geng, H.; Vilms Pedersen, S.; Ma, Y.; Haghighi, T.; Dai, H.; Howes, P.D.; Stevens, M.M. Noble Metal, Anoparticle Biosensors: From Fundamental Studies toward Point-of-Care Diagnostics. Acc. Chem. Res. 2022, 55, 593–604. [Google Scholar] [CrossRef]
- Fedi, A.; Vitale, C.; Giannoni, P.; Caluori, G.; Marrella, A. Biosensors to Monitor Cell Activity in 3D Hydrogel-Based Tissue Models. Sensors 2022, 22, 1517. [Google Scholar] [CrossRef]
- Farrell, M.J.; Shin, J.I.; Smith, L.J.; Mauck, R.L. Functional Consequences of Glucose and Oxygen Deprivation Onengineered Mesenchymal Stem Cell-Based Cartilage Constructs. Osteoarthr. Cartil. 2015, 23, 134–142. [Google Scholar] [CrossRef]
- Zirath, H.; Rothbauer, M.; Spitz, S.; Bachmann, B.; Jordan, C.; Müller, B.; Ehgartner, J.; Priglinger, E.; Mühleder, S.; Redl, H.; et al. Every Breath You Take: Non-Invasive Real-Time Oxygen Biosensing in Two- and Three-Dimensional Microfluidic Cell Models. Front. Physiol. 2018, 9, 815. [Google Scholar] [CrossRef]
- Tauffenberger, A.; Magistretti, P.J. Reactive Oxygen Species: Beyond Their Reactive Behavior. Neurochem. Res. 2021, 46, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Hensley, K.; Robinson, K.A.; Gabbita, S.P.; Salsman, S.; Floyd, R.A. Reactive Oxygen Species, Cell Signaling, and Cell Injury. Free Radic. Biol. Med. 2000, 28, 1456–1462. [Google Scholar] [CrossRef] [PubMed]
- Crulhas, B.P.; Recco, L.C.; Delella, F.K.; Pedrosa, V.A. A Novel Superoxide Anion Biosensor for Monitoring Reactive Species of Oxygen Released by Cancer Cells. Electroanalysis 2017, 29, 1252–1257. [Google Scholar] [CrossRef]
- Ma, Z.; Luo, Y.; Zhu, Q.; Jiang, M.; Pan, M.; Xie, T.; Huang, X.; Chen, D. In-Situ Monitoring of Glucose Metabolism in Cancer Cell Microenvironments Based on Hollow Fiber Structure. Biosens. Bioelectron. 2020, 162, 112261. [Google Scholar] [CrossRef]
- Hu, N.; Wu, C.; Ha, D.; Wang, T.; Liu, Q.; Wang, P. A Novel Microphysiometer Based on High Sensitivity LAPS and Microfluidic System for Cellular Metabolism Study and Rapid Drug Screening. Biosens. Bioelectron. 2013, 40, 167–173. [Google Scholar] [CrossRef]
- Lehmann, M.; Baumann, W.; Brischwein, M.; Gahle, H.J.; Freund, I.; Ehret, R.; Drechsler, S.; Palzer, H.; Kleintges, M.; Sieben, U.; et al. Simultaneous Measurement of Cellular Respiration and Acidification with a Single CMOS ISFET. Biosens. Bioelectron. 2001, 16, 195–203. [Google Scholar] [CrossRef]
- Yang, C.-M.; Yen, T.; Liu, H.-L.; Lin, Y.-J.; Lin, P.-Y.; Tsui, L.S.; Chen, C.-H.; Chen, Y.-P.; Hsu, Y.-C.; Lo, C.-H.; et al. A Real-Time Mirror-LAPS Mini System for Dynamic Chemical Imaging and Cell Acidification Monitoring. Sens. Actuators B Chem. 2021, 341, 130003. [Google Scholar] [CrossRef]
- Figueiredo, L.; Pace, R.; D’Arros, C.; Réthoré, G.; Guicheux, J.; le Visage, C.; Weiss, P. Assessing Glucose and Oxygen Diffusion in Hydrogels for the Rational Design of 3D Stem Cell Scaffolds in Regenerative Medicine. J. Tissue Eng. Regen. Med. 2018, 12, 1238–1246. [Google Scholar] [CrossRef]
- Bavli, D.; Prill, S.; Ezra, E.; Levy, G.; Cohen, M.; Vinken, M.; Vanfleteren, J.; Jaeger, M.; Nahmias, Y. Real-Time Monitoring of Metabolic Function in Liver-on-Chip Microdevices Tracks the Dynamics of Mitochondrial Dysfunction. Proc. Natl. Acad. Sci. USA 2016, 113, E2231–E2240. [Google Scholar] [CrossRef]
- Shaibani, P.M.; Etayash, H.; Naicker, S.; Kaur, K.; Thundat, T. Metabolic Study of Cancer Cells Using a PH Sensitive Hydrogel Nanofiber Light Addressable Potentiometric Sensor. ACS Sens. 2017, 2, 151–156. [Google Scholar] [CrossRef]
- Pan, Y.; Hu, N.; Wei, X.; Gong, L.; Zhang, B.; Wan, H.; Wang, P. 3D Cell-Based Biosensor for Cell Viability and Drug Assessment by 3D Electric Cell/Matrigel-Substrate Impedance Sensing. Biosens. Bioelectron. 2019, 130, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Lei, K.F.; Wu, Z.-M.; Huang, C.-H. Impedimetric Quantification of the Formation Process and the Chemosensitivity of Cancer Cell Colonies Suspended in 3D Environment. Biosens. Bioelectron. 2015, 74, 878–885. [Google Scholar] [CrossRef]
- Wu, R.; Li, L.; Pan, L.; Yan, K.; Shi, Y.; Jiang, L.; Zhu, J.-J. Long-Term Cell Culture and Electrically in Situ Monitoring of Living Cells Based on a Polyaniline Hydrogel Sensor. J. Mater. Chem. B 2021, 9, 9514–9523. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-W.; Hu, X.-B.; Qin, Y.; Jin, Z.-H.; Zhang, X.-W.; Liu, Y.-L.; Huang, W.-H. Conductive Polymer Coated Scaffold to Integrate 3D Cell Culture with Electrochemical Sensing. Anal. Chem. 2019, 91, 4838–4844. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xie, J.; Gao, L.; Li, C.M. Au Nanoparticles–3D Graphene Hydrogel Nanocomposite to Boost Synergistically in Situ Detection Sensitivity toward Cell-Released Nitric Oxide. ACS Appl. Mater. Interfaces 2015, 7, 2726–2734. [Google Scholar] [CrossRef] [PubMed]
- Manickam, P.; Vashist, A.; Madhu, S.; Sadasivam, M.; Sakthivel, A.; Kaushik, A.; Nair, M. Gold Nanocubes Embedded Biocompatible Hybrid Hydrogels for Electrochemical Detection of H2O2. Bioelectrochemistry 2020, 131, 107373. [Google Scholar] [CrossRef] [PubMed]
- Trampe, E.; Koren, K.; Akkineni, A.R.; Senwitz, C.; Krujatz, F.; Lode, A.; Gelinsky, M.; Kühl, M. Functionalized Bioink with Optical Sensor Nanoparticles for O2 Imaging in 3D-Bioprinted Constructs. Adv. Funct. Mater. 2018, 28, 1804411. [Google Scholar] [CrossRef]
- Lemon, C.M.; Karnas, E.; Han, X.; Bruns, O.T.; Kempa, T.J.; Fukumura, D.; Bawendi, M.G.; Jain, R.K.; Duda, D.G.; Nocera, D.G. Micelle-Encapsulated Quantum Dot-Porphyrin Assemblies as in Vivo Two-Photon Oxygen Sensors. J. Am. Chem. Soc. 2015, 137, 9832–9842. [Google Scholar] [CrossRef]
- Rivera, K.R.; Pozdin, V.A.; Young, A.T.; Erb, P.D.; Wisniewski, N.A.; Magness, S.T.; Daniele, M. Integrated Phosphorescence-Based Photonic Biosensor (IPOB) for Monitoring Oxygen Levels in 3D Cell Culture Systems. Biosens. Bioelectron. 2019, 123, 131–140. [Google Scholar] [CrossRef]
- Koduri, M.P.S.; Goudar, V.; Shao, Y.-W.; Hunt, J.A.; Henstock, J.R.; Curran, J.; Tseng, F.G. Oxygen Particles for Spatiometric Monitoring of Cell Physiological Conditions. ACS Appl. Mater. Interfaces 2018, 10, 30163–30171. [Google Scholar] [CrossRef]
- Dmitriev, R.I.; Borisov, S.M.; Kondrashina, A.V.; Pakan, J.M.P.; Anilkumar, U.; Prehn, J.H.M.; Zhdanov, A.V.; McDermott, K.W.; Klimant, I.; Papkovsky, D.B. Imaging Oxygen in Neural Cell and Tissue Models by Means of Anionic Cell-Permeable Phosphorescent Nanoparticles. Cell. Mol. Life Sci. 2015, 72, 367–381. [Google Scholar] [CrossRef] [PubMed]
- Wolfbeis, O.S. Luminescent Sensing and Imaging of Oxygen: Fierce Competition to the Clark Electrode. BioEssays 2015, 37, 921–928. [Google Scholar] [CrossRef] [PubMed]
- Moldero, I.L.; Chandra, A.; Cavo, M.; Mota, C.; Kapsokalyvas, D.; Gigli, G.; Moroni, L.; del Mercato, L.L. Probing the PH Microenvironment of Mesenchymal Stromal Cell Cultures on Additive-Manufactured Scaffolds. Small 2020, 16, e2002258. [Google Scholar] [CrossRef] [PubMed]
- Kenney, R.M.; Boyce, M.W.; Whitman, N.A.; Kromhout, B.P.; Lockett, M.R. A PH-Sensing Optode for Mapping Spatiotemporal Gradients in 3D Paper-Based Cell Cultures. Anal. Chem. 2018, 90, 2376–2383. [Google Scholar] [CrossRef] [PubMed]
- Koo Lee, Y.-E.; Ulbrich, E.E.; Kim, G.; Hah, H.; Strollo, C.; Fan, W.; Gurjar, R.; Koo, S.; Kopelman, R. Near Infrared Luminescent Oxygen Nanosensors with Nanoparticle Matrix Tailored Sensitivity. Anal. Chem. 2010, 82, 8446–8455. [Google Scholar] [CrossRef]
- Acosta, M.A.; Ymele-Leki, P.; Kostov, Y.V.; Leach, J.B. Fluorescent Microparticles for Sensing Cell Microenvironment Oxygen Levels within 3D Scaffolds. Biomaterials 2009, 30, 3068–3074. [Google Scholar] [CrossRef]
- Jooken, S.; de Coene, Y.; Deschaume, O.; Zámbó, D.; Aubert, T.; Hens, Z.; Dorfs, D.; Verbiest, T.; Clays, K.; Callewaert, G.; et al. Enhanced Electric Field Sensitivity of Quantum Dot/Rod Two-Photon Fluorescence and Its Relevance for Cell Transmembrane Voltage Imaging. Nanophotonics 2021, 10, 2407–2420. [Google Scholar] [CrossRef]
- Park, C.H.; Lee, S.; Pornnoppadol, G.; Nam, Y.S.; Kim, S.-H.; Kim, B.J. Microcapsules Containing PH-Responsive, Fluorescent Polymer-Integrated MoS2: An Effective Platform for in Situ PH Sensing and Photothermal Heating. ACS Appl. Mater. Interfaces 2018, 10, 9023–9031. [Google Scholar] [CrossRef]
- Chu, B.; Wang, H.; Song, B.; Peng, F.; Su, Y.; He, Y. Fluorescent and Photostable Silicon Nanoparticles Sensors for Real-Time and Long-Term Intracellular PH Measurement in Live Cells. Anal. Chem. 2016, 88, 9235–9242. [Google Scholar] [CrossRef]
- Gomes, A.; Fernandes, E.; Lima, J.L.F.C. Fluorescence Probes Used for Detection of Reactive Oxygen Species. J. Biochem. Biophys. Methods 2005, 65, 45–80. [Google Scholar] [CrossRef]
- Kundu, K.; Knight, S.F.; Willett, N.; Lee, S.; Taylor, W.R.; Murthy, N. Hydrocyanines: A Class of Fluorescent Sensors That Can Image Reactive Oxygen Species in Cell Culture, Tissue, and in Vivo. Angew. Chem. Int. Ed. 2009, 48, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Setsukinai, K.I.; Urano, Y.; Kakinuma, K.; Majima, H.J.; Nagano, T. Development of Novel Fluorescence Probes That Can Reliably Detect Reactive Oxygen Species and Distinguish Specific Species. J. Biol. Chem. 2003, 278, 3170–3175. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.; Lee, Y.-E.K.; Xu, H.; Philbert, M.A.; Kopelman, R. Nanoencapsulation Method for High Selectivity Sensing of Hydrogen Peroxide inside Live Cells. Anal. Chem. 2010, 82, 2165–2169. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Khaja, S.; Velasquez-Castano, J.C.; Dasari, M.; Sun, C.; Petros, J.; Taylor, W.R.; Murthy, N. In Vivo Imaging of Hydrogen Peroxide with Chemiluminescent Nanoparticles. Nat. Mater. 2007, 6, 765–769. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Acosta, M.A.; Leach, J.B.; Carrier, R.L. Spatially Monitoring Oxygen Level in 3D Microfabricated Cell Culture Systems Using Optical Oxygen Sensing Beads. Lab Chip 2013, 13, 1586. [Google Scholar] [CrossRef]
- Skylaki, S.; Hilsenbeck, O.; Schroeder, T. Challenges in Long-Term Imaging and Quantification of Single-Cell Dynamics. Nat. Biotechnol. 2016, 34, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Icha, J.; Weber, M.; Waters, J.C.; Norden, C. Phototoxicity in Live Fluorescence Microscopy, and How to Avoid It. BioEssays 2017, 39, 1700003. [Google Scholar] [CrossRef]
- Chandra, A.; Singh, N. Cell Microenvironment Ph Sensing in 3D Microgels Using Fluorescent Carbon Dots. ACS Biomater. Sci. Eng. 2017, 3, 3620–3627. [Google Scholar] [CrossRef]
- Tsai, E.S.; Himmelstoß, S.F.; Wiesholler, L.M.; Hirsch, T.; Hall, E.A.H. Upconversion Nanoparticles for Sensing PH. Analyst 2019, 144, 5547–5557. [Google Scholar] [CrossRef]
- Liu, J.; Liu, Y.; Bu, W.; Bu, J.; Sun, Y.; Du, J.; Shi, J. Ultrasensitive Nanosensors Based on Upconversion Nanoparticles for Selective Hypoxia Imaging in Vivo upon Near-Infrared Excitation. J. Am. Chem. Soc. 2014, 136, 9701–9709. [Google Scholar] [CrossRef]
- Acosta, M.A.; Velasquez, M.; Williams, K.; Ross, J.M.; Leach, J.B. Fluorescent Silica Particles for Monitoring Oxygen Levels in Three-Dimensional Heterogeneous Cellular Structures. Biotechnol. Bioeng. 2012, 109, 2663–2670. [Google Scholar] [CrossRef]
- Tang, B.; Zhang, N.; Chen, Z.; Xu, K.; Zhuo, L.; An, L.; Yang, G. Probing Hydroxyl Radicals and Their Imaging in Living Cells by Use of FAM–DNA–Au Nanoparticles. Chem. Eur. J. 2008, 14, 522–528. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Li, Y.; Jooken, S.; Deschaume, O.; Liu, F.; Wang, S.; Bartic, C. Second-Order Optimized Regularized Structured Illumination Microscopy (SorSIM) for High-Quality and Rapid Super Resolution Image Reconstruction with Low Signal Level. Opt. Express 2020, 28, 16708–16724. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Hasan, A.; Attar, F.; Sharifi, M.; Siddique, R.; Mraiche, F.; Falahati, M. Gold Nanoparticle-Based Platforms for Diagnosis and Treatment of Myocardial Infarction. ACS Biomater. Sci. Eng. 2020, 6, 6460–6477. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Mehrotra, S.; Zare-Eelanjegh, E.; Rodrigues, R.O.; Akbarinejad, A.; Ge, D.; Amato, L.; Kiaee, K.; Fang, Y.; Rosenkranz, A.; et al. A Heart-breast Cancer-on-a-chip Platform for Disease Modeling and Monitoring of Cardiotoxicity Induced by Cancer Chemotherapy. Small 2021, 17, 2004258. [Google Scholar] [CrossRef] [PubMed]
- Berthuy, O.; Blum, L.; Marquette, C. Cancer-Cells on Chip for Label-Free Detection of Secreted Molecules. Biosensors 2016, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.S.; Aleman, J.; Shin, S.R.; Kilic, T.; Kim, D.; Mousavi Shaegh, S.A.; Massa, S.; Riahi, R.; Chae, S.; Hu, N.; et al. Multisensor-Integrated Organs-on-Chips Platform for Automated and Continual in Situ Monitoring of Organoid Behaviors. Proc. Natl. Acad. Sci. USA 2017, 114, E2293–E2302. [Google Scholar] [CrossRef] [PubMed]
- Ortega, M.A.; Fernández-Garibay, X.; Castaño, A.G.; de Chiara, F.; Hernández-Albors, A.; Balaguer-Trias, J.; Ramón-Azcón, J. Muscle-on-a-Chip with an on-Site Multiplexed Biosensing System for in Situ Monitoring of Secreted IL-6 and TNF-α. Lab Chip 2019, 19, 2568–2580. [Google Scholar] [CrossRef]
- Son, K.J.; Rahimian, A.; Shin, D.-S.; Siltanen, C.; Patel, T.; Revzin, A. Microfluidic Compartments with Sensing Microbeads for Dynamic Monitoring of Cytokine and Exosome Release from Single Cells. Analyst 2016, 141, 679–688. [Google Scholar] [CrossRef]
- Matta, D.P.; Tripathy, S.; Krishna Vanjari, S.R.; Sharma, C.S.; Singh, S.G. An Ultrasensitive Label Free Nanobiosensor Platform for the Detection of Cardiac Biomarkers. Biomed. Microdevices 2016, 18, 111. [Google Scholar] [CrossRef]
- Rezaei, Z.; Ranjbar, B. Ultra-Sensitive, Rapid Gold Nanoparticle-Quantum Dot Plexcitonic Self-Assembled Aptamer-Based Nanobiosensor for the Detection of Human Cardiac Troponin I. Eng. Life Sci. 2017, 17, 165–174. [Google Scholar] [CrossRef]
- Kratz, S.; Höll, G.; Schuller, P.; Ertl, P.; Rothbauer, M. Latest Trends in Biosensing for Microphysiological Organs-on-a-Chip and Body-on-a-Chip Systems. Biosensors 2019, 9, 110. [Google Scholar] [CrossRef]
- Plou, J.; Molina-Martínez, B.; García-Astrain, C.; Langer, J.; García, I.; Ercilla, A.; Perumal, G.; Carracedo, A.; Liz-Marzán, L.M. Nanocomposite Scaffolds for Monitoring of Drug Diffusion in Three-Dimensional Cell Environments by Surface-Enhanced Raman Spectroscopy. Nano Lett. 2021, 21, 8785–8793. [Google Scholar] [CrossRef]
- García-Astrain, C.; Lenzi, E.; Jimenez de Aberasturi, D.; Henriksen-Lacey, M.; Binelli, M.R.; Liz-Marzán, L.M. 3D-printed Biocompatible Scaffolds with Built-in Nanoplasmonic Sensors. Adv. Funct. Mater. 2020, 30, 2005407. [Google Scholar] [CrossRef]
- Kim, T.H.; Lee, K.B.; Choi, J.W. 3D Graphene Oxide-Encapsulated Gold Nanoparticles to Detect Neural Stem Cell Differentiation. Biomaterials 2013, 34, 8660–8670. [Google Scholar] [CrossRef] [PubMed]
- El-Said, W.A.; Kim, S.U.; Choi, J.W. Monitoring in Vitro Neural Stem Cell Differentiation Based on Surface-Enhanced Raman Spectroscopy Using a Gold Nanostar Array. J. Mater. Chem. C Mater. 2015, 3, 3848–3859. [Google Scholar] [CrossRef]
- Kim, K.; Ahn, J.; Yoon, K.; Ko, M.; Ahn, J.; Kim, H.; Park, J.; Lee, C.; Chang, D.; Oh, S. In Vivo Magnetic Resonance Thermometry for Brain and Body Temperature Variations in Canines under General Anesthesia. Sensors 2022, 22, 4034. [Google Scholar] [CrossRef] [PubMed]
- Rieke, V.; Pauly, K.B. MR Thermometry. J. Magn. Reson. Imaging 2008, 27, 376–390. [Google Scholar] [CrossRef] [PubMed]
- Hankiewicz, J.H.; Stoll, J.A.; Stroud, J.; Davidson, J.; Livesey, K.L.; Tvrdy, K.; Roshko, A.; Russek, S.E.; Stupic, K.; Bilski, P.; et al. Nano-Sized Ferrite Particles for Magnetic Resonance Imaging Thermometry. J. Magn. Magn. Mater. 2019, 469, 550–557. [Google Scholar] [CrossRef]
- Kim, J.; Choi, W.; Park, E.Y.; Kang, Y.; Lee, K.J.; Kim, H.H.; Kim, W.J.; Kim, C. Real-Time Photoacoustic Thermometry Combined with Clinical Ultrasound Imaging and High-Intensity Focused Ultrasound. IEEE Trans. Biomed. Eng. 2019, 66, 3330–3338. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhu, W.; Hui, F.; Lanza, M.; Borca-Tasciuc, T.; Rojo, M.M.; Zhang, Y.; Zhu, W.; Hui, F.; Lanza, M.; et al. A Review on Principles and Applications of Scanning Thermal Microscopy (SThM). Adv. Funct. Mater. 2020, 30, 1900892. [Google Scholar] [CrossRef]
- Oh, J.; Lee, G.H.; Rho, J.; Shin, S.; Lee, B.J.; Nam, Y.; Park, Y. Optical Measurements of Three-Dimensional Microscopic Temperature Distributions Around Gold Nanorods Excited by Localized Surface Plasmon Resonance. Phys. Rev. Appl. 2019, 11, 44079. [Google Scholar] [CrossRef]
- Lu, J.; Ni, C.; Huang, J.; Liu, Y.; Tao, Y.; Hu, P.; Wang, Y.; Zheng, S.; Shi, M. Biocompatible Mesoporous Silica–Polydopamine Nanocomplexes as MR/Fluorescence Imaging Agent for Light-Activated Photothermal–Photodynamic Cancer Therapy in Vivo. Front. Bioeng. Biotechnol. 2021, 9, 752982. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; del Rosal, B.; Jaque, D.; Uchiyama, S.; Jin, D. Advances and Challenges for Fluorescence Nanothermometry. Nat. Methods 2020, 17, 967–980. [Google Scholar] [CrossRef] [PubMed]
- Jaque, D.; Vetrone, F. Luminescence Nanothermometry. Nanoscale 2012, 4, 4301. [Google Scholar] [CrossRef]
- Gota, C.; Okabe, K.; Funatsu, T.; Harada, Y.; Uchiyama, S. Hydrophilic Fluorescent Nanogel Thermometer for Intracellular Thermometry. J. Am. Chem. Soc. 2009, 131, 2766–2767. [Google Scholar] [CrossRef]
- Suzuki, M.; Tseeb, V.; Oyama, K.; Ishiwata, S. Microscopic Detection of Thermogenesis in a Single Hela Cell. Biophys. J. 2007, 92, L46–L48. [Google Scholar] [CrossRef]
- Ye, F.; Wu, C.; Jin, Y.; Chan, Y.-H.; Zhang, X.; Chiu, D.T. Ratiometric Temperature Sensing with Semiconducting Polymer Dots. J. Am. Chem. Soc. 2011, 133, 8146–8149. [Google Scholar] [CrossRef]
- Zhegalova, N.G.; Dergunov, S.A.; Wang, S.T.; Pinkhassik, E.; Berezin, M.Y. Design of Fluorescent Nanocapsules as Ratiometric Nanothermometers. Chem. Eur. J. 2014, 20, 10292–10297. [Google Scholar] [CrossRef]
- Hatanaka, M.; Hirai, Y.; Kitagawa, Y.; Nakanishi, T.; Hasegawa, Y.; Morokuma, K. Organic Linkers Control the Thermosensitivity of the Emission Intensities from Tb(iii) and Eu(iii) in a Chameleon Polymer. Chem. Sci. 2017, 8, 423–429. [Google Scholar] [CrossRef]
- Zhao, Y.; Shi, C.; Yang, X.; Shen, B.; Sun, Y.; Chen, Y.; Xu, X.; Sun, H.; Yu, K.; Yang, B.; et al. Ph- and Temperature-Sensitive Hydrogel Nanoparticles with Dual Photoluminescence for Bioprobes. ACS Nano 2016, 10, 5856–5863. [Google Scholar] [CrossRef] [PubMed]
- Iyisan, B.; Thiramanas, R.; Nazarova, N.; Avlasevich, Y.; Mailänder, V.; Baluschev, S.; Landfester, K. Temperature Sensing in Cells Using Polymeric Upconversion Nanocapsules. Biomacromolecules 2020, 21, 4469–4478. [Google Scholar] [CrossRef] [PubMed]
- Drabik, J.; Ledwa, K.; Marciniak, Ł. Implementing Defects for Ratiometric Luminescence Thermometry. Nanomaterials 2020, 10, 1333. [Google Scholar] [CrossRef] [PubMed]
- Runowski, M.; Shyichuk, A.; Tymiński, A.; Grzyb, T.; Lavín, V.; Lis, S. Multifunctional Optical Sensors for Nanomanometry and Nanothermometry: High-Pressure and High-Temperature Upconversion Luminescence of Lanthanide-Doped Phosphates—LaPO 4 /YPO 4 :Yb 3+ –Tm 3+. ACS Appl. Mater. Interfaces 2018, 10, 17269–17279. [Google Scholar] [CrossRef] [PubMed]
- Brites, C.D.S.; Balabhadra, S.; Carlos, L.D. Lanthanide-based Thermometers: At the Cutting-edge of Luminescence Thermometry. Adv. Opt. Mater. 2019, 7, 1801239. [Google Scholar] [CrossRef]
- Sekiguchi, T.; Sotoma, S.; Harada, Y. Fluorescent Nanodiamonds as a Robust Temperature Sensor inside a Single Cell. Biophys. Phys. 2018, 15, 229–234. [Google Scholar] [CrossRef]
- An, H.; Yin, Z.; Mitchell, C.; Semnani, A.; Hajrasouliha, A.R.; Hosseini, M. Nanodiamond Ensemble-Based Temperature Measurement in Living Cells and Its Limitations. Meas. Sci. Technol. 2021, 32, 15701. [Google Scholar] [CrossRef]
- Lu, D.-F.; Hong, Z.-F.; Xie, J.; Kong, X.-J.; Long, L.-S.; Zheng, L.-S. High-Nuclearity Lanthanide–Titanium Oxo Clusters as Luminescent Molecular Thermometers with High Quantum Yields. Inorg. Chem. 2017, 56, 12186–12192. [Google Scholar] [CrossRef]
- Albers, A.E.; Chan, E.M.; McBride, P.M.; Ajo-Franklin, C.M.; Cohen, B.E.; Helms, B.A. Dual-Emitting Quantum Dot/Quantum Rod-Based Nanothermometers with Enhanced Response and Sensitivity in Live Cells. J. Am. Chem. Soc. 2012, 134, 9565–9568. [Google Scholar] [CrossRef]
- Sadeghi, S.M. Plasmonic Metaresonance Nanosensors: Ultrasensitive Tunable Optical Sensors Based on Nanoparticle Molecules. IEEE Trans. Nanotechnol. 2011, 10, 566–571. [Google Scholar] [CrossRef]
- Lee, J.; Chen, H.; Koh, K.; Chang, C.L.; Kim, C.-M.; Kim, S.-H. Nanoassembly of CdTe Nanowires and Au Nanoparticles: PH Dependence and Reversibility of Photoluminescence. Korean J. Chem. Eng. 2009, 26, 417–421. [Google Scholar] [CrossRef]
- Bradac, C.; Lim, S.F.; Chang, H.C.; Aharonovich, I. Optical Nanoscale Thermometry: From Fundamental Mechanisms to Emerging Practical Applications. Adv. Opt. Mater. 2020, 8, 2000183. [Google Scholar] [CrossRef]
- Brites, C.D.S.; Lima, P.P.; Silva, N.J.O.; Millán, A.; Amaral, V.S.; Palacio, F.; Carlos, L.D. A Luminescent Molecular Thermometer for Long-Term Absolute Temperature Measurements at the Nanoscale. Adv. Mater. 2010, 22, 4499–4504. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Zhou, Y.; Zhang, Z.; Liu, Y.; Zhai, D.; Zhuang, H.; Li, Q.; Yuye, J.; Wu, C.; Chang, J. Multifunctional Bioactive Nd-Ca-Si Glasses for Fluorescence Thermometry, Photothermal Therapy, and Burn Tissue Repair. Sci. Adv. 2020, 6, eabb1311. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.H.; McKinney, R.L.; Hasan, M.T.; Naumov, A.V. Graphene Quantum Dots as Intracellular Imaging-Based Temperature Sensors. Materials 2021, 14, 616. [Google Scholar] [CrossRef] [PubMed]
- Maestro, L.M.; Rodriguez, E.M.; Rodriguez, F.S.; la Cruz, M.C.I.; Juarranz, A.; Naccache, R.; Vetrone, F.; Jaque, D.; Capobianco, J.A.; Solé, J.G. CdSe Quantum Dots for Two-Photon Fluorescence Thermal Imaging. Nano Lett. 2010, 10, 5109–5115. [Google Scholar] [CrossRef]
- Wang, C.; He, Y.; Xu, Y.; Sui, L.; Jiang, T.; Ran, G.; Song, Q. “Light on” Fluorescence Carbon Dots with Intramolecular Hydrogen Bond-Regulated Co-Planarization for Cell Imaging and Temperature Sensing. J. Mater. Chem. A Mater. 2022, 10, 2085–2095. [Google Scholar] [CrossRef]
- Mekhdjian, A.H.; Kai, F.; Rubashkin, M.G.; Prahl, L.S.; Przybyla, L.M.; McGregor, A.L.; Bell, E.S.; Barnes, J.M.; DuFort, C.C.; Ou, G.; et al. Integrin-Mediated Traction Force Enhances Paxillin Molecular Associations and Adhesion Dynamics That Increase the Invasiveness of Tumor Cells into a Three-Dimensional Extracellular Matrix. Mol. Biol. Cell 2017, 28, 1467–1488. [Google Scholar] [CrossRef]
- Owen, L.M.; Adhikari, A.S.; Patel, M.; Grimmer, P.; Leijnse, N.; Kim, M.C.; Notbohm, J.; Franck, C.; Dunn, A.R. A Cytoskeletal Clutch Mediates Cellular Force Transmission in a Soft, Three-Dimensional Extracellular Matrix. Mol. Biol. Cell 2017, 28, 1959–1974. [Google Scholar] [CrossRef]
- Vogel, V.; Sheetz, M. Local Force and Geometry Sensing Regulate Cell Functions. Nat. Rev. Mol. Cell Biol. 2006, 7, 265–275. [Google Scholar] [CrossRef]
- Humphrey, J.D.; Dufresne, E.R.; Schwartz, M.A. Mechanotransduction and Extracellular Matrix Homeostasis. Nat. Rev. Mol. Cell Biol. 2014, 15, 802–812. [Google Scholar] [CrossRef] [PubMed]
- Gómez-González, M.; Latorre, E.; Arroyo, M.; Trepat, X. Measuring Mechanical Stress in Living Tissues. Nat. Rev. Phys. 2020, 2, 300–317. [Google Scholar] [CrossRef]
- Lavrenyuk, K.; Conway, D.; Dahl, K.N. Imaging Methods in Mechanosensing: A Historical Perspective and Visions for the Future. Mol. Biol. Cell 2021, 32, 842–854. [Google Scholar] [CrossRef] [PubMed]
- Banda, O.A.; Sabanayagam, C.R.; Slater, J.H. Reference-Free Traction Force Microscopy Platform Fabricated via Two-Photon Laser Scanning Lithography Enables Facile Measurement of Cell-Generated Forces. ACS Appl. Mater. Interfaces 2019, 11, 18233–18241. [Google Scholar] [CrossRef]
- Hazlett, L.; Landauer, A.K.; Patel, M.; Witt, H.A.; Yang, J.; Reichner, J.S.; Franck, C. Epifluorescence-Based Three-Dimensional Traction Force Microscopy. Sci. Rep. 2020, 10, 16599. [Google Scholar] [CrossRef]
- Huang, J.; Wang, L.; Xiong, C.; Yuan, F. Elastic Hydrogel as a Sensor for Detection of Mechanical Stress Generated by Single Cells Grown in Three-Dimensional Environment. Biomaterials 2016, 98, 103–112. [Google Scholar] [CrossRef]
- Khalilgharibi, N.; Fouchard, J.; Asadipour, N.; Barrientos, R.; Duda, M.; Bonfanti, A.; Yonis, A.; Harris, A.; Mosaffa, P.; Fujita, Y.; et al. Stress Relaxation in Epithelial Monolayers Is Controlled by the Actomyosin Cortex. Nat. Phys. 2019, 15, 839–847. [Google Scholar] [CrossRef]
- Latorre, E.; Kale, S.; Casares, L.; Gómez-González, M.; Uroz, M.; Valon, L.; Nair, R.V.; Garreta, E.; Montserrat, N.; del Campo, A.; et al. Active Superelasticity in Three-Dimensional Epithelia of Controlled Shape. Nature 2018, 563, 203–208. [Google Scholar] [CrossRef]
- Arlett, J.L.; Myers, E.B.; Roukes, M.L. Comparative Advantages of Mechanical Biosensors. Nat. Nanotechnol. 2011, 6, 203–215. [Google Scholar] [CrossRef]
- Raja, S.N.; Zherebetskyy, D.; Wu, S.; Ercius, P.; Powers, A.; Olson, A.C.K.; Du, D.X.; Lin, L.; Govindjee, S.; Wang, L.-W.; et al. Mechanisms of Local Stress Sensing in Multifunctional Polymer Films Using Fluorescent Tetrapod Nanocrystals. Nano Lett. 2016, 16, 5060–5067. [Google Scholar] [CrossRef]
- Raja, S.N.; Olson, A.C.K.; Thorkelsson, K.; Luong, A.J.; Hsueh, L.; Chang, G.; Gludovatz, B.; Lin, L.; Xu, T.; Ritchie, R.O.; et al. Tetrapod Nanocrystals as Fluorescent Stress Probes of Electrospun Nanocomposites. Nano Lett. 2013, 13, 3915–3922. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.L.; Koski, K.J.; Olson, A.C.K.; Alivisatos, A.P. Luminescent Nanocrystal Stress Gauge. Proc. Natl. Acad. Sci. USA 2010, 107, 21306–21310. [Google Scholar] [CrossRef] [PubMed]
- Lay, A.; Siefe, C.; Fischer, S.; Mehlenbacher, R.D.; Ke, F.; Mao, W.L.; Alivisatos, A.P.; Goodman, M.B.; Dionne, J.A. Bright, Mechanosensitive Upconversion with Cubic-Phase Heteroepitaxial Core–Shell Nanoparticles. Nano Lett. 2018, 18, 4454–4459. [Google Scholar] [CrossRef] [PubMed]
- Lay, A.; Wang, D.S.; Wisser, M.D.; Mehlenbacher, R.D.; Lin, Y.; Goodman, M.B.; Mao, W.L.; Dionne, J.A. Upconverting Nanoparticles as Optical Sensors of Nano- to Micro-Newton Forces. Nano Lett. 2017, 17, 4172–4177. [Google Scholar] [CrossRef] [PubMed]
- Lay, A.; Sheppard, O.H.; Siefe, C.; McLellan, C.A.; Mehlenbacher, R.D.; Fischer, S.; Goodman, M.B.; Dionne, J.A. Optically Robust and Biocompatible Mechanosensitive Upconverting Nanoparticles. ACS Cent. Sci. 2019, 5, 1211–1222. [Google Scholar] [CrossRef] [PubMed]
- McLellan, C.A.; Siefe, C.; Casar, J.R.; Peng, C.S.; Fischer, S.; Lay, A.; Parakh, A.; Ke, F.; Gu, X.W.; Mao, W.; et al. Engineering Bright and Mechanosensitive Alkaline-Earth Rare-Earth Upconverting Nanoparticles. J. Phys. Chem. Lett. 2022, 13, 1547–1553. [Google Scholar] [CrossRef]
- Casar, J.R.; McLellan, C.A.; Siefe, C.; Dionne, J.A. Lanthanide-Based Nanosensors: Refining Nanoparticle Responsiveness for Single Particle Imaging of Stimuli. ACS Photonics 2021, 8, 3–17. [Google Scholar] [CrossRef]
- Rifaie-Graham, O.; Apebende, E.A.; Bast, L.K.; Bruns, N. Self-Reporting Fiber-Reinforced Composites That Mimic the Ability of Biological Materials to Sense and Report Damage. Adv. Mater. 2018, 30, 1705483. [Google Scholar] [CrossRef]
- Mehlenbacher, R.D.; Kolbl, R.; Lay, A.; Dionne, J.A. Nanomaterials for in Vivo Imaging of Mechanical Forces and Electrical Fields. Nat. Rev. Mater. 2018, 3, 17080. [Google Scholar] [CrossRef]
- Demircan Yalcin, Y.; Luttge, R. Electrical Monitoring Approaches in 3-Dimensional Cell Culture Systems: Toward Label-Free, High Spatiotemporal Resolution, and High-Content Data Collection in Vitro. Organs-On-A-Chip 2021, 3, 100006. [Google Scholar] [CrossRef]
- Tian, B.; Liu, J.; Dvir, T.; Jin, L.; Tsui, J.H.; Qing, Q.; Suo, Z.; Langer, R.; Kohane, D.S.; Lieber, C.M. Macroporous Nanowire Nanoelectronic Scaffolds for Synthetic Tissues. Nat. Mater. 2012, 11, 986–994. [Google Scholar] [CrossRef] [PubMed]
- Feiner, R.; Engel, L.; Fleischer, S.; Malki, M.; Gal, I.; Shapira, A.; Shacham-Diamand, Y.; Dvir, T. Engineered Hybrid Cardiac Patches with Multifunctional Electronics for Online Monitoring and Regulation of Tissue Function. Nat. Mater. 2016, 15, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Hong, G.; Gao, T.; Lieber, C.M. Mesh Nanoelectronics: Seamless Integration of Electronics with Tissues. Acc. Chem. Res. 2018, 51, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Hong, G.; Yang, X.; Zhou, T.; Lieber, C.M. Mesh Electronics: A New Paradigm for Tissue-like Brain Probes. Curr. Opin. Neurobiol. 2018, 50, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Atay, T.; Nurmikko, A.V. Optical Detection of Brain Cell Activity Using Plasmonic Gold Nanoparticles. Nano Lett. 2009, 9, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, A.; Serna, P.; Morgenstein, L.; Yang, G.; Bar-Elli, O.; Ortiz, G.; Miller, E.; Oron, D.; Grupi, A.; Weiss, S.; et al. Development of Lipid-Coated Semiconductor Nanosensors for Recording of Membrane Potential in Neurons. ACS Photonics 2020, 7, 1141–1152. [Google Scholar] [CrossRef]
- Caglar, M.; Pandya, R.; Xiao, J.; Foster, S.K.; Divitini, G.; Chen, R.Y.S.; Greenham, N.C.; Franze, K.; Rao, A.; Keyser, U.F. All-Optical Detection of Neuronal Membrane Depolarization in Live Cells Using Colloidal Quantum Dots. Nano Lett. 2019, 19, 8539–8549. [Google Scholar] [CrossRef]
- Jooken, S.; de Coene, Y.; Deschaume, O.; Krylychkina, O.; Verbiest, T.; Clays, K.; Callewaert, G.; Bartic, C. Quantum Dot-Functionalized Extracellular Matrices for in Situ Monitoring of Cardiomyocyte Activity. ACS Appl. Nano Mater. 2020, 3, 6118–6126. [Google Scholar] [CrossRef]
- Nag, O.K.; Muroski, M.E.; Hastman, D.A.; Almeida, B.; Medintz, I.L.; Huston, A.L.; Delehanty, J.B. Nanoparticle-Mediated Visualization and Control of Cellular Membrane Potential: Strategies, Progress, and Remaining Issues. ACS Nano 2020, 14, 2659–2677. [Google Scholar] [CrossRef]
- Chen, G.; Zhang, Y.; Peng, Z.; Huang, D.; Li, C.; Wang, Q. Glutathione-Capped Quantum Dots for Plasma Membrane Labeling and Membrane Potential Imaging. Nano Res. 2019, 12, 1321–1326. [Google Scholar] [CrossRef]
- Guduru, R.; Liang, P.; Yousef, M.; Horstmyer, J.; Khizroev, S. Mapping the Brain’s Electric Fields with Magnetoelectric Nanoparticles. Bioelectron. Med. 2018, 4, 10. [Google Scholar] [CrossRef] [PubMed]
- Bok, I.; Haber, I.; Qu, X.; Hai, A. In Silico Assessment of Electrophysiological Neuronal Recordings Mediated by Magnetoelectric Nanoparticles. Sci. Rep. 2022, 12, 8386. [Google Scholar] [CrossRef] [PubMed]
- Orendorff, R.; Peck, A.J.; Zheng, B.; Shirazi, S.N.; Matthew Ferguson, R.; Khandhar, A.P.; Kemp, S.J.; Goodwill, P.; Krishnan, K.M.; Brooks, G.A.; et al. First in Vivo Traumatic Brain Injury Imaging via Magnetic Particle Imaging. Phys. Med. Biol. 2017, 62, 3501–3509. [Google Scholar] [CrossRef] [PubMed]
- Scanziani, M.; Häusser, M. Electrophysiology in the Age of Light. Nature 2009, 461, 930–939. [Google Scholar] [CrossRef] [PubMed]
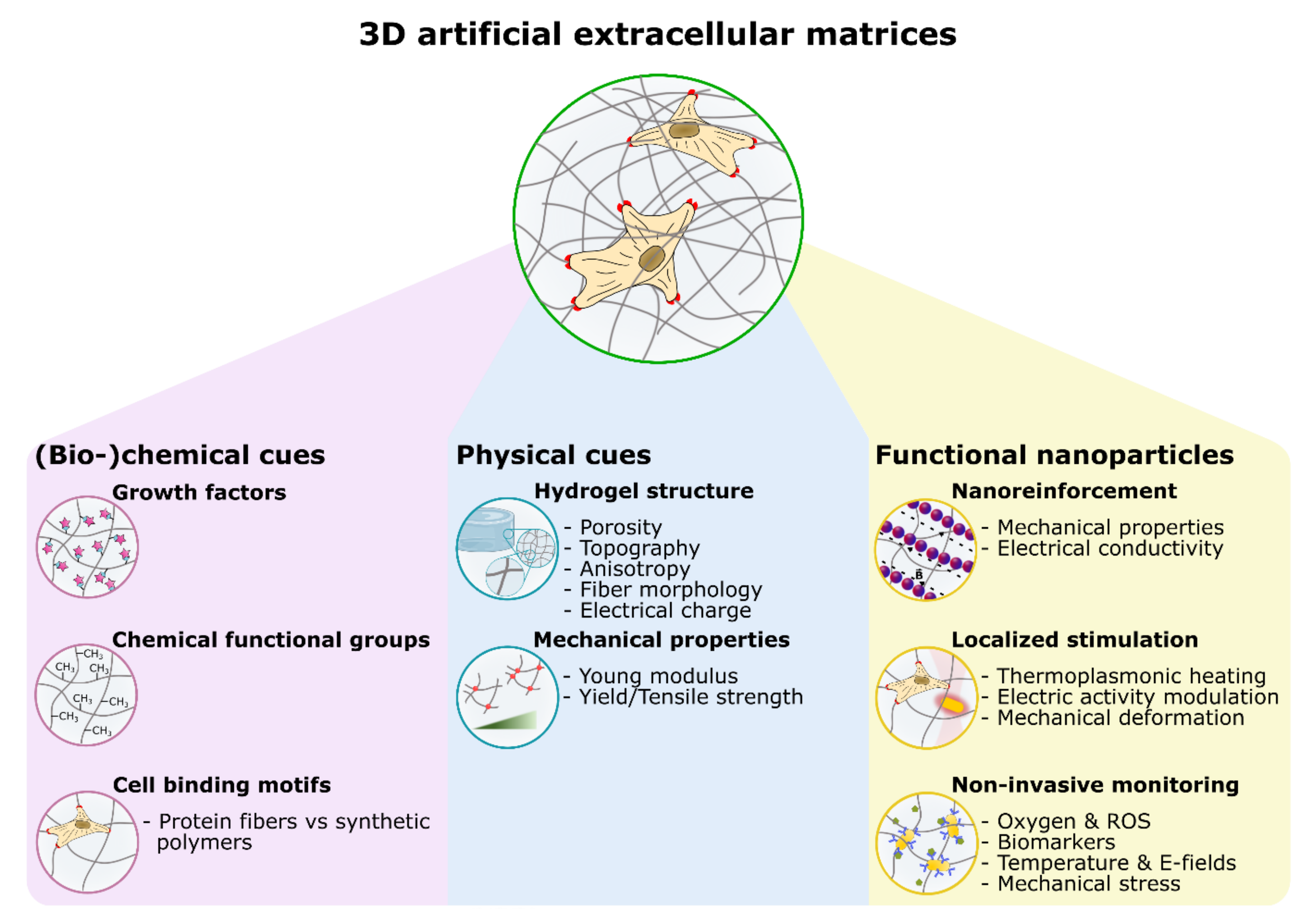
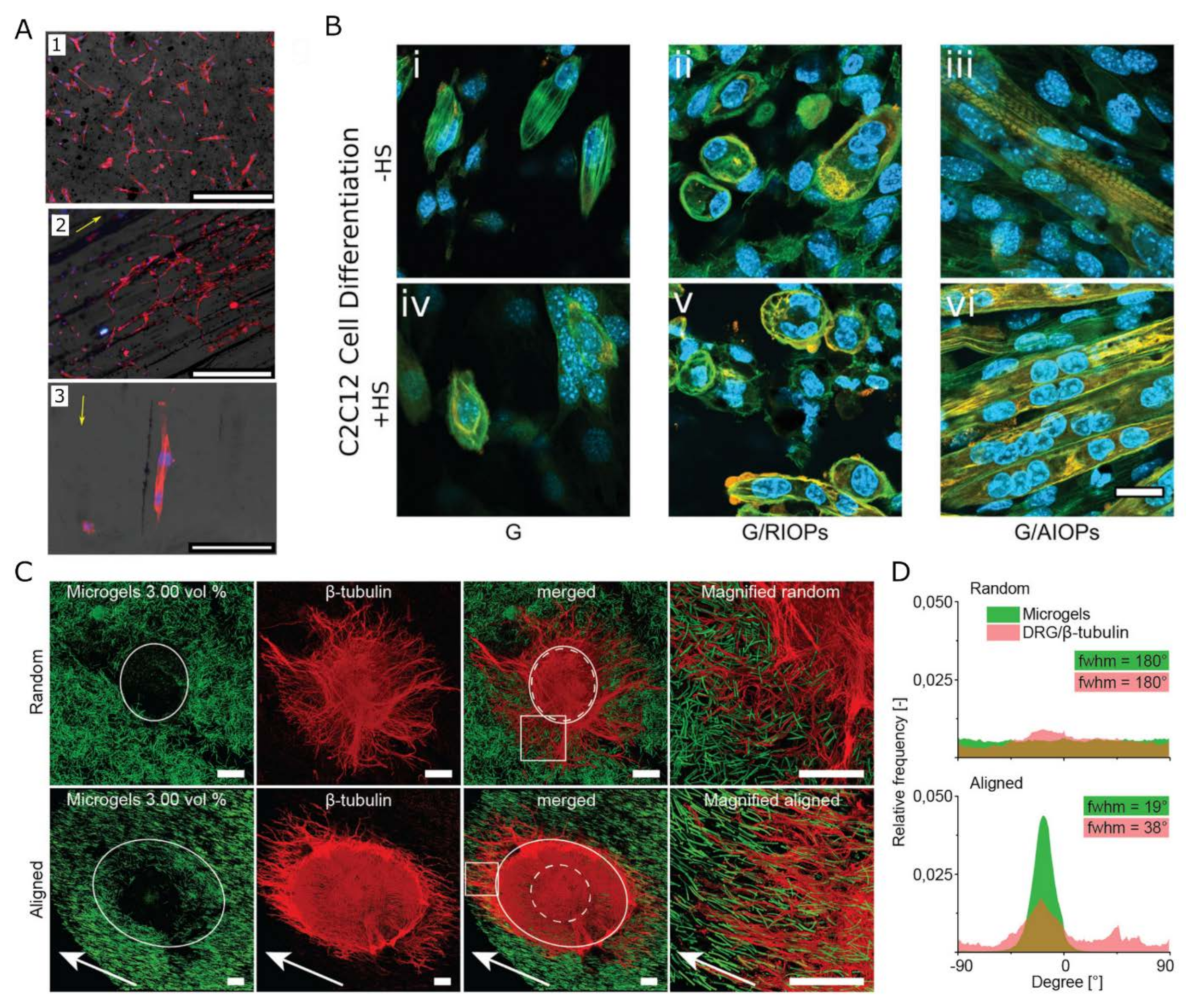


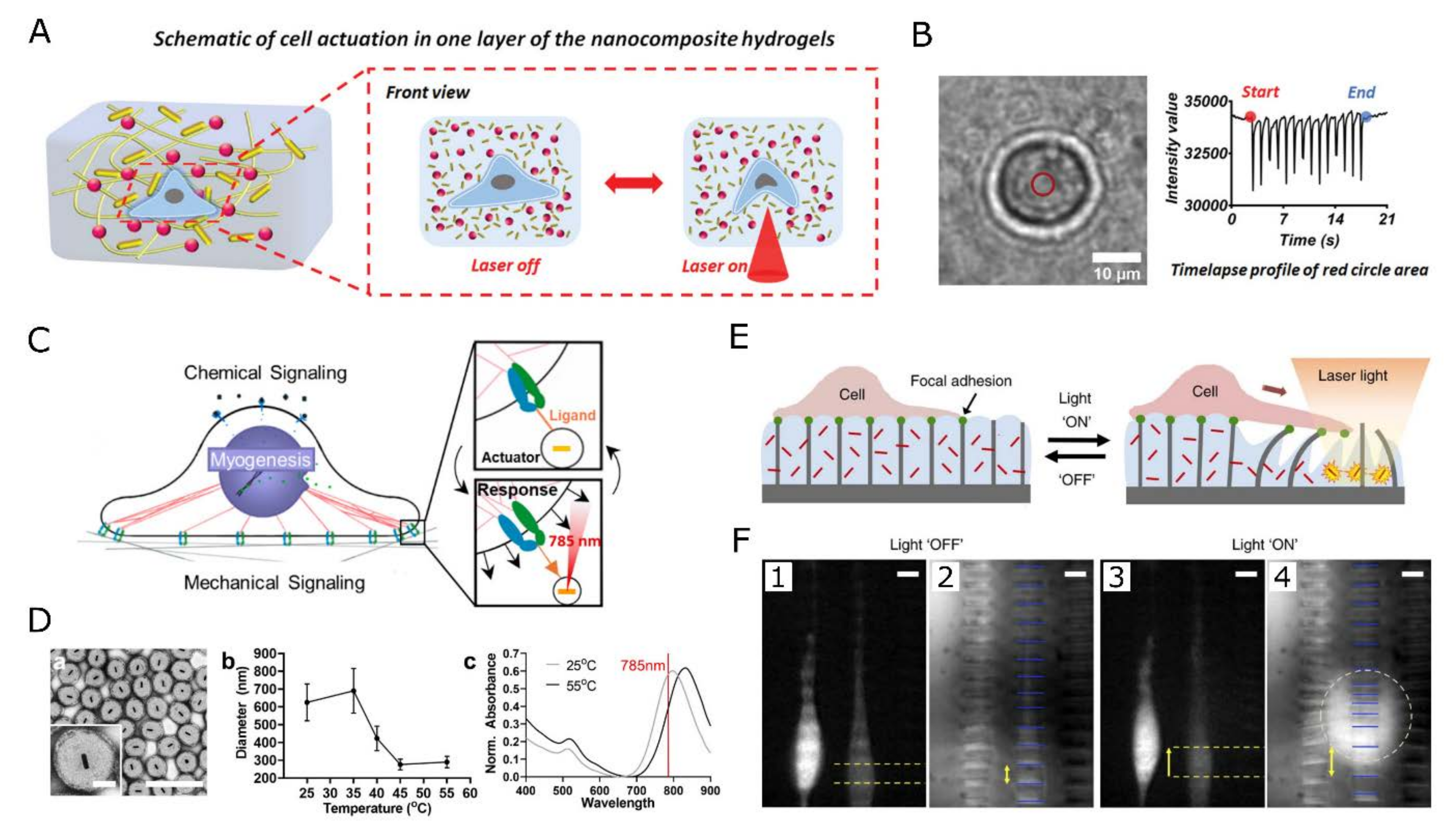
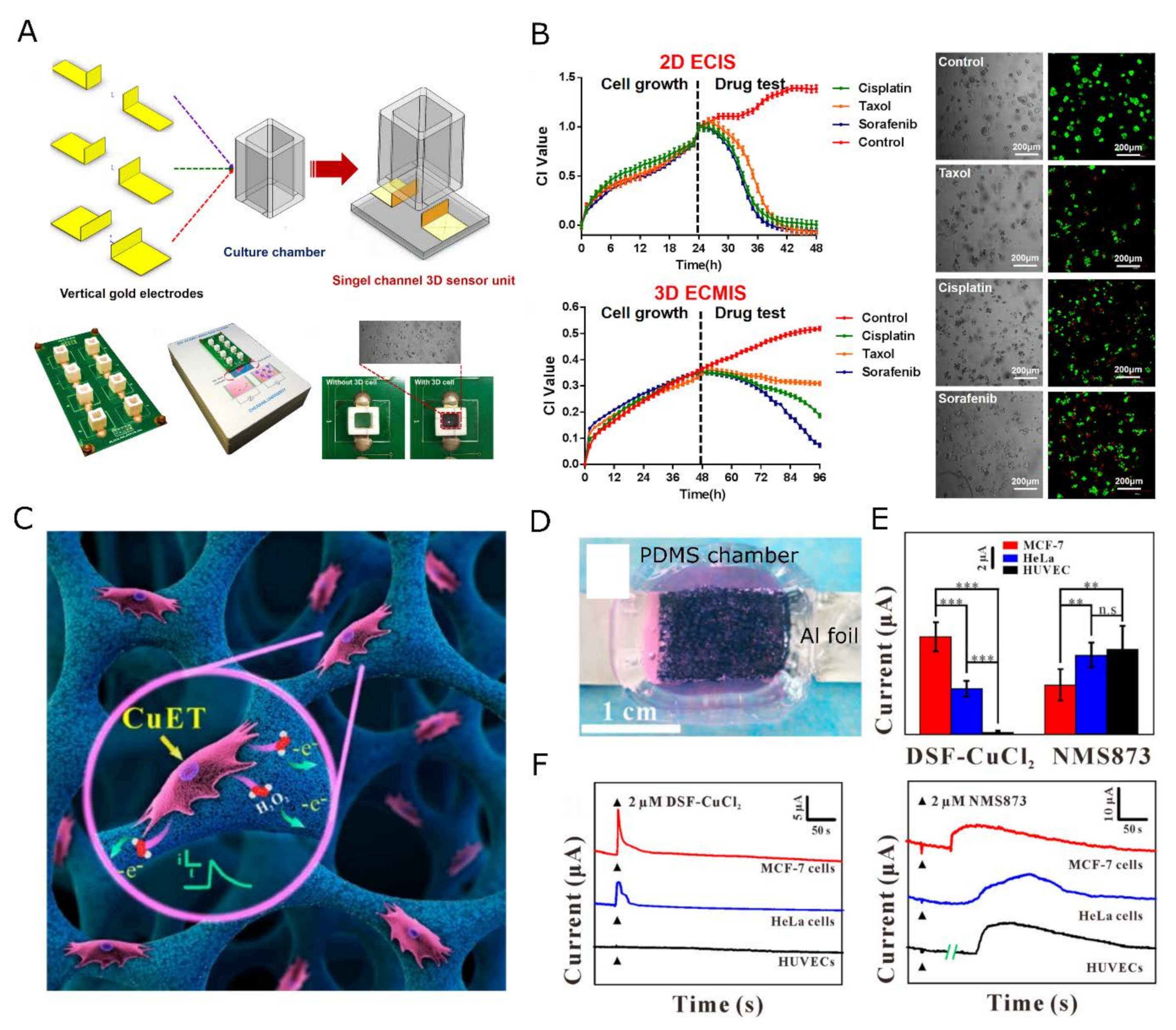
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jooken, S.; Deschaume, O.; Bartic, C. Nanocomposite Hydrogels as Functional Extracellular Matrices. Gels 2023, 9, 153. https://doi.org/10.3390/gels9020153
Jooken S, Deschaume O, Bartic C. Nanocomposite Hydrogels as Functional Extracellular Matrices. Gels. 2023; 9(2):153. https://doi.org/10.3390/gels9020153
Chicago/Turabian StyleJooken, Stijn, Olivier Deschaume, and Carmen Bartic. 2023. "Nanocomposite Hydrogels as Functional Extracellular Matrices" Gels 9, no. 2: 153. https://doi.org/10.3390/gels9020153
APA StyleJooken, S., Deschaume, O., & Bartic, C. (2023). Nanocomposite Hydrogels as Functional Extracellular Matrices. Gels, 9(2), 153. https://doi.org/10.3390/gels9020153








