Aspergillus Section Terrei and Antifungals: From Broth to Agar-Based Susceptibility Testing Methods
Abstract
1. Introduction
2. Materials and Methods
2.1. Fungal Strains
2.2. Antifungal Agents
2.3. Inoculum Preparation and AFST
2.4. Interpretation of Results
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kontoyiannis, D.P.; Marr, K.A.; Park, B.J.; Alexander, B.D.; Anaissie, E.J.; Walsh, T.J.; Ito, J.; Andes, D.R.; Baddley, J.W.; Brown, J.M.; et al. Prospective surveillance for invasive fungal infections in hematopoietic stem cell transplant recipients, 2001–2006: Overview of the Transplant-Associated Infection Surveillance Network (TRANSNET) Database. Clin. Infect Dis. 2010, 50, 1091–1100. [Google Scholar] [CrossRef] [PubMed]
- Pappas, P.G.; Alexander, B.D.; Andes, D.; Hadley, S.; Kauffman, C.A.; Freifeld, A.; Anaissie, E.J.; Brumble, L.M.; Herwaldt, L.; Ito, J.; et al. Invasive fungal infections among organ transplant recipients: Results of the Transplant-Associated Infection Surveillance Network (TRANSNET). Clin. Infect Dis. 2010, 50, 1101–1111. [Google Scholar] [CrossRef] [PubMed]
- Taccone, F.S.; Abeele, A.-M.; den Abeele, V.; Bulpa, P.; Misset, B.; Meersseman, W.; Cardoso, T.; Paiva, J.-A.; Blasco-Navalpotro, M.; De Laere, E.; et al. Epidemiology of invasive aspergillosis in critically ill patients: Clinical presentation, underlying conditions, and outcomes. Crit. Care 2015, 19, 7. [Google Scholar] [CrossRef] [PubMed]
- Rüping, M.J.G.T.; Gerlach, S.; Fischer, G.; Lass-Flörl, C.; Hellmich, M.; Vehreschild, J.J.; Cornely, O.A. Environmental and clinical epidemiology of Aspergillus terreus: Data from a prospective surveillance study. J. Hosp. Infect. 2011, 78, 226–230. [Google Scholar] [CrossRef]
- Tritz, D.M.; Woods, G.L. Fatal disseminated infection with Aspergillus terreus in immunocompromised hosts. Clin. Infect Dis. 1993, 16, 118–122. [Google Scholar] [CrossRef]
- Lass-Flörl, C.; Dietl, A.M.; Kontoyiannis, D.P.; Brock, M. Aspergillus terreus Species Complex. Clin. Microbiol. Rev. 2021, 34, e0031120. [Google Scholar] [CrossRef]
- Vahedi Shahandashti, R.; Lass-Flörl, C. Antifungal resistance in Aspergillus terreus: A current scenario. Fungal Genet Biol. 2019, 131, 103247. [Google Scholar] [CrossRef]
- Cavassin, F.B.; Baú-Carneiro, J.L.; Vilas-Boas, R.R.; Queiroz-Telles, F. Sixty years of Amphotericin B: An overview of the main antifungal agent used to treat invasive fungal infections. Infect. Dis. Ther. 2021, 10, 115–147. [Google Scholar] [CrossRef]
- Escribano, P.; Peláez, T.; Recio, S.; Bouza, E.; Guinea, J. Characterization of clinical strains of Aspergillus terreus complex: Molecular identification and antifungal susceptibility to azoles and amphotericin B. Clin. Microbiol. Infect. 2012, 18, E24–E26. [Google Scholar] [CrossRef]
- Walsh, T.J.; Petraitis, V.; Petraitiene, R.; Field-Ridley, A.; Sutton, D.; Ghannoum, M.; Sein, T.; Schaufele, R.; Peter, J.; Bacher, J.; et al. Experimental pulmonary aspergillosis due to Aspergillus terreus: Pathogenesis and treatment of an emerging fungal pathogen resistant to amphotericin B. J. Infect. Dis. 2003, 188, 305–319. [Google Scholar] [CrossRef]
- Tortorano, A.; Prigitano, A.; Dho, G.; Biraghi, E.; Stevens, D.; Ghannoum, M.; Nolard, N.; Viviani, M. In vitro activity of amphotericin B against Aspergillus terreus isolates from different countries and regions. J. Chemother. Florence Italy 2008, 20, 756–757. [Google Scholar] [CrossRef] [PubMed]
- Heo, M.S.; Shin, J.H.; Choi, M.J.; Park, Y.-J.; Lee, H.S.; Koo, S.H.; Gil Lee, W.; Kim, S.H.; Shin, M.-G.; Suh, S.-P.; et al. Molecular identification and Amphotericin B susceptibility testing of clinical isolates of Aspergillus from 11 hospitals in Korea. Ann. Lab. Med. 2015, 35, 602–610. [Google Scholar] [CrossRef] [PubMed]
- Perfect, J.R. The antifungal pipeline: A reality check. Nat. Rev. Drug Discov. 2017, 16, 603–616. [Google Scholar] [CrossRef] [PubMed]
- Zoran, T.; Sartori, B.; Sappl, L.; Aigner, M.; Sánchez-Reus, F.; Rezusta, A.; Chowdhary, A.; Taj-Aldeen, S.J.; Arendrup, M.C.; Oliveri, S.; et al. Azole-Resistance in Aspergillus terreus and related species: An emerging problem or a rare phenomenon? Front. Microbiol. 2018, 9, 516. [Google Scholar] [CrossRef] [PubMed]
- Arendrup, M.C.; Jensen, R.H.; Grif, K.; Skov, M.; Pressler, T.; Johansen, H.K.; Lass-Flörl, C. In vivo emergence of Aspergillus terreus with reduced azole susceptibility and a Cyp51a M217I alteration. J. Infect. Dis. 2012, 206, 981–985. [Google Scholar] [CrossRef]
- Herbrecht, R.; Denning, D.W.; Patterson, T.F.; Bennett, J.E.; Greene, R.E.; Oestmann, J.W.; Kern, W.V.; Marr, K.A.; Ribaud, P.; Lortholary, O.; et al. Invasive fungal infections group of the European Organisation for Research and Treatment of Cancer and the Global Aspergillus Study group. Voriconazole versus amphotericin B for primary therapy of invasive aspergillosis. N. Engl. J. Med. 2002, 347, 408–415. [Google Scholar] [CrossRef]
- Stewart, E.R.; Thompson, G.R. Treatment of primary pulmonary aspergillosis: An assessment of the evidence. J. Fungi 2016, 2, 25. [Google Scholar] [CrossRef]
- Thompson, G.R.; Wiederhold, N.P. Isavuconazole: A comprehensive review of spectrum of activity of a new triazole. Mycopathologia 2010, 170, 291–313. [Google Scholar] [CrossRef]
- Berkow, E.L.; Lockhart, S.R.; Ostrosky-Zeichner, L. Antifungal susceptibility testing: Current approaches. Clin. Microbiol. Rev. 2020, 33, e00069-19. [Google Scholar] [CrossRef]
- Rex, J.; Pfaller, M.A.; Galgiani, J.N.; Bartlett, M.S.; Espinel-Ingroff, A.; Ghannoum, M.A.; Lancaster, M.; Odds, F.C.; Rinaldi, M.G.; Walsh, T.J.; et al. Development of interpretive breakpoints for antifungal susceptibility testing: Conceptual framework and analysis of in vitro-in vivo correlation data for fluconazole, itraconazole, and Candida infections. Clin. Infect. Dis. 1997, 24, 235–247. [Google Scholar] [CrossRef]
- CLSI. Performance Standards for Antimicrobial Susceptibility Testing, 2nd ed.; CLSI supplement M61; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2013. [Google Scholar]
- Arendrup, M.C.; Guinea, J.; Cuenca-Estrella, M.; Meletiadis, J.; Mouton, J.W.; Lagrou, k.; Howard, S.J.; the Subcommittee on Antifungal Susceptibility Testing (AFST) of the ESCMID European Committee for Antimicrobial Susceptibility Testing (EUCAST). Method for the Determination of Broth Dilution Minimum Inhibitory Concentrations of Antifungal Agents for Conidia Forming Moulds. EUCAST Definitive Document DEF 9.3.2. Available online: https://www.aspergillus.org.uk/wp-content/uploads/2016/03/EUCAST_E_Def_9_3_Mould_testing_definitive_0.pdf (accessed on 1 February 2023).
- Gupta, P.; Khare, V.; Kumar, D.; Ahmad, A.; Banerjee, G.; Singh, M. Comparative evaluation of disc diffusion and E-test with broth micro-dilution in susceptibility testing of amphotericin b, voriconazole and caspofungin against clinical Aspergillus isolates. J. Clin. Diagn. Res. 2015, 9, DC04–DC07. [Google Scholar] [CrossRef] [PubMed]
- Risslegger, B.; Zoran, T.; Lackner, M.; Aigner, M.; Sánchez-Reus, F.; Rezusta, A.; Chowdhary, A.; Taj-Aldeen, S.; Arendrup, M.; Oliveri, S.; et al. A prospective international Aspergillus terreus survey: An EFISG, ISHAM and ECMM joint study. Clin. Microbiol. Infect. 2017, 23, 776.e1–776.e5. [Google Scholar] [CrossRef] [PubMed]
- Houbraken, J.; Kocsubé, S.; Visagie, C.M.; Yilmaz, N.; Wang, X.-C.; Meijer, M.; Kraak, B.; Hubka, V.; Bensch, K.; Samson, R.A.; et al. Classification of Aspergillus, Penicillium, Talaromyces and related genera (Eurotiales): An overview of families, genera, subgenera, sections, series and species. Stud. Mycol. 2020, 95, 5–169. [Google Scholar] [CrossRef] [PubMed]
- Espinel-Ingroff, A.; Rezusta, A. E-Test Method for testing susceptibilities of Aspergillus spp. to the new triazoles voriconazole and posaconazole and to established antifungal agents: Comparison with NCCLS broth microdilution method. J. Clin. Microbiol. 2002, 40, 2101–2107. [Google Scholar] [CrossRef]
- Espinel-Ingroff, A. Comparison of the e-test with the NCCLS M38-p method for antifungal susceptibility testing of common and emerging pathogenic filamentous fungi. J. Clin. Microbiol. 2001, 39, 1360–1367. [Google Scholar] [CrossRef]
- Dannaoui, E.; Espinel-Ingroff, A. Antifungal susceptibly testing by concentration gradient strip Etest method for fungal isolates: A review. J. Fungi 2019, 5, 108. [Google Scholar] [CrossRef]
- CLSI. Epidemiological Cutoff Values for Antifungal Susceptibility Testing, 3rd ed.; CLSI supplement M59; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2020. [Google Scholar]
- Vahedi-Shahandashti, R.; Dietl, A.M.; Binder, U.; Nagl, M.; Würzner, R.; Lass-Flörl, C. Aspergillus terreus and the interplay with amphotericin b: From resistance to tolerance? Antimicrob. Agents Chemother. 2022, 66, e0227421. [Google Scholar] [CrossRef]
- Meletiadis, J.; Geertsen, E.; Curfs-Breuker, I.; Meis, J.F.; Mouton, J.W. Intra- and interlaboratory agreement in assessing the in vitro activity of micafungin against common and rare Candida species with the EUCAST, CLSI, and Etest methods. Antimicrob. Agents Chemother. 2016, 60, 6173–6178. [Google Scholar] [CrossRef]
- Lamoth, F.; Lewis, R.E.; Kontoyiannis, D.P. Role and interpretation of antifungal susceptibility testing for the management of invasive fungal infections. J. Fungi 2021, 7, 17. [Google Scholar] [CrossRef]
- Chryssanthou, E.; Cuenca-Estrella, M. Comparison of the EUCAST-AFST broth dilution method with the CLSI reference broth dilution method (M38-A) for susceptibility testing of posaconazole and voriconazole against Aspergillus spp. Clin. Microbiol. Infect. 2006, 12, 901–904. [Google Scholar] [CrossRef][Green Version]
- Sanguinetti, M.; Posteraro, B. Susceptibility testing of fungi to antifungal drugs. J. Fungi 2018, 4, 110. [Google Scholar] [CrossRef]
- McCarty, T.P.; Luethy, P.M.; Baddley, J.W.; Pappas, P.G. Clinical utility of antifungal susceptibility testing. JAC-Antimicrobal Resist. 2022, 4, dlac067. [Google Scholar] [CrossRef]
- Posteraro, B.; Sanguinetti, M. The future of fungal susceptibility testing. Future Microbiol. 2014, 9, 947–967. [Google Scholar] [CrossRef] [PubMed]
- Cuenca-Estrella, M.; Gomez-Lopez, A.; Mellado, E.; Rodriguez-Tudela, J.L. Correlation between the procedure for antifungal susceptibility testing for Candida spp. of the European Committee on Antibiotic Susceptibility Testing (EUCAST) and four commercial techniques. Clin. Microbiol. Infect. 2005, 11, 486–492. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Espinel-Ingroff, A.; Turnidge, J. The role of epidemiological cutoff values (ECVs/ECOFFs) in antifungal susceptibility testing and interpretation for uncommon yeasts and moulds. Rev. Iberoam. Micol. 2016, 33, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Lockhart, S.R.; Ghannoum, M.A.; Alexander, B.D. Establishment and use of epidemiological cutoff values for molds and yeasts by use of the clinical and laboratory standards institute M57 standard. J. Clin. Microbiol. 2017, 55, 1262–1268. [Google Scholar] [CrossRef] [PubMed]
- Espinel-Ingroff, A.; Turnidge, J.; Alastruey-Izquierdo, A.; Botterel, F.; Canton, E.; Castro, C.; Chen, Y.-C.; Chryssanthou, E.; Dannaoui, E.; Garcia-Effron, G.; et al. Method-dependent epidemiological cutoff values for detection of triazole resistance in Candida and Aspergillus Species for the sensititre YeastOne colorimetric broth and Etest agar diffusion methods. Antimicrob. Agents Chemother. 2018, 63, e01651-18. [Google Scholar] [CrossRef]
- Jenks, J.D.; Salzer, H.J.; Prattes, J.; Krause, R.; Buchheidt, D.; Hoenigl, M. Spotlight on isavuconazole in the treatment of invasive aspergillosis and mucormycosis: Design, development, and place in therapy. Drug Des. Dev. Ther. 2018, 12, 1033–1044. [Google Scholar] [CrossRef]
- Cascio, G.L.; Bazaj, A.; Trovato, L.; Sanna, S.; Andreoni, S.; Blasi, E.; Conte, M.; Fazii, P.; Oliva, E.; Lepera, V.; et al. Multicenter Italian study on “In Vitro Activities” of isavuconazole, voriconazole, amphotericin b, and caspofungin for Aspergillus species: Comparison between SensititreTM YeastOneTM and MIC test strip. Infect. Drug Resist. 2022, 15, 5839–5848. [Google Scholar] [CrossRef]
- Lamoth, F.; Alexander, B.D. Comparing Etest and broth microdilution for antifungal susceptibility testing of the most-relevant pathogenic molds. J. Clin. Microbiol. 2015, 53, 3176–3181. [Google Scholar] [CrossRef]
- Oakley, K.L.; Moore, C.B.; Denning, D.W. Comparison of in vitro activity of liposomal nystatin against Aspergillus species with those of nystatin, amphotericin B (AB) deoxycholate, AB colloidal dispersion, liposomal AB, AB lipid complex, and itraconazole. Antimicrob. Agents Chemother. 1999, 43, 1264–1266. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Swenson, C.E.; Perkins, W.R.; Roberts, P.; Ahmad, I.; Stevens, R.; Stevens, D.A.; Janoff, A.S. In vitro and in vivo antifungal activity of amphotericin B lipid complex: Are phospholipases important? Antimicrob. Agents Chemother. 1998, 42, 767–771. [Google Scholar] [CrossRef] [PubMed]
- Salsé, M.; Gangneux, J.-P.; Cassaing, S.; Delhaes, L.; Fekkar, A.; Dupont, D.; Botterel, F.; Costa, D.; Bourgeois, N.; Bouteille, B.; et al. Multicentre study to determine the Etest epidemiological cut-off values of antifungal drugs in Candida spp. and Aspergillus fumigatus species complex. Clin. Microbiol. Infect. 2019, 25, 1546–1552. [Google Scholar] [CrossRef] [PubMed]
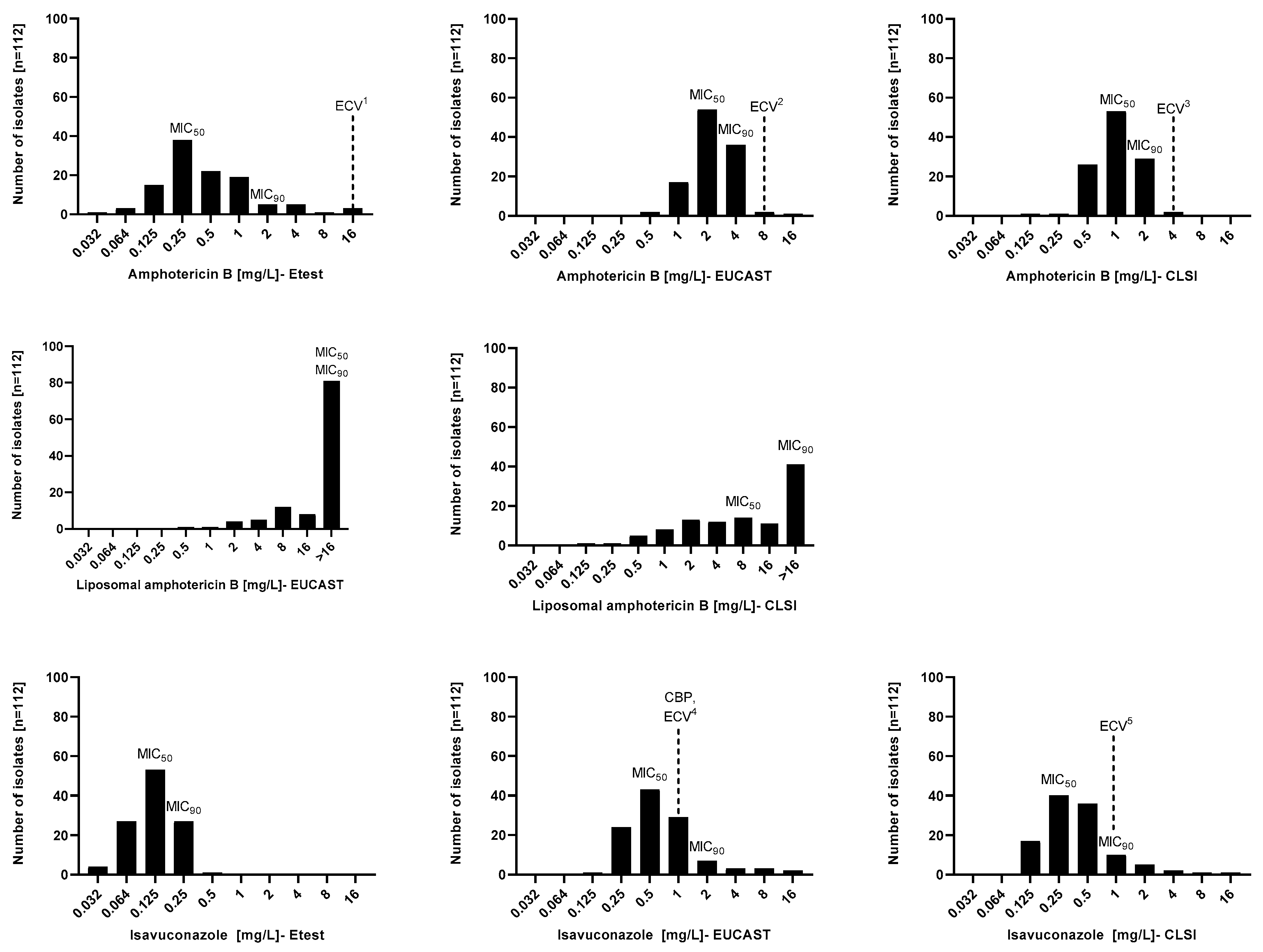
| Species (Number of Isolates) | Method | MIC [mg/L] | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AmB | L-AmB | ISA | |||||||||||
| MIC50 | MIC90 | Range | GM | MIC50 | MIC90 | Range | GM | MIC50 | MIC90 | Range | GM | ||
| Aspergillus terreus (n = 50) | Etest | 0.25 | 1 | 0.032–2 | 0.27 | - | - | - | - | 0.125 | 0.25 | 0.064–0.25 | 0.14 |
| EUCAST | 2 | 4 | 0.5–4 | 1.84 | >16 | >16 | 0.5–>16 | 6.28 | 0.5 | 1 | 0.25–8 | 0.62 | |
| CLSI | 1 | 2 | 0.125–2 | 0.91 | 4 | >16 | 0.125–>16 | 2.96 | 0.25 | 1 | 0.125–4 | 0.36 | |
| Aspergillus hortai (n = 11) | Etest | 0.5 | 0.5 | 0.064–1 | 0.37 | - | - | - | - | 0.125 | 0.25 | 0.064–0.25 | 0.133 |
| EUCAST | 4 | 4 | 2–4 | 3.31 | >16 | >16 | 8–>16 | 8 | 0.5 | 1 | 0.25–2 | 0.53 | |
| CLSI | 2 | 2 | 0.5–2 | 1.29 | 16 | >16 | 2–>16 | 5.66 | 0.25 | 0.5 | 0.125–0.5 | 0.28 | |
| Aspergillus citrinoterreus (n = 34) | Etest | 0.5 | 4 | 0.125–16 | 0.61 | - | - | - | - | 0.064 | 0.25 | 0.032–0.25 | 0.09 |
| EUCAST | 2 | 4 | 1–16 | 2.89 | >16 | >16 | 2–>16 | 7.25 | 0.5 | 1 | 0.125–4 | 0.48 | |
| CLSI | 1 | 2 | 0.5–4 | 1.28 | >16 | >16 | 0.25–>16 | 2.59 | 0.25 | 0.5 | 0.125–2 | 0.32 | |
| Aspergillus alabamensis (n = 9) | Etest | 2 | 12 | 0.5–16 | 2.64 | - | - | - | . | 0.064 | 0.125 | 0.064–0.125 | 0.09 |
| EUCAST | 2 | 4 | 2–4 | 2.72 | >16 | >16 | 2–>16 | 2 | 1 | 2 | 0.5–2 | 0.93 | |
| CLSI | 1 | 1 | 0.5–2 | 1 | 8 | >16 | 1–>16 | 6.56 | 0.5 | 1 | 0.25–1 | 0.43 | |
| Aspergillus iranicus (n = 5) | Etest | 0.25 | 1 | 0.25–2 | 0.66 | - | - | - | - | 0.25 | 0.25 | 0.125–0.5 | 0.25 |
| EUCAST | 1 | 2 | 1–4 | 1.52 | >16 | >16 | 2–>16 | 2 | 4 | 16 | 2–16 | 6.96 | |
| CLSI | 0.5 | 0.5 | 0.5–1 | 0.57 | 8 | >16 | 1–>16 | 4 | 2 | 8 | 0.25–16 | 3.03 | |
| Aspergillus niveus (n = 2) | Etest | - | - | 0.125–0.5 | 0.25 | - | - | - | - | - | - | 0.125–0.25 | 0.18 |
| EUCAST | - | - | 1–2 | 1.41 | - | - | 8–>16 | 8 | - | - | 2–4 | 2.83 | |
| CLSI | - | - | 0.25–0.5 | 0.35 | - | - | 4–8 | 5.66 | - | - | 2 | 2 | |
| Aspergillus neoafricanus (n = 1) | Etest | - | - | 0.25 | - | - | - | - | - | - | - | 0.25 | - |
| EUCAST | - | - | 4 | - | - | - | >16 | - | - | - | 1 | - | |
| CLSI | - | - | 1 | - | - | - | >16 | - | - | - | 1 | - | |
| All isolates (n = 112) | Etest | 0.25 | 2 | 0.032–16 | 0.45 | - | - | - | - | 0.125 | 0.25 | 0.032–0.5 | 0.12 |
| EUCAST | 2 | 4 | 0.5–16 | 2.29 | >16 | >16 | 0.5–>16 | 6.12 | 0.5 | 2 | 0.125–16 | 0.67 | |
| CLSI | 1 | 2 | 0.125–4 | 1.01 | 8 | >16 | 0.125–>16 | 3.39 | 0.25 | 1 | 0.125–16 | 0.39 | |
| Species | Antifungal Agents | Essential Agreement | ||
|---|---|---|---|---|
| Aspergillus terreus s.s. (n = 50) | Etest vs. CLSI | Etest vs. EUCAST | CLSI vs. EUCAST | |
| AmB | 74.0% | 44.0% | 100% | |
| L-AmB | - | - | 54.0% | |
| ISA | 62.0% | 58.0% | 96.0% | |
| Aspergillus terreus non-s.s. (n = 62) | AmB | 82.2% | 54.8% | 96.7% |
| L-AmB | - | - | 79.0% | |
| ISA | 67.7% | 54.1% | 95.1% | |
| All species (n = 112) | AmB | 79.6% | 49.5% | 98.2% |
| L-AmB | - | - | 71.4% | |
| ISA | 77.6% | 46.4% | 95.5% | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vahedi-Shahandashti, R.; Hahn, L.; Houbraken, J.; Lass-Flörl, C. Aspergillus Section Terrei and Antifungals: From Broth to Agar-Based Susceptibility Testing Methods. J. Fungi 2023, 9, 306. https://doi.org/10.3390/jof9030306
Vahedi-Shahandashti R, Hahn L, Houbraken J, Lass-Flörl C. Aspergillus Section Terrei and Antifungals: From Broth to Agar-Based Susceptibility Testing Methods. Journal of Fungi. 2023; 9(3):306. https://doi.org/10.3390/jof9030306
Chicago/Turabian StyleVahedi-Shahandashti, Roya, Lisa Hahn, Jos Houbraken, and Cornelia Lass-Flörl. 2023. "Aspergillus Section Terrei and Antifungals: From Broth to Agar-Based Susceptibility Testing Methods" Journal of Fungi 9, no. 3: 306. https://doi.org/10.3390/jof9030306
APA StyleVahedi-Shahandashti, R., Hahn, L., Houbraken, J., & Lass-Flörl, C. (2023). Aspergillus Section Terrei and Antifungals: From Broth to Agar-Based Susceptibility Testing Methods. Journal of Fungi, 9(3), 306. https://doi.org/10.3390/jof9030306







