Central Composite Design (CCD) for the Optimisation of Ethosomal Gel Formulation of Punica granatum Extract: In Vitro and In Vivo Evaluations
Abstract
:1. Introduction
2. Results
2.1. Optimisation of Formulation
2.2. Response Analysis through Polynomial Equations
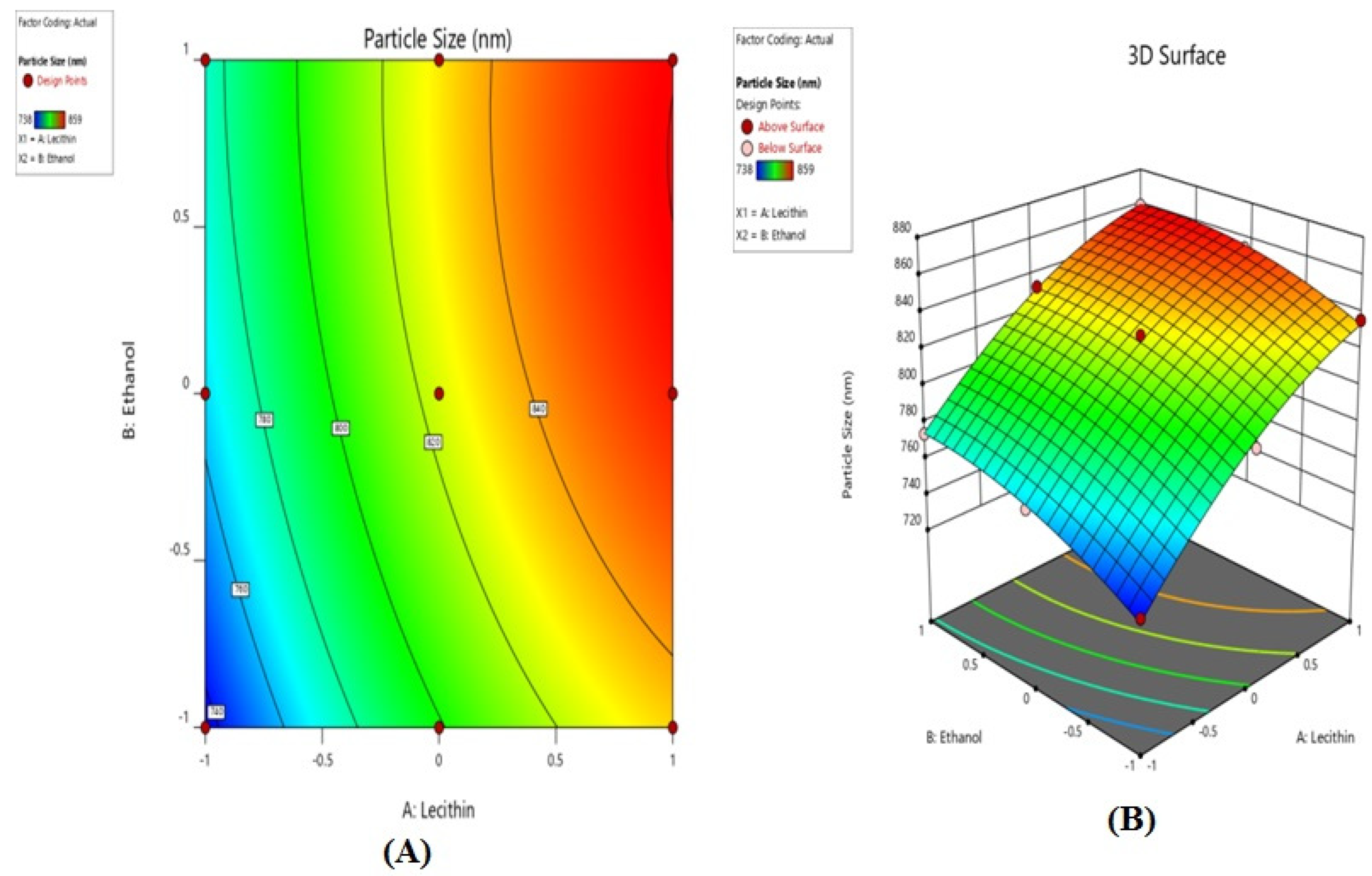
2.2.1. Effect of Variables on Particle Size
2.2.2. Effect of Variables on Entrapment Efficiency
2.3. Gel Punica granatum
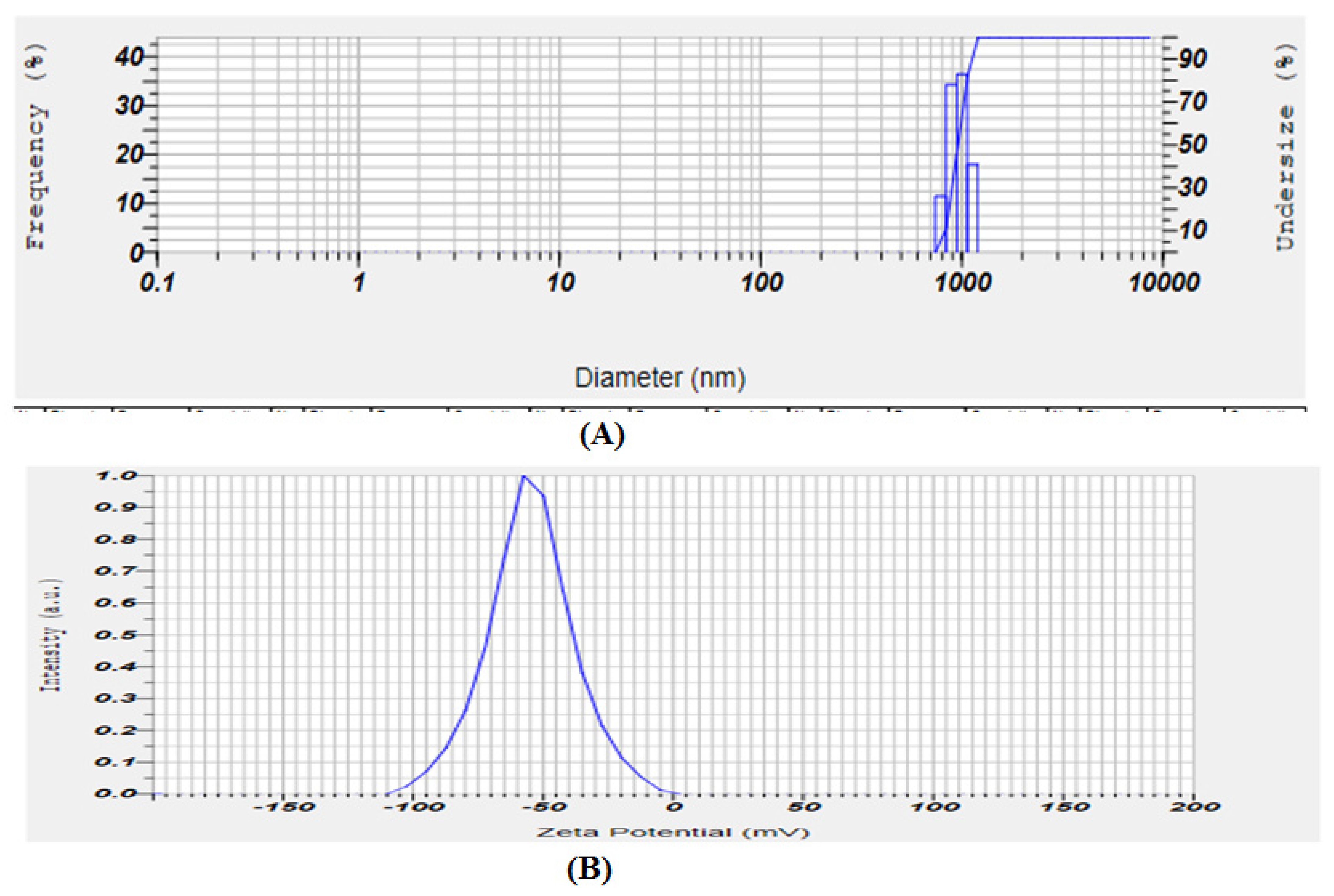
2.3.1. Vesicle Size, Particle Size Distribution, and Zeta Potential (ZP)
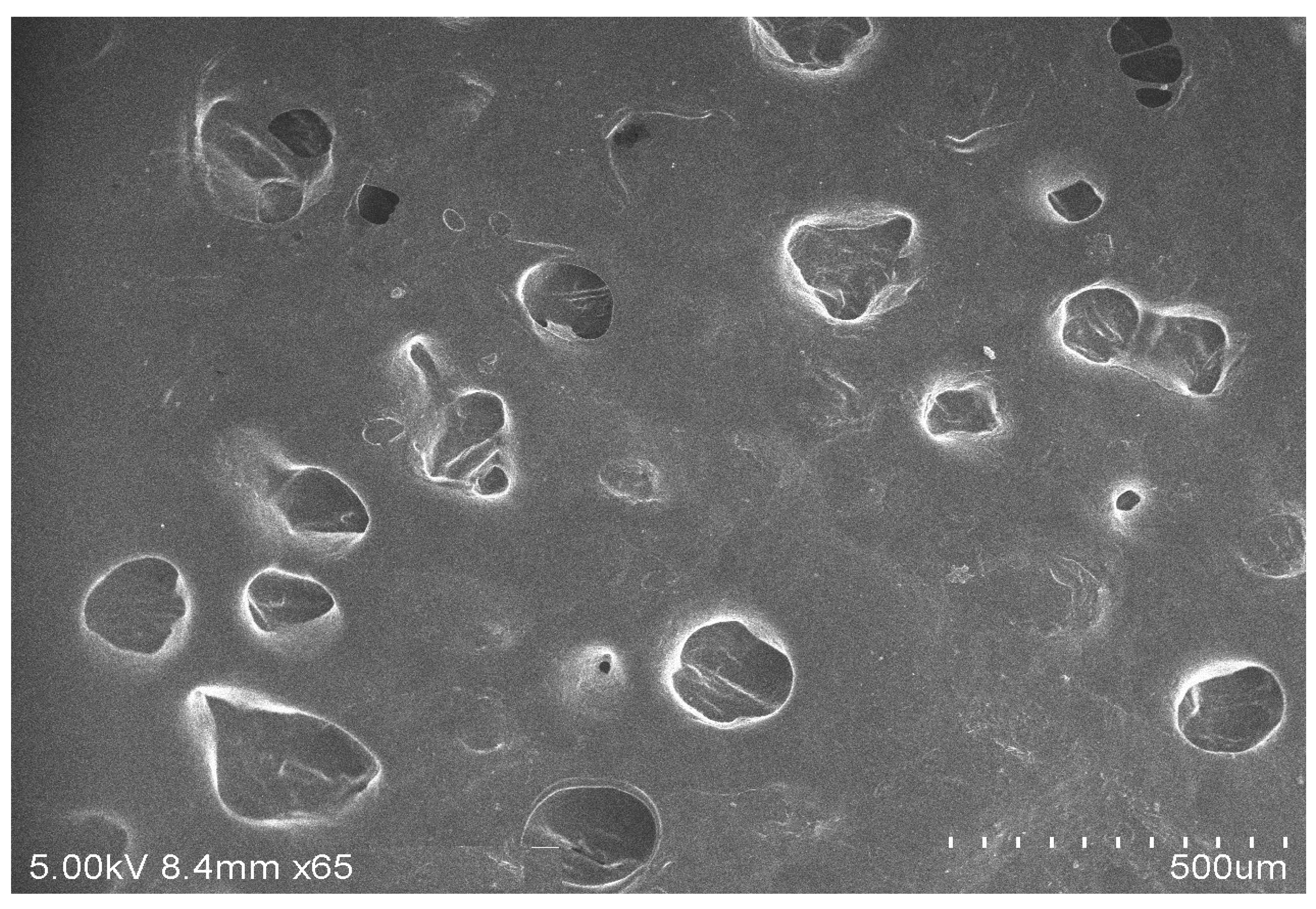
2.3.2. Scanning Electron Microscopy (SEM)
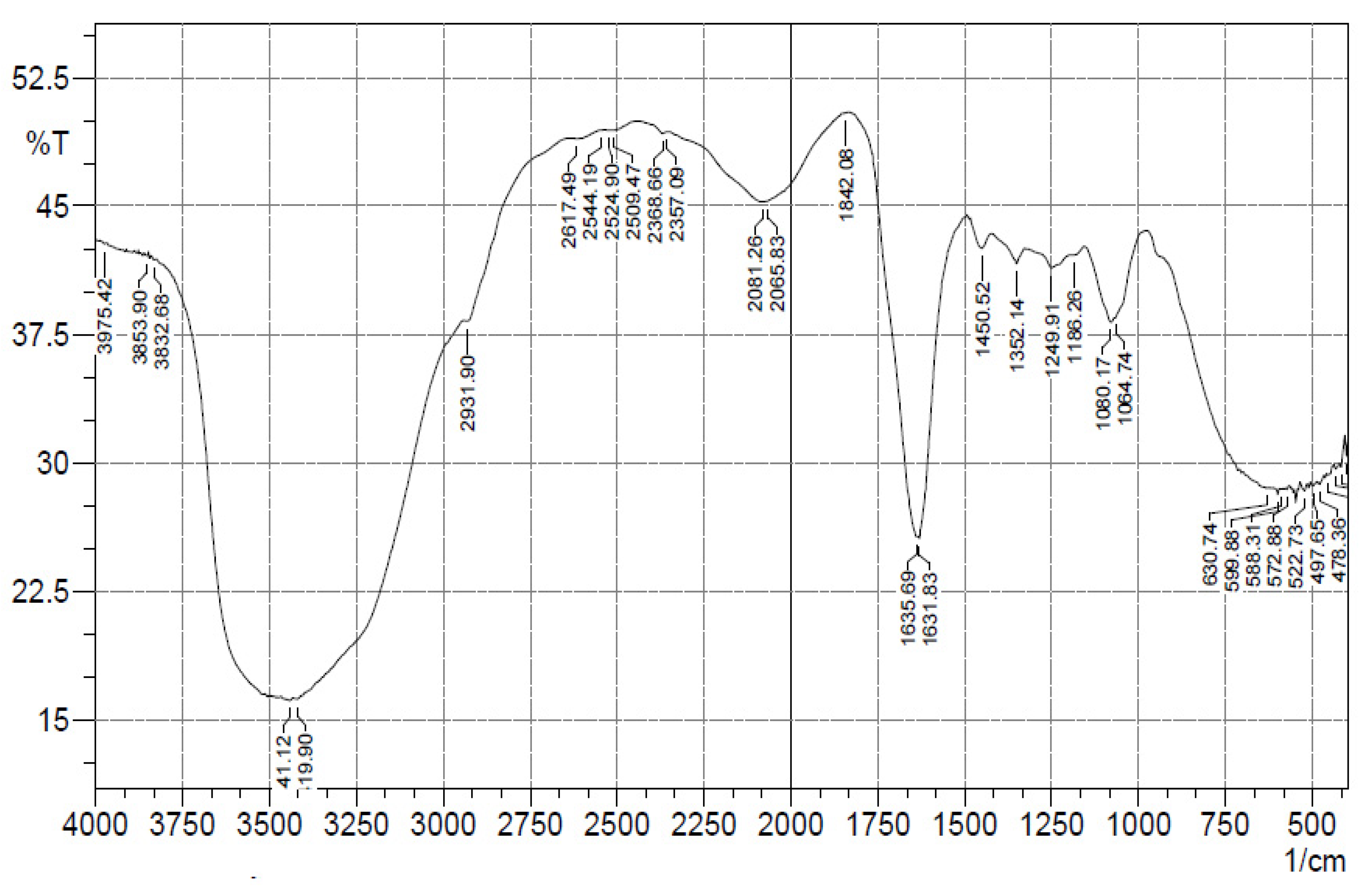
2.3.3. FTIR
2.4. Gel Evaluations
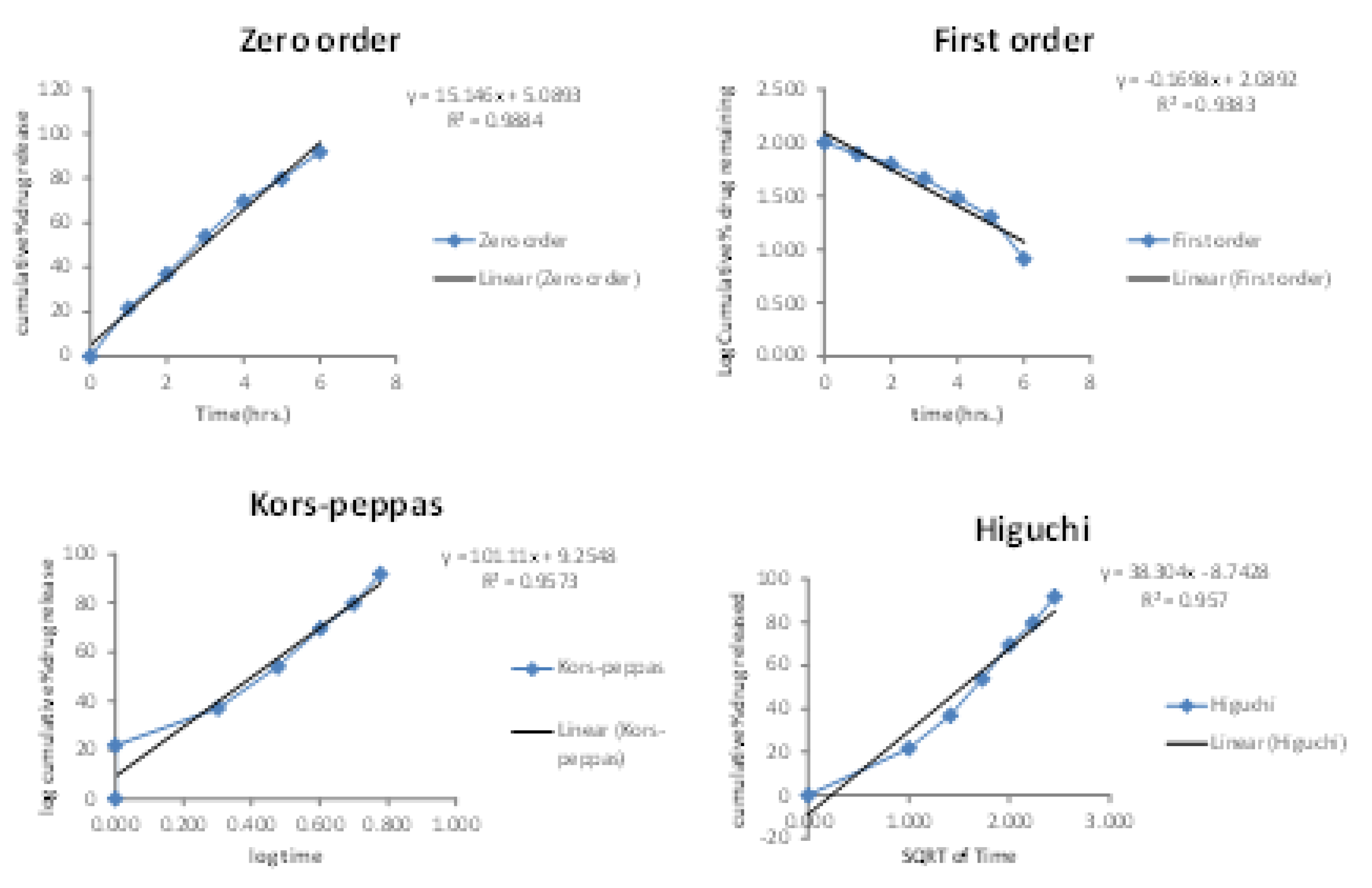
2.5. In Vitro Diffusion Studies
2.6. Stability Studies
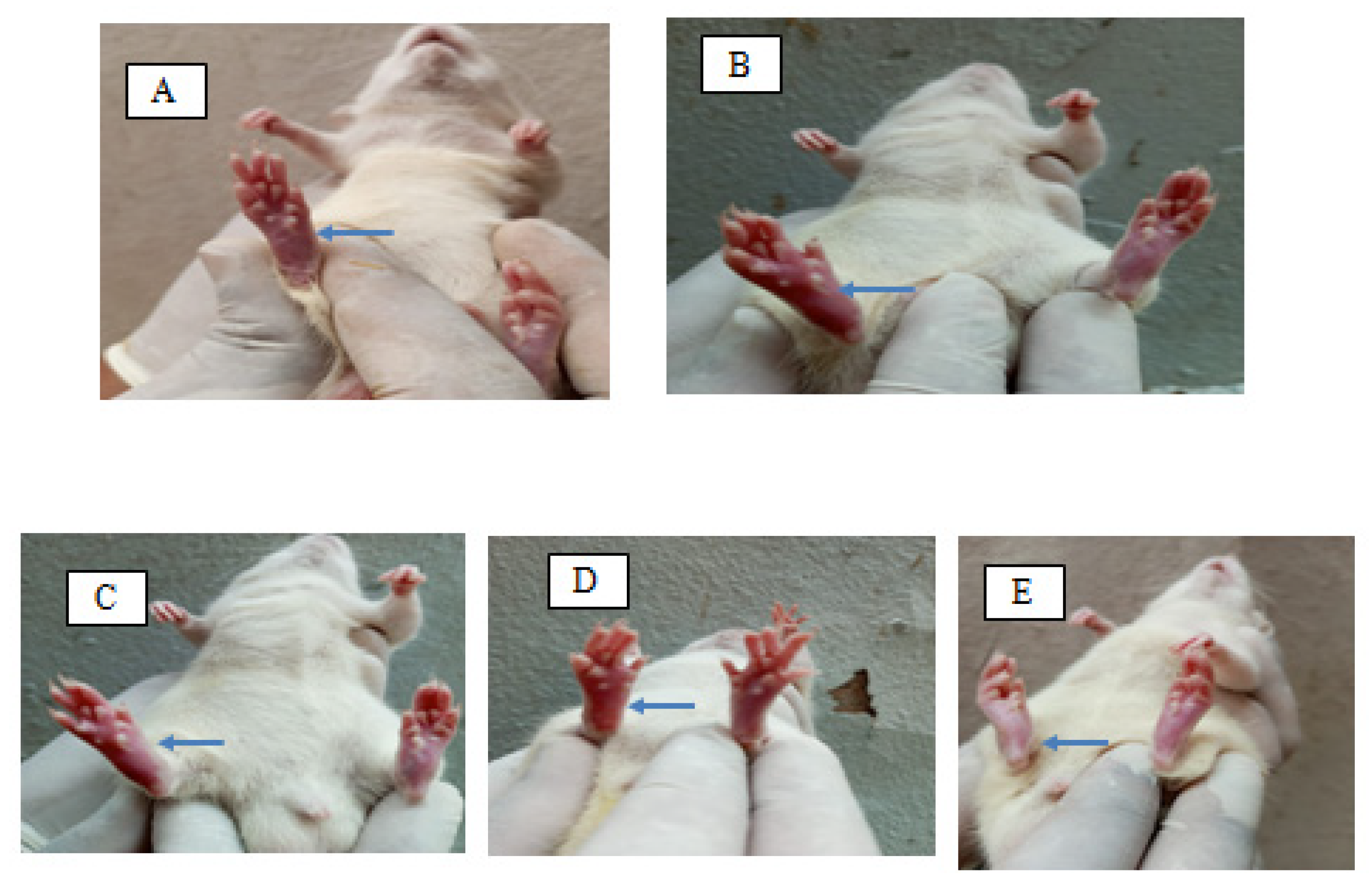
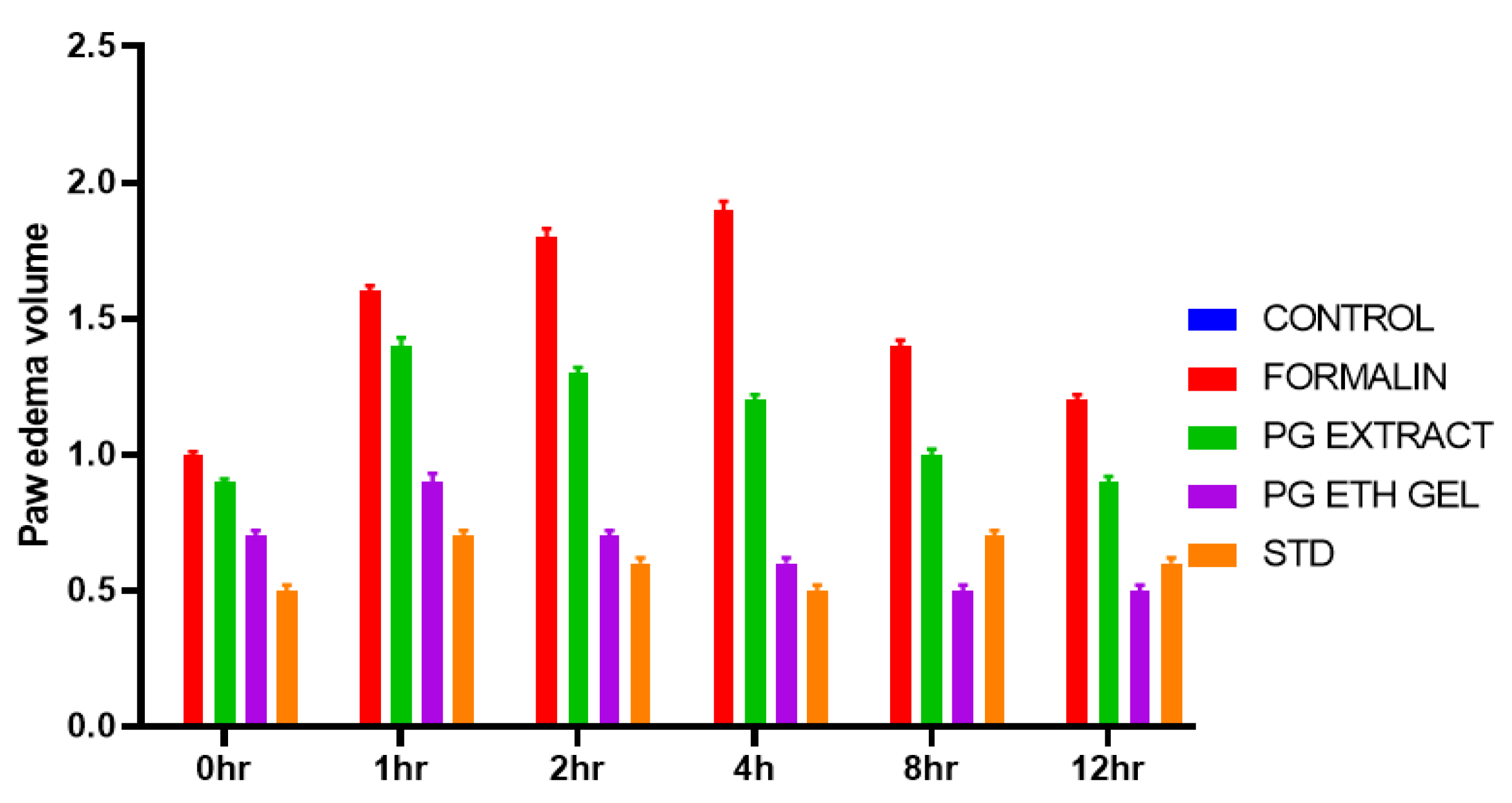
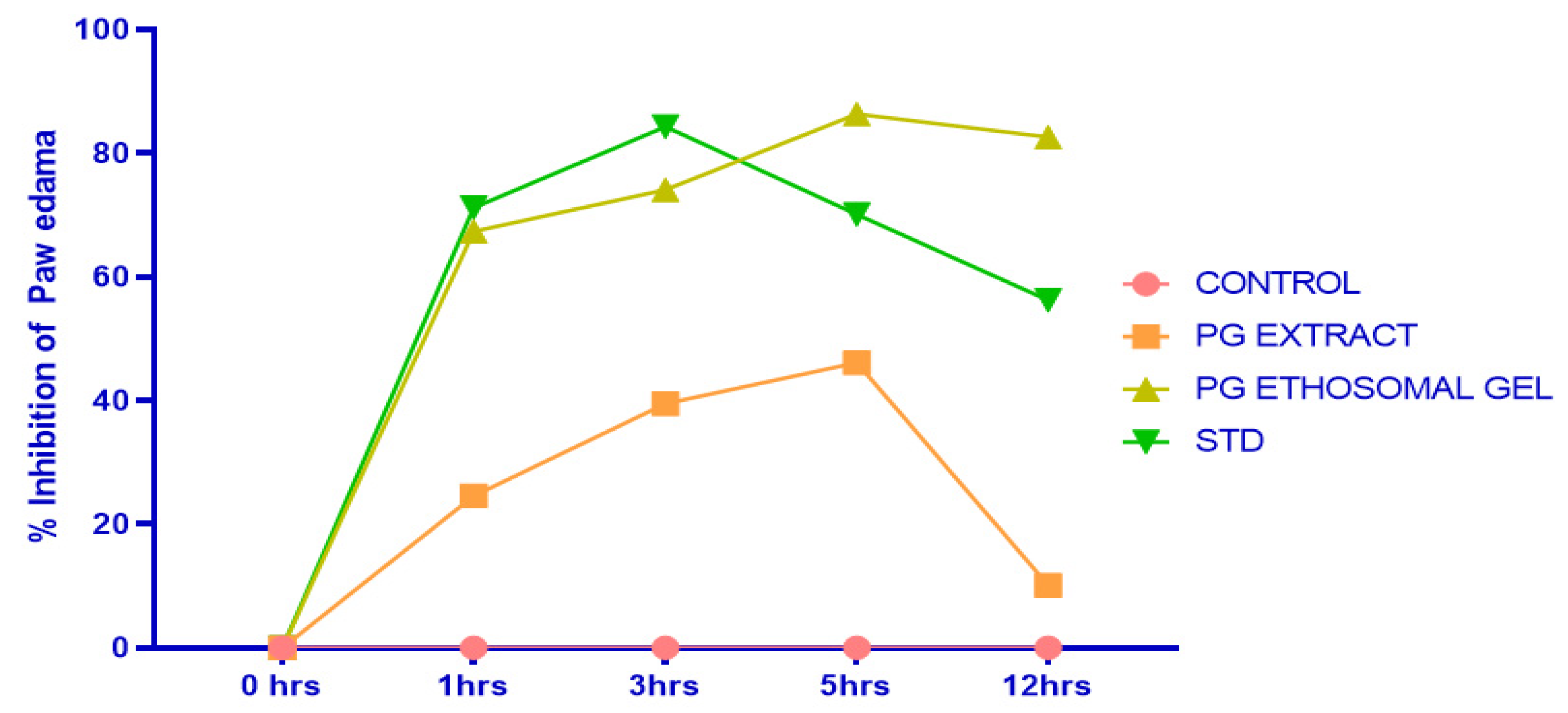
2.7. Formalin-Induced Rat Paw Oedema
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Plant Material and Chemicals
5.2. Instrument Details
5.3. Animals
5.4. Preparation of Peel Extract
5.5. Preparation of Ethosomes Suspensions
5.6. Preparation of Ethosomal Gel
5.7. Experimental Design
5.8. Punica granatum Gel Characterisation
5.8.1. Vesicle Size, Particle Size Distribution, and ZP
5.8.2. SEM
5.8.3. FTIR
5.9. Evaluations of Ethosomal Gel
5.9.1. Viscosity
5.9.2. Spreadability
5.9.3. pH Measurement
5.10. In Vitro Diffusion Studies
5.11. Stability Studies
5.12. Formalin-Induced Albino Rats Paw Oedema
5.13. Statistical Analysis of Results
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Adu-Frimpong, M.; Firempong, C.K.; Omari-Siaw, E.; Wang, Q.; Mukhtar, Y.M.; Deng, W.; Yu, Q.; Xu, X.; Yu, J. Preparation, optimization, and pharmacokinetic study of nanoliposomes loaded with triacylglycerol-bound punicic acid for increased antihepatotoxic activity. Drug Dev. Res. 2019, 80, 230–245. [Google Scholar] [CrossRef] [PubMed]
- Middha, S.K.; Usha, T.; Pande, V. A review on antihyperglycemic and antihepatoprotective activity of eco-friendly Punica granatum peel waste. Evid. Based Compl. Alt. Med. 2013, 2013, 656172. [Google Scholar]
- Khan, J.A.; Hanee, S. Antibacterial properties of Punica granatum peels. Int. J. Appl. Biol. Pharm. Technol. 2011, 2, 23–27. [Google Scholar]
- Dkhil, M.A. Anti-coccidial, anthelmintic and antioxidant activities of pomegranate (Punica granatum) peel extract. Parasitol. Res. 2013, 112, 2639–2646. [Google Scholar] [CrossRef] [PubMed]
- Abdollahzadeh, S.; Mashouf, R.Y.; Moghaddam, M.H.; Roozbahani, N.; Vahedi, M. Antibacterial and antifungal activities of Punica granatum peel extracts against oral pathogens. J. Dent. 2011, 8, 1–6. [Google Scholar]
- Devanesan, S.; AlSalhi, M.S.; Balaji, R.V.; Ranjitsingh, A.J.A.; Ahamed, A.; Alfuraydi, A.A.; AlQahatani, F.Y.; Aleanizy, F.S.; Othman, A.H. Antimicrobial and cytotoxicity effects of synthesized silver nanoparticles from Punica granatum peel extract. Nanoscale Res. Lett. 2018, 13, 315. [Google Scholar] [CrossRef]
- Modaeinama, S.; Abasi, M.; Abbasi, M.M.; Jahanban-Esfahlan, R. Anti tumoral properties of Punica granatum (pomegranate) peel extract on different human cancer cells. Asian Pac. J. Cancer Prev. 2015, 16, 5697–5701. [Google Scholar] [CrossRef]
- Moghaddam, G.; Sharifzadeh, M.; Hassanzadeh, G.; Khanavi, M.; Hajimahmoodi, M. Anti-ulcerogenic activity of the pomegranate peel (Punica granatum) methanol extract. Food Nutr. Sci. 2013, 4, 43–48. [Google Scholar]
- Khan, I.; Rahman, H.; Abd El-Salam, N.M.; Tawab, A.; Hussain, A.; Khan, T.A.; Khan, U.A.; Qasim, M.; Adnan, M.; Azizullah, A.; et al. Punica granatum peel extracts: HPLC fractionation and LC MS analysis to quest compounds having activity against multidrug resistant bacteria. BMC Complement Alt. Med. 2017, 17, E247. [Google Scholar] [CrossRef]
- Wang, D.; Özen, C.; Abu-Reidah, I.M.; Chigurupati, S.; Patra, J.K.; Horbanczuk, J.O.; Jozwik, A.; Tzvetkov, N.T.; Uhrin, P.; Atanasov, A.G. Vasculoprotective effects of pomegranate (Punica granatum L.). Front. Pharmacol. 2018, 9, 544. [Google Scholar] [CrossRef]
- Aslam, S.; Jahan, N.; Khalil-Ur-Rehman; Ali, S. Formulation, optimisation and in-vitro, in-vivo evaluation of surfactant stabilised nanosuspension of Ginkgo biloba. J. Microencapsul. 2019, 36, 576–590. [Google Scholar]
- Saraf, S. Applications of novel drug delivery system for herbal formulations. Fitoterapia 2010, 81, 680–689. [Google Scholar]
- Bailey, M.M.; Berkland, C.J. Nanoparticle formulations in pulmonary drug delivery. Med. Res. Rev. 2009, 29, 196–212. [Google Scholar] [CrossRef] [PubMed]
- Mahdi, W.A.; Alam, P.; Alshetaili, A.; Alshehri, S.; Ghoneim, M.M.; Shakeel, F. Product development studies cranberry seed oil nanoemulsion. Processes 2022, 10, 393. [Google Scholar] [CrossRef]
- Shoaib, A.; Azmi, L.; Pal, S.; Alqahtani, S.S.; Rahamathulla, M.; Hani, U.; Alshehri, S.; Ghoneim, M.M.; Shakeel, F. Integrating nanotechnology with naturally occurring phytochemicals in neuropathy induced by diabetes. J. Mol. Liq. 2022, 350, 118189. [Google Scholar] [CrossRef]
- Moradi, S.Z.; Momtaz, S.; Bayrami, Z.; Farzaei, M.H.; Abdollahi, M. Nanoformulations of herbal extracts in treatment of neurodegenerative disorders. Front. Bioeng. Biotechnol. 2020, 8, 238. [Google Scholar] [CrossRef]
- Khogta, S.; Patel, J.; Barve, K.; Londhe, V. Herbal nano-formulations for topical delivery. J. Herbal Med. 2020, 20, 100300. [Google Scholar] [CrossRef]
- Wickramasinghe, A.S.D.; Kalansuriya, P.; Attanayake, A.P. Nanoformulation of plant-based natural products for type 2 diabetes mellitus: From formulation design to therapeutic applications. Curr. Ther. Res. 2022, 96, 100672. [Google Scholar] [CrossRef]
- Jain, P.; Taleuzzaman, M.; Kala, C.; Kumar Gupta, D.; Ali, A.; Aslam, M. Quality by design (Qbd) assisted development of phytosomal gel of Aloe vera extract for topical delivery. J. Liposome Res. 2021, 31, 381–388. [Google Scholar] [CrossRef]
- Kaur, J.; Anwer, M.K.; Sartaj, A.; Panda, B.P.; Ali, A.; Zafar, A.; Kumar, V.; Gilani, S.J.; Kala, C.; Taleuzzaman, M. ZnO Nanoparticles of Rubia cordifolia extract formulation developed and optimized with QbD application, considering ex vivo skin permeation, antimicrobial and antioxidant properties. Molecules 2022, 27, 1450. [Google Scholar] [CrossRef]
- Mekjaruskul, C.; Sripanidkulchai, B. Kaempferia parviflora nanosuspension formulation for scalability and improvement of dissolution profiles and intestinal absorption. AAPS PharmSciTech. 2019, 21, 52. [Google Scholar] [CrossRef] [PubMed]
- Bose, A.; Wong, T.W.; Singh, N. Formulation development and optimization of sustained release matrix tablet of Itopride HCl by response surface methodology and its evaluation of release kinetics. Saudi Pharm. J. 2013, 21, 201–213. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.O.; Jeong, Y.S. Effects of different concentrations of formalin on paw edema and pain behaviors in rats. J. Korean Med. Sci. 2002, 17, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Németh, Z.; Pallagi, E.; Dobó, D.G.; Kozma, G.; Kónya, Z.; Csóka, I. An updated risk assessment as part of the QbD-based liposome design and development. Pharmaceutics 2021, 13, 1071. [Google Scholar] [CrossRef] [PubMed]
- Baboota, S.; Shakeel, F.; Kohli, K. Formulation and evaluation of once-a-day transdermal gels of diclofenac diethylamine. Methods Find. Exp. Clin. Pharmacol. 2006, 28, 109–114. [Google Scholar] [CrossRef]
- Wong, Y.C.; Ahmad-Mudzaqqir, M.Y.; Wan-Nurdiyana, W.A. Extraction of essential oil from cinnamon (Cinnamomum zeylanicum). Oriental J. Chem. 2014, 30, 37–47. [Google Scholar] [CrossRef]
- Barupal, A.K.; Gupta, V.; Ramteke, S. Preparation and characterization of ethosomes for topical delivery of aceclofenac. Indian J. Pharm. Sci. 2010, 72, 582–586. [Google Scholar]
- Amarachinta, P.R.; Sharma, G.; Samed, N.; Chettupalli, A.K.; Alle, M.; Kim, J.-C. Central composite design for the development of carvedilol-loaded transdermal ethosomal hydrogel for extended and enhanced anti-hypertensive effect. J. Nanobiotechnol. 2021, 19, 100. [Google Scholar] [CrossRef]
- Khan, P.; Akhtar, N. Phytochemical investigations and development of ethosomal gel with Brassica oleraceae L. (Brassicaceae) extract: An innovative nano approach towards cosmetic and pharmaceutical industry. Ind. Crops Prod. 2022, 183, 114905. [Google Scholar] [CrossRef]
- Kumar, R.; Mirza, M.A.; Naseef, P.P.; Kuruniyan, M.S.; Zakir, F.; Aggarwal, G. Exploring the potential of natural product-based nanomedicine for maintaining oral health. Molecules 2022, 27, 1725. [Google Scholar] [CrossRef]
- Sudhakar, K.; Mishra, V.; Jain, S.; Rompicherla, N.C.; Malviya, N.; Tambuwala, M.M. Development and evaluation of the effect of ethanol and surfactant in vesicular carriers on lamivudine permeation through the skin. Int. J. Pharm. 2021, 610, 121226. [Google Scholar] [CrossRef] [PubMed]
- Graily-Moradi, F.; Asgari Lajayer, B. Nanoinsecticides: Preparation, application, and mode of action. In Nanotechnology Applications in Health and Environmental Sciences. Nanotechnology in the Life Sciences; Saglam, N., Korkusuz, F., Prasad, R., Eds.; Springer: Cham, Switzerland, 2021. [Google Scholar]
- Ghidan, A.Y.; Al-Antary, T.M.; Awwad, A.M. Green synthesis of copper oxide nanoparticles using Punica granatum peels extract: Effect on green peach aphid. Env. Nanotechnol. Monit. Manag. 2016, 6, 95–98. [Google Scholar] [CrossRef]
- He, Y.; Du, Z.; Lv, H.; Jia, Q.; Tang, Z.; Zheng, X.; Zhang, K.; Zhao, F. Green synthesis of silver nanoparticles by Chrysanthemum morifolium Ramat. extract and their application in clinical ultrasound gel. Int. J. Nanomed. 2013, 8, 1809–1815. [Google Scholar] [CrossRef]
- Gavan, A.; Colobatiu, L.; Hanganu, D.; Bogdan, C.; Olah, N.K.; Achim, M.; Mirel, S. Development and evaluation of hydrogel wound dressings loaded with herbal extracts. Processes 2022, 10, 242. [Google Scholar] [CrossRef]
- Akhtar, N.; Akhtar, N.; Menaa, F.; Alharbi, W.; Alaryani, F.S.S.; Alqahtani, A.M.; Ahmad, F. Fabrication of ethosomes containing tocopherol acetate to enhance transdermal permeation: In vitro and ex vivo characterizations. Gels 2022, 8, 335. [Google Scholar] [CrossRef]
- Salamanca, C.H.; Barrera-Ocampo, A.; Lasso, J.C.; Camacho, N.; Yarce, C.J. Franz diffusion cell approach for pre-formulation characterisation of ketoprofen semi-solid dosage forms. Pharmaceutics 2018, 10, 148. [Google Scholar] [CrossRef]
- Yusefi, M.; Lee-Kiun Soon, M.; Teow, S.Y.; Monchouguy, E.I.; Neerooa, B.N.H.M.; Izadiyan, Z.; Jahangirian, H.; Rafiee-Moghaddam, R.; Webster, T.J.; Shameli, K. Fabrication of cellulose nanocrystals as potential anticancer drug delivery systems for colorectal cancer treatment. Int. J. Biol. Macromol. 2022, 199, 372–385. [Google Scholar] [CrossRef]
- Ajazuddin, A.A.; Khichariya, A.; Gupta, S.; Patel, R.J.; Giri, T.K.; Tripathi, D.K. Recent expansions in an emergent novel drug delivery technology: Emulgel. J. Control. Release 2013, 171, 122–132. [Google Scholar] [CrossRef]
- Dave, V.; Bhardwaj, N.; Gupta, N.; Tak, K. Herbal ethosomal gel containing luliconazole for productive relevance in the field of biomedicine. 3 Biotech. 2020, 10, 97. [Google Scholar] [CrossRef]
- Bekhit, A.A.; Nasralla, S.N.; El-Agroudy, E.J.; Hamouda, N.; El-Fattah, A.A.; Bekhit, S.A.; Amagase, K.; Ibrahim, T.M. Investigation of the anti-inflammatory and analgesic activities of promising pyrazole derivative. Eur. J. Pharm. Sci. 2022, 168, 106080. [Google Scholar] [CrossRef]
- Kumar, T.; Jain, V. Antinociceptive and anti-Inflammatory activities of Bridelia retusa methanolic fruit extract in experimental animals. Scient. World J. 2014, 2014, 890151. [Google Scholar] [CrossRef] [PubMed]
- Almeida, D.S.; da Silva, D.; Moreira, L.; Menegatti, R.; Lião, L.M.; Sanz, G.; Vaz, B.G.; Ghedini, P.C.; Costa, E.A.; Florentino, I.F. Investigation of anti-inflammatory potential of 5-(3,5-di-tert-butyl-4-hydroxybenzylidene)-2-thioxodihydropyrimidine-4,6(1H,5H)-dione compound. Eur. J. Pharmacol. 2020, 886, 173388. [Google Scholar] [CrossRef] [PubMed]
- Aziz, T.A.; Kareem, A.A.; Othman, H.H.; Ahmed, Z.A. The anti-inflammatory effect of different doses of aliskiren in rat models of inflammation. Drug Des. Dev. Ther. 2020, 14, 2841–2851. [Google Scholar] [CrossRef] [PubMed]
- Eltom, S.E.M.; Abdellatif, A.A.H.; Maswadeh, H.; Al-Omar, M.S.; Abdel-Hafez, A.A.; Mohammed, H.A.; Agabein, E.M.; Alqasoomi, I.; Alrashidi, S.A.; Sajid, M.S.M.; et al. The anti-Inflammatory effect of a γ-lactone isolated from ostrich oil of Struthio camelus (Ratite) and its formulated nano-emulsion in formalin-induced paw edema. Molecules 2021, 26, 3701. [Google Scholar] [CrossRef]



| Coded Values Level | Independent Variables | |
|---|---|---|
| X1, Lecithin | X2, Ethanol | |
| −1 | 1 | 15 |
| 0 | 1.5 | 20 |
| +1 | 2 | 30 |
| Formulation Code | X1, Lecithin | X2, Ethanol | Y1, Particle Size (nm) | Y2, Entrapment Efficiency (%) |
|---|---|---|---|---|
| F1 | 1 | 15 | 738 | 53.51 |
| F2 | 1 | 20 | 762 | 57.57 |
| F3 | 1 | 30 | 774 | 59.20 |
| F4 | 1.5 | 15 | 794 | 57.74 |
| F5 | 1.5 | 20 | 828 | 62.20 |
| F6 | 1.5 | 30 | 832 | 64.25 |
| F7 | 2 | 15 | 836 | 68.39 |
| F8 | 2 | 20 | 854 | 72.20 |
| F9 | 2 | 30 | 859 | 74.12 |
| Formulation | pH | Viscosity (CPS) | Spreadability (g.cm/s) | Grittiness |
|---|---|---|---|---|
| PGE1 | 7.1 | 34,015 | 35.15 | No |
| PGE2 | 7.0 | 35,125 | 33.58 | |
| PGE3 | 6.9 | 38,478 | 31.35 | |
| PGE4 | 6.9 | 30,975 | 36.28 | |
| PGE5 | 6.8 | 35,750 | 34.36 | |
| PGE6 | 6.8 | 36,435 | 33.65 | |
| PGE7 | 7.1 | 34,019 | 35.95 | |
| PGE8 | 7.1 | 33,735 | 32.18 | |
| PGE9 | 7.1 | 32,158 | 31.55 |
| Stability Study Period | Drug Content (%) | |
|---|---|---|
| Initial | 4 ± 2 °C | 98.5 |
| 30 ± 2 °C | 98.6 | |
| After 3 weeks | 4 ± 2 °C | 97.4 |
| 30 ± 2 °C | 96.7 | |
| After 6 weeks | 4 ± 2 °C | 97.2 |
| 30 ± 2 °C | 96.3 | |
| After 9 weeks | 4 ± 2 °C | 97.6 |
| 30 ± 2 °C | 96.3 | |
| After 12 weeks | 4 ± 2 °C | 97.5 |
| 30 ± 2 °C | 96.6 | |
| Groups | 0 h | 1 h | 2 h | 4 h | 8 h | 12 h |
|---|---|---|---|---|---|---|
| Control | 0.0 ± 0 | 0.0 ± 0 | 0.0 ± 0 | 0.0 ± 0 | 0.0 ± 0 | 0.0 ± 0 |
| Formalin | 1.0 ± 0.01 | 1.6 ± 0.02 | 1.8 ± 0.03 | 1.9 ± 0.03 | 1.4 ± 0.02 | 1.2 ± 0.02 |
| PG Extract | 0.9 ± 0.01 | 1.4 ± 0.01 | 1.3 ± 0.01 * | 1.2 ± 0.01 * | 1.0 ± 0.01 * | 0.9 ± 0.01 * |
| PG EthoGel | 0.7 ± 0.01 | 0.9 ± 0.01 ** | 0.7 ± 0.01 ** | 0.6 ± 0.01 *** | 0.5 ± 0.01 *** | 0.5 ± 0.01 ** |
| Standard | 0.5 ± 0.01 | 0.7 ± 0.01 ** | 0.6 ± 0.01 *** | 0.5 ± 0.01 *** | 0.7 ± 0.01 ** | 0.6 ± 0.01 *** |
| Groups | 0 h | 1 h | 3 h | 5 h | 12 h |
|---|---|---|---|---|---|
| Control | 0.0% | 0.0% | 0.0% | 0.0% | 0.0% |
| PG EXTRACT | 0.0% | 24.6% | 39.50% | 46.13% | 10.10% |
| PG ET GEL | 0.0% | 67.40% | 74.10% | 86.34% | 82.64% |
| Standard | 0.0% | 71.20% | 84.30% | 70.12% | 56.24% |
| Quantities in w/w % (100 gm) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Code | EAP Extract | Carbapol 934 (% w/v) gms | Lecithin (w/v) (%) | Ethanol (v/v) mL | Cholesterol (%) | PEG 400 (v/v) mL | TEM (w/v) (%) | Water QS mL |
| EPG1 | 10 | 1.5 | 1 | 15 | 0.2 | 1 | 0.5 | 100 |
| EPG2 | 10 | 1.5 | 1.5 | 15 | 0.2 | 1 | 0.5 | 100 |
| EPG3 | 10 | 1.5 | 2 | 15 | 0.2 | 1 | 0.5 | 100 |
| EPG4 | 10 | 1.5 | 1 | 20 | 0.2 | 1 | 0.5 | 100 |
| EPG5 | 10 | 1.5 | 1.5 | 20 | 0.2 | 1 | 0.5 | 100 |
| EPG6 | 10 | 1.5 | 2 | 20 | 0.2 | 1 | 0.5 | 100 |
| EPG7 | 10 | 1.5 | 1 | 30 | 0.2 | 1 | 0.5 | 100 |
| EPG8 | 10 | 1.5 | 1.5 | 30 | 0.2 | 1 | 0.5 | 100 |
| EPG9 | 10 | 1.5 | 2 | 30 | 0.2 | 1 | 0.5 | 100 |
| Formulation Code | Lecithin (% w/v) | Ethanol (mL) | Cholesterol (%) |
|---|---|---|---|
| F1 | 1 | 15 | 0.2 |
| F2 | 1 | 20 | 0.2 |
| F3 | 1 | 30 | 0.2 |
| F4 | 1.5 | 15 | 0.2 |
| F5 | 1.5 | 20 | 0.2 |
| F6 | 1.5 | 30 | 0.2 |
| F7 | 2 | 15 | 0.2 |
| F8 | 2 | 20 | 0.2 |
| F9 | 2 | 30 | 0.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alam, P.; Shakeel, F.; Foudah, A.I.; Alshehri, S.; Salfi, R.; Alqarni, M.H.; Aljarba, T.M. Central Composite Design (CCD) for the Optimisation of Ethosomal Gel Formulation of Punica granatum Extract: In Vitro and In Vivo Evaluations. Gels 2022, 8, 511. https://doi.org/10.3390/gels8080511
Alam P, Shakeel F, Foudah AI, Alshehri S, Salfi R, Alqarni MH, Aljarba TM. Central Composite Design (CCD) for the Optimisation of Ethosomal Gel Formulation of Punica granatum Extract: In Vitro and In Vivo Evaluations. Gels. 2022; 8(8):511. https://doi.org/10.3390/gels8080511
Chicago/Turabian StyleAlam, Prawez, Faiyaz Shakeel, Ahmed I. Foudah, Sultan Alshehri, Roshan Salfi, Mohammed H. Alqarni, and Tariq M. Aljarba. 2022. "Central Composite Design (CCD) for the Optimisation of Ethosomal Gel Formulation of Punica granatum Extract: In Vitro and In Vivo Evaluations" Gels 8, no. 8: 511. https://doi.org/10.3390/gels8080511








