Polymeric Hydrogels as Technology Platform for Drug Delivery Applications
Abstract
:1. Introduction
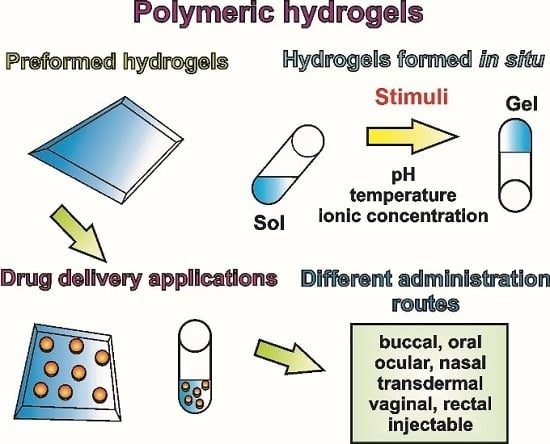
2. Polymeric Hydrogels for Drug Delivery Applications
3. Preformed Hydrogels versus Hydrogels Formed Upon Implantation (In Situ)
3.1. Preformed Hydrogels
3.2. Hydrogels Formed In Situ
Smart Hydrogels
4. Nanogels and Microgels
5. Administration Routes of Hydrogels
5.1. Minimally-Invasive Administration Routes
5.2. Invasive Administration Routes
6. Conclusions and Future Directions
Acknowledgments
Conflicts of Interest
References
- Anumolu, S.S.; Singh, Y.; Gao, D.; Stein, S.; Sinko, P.J. Design and evaluation of novel fast forming pilocarpine-loaded ocular hydrogels for sustained pharmacological response. J. Control Release 2009, 137, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Gaharwar, A.K.; Peppas, N.A.; Khademhosseini, A. Nanocomposite hydrogels for biomedical applications. Biotechnol. Bioeng. 2014, 111, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Onuki, Y.; Hasegawa, N.; Kida, C.; Obata, Y.; Takayama, K. Study of the contribution of the state of water to the gel properties of a photocrosslinked polyacrylic acid hydrogel using magnetic resonance imaging. J. Pharm. Sci. 2014, 103, 3532–3541. [Google Scholar] [CrossRef] [PubMed]
- Rossi, B.; Venuti, V.; D’Amico, F.; Gessini, A.; Mele, A.; Punta, C.; Melone, L.; Crupi, V.; Majolino, D.; Masciovecchio, C. Guest-matrix interactions affect the solvation of cyclodextrin-based polymeric hydrogels: A UV Raman scattering study. Soft Matter 2016, 12, 8861–8868. [Google Scholar] [CrossRef] [PubMed]
- Schoener, C.A.; Hutson, H.N.; Peppas, N.A. pH-Responsive Hydrogels with Dispersed Hydrophobic Nanoparticles for the Oral Delivery of Chemotherapeutics. J. Biomed. Mater. Res. A 2013, 101, 2229–2236. [Google Scholar] [CrossRef] [PubMed]
- Hou, K.; Wang, H.; Lin, Y.; Chen, S.; Yang, S.; Cheng, Y.; Hsiao, B.S.; Zhu, M. Large Scale Production of Continuous Hydrogel Fibers with Anisotropic Swelling Behavior by Dynamic-Crosslinking-Spinning. Macromol. Rapid Commun. 2016, 37, 1795–1801. [Google Scholar] [CrossRef] [PubMed]
- Concheiro, A.; Alvarez-Lorenzo, C. Chemically cross-linked and grafted cyclodextrin hydrogels: From nanostructures to drug-eluting medical devices. Adv. Drug Deliv. Rev. 2013, 65, 1188–1203. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Yin, H.; Lu, Y.; Zhang, H.; Wang, H. Evaluation of Hydrogel Suppositories for Delivery of 5-Aminolevulinic Acid and Hematoporphyrin Monomethyl Ether to Rectal Tumors. Molecules 2016, 21, 1347. [Google Scholar] [CrossRef] [PubMed]
- Siepmann, J.; Peppas, N.A. Higuchi equation: Derivation, applications, use and misuse. Int. J. Pharm. 2011, 418, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N.A.; Moynihan, H.J. Solute diffusion in swollen membranes. IV. Theories for moderately swollen networks. J. Appl. Polym. Sci. 1985, 30, 2589–2606. [Google Scholar] [CrossRef]
- Harland, R.S.; Peppas, N.A. Solute diffusion in swollen membranes. VII. Diffusion in semicrystalline networks. Colloid Polym. Sci. 1989, 267, 218–225. [Google Scholar] [CrossRef]
- Caldorera-Moore, A.; Peppas, N.A. Micro- and Nanotechnologies for Intelligent and Responsive Biomaterial-Based Medical Systems. Adv. Drug Deliv. Rev. 2009, 61, 1391–1401. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Peppas, N.A. Hydrogels and Scaffolds for Immunomodulation. Adv. Mater. 2014, 26, 6530–6541. [Google Scholar] [CrossRef] [PubMed]
- Elviri, L.; Bianchera, A.; Bergonzi, C.; Bettini, R. Controlled local drug delivery strategies from chitosan hydrogels for wound healing. Expert Opin. Drug Deliv. 2016. [Google Scholar] [CrossRef] [PubMed]
- Pérez, E.; Fernández, A.; Olmo, R.; Teijón, J.M.; Blanco, M.D. pH and glutathion-responsive hydrogel for localized delivery of paclitaxel. Colloids Surf. B Biointerfaces 2014, 116, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.; Fabry, B.; Boccaccini, A.R. Fibrous protein-based hydrogels for cell encapsulation. Biomaterials 2014, 35, 6727–6738. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, L.; Song, L.; Niu, G.; Cao, H.; Wang, G.; Yang, H.; Zhu, S. Controllable properties and microstructure of hydrogels based on crosslinked poly(ethylene glycol) diacrylates with different molecular weights. J. Appl. Polym. Sci. 2010, 121, 531–540. [Google Scholar] [CrossRef]
- Slaughter, B.V.; Khurshid, S.S.; Fisher, O.Z.; Khademhosseini, A.; Peppas, N.A. Hydrogels in Regenerative Medicine. Adv. Mater. 2009, 21, 3307–3329. [Google Scholar] [CrossRef] [PubMed]
- Naseri, N.; Deepa, B.; Mathew, A.P.; Oksman, K.; Girandon, L. Nanocellulose-Based Interpenetrating Polymer Network (IPN) Hydrogels for Cartilage Applications. Biomacromolecules 2016, 17, 3714–3723. [Google Scholar] [CrossRef] [PubMed]
- Straccia, M.C.; d’Ayala, G.G.; Romano, I.; Oliva, A.; Laurienzo, P. Alginate hydrogels coated with chitosan for wound dressing. Mar. Drugs 2015, 13, 2890–2908. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Lorenzo, C.; dos Santos, F.R.; Sosnik, A.; Torres-Labandeira, J.; Concheiro, A. Cyclodextrin-based hydrogels as highly versatile drug delivery systems. In Handbook of Hydrogels: Properties, Preparation and Applications; Stein, D.B., Ed.; Nova Publishers: Hauppauge, NY, USA, 2009; pp. 61–102. [Google Scholar]
- Machín, R.; Isasi, J.R.; Vélaz, I. β-Cyclodextrin hydrogels as potential drug delivery systems. Carbohydr. Polym. 2012, 87, 2024–2030. [Google Scholar] [CrossRef]
- Mennini, N.; Casella, G.; Cirri, M.; Maestrelli, F.; Mura, P. Development of cyclodextrin hydrogels for vaginal delivery of dehydroepiandrosterone. J. Pharm. Pharmacol. 2016, 68, 762–771. [Google Scholar] [CrossRef] [PubMed]
- Sosnik, A. Design of Injectable biomaterials for biomedical and pharmaceutical applications: Past, present and future of the in situ generated implants. ARS Pharm. 2007, 48, 83–102. [Google Scholar]
- Tsitsilianis, C. Responsive reversible hydrogels from associative “smart” macromolecules. Soft Matter 2010, 6, 2372–2388. [Google Scholar] [CrossRef]
- Gupta, M.K.; Martin, J.R.; Werfel, T.A.; Shen, T.; Page, J.M.; Duvall, C.L. Cell protective, ABC triblock polymer-based thermoresponsive hydrogels with ROS-triggered degradation and drug release. J. Am. Chem. Soc. 2014, 136, 14896–14902. [Google Scholar] [CrossRef] [PubMed]
- Marchesan, S.; Qu, Y.; Waddington, L.J.; Easton, C.D.; Glattauer, V.; Lithgow, T.J.; McLean, K.M.; Forsythe, J.S.; Hartley, P.G. Self-assembly of ciprofloxacin and a tripeptide into an antimicrobial nanostructured hydrogel. Biomaterials 2013, 34, 3678–3687. [Google Scholar] [CrossRef] [PubMed]
- Slaughter, B.V.; Blanchard, A.T.; Maass, K.F.; Peppas, N.A. Dynamic swelling behavior of interpenetrating polymer networks in response to temperature and pH. J. Appl. Polym. Sci. 2015, 132, 42076. [Google Scholar] [CrossRef] [PubMed]
- Chiappetta, D.A.; Sosnik, A. Poly(ethylene oxide)-poly(propylene oxide) block copolymer micelles as drug delivery agents: Improved hydrosolubility, stability and bioavailability of drugs. Eur. J. Pharm. Biopharm. 2007, 66, 303–317. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Y.; Wu, H.C.; Sun, J.S.; Dong, G.C.; Wang, T.W. Injectable and thermoresponsive self-assembled nanocomposite hydrogel for long-term anticancer drug delivery. Langmuir 2013, 29, 3721–3729. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.G.; Lee, S.E.; Kang, B.S.; Ng, C.L.; Davaa, E.; Park, J.S. Thermosensitive and mucoadhesive sol-gel composites of paclitaxel/dimethyl-β-cyclodextrin for buccal delivery. PLoS ONE 2014, 9, e109090. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Burger, C.; Li, C.; Li, J.; Lin, M.Y.; Colby, R.H.; Rafailovich, M.H.; Sokolov, J.C. Shear-Induced Layered Structure of Polymeric Micelles by SANS. Macromolecules 2007, 40, 4016–4022. [Google Scholar] [CrossRef]
- Varshosaz, J.; Tabbakhian, M.; Salmani, Z. Designing of a Thermosensitive Chitosan/Poloxamer In Situ Gel for Ocular Delivery of Ciprofloxacin. Open Drug Deliv. J. 2008, 2, 61–70. [Google Scholar] [CrossRef]
- Esposito, E.; Carotta, V.; Scabbia, A.; Trombelli, L.; D’Antona, P.; Menegatti, E.; Nastruzzi, C. Comparative analysis of tetracycline-containing dental gels: Poloxamer- and monoglyceride-based formulations. Int. J. Pharm. 1996, 142, 9–23. [Google Scholar] [CrossRef]
- Sosnik, A.; Cohn, D. Poly(ethylene glycol)-poly(epsilon-caprolactone) block oligomers as injectable materials. Polymer 2003, 44, 7033–7042. [Google Scholar] [CrossRef]
- Sosnik, A.; Cohn, D.; San Román, J.; Abraham, G.A. Crosslinkable PEO-PPO-PEO-based reverse thermo-responsive gels as potentially injectable materials. J. Biomater. Sci. Polym. Ed. 2003, 14, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Cohn, D.; Sosnik, A.; Levy, A. Improved reverse thermo-responsive polymeric systems. Biomaterials 2003, 24, 3707–3714. [Google Scholar] [CrossRef]
- Kumar, S.; Himmelstein, K.J. Modification of in situ gelling behavior of carbopol solutions by hydroxypropyl methylcellulose. J. Pharm. Sci. 1995, 84, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.C.; Huang, K.Y.; Yang, B.Y.; Ko, C.H.; Huang, H.M. Fabrication of Novel Hydrogel with Berberine-Enriched Carboxymethylcellulose and Hyaluronic Acid as an Anti-Inflammatory Barrier Membrane. Biomed Res. Int. 2016, 2016, 3640182. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Altunbas, A.; Yucel, T.; Nagarkar, R.P.; Schneider, J.P.; Pochan, D.J. Injectable solid hydrogel: Mechanism of shear-thinning and immediate recovery of injectable β-hairpin peptide hydrogels. Soft Matter 2010, 6, 5143–5156. [Google Scholar] [CrossRef] [PubMed]
- Riber, L.; Burmølle, M.; Alm, M.; Milani, S.M.; Thomsen, P.; Hansen, L.H.; Sørensen, S.J. Enhanced plasmid loss in bacterial populations exposed to the antimicrobial compound irgasan delivered from interpenetrating polymer network silicone hydrogels. Plasmid 2016, 87–88, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Tummala, G.K.; Rojas, R.; Mihranyan, A. Poly(vinyl alcohol) Hydrogels Reinforced with Nanocellulose for Ophthalmic Applications: General Characteristics and Optical Properties. J. Phys. Chem. B 2016, 120, 13094–13101. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, J.F.; Alvarez-Lorenzo, C.; Silva, M.; Balsa, L.; Couceiro, J.; Torres-Labandeira, J.J.; Concheiro, A. Soft contact lenses functionalized with pendant cyclodextrins for controlled drug delivery. Biomaterials 2009, 30, 1348–1355. [Google Scholar] [CrossRef] [PubMed]
- Chassenieux, C.; Tsitsilianis, C. Recent trends in pH/thermo-responsive self-assembling hydrogels: From polyions to peptide-based polymeric gelators. Soft Matter 2016, 12, 1344–1359. [Google Scholar] [CrossRef] [PubMed]
- Anumolu, S.S.; DeSantis, A.S.; Menjoge, A.R.; Hahn, R.A.; Beloni, J.A.; Gordon, M.K.; Sinko, P.J. Doxycycline loaded poly(ethylene glycol) hydrogels for healing vesicant-induced ocular wounds. Biomaterials 2010, 31, 964–974. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Shoichet, M.S. A photolabile hydrogel for guided three-dimensional cell growth and migration. Nat. Mater. 2004, 3, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Liu, C.; Hai, B.; Ma, T.; Zhang, W.; Tan, J.; Fu, X.; Wang, H.; Xu, Y.; Song, C. Chitosan conduits filled with simvastatin/Pluronic F-127 hydrogel promote peripheral nerve regeneration in rats. J. Biomed. Mater. Res. B Appl. Biomater. 2017. [Google Scholar] [CrossRef] [PubMed]
- Osman, A.; Oner, E.T.; Eroglu, M.S. Novel levan and pNIPA temperature sensitive hydrogels for 5-ASA controlled release. Carbohydr. Polym. 2017, 165, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.H.; Kim, A.; Shin, W.; Heo, M.B.; Noh, H.J.; Hong, K.S.; Cho, J.H.; Lim, Y.T. Photothermal-modulated drug delivery and magnetic relaxation based on collagen/poly(γ-glutamic acid) hydrogel. Int. J. Nanomed. 2017, 12, 2607–2620. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; He, Y.; Zhao, Y.; Xie, H.; Lin, Q.; He, Z.; Wang, X.; Li, J.; Zhang, H.; Wang, C.; et al. A Thermosensitive Heparin-Poloxamer Hydrogel Bridges aFGF to Treat Spinal Cord Injury. ACS Appl. Mater. Interfaces 2017, 9, 6725–6745. [Google Scholar] [CrossRef] [PubMed]
- Marek, S.R.; Conn, C.A.; Peppas, N.A. Cationic Nanogels Based On Diethylaminoethyl Methacrylate. Polymer 2010, 51, 1237–1243. [Google Scholar] [CrossRef] [PubMed]
- Mekkawy, A.I.; El-Mokhtar, M.A.; Nafady, N.A.; Yousef, N.; Hamad, M.A.; El-Shanawany, S.M.; Ibrahim, E.H.; Elsabahy, M. In vitro and in vivo evaluation of biologically synthesized silver nanoparticles for topical applications: Effect of surface coating and loading into hydrogels. Int. J. Nanomed. 2017, 12, 759–777. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Song, H.; Zhang, Y.; Liu, J.; Zhang, J.; Wang, W.; Liu, J.; Li, C.; Kong, D. Bridging the Gap between Macroscale Drug Delivery Systems and Nanomedicines: A Nanoparticle-Assembled Thermosensitive Hydrogel for Peritumoral Chemotherapy. ACS Appl. Mater. Interfaces 2016, 8, 29323–29333. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.; Mohd Amin, M.C.; Ismail, I.; Buang, F. Enhancement of oral insulin bioavailability: In vitro and in vivo assessment of nanoporous stimuli-responsive hydrogel microparticles. Expert Opin. Drug Deliv. 2016, 13, 621–632. [Google Scholar] [CrossRef] [PubMed]
- Kamel, R.; Mahmoud, A.; El-Feky, G. Double-phase hydrogel for buccal delivery of tramadol. Drug Dev. Ind. Pharm. 2012, 38, 468–483. [Google Scholar] [CrossRef] [PubMed]
- Forbes, D.C.; Peppas, N.A. Oral delivery of small RNA and DNA. J. Control. Release 2012, 162, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Dafe, A.; Etemadi, H.; Dilmaghani, A.; Mahdavinia, G.R. Investigation of pectin/starch hydrogel as a carrier for oral delivery of probiotic bacteria. Int. J. Biol. Macromol. 2017, 97, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Sande, M.; Csaba, N.S.; Alonso, M.J. New perspectives in oral peptide and protein delivery: From nanocarrier design to in vivo effectiveness. In Advances and Challenges Oral Delivery of Macromolecules; Russell-Jones, G., Ed.; Future Medicine: London, UK, 2016; pp. 46–64. [Google Scholar] [CrossRef]
- Brayden, D.J.; Alonso, M.J. Oral delivery of peptides: Opportunities and issues for translation. Adv. Drug Deliv. Rev. 2016, 106, 193–195. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, C.; Steichen, S.; Peppas, N.A. Development and characterization of stimuli-responsive hydrogel microcarriers for oral protein delivery. J. Biomed. Mater. Res. A 2017, 105, 1243–1251. [Google Scholar] [CrossRef] [PubMed]
- Treenate, P.; Monvisade, P. In vitro drug release profiles of pH-sensitive hydroxyethylacryl chitosan/sodium alginate hydrogels using paracetamol as a soluble model drug. Int. J. Biol. Macromol. 2017, 99, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Ferreiro, A.; Silva-Rodriguez, J.; Otero-Espinar, F.J.; González-Barcia, M.; Lamas, M.J.; Ruibal, A.; Luaces-Rodríguez, A.; Vieites-Prado, A.; Lema, I.; Herranz, M.; et al. In vivo eye surface residence determination by high-resolution scintigraphy of a novel ion-sensitive hydrogel based on gellan gum and kappa-carrageenan. Eur. J. Pharm. Biopharm. 2017, 114, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Zhang, N.; Hua, H.; Liu, T.; Tang, Y.; Fu, L.; Yang, Y.; Ma, X.; Zhao, Y. Preparation, pharmacokinetics and pharmacodynamics of ophthalmic thermosensitive in situ hydrogel of betaxolol hydrochloride. Biomed. Pharmacother. 2016, 83, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Fabiano, A.; Bizzarri, R.; Zambito, Y. Thermosensitive hydrogel based on chitosan and its derivatives containing medicated nanoparticles for transcorneal administration of 5-fluorouracil. Int. J. Nanomed. 2017, 12, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Zaki, N.M.; Awad, G.A.; Mortada, N.D.; Abd El Hady, S.S. Enhanced bioavailability of metoclopramide HCl by intranasal administration of a mucoadhesive in situ gel with modulated rheological and mucociliary transport properties. Eur. J. Pharm. Sci. 2007, 32, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Mistry, A.; Stolnik, S.; Illum, L. Nanoparticles for direct nose-to-brain delivery of drugs. Int. J. Pharm. 2009, 379, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Patil, K.; Bobade, N.; Yeole, P.; Gaikwad, R. Formulation of intranasal mucoadhesive temperature-mediated in situ gel containing ropinirole and evaluation of brain targeting efficiency in rats. J. Drug Target. 2010, 18, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, H.S.; Gattani, S. In situ gels of Metoclopramide Hydrochloride for intranasal delivery: In vitro evaluation and in vivo pharmacokinetic study in rabbits. Drug Deliv. 2010, 17, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Arafa, M.G.; Ayoub, B.M. DOE Optimization of Nano-based Carrier of Pregabalin as Hydrogel: New Therapeutic & Chemometric Approaches for Controlled Drug Delivery Systems. Sci. Rep. 2017, 7, 41503. [Google Scholar] [CrossRef] [PubMed]
- Furst, T.; Piette, M.; Lechanteur, A.; Evrard, B.; Piel, G. Mucoadhesive cellulosic derivative sponges as drug delivery system for vaginal application. Eur. J. Pharm. Biopharm. 2015, 95, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Malli, S.; Bories, C.; Pradines, B.; Loiseau, P.M.; Ponchel, G.; Bouchemal, K. In situ forming pluronic® F127/chitosan hydrogel limits metronidazole transmucosal absorption. Eur. J. Pharm. Biopharm. 2017, 112, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Bouchemal, K.; Aka-Any-Grah, A.; Dereuddre-Bosquet, N.; Martin, L.; Lievin-Le-Moal, V.; Le Grand, R.; Nicolas, V.; Gibellini, D.; Lembo, D.; Poüs, C.; et al. Thermosensitive and mucoadhesive pluronic-hydroxypropylmethylcellulose hydrogel containing the mini-CD4 M48U1 is a promising efficient barrier against HIV diffusion through macaque cervicovaginal mucus. Antimicrob. Agents Chemother. 2015, 59, 2215–2222. [Google Scholar] [CrossRef] [PubMed]
- Cole, L.; Hanning, C.D.; Robertson, S.; Quinn, K. Further development of a morphine hydrogel suppository. Br. J. Clin. Pharmacol. 1990, 30, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Seib, F.P.; Pritchard, E.M.; Kaplan, D.L. Self-assembling doxorubicin silk hydrogels for the focal treatment of primary breast cancer. Adv. Funct. Mater. 2013, 23, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Wang, K.; Wang, H.; Chen, F.; Huang, W.; Chen, Y.; Chen, J.; Tao, J.; Wen, X.; Xiong, S. Novel self-assembled tacrolimus nanoparticles cross-linking thermosensitive hydrogels for local rheumatoid arthritis therapy. Colloids Surf. B Biointerfaces 2017, 149, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Lei, K.; Chen, Y.; Wang, J.; Peng, X.; Yu, L.; Ding, J. Non-invasive Monitoring of In Vivo Degradation of a Radiopaque Thermoreversible Hydrogel and Its Efficacy in Preventing Post-operative Adhesions. Acta Biomater. 2017. [Google Scholar] [CrossRef] [PubMed]













© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sosnik, A.; Seremeta, K.P. Polymeric Hydrogels as Technology Platform for Drug Delivery Applications. Gels 2017, 3, 25. https://doi.org/10.3390/gels3030025
Sosnik A, Seremeta KP. Polymeric Hydrogels as Technology Platform for Drug Delivery Applications. Gels. 2017; 3(3):25. https://doi.org/10.3390/gels3030025
Chicago/Turabian StyleSosnik, Alejandro, and Katia P. Seremeta. 2017. "Polymeric Hydrogels as Technology Platform for Drug Delivery Applications" Gels 3, no. 3: 25. https://doi.org/10.3390/gels3030025