Metal Oxide/TiO2 Hybrid Nanotubes Fabricated through the Organogel Route
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation of Gelator 1
2.2. Fabrication of TiO2 Nanotubes
2.3. Effect of Calcination Temperatures on TiO2 Nanotubes
2.4. Fabrication of Other Metal Oxides
2.5. Fabrication of Ta2O5/TiO2 Hybrid Nanotubes
2.6. Hybridization of Other Metal Oxides into TiO2
2.7. Hybridization of Metal Oxides Except for TiO2
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Sol-Gel Polymerization
4.3. Instrumentation and Techniques
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Terech, P.; Weiss, R.G. Low Molecular Weight Gelators of Organic Liquids and the Properties of Their Gels. Chem. Rev. 1997, 97, 3133–3159. [Google Scholar] [CrossRef] [PubMed]
- Estroff, L.A.; Hamilton, A.D. Water Gelation by Small Organic Molecules. Chem. Rev. 2004, 204, 1201–1217. [Google Scholar] [CrossRef] [PubMed]
- De Loos, M.; Feringa, B.L.; van Esch, J.H. Design and Application of Self-Assembled Low Molecular Weight Hydrogels. Eur. J. Org. Chem. 2005, 3615–3631. [Google Scholar] [CrossRef]
- Dastidar, P. Supramolecular gelling agents: Can they be designed? Chem. Soc. Rev. 2008, 37, 2699–2715. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Hanabusa, K. L-Lysine-based low-molecular-weight gelators. Chem. Soc. Rev. 2009, 38, 967–975. [Google Scholar] [CrossRef] [PubMed]
- Steed, J.W. Supramolecular gel chemistry: Developments over the last decade. Chem. Commun. 2011, 47, 1379–1383. [Google Scholar] [CrossRef] [PubMed]
- Weiss, R.G. The Past, Present, and Future of Molecular Gels. What Is the Status of the Field, and Where Is It Going? J. Am. Chem. Soc. 2014, 136, 7519–7530. [Google Scholar] [CrossRef] [PubMed]
- Okesola, B.O.; Smith, D.K. Applying low molecular weight supramolecular gelators in an environmental setting–self-assembled gels as smart materials for pollutant removal. Chem. Soc. Rev. 2016, 45, 4226–4251. [Google Scholar] [CrossRef] [PubMed]
- Hanabusa, K.; Suzuki, M. Physical Gelation by Low-Molecular-Weight Compounds and Development of Gelators. Bull. Chem. Soc. Jpn. 2016, 89, 174–189. [Google Scholar] [CrossRef]
- Terech, P.; Weiss, R.G. Molecular Gels Materials with Self-Assembled Fibrillar Networks; Springer: Dordrecht, The Netherlands, 2006. [Google Scholar]
- Escuder, B.; Miravet, J.F. Functional Molecular Gels; RSC: Cambridge, UK, 2014. [Google Scholar]
- Piepenbrock, M.-O.M.; Lloyd, G.O.; Clarke, N.; Steed, J.W. Metal- and Anion-Binding Supramolecular Gels. Chem. Rev. 2010, 110, 1960–2004. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Chen, L.; Zhang, M.; Yi, T. Low-molecular-mass gels responding to ultrasound and mechanical stress: Towards self-healing materials. Chem. Soc. Rev. 2014, 43, 5346–5371. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.D.; Steed, J.W. Gels with sense: Supramolecular materials that respond to heat, light and sound. Chem. Soc. Rev. 2016, 45, 6546–6596. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Ghosh, S. H-bonding directed programmed supramolecular assembly of naphthalene-diimide (DNI) derivatives. Chem. Commun. 2016, 52, 6860–6872. [Google Scholar] [CrossRef] [PubMed]
- Aparicio, F.; Cherumukkil, S.; Ajayagosh, A.; Sánchez, L. Color-Tunable Cyano-Substituted Divinylene Arene Luminogens as Fluorescent π-Gelators. Langmuir 2016, 32, 284–489. [Google Scholar] [CrossRef] [PubMed]
- Draper, E.R.; Lee, J.R.; Wallace, M.; Jäckel, F.; Cowan, A.J.; Adam, D.J. Self-sorted photoconductive xerogels. Chem. Sci. 2016, 7, 6499–6505. [Google Scholar] [CrossRef] [PubMed]
- Van Herpt, J.T.; Areephong, J.; Stuart, M.C.A.; Browne, W.R.; Feringa, B.L. Light-Controlled Formation of Vesicles and Supramolecular Organogels by a Cholesterol-Bearing Amphiphilic Molecular Switch. Chem. Eur. J. 2014, 20, 1737–1742. [Google Scholar] [CrossRef] [PubMed]
- Latxague, L.; Ramin, M.A.; Appavoo, A.; Berto, P.; Maisani, M.; Ehret, C.; Chassande, O.; Barthélémy, P. Control of Stem-Cell Behavior by Fine Tuning the Supramolecular Assemblies of Low-Molecular-Weight Gelators. Angew. Chem. Int. Ed. 2015, 54, 4517–4521. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Zhou, J.; Junfeng, S.; Xu, B. Supramolecular Hydrogelators and Hydrogels: From Soft Matter to Molecular Biomaterials. Chem. Rev. 2015, 115, 13165–13307. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.; Dastidar, P. Multidrug-Containing, Salt-Based, Injectable Supramolecular Gels for Self-Delivery, Cell Imaging and Other Materials Applications. Chem. Eur. J. 2016, 22, 14929–14939. [Google Scholar] [CrossRef] [PubMed]
- Shigemitsu, H.; Hamachi, I. Design Strategies of Stimuli-Responsive Supramolecular Hydrogels Relying on Structural Analyses and Cell-Mimicking Approaches. Acc. Chem. Res. 2017, 50, 740–750. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Hayakawa, Y.; Hanabusa, K. Thixotropic Supramolecular Gels Based on l-Lysine Derivatives. Gels 2015, 1, 81–93. [Google Scholar] [CrossRef]
- Liu, G.-F.; Ji, W.; Wang, W.-L.; Feng, C.-L. Multiresponsive Hydrogel Coassembled from Phenylalanine and Azobenzene Derivatives as 3D Scaffolds for Photoguiding Cell Adhesion and Release. ACS Appl. Mater. Interfaces 2015, 7, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Hanabusa, K.; Takata, S.; Fujisaki, M.; Nomura, Y.; Suzuki, M. Fluorescent Gelators of Detection of Explosives. Bull. Chem. Soc. Jpn. 2016, 89, 1391–1401. [Google Scholar] [CrossRef]
- Draper, E.R.; Dietrich, B.; Adam, D.J. Self-assembly, self-sorting, and electronic properties of a diketopyrrolopyyrole hydrogelator. Chem. Commun. 2017, 53, 1864–1867. [Google Scholar] [CrossRef] [PubMed]
- Ren, C.; Shen, J.; Chen, F.; Zeng, H. Rapid Room-temperature Gelation of Crude Oils by a Wetted Powder Gelator. Angew. Chem. Int. Ed. 2017, 56, 3847–3851. [Google Scholar] [CrossRef] [PubMed]
- Fox, M.A.; Dulay, M.T. Heterogeneous photocatalysts. Chem. Rev. 1993, 93, 341–357. [Google Scholar] [CrossRef]
- Hoffmann, M.R.; Martin, S.T.; Choi, W.Y.; Bahnemann, D.W. Environmental Applications of Semiconductor Photocatalyst. Chem. Rev. 1995, 95, 69–96. [Google Scholar] [CrossRef]
- Ono, Y.; Nakashima, K.; Sano, M.; Kanekiyo, Y.; Inoue, K.; Hojo, J.; Shinkai, S. Organic gels are useful as a template for the preparation of hollow fiber silica. Chem. Commun. 1998, 34, 1477–1479. [Google Scholar] [CrossRef]
- Bommel, K.J.C.; Frigeri, A.; Shinkai, S. Organic templates for the generation of inorganic materials. Angew. Chem. Int. Ed. 2003, 42, 980–999. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.H.; Ono, Y.; Hanabusa, K.; Shinkai, S. Creation of Both Right-Handed and Left-Handed Silica Structures by Sol-Gel Transcription of Organogel Fibers Comprised of Chiral Doaminocyclohexane Derivatives. J. Am. Chem. Soc. 2000, 122, 5008–5009. [Google Scholar] [CrossRef]
- Yang, Y.; Suzuki, M.; Kimura, M.; Shirai, H.; Kurose, A.; Hanabusa, K. Nanofiberization of inner helical mesoporous silica using chiral gelato as template under a shear flow. Chem. Commun. 2005, 2032–2034. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Hamasaki, N.; Suzuki, M.; Kimura, M.; Shirai, H.; Hanabusa, K. Preparation of Helical Transition-Metal Oxide Tubes Using Organogelators as Structure-Directing Agents. J. Am. Chem. Soc. 2002, 124, 6550–6551. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Nakajima, Y.; Sato, T.; Shirai, H.; Hanabusa, K. Fabrication of TiO2 using L-lysine-based organogelators as organic templates: Control of the nanostructures. Chem. Commun. 2006, 377–379. [Google Scholar] [CrossRef] [PubMed]
- Hanabusa, K.; Numazawa, T.; Kobayashi, S.; Suzuki, M.; Shirai, H. Preparation of Metal Oxide Nanotubes Using Gelators as Structure-Directing Agents. Macromol. Symp. 2006, 235, 52–56. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, S.; Huo, H.; Huang, Z.; Li, Y.; Li, B.; Yang, Y. Preparation of helical mesoporous tantalum oxide nanotubes through a sol-gel transcription approach. Chem. Asian J. 2013, 8, 709–712. [Google Scholar] [CrossRef] [PubMed]
- Huo, H.; Wang, S.; Li, Y.; Li, B.; Yang, Y. Chiral zirconia nanotubes prepared through a sol-gel transcription approach. J. Mater. Chem. A 2014, 2, 333–338. [Google Scholar] [CrossRef]
- Suzuki, M.; Kikuchi, M.; Hanabusa, K. Photocatalytic Activity of Titanium Oxide Nano-Networks Fabricated through Organogel Route Using L-Isoleucine-Based Gelator. SEN’I GAKKAISHI 2014, 70, 213–217. [Google Scholar] [CrossRef]
- Liu, X.Y.; Li, J.-L. Molecular Gels for Controlled Formation of Micro-/Nano-Structures. In Soft Fibrillar Materials; Fabrication and Applications; Wiley-VCH: Weinheim, Germany, 2013; Section I: Small Molecule Gels, Chapter 5; pp. 163–181. [Google Scholar]
- Chen, X.; Mao, S.S. Titanium Dioxide Nanomaterials: Synthesis, Properties, Modifications, and Applications. Chem. Rev. 2007, 107, 2891–2959. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Selloni, A. Special issue for Titanium dioxide nanomaterials. Chem. Rev. 2014, 114, 9281–10216. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.C.; Yu, J.; Ho, W.; Zhang, L. Preparation of highly photocatalytic active nano-sized TiO2 particles via ultrasonic irradiation. Chem. Commun. 2001, 1942–1943. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhong, X.; Liu, S.; Li, D.; Han, M. Aminolysis Route to Monodisperse Titania Nanorods with Tunable Aspect Ratio. Angew. Chem. Int. Ed. 2005, 44, 3466–3470. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-L.; Peng, Q.; Yi, J.-X.; Wang, X.; Li, Y. Near Monodisperse TiO2 Nanoparticles and Nanorods. Chem. Eur. J. 2006, 12, 2383–2391. [Google Scholar] [CrossRef] [PubMed]
- Zare, M.; Mortezaali, A.; Shafiekhani, A. Photoelectrochemical Determination of Shallow and Deep Trap States of Platinum-Decorated TiO2 Nanotube Arrays for Photocatalytic Applications. J. Phys. Chem. C 2016, 120, 9017–9027. [Google Scholar] [CrossRef]
- Méndez-Mendrano, M.G.; Kowalska, E.; Lehoux, A.; Herissan, A.; Ohtani, B.; Bahena, D.; Briois, V.; Colbeau-Justin, C.; Rodríguez-López, J.L.; Remita, H. Surface Modification of TiO2 with Ag nanoparticles and CuO nanoclusters for Application in Photocatalysis. J. Phys. Chem. C 2016, 120, 5143–5154. [Google Scholar] [CrossRef]
- Chen, Z.; Zhao, J.; Yang, Z.; Ye, Q.; Huang, K.; Hou, C.; Zhao, Z.; You, J.; Li, Y. Fabrication of TiO2/WO3 Composite Nanofibers by Electrospinning and Photocatalytic Performance of the Resultant Fabrics. Ind. Eng. Chem. Res. 2016, 55, 80–85. [Google Scholar] [CrossRef]
- Cai, J.; Wu, X.; Li, S.; Zheng, F. Synthesis of TiO2@WO3/Au Nanocomposite Hollow Spheres with Controllable Size and High Visible-Light-Driven Photocatalytic Activity. ACS Sustain. Chem. Eng. 2016, 4, 1581–1590. [Google Scholar] [CrossRef]
- Lee, K.; Mazare, A.; Schmuki, P. One-Dimensional Titanium Dioxide Nanomaterials: Nanotubes. Chem. Rev. 2014, 114, 9385–9454. [Google Scholar] [CrossRef] [PubMed]
- Ku, S.J.; Jo, G.C.; Bak, C.H.; Kim, S.M.; Shin, Y.R.; Kim, K.H.; Kwon, S.H.; Kim, J.-B. Highly ordered freestanding titanium oxide nanotube arrays using Si-containing block copolymer lithography and atomic layer deposition. Nanotechnology 2013, 24, 085301. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Nakajima, Y.; Yumoto, M.; Kimura, M.; Shirai, H.; Hanabusa, K. Effects of Hydrogel Bonding and van der Waals Interactions on Organogelation Using Designed Low-Molecular-Weight Gelators and Gel Formation at Room Temperature. Langmuir 2003, 19, 8622–8624. [Google Scholar] [CrossRef]



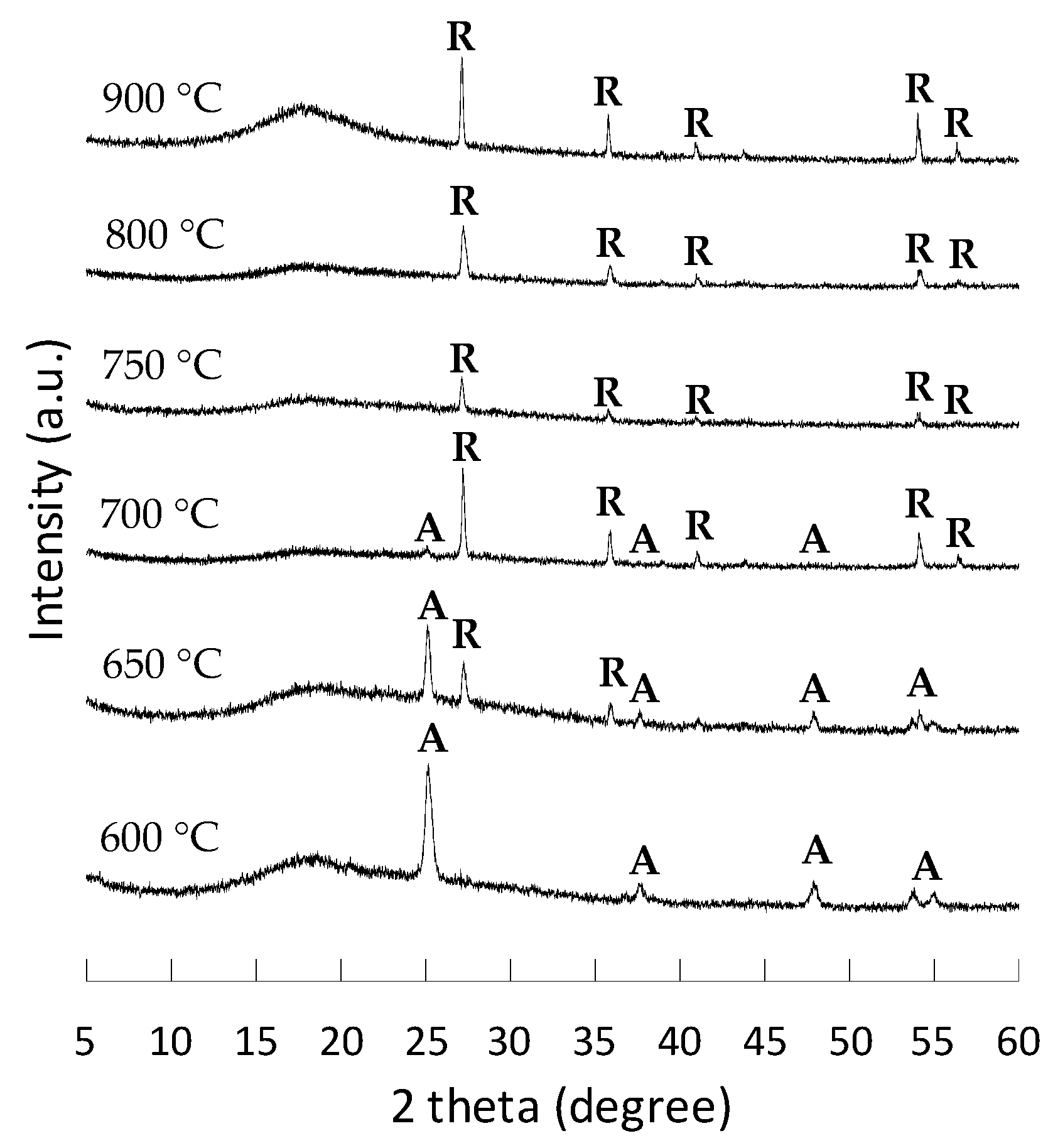




| Calcination | Nanostructure | Crystal | BET Surface Area |
|---|---|---|---|
| 550 °C | Nanotube | Anatase | 19 m2/g |
| 600 °C | Nanotube | Anatase | 18 m2/g |
| 650 °C | Nanotube | Anatase/Rutile | 13 m2/g |
| 700 °C | Nanotube | Anatase/Rutile | 12 m2/g |
| 750 °C | Nanotube | Rutile | 10 m2/g |
| 800 °C | Partly collapse | Rutile | 5 m2/g |
| 900 °C | Collapse | Rutile | 4 m2/g |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suzuki, M.; Tanaka, K.; Kato, Y.; Hanabusa, K. Metal Oxide/TiO2 Hybrid Nanotubes Fabricated through the Organogel Route. Gels 2017, 3, 24. https://doi.org/10.3390/gels3030024
Suzuki M, Tanaka K, Kato Y, Hanabusa K. Metal Oxide/TiO2 Hybrid Nanotubes Fabricated through the Organogel Route. Gels. 2017; 3(3):24. https://doi.org/10.3390/gels3030024
Chicago/Turabian StyleSuzuki, Masahiro, Keita Tanaka, Yukie Kato, and Kenji Hanabusa. 2017. "Metal Oxide/TiO2 Hybrid Nanotubes Fabricated through the Organogel Route" Gels 3, no. 3: 24. https://doi.org/10.3390/gels3030024





