Evaluation of Antifungal Selective Toxicity Using Candida glabrata ERG25 and Human SC4MOL Knock-In Strains
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains, Plasmids, and Media
2.2. Construction of Sc(hERG25) and Sc(CgERG25)
2.3. Liquid Growth Assays and Determination of IC50
2.4. Spotting Assay
2.5. Determination of Minimum Inhibitory Concentration (MIC)
2.6. Evaluation of Cytotoxicity
2.7. Statistical Analyses
3. Results
3.1. Complementarity of hERG25 and CgERG25 in S. cerevisiae
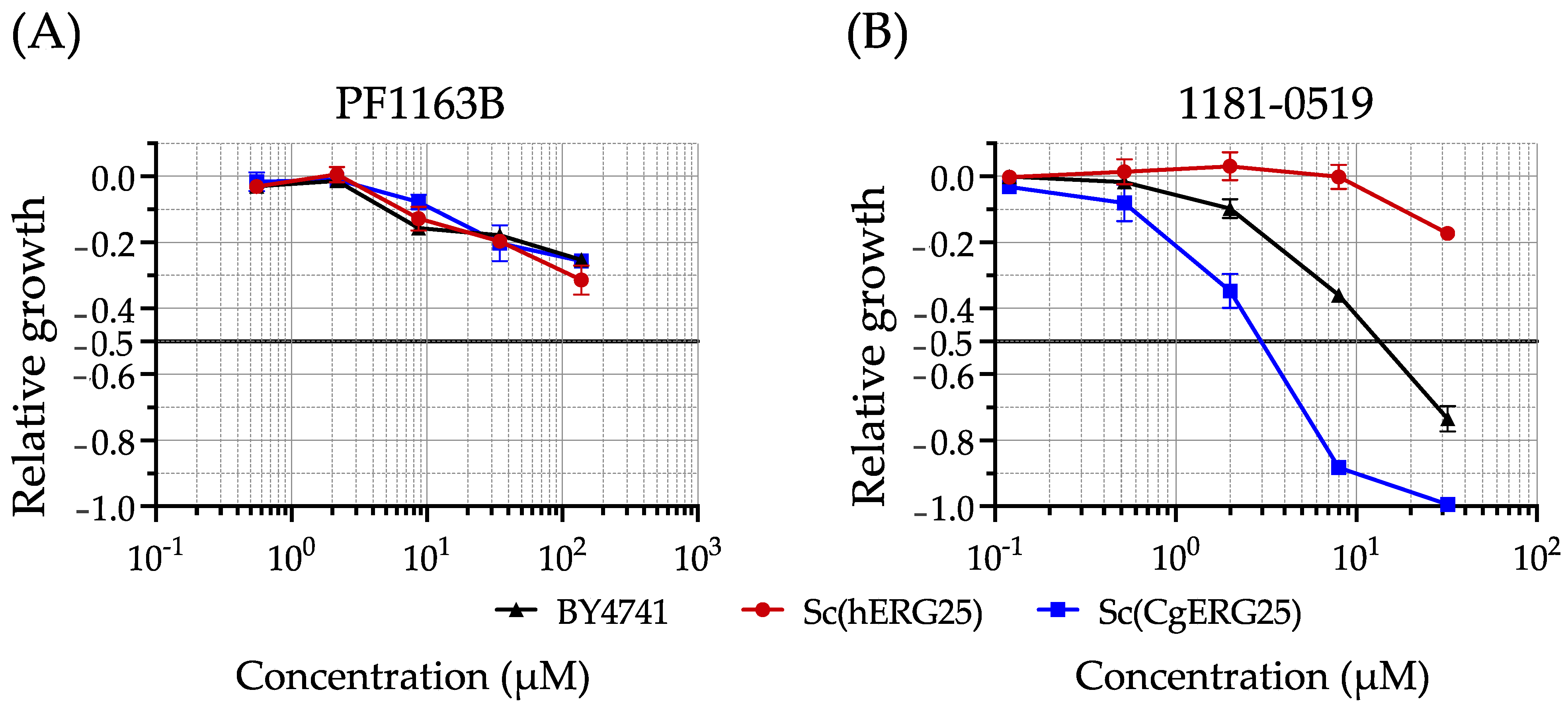
3.2. Drug Susceptibility of Knock-In Strains with Liquid Growth Assays
3.3. Evaluation of 1181-0519 Spectrum
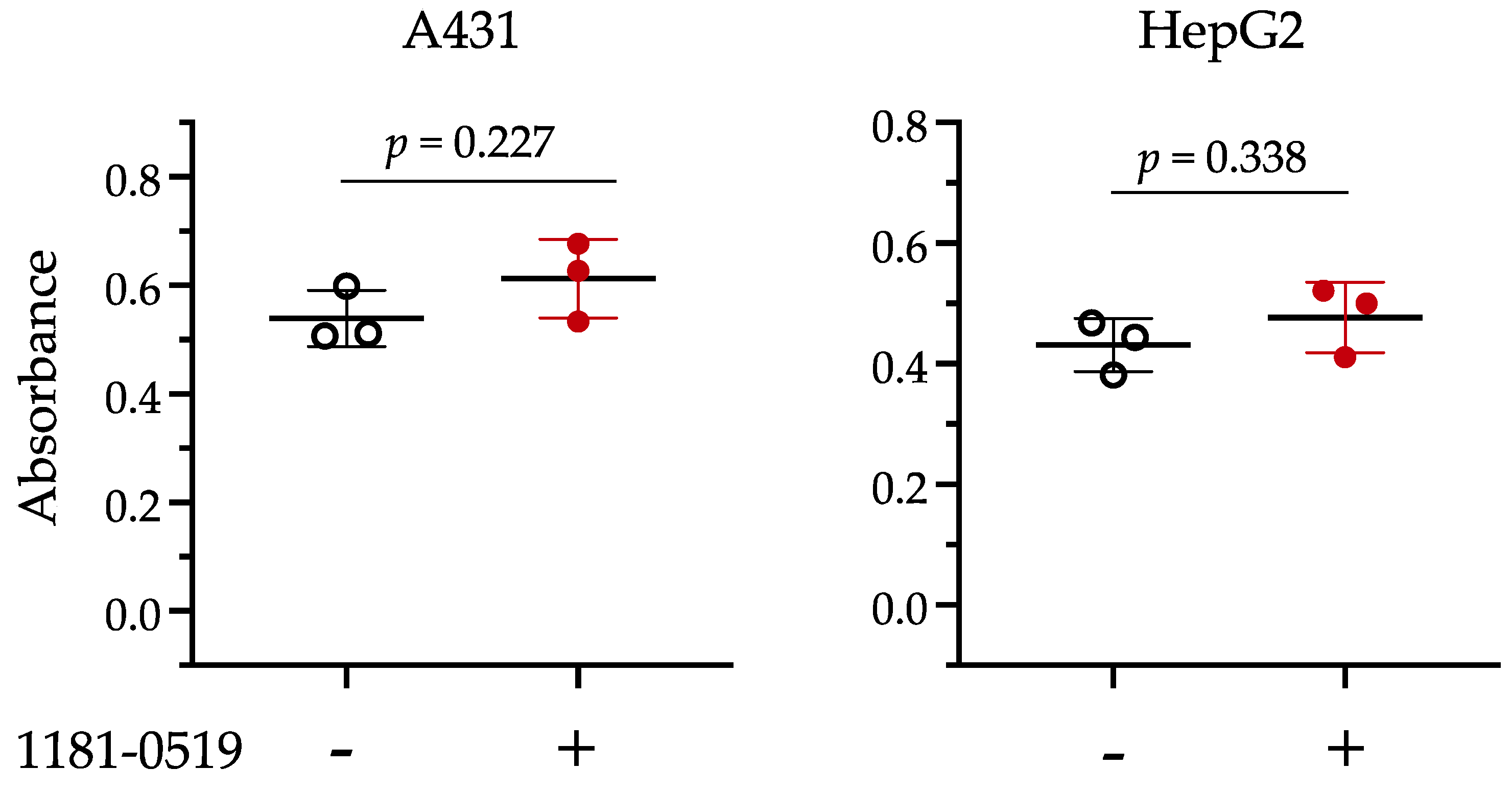
3.4. Cytotoxicity of 1181-0519
3.5. Homology Analysis of Erg25p
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rehman, S. A Parallel and Silent Emerging Pandemic: Antimicrobial Resistance (AMR) amid COVID-19 Pandemic. J. Infect. Public Health 2023, 16, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.-C.; Chen, S.-Y.; Ko, W.-C.; Hsueh, P.-R. Increased Antimicrobial Resistance during the COVID-19 Pandemic. Int. J. Antimicrob. Agents 2021, 57, 106324. [Google Scholar] [CrossRef]
- Kariyawasam, R.M.; Julien, D.A.; Jelinski, D.C.; Larose, S.L.; Rennert-May, E.; Conly, J.M.; Dingle, T.C.; Chen, J.Z.; Tyrrell, G.J.; Ronksley, P.E.; et al. Antimicrobial Resistance (AMR) in COVID-19 Patients: A Systematic Review and Meta-Analysis (November 2019–June 2021). Antimicrob. Resist. Infect. Control 2022, 11, 45. [Google Scholar] [CrossRef] [PubMed]
- Giacobbe, D.R.; Maraolo, A.E.; Simeon, V.; Magnè, F.; Pace, M.C.; Gentile, I.; Chiodini, P.; Viscoli, C.; Sanguinetti, M.; Mikulska, M.; et al. Changes in the Relative Prevalence of Candidaemia Due to Non-albicans Candida Species in Adult In-patients: A Systematic Review, Meta-analysis and Meta-regression. Mycoses 2020, 63, 334–342. [Google Scholar] [CrossRef] [PubMed]
- Campoy, S.; Adrio, J.L. Antifungals. Biochem. Pharmacol. 2017, 133, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.N.; De Mello, T.P.; De Souza Ramos, L.; Branquinha, M.H.; Dos Santos, A.L.S. New and Promising Chemotherapeutics for Emerging Infections Involving Drug-Resistant Non-Albicans Candida Species. Curr. Top. Med. Chem. 2019, 19, 2527–2553. [Google Scholar] [CrossRef]
- Houšť, J.; Spížek, J.; Havlíček, V. Antifungal Drugs. Metabolites 2020, 10, 106. [Google Scholar] [CrossRef] [PubMed]
- Gray, K.C.; Palacios, D.S.; Dailey, I.; Endo, M.M.; Uno, B.E.; Wilcock, B.C.; Burke, M.D. Amphotericin Primarily Kills Yeast by Simply Binding Ergosterol. Proc. Natl. Acad. Sci. USA 2012, 109, 2234–2239. [Google Scholar] [CrossRef] [PubMed]
- Saravolatz, L.D.; Ostrosky-Zeichner, L.; Marr, K.A.; Rex, J.H.; Cohen, S.H. Amphotericin B: Time for a New “Gold Standard”. Clin. Infect. Dis. 2003, 37, 415–425. [Google Scholar] [CrossRef]
- Singh, A.; Singh, K.; Sharma, A.; Kaur, K.; Chadha, R.; Bedi, P.M.S. Recent Advances in Antifungal Drug Development Targeting Lanosterol 14α-demethylase ( CYP51 ): A Comprehensive Review with Structural and Molecular Insights. Chem. Biol. Drug Des. 2023, 102, 606–639. [Google Scholar] [CrossRef]
- Bossche, H.V.; Marichal, P.; Gorrens, J.; Bellens, D.; Moereels, H.; Janssen, P.A.J. Mutation in Cytochrome P-450-Dependent 14α-Demethylase Results in Decreased Affinity for Azole Antifungals. Biochem. Soc. Trans. 1990, 18, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Hitchcock, C.A. Cytochrome P-450-Dependent 14α-Sterol Demethylase of Candida albicans and Its Interaction with Azole Antifungals. Biochem. Soc. Trans. 1991, 19, 782–787. [Google Scholar] [CrossRef] [PubMed]
- Hitchcock, C.A.; Adams, D.J. Interaction of Azole Antifungal Antibiotics with Cytochrome P-450-Dependent 14a-Sterol Demethylase Purified from Candida albicans. Biochem. J. 1990, 266, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Jamzivar, F.; Shams-Ghahfarokhi, M.; Khoramizadeh, M.; Yousefi, N.; Gholami-Shabani, M. Unraveling the Importance of Molecules of Natural Origin in Antifungal Drug Development through Targeting Ergosterol Biosynthesis Pathway. Iran. J. Microbiol. 2020, 11, 448. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Esquivel, B.D.; White, T.C. Overexpression or Deletion of Ergosterol Biosynthesis Genes Alters Doubling Time, Response to Stress Agents, and Drug Susceptibility in Saccharomyces cerevisiae. mBio 2018, 9, e01291-18. [Google Scholar] [CrossRef]
- Okamoto, M.; Takahashi-Nakaguchi, A.; Tejima, K.; Sasamoto, K.; Yamaguchi, M.; Aoyama, T.; Nagi, M.; Tanabe, K.; Miyazaki, Y.; Nakayama, H.; et al. Erg25 Controls Host-Cholesterol Uptake Mediated by Aus1p-Associated Sterol-Rich Membrane Domains in Candida glabrata. Front. Cell Dev. Biol. 2022, 10, 820675. [Google Scholar] [CrossRef]
- Sanglard, D.; Kuchler, K.; Ischer, F.; Pagani, J.L.; Monod, M.; Bille, J. Mechanisms of Resistance to Azole Antifungal Agents in Candida albicans Isolates from AIDS Patients Involve Specific Multidrug Transporters. Antimicrob. Agents Chemother. 1995, 39, 2378–2386. [Google Scholar] [CrossRef]
- Sanglard, D.; Ischer, F.; Monod, M.; Bille, J. Cloning of Candida albicans Genes Conferring Resistance to Azole Antifungal Agents: Characterization of CDR2, a New Multidrug ABC Transporter Gene. Microbiology 1997, 143, 405–416. [Google Scholar] [CrossRef]
- Miyazaki, H.; Miyazaki, Y.; Geber, A.; Parkinson, T.; Hitchcock, C.; Falconer, D.J.; Ward, D.J.; Marsden, K.; Bennett, J.E. Fluconazole Resistance Associated with Drug Efflux and Increased Transcription of a Drug Transporter Gene, PDH1, in Candida glabrata. Antimicrob. Agents Chemother. 1998, 42, 1695–1701. [Google Scholar] [CrossRef]
- Redding, S.W.; Kirkpatrick, W.R.; Saville, S.; Coco, B.J.; White, W.; Fothergill, A.; Rinaldi, M.; Eng, T.; Patterson, T.F.; Lopez-Ribot, J. Multiple Patterns of Resistance to Fluconazole in Candida glabrata Isolates from a Patient with Oropharyngeal Candidiasis Receiving Head and Neck Radiation. J. Clin. Microbiol. 2003, 41, 619–622. [Google Scholar] [CrossRef]
- Bennett, J.E.; Izumikawa, K.; Marr, K.A. Mechanism of Increased Fluconazole Resistance in Candida glabrata during Prophylaxis. Antimicrob. Agents Chemother. 2004, 48, 1773–1777. [Google Scholar] [CrossRef] [PubMed]
- Vermitsky, J.-P.; Edlind, T.D. Azole Resistance in Candida glabrata: Coordinate Upregulation of Multidrug Transporters and Evidence for a Pdr1-Like Transcription Factor. Antimicrob. Agents Chemother. 2004, 48, 3773–3781. [Google Scholar] [CrossRef]
- Torelli, R.; Posteraro, B.; Ferrari, S.; La Sorda, M.; Fadda, G.; Sanglard, D.; Sanguinetti, M. The ATP-Binding Cassette Transporter–Encoding Gene CgSNQ2 Is Contributing to the CgPDR1-Dependent Azole Resistance of Candida glabrata. Mol. Microbiol. 2008, 68, 186–201. [Google Scholar] [CrossRef] [PubMed]
- Sanguinetti, M.; Posteraro, B.; Fiori, B.; Ranno, S.; Torelli, R.; Fadda, G. Mechanisms of Azole Resistance in Clinical Isolates of Candida glabrata Collected during a Hospital Survey of Antifungal Resistance. Antimicrob. Agents Chemother. 2005, 49, 668–679. [Google Scholar] [CrossRef] [PubMed]
- Sanglard, D.; Ischer, F.; Bille, J. Role of ATP-Binding-Cassette Transporter Genes in High-Frequency Acquisition of Resistance to Azole Antifungals in Candida glabrata. Antimicrob. Agents Chemother. 2001, 45, 1174–1183. [Google Scholar] [CrossRef] [PubMed]
- Badrane, H.; Cheng, S.; Dupont, C.; Hao, B.; Driscoll, E.; Morder, K.; Liu, G.; Newbrough, A.; Fleres, G.; Kaul, D.; et al. Genotypic Diversity and Unrecognized Antifungal Resistance among Populations of Candida glabrata from Positive Blood Cultures. Nat. Commun. 2023, 14, 5918. [Google Scholar] [CrossRef]
- Okamoto, M.; Nakano, K.; Takahashi-Nakaguchi, A.; Sasamoto, K.; Yamaguchi, M.; Teixeira, M.C.; Chibana, H. In Candida glabrata, ERMES Component GEM1 Controls Mitochondrial Morphology, mtROS, and Drug Efflux Pump Expression, Resulting in Azole Susceptibility. J. Fungi 2023, 9, 240. [Google Scholar] [CrossRef]
- Brun, S.; Aubry, C.; Lima, O.; Filmon, R.; Bergès, T.; Chabasse, D.; Bouchara, J.-P. Relationships between Respiration and Susceptibility to Azole Antifungals in Candida glabrata. Antimicrob. Agents Chemother. 2003, 47, 847–853. [Google Scholar] [CrossRef]
- Vandeputte, P.; Tronchin, G.; Rocher, F.; Renier, G.; Bergès, T.; Chabasse, D.; Bouchara, J.-P. Hypersusceptibility to Azole Antifungals in a Clinical Isolate of Candida glabrata with Reduced Aerobic Growth. Antimicrob. Agents Chemother. 2009, 53, 3034–3041. [Google Scholar] [CrossRef]
- Peng, Y.; Dong, D.; Jiang, C.; Yu, B.; Wang, X.; Ji, Y. Relationship between Respiration Deficiency and Azole Resistance in Clinical Candida glabrata. FEMS Yeast Res. 2012, 12, 719–727. [Google Scholar] [CrossRef]
- Bard, M.; Sturm, A.M.; Pierson, C.A.; Brown, S.; Rogers, K.M.; Nabinger, S.; Eckstein, J.; Barbuch, R.; Lees, N.D.; Howell, S.A.; et al. Sterol Uptake in Candida glabrata: Rescue of Sterol Auxotrophic Strains. Diagn. Microbiol. Infect. Dis. 2005, 52, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Hazen, K.C.; Stei, J.; Darracott, C.; Breathnach, A.; May, J.; Howell, S.A. Isolation of Cholesterol-Dependent Candida glabrata from Clinical Specimens. Diagn. Microbiol. Infect. Dis. 2005, 52, 35–37. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.; Ahmad, S.; Joseph, L.; Al-Obaid, K. Isolation of Cholesterol-Dependent, Multidrug-Resistant Candida glabrata strains from Blood Cultures of a Candidemia Patient in Kuwait. BMC Infect. Dis. 2014, 14, 188. [Google Scholar] [CrossRef] [PubMed]
- Nagi, M.; Tanabe, K.; Tanaka, K.; Ueno, K.; Nakayama, H.; Ishikawa, J.; Abe, M.; Yamagoe, S.; Umeyama, T.; Nakamura, S.; et al. Exhibition of Antifungal Resistance by Sterol-Auxotrophic Strains of Candida glabrata with Intact Virulence. JAC-Antimicrob. Resist. 2022, 4, dlac018. [Google Scholar] [CrossRef] [PubMed]
- Langer, G. Implementation and Use of State-of-the-Art, Cell-Based In Vitro Assays. In New Approaches to Drug Discovery; Nielsch, U., Fuhrmann, U., Jaroch, S., Eds.; Handbook of Experimental Pharmacology; Springer International Publishing: Cham, Switzerland, 2015; Volume 232, pp. 171–190. ISBN 978-3-319-28912-0. [Google Scholar]
- Mo, C.; Valachovic, M.; Randall, S.K.; Nickels, J.T.; Bard, M. Protein–Protein Interactions among C-4 Demethylation Enzymes Involved in Yeast Sterol Biosynthesis. Proc. Natl. Acad. Sci. USA 2002, 99, 9739–9744. [Google Scholar] [CrossRef]
- Mo, C.; Bard, M. Erg28p is a key protein in the yeast sterol biosynthetic enzyme complex. J. Lipid Res. 2005, 46, 1991–1998. [Google Scholar] [CrossRef]
- Kachroo, A.H.; Laurent, J.M.; Yellman, C.M.; Meyer, A.G.; Wilke, C.O.; Marcotte, E.M. Systematic Humanization of Yeast Genes Reveals Conserved Functions and Genetic Modularity. Science 2015, 348, 921–925. [Google Scholar] [CrossRef]
- Kuchta, T.; Barková, K.; Kubinec, R. Ergosterol Depletion and 4-Methyl Sterols Accumulation in the Yeast Saccharomyces cerevisiae Treated with an Antifungal, 6-Amino-2-n-Pentylthiobenzothiazole. Biochem. Biophys. Res. Commun. 1992, 189, 85–91. [Google Scholar] [CrossRef]
- Nose, H.; Seki, A.; Yaguchi, T.; Hosoya, A.; Sasaki, T.; Hoshiko, S.; Shomura, T. PF1163A and B, New Antifungal Antibiotics Produced by Penicillium Sp. I. Taxonomy of Producing Strain, Fermentation, Isolation and Biological Activities. J. Antibiot. 2000, 53, 33–37. [Google Scholar] [CrossRef][Green Version]
- Nose, H.; Fushimi, H.; Seki, A.; Sasaki, T.; Watabe, H.; Hoshiko, S. PF1163A, a Novel Antifungal Agent, Inhibit Ergosterol Biosynthesis at C-4 Sterol Methyl Oxidase. J. Antibiot. 2002, 55, 969–974. [Google Scholar] [CrossRef]
- Kim, S.H.; Steere, L.; Zhang, Y.-K.; McGregor, C.; Hahne, C.; Zhou, Y.; Liu, C.; Cai, Y.; Zhou, H.; Chen, X.; et al. Inhibiting C-4 Methyl Sterol Oxidase with Novel Diazaborines to Target Fungal Plant Pathogens. ACS Chem. Biol. 2022, 17, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.Y.; St. Onge, R.P.; Proctor, M.J.; Wallace, I.M.; Nile, A.H.; Spagnuolo, P.A.; Jitkova, Y.; Gronda, M.; Wu, Y.; Kim, M.K.; et al. Mapping the Cellular Response to Small Molecules Using Chemogenomic Fitness Signatures. Science 2014, 344, 208–211. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.D.; Suresh, S.; Schlecht, U.; Wu, M.; Wagih, O.; Peltz, G.; Davis, R.W.; Steinmetz, L.M.; Parts, L.; St. Onge, R.P. Quantitative CRISPR Interference Screens in Yeast Identify Chemical-Genetic Interactions and New Rules for Guide RNA Design. Genome Biol. 2016, 17, 45. [Google Scholar] [CrossRef] [PubMed]
- Abe, H. In Vitro Oligosaccharide Synthesis Using Intact Yeast Cells That Display Glycosyltransferases at the Cell Surface through Cell Wall-Anchored Protein Pir. Glycobiology 2003, 13, 87–95. [Google Scholar] [CrossRef]
- Nozaki, S.; Niki, H. Exonuclease III (XthA) Enforces In Vivo DNA Cloning of Escherichia coli to Create Cohesive Ends. J. Bacteriol. 2019, 201, 10–1128. [Google Scholar] [CrossRef]
- Ueno, K.; Uno, J.; Nakayama, H.; Sasamoto, K.; Mikami, Y.; Chibana, H. Development of a Highly Efficient Gene Targeting System Induced by Transient Repression of YKU80 Expression in Candida glabrata. Eukaryot. Cell 2007, 6, 1239–1247. [Google Scholar] [CrossRef]
- Schlecht, U.; Miranda, M.; Suresh, S.; Davis, R.W.; St. Onge, R.P. Multiplex Assay for Condition-Dependent Changes in Protein–Protein Interactions. Proc. Natl. Acad. Sci. USA 2012, 109, 9213–9218. [Google Scholar] [CrossRef]
- Shanklin, J.; Whittle, E.; Fox, B.G. Eight Histidine Residues Are Catalytically Essential in a Membrane-Associated Iron Enzyme, Stearoyl-CoA Desaturase, and Are Conserved in Alkane Hydroxylase and Xylene Monooxygenase. Biochemistry 1994, 33, 12787–12794. [Google Scholar] [CrossRef]
- Li, L.; Kaplan, J. Characterization of Yeast Methyl Sterol Oxidase (ERG25) and Identification of a Human Homologue. J. Biol. Chem. 1996, 271, 16927–16933. [Google Scholar] [CrossRef] [PubMed]
- Pierson, C.A.; Eckstein, J.; Barbuch, R.; Bard, M. Ergosterol Gene Expression in Wild-Type and Ergosterol-Deficient Mutants of Candida albicans. Med. Mycol. 2004, 42, 385–389. [Google Scholar] [CrossRef][Green Version]
- Sukhanova, A.; Gorin, A.; Serebriiskii, I.G.; Gabitova, L.; Zheng, H.; Restifo, D.; Egleston, B.L.; Cunningham, D.; Bagnyukova, T.; Liu, H.; et al. Targeting C4-Demethylating Genes in the Cholesterol Pathway Sensitizes Cancer Cells to EGF Receptor Inhibitors via Increased EGF Receptor Degradation. Cancer Discov. 2013, 3, 96–111. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Smith, L.D.; Chang, R.; Li, X.; Vockley, J. The Role of Sterol-C4-Methyl Oxidase in Epidermal Biology. Biochim. Biophys. Acta BBA-Mol. Cell Biol. Lipids 2014, 1841, 331–335. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Kratz, L.E.; Michel, J.J.; Vallejo, A.N.; Ferris, L.; Kelley, R.I.; Hoover, J.J.; Jukic, D.; Gibson, K.M.; Wolfe, L.A.; et al. Mutations in the Human SC4MOL Gene Encoding a Methyl Sterol Oxidase Cause Psoriasiform Dermatitis, Microcephaly, and Developmental Delay. J. Clin. Investig. 2011, 121, 976–984. [Google Scholar] [CrossRef] [PubMed]
- Frisso, G.; Gelzo, M.; Procopio, E.; Sica, C.; Lenza, M.P.; Dello Russo, A.; Donati, M.A.; Salvatore, F.; Corso, G. A Rare Case of Sterol-C4-Methyl Oxidase Deficiency in a Young Italian Male: Biochemical and Molecular Characterization. Mol. Genet. Metab. 2017, 121, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Kalay Yildizhan, I.; Gökpınar İli, E.; Onoufriadis, A.; Kocyigit, P.; Kesidou, E.; Simpson, M.A.; McGrath, J.A.; Kutlay, N.Y.; Kundakci, N. New Homozygous Missense MSMO1 Mutation in Two Siblings with SC4MOL Deficiency Presenting with Psoriasiform Dermatitis. Cytogenet. Genome Res. 2020, 160, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Byskov, A.G.; Andersen, C.Y.; Nordholm, L.; Thogersen, H.; Guoliang, X.; Wassmann, O.; Andersen, J.V.; Guddal, E.; Roed, T. Chemical Structure of Sterols That Activate Oocyte Meiosis. Nature 1995, 374, 559–562. [Google Scholar] [CrossRef]
- Baltsen, M.; Byskov, A.G. Quantitation of Meiosis Activating Sterols in Human Follicular Fluid Using HPLC and Photodiode Array Detection. Biomed. Chromatogr. 1999, 13, 382–388. [Google Scholar] [CrossRef]
- Tsafriri, A.; Cao, X.; Vaknin, K.M.; Popliker, M. Is Meiosis Activating Sterol (MAS) an Obligatory Mediator of Meiotic Resumption in Mammals. Mol. Cell. Endocrinol. 2002, 187, 197–204. [Google Scholar] [CrossRef]

| Strain | Parental Strain | Genotype and (Plasmid) | Resource |
|---|---|---|---|
| Sc(erg25Δ/ERG25) | BY4743 | MATa/α his3Δ1/his3Δ1 leu2Δ0/leu2Δ0 LYS2/lys2Δ0 met15Δ0/MET15 ura3Δ0/ura3Δ0 erg25Δ::KanMX/ERG25 | In this study |
| BY4741 | S. cerevisiae | MATa his3Δ1 leu2Δ0 met15Δ0 ura3Δ0 | Euroscarf |
| Sc(hERG25) | Sc(erg25Δ/ERG25) | erg25Δ::KanMX(YEp352-GAPII-hERG25) | In this study |
| Sc(CgERG25) | Sc(hERG25) | erg25Δ::KanMX(YEp351-GAPII-CgERG25) | In this study |
| C. albicans SC5314 | WT | NBRP, Chiba, Japan | |
| C. glabrata CBS 138 | WT | NBRP, Chiba, Japan | |
| C. auris CBS 10913 | WT | NBRP, Chiba, Japan | |
| C. tropcalis CBS 94 | WT | NBRP, Chiba, Japan | |
| C. parapsilosis CBS 604 | WT | NBRP, Chiba, Japan | |
| C. krusei CBS 573 | WT | NBRP, Chiba, Japan |
| Strain | IC50 (µM) | |
|---|---|---|
| PF1163B | 1181-0519 | |
| BY4741 | >138 | 13 |
| Sc(hERG25) | >138 | >32 |
| Sc(CgERG25) | >138 | 3 |
| Strain | MIC (µM) |
|---|---|
| C. albicans SC5314 | 2 |
| C. glabrata CBS 138 | 2 |
| C. auris CBS 10913 | 1 |
| C. tropicalis CBS 94 | 16 |
| C. parapsilosis CBS 604 | 1 |
| C. krusei CBS 573 | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakano, K.; Okamoto, M.; Takahashi-Nakaguchi, A.; Sasamoto, K.; Yamaguchi, M.; Chibana, H. Evaluation of Antifungal Selective Toxicity Using Candida glabrata ERG25 and Human SC4MOL Knock-In Strains. J. Fungi 2023, 9, 1035. https://doi.org/10.3390/jof9101035
Nakano K, Okamoto M, Takahashi-Nakaguchi A, Sasamoto K, Yamaguchi M, Chibana H. Evaluation of Antifungal Selective Toxicity Using Candida glabrata ERG25 and Human SC4MOL Knock-In Strains. Journal of Fungi. 2023; 9(10):1035. https://doi.org/10.3390/jof9101035
Chicago/Turabian StyleNakano, Keiko, Michiyo Okamoto, Azusa Takahashi-Nakaguchi, Kaname Sasamoto, Masashi Yamaguchi, and Hiroji Chibana. 2023. "Evaluation of Antifungal Selective Toxicity Using Candida glabrata ERG25 and Human SC4MOL Knock-In Strains" Journal of Fungi 9, no. 10: 1035. https://doi.org/10.3390/jof9101035
APA StyleNakano, K., Okamoto, M., Takahashi-Nakaguchi, A., Sasamoto, K., Yamaguchi, M., & Chibana, H. (2023). Evaluation of Antifungal Selective Toxicity Using Candida glabrata ERG25 and Human SC4MOL Knock-In Strains. Journal of Fungi, 9(10), 1035. https://doi.org/10.3390/jof9101035






