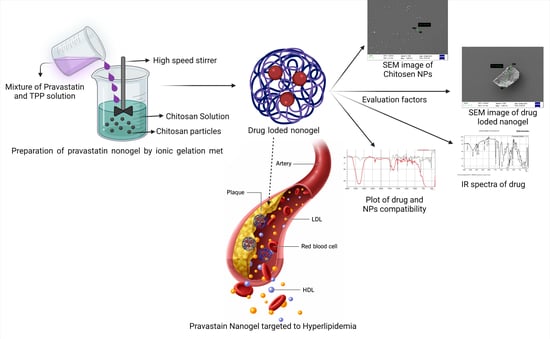
Formulation Development and Evaluation of Pravastatin-Loaded Nanogel for Hyperlipidemia Management
Abstract
:1. Introduction
2. Results and Discussion
2.1. Experimental Design for Optimization of Nanoparticles
2.2. Characterization of Optimized Nanoparticles
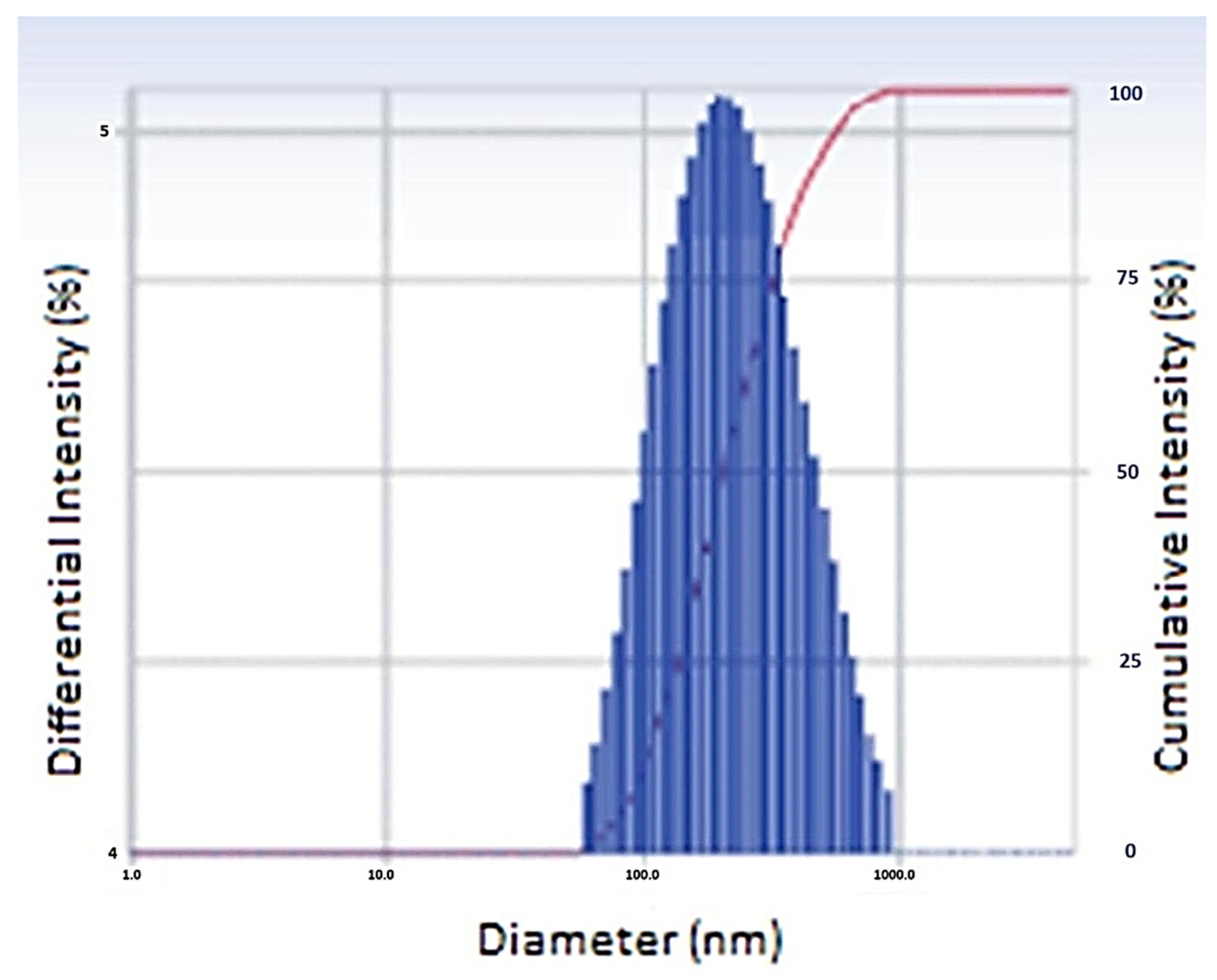
2.2.1. Particle Size
2.2.2. Entrapment Efficiency
2.2.3. Scanning Electron Microscopy (SEM) of Nanoparticles
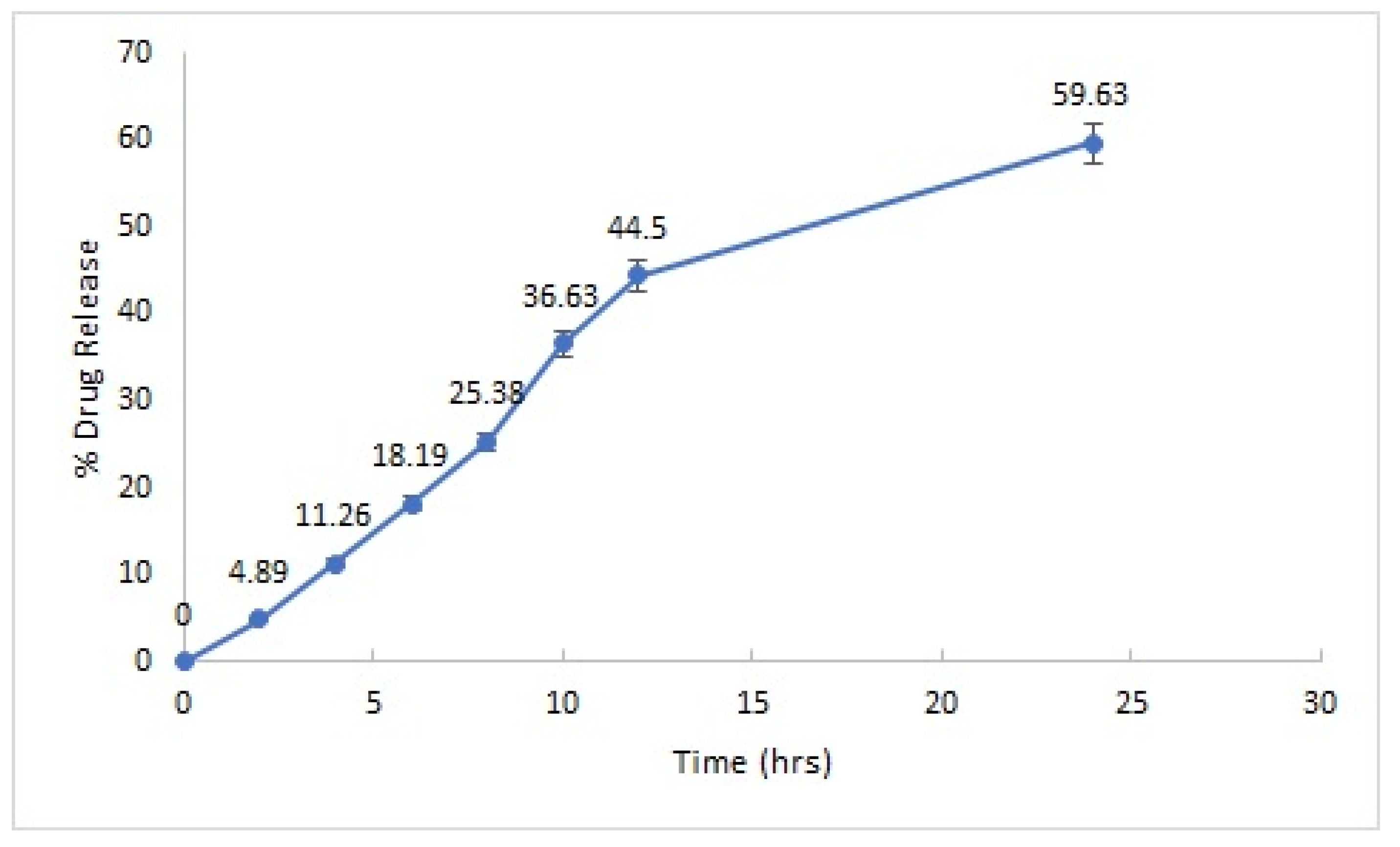
2.2.4. In Vitro Drug Release
2.2.5. Swelling Ratio
2.2.6. SEM Image of Structural Network of Nanogel
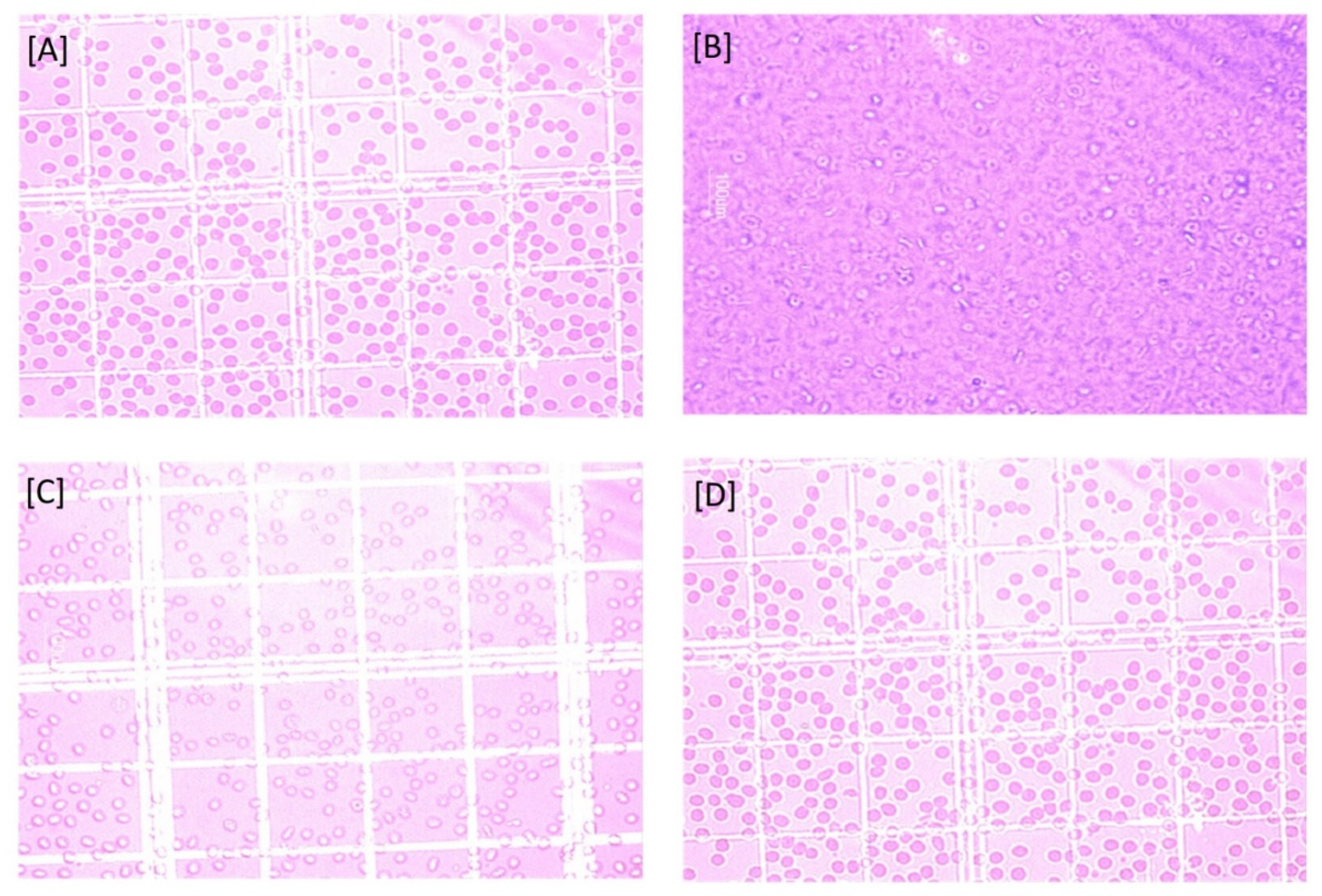
2.2.7. Effect on Surface Morphology of Erythrocytes
2.2.8. Hemolytic Toxicity
2.2.9. Pharmacokinetic Studies
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Methods
4.2.1. Preparation of Pravastatin-Loaded Chitosan Nanogel
4.2.2. Experimental Design for Optimization of Nanogel
4.2.3. Characterization and Evaluation of Optimized Nanoparticles/Nanogel
Particle Size and Zeta Potential
Entrapment Efficiency
Morphology Observation
Fourier-Transform Infrared Spectroscopy
In Vitro Drug Release
Swelling Ratio
Hemolytic Toxicity
Effect on Surface Morphology of Erythrocytes
Pharmacokinetics Studies
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ghaywat, S.D.; Mate, P.S.; Parsutkar, Y.M.; Chandimeshram, A.D.; Umekar, M.J. Overview of nanogel and its applications. GSC Biol. Pharm. Sci. 2021, 16, 040–061. [Google Scholar] [CrossRef]
- Ansari, S.; Karimi, M. Novel developments and trends of analytical methods for drug analysis in biological and environmental samples by molecularly imprinted polymers. TrAC Trends Anal. Chem. 2017, 89, 146–162. [Google Scholar] [CrossRef]
- Sivaram, A.J.; Rajitha, P.; Maya, S.; Jayakumar, R.; Sabitha, M. Nanogels for delivery, imaging and therapy. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2015, 7, 509–533. [Google Scholar] [CrossRef] [PubMed]
- Vashist, A.; Kaushik, A.; Vashist, A.; Bala, J.; Nikkhah-Moshaie, R.; Sagar, V.; Nair, M. Nanogels as potential drug nanocarriers for CNS drug delivery. Drug Discov. Today 2018, 23, 1436–1443. [Google Scholar] [CrossRef] [PubMed]
- Anooj, E.S.; Charumathy, M.; Sharma, V.; Vibala, B.V.; Gopukumar, S.T.; Jainab, S.B.; Vallinayagam, S. Nanogels: An overview of properties, biomedical applications, future research trends and developments. J. Mol. Struct. 2021, 1239, 130446. [Google Scholar] [CrossRef]
- Grimaudo, M.A.; Concheiro, A.; Alvarez-Lorenzo, C. Nanogels for regenerative medicine. J. Control. Release 2019, 313, 148–160. [Google Scholar] [CrossRef] [PubMed]
- Cuggino, J.C.; Blanco, E.R.; Gugliotta, L.M.; Igarzabal, C.I.; Calderón, M. Crossing biological barriers with nanogels to improve drug delivery performance. J. Control. Release 2019, 307, 221–246. [Google Scholar] [CrossRef]
- Yin, Y.; Hu, B.; Yuan, X.; Cai, L.; Gao, H.; Yang, Q. Nanogel: A versatile nano-delivery system for biomedical applications. Pharmaceutics 2020, 12, 290. [Google Scholar] [CrossRef] [Green Version]
- Korani, S.; Korani, M.; Bahrami, S.; Johnston, T.P.; Butler, A.E.; Banach, M.; Sahebkar, A. Application of nanotechnology to improve the therapeutic benefits of statins. Drug Discov. Today 2019, 24, 567–574. [Google Scholar] [CrossRef]
- Hill, M.F.; Bordoni, B. Hyperlipidemia. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK559182/ (accessed on 30 September 2021).
- Nelson, R.H. Hyperlipidemia as a risk factor for cardiovascular disease. Prim. Care Clin. Off. Pract. 2013, 40, 195–211. [Google Scholar] [CrossRef] [Green Version]
- Ludwig, A. The use of mucoadhesive polymers in ocular drug delivery. Adv. Drug Deliv. Rev. 2005, 57, 1595–1639. [Google Scholar] [CrossRef] [PubMed]
- Barbu, E.; Verestiuc, L.; Iancu, M.; Jatariu, A.; Lungu, A.; Tsibouklis, J. Hybrid polymeric hydrogels for ocular drug delivery: Nanoparticulate systems from copolymers of acrylic acid-functionalized chitosan and N-isopropylacrylamide or 2-hydroxyethyl methacrylate. Nanotechnology 2009, 20, 225108. [Google Scholar] [CrossRef] [PubMed]
- Kao, H.J.; Lo, Y.L.; Lin, H.R.; Yu, S.P. Characterization of pilocarpine-loaded chitosan/Carbopol nanoparticles. J. Pharm. Pharmacol. 2006, 58, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Järvinen, K.; Järvinen, T.; Thompson, D.O.; Stella, V.J. The effect of a modified β-cyclodextrin, SBE4-β-CD, on the aqueous stability and ocular absorption of pilocarpine. Curr. Eye Res. 1994, 13, 897–905. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.-G. Preface. Curr. Drug Deliv. 2021, 18, 2–3. [Google Scholar] [CrossRef]
- Xu, H.; Xu, X.; Li, S.; Song, W.-L.; Yu, D.-G.; Annie Bligh, S.W. The Effect of Drug Heterogeneous Distributions within Core-Sheath Nanostructures on Its Sustained Release Profiles. Biomolecules 2021, 11, 1330. [Google Scholar] [CrossRef]
- Kang, S.; He, Y.; Yu, D.-G.; Li, W.; Wang, K. Drug–zein@ lipid hybrid nanoparticles: Electrospraying preparation and drug extended release application. Colloids Surf. B Biointerfaces 2021, 201, 111629. [Google Scholar] [CrossRef]
- Borujeni, S.H.; Mirdamadian, S.Z.; Varshosaz, J.; Taheri, A. Three-dimensional (3D) printed tablets using ethyl cellulose and hydroxypropyl cellulose to achieve zero order sustained release profile. Cellulose 2020, 27, 1573–1589. [Google Scholar] [CrossRef]
- Kaur, M.; Sudhakar, K.; Mishra, V. Fabrication and biomedical potential of nanogels: An overview. Int. J. Polym. Mater. Polym. Biomater. 2019, 68, 287–296. [Google Scholar] [CrossRef]
- Kesharwani, P.; Jain, A.; Srivastava, A.K.; Keshari, M.K. Systematic development and characterization of curcumin-loaded nanogel for topical application. Drug Dev. Ind. Pharm. 2020, 46, 1443–1457. [Google Scholar] [CrossRef]
- Sharma, A.; Garg, T.; Aman, A.; Panchal, K.; Sharma, R.; Kumar, S.; Markandeywar, T. Nanogel—An advanced drug delivery tool: Current and future. Artif. Cells Nanomed. Biotechnol. 2016, 44, 165–177. [Google Scholar] [CrossRef]
- Endo, A. A historical perspective on the discovery of statins. Proc. Jpn. Acad. Ser. B 2010, 86, 484–493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murphy, C.; Deplazes, E.; Cranfield, C.G.; Garcia, A. The role of structure and biophysical properties in the pleiotropic effects of statins. Int. J. Mol. Sci. 2020, 21, 8745. [Google Scholar] [CrossRef]
- Manzoni, M.; Rollini, M. Biosynthesis and biotechnological production of statins by filamentous fungi and application of these cholesterol-lowering drugs. Appl. Microbiol. Biotechnol. 2002, 58, 555–564. [Google Scholar]
- Cuggino, J.C.; Molina, M.; Wedepohl, S.; Igarzabal, C.I.; Calderón, M.; Gugliotta, L.M. Responsive nanogels for application as smart carriers in endocytic pH-triggered drug delivery systems. Eur. Polym. J. 2016, 78, 14–24. [Google Scholar] [CrossRef]
- Shidhaye, S.S.; Thakkar, P.V.; Dand, N.M.; Kadam, V.J. Buccal drug delivery of pravastatin sodium. AAPS PharmSciTech 2010, 11, 416–424. [Google Scholar] [CrossRef] [Green Version]
- Desai, K.G. Chitosan nanoparticles prepared by ionotropic gelation: An overview of recent advances. Crit. Rev. Ther. Drug Carr. Syst. 2016, 33, 107–158. [Google Scholar] [CrossRef]
- Wang, H.; Deng, H.; Gao, M.; Zhang, W. Self-assembled nanogels based on ionic gelation of natural polysaccharides for drug delivery. Front. Bioeng. Biotechnol. 2021, 9, 703559. [Google Scholar] [CrossRef]
- Asadian-Birjand, M.; Bergueiro, J.; Rancan, F.; Cuggino, J.C.; Mutihac, R.C.; Achazi, K.; Dernedde, J.; Blume-Peytayi, U.; Vogt, A.; Calderón, M. Engineering thermoresponsive polyether-based nanogels for temperature dependent skin penetration. Polym. Chem. 2015, 6, 5827–5831. [Google Scholar] [CrossRef] [Green Version]
- Durán-Lobato, M.; Carrillo-Conde, B.; Khairandish, Y.; Peppas, N.A. Surface-modified P (HEMA-co-MAA) nanogel carriers for oral vaccine delivery: Design, characterization, and in vitro targeting evaluation. Biomacromolecules 2014, 15, 2725–2734. [Google Scholar] [CrossRef] [Green Version]
- Mishra, V.; Jain, N.K. Acetazolamide encapsulated dendritic nano-architectures for effective glaucoma management in rabbits. Int. J. Pharm. 2014, 461, 380–390. [Google Scholar] [CrossRef] [PubMed]
- Mishra, V.; Gupta, U.; Jain, N.K. Influence of different generations of poly (propylene imine) dendrimers on human erythrocytes. Die Pharm. 2010, 65, 891–895. [Google Scholar]
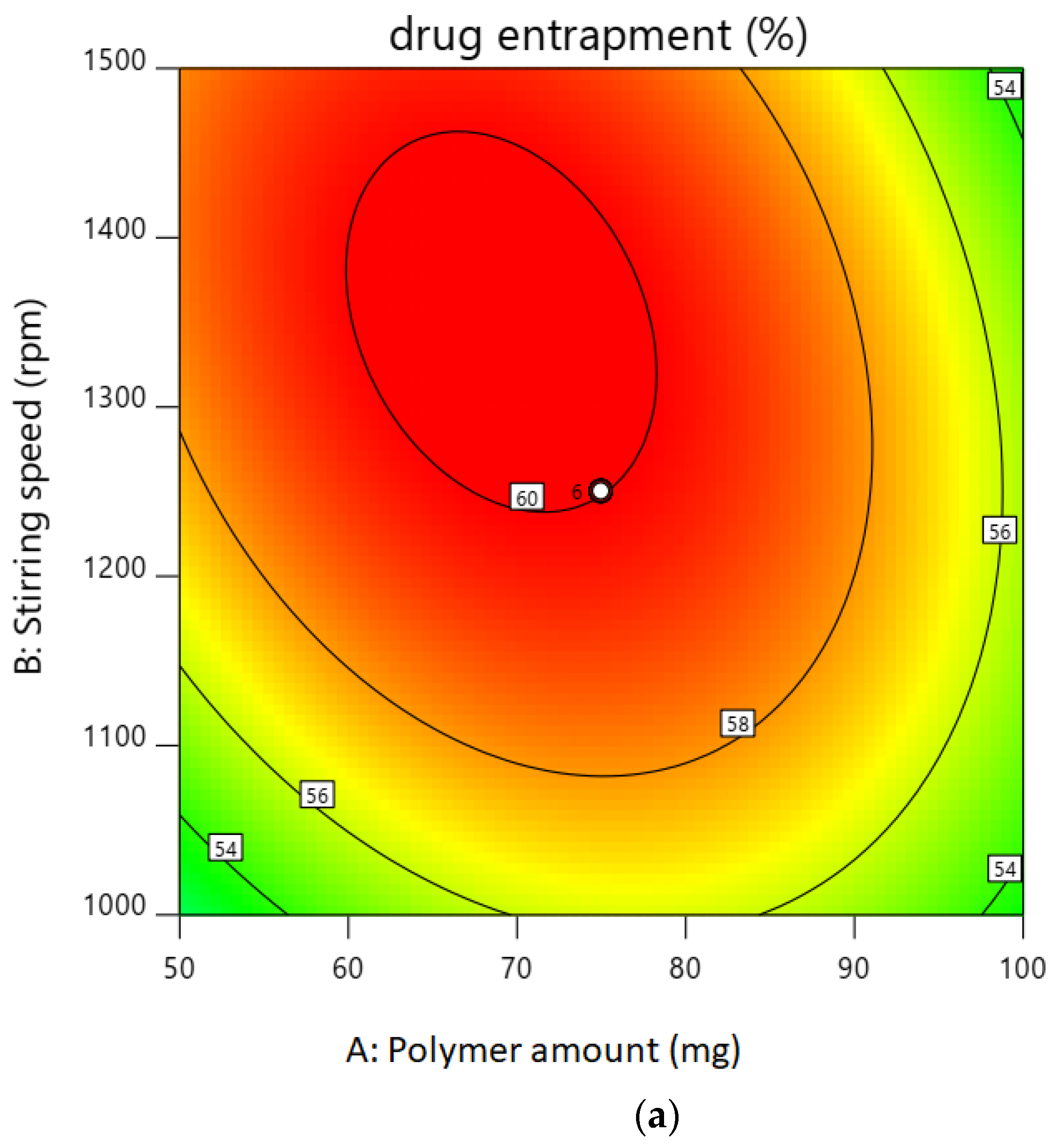
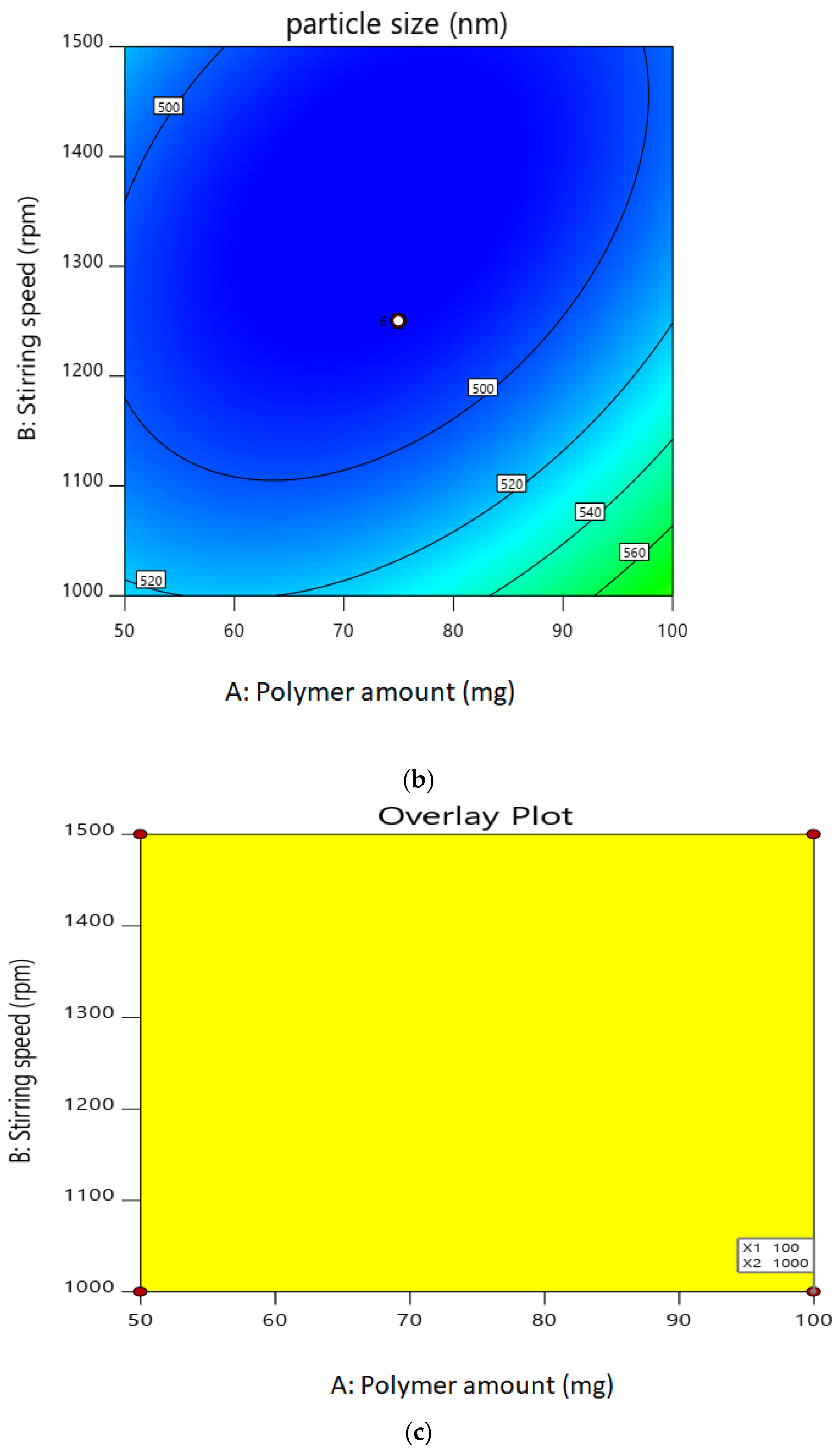


| Factor 1 | Factor 2 | Factor 3 | Response 1 | Response 2 | ||
|---|---|---|---|---|---|---|
| Std | Run | A: Polymer Amount | B: Stirring Speed | C: Sonication Time | Drug Entrapment | Particle Size |
| (mg) | (rpm) | (min) | % | (nm) | ||
| 1 | 10 | 50 | 1000 | 5 | 53.6 | 556 |
| 2 | 11 | 100 | 1000 | 5 | 56 | 602 |
| 3 | 17 | 50 | 1500 | 5 | 58.95 | 604 |
| 4 | 16 | 100 | 1500 | 5 | 47 | 614 |
| 5 | 15 | 50 | 1000 | 10 | 49.5 | 556 |
| 6 | 5 | 100 | 1000 | 10 | 47 | 655 |
| 7 | 7 | 50 | 1500 | 10 | 55 | 500 |
| 8 | 6 | 100 | 1500 | 10 | 54.6 | 498 |
| 9 | 14 | 33 | 1250 | 7.5 | 51 | 543 |
| 10 | 13 | 117 | 1250 | 7.5 | 50.33 | 543 |
| 11 | 20 | 75 | 830 | 7.5 | 50.68 | 578 |
| 12 | 1 | 75 | 1670 | 7.5 | 56.95 | 502 |
| 13 | 4 | 75 | 1250 | 3 | 58.2 | 566 |
| 14 | 12 | 75 | 1250 | 12 | 54 | 601 |
| 15 | 8 | 75 | 1250 | 7.5 | 60 | 486 |
| 16 | 18 | 75 | 1250 | 7.5 | 60 | 486 |
| 17 | 3 | 75 | 1250 | 7.5 | 60 | 486 |
| 18 | 9 | 75 | 1250 | 7.5 | 60 | 486 |
| 19 | 2 | 75 | 1250 | 7.5 | 60 | 486 |
| 20 | 19 | 75 | 1250 | 7.5 | 60 | 486 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| Model | 356.02 | 9 | 39.56 | 9.40 | 0.0008 | Significant |
| A—Polymer amount | 13.50 | 1 | 13.50 | 3.21 | 0.1035 | |
| B—Stirring speed | 29.27 | 1 | 29.27 | 6.96 | 0.0248 | |
| C—Sonication time | 19.97 | 1 | 19.97 | 4.75 | 0.0544 | |
| AB | 18.76 | 1 | 18.76 | 4.46 | 0.0609 | |
| AC | 5.53 | 1 | 5.53 | 1.31 | 0.2784 | |
| BC | 35.07 | 1 | 35.07 | 8.34 | 0.0162 | |
| A2 | 165.68 | 1 | 165.68 | 39.38 | <0.0001 | |
| B2 | 74.72 | 1 | 74.72 | 17.76 | 0.0018 | |
| C2 | 31.10 | 1 | 31.10 | 7.39 | 0.0216 | |
| Residual | 42.07 | 10 | 4.21 | |||
| Lack of Fit | 42.07 | 5 | 8.41 | |||
| Pure Error | 0.0000 | 5 | 0.0000 | |||
| Cor Total | 398.09 | 19 |
| Response | Predicted Mean | Predicted Median | Std Deviation | N | SE Prediction | 95% Low | Mean | 95% High |
|---|---|---|---|---|---|---|---|---|
| Entrapment efficiency | 49.4784 | 49.4784 | 2.05155 | 1 | 2.65049 | 49.368 | 49.4784 | 55.384 |
| Particle size | 647.994 | 647.994 | 23.6573 | 1 | 30.5699 | 579.88 | 647.994 | 716.108 |
| S. No. | Time (h) | Pravastatin Oral Solution (µg/mL) | Pravastatin Oral Nanogel (µg/mL) |
|---|---|---|---|
| 1 | 0 | 0 | 0 |
| 2 | 1 | 68 | 14 |
| 3 | 2 | 47 | 26 |
| 4 | 3 | 23 | 39 |
| 5 | 4 | 11 | 79 |
| 6 | 5 | 0 | 65 |
| 7 | 6 | 47 | |
| 8 | 7 | 31 | |
| 9 | 8 | 9 | |
| 10 | 10 | 0 | |
| Cmax | 68 µg/mL | 79 µg/mL | |
| tmax | 1 h | 4 h | |
| AUC last | 140.707 (µg·h/mL) | 302.022 (µg·h/mL) | |
| AUC total | 155.85 (µg·h/mL) | 312.912 (µg·h/mL) | |
| T half | 0.98 h | 0.83 h | |
| MRT | 2.35 h | 4.62 h |
| Parameters | Result | Inference |
|---|---|---|
| Particle size | 486.2 nm | Desired and acceptable size |
| Polydispersity index | 0.303 | Uniform distribution |
| Zeta potential | 43.4 mV | Positively and evenly distributed |
| Entrapment efficiency | 50% | Nanoparticles leads for higher entrapment of drug |
| Drug release | 59.63% (24 h) | Sustained release of the drug was obtained |
| SEM studies | - | Regular shape |
| Compatibility study | Characteristic peak is obtained | Overlay plot confirms the characteristic peaks of drug |
| Toxicity study | Less toxicity is exhibited | Safer to use |
| Pharmacokinetic study | Higher bioavailability |
| CQAs | Polymer Amount | Stirring Speed | Sonication Time |
|---|---|---|---|
| Particle size | Low | Medium | High |
| % Entrapment efficiency | Low | Medium | High |
| Profile | Target | Justification |
|---|---|---|
| Dosage form | Nanogel | Novel dosage form for targeted drug delivery |
| Dosage design | Sustained release oral nanogel | For increasing residence time of pravastatin |
| Therapeutic indication | Antihyperlipidemia | Pravastatin acts by inhibition of cholesterol producing enzymes |
| Route of administration | Oral | Most suitable route of administration and can be well absorbed in intestine |
| Particle size | 10–1000 nm | Drug absorption and uniform biodistribution |
| Zeta potential | −200 to 200 mV | Needed to ensure stability |
| Entrapment efficiency | >50% | Nanogel entraps higher amount of drug |
| Independent Variables | Low | High |
|---|---|---|
| Coded Values | (−1) | (+1) |
| A = Polymer amount (mg) | 50 | 100 |
| B = Stirring speed (rpm) | 1000 | 1500 |
| C = Sonication time (min) | 4 | 8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saraogi, G.K.; Tholiya, S.; Mishra, Y.; Mishra, V.; Albutti, A.; Nayak, P.; Tambuwala, M.M. Formulation Development and Evaluation of Pravastatin-Loaded Nanogel for Hyperlipidemia Management. Gels 2022, 8, 81. https://doi.org/10.3390/gels8020081
Saraogi GK, Tholiya S, Mishra Y, Mishra V, Albutti A, Nayak P, Tambuwala MM. Formulation Development and Evaluation of Pravastatin-Loaded Nanogel for Hyperlipidemia Management. Gels. 2022; 8(2):81. https://doi.org/10.3390/gels8020081
Chicago/Turabian StyleSaraogi, Gaurav Kant, Siddharth Tholiya, Yachana Mishra, Vijay Mishra, Aqel Albutti, Pallavi Nayak, and Murtaza M. Tambuwala. 2022. "Formulation Development and Evaluation of Pravastatin-Loaded Nanogel for Hyperlipidemia Management" Gels 8, no. 2: 81. https://doi.org/10.3390/gels8020081
APA StyleSaraogi, G. K., Tholiya, S., Mishra, Y., Mishra, V., Albutti, A., Nayak, P., & Tambuwala, M. M. (2022). Formulation Development and Evaluation of Pravastatin-Loaded Nanogel for Hyperlipidemia Management. Gels, 8(2), 81. https://doi.org/10.3390/gels8020081