Engineered Vesicles and Hydrogel Technologies for Myocardial Regeneration
Abstract
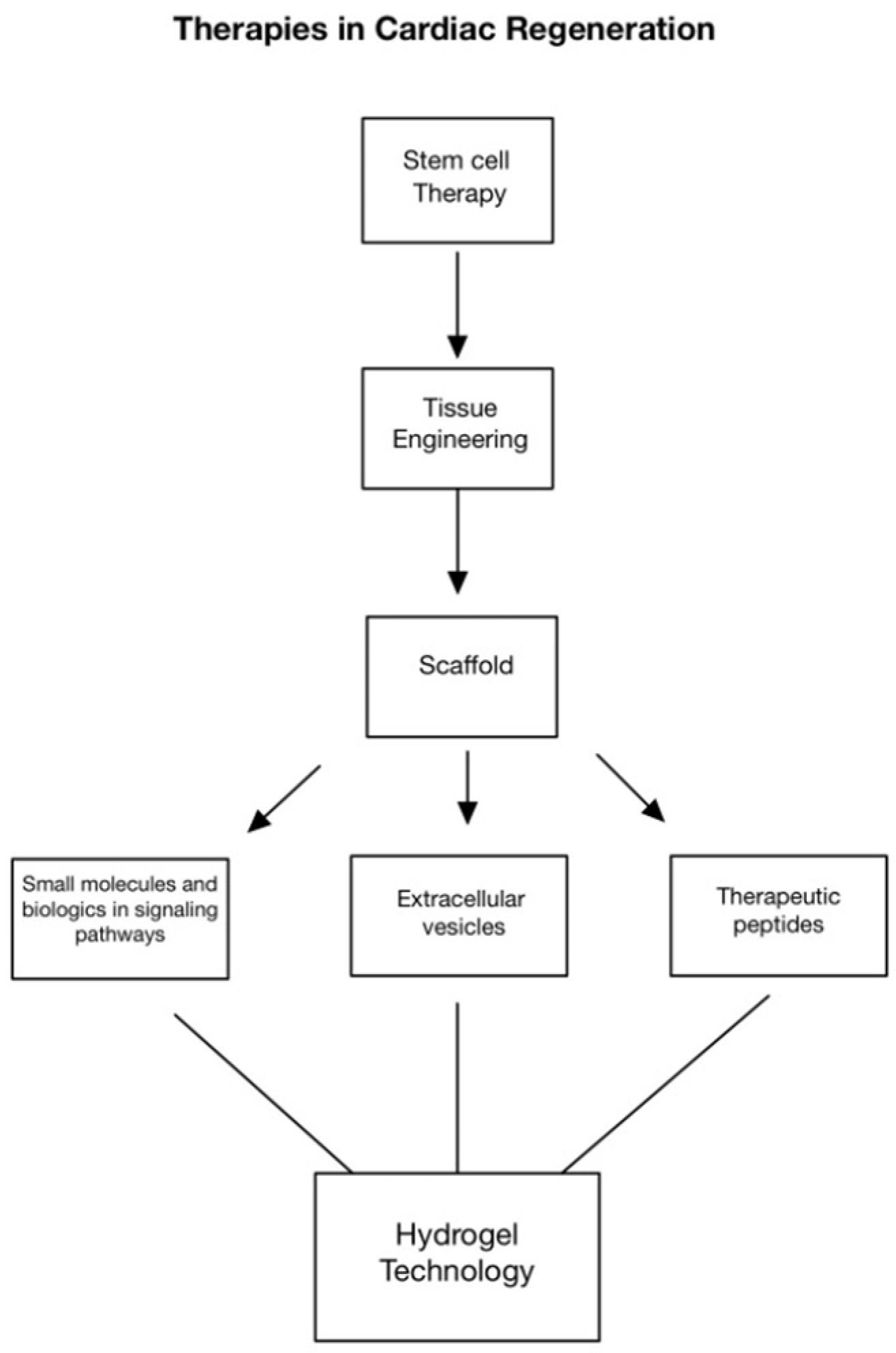
:1. Introduction
2. Extracellular Vesicles (EVs) in Myocardial Injury and Healing Response
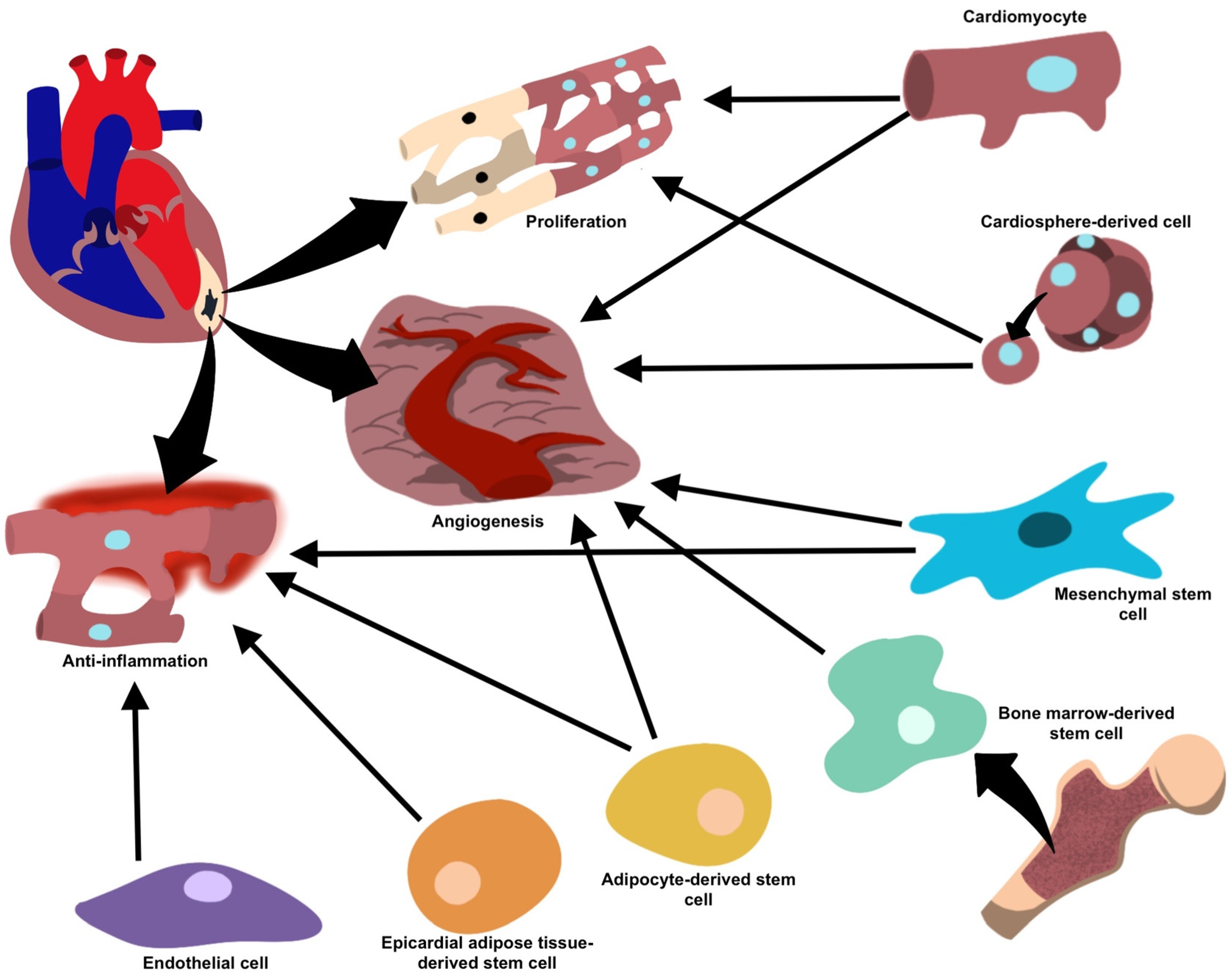
2.1. Vesicle-Mediated Cardiac Regeneration
2.2. Immune Cell-Derived EVs
2.3. Mesenchymal Stem Cells (MSCs)-Derived EVs
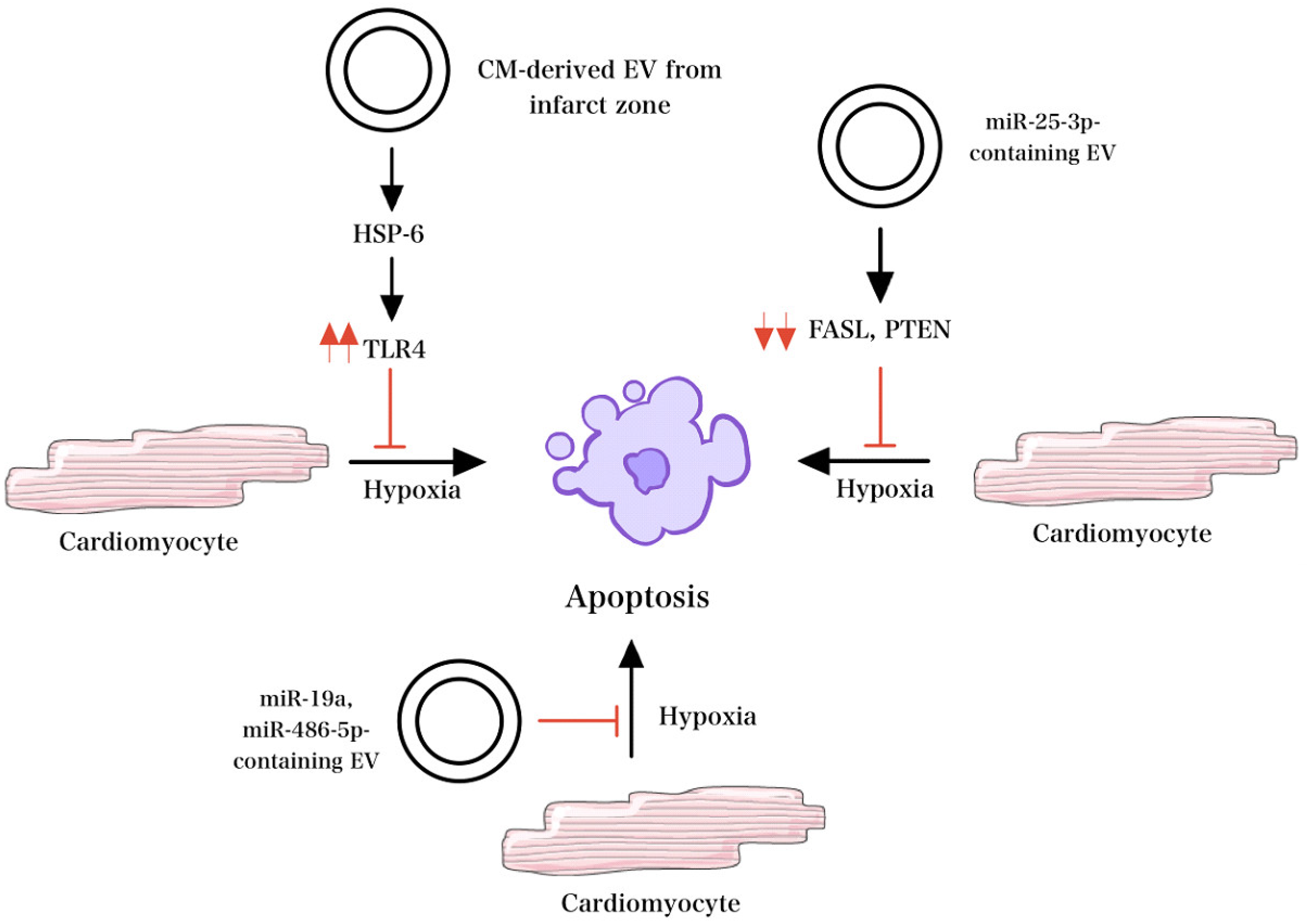
2.4. EVs from Cardiomyocytes (CMs) and Cardiac Fibroblasts (CF)
2.5. EVs from Cardiosphere-Derived Cells (CDCs)
2.6. Cardiac Endothelial Cell (CEC)-Derived EVs
2.7. Adipose-Derived Stem Cells (ADSC)-Derived EVs
2.8. Bone Marrow-Derived Stem Cells (BMSC)-Derived EVs
2.9. Epicardial Adipose-Derived Stem Cells-Derived EVs
3. Vesicle-Derived Signaling for Cardiac Regeneration
| Animal Model | Target | Function | References |
|---|---|---|---|
| Amphibian and in vitro | miR-1 | Promote differentiation into cardiac muscle. | [69] |
| Amphibian and in vitro | miR-133 | Promote myoblast proliferation. | [69] |
| Mouse | miR-29 | Prevents fibrosis by reducing the expression of collagen. | [54] |
| Mouse | miR-24 | Inhibition of cardiomyocyte apoptosis, attenuation of infarct size, reduced cardiac dysfunction. | [54] |
| Mouse | miR-182-5p | Inhibition of TLR4 activity. | [55,56] |
| Mouse | miR-212-5p | reduced levels of α-SMA, Collagen I, TGF-β1, and IL-1β. | [22] |
| Human | miR-93-5p | Promotes proliferation of tumor cells via effects on the PTEN gene. | [70] |
| Rat | miR-146a | suppressed hypoxic-induced H9c2 injury by suppressing EGR1 and regulating inflammation, apoptosis, and fibrosis by promoting the TLR4/NFκB signaling pathway. | [58] |
| Human | miR-125b-5p | Suppression of p53 and BAK1 and regulation of apoptosis. | [59] |
| Rat | miR-19 | Prevention of hypoxia/reoxygenation-induced apoptosis and alleviating injury via AKT pathway. | [60] |
| Mice | miR-25-3p | Targets PTEN and FASL to inhibit apoptosis. | [61] |
| Rat | miR-486-5p | Suppression of hypoxia/reoxygenation-triggered apoptosis and P13K/AKT pathway activation. | [62] |
| Human | miR-146 | Potentially protective against oxidative stress. | [71] |
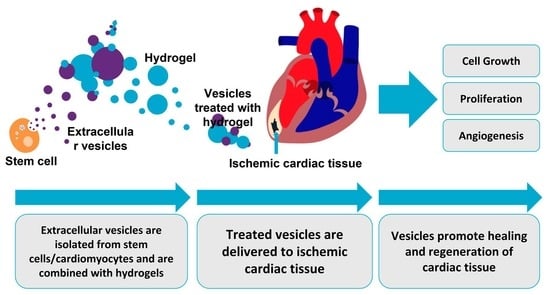
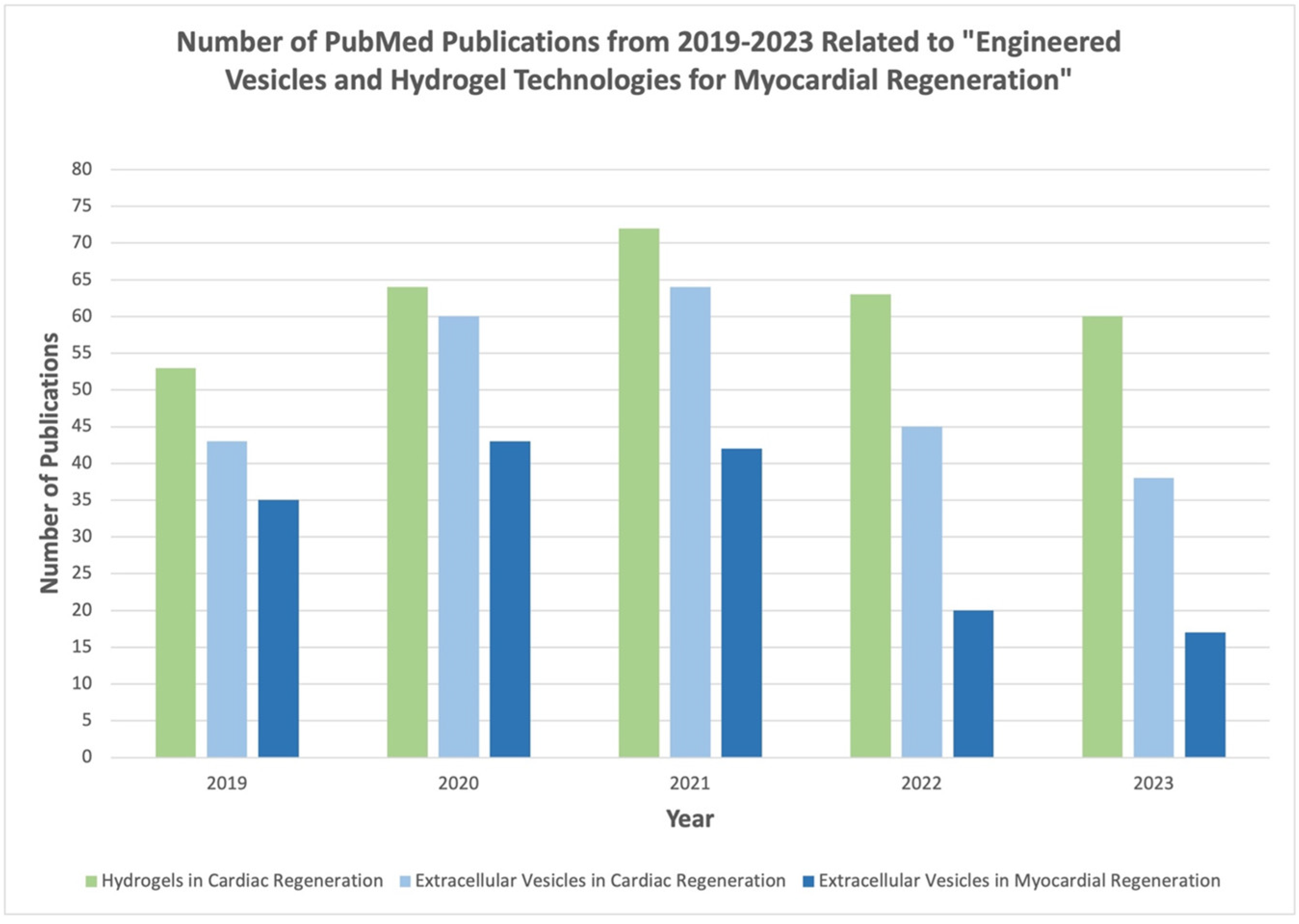
4. Engineered EVs and Hydrogel Technologies for Cardiac Healing
5. EV-Loaded Hydrogels for Cardiac Regeneration
6. Natural Hydrogels
7. Composite and Functionalized Hydrogels
8. Translational Implications, Limitations and Future
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tzahor, E.; Poss, K.D. Cardiac Regeneration Strategies: Staying Young at Heart. Science 2017, 356, 1035–1039. [Google Scholar] [CrossRef] [PubMed]
- Sheng, K.; Nie, Y.; Gao, B. Recent Advances in Myocardial Regeneration Strategy. J. Int. Med. Res. 2019, 47, 5453–5464. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; LeBleu, V.S. The Biology, Function, and Biomedical Applications of Exosomes. Science 2020, 367, eaau6977. [Google Scholar] [CrossRef] [PubMed]
- Joladarashi, D.; Kishore, R. Mesenchymal Stromal Cell Exosomes in Cardiac Repair. Curr. Cardiol. Rep. 2022, 24, 405–417. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.; Chen, T. Mesenchymal Stem Cell-Derived Exosomes and Their Potential Agents in Hematological Diseases. Oxid. Med. Cell Longev. 2021, 2021, 4539453. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.L.; Lai, T.C.; Lin, S.R.; Lin, S.W.; Chen, Y.C.; Pu, C.M.; Lee, I.T.; Tsai, J.S.; Lee, C.W.; Chen, Y.L. Conditioned Medium from Adipose-Derived Stem Cells Attenuates Ischemia/Reperfusion-Induced Cardiac Injury through the MicroRNA-221/222/PUMA/ETS-1 Pathway. Theranostics 2021, 11, 3131–3149. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Jin, L. Mechanics Underpinning Phase Separation of Hydrogels. Macromolecules 2023, 56, 426–439. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.; Nyman, J.N.; Reinholdt, R.; Cai, J.; Schaedel, A.-L.; van der Plas, M.J.A.; Malmsten, M.; Rades, T.; Heinz, A. In Situ Transformation of Electrospun Nanofibers into Nanofiber-Reinforced Hydrogels. Nanomaterials 2022, 12, 2437. [Google Scholar] [CrossRef]
- Yang, J.; Rao, L.; Wang, Y.; Zhao, Y.; Liu, D.; Wang, Z.; Fu, L.; Wang, Y.; Yang, X.; Li, Y.; et al. Recent Advances in Smart Hydrogels Prepared by Ionizing Radiation Technology for Biomedical Applications. Polymers 2022, 14, 4377. [Google Scholar] [CrossRef]
- Sahoo, S.; Losordo, D. Cardiac Repair and Regeneration after MI: What Is Known? Circ. Res. 2014, 114, 333–344. [Google Scholar] [CrossRef]
- Ong, S.G.; Wu, J.C. Exosomes as Potential Alternatives to Stem Cell Therapy in Mediating Cardiac Regeneration. Circ. Res. 2015, 117, 7–9. [Google Scholar] [CrossRef]
- Kolios, G.; Moodley, Y. Introduction to Stem Cells and Regenerative Medicine. Respiration 2012, 85, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaka, Y.; Yashiro, R. Therapeutic Strategy of Mesenchymal-Stem-Cell-Derived Extracellular Vesicles as Regenerative Medicine. Int. J. Mol. Sci. 2022, 23, 6480. [Google Scholar] [CrossRef] [PubMed]
- Hegyesi, H.; Pallinger, É.; Mecsei, S.; Hornyák, B.; Kovácsházi, C.; Brenner, G.B.; Giricz, Z.; Pálóczi, K.; Kittel, Á.; Tóvári, J.; et al. Circulating Cardiomyocyte-Derived Extracellular Vesicles Reflect Cardiac Injury during Systemic Inflammatory Response Syndrome in Mice. Cell. Mol. Life Sci. 2022, 79, 84. [Google Scholar] [CrossRef] [PubMed]
- Ju, Y.; Hu, Y.; Yang, P.; Xie, X.; Fang, B. Extracellular Vesicle-Loaded Hydrogels for Tissue Repair and Regeneration. Mater. Today Bio 2023, 18, 100522. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gu, H.; Qin, D.; Yang, L.; Huang, W.; Essandoh, K.; Wang, Y.; Caldwell, C.C.; Peng, T.; Zingarelli, B.; et al. Exosomal MIR-223 Contributes to Mesenchymal Stem Cell-Elicited Cardioprotection in Polymicrobial Sepsis. Sci. Rep. 2015, 5, 13721. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chen, Y.; Zhao, Z.; Meng, Q.; Yu, Y.; Sun, J.; Yang, Z.; Chen, Y.; Li, J.; Ma, T.; et al. Engineered Exosomes with Ischemic Myocardium-Targeting Peptide for Targeted Therapy in Myocardial Infarction. J. Am. Heart Assoc. 2018, 7, e008737. [Google Scholar] [CrossRef]
- Wang, H.; Xie, Y.; Salvador, A.M.; Zhang, Z.; Chen, K.; Li, G.; Xiao, J. Exosomes: Multifaceted Messengers in Atherosclerosis. Curr. Atheroscler. Rep. 2020, 22, 57. [Google Scholar] [CrossRef]
- Wang, X.; Bai, L.; Liu, X.; Shen, W.; Tian, H.; Liu, W.; Yu, B. Cardiac Microvascular Functions Improved by MSC-Derived Exosomes Attenuate Cardiac Fibrosis after Ischemia–Reperfusion via PDGFR-β Modulation. Int. J. Cardiol. 2021, 344, 13–24. [Google Scholar] [CrossRef]
- Dong, J.; Wu, B.; Tian, W. Exosomes Derived from Hypoxia-Preconditioned Mesenchymal Stem Cells (HypoMSCs-Exo): Advantages in Disease Treatment. Cell Tissue Res. 2023, 392, 621–629. [Google Scholar] [CrossRef]
- Wen, H.; Peng, L.; Chen, Y. The Effect of Immune Cell-Derived Exosomes in the Cardiac Tissue Repair after Myocardial Infarction: Molecular Mechanisms and Pre-Clinical Evidence. J. Cell. Mol. Med. 2021, 25, 6500–6510. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Peng, W.; Fang, M.; Wu, M.; Wu, M. MSCs-Derived Extracellular Vesicles Carrying MiR-212-5p Alleviate Myocardial Infarction-Induced Cardiac Fibrosis via NLRC5/VEGF/TGF-Β1/SMAD Axis. J. Cardiovasc. Transl. Res. 2022, 15, 302–316. [Google Scholar] [CrossRef] [PubMed]
- Fang, W.H.; Agrawal, D.K.; Thankam, F.G. Smart Exosomes: A Smart Approach for Tendon Regeneration. Tissue Eng. Part. B Rev. 2022, 28, 613–625. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Gao, W.; Yao, K.; Ge, J. Roles of Exosomes Derived from Immune Cells in Cardiovascular Diseases. Front. Immunol. 2019, 10, 648. [Google Scholar] [CrossRef] [PubMed]
- Mihailovic, P.M.; Lio, W.M.; Herscovici, R.; Chyu, K.Y.; Yano, J.; Zhao, X.; Zhou, J.; Zhou, B.; Freeman, M.R.; Yang, W.; et al. Keratin 8 Is a Potential Self-Antigen in the Coronary Artery Disease Immunopeptidome: A Translational Approach. PLoS ONE 2019, 14, e0213025. [Google Scholar] [CrossRef] [PubMed]
- Lazar, E.; Benedek, T.; Korodi, S.; Rat, N.; Lo, J.; Benedek, I. Stem Cell-Derived Exosomes—An Emerging Tool for Myocardial Regeneration. World J. Stem Cells 2018, 10, 106–115. [Google Scholar] [CrossRef]
- Shafei, S.; Khanmohammadi, M.; Ghanbari, H.; Nooshabadi, V.T.; Tafti, S.H.A.; Rabbani, S.; Kasaiyan, M.; Basiri, M.; Tavoosidana, G. Effectiveness of Exosome Mediated MiR-126 and MiR-146a Delivery on Cardiac Tissue Regeneration. Cell Tissue Res. 2022, 390, 71–92. [Google Scholar] [CrossRef] [PubMed]
- Nasser, M.I.; Masood, M.; Adlat, S.; Gang, D.; Zhu, S.; Li, G.; Li, N.; Chen, J.; Zhu, P. Mesenchymal Stem Cell-Derived Exosome MicroRNA as Therapy for Cardiac Ischemic Injury. Biomed. Pharmacother. 2021, 143, 112118. [Google Scholar] [CrossRef]
- Sun, S.J.; Wei, R.; Li, F.; Liao, S.Y.; Tse, H.F. Mesenchymal Stromal Cell-Derived Exosomes in Cardiac Regeneration and Repair. Stem Cell Rep. 2021, 16, 1662–1673. [Google Scholar] [CrossRef]
- He, C.; Zheng, S.; Luo, Y.; Wang, B. Exosome Theranostics: Biology and Translational Medicine. Theranostics 2018, 8, 237–255. [Google Scholar] [CrossRef]
- Zhang, P.; Su, J.; Mende, U. Cross Talk between Cardiac Myocytes and Fibroblasts: From Multiscale Investigative Approaches to Mechanisms and Functional Consequences. Am. J. Physiol. Heart Circ. Physiol. 2012, 303, H1385–H1396. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Tu, H.; Tang, K.; Huang, H.; Ou, S.; Wu, J. MiR-3064 in Epicardial Adipose-Derived Exosomes Targets Neuronatin to Regulate Adipogenic Differentiation of Epicardial Adipose Stem Cells. Front. Cardiovasc. Med. 2021, 8, 709079. [Google Scholar] [CrossRef] [PubMed]
- Wojciechowska, A.; Braniewska, A.; Kozar-Kamińska, K. MicroRNA in Cardiovascular Biology and Disease. Adv. Clin. Exp. Med. 2017, 26, 865–874. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, T.; Barone, A.; Salatino, A.; Molinaro, C.; Marino, F.; Scalise, M.; Torella, M.; De Angelis, A.; Urbanek, K.; Torella, D.; et al. Unravelling the Biology of Adult Cardiac Stem Cell-Derived Exosomes to Foster Endogenous Cardiac Regeneration and Repair. Int. J. Mol. Sci. 2020, 21, 3725. [Google Scholar] [CrossRef]
- Smyth, T.; Kullberg, M.; Malik, N.; Smith-Jones, P.; Graner, M.W.; Anchordoquy, T.J. Biodistribution and Delivery Efficiency of Unmodified Tumor-Derived Exosomes. J. Control. Release 2015, 199, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Maring, J.A.; Beez, C.M.; Falk, V.; Seifert, M.; Stamm, C. Myocardial Regeneration via Progenitor Cell-Derived Exosomes. Stem Cells Int. 2017, 2017, 7849851. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, C.; Ma, J. Role of Cardiac Endothelial Cells-Derived MicroRNAs in Cardiac Remodeling. Discov. Med. 2019, 28, 95–105. [Google Scholar] [PubMed]
- Roncarati, R.; Viviani Anselmi, C.; Losi, M.A.; Papa, L.; Cavarretta, E.; Da Costa Martins, P.; Contaldi, C.; Saccani Jotti, G.; Franzone, A.; Galastri, L.; et al. Circulating MiR-29a, among Other up-Regulated MicroRNAs, Is the Only Biomarker for Both Hypertrophy and Fibrosis in Patients with Hypertrophic Cardiomyopathy. J. Am. Coll. Cardiol. 2014, 63, 920–927. [Google Scholar] [CrossRef]
- Ono, K.; Kuwabara, Y.; Han, J. MicroRNAs and Cardiovascular Diseases. FEBS J. 2011, 278, 1619–1633. [Google Scholar] [CrossRef]
- Song, J.; Chen, X.; Wang, M.; Xing, Y.; Zheng, Z.; Hu, S. Cardiac Endothelial Cell-Derived Exosomes Induce Specific Regulatory B Cells. Sci. Rep. 2014, 4, 7583. [Google Scholar] [CrossRef]
- Lee, C.; Mitsialis, S.A.; Aslam, M.; Vitali, S.H.; Vergadi, E.; Konstantinou, G.; Sdrimas, K.; Fernandez-Gonzalez, A.; Kourembanas, S. Exosomes Mediate the Cytoprotective Action of Mesenchymal Stromal Cells on Hypoxia-Induced Pulmonary Hypertension. Circulation 2012, 126, 2601–2611. [Google Scholar] [CrossRef] [PubMed]
- Luo, Q.; Guo, D.; Liu, G.; Chen, G.; Hang, M.; Jin, M. Exosomes from MiR-126-Overexpressing Adscs Are Therapeutic in Relieving Acute Myocardial Ischaemic Injury. Cell. Physiol. Biochem. 2017, 44, 2105–2116. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.; Thorne, T.; Losordo, D.W.; Yoon, Y.S. Repair of Ischemic Heart Disease with Novel Bone Marrow-Derived Multipotent Stem Cells. Cell Cycle 2005, 4, 861–864. [Google Scholar] [CrossRef] [PubMed]
- Müller, P.; Lemcke, H.; David, R. Stem Cell Therapy in Heart Diseases-Cell Types, Mechanisms and Improvement Strategies. Cell. Physiol. Biochem. 2018, 48, 2607–2655. [Google Scholar] [CrossRef] [PubMed]
- Azari, Z.; Nazarnezhad, S.; Webster, T.J.; Hoseini, S.J.; Brouki Milan, P.; Baino, F.; Kargozar, S. Stem cell-mediated Angiogenesis in Skin Tissue Engineering and Wound Healing. Wound Repair. Regen. 2022, 30, 421–435. [Google Scholar] [CrossRef] [PubMed]
- Mangialardi, G.; Madeddu, P. Bone Marrow-Derived Stem Cells: A Mixed Blessing in the Multifaceted World of Diabetic Complications. Curr. Diabetes Rep. 2016, 16, 43. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Shaw, G.; Murphy, M.; Barry, F. Induced Pluripotent Stem Cell-Derived Mesenchymal Stromal Cells Are Functionally and Genetically Different From Bone Marrow-Derived Mesenchymal Stromal Cells. Stem Cells 2019, 37, 754–765. [Google Scholar] [CrossRef] [PubMed]
- Korf-Klingebiel, M.; Reboll, M.R.; Klede, S.; Brod, T.; Pich, A.; Polten, F.; Napp, L.C.; Bauersachs, J.; Ganser, A.; Brinkmann, E.; et al. Myeloid-Derived Growth Factor (C19orf10) Mediates Cardiac Repair Following Myocardial Infarction. Nat. Med. 2015, 21, 140–149. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Y.; Feng, J.; Liu, W.; Li, Y.; Liu, J.; Yin, Q.; Lian, H.; Liu, L.; Nie, Y. Mydgf Promotes Cardiomyocyte Proliferation and Neonatal Heart Regeneration. Theranostics 2020, 10, 9100–9112. [Google Scholar] [CrossRef]
- Özkaynak, B.; Şahin, I.; Özenc, E.; Subaşı, C.; Oran, D.S.; Totoz, T.; Tetikkurt, Ü.S.; Mert, B.; Polat, A.; Okuyan, E.; et al. Mesenchymal Stem Cells Derived from Epicardial Adipose Tissue Reverse Cardiac Remodeling in a Rabbit Model of Myocardial Infarction. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 4372–4384. [Google Scholar]
- Thankam, F.G.; Agrawal, D.K. Single Cell Genomics Identifies Unique Cardioprotective Phenotype of Stem Cells Derived from Epicardial Adipose Tissue under Ischemia. Stem Cell Rev. Rep. 2022, 18, 294–335. [Google Scholar] [CrossRef] [PubMed]
- Lambert, C.; Arderiu, G.; Bejar, M.T.; Crespo, J.; Baldellou, M.; Juan-Babot, O.; Badimon, L. Stem Cells from Human Cardiac Adipose Tissue Depots Show Different Gene Expression and Functional Capacities. Stem Cell Res. Ther. 2019, 10, 361. [Google Scholar] [CrossRef] [PubMed]
- Singla, D.K. Stem Cells and Exosomes in Cardiac Repair. Curr. Opin. Pharmacol. 2016, 27, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Shao, L.; Zhang, Y.; Lan, B.; Wang, J.; Zhang, Z.; Zhang, L.; Xiao, P.; Meng, Q.; Geng, Y.; Yu, X.; et al. MiRNA-Sequence Indicates That Mesenchymal Stem Cells and Exosomes Have Similar Mechanism to Enhance Cardiac Repair. Biomed. Res. Int. 2017, 2017, 4150705. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Li, X.; Hu, J.; Chen, F.; Qiao, S.; Sun, X.; Gao, L.; Xie, J.; Xu, B. Mesenchymal Stromal Cell-Derived Exosomes Attenuate Myocardial Ischaemia-Reperfusion Injury through MiR-182-Regulated Macrophage Polarization. Cardiovasc. Res. 2019, 115, 1205–1216. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Li, W.; Li, Y.; Chen, J.; An, H.; Zeng, G.; Wang, T.; Guo, Y.; Wang, C. MiR-182-5p Mediated by Exosomes Derived From Bone Marrow Mesenchymal Stem Cell Attenuates Inflammatory Responses by Targeting TLR4 in a Mouse Model of Myocardial Infraction. Immune Netw. 2022, 22, e49. [Google Scholar] [CrossRef]
- Liu, S.; Chen, J.; Shi, J.; Zhou, W.; Wang, L.; Fang, W.; Zhong, Y.; Chen, X.; Chen, Y.; Sabri, A.; et al. M1-like Macrophage-Derived Exosomes Suppress Angiogenesis and Exacerbate Cardiac Dysfunction in a Myocardial Infarction Microenvironment. Basic. Res. Cardiol. 2020, 115, 22. [Google Scholar] [CrossRef]
- Pan, J.; Alimujiang, M.; Chen, Q.; Shi, H.; Luo, X. Exosomes Derived from MiR-146a-modified Adipose-derived Stem Cells Attenuate Acute Myocardial Infarction−induced Myocardial Damage via Downregulation of Early Growth Response Factor 1. J. Cell Biochem. 2019, 120, 4433–4443. [Google Scholar] [CrossRef]
- Zhu, L.-P.; Tian, T.; Wang, J.-Y.; He, J.-N.; Chen, T.; Pan, M.; Xu, L.; Zhang, H.; Qiu, X.-T.; Li, C.-C.; et al. Hypoxia-Elicited Mesenchymal Stem Cell-Derived Exosomes Facilitates Cardiac Repair through MiR-125b-Mediated Prevention of Cell Death in Myocardial Infarction. Theranostics 2018, 8, 6163–6177. [Google Scholar] [CrossRef]
- Huang, L.; Yang, L.; Ding, Y.; Jiang, X.; Xia, Z.; You, Z. Human Umbilical Cord Mesenchymal Stem Cells-Derived Exosomes Transfers MicroRNA-19a to Protect Cardiomyocytes from Acute Myocardial Infarction by Targeting SOX6. Cell Cycle 2020, 19, 339–353. [Google Scholar] [CrossRef]
- Peng, Y.; Zhao, J.-L.; Peng, Z.-Y.; Xu, W.-F.; Yu, G.-L. Exosomal MiR-25-3p from Mesenchymal Stem Cells Alleviates Myocardial Infarction by Targeting pro-Apoptotic Proteins and EZH2. Cell Death Dis. 2020, 11, 317. [Google Scholar] [CrossRef]
- Sun, X.-H.; Wang, X.; Zhang, Y.; Hui, J. Exosomes of Bone-Marrow Stromal Cells Inhibit Cardiomyocyte Apoptosis under Ischemic and Hypoxic Conditions via MiR-486-5p Targeting the PTEN/PI3K/AKT Signaling Pathway. Thromb. Res. 2019, 177, 23–32. [Google Scholar] [CrossRef]
- Thankam, F.G.; Agrawal, D.K. Infarct Zone: A Novel Platform for Exosome Trade in Cardiac Tissue Regeneration. J. Cardiovasc. Transl. Res. 2020, 13, 686–701. [Google Scholar] [CrossRef] [PubMed]
- Arroyo-Campuzano, M.; Gil-Hernández, A.; Silva-Palacios, A. Cardiosome-Mediated Protection in Myocardial Ischemia. Clin. Chim. Acta 2023, 545, 117374. [Google Scholar] [CrossRef]
- Morelli, M.B.; Shu, J.; Sardu, C.; Matarese, A.; Santulli, G. Cardiosomal MicroRNAs Are Essential in Post-Infarction Myofibroblast Phenoconversion. Int. J. Mol. Sci. 2019, 21, 201. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Huang, M.; Wu, J.; Jiang, Q.; Zheng, X. Exosomes Isolated from the Plasma of Remote Ischemic Conditioning Rats Improved Cardiac Function and Angiogenesis after Myocardial Infarction through Targeting Hsp70. Aging 2020, 12, 3682–3693. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Li, X.; Li, T.; Zhao, L.; He, J.; Zha, L.; Qi, Q.; Yu, Z. MicroRNA-423-3p Exosomes Derived from Cardiac Fibroblasts Mediates the Cardioprotective Effects of Ischaemic Post-Conditioning. Cardiovasc. Res. 2019, 115, 1189–1204. [Google Scholar] [CrossRef] [PubMed]
- Barile, L.; Moccetti, T.; Marbán, E.; Vassalli, G. Roles of Exosomes in Cardioprotection. Eur. Heart J. 2016, 38, 1372–1379. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-F.; Mandel, E.M.; Thomson, J.M.; Wu, Q.; Callis, T.E.; Hammond, S.M.; Conlon, F.L.; Wang, D.-Z. The Role of MicroRNA-1 and MicroRNA-133 in Skeletal Muscle Proliferation and Differentiation. Nat. Genet. 2006, 38, 228–233. [Google Scholar] [CrossRef]
- Liu, B.; Lee, B.W.; Nakanishi, K.; Villasante, A.; Williamson, R.; Metz, J.; Kim, J.; Kanai, M.; Bi, L.; Brown, K.; et al. Cardiac Recovery via Extended Cell-Free Delivery of Extracellular Vesicles Secreted by Cardiomyocytes Derived from Induced Pluripotent Stem Cells. Nat. Biomed. Eng. 2018, 2, 293–303. [Google Scholar] [CrossRef]
- Beg, F.; Wang, R.; Saeed, Z.; Devaraj, S.; Masoor, K.; Nakshatri, H. Inflammation-Associated MicroRNA Changes in Circulating Exosomes of Heart Failure Patients. BMC Res. Notes 2017, 10, 751. [Google Scholar] [CrossRef] [PubMed]
- Xitong, D.; Xiaorong, Z. Targeted Therapeutic Delivery Using Engineered Exosomes and Its Applications in Cardiovascular Diseases. Gene 2016, 575, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Ohno, S.; Takanashi, M.; Sudo, K.; Ueda, S.; Ishikawa, A.; Matsuyama, N.; Fujita, K.; Mizutani, T.; Ohgi, T.; Ochiya, T.; et al. Systemically Injected Exosomes Targeted to EGFR Deliver Antitumor MicroRNA to Breast Cancer Cells. Mol. Ther. 2013, 21, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Erviti, L.; Seow, Y.; Yin, H.; Betts, C.; Lakhal, S.; Wood, M.J.A. Delivery of SiRNA to the Mouse Brain by Systemic Injection of Targeted Exosomes. Nat. Biotechnol. 2011, 29, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Ling, X.; Yang, Y.; Zhang, J.; Li, Q.; Niu, X.; Hu, G.; Chen, B.; Li, H.; Wang, Y.; et al. Embryonic Stem Cells-Derived Exosomes Endowed with Targeting Properties as Chemotherapeutics Delivery Vehicles for Glioblastoma Therapy. Adv. Sci. 2019, 6, 1801899. [Google Scholar] [CrossRef]
- De Abreu, R.C.; Fernandes, H.; da Costa Martins, P.A.; Sahoo, S.; Emanueli, C.; Ferreira, L. Native and Bioengineered Extracellular Vesicles for Cardiovascular Therapeutics. Nat. Rev. Cardiol. 2020, 17, 685–697. [Google Scholar] [CrossRef] [PubMed]
- Nakase, I.; Noguchi, K.; Fujii, I.; Futaki, S. Vectorization of Biomacromolecules into Cells Using Extracellular Vesicles with Enhanced Internalization Induced by Macropinocytosis. Sci. Rep. 2016, 6, 34937. [Google Scholar] [CrossRef] [PubMed]
- Aslan, C.; Kiaie, S.H.; Zolbanin, N.M.; Lotfinejad, P.; Ramezani, R.; Kashanchi, F.; Jafari, R. Exosomes for MRNA Delivery: A Novel Biotherapeutic Strategy with Hurdles and Hope. BMC Biotechnol. 2021, 21, 20. [Google Scholar] [CrossRef]
- Armstrong, J.P.K.; Stevens, M.M. Strategic Design of Extracellular Vesicle Drug Delivery Systems. Adv. Drug Deliv. Rev. 2018, 130, 12–16. [Google Scholar] [CrossRef]
- Gallet, R.; Dawkins, J.; Valle, J.; Simsolo, E.; de Couto, G.; Middleton, R.; Tseliou, E.; Luthringer, D.; Kreke, M.; Smith, R.R.; et al. Exosomes Secreted by Cardiosphere-Derived Cells Reduce Scarring, Attenuate Adverse Remodelling, and Improve Function in Acute and Chronic Porcine Myocardial Infarction. Eur. Heart J. 2016, 38, 201–211. [Google Scholar] [CrossRef]
- Hu, W.; Yang, C.; Guo, X.; Wu, Y.; Loh, X.J.; Li, Z.; Wu, Y.-L.; Wu, C. Research Advances of Injectable Functional Hydrogel Materials in the Treatment of Myocardial Infarction. Gels 2022, 8, 423. [Google Scholar] [CrossRef] [PubMed]
- Lv, K.; Li, Q.; Zhang, L.; Wang, Y.; Zhong, Z.; Zhao, J.; Lin, X.; Wang, J.; Zhu, K.; Xiao, C.; et al. Incorporation of Small Extracellular Vesicles in Sodium Alginate Hydrogel as a Novel Therapeutic Strategy for Myocardial Infarction. Theranostics 2019, 9, 7403–7416. [Google Scholar] [CrossRef]
- Zhang, Y.; Cai, Z.; Shen, Y.; Lu, Q.; Gao, W.; Zhong, X.; Yao, K.; Yuan, J.; Liu, H. Hydrogel-Load Exosomes Derived from Dendritic Cells Improve Cardiac Function via Treg Cells and the Polarization of Macrophages Following Myocardial Infarction. J. Nanobiotechnol. 2021, 19, 271. [Google Scholar] [CrossRef] [PubMed]
- Cheng, G.; Zhu, D.; Huang, K.; Caranasos, T.G. Minimally Invasive Delivery of a Hydrogel-Based Exosome Patch to Prevent Heart Failure. J. Mol. Cell Cardiol. 2022, 169, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Cui, X.; Zhang, Z.; Xu, Y.; Guo, J.; Soliman, B.G.; Lu, Y.; Qin, Z.; Wang, Q.; Zhang, H.; et al. Injection-Free Delivery of MSC-Derived Extracellular Vesicles for Myocardial Infarction Therapeutics. Adv. Healthc. Mater. 2022, 11, 2100312. [Google Scholar] [CrossRef] [PubMed]
- Gil-Cabrerizo, P.; Saludas, L.; Prósper, F.; Abizanda, G.; Echanove-González de Anleo, M.; Ruiz-Villalba, A.; Garbayo, E.; Blanco-Prieto, M.J. Development of an Injectable Alginate-Collagen Hydrogel for Cardiac Delivery of Extracellular Vesicles. Int. J. Pharm. 2022, 629, 122356. [Google Scholar] [CrossRef]
- Gómez-Cid, L.; López-Donaire, M.L.; Velasco, D.; Marín, V.; González, M.I.; Salinas, B.; Cussó, L.; García, Á.; Bravo, S.B.; Fernández-Santos, M.E.; et al. Cardiac Extracellular Matrix Hydrogel Enriched with Polyethylene Glycol Presents Improved Gelation Time and Increased On-Target Site Retention of Extracellular Vesicles. Int. J. Mol. Sci. 2021, 22, 9226. [Google Scholar] [CrossRef]
- Monguió-Tortajada, M.; Prat-Vidal, C.; Moron-Font, M.; Clos-Sansalvador, M.; Calle, A.; Gastelurrutia, P.; Cserkoova, A.; Morancho, A.; Ramírez, M.Á.; Rosell, A.; et al. Local Administration of Porcine Immunomodulatory, Chemotactic and Angiogenic Extracellular Vesicles Using Engineered Cardiac Scaffolds for Myocardial Infarction. Bioact. Mater. 2021, 6, 3314–3327. [Google Scholar] [CrossRef]
- Chen, C.W.; Wang, L.L.; Zaman, S.; Gordon, J.; Arisi, M.F.; Venkataraman, C.M.; Chung, J.J.; Hung, G.; Gaffey, A.C.; Spruce, L.A.; et al. Sustained Release of Endothelial Progenitor Cell-Derived Extracellular Vesicles from Shear-Thinning Hydrogels Improves Angiogenesis and Promotes Function after Myocardial Infarction. Cardiovasc. Res. 2018, 114, 1029–1040. [Google Scholar] [CrossRef]
- Cattelan, G.; Guerrero Gerbolés, A.; Foresti, R.; Pramstaller, P.P.; Rossini, A.; Miragoli, M.; Caffarra Malvezzi, C. Alginate Formulations: Current Developments in the Race for Hydrogel-Based Cardiac Regeneration. Front. Bioeng. Biotechnol. 2020, 8, 414. [Google Scholar] [CrossRef]
- Plotkin, M.; Vaibavi, S.R.; Rufaihah, A.J.; Nithya, V.; Wang, J.; Shachaf, Y.; Kofidis, T.; Seliktar, D. The Effect of Matrix Stiffness of Injectable Hydrogels on the Preservation of Cardiac Function after a Heart Attack. Biomaterials 2014, 35, 1429–1438. [Google Scholar] [CrossRef]
- Kharkar, P.M.; Kiick, K.L.; Kloxin, A.M. Designing Degradable Hydrogels for Orthogonal Control of Cell Microenvironments. Chem. Soc. Rev. 2013, 42, 7335–7372. [Google Scholar] [CrossRef] [PubMed]
- Raia, N.R.; Partlow, B.P.; McGill, M.; Kimmerling, E.P.; Ghezzi, C.E.; Kaplan, D.L. Enzymatically Crosslinked Silk-Hyaluronic Acid Hydrogels. Biomaterials 2017, 131, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Bonafè, F.; Govoni, M.; Giordano, E.; Caldarera, C.M.; Guarnieri, C.; Muscari, C. Hyaluronan and Cardiac Regeneration. J. Biomed. Sci. 2014, 21, 100. [Google Scholar] [CrossRef] [PubMed]
- Bu, W.; Wu, Y.; Ghaemmaghami, A.M.; Sun, H.; Mata, A. Rational Design of Hydrogels for Immunomodulation. Regen. Biomater. 2022, 9, rbac009. [Google Scholar] [CrossRef] [PubMed]
- Patil, V.A.; Masters, K.S. Engineered Collagen Matrices. Bioengineering 2020, 7, 163. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Li, Q.; Chen, P.; Duan, Q.; Zhan, J.; Cai, X.; Wang, L.; Hou, H.; Qiu, X. A Smart Adhesive Janus Hydrogel for Non-Invasive Cardiac Repair and Tissue Adhesion Prevention. Nat. Commun. 2022, 13, 7666. [Google Scholar] [CrossRef] [PubMed]
- Klotz, B.J.; Gawlitta, D.; Rosenberg, A.J.W.P.; Malda, J.; Melchels, F.P.W. Gelatin-Methacryloyl Hydrogels: Towards Biofabrication-Based Tissue Repair. Trends Biotechnol. 2016, 34, 394–407. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Kaur, H. Sprayed In-Situ Synthesis of Polyvinyl Alcohol/Chitosan Loaded Silver Nanocomposite Hydrogel for Improved Antibacterial Effects. Int. J. Biol. Macromol. 2020, 145, 950–964. [Google Scholar] [CrossRef]
- Ganguly, S.; Das, P.; Maity, P.P.; Mondal, S.; Ghosh, S.; Dhara, S.; Das, N.C. Green Reduced Graphene Oxide Toughened Semi-IPN Monolith Hydrogel as Dual Responsive Drug Release System: Rheological, Physicomechanical, and Electrical Evaluations. J. Phys. Chem. B 2018, 122, 7201–7218. [Google Scholar] [CrossRef]
- Ohsedo, Y.; Ueno, W. Creation of Polymer Hydrogelator/Poly(Vinyl Alcohol) Composite Molecular Hydrogel Materials. Gels 2023, 9, 679. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Yao, D.; Guo, R.; Deng, L.; Dong, A.; Zhang, J. Composites of Polymer Hydrogels and Nanoparticulate Systems for Biomedical and Pharmaceutical Applications. Nanomaterials 2015, 5, 2054–2130. [Google Scholar] [CrossRef] [PubMed]
- KC, P.; Hong, Y.; Zhang, G. Cardiac Tissue-Derived Extracellular Matrix Scaffolds for Myocardial Repair: Advantages and Challenges. Regen. Biomater. 2019, 6, 185–199. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chen, X.; Hong, H.; Hu, R.; Liu, J.; Liu, C. Decellularized Extracellular Matrix Scaffolds: Recent Trends and Emerging Strategies in Tissue Engineering. Bioact. Mater. 2022, 10, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Sun, Q.; Li, Q.; Kawazoe, N.; Chen, G. Functional Hydrogels with Tunable Structures and Properties for Tissue Engineering Applications. Front. Chem. 2018, 6, 499. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Duan, L.; Zhang, Y.; Cao, J.; Zhang, K. Current Hydrogel Advances in Physicochemical and Biological Response-Driven Biomedical Application Diversity. Signal Transduct. Target. Ther. 2021, 6, 426. [Google Scholar] [CrossRef] [PubMed]
- Cambier, L.; Giani, J.F.; Liu, W.; Ijichi, T.; Echavez, A.K.; Valle, J.; Marbán, E. Angiotensin II–Induced End-Organ Damage in Mice Is Attenuated by Human Exosomes and by an Exosomal Y RNA Fragment. Hypertension 2018, 72, 370–380. [Google Scholar] [CrossRef]
- Monguió-Tortajada, M.; Prat-Vidal, C.; Martínez-Falguera, D.; Teis, A.; Soler-Botija, C.; Courageux, Y.; Munizaga-Larroudé, M.; Moron-Font, M.; Bayes-Genis, A.; Borràs, F.E.; et al. Acellular Cardiac Scaffolds Enriched with MSC-Derived Extracellular Vesicles Limit Ventricular Remodelling and Exert Local and Systemic Immunomodulation in a Myocardial Infarction Porcine Model. Theranostics 2022, 12, 4656–4670. [Google Scholar] [CrossRef]
- Li, G.; Chen, T.; Dahlman, J.; Eniola-Adefeso, L.; Ghiran, I.C.; Kurre, P.; Lam, W.A.; Lang, J.K.; Marbán, E.; Martín, P.; et al. Current Challenges and Future Directions for Engineering Extracellular Vesicles for Heart, Lung, Blood and Sleep Diseases. J. Extracell. Vesicles 2023, 12, 12305. [Google Scholar] [CrossRef]
- Alamdari, S.G.; Alibakhshi, A.; de la Guardia, M.; Baradaran, B.; Mohammadzadeh, R.; Amini, M.; Kesharwani, P.; Mokhtarzadeh, A.; Oroojalian, F.; Sahebkar, A. Conductive and Semiconductive Nanocomposite-Based Hydrogels for Cardiac Tissue Engineering. Adv. Healthc. Mater. 2022, 11, 202200526. [Google Scholar] [CrossRef]
- Fan, L.; Liu, C.; Chen, X.; Zheng, L.; Zou, Y.; Wen, H.; Guan, P.; Lu, F.; Luo, Y.; Tan, G.; et al. Exosomes-loaded Electroconductive hydrogel synergistically promotes tissue repair after spinal cord injury via Immunoregulation and enhancement of myelinated axon growth. Adv. Sci. 2022, 9, 2105586. [Google Scholar] [CrossRef]

| Target Cell | Mechanism Of Repair/Regeneration and Mediators | References |
|---|---|---|
| Mesenchymal stem cells | Anti-inflammatory (miR-182, miR-233, miR-181c, miR-19a, miR-22, miR-199a, miR-214), anti-fibrotic (miR-19a, miR-29, miR-133) and pro-angiogenic (miR-126, miR-210, miR-20a, VEGF) processes facilitating repair and regeneration and inhibits the formation of fibrotic scar tissue. | [27] |
| Cardiomyocytes | Cell growth (miR-17, miR-20a, miR-23b) under normal conditions, and enhanced angiogenesis and decreased collagen deposition (miR-16, miR-19a, miR-19b, miR-23a, miR-23b) under stress. | [33] |
| Cardiosphere-derived cells | Pro-angiogenic and pro-apoptotic properties (miR-210, miR-132, miR-146a-3p). | [26] |
| Endothelial cells | Reduce inflammation and facilitate healing (integrin avβ6). | [40] |
| Adipose-derived stem cells | Anti-inflammatory properties (miR-126) prevent fibrosis and favor angiogenesis, facilitating repair. | [42] |
| Bone marrow-derived stem cells | Neovascularization and vasculogenesis, once implanted into ischemic cardiac tissue. | [43,44] |
| Epicardial adipose tissue-derived stem cells | Upregulation in regenerative properties and proliferative/anti-inflammatory proteins during periods of cellular stress or ischemia, as well as differentiation of cell types. | [51] |
| Hydrogel Materials | EV Source | EVs Retention Time (In Vivo) | Application | References |
|---|---|---|---|---|
| Alginate | MSCs | 14 days | Injection | [82] |
| Alginate | Dendritic cells | 14 days | Injection | [83] |
| Hyaluronic acid (HA) | MSCs | 2 days | Injection | [84] |
| Collagen | iPSCs-derived cardiomyocytes | 7 days | Patch | [70] |
| Gelatin methacryloyl (GelMA) | MSCs | 2 days | Spraying | [85] |
| Alginate and collagen | ADSCs | 7 days | Injection | [86] |
| Cardiac extracellular matrix and polyethylene glycol (PEG) | Cardiospheres | 1 day | Injection | [87] |
| Decellularized cardiac scaffolds and self-assembling peptide hydrogel | MSCs | 6 days | 3D scaffold | [88] |
| Distearoyl phosphoethanolamine, polypeptide, and PEG | Regulatory T cells | 7 days | injection | [84] |
| Adamantane and β-cyclodextrin modified HA | Endothelial progenitor cells | 21 days (in vitro) | injection | [89] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghassemi, K.; Inouye, K.; Takhmazyan, T.; Bonavida, V.; Yang, J.-W.; de Barros, N.R.; Thankam, F.G. Engineered Vesicles and Hydrogel Technologies for Myocardial Regeneration. Gels 2023, 9, 824. https://doi.org/10.3390/gels9100824
Ghassemi K, Inouye K, Takhmazyan T, Bonavida V, Yang J-W, de Barros NR, Thankam FG. Engineered Vesicles and Hydrogel Technologies for Myocardial Regeneration. Gels. 2023; 9(10):824. https://doi.org/10.3390/gels9100824
Chicago/Turabian StyleGhassemi, Kaitlyn, Keiko Inouye, Tatevik Takhmazyan, Victor Bonavida, Jia-Wei Yang, Natan Roberto de Barros, and Finosh G. Thankam. 2023. "Engineered Vesicles and Hydrogel Technologies for Myocardial Regeneration" Gels 9, no. 10: 824. https://doi.org/10.3390/gels9100824
APA StyleGhassemi, K., Inouye, K., Takhmazyan, T., Bonavida, V., Yang, J.-W., de Barros, N. R., & Thankam, F. G. (2023). Engineered Vesicles and Hydrogel Technologies for Myocardial Regeneration. Gels, 9(10), 824. https://doi.org/10.3390/gels9100824