On the Mechanism of Drug Release from Polysaccharide Hydrogels Cross-Linked with Magnetite Nanoparticles by Applying Alternating Magnetic Fields: the Case of DOXO Delivery
Abstract
:1. Introduction
2. Results and Discussion
- a)
- functionalization of the magnetite (Fe3O4) NPs with APTMS in order to introduce -NH2 groups on the NPs surface (NPs-NH2); and
- b)
- binding of CMC polymer to the (Fe3O4) NPs-NH2, via the formation of an amide bond between the CMC carboxylic groups and the -NH2 groups on the NPs surface in order to get the hybrid hydrogel.
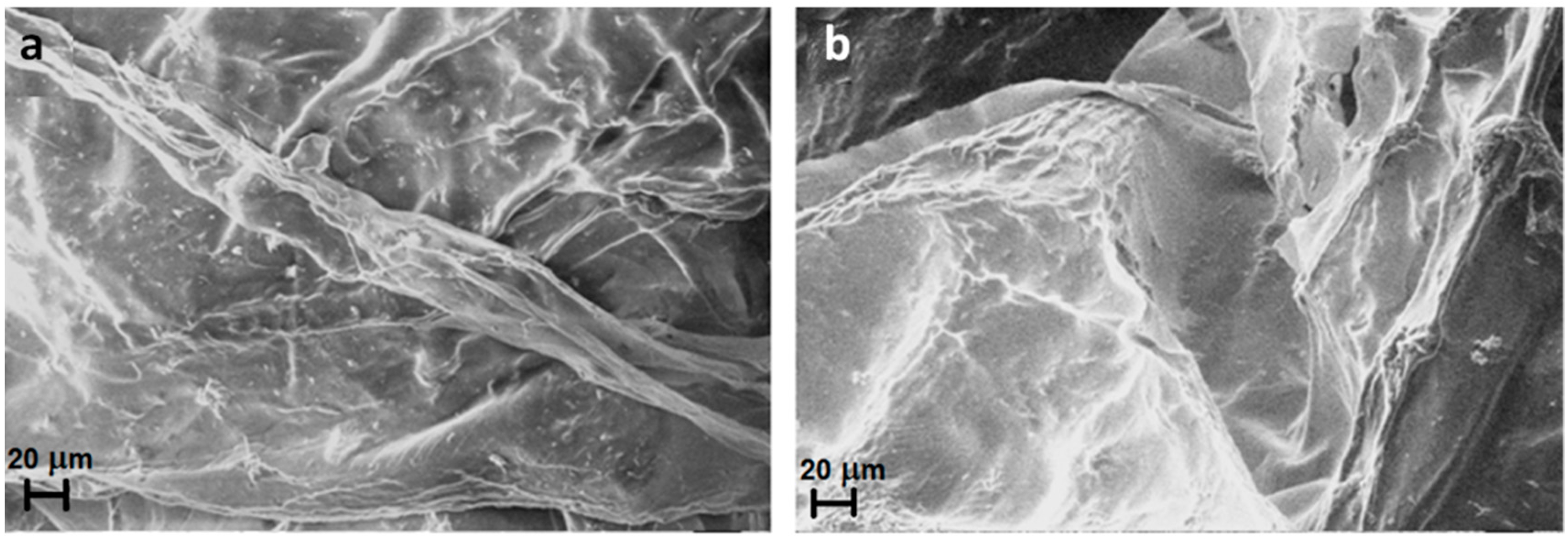
2.1. Characterization and Morphological Analysis of the NPs-NH2 and the Hydrogel CMC-NPs
| Fe3O4 NPs | Fe3O4 NPs-NH2 | |
|---|---|---|
| Size (nm) | 2004 ± 207 | 499 ± 10 |
| PDI ζ-potential | 0.88 ± 0.09 17 ± 2 | 0.35 ± 0.08 23 ± 1 |
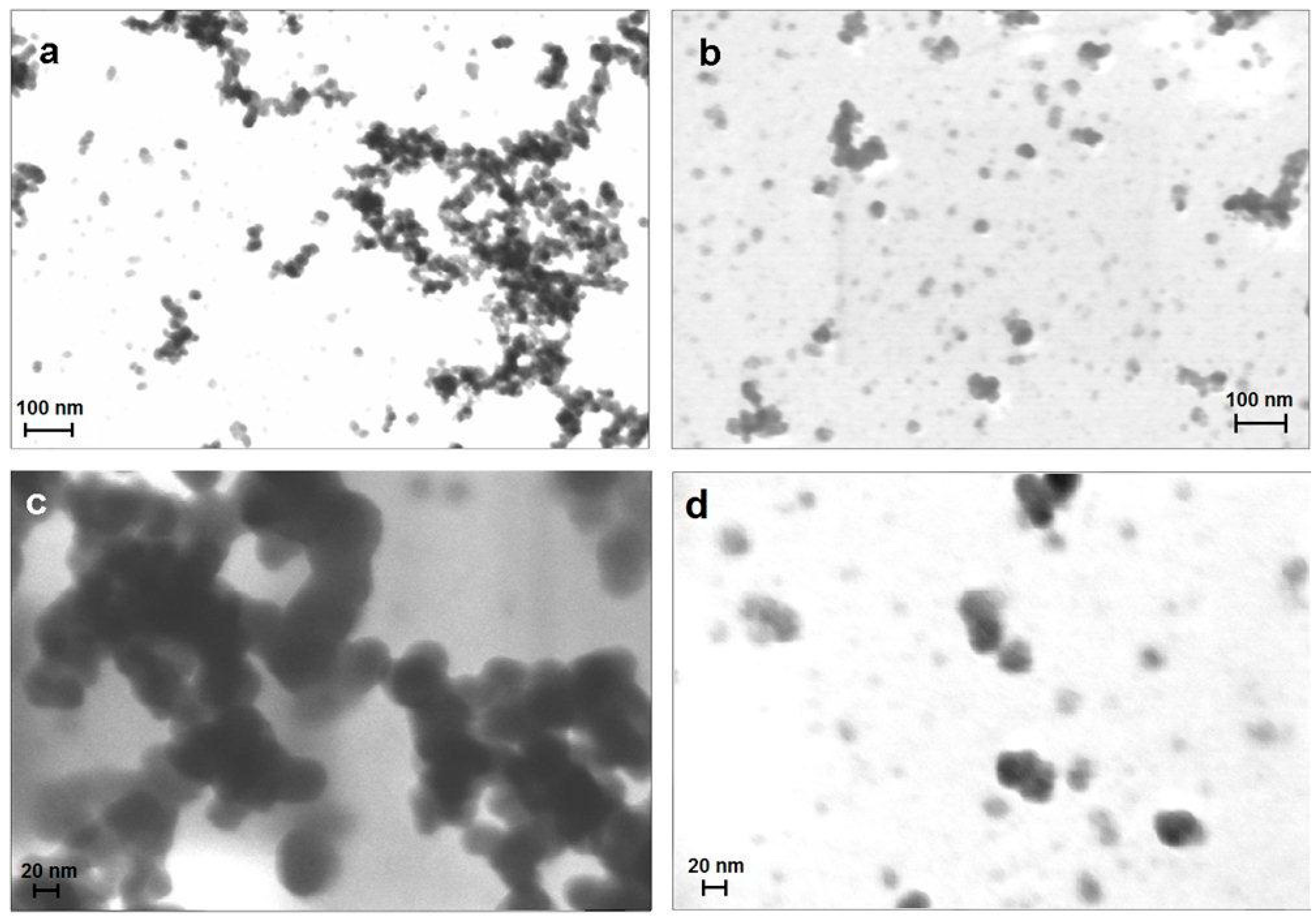
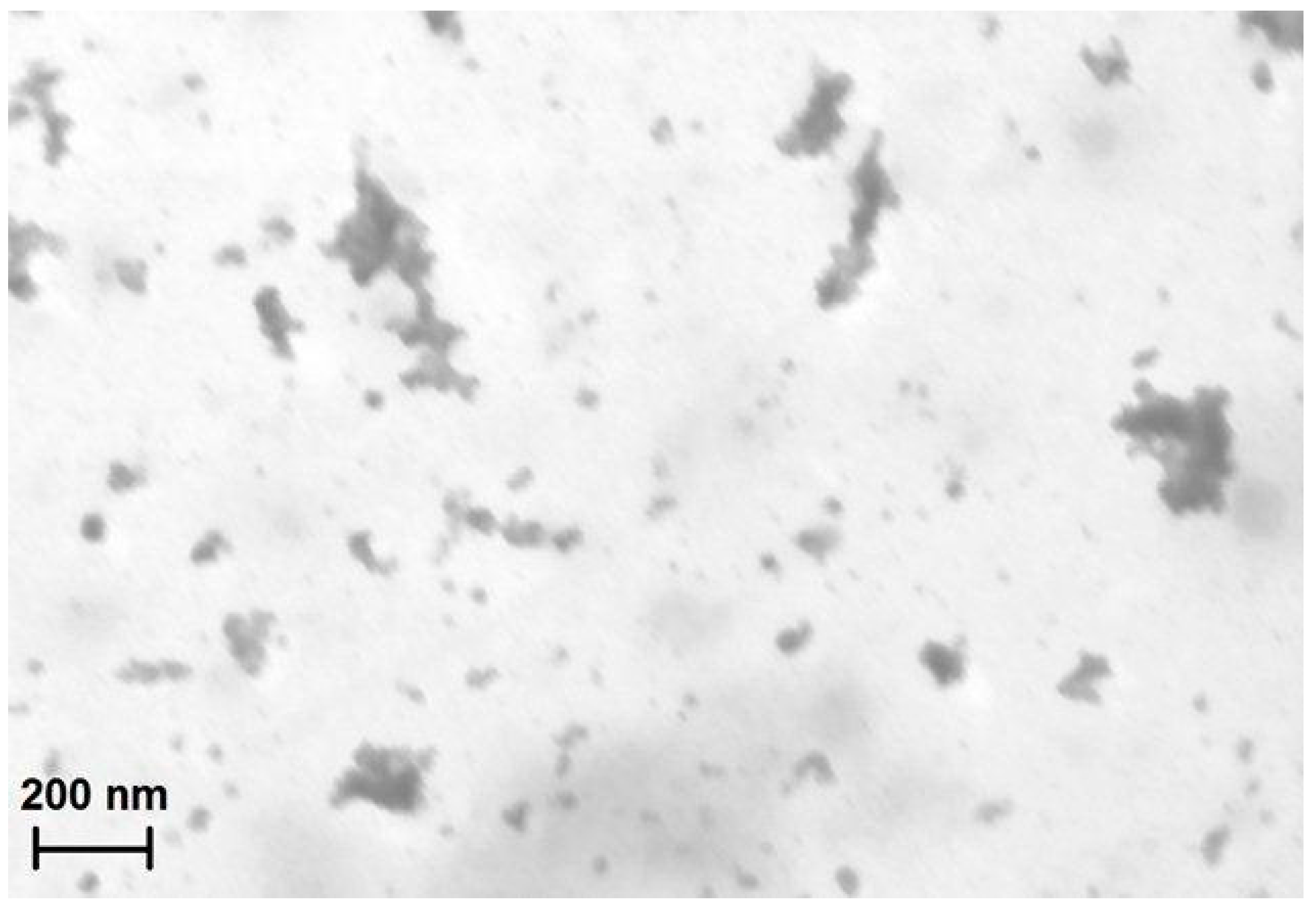
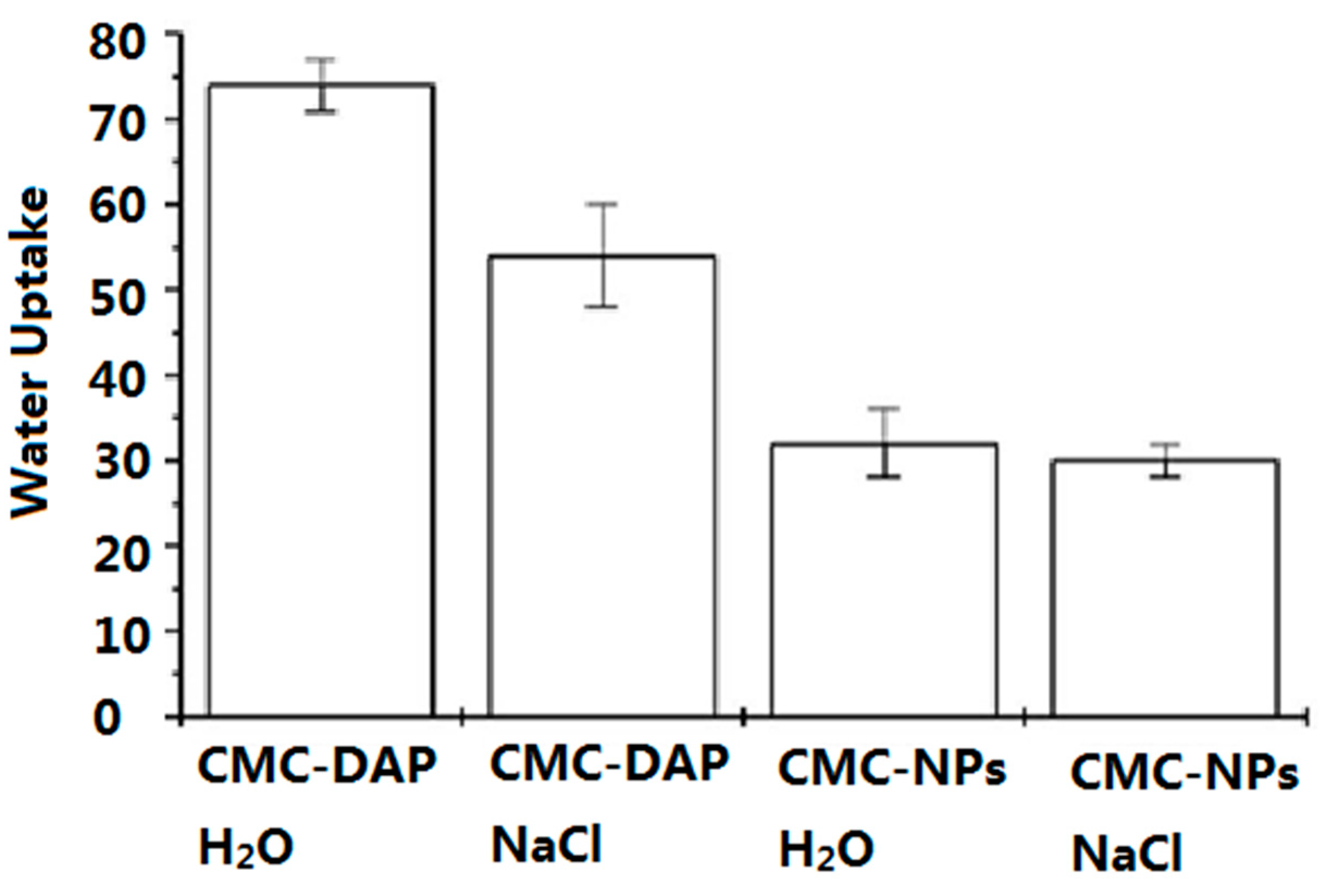
2.2. Water Uptake (WU) and Rheological Measurements
| Sample | G' (Pa) | G" (Pa) |
|---|---|---|
| CMC-DAP | 1025 ± 300 | 300 ± 90 |
| CMC-NPs (Fe3O4) | 3250 ± 120 | 258 ± 30 |
2.3. Magnetic Properties
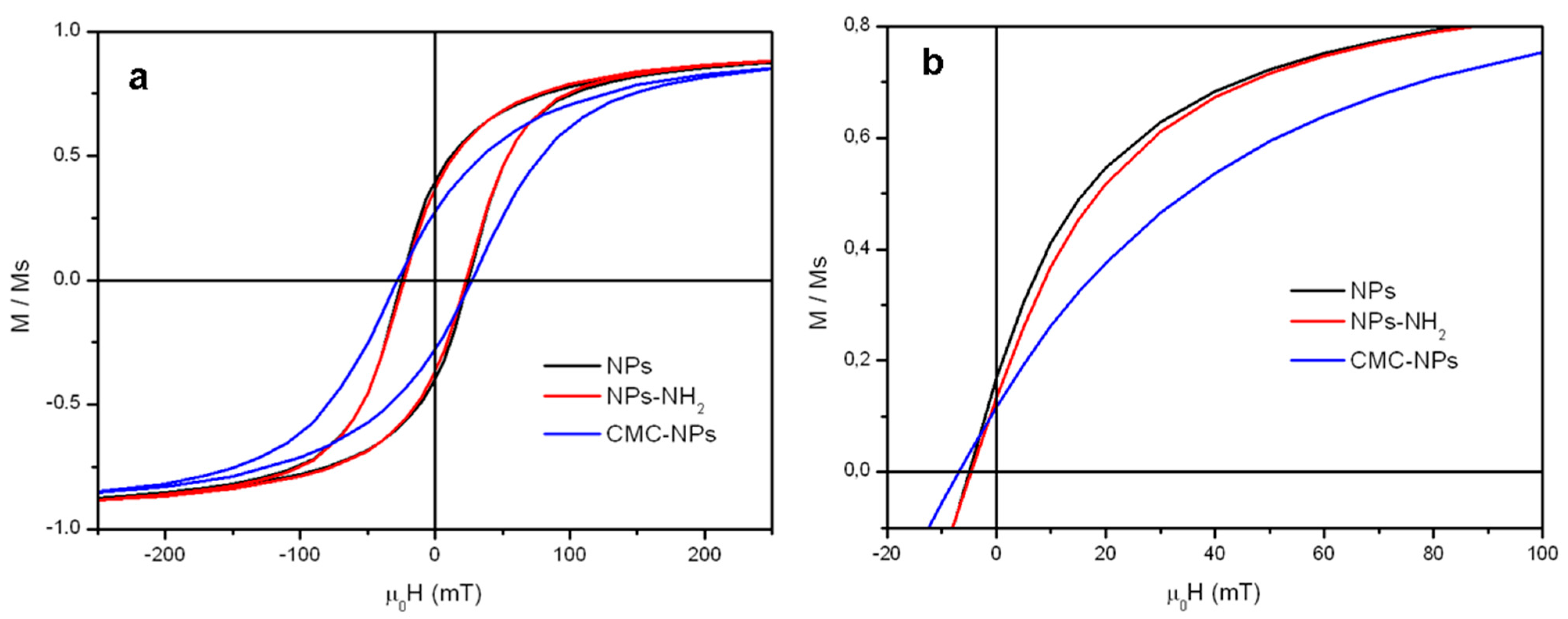
| Sample | 0Hc 2.5 K (mT) a | MS 2.5 K (Am2/kg) b | MR 2.5 K c | 0Hc 300 K (mT) d | MS 300 K (Am2/kg) e | MR 300 K f |
|---|---|---|---|---|---|---|
| NPs | 24.6 | 86 | 0.39 | 5.0 | 76 | 0.17 |
| NPs-NH2 | 23.2 | 83 | 0.37 | 4.6 | 74 | 0.13 |
| CMC-NPs hydrogel | 27.8 | 16 | 0.27 | 6.8 | 14 | 0.12 |
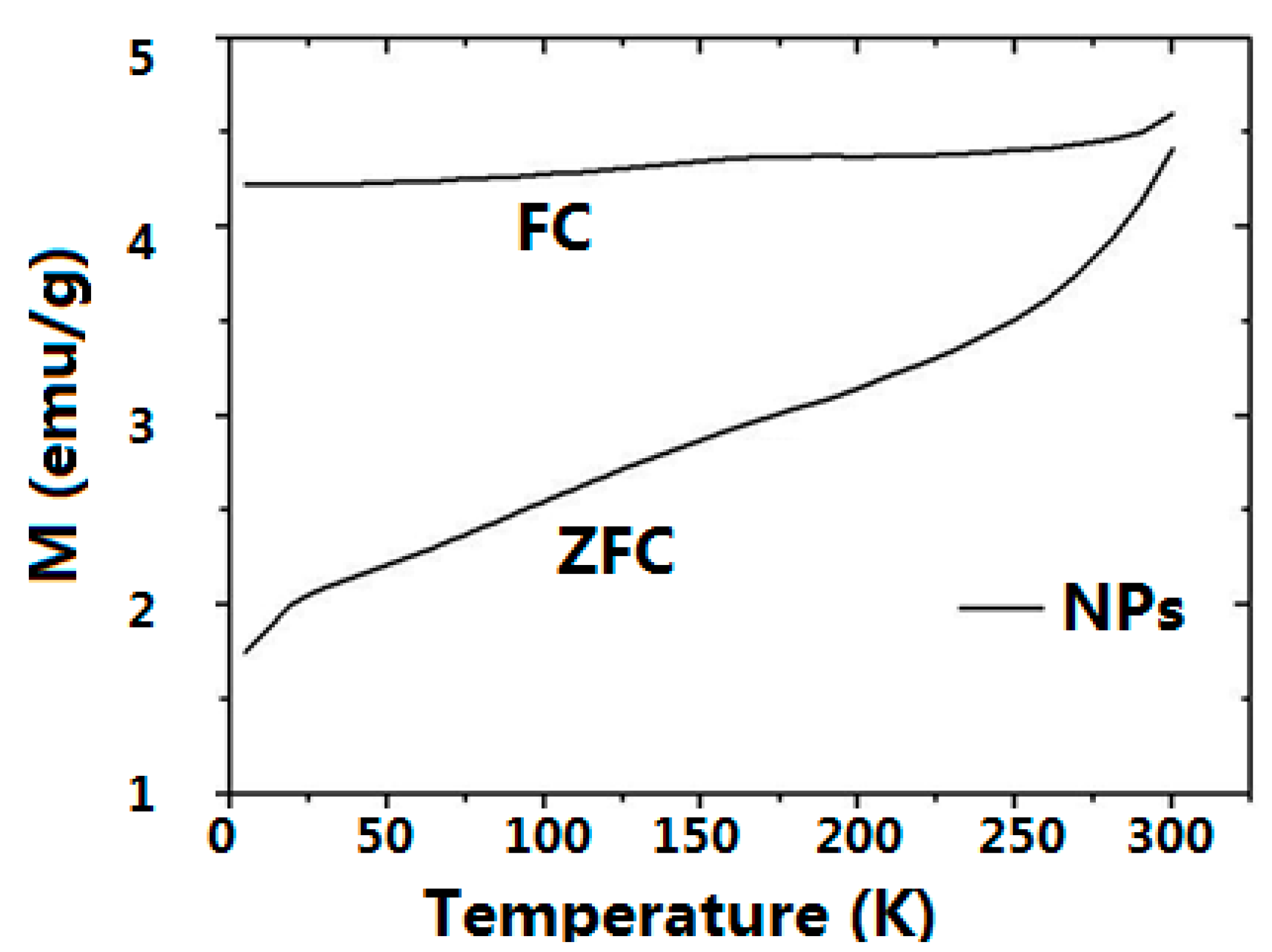
2.4. Biological Tests
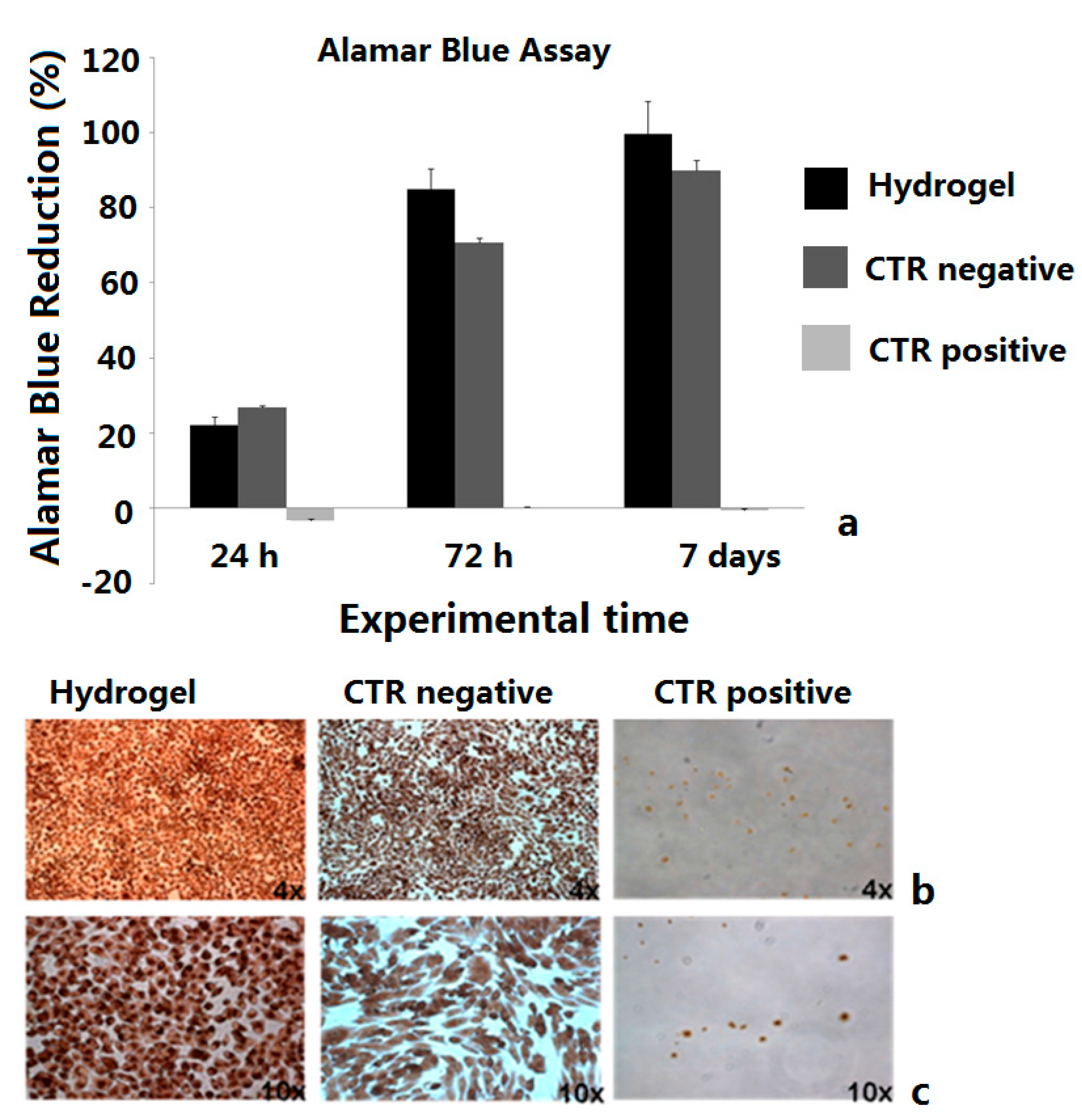
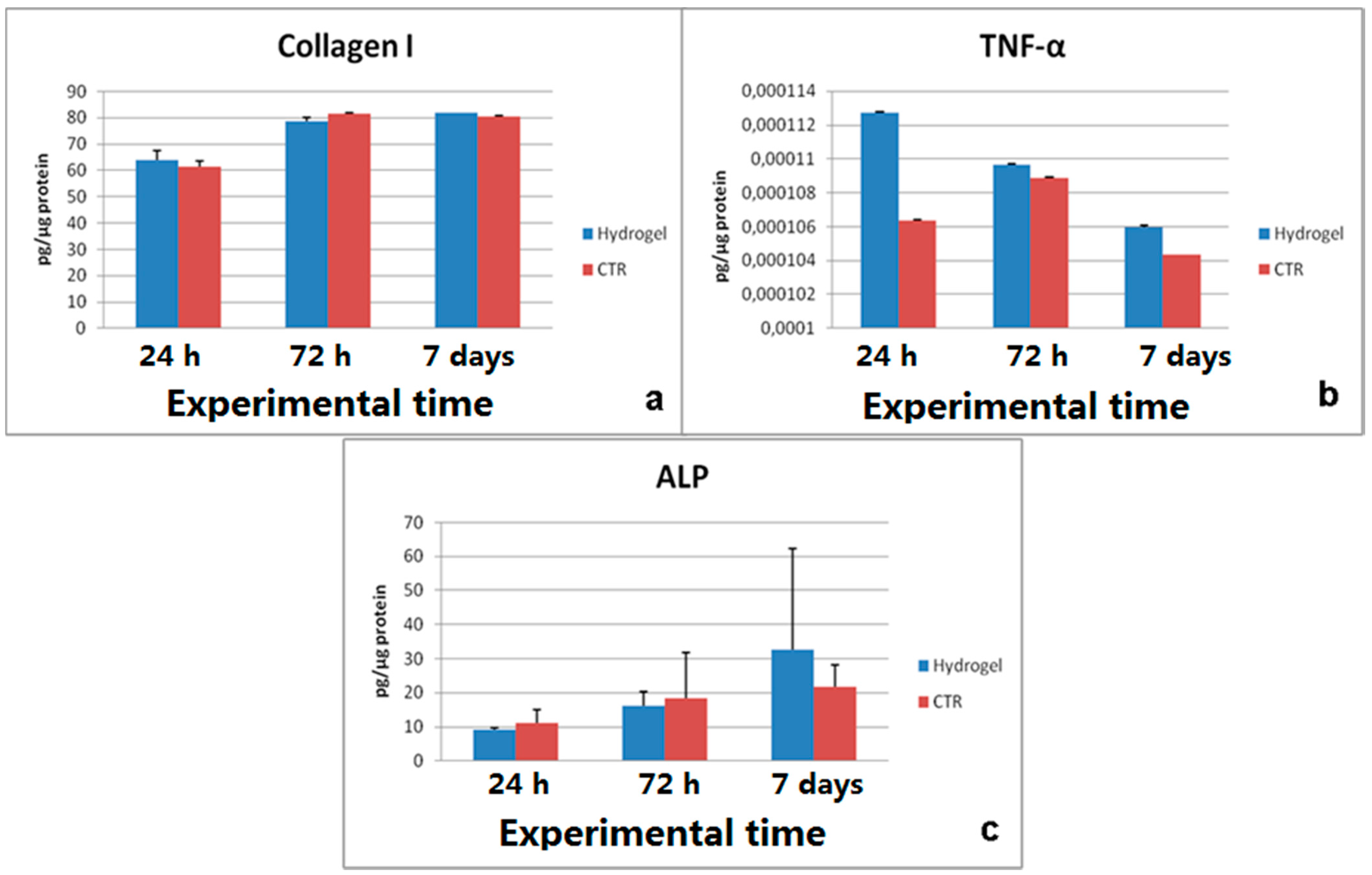
2.5. Release of Doxorubicin from Drug-Loaded CMC-NPs Hydrogel
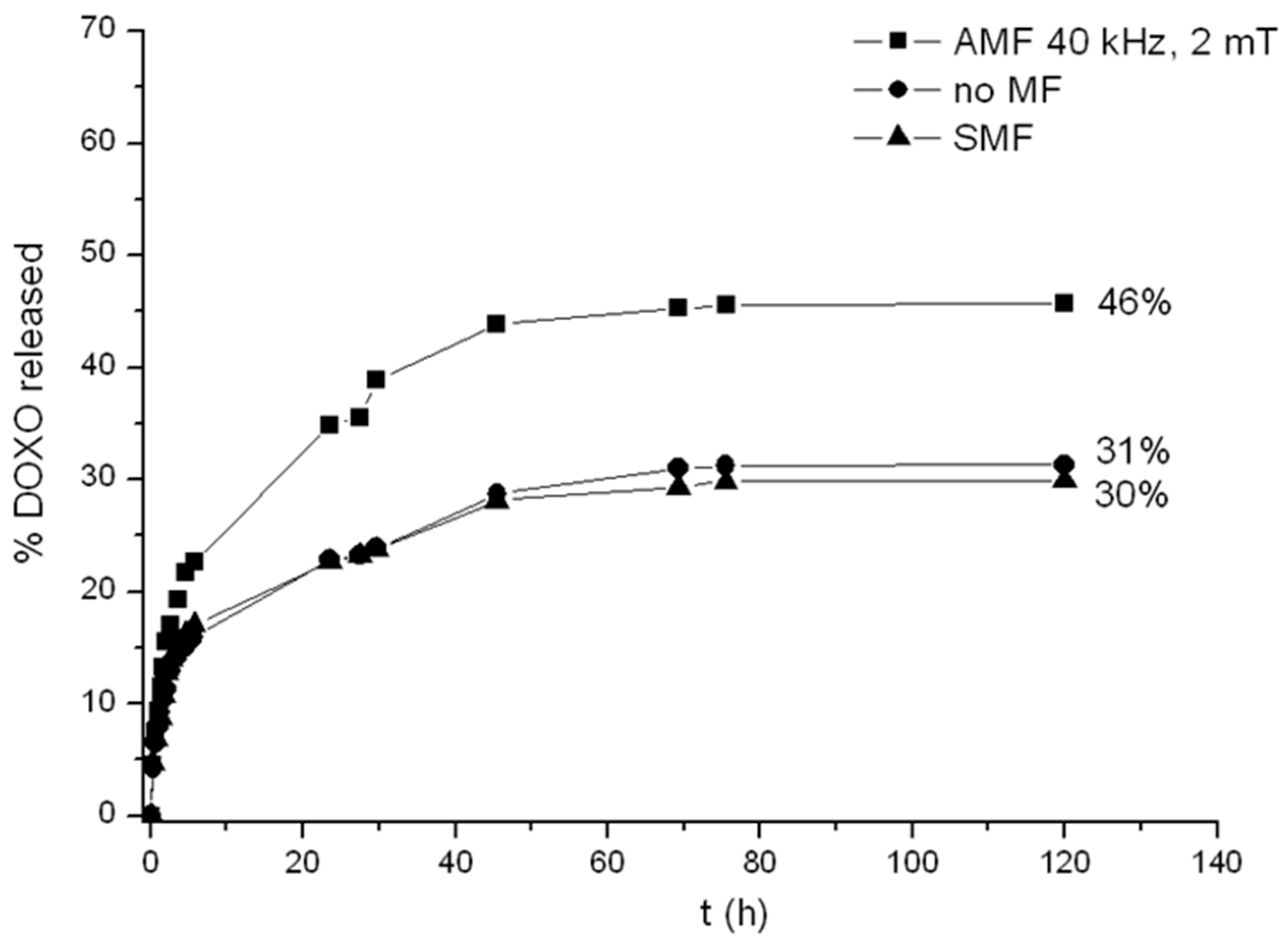

3. Conclusions
4. Materials and methods
4.1. Materials
4.2. Methods
4.2.1. Functionalization of Fe3O4 NPs
4.2.2. Synthesis of the Hybrid Hydrogel
4.2.3. FT-IR and UV-Visible Spectroscopy
4.2.4. Dynamic Light Scattering (DLS)
4.2.5. FESEM and STEM
4.2.6. Water Uptake
4.2.7. Rheological Measurements
4.2.8. Magnetic Properties
4.2.9. Biological Tests
4.2.10. Drug Loading and Release
Acknowledgements
Conflicts of Interest
References
- Mitra, S.; Gaur, U.; Ghosh, P.C.; Maitra, A.N. Tumour targeted delivery of encapsulated dextran-doxorubicin conjugate using chitosan nanoparticles as carrier. J. Control. Release 2001, 74, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Na, K.; Lee, E.S.; Bae, Y.H. Adriamycin loaded pullulan acetate/sulfonamide conjugate nanoparticles responding to tumor pH: pH-dependent cell interaction, internalization and cytotoxicity. J. Control. Release 2003, 87, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Orive, G.; Hernández, R.M.; Gascon, A.R.; Luis Pedraz, J. Micro and nano drug delivery systems in cancer therapy. Cancer Ther. 2005, 3, 131–138. [Google Scholar]
- Panyam, J.; Labhasetwar, V. Biodegradable nanoparticles for drug and gene delivery to cells and tissue. Adv. Drug Deliv. Rev. 2003, 55, 329–347. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.S.; Chen, D.H. Magnetic nanoparticles grafted with cyclodextrin for hydrophobic drug delivery. Chem. Mater. 2007, 19, 6345–6349. [Google Scholar] [CrossRef]
- Jain, T.K.; Reddy, M.K.; Morales, M.A.; Leslie-Pelecky, D.L.; Labhasetwar, V. Biodistribution, clearance, and biocompatibility of iron oxide magnetic nanoparticles in rats. Mol. Pharm. 2008, 5, 316–327. [Google Scholar] [CrossRef] [PubMed]
- Jain, T.K.; Richey, J.; Strand, M.; Leslie-Pelecky, D.L.; Flask, C.A.; Labhasetwar, V. Magnetic nanoparticles with dual functional properties: Drug delivery and magnetic resonance imaging. Biomaterials 2008, 29, 4012–4021. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.J.; Hoffman, J.M.; Ebara, M.; Hoffman, A.S.; Estournès, C.; Wattiaux, A.; Stayton, P.S. Dual magnetic-/temperature-responsive nanoparticles for microfluidic separations and assays. Langmuir 2007, 23, 7385–7391. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.H.; Liu, T.Y.; Liu, D.M.; Chen, S.Y. Nano-ferrosponges for controlled drug release. J. Control. Release 2007, 121, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Yong, Y.; Bai, Y.; Li, Y.; Cui, Y.; Xia, C. Preparation and application of polymer-grafted magnetic nanoparticles for lipase immobilization. J. Magn. Magn. Mater. 2008, 320, 2350–2355. [Google Scholar] [CrossRef]
- Rahimi, M.; Wadajkar, A.; Subramanian, K.; Yousef, M.; Cui, W.; Hsieh, J.T.; Nguyen, K.T. In vitro evaluation of novel polymer-coated magnetic nanoparticles for controlled drug delivery. Nanomedicine 2010, 6, 672–680. [Google Scholar]
- Wu, C.L.; He, H.; Gao, H.J.; Gan, L.; Ma, J.R.; An, Y.L.; Shi, L.Q. Synthesis of Fe3O4@SiO2@polymer nanoparticles for controlled drug release. Sci. China Chem. 2010, 53, 514–518. [Google Scholar] [CrossRef]
- Nattama, S.; Rahimi, M.; Wadajkar, A.S.; Koppolu, B.; Hua, J.; Nwariaku, F.; Nguyen, K.T. Characterization of polymer coated magnetic nanoparticles for targeted treatment of cancer. In Engineering in Medicine and Biology Workshop; IEEE: Dallas, TX, USA, 2007; pp. 35–38. [Google Scholar]
- Butoescu, N.; Jordan, O.; Burdet, P.; Stadelmann, P.; Petri-Fink, A.; Hofmann, H.; Doelker, E. Dexamethasone-containing biodegradable superparamagnetic microparticles for intra-articular administration: Physicochemical and magnetic properties, in vitro and in vivo drug release. Eur. J. Pharm. Biopharm. 2009, 72, 529–538. [Google Scholar] [CrossRef] [PubMed]
- Butoescu, N.; Seemayer, C.A.; Foti, M.; Jordan, O.; Doelker, E. Dexamethasone-containing PLGA Zuperparamagnetic microparticles as carriers for the local treatment of arthritis. Biomaterials 2009, 30, 1772–1780. [Google Scholar] [CrossRef] [PubMed]
- Perez, J.M.; O’Loughin, T.; Simeone, F.J.; Weissleder, R.; Josephson, L. DNA-based magnetic nanoparticle assembly acts as a magnetic relaxation nanoswitch allowing screening of DNA-cleaving agents. J. Am. Chem. Soc. 2002, 124, 2856–2857. [Google Scholar] [CrossRef] [PubMed]
- Kinsella, J.M.; Ivanisevic, A. Enzymatic clipping of DNA wires coated with magnetic nanoparticles. J. Am. Chem. Soc. 2005, 127, 3276–3277. [Google Scholar] [CrossRef]
- Xu, C.J.; Xu, K.M.; Gu, H.W.; Zheng, R.K.; Liu, H.; Zhang, X.X.; Guo, Z.H.; Xu, B. Dopamine as a robust anchor to immobilize functional molecules on the iron oxide shell of magnetic nanoparticles. J. Am. Chem. Soc. 2004, 126, 9938–9939. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, J.E.; Lee, J.; Yu, J.H.; Kim, B.C.; An, K.; Hwang, Y.; Shin, C.-H.; Park, J.-G.; Kim, J.; et al. Magnetic fluorescent delivery vehicle using uniform mesoporous silica spheres embedded with monodispersed magnetic and semiconductor nanocrystals. J. Am. Chem. Soc. 2006, 128, 688–689. [Google Scholar] [CrossRef] [PubMed]
- Lu, A.H.; Salabas, E.L.; Schüth, F. Magnetic nanoparticles: synthesis, protection, functionalization, and application. Angew. Chem. Int. Ed. Engl. 2007, 46, 1222–1244. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.Q.; Chen, Y.H.; Yuan, R.X.; Chen, G.H.; Blanco, E.; Gao, J.M.; Shuai, X.T. Folate-encoded and Fe3O4-loaded polymeric micelles for dual targeting of cancer cells. Polymer 2008, 48, 3477–3485. [Google Scholar] [CrossRef]
- Peppas, N.A. Hydrogels. In Biomaterials Science: An Introduction to Materials in Medicine, 2th ed.; Ratner, B.D., Hoffman, A.S., Schoen, F.J., Lemons, J.E., Eds.; Academic Press: New York, 2004; pp. 100–107. [Google Scholar]
- Drury, L.; Mooney, D. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef] [PubMed]
- Giani, G.; Fedi, S.; Barbucci, R. Hybrid Magnetic Hydrogel: A Potential System for Controlled Drug Delivery by Means of Alternating Magnetic Fields. Polymers 2012, 4, 1157–1169. [Google Scholar] [CrossRef]
- Liang, Y.; Zhang, L.; Jiang, W.; Lei, W. Embedding magnetic nanoparticles into polysaccharide-based hydrogels for magnetically assisted bioseparation. Chem. Phys. Chem. 2007, 8, 2367–2372. [Google Scholar] [PubMed]
- Heim, E.; Harling, S.; Pöhlig, K.; Ludwig, F.; Menzel, H.; Schilling, M. Fluxgate magnetorelaxometry of superparamagnetic nanoparticles for hydrogel characterization. J. Magn. Magn. Mater. 2007, 311, 150–154. [Google Scholar] [CrossRef]
- Barbucci, R.; Pasqui, D.; Giani, G.; De Cagna, M.; Fini, M.; Giardino, R.; Atrei, A. A novel strategy for engineering hydrogels with ferromagnetic nanoparticles as crosslinkers of the polymer chains Potential applications as a targeted drug delivery system. Soft Matter 2011, 7, 5558–5565. [Google Scholar] [CrossRef]
- Uva, M.; Pasqui, D.; Mencuccini, L.; Fedi, S.; Barbucci, R. Influence of alternating and static magnetic fields on drug release from hybrid hydrogels containing magnetic nanoparticles. J. Biomater. Nanobiotechnol. 2014, 5, 116–127. [Google Scholar] [CrossRef]
- Ahmed, N.; Jaafar-Maalej, C.; Eissa, M.M.; Fessi, H.; Elaissari, A. New oil-in-water magnetic emulsion as contrast agent for in vivo magnetic resonance imaging (MRI). J. Biomed. Nanotechnol. 2013, 9, 1579–1585. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, B.; Liu, F.; Luo, J. In vivo tomographic imaging with fluorescence and MRI using tumor-targeted dual-labeled nanoparticles. Int. J. Nanomed. 2014, 9, 33–41. [Google Scholar]
- Koppolu, B.; Bhavsar, Z.; Wadajkar, A.S.; Nattama, S.; Rahimi, M.; Nwariaku, F.; Nguyen, K.T. Temperature-sensitive polymer-coated magnetic nanoparticles as a potential drug delivery system for targeted therapy of thyroid cancer. J. Biomed. Nanotechnol. 2012, 8, 983–890. [Google Scholar] [CrossRef] [PubMed]
- Wahajuddin; Arora, S. Superparamagnetic iron oxide nanoparticles: magnetic nanoplatforms as drug carriers. Int. J. Nanomed. 2012, 9, 3445–3471. [Google Scholar] [CrossRef]
- Wang, C.; Xu, R.; Tang, L. The local heating effect by magnetic nanoparticles aggregate on support lipid bilayers. J. Biomed. Nanotechnol. 2013, 9, 1210–1215. [Google Scholar] [CrossRef] [PubMed]
- Samanta, B.; Yan, H.; Fischer, N.O.; Shi, J.; Jerry, D.J.; Rotello, V.M. Protein-passivated Fe3O4 nanoparticles: Low toxicity and rapid heating for thermal therapy. J. Mater. Chem. 2008, 9, 1204–1208. [Google Scholar] [CrossRef]
- Ta, H.T.; Dass, C.R.; Larson, I.; Choong, P.F.M.; Dunstan, D.E. A chitosan–dipotassium orthophosphate hydrogel for the delivery of doxorubicin in the treatment of osteosarcoma. Biomaterials 2009, 30, 3605–3613. [Google Scholar] [CrossRef] [PubMed]
- Barbucci, R.; Giani, G.; Fedi, S.; Bottari, S.; Casolaro, M. Biohydrogels with Magnetic Nanoparticles as Crosslinker: Characteristics and Potential Use for Controlled Antitumor Drug-Delivery. Acta Biomater. 2012, 8, 4244–4252. [Google Scholar] [CrossRef] [PubMed]
- Xu, R. Progress in nanoparticles characterization: Sizing and zeta potential measurement. Particuology 2008, 6, 112–115. [Google Scholar] [CrossRef]
- Rodríguez-Arco, L.; López-López, M.; González-Caballero, F.; Durán, J. Steric repulsion as a way to achieve the required stability for the preparation of ionic liquid-based ferrofluids. J. Colloid Interf. Sci. 2011, 357, 252–254. [Google Scholar] [CrossRef]
- Pasqui, D.; Torricelli, P.; De Cagna, M.; Fini, M.; Barbucci, R. Carboxymethyl cellulose-hydroxyapatite hybrid hydrogel as a composite material for bone tissue engineering applications. J. Biomed. Mater. Res. Part A 2014, 102, 1568–1579. [Google Scholar] [CrossRef]
- Wu, Y.; Hussain, M.; Fassihi, R. Development of a simple analytical methodology for determination of glucosamine release from modified release matrix tablets. J. Pharm. Biomed. Anal. 2005, 38, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Goya, G.F.; Berquó, T.S.; Fonseca, F.C.; Morales, M.P. Static and dynamic magnetic properties of spherical magnetite nanoparticles. J. Appl. Phys. 2003, 94, 3520–3528. [Google Scholar] [CrossRef]
- Kechrakos, D.; Trohidou, K.N. Competition between dipolar and exchange interparticle interactions in magnetic nanoparticle films. J. Magn. Magn. Mat. 2003, 262, 107–110. [Google Scholar] [CrossRef]
- Rosensweig, R.E. Heating magnetic fluid with alternating magnetic field. J. Magn. Magn. Mat. 2002, 252, 370–374. [Google Scholar] [CrossRef]
- Riedinger, A.; Guardia, P.; Curcio, A.; Garcia, M.A.; Cingolani, R.; Manna, L.; Pellegrino, T. Subnanometer Local Temperature Probing and Remotely Controlled Drug Release Based on Azo-Functionalized Iron Oxide Nanoparticles. Nano Lett. 2013, 13, 2399–2406. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Xu, Z.; Nystrom, A.M.; Zhang, K.; Leonard, J.R.; Wooley, K.L. 19F- and fluorescently labeled micelles as nanoscopic assemblies for chemotherapeutic delivery. Bioconjug. Chem. 2008, 19, 2492–2498. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Li, D.; Zhang, L.; Li, J.; Wang, E. Monodisperse mesoporous superparamagnetic single-crystal magnetite nanoparticles for drug delivery. Biomaterials 2009, 30, 1881–1889. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Xie, J.; Tong, Y.W.; Wang, C.H. Effect of PEG conformation and particle size on the cellular uptake efficiency of nanoparticles with the HepG2 cells. J. Control. Release 2007, 118, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Ilg, P. Stimuli-responsive hydrogels cross-linked by magnetic nanoparticles. Soft Matter 2013, 9, 3465–3468. [Google Scholar] [CrossRef]
- Cheng, D.; Li, X.; Zhang, G.; Shi, H. Morphological effect of oscillating magnetic nanoparticles in killing tumor cells. Nanoscale Res. Lett. 2014, 9, 195. [Google Scholar] [CrossRef] [PubMed]
- Pasqui, D.; De Cagna, M.; Barbucci, R. Polysaccharide-Based Hydrogels: The Key Role of Water in Affecting Mechanical Properties. Polymers 2012, 4, 1517–1534. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uva, M.; Mencuccini, L.; Atrei, A.; Innocenti, C.; Fantechi, E.; Sangregorio, C.; Maglio, M.; Fini, M.; Barbucci, R. On the Mechanism of Drug Release from Polysaccharide Hydrogels Cross-Linked with Magnetite Nanoparticles by Applying Alternating Magnetic Fields: the Case of DOXO Delivery. Gels 2015, 1, 24-43. https://doi.org/10.3390/gels1010024
Uva M, Mencuccini L, Atrei A, Innocenti C, Fantechi E, Sangregorio C, Maglio M, Fini M, Barbucci R. On the Mechanism of Drug Release from Polysaccharide Hydrogels Cross-Linked with Magnetite Nanoparticles by Applying Alternating Magnetic Fields: the Case of DOXO Delivery. Gels. 2015; 1(1):24-43. https://doi.org/10.3390/gels1010024
Chicago/Turabian StyleUva, Marianna, Lorenzo Mencuccini, Andrea Atrei, Claudia Innocenti, Elvira Fantechi, Claudio Sangregorio, Melania Maglio, Milena Fini, and Rolando Barbucci. 2015. "On the Mechanism of Drug Release from Polysaccharide Hydrogels Cross-Linked with Magnetite Nanoparticles by Applying Alternating Magnetic Fields: the Case of DOXO Delivery" Gels 1, no. 1: 24-43. https://doi.org/10.3390/gels1010024
APA StyleUva, M., Mencuccini, L., Atrei, A., Innocenti, C., Fantechi, E., Sangregorio, C., Maglio, M., Fini, M., & Barbucci, R. (2015). On the Mechanism of Drug Release from Polysaccharide Hydrogels Cross-Linked with Magnetite Nanoparticles by Applying Alternating Magnetic Fields: the Case of DOXO Delivery. Gels, 1(1), 24-43. https://doi.org/10.3390/gels1010024









