Nanocomposite Perfluorosulfonic Acid/Montmorillonite-Na+ Polymer Membrane as Gel Electrolyte in Hybrid Supercapacitors
Abstract
1. Introduction
2. Results and Discussion
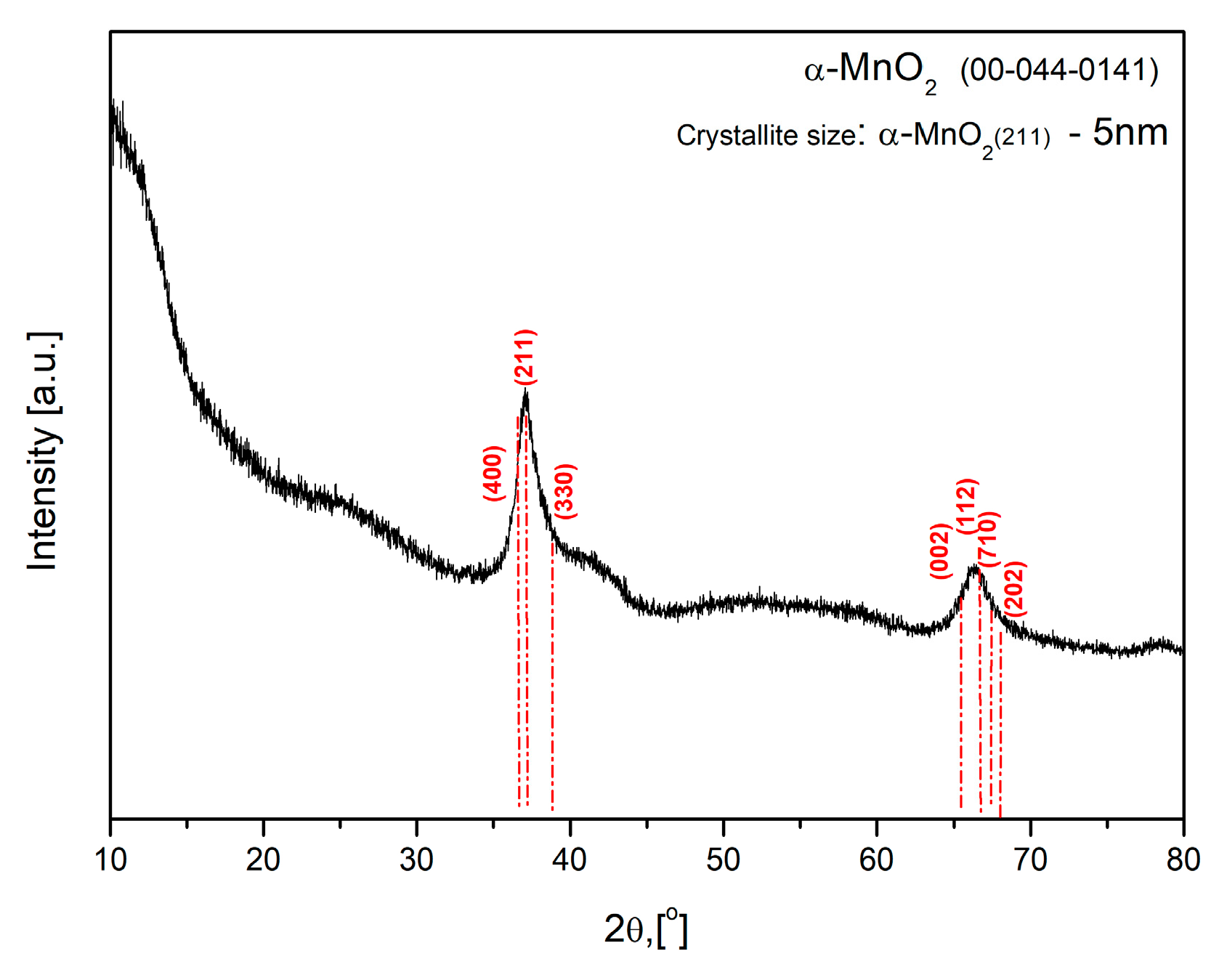
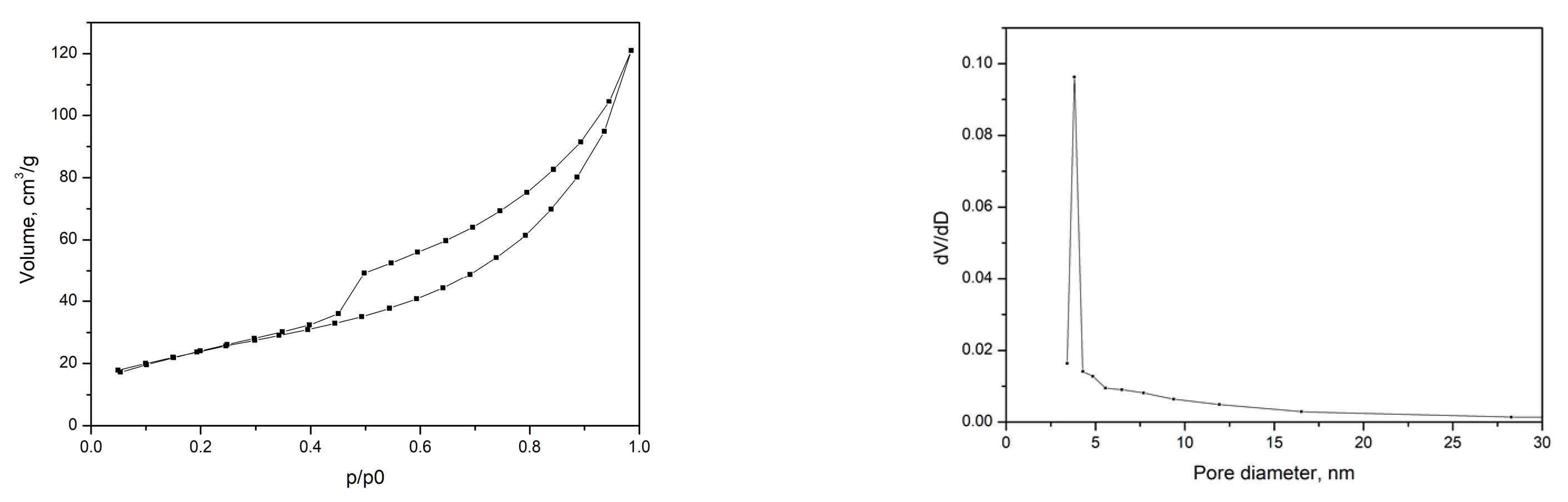
2.1. Physicochemical Characterization of the MnO2 and the Polymer Membrane
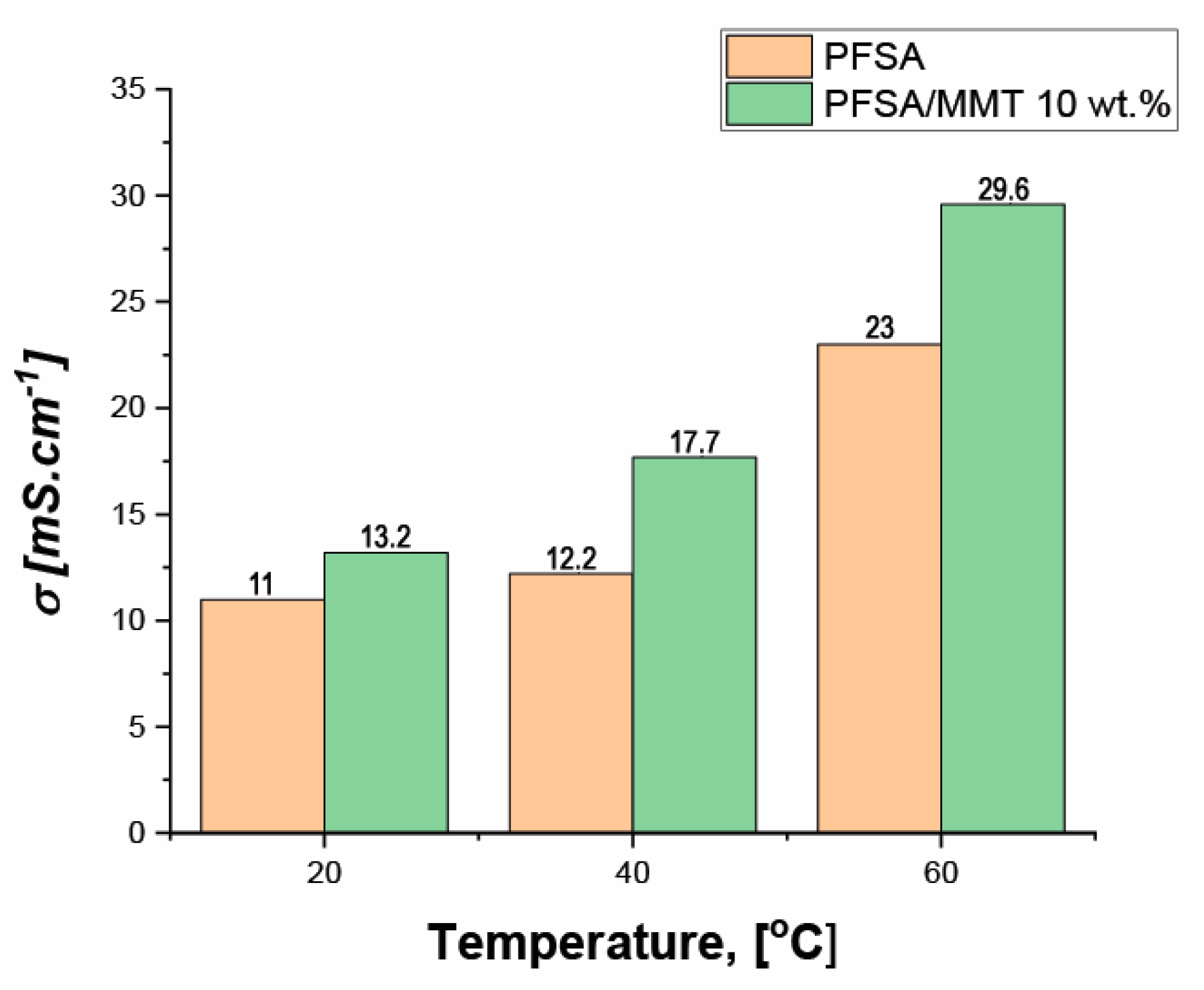
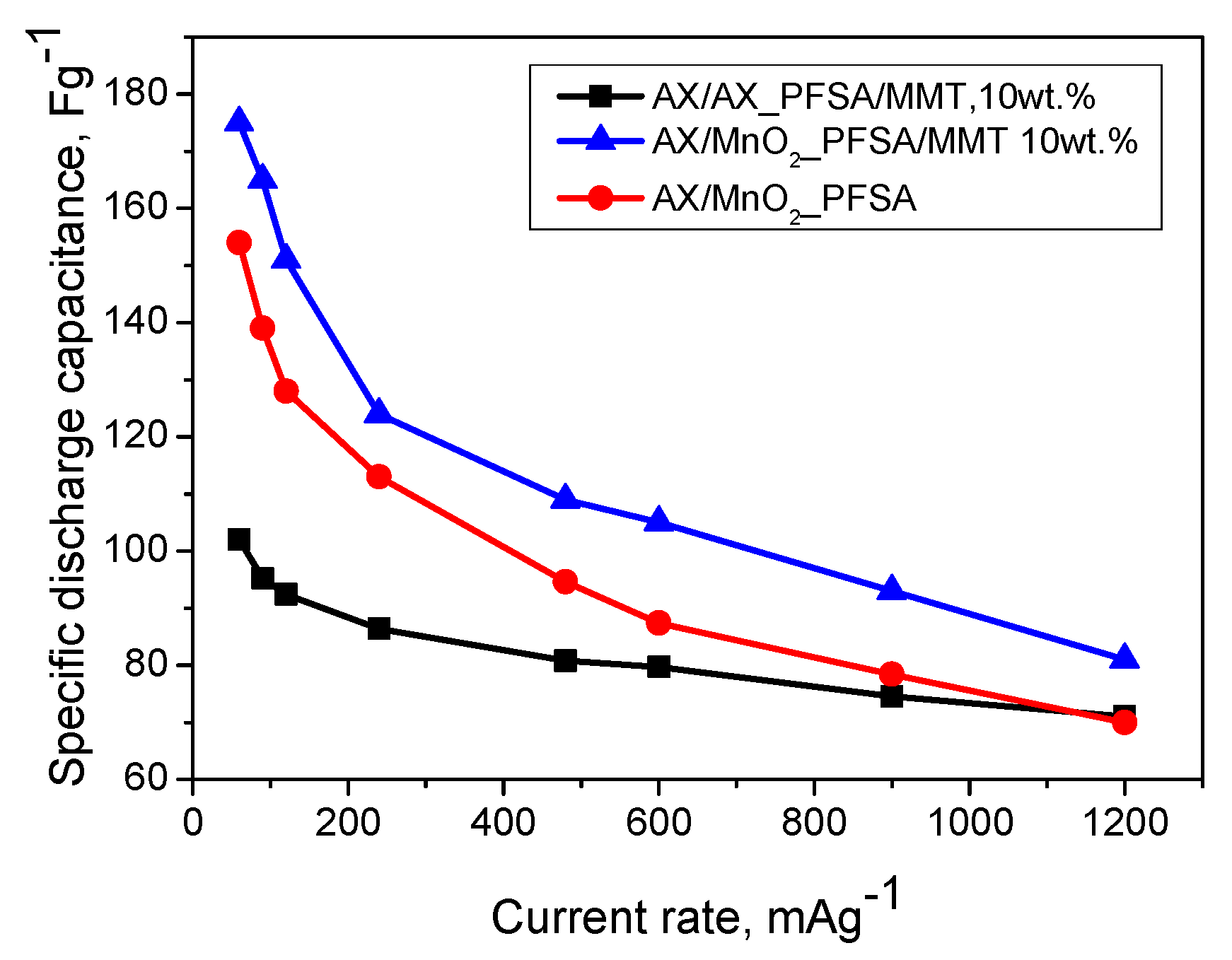
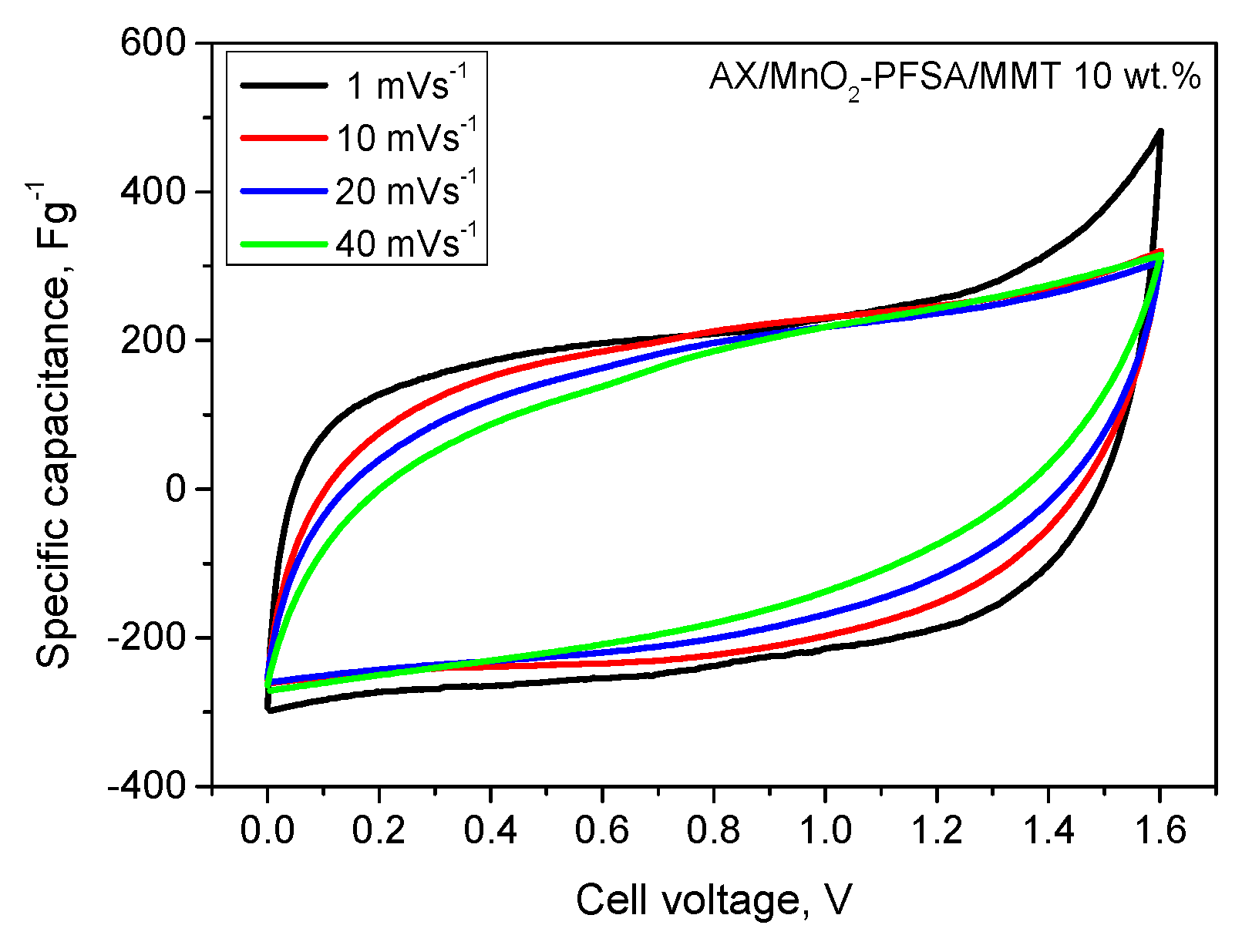
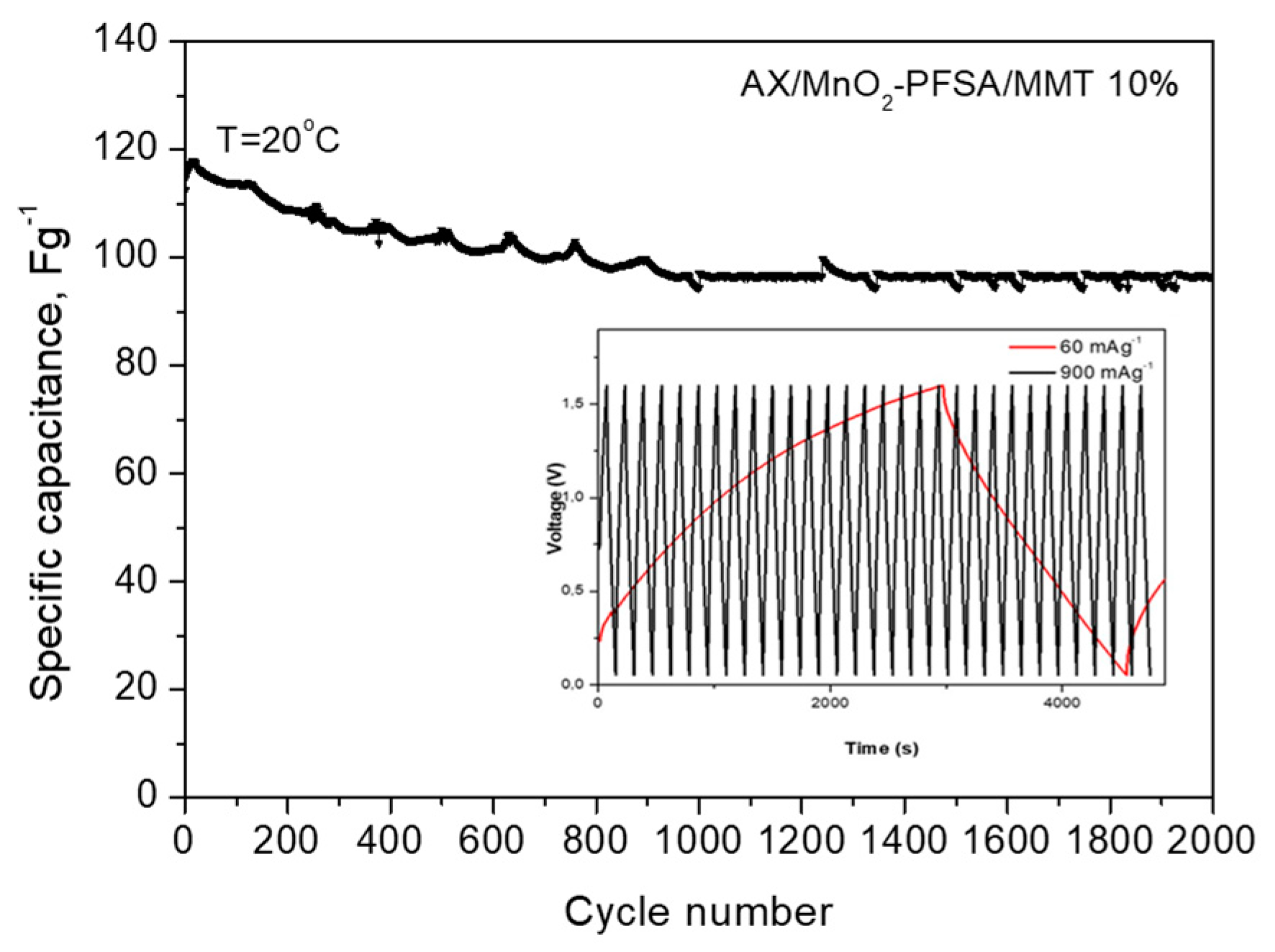
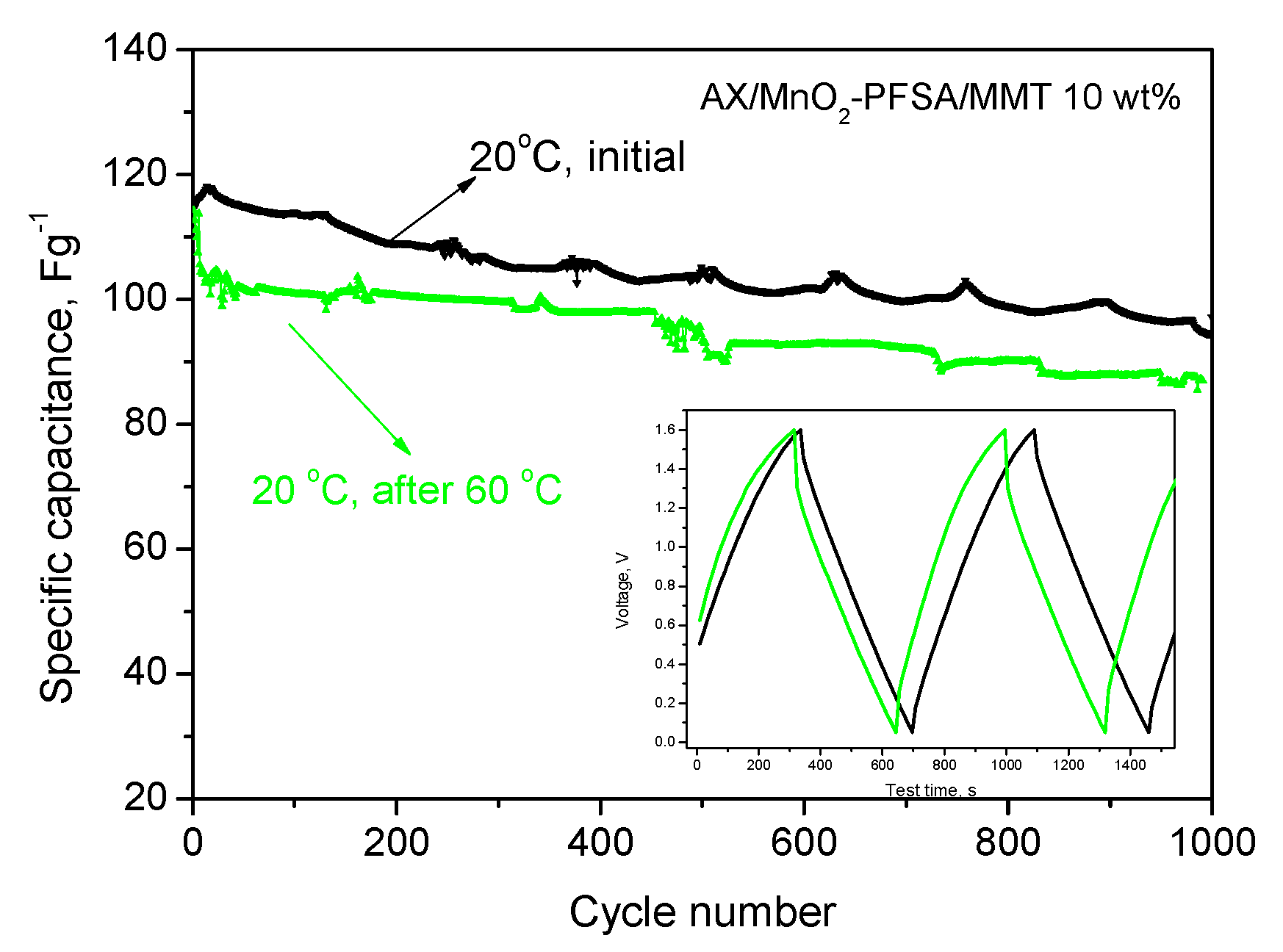
2.2. Electrochemical Results
3. Conclusions
4. Materials and Methods
4.1. Preparation of the MnO2
4.2. Preparation of Composite PFSA/MMT Membranes
4.3. Electrode Preparation and Supercapacitor Cell Assembly
4.4. Physicochemical Characterization of Materials
4.5. Electrochemical Characterization
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Adil, M.; Abdelkareem, M.A.; Sayed, E.T.; Rodriguez, C.; Ramadan, M.; Olabi, A.-G. Progress of Metal Chalcogenides in Supercapacitors. Encycl. Smart Mater. 2022, 2, 424–433. [Google Scholar]
- Feenstra, J.; Granstrom, J.; Sodano, H. Energy harvesting through a backpack employing a mechanically amplified piezoelectric stack. MSSP 2008, 22, 721–734. [Google Scholar] [CrossRef]
- Hunang, C.; Shen, C.; Tang, C.; Chang, S. A wearable yarn-based piezo-resistive sensor. Sens. Actuators A Phys. 2008, 141, 396–403. [Google Scholar] [CrossRef]
- Wu, J.; Zhou, D.; Too, C.O.; Wallace, G.G. Conducting polymer coated lycra. Synth. Met. 2005, 155, 698–701. [Google Scholar] [CrossRef]
- Conway, B.E.; Pell, W. Efficiency Aspects of Charge/Discharge Cycling at Porous Capacitor Electrodes: Behavior of a Hardware Model Circuit. In Proceedings of the 8th International Seminar on Double-Layer Capacitors and Similar Energy Storage Devices, Deerfield Beach, FL, USA, 7–9 December 1998; p. 7. [Google Scholar]
- Arbizzani, C.; Mastragostino, M.; Scosati, B. Handbook of Organic Conductive Molecules and Polymers; Nalwa, H.S., Ed.; Wiley: Chichester, UK, 1997; Volume 4, p. 595. [Google Scholar]
- Holla, S.; Selvakumar, M. Effect of Different Electrolytes on the Supercapacitor Behavior of Single and Multilayered Electrode Materials Based on Multiwalled Carbon Nanotube/Polyaniline Composite. Macromol. Chem. Phys. 2018, 219, 1800213. [Google Scholar] [CrossRef]
- Yuan, Y.; Chen, L.; Li, Y. Functional LiTaO3 filler with tandem conductivity and ferroelectricity for PVDF-based composite solid-state electrolyte. Energy Mater. Devices 2023, 1, 9370004. [Google Scholar] [CrossRef]
- Peng, J.; Lu, D.; Wu, S.; Yang, N.; Cui, Y.; Ma, Z.; Liu, M.; Shi, Y.; Sun, Y.; Niu, J.; et al. Lithium Superionic Conductive Nanofiber-Reinforcing High-Performance Polymer Electrolytes for Solid-State Batteries. J. Am. Chem. Soc. 2024, 146, 11897–11905. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Cui, Y.; Su, H.; Peng, J.; Shi, Y.; Niu, J.; Wang, F. A Chemically Bonded Ultraconformal Layer between the Elastic Solid Electrolyte and Lithium Anode for High-performance Lithium Metal Batteries. Angew. Chem. 2023, 62, e202304339. [Google Scholar] [CrossRef]
- Zhu, M.; Wu, J.; Wang, Y.; Song, M.; Long, L.; Siyal, S.H.; Yang, X.; Sui, G. Recent advances in gel polymer electrolyte for high-performance lithium batteries. J. Energy Chem. 2019, 37, 126–142. [Google Scholar] [CrossRef]
- Quartarone, E.; Mustarelli, P. Electrolytes for solid-state lithium rechargeable batteries: Recent advances and perspectives. Chem. Soc. Rev. 2011, 40, 2525–2540. [Google Scholar] [CrossRef]
- Fan, L.; Wei, S.; Li, S.; Li, Q.; Lu, Y. Recent progress of the solid-state electrolytes for high-energy metal-based batteries. Adv. Energy Mater. 2018, 8, 1702657. [Google Scholar] [CrossRef]
- Du, Y.; Liu, X.; Chen, L.; Yin, S.; Xie, Y.; Li, A.; Liang, X.; Luo, Y.; Wu, F.; Mei, Y.; et al. 3D hierarchical fireproof gel polymer electrolyte towards high-performance and comprehensive safety lithium-ion batteries. Chem. Eng. J. 2023, 476, 146605. [Google Scholar] [CrossRef]
- Lin, Y.; Shang, J.; Liu, Y.; Wang, Z.; Bai, Z.; Ou, X.; Tang, Y. Chlorination Design for Highly Stable Electrolyte toward High Mass Loading and Long Cycle Life Sodium-Based Dual-Ion Battery. Adv. Mater. 2024, 36, 2402702. [Google Scholar] [CrossRef] [PubMed]
- Novakov, C.; Kalinova, R.; Veleva, S.; Ublekov, F.; Dimitrov, I.; Stoyanova, A. Flexible Polymer-Ionic Liquid Films for Supercapacitor Applications. Gels 2023, 9, 338. [Google Scholar] [CrossRef] [PubMed]
- Lamba, P.; Singh, P.; Singh, P.; Singh, P.; Bharti; Kumar, A.; Gupta, M.; Kumar, Y. Recent advancements in supercapacitors based on different electrode materials: Classifications, synthesis methods and comparative performance. J. Energy Storage 2022, 48, 103871. [Google Scholar] [CrossRef]
- Navarra, M.A.; Croce, F.; Scrosati, B. New, high temperature superacid zirconia-doped Nafion™ composite membranes. J. Mater. Chem. 2007, 17, 3210–3215. [Google Scholar] [CrossRef]
- Allodi, V.; Brutti, S.; Giarola, M.; Sgambetterra, M.; Navarra, M.A.; Panero, S.; Mariotto, G. Structural and Spectroscopic Characterization of A Nanosized Sulfated TiO2 Filler and of Nanocomposite Nafion Membranes. Polymers 2016, 8, 68. [Google Scholar] [CrossRef]
- Lufrano, F.; Staiti, P. Performance improvement of Nafion based solid state electrochemical supercapacitor. Electrochim. Acta 2004, 49, 2683–2689. [Google Scholar] [CrossRef]
- RSong, Y.; Park, J.H.; Sivakkumar, S.R.; Kim, S.H.; Ko, J.M.; Park, D.Y.; Jo, S.M.; Kim, D.Y. Supercapacitive properties of polyaniline/Nafion/hydrous RuO2 composite electrodes. J. Power Sources 2007, 166, 297–301. [Google Scholar]
- Kim, B.C.; Ko, J.M.; Wallace, G.G. A novel capacitor material based on Nafion-doped polypyrrole. J. Power Sources 2008, 177, 665–668. [Google Scholar] [CrossRef]
- Kaur, N.; Kishore, D. Montmorillonite: An efficient, heterogeneous and green catalyst for organic synthesis. J. Chem. Pharm. Res. 2012, 4, 991–1015. [Google Scholar]
- Jin, T.; Zhang, S.; Li, T. Transesterification of β-ketoesters with alcohols catalyzed by montmorillonite K-10. Green Chem. 2002, 4, 32–34. [Google Scholar] [CrossRef]
- Jha, A.; Garade, A.C.; Shirai, M.; Rode, C.V. Metal cation-exchanged montmorillonite clay as catalysts for hydroxyalkylation reaction. Appl. Clay Sci. 2013, 74, 141–146. [Google Scholar] [CrossRef]
- Tombácz, E.; Szekeres, M. Colloidal behavior of aqueous montmorillonite suspensions: The specific role of pH in the presence of indifferent electrolytes. Appl. Clay Sci. 2004, 27, 75–94. [Google Scholar] [CrossRef]
- Maiti, S.; Pramanik, A.; Chattopadhyay, S.; De, G.; Mahanty, S. Electrochemical energy storage in montmorillonite K10 clay based composite as supercapacitor using ionic liquid electrolyte. J. Colloid Interface Sci. 2016, 464, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.W.; Ji, K.S.; Lee, J.P.; Park, J.W. Electrochemical characteristics of two types of PEO-based composite electrolyte with functional SiO2. J. Power Sources 2003, 119–121, 415–421. [Google Scholar] [CrossRef]
- Sing, K.S.W. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984). Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Ray, S.S.; Okamoto, M. Polymer/layered silicate nanocomposites: A review from preparation to processing. Prog. Polym. Sci. 2003, 28, 1539–1641. [Google Scholar]
- Uddin, F. Clays, Nanoclays, and Montmorillonite Minerals. Metall. Mater. Trans. A 2008, 39, 2804–2814. [Google Scholar] [CrossRef]
- Rey-Raap, N.; Flores-López, S.; Santos-Gómez, L.; Brigandì, A.; Thomas, M.; Stoyanova, A.; Lufrano, A.; Arenillas, A. Graphene Doped Carbon-Gels and MnO2 for Next Generation of Solid-State Asymmetric Supercapacitors. ChemElectroChem 2023, 10, e202300161. [Google Scholar] [CrossRef]
- Rey-Raap, N.; Angel Menéndez, J.; Arenillas, A. RF xerogels with tailored porosity over the entire nanoscale. Microporous Mesoporous Mater. 2014, 195, 266–275. [Google Scholar] [CrossRef]
- Wang, J.; Yu, X.; Wang, C.; Xiang, K.; Deng, M.; Yin, H. PAMPS/MMT composite hydrogel electrolyte for solid-state supercapacitors. J. Alloys Compd. 2017, 709, 596–601. [Google Scholar] [CrossRef]
- Thomas, M.; Veleva, S.; Karamanova, B.; Brigandì, A.; Rey-Raap, N.; Arenillas, A.; Stoyanova, A.; Lufrano, F. Highly stable and reliable asymmetric solid-state supercapacitors with low self-discharge rates. SM&T 2023, 38, e00770. [Google Scholar]
- Wang, J.; Feng, S.P.; Yang, Y.; Hau, N.Y.; Munro, M.; Ferreira-Yang, E.; Chen, G. Thermal charging phenomenon in electrical double layer capacitors. Nano Lett. 2015, 15, 5784–5790. [Google Scholar] [CrossRef] [PubMed]
- Conway, B.E. Electrochemical Supercapacitors: Scientific Fundamentals and Technological Applications; Springer: Berlin/Heidelberg, Germany, 1999. [Google Scholar] [CrossRef]
- Karamanova, B.; Mladenova, E.; Thomas, M.; Rey-Raap, N.; Arenillas, A.; Lufrano, F.; Stoyanova, A. Electrochemical Performance of Symmetric Solid-State Supercapacitors Based on Carbon Xerogel Electrodes and Solid Polymer Electrolytes. Gels 2023, 9, 983. [Google Scholar] [CrossRef]
- Wang, T.; Zhang, S.; Yan, X.; Lyu, M.; Wang, L.; Bell, J. and and Wang, H.; 2-Methylimidazole-Derived Ni–Co Layered Double Hydroxide Nanosheets as High Rate Capability and High Energy Density Storage Material in Hybrid Supercapacitors. ACS Appl. Mater. Interfaces 2017, 9, 15510–15524. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mladenova, B.; Borisov, G.; Dimitrova, M.; Budurova, D.; Staneva, M.; Ublekov, F.; Stoyanova, A. Nanocomposite Perfluorosulfonic Acid/Montmorillonite-Na+ Polymer Membrane as Gel Electrolyte in Hybrid Supercapacitors. Gels 2024, 10, 452. https://doi.org/10.3390/gels10070452
Mladenova B, Borisov G, Dimitrova M, Budurova D, Staneva M, Ublekov F, Stoyanova A. Nanocomposite Perfluorosulfonic Acid/Montmorillonite-Na+ Polymer Membrane as Gel Electrolyte in Hybrid Supercapacitors. Gels. 2024; 10(7):452. https://doi.org/10.3390/gels10070452
Chicago/Turabian StyleMladenova, Borislava, Galin Borisov, Mariela Dimitrova, Desislava Budurova, Maya Staneva, Filip Ublekov, and Antonia Stoyanova. 2024. "Nanocomposite Perfluorosulfonic Acid/Montmorillonite-Na+ Polymer Membrane as Gel Electrolyte in Hybrid Supercapacitors" Gels 10, no. 7: 452. https://doi.org/10.3390/gels10070452
APA StyleMladenova, B., Borisov, G., Dimitrova, M., Budurova, D., Staneva, M., Ublekov, F., & Stoyanova, A. (2024). Nanocomposite Perfluorosulfonic Acid/Montmorillonite-Na+ Polymer Membrane as Gel Electrolyte in Hybrid Supercapacitors. Gels, 10(7), 452. https://doi.org/10.3390/gels10070452






