Comparative Effectiveness of Filamentous Fungi in Biocontrol of Meloidogyne javanica and Activated Defense Mechanisms on Tomato
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Nematode Inoculum
2.3. Isolation, Phenotypic and Molecular Identification of Fungi
2.4. In Vitro Evaluation of Direct Fungal Parasitism of M. javanica
2.5. Scanning Electron Microscopy (SEM)
2.6. Pot Experiment
2.6.1. Antioxidant Enzyme Activities in Tomato Roots
Peroxidase Activity (POD)
Catalase Activity (CAT)
Ascorbate Peroxidase (APX)
Polyphenol Oxidase (PPO)
2.6.2. Total Soluble Protein Content
2.6.3. Total Phenol Content
2.6.4. Lipid Peroxidation (MDA Content)
2.6.5. Extraction and Enumeration of Nematodes and Assessment of Their Development
2.7. Statistical Analysis
3. Results
3.1. Isolation and Identification of Filamentous Fungi
3.2. In Vitro Experiments
3.3. Parasitism Observation
3.4. Plant Growth Parameters
3.5. Antioxidant Enzymes Activity
3.6. Phenols, MDA, and Protein Content in Tomato Roots after Treatment
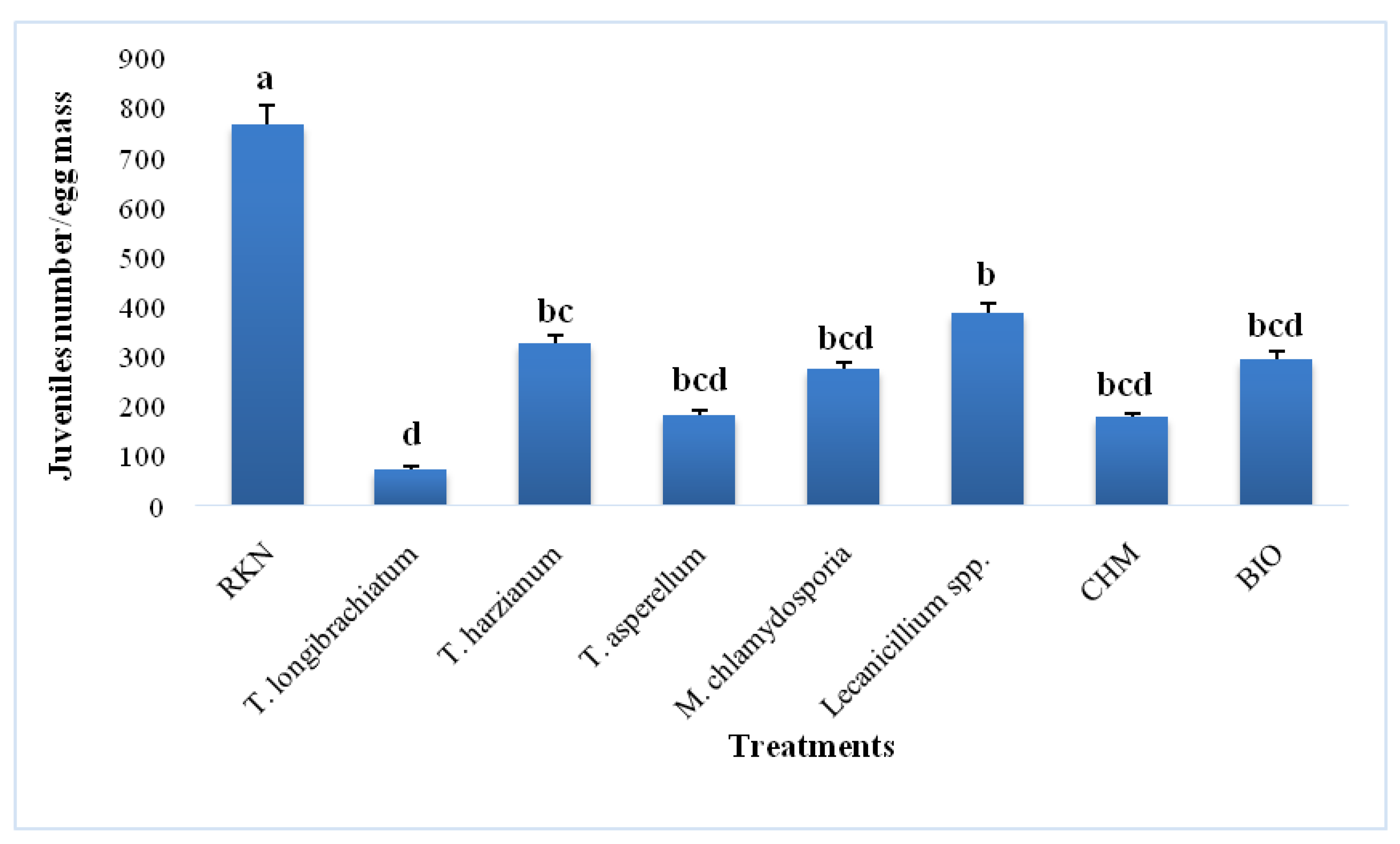
3.7. Nematode Reproduction
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moens, M.; Perry, R.N.; Starr, J.L. Meloidogyne species: A diverse group of novel and important plant parasites. In Root-Knot Nemadodes; Perry, R.N., Moens, M., Starr, J.L., Eds.; CABI Publishing: Wallingford, UK, 2009; pp. 1–17. [Google Scholar]
- Nicol, J.M.; Turner, S.J.; Coyne, D.L.; Nijs, L.D.; Hockland, S.; Maafi, Z.T. Current nematode threats to world agriculture. In Genomics and Molecular Genetics of Plant-Nematode Interactions; Jones, J., Gheysen, G., Fenoll, C., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 21–43. [Google Scholar]
- Jones, J.T.; Haegeman, A.; Danchin, E.G.; Gaur, H.S.; Helder, J.; Jones, M.G.; Kikuchi, T.; Manzanilla-López, R.; Palomares-Rius, J.E.; Wesemael, W.M.; et al. Top 10 plant-parasitic nematodes in molecular plant pathology. Mol. Plant Pathol. 2013, 14, 946–961. [Google Scholar] [CrossRef] [PubMed]
- Horrigue-Raouani, N. Variabilité de la Relation Hôte Parasite Dans le Cas Des Meloidogyne spp. (Nematoda: Meloidogynidae). Ph.D. Thesis, Université Tunis-El Manar, Faculté des Sciences de Tunis, Tunis, Tunisie, 2003; p. 222. [Google Scholar]
- Zasada, I.A.; Halbrendt, J.M.; Kokalis-Burelle, N.; LaMondia, J.; McKenry, M.V.; Noling, J.W. Managing nematodes without methyl bromide. Annu. Rev. Phytopathol. 2010, 48, 311–328. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.A.; Azeem, F.; Abbas, A.; Joyia, F.A.; Li, H.; Dababat, A.A. Transgenic strategies for enhancement of nematode resistance in plants. Front. Plant Sci. 2017, 8, 750. [Google Scholar] [CrossRef] [PubMed]
- Keswani, C.; Mishra, S.; Sarma, B.; Singh, S.; Singh, H. Unraveling the efficient applications of secondary metabolites of various Trichoderma spp. Appl. Microbiol. Biotechnol. 2014, 98, 533–544. [Google Scholar] [CrossRef]
- Jogaiah, S.; Kurjogi, M.; Govind, S.R.; Shetty, H.S.; Basappa, V.A.; Tran, L.S.P. Isolation and evaluation of proteolytic actinomycete isolates as novel inducers of pearl millet downy mildew disease protection. Sci. Rep. 2016, 6, 30789. [Google Scholar] [CrossRef]
- Waghunde, R.R.; Shelake, R.M.; Sabalpara, A.N. Trichoderma: A significant fungus for agriculture and environment. Afr. J. Agric. Res. 2016, 11, 1952–1965. [Google Scholar]
- Martinuz, A.; Schouten, A.; Sikora, R.A. Post-infection development of Meloidogyne incognita on tomato treated with the endophytes Fusarium oxysporum strain Fo162 and Rhizobium et li strain G12. Biocontrol 2012, 58, 95–104. [Google Scholar] [CrossRef]
- Schouteden, N.; De Waele, D.; Dirk, P.; Panis, B.; Vos, M.C. Arbuscular mycorrhizal fungi for the biocontrol of plant-parasitic nematodes: A review of the mechanisms involved. Front. Microbiol. 2015, 6, 1280. [Google Scholar] [CrossRef]
- Gupta, R.; Singh, A.; Srivastava, M.; Shanker, K.; Pandey, R. Plant-microbe interactions endorse growth by uplifting microbial community structure of Bacopa monnieri rhizosphere under nematode stress. Microbiol. Res. 2019, 218, 87–96. [Google Scholar] [CrossRef]
- Gupta, R.; Singh, A.; Ajayakumar, P.V.; Pandey, R. Microbial interference mitigates Meloidogyne incognita mediated oxidative stress and augments bacoside content in Bacopa monnieri L. Microbiol. Res. 2017, 199, 67–78. [Google Scholar] [CrossRef]
- Gupta, R.; Saikia, S.K.; Pandey, R. Bioconsortia augments antioxidant and yield in Matricaria recutita L. against Meloidogyne incognita (Kofoid and White) Chitwood Infestation. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2015, 7, 335–342. [Google Scholar] [CrossRef]
- Gupta, R.; Singh, A.; Gupta, M.M.; Pandey, R. Cumulative role of bio-inoculants on growth: Antioxidant potential and artemisin in content in Artemisia annua L. under organic field conditions. World J. Microbiol. Biotechnol. 2016, 32, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Aminuzzaman, F.M.; Xie, H.Y.; Duan, W.J.; Sun, B.D.; Liu, X.Z. Isolation of nematophagous fungi from eggs and females of Meloidogyne spp. and evaluation of their biological control potential. Biocontrol. Sci. Techol. 2013, 23, 170–182. [Google Scholar] [CrossRef]
- Hajji-Hedfi, L.; M’Hamdi-Boughalleb, N.; Horrigue-Raouani, N. Fungal diversity in rhizosphere of root-knot nematode infected tomatoes in Tunisia. Symbiosis 2019, 79, 171–181. [Google Scholar] [CrossRef]
- Haarith, D.; Bushley, K.E.; Chen, S. Fungal communities associated with Heterodera glycines and their potential in biological control: A current update. J. Nematol. 2020, 52, 1–17. [Google Scholar] [CrossRef]
- Moreno-Gavíra, A.; Huertas, V.; Diánez, F.; Sánchez-Montesinos, B.; Santos, M. Paecilomyces and its importance in the biological control of agricultural pests and diseases. Plants 2020, 9, 1746. [Google Scholar] [CrossRef]
- Sood, M.; Kapoor, D.; Kumar, V.; Sheteiwy, M.S.; Ramakrishnan, M.; Landi, M.; Araniti, F.; Sharma, A. Trichoderma: The “Secrets” of a Multitalented Biocontrol Agent. Plants 2020, 9, 762. [Google Scholar] [CrossRef]
- Hajji-Hedfi, L.; Regaieg, H.; Larayedh, A.; Chihani, N.; Horrigue-Raouani, N. Biological control of wilt disease complex on tomato crop caused by Meloidogyne javanica and Fusarium oxysporum f. sp. lycopersici by Verticillium leptobactrum. Environ. Sci. Pollut. Res. 2018, 25, 18297–18302. [Google Scholar] [CrossRef]
- Moosavi, M.R.; Zare, R.; Zamanizadeh, H.R.; Fatemy, S. Pathogenicity of Pochonia species on eggs of Meloidogyne javanica. J. Invertebr. Pathol. 2010, 104, 125–133. [Google Scholar] [CrossRef]
- Manzanilla-López, R.H.; Esteves, I.; Finetti-Sialer, M.M.; Hirsch, P.R.; Ward, E.; Devonshire, J.; Hidalgo-Díaz, L. Pochonia chlamydosporia: Advances and challenges to improve its performance as a biological control agent of sedentary endo-parasitic nematodes. J. Nematol. 2013, 45, 1–7. [Google Scholar]
- Zavala-González, E.A.; Escudero, N.; Lopez-Moya, F.; Aranda-Martinez, A.; Exposito, A.; Lopez-Llorca, L.V. Some isolates of the nematophagous fungus Pochonia chlamydosporia promote root growth and reduce flowering time in tomato. Ann. Appl. Biol. 2015, 166, 472–483. [Google Scholar] [CrossRef]
- Ross, I.; Alami, Y.; Harvey, P.; Achouak, W.; Ryder, M. Genetic diversity and biological control activity of novel species of closely related Pseudomonads isolated from wheat field soils in South Australia. Appl. Environ. Microbiol. 2000, 37, 473–491. [Google Scholar] [CrossRef] [PubMed]
- Abd-Elgawad, M.M.M.; Askary, T.H. Factors affecting success of biological agents used in controlling the plant-parasitic nematodes. Egypt. J. Biol. Pest Control. 2020, 30, 17. [Google Scholar] [CrossRef]
- Aly, A.H.; Debbab, A.; Proksch, P. Fungal endophytes: Unique plant inhabitants with great promises. Appl. Microbiol. Biotechnol. 2011, 90, 1829–1845. [Google Scholar] [CrossRef]
- Daguerre, Y.; Siegel, K.; Edel-Hermann, V.; Steinberg, C. Fungal proteins and genes associated with biocontrol mechanisms of soil-borne pathogens: A review. Fungal Biol. Rev. 2014, 28, 97–125. [Google Scholar] [CrossRef]
- Köhl, J.; Kolnaar, R.; Ravensberg, W.J. Mode of action of microbial biological control agents against plant diseases: Relevance beyond efficacy. Front. Plant. Sci. 2019, 10, 845. [Google Scholar] [CrossRef]
- Hajji, L.; Elouaer, M.A.; Regaieg, H.; Boughalleb, N. Biochemical and plant nutrient alterations induced by Meloidogyne javanica and Fusarium oxysporum f. sp. radicis lycopersici co-infection on tomato cultivars with differing level of resistance to M. javanica. Eur. J. Plant Pathol. 2017, 148, 463–472. [Google Scholar]
- Sato, K.; Kadota, Y.; Shirasu, K. Plant immune responses to parasitic nematodes. Front. Plant Sci. 2019, 10, 1165. [Google Scholar] [CrossRef]
- Pitzschke, A.; Forzani, C.; Hirt, H. Reactive oxygen species signaling in plants. Forum Rev. Antioxid. Redox Signal. 2006, 8, 9–10. [Google Scholar] [CrossRef]
- Møller, I.M.; Jensen, P.E.; Hansson, A. Oxidative modifications to cellular components in plants. Annu. Rev. Plant Biol. 2007, 58, 459–481. [Google Scholar] [CrossRef]
- Rejeb, I.B.; Pastor, V.; Mauch-Mani, B. Plant responses to simultaneous biotic and abiotic stress: Molecular mechanisms. Plants 2014, 3, 458–475. [Google Scholar] [CrossRef] [PubMed]
- Del Río, L.A. ROS and RNS in plant physiology: An overview. J. Exp. Bot. Adv. Access 2015, 66, 2827–2837. [Google Scholar] [CrossRef] [PubMed]
- Jeandroz, S.; Lamotte, O. Editorial: Plant responses to biotic and abiotic stresses: Lessons from cell signaling. Front. Plant Sci. 2017, 8, 1772. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Ullah, F.; Zhou, D.X.; Yi, M.; Zhao, Y. Mechanisms of ROS regulation of plant development and stress responses. Front. Plant Sci. 2019, 10, 800. [Google Scholar] [CrossRef]
- Al Sammarraie, O.N.; Khalid, Y.; Alsharafa, K.Y.; Muhamad, O.; Al Limoun, M.O.; Khleifat, K.M.; Al Sarayreh, S.A.; Al Shuneigat, J.M.; Kalaji, H.M. Effect of various abiotic stressors on some biochemical indices of Lepidium sativum plants. Nature Res. Sci. Rep. 2020, 10, 21131. [Google Scholar] [CrossRef]
- Dvořák, P.; Krasylenko, Y.; Zeiner, A.; Šamaj, J.; Takáč, T. Signaling toward reactive oxygen species-scavenging enzymes in plants. Front. Plant Sci. 2021, 11, 618835. [Google Scholar] [CrossRef]
- Miura, E.; Kato, Y.; Sakamoto, W. Comparative transcriptome analysis of green/white variegated sectors in Arabidopsis yellow variegated2: Responses to oxidative and other stresses in white sectors. J. Exp. Bot. 2010, 61, 2433–2445. [Google Scholar] [CrossRef]
- Samalova, M.; Meyer, A.J.; Gurr, S.J.; Fricker, M.D. Robust anti-oxidant defenses in the rice blast fungus Magnaporthe oryzae confer tolerance to the host oxidative burst. New Phytol. 2014, 201, 556–573. [Google Scholar] [CrossRef]
- Taylor, A.; Sasser, J. Biology, Identification and Control of Root-Knot Nematodes (Meloidogyne species) Raleigh, 1st ed.; Department of Plant Pathology North Carolina State University and the United States Agency for International Development: Raleih, NC, USA, 1978. [Google Scholar]
- Hussey, R.; Barker, K. Comparison of methods of collecting inocula of Meloidogyne spp., including a new technique. Plant Dis. Rep. 1973, 57, 1025–1028. [Google Scholar]
- Whitehead, A.G.; Hemming, J.R. A comparison of some quantitative methods of extracting small vermiform nematodes from soil. Ann. Appl. Biol. 1965, 55, 25–38. [Google Scholar] [CrossRef]
- Garrett, S.D. Colonization of unsterilized filter paper by cereal foot-rot fungi. Trans. Br. Mycol. Soc. 1980, 74, 259–263. [Google Scholar] [CrossRef]
- Smith, N.R.; Dawson, V.T. The bacteriostatic action of rose bengal in media used for plate counts of soil fungi. Soil Sci. 1944, 58, 467–472. [Google Scholar] [CrossRef]
- Zare, R.; Gams, W. A revision of Verticillium section Prostrata. IV. The genera Lecanicillium and Simplicillium gen. nov. Nova Hedwig. 2001, 73, 1–50. [Google Scholar] [CrossRef]
- Zare, R.; Gams, W.; Evans, H.C. A revision of Verticillium section Prostrata. V. The genus Pochonia, with notes on Rotiferophthora. Nova Hedwig. 2001, 73, 51–86. [Google Scholar] [CrossRef]
- Samuels, G.J.; Hebbar, P.K. Trichoderma: Identification and Agricultural Applications; APS Press: Eagan, MN, USA, 2015. [Google Scholar]
- Humber, R.A. Entomopathogenic Fungal Identification; USDA-ARS Plant Protection Research Unit, US Plant, Soil & Nutrition Laboratory Tower Road: Ithaca, NY, USA, 2005. [Google Scholar]
- Kirk, P.M.; Cannon, P.F.; Minter, D.W.; Stalpers, J.A. (Eds.) Ainswoth & Bisby’s Dictionary of the Fungi, 10th ed.; CABI Europe: Wallingford, UK, 2008; p. 445. ISBN 978 0 85199 826 8. [Google Scholar]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; Volume 315, p. 322. [Google Scholar]
- Glass, N.L.; Donaldson, G.C. Development of primer sets designed for use with the PCR to amplify conserved genes from filamentous ascomycetes. Appl. Environ. Microbiol. 1995, 61, 1323–1330. [Google Scholar] [CrossRef]
- Karnovsky, M.J. A formaldehyde-glutaraldehyde fixative of high osmolarity for use in electron microscopy. J. Cell Biol. 1965, 27, 137A. [Google Scholar]
- Pharand, B.; Carisse, O.; Benhamou, N. Cytological aspects of compost-mediated induced resistance against Fusarium crown and root rot in tomato. Phytopathology 2002, 92, 424–438. [Google Scholar] [CrossRef]
- Havir, E.A.; McHale, N.A. Biochemical and developmental characterization of multiple forms of catalase in tobacco leaves. Plant Physiol. 1987, 84, 450–455. [Google Scholar] [CrossRef]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar]
- Mayer, A.M.; Harel, E.; Shaul, R.B. Assay of catechol oxidase: A critical comparison of methods. Phytochemistry 1965, 5, 783–789. [Google Scholar] [CrossRef]
- Xu, C.; Xu, Y.; Huang, B. Protein extraction for two-dimensional gel electrophoresis of proteomic profiling in turfgrass. Crop Sci. 2008, 48, 1608–1614. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic–phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Hodges, D.M.; DeLong, J.M.; Forney, C.F.; Prange, R.K. Improving the thiobarbituric acid-reactive-substances assay for estimating lipid per-oxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 1999, 207, 604–611. [Google Scholar] [CrossRef]
- Hussey, R.S.; Janssen, G.J.W. Root-knot nematode: Meloidogyne species. In Plant Resistance to Parasitic Nematodes; Starr, J.L., Cook, R., Bridge, J., Eds.; CAB International: Wallingford, UK, 2002; pp. 43–70. [Google Scholar]
- Coolen, W.A. Methods for the extraction of Meloidogyne spp. and other nematodes form roots and soil. In Root-Knot Nematodes (Meloidogyne species) Systematics, Biology and Control; Lamberti, F., Taylor, C.E., Eds.; Academic Press: London, UK; New York, NY, USA, 1979; pp. 317–329. [Google Scholar]
- De Grisse, A.T. Re-Description ou Modification de Quelques Techniques Utilisées Dans L’étude des Nématodes Phyto Parasitaires; Mededelingen Rijksfaculteit der Landbouwetinschap: Gent, Belgium, 1969; pp. 351–369. [Google Scholar]
- McKenry, M.V.; Roberts, P.A. Phytonematology Study Guide; Publication 4045; University of California, Division of Agriculture and Natural Resources: Los Angeles, CA, USA, 1985. [Google Scholar]
- Ahmad, L.; Siddiqui, Z.A.; Abd_Allah, E.F. Effects of interaction of Meloidogyne incognita, Alternaria dauci and Rhizoctonia solani on the growth, chlorophyll, carotenoid and proline contents of carrot in three types of soil. Acta Agric. Scand. Sect. B—Soil Plant Sci. 2019, 69, 324–331. [Google Scholar] [CrossRef]
- Makumbi-Kidza, N.N.; Speijer, P.R.; Sikora, R.A. Effects of Meloidogyne incognita on growth and storage-root formation of cassava (Manihot esculenta). J. Nematol. 2000, 32, 475–477. [Google Scholar]
- Kayani, M.Z.; Tariq Mukhtar, T.; Hussain, M.A. Interaction between nematode inoculum density and plant age on growth and yield of cucumber and reproduction of Meloidogyne incognita. Pak. J. Zool. 2018, 50, 897–902. [Google Scholar] [CrossRef]
- Mukhtar, T.; Kayani, M.Z. Growth and yield responses of fifteen cucumber cultivars to root-knot nematode (Meloidogyne incognita). Acta Sci. Pol. Hortorum Cultus 2019, 18, 45–52. [Google Scholar] [CrossRef]
- Strajnar, P.; Širca, S.; Urek, G.; Šircelj, H.; Železnik, P.; Vodnik, D. Effect of Meloidogyne ethiopica parasitism on water management and physiological stress in tomato. Eur. J. Plant Pathol. 2012, 132, 49–57. [Google Scholar] [CrossRef]
- Hlaoua, W.; Horrigue Raouani, N. Effet de Meloidogyne incognita sur la culture de pomme de terre. Nematol. Mediterr. 2007, 35, 213–220. [Google Scholar]
- Abd-Elgawad, M.M.M.; Kabeil, S.S.A. Biological control of Meloidogyne incognita by Trichoderma harzianum and Serratia marcescens and their related enzymatic changes in tomato roots. Afr. J. Biotechnol. 2012, 11, 16247–16252. [Google Scholar]
- Harman, G.E.; Doni, F.; Khadka, R.B.; Uphoff, N. Endophytic strains of Trichoderma increase plants’ photosynthetic capability. J. Appl. Microbiol. 2019, 130, 529–546. [Google Scholar] [CrossRef] [PubMed]
- Muthulakshmi, M.; Kumar, S.; Subramanian, S.; Anita, B. Compatibility of Pochonia chlamydosporia with other biocontrol agents and carbofuran. J. Biopestic. 2012, 5, 243–245. [Google Scholar]
- Molinari, S.; Leonetti, P. Bio-control agents activate plant immune response and prime susceptible tomato against root-knot nematodes. PLoS ONE 2019, 14, e0213230. [Google Scholar] [CrossRef]
- Siddaiah, C.N.; Satyanarayana, N.R.; Mudili, V.; Kumar Gupta, V.; Gurunathan, S.; Rangappa, S.; Huntrike, S.S.; Srivastava, R.K. Elicitation of resistance and associated defense responses in Trichoderma hamatum induced protection against pearl millet downy mildew pathogen. Sci. Rep. 2017, 7, 43991. [Google Scholar] [CrossRef]
- Khan, M.R.; Ahmad, I.; Ahamad, F. Effect of pure culture and culture filtrates of Trichoderma species on root-knot nematode, Meloidogyne incognita infesting tomato. Indian Phytopathol. 2018, 71, 265–274. [Google Scholar] [CrossRef]
- Poveda, J.; Abril-Urias, P.; Escobar, C. Biological control of plant-parasitic nematodes by filamentous fungi inducers of resistance: Trichoderma, mycorrhizal and endophytic fungi. Front. Microbiol. 2020, 11, 992. [Google Scholar] [CrossRef]
- Gillet, F.X.; Bournaud, C.; Antonino, J.D.; Junior, S.; Grossi-de-Sa, F.M. Plant-parasitic nematodes: Towards understanding molecular players in stress responses. Ann. Bot. 2017, 119, 775–789. [Google Scholar] [CrossRef]
- Melillo, M.T.; Leonetti, P.; Leone, A.; Veronico, P.; Bleve-Zacheo, T. ROS and NO production in compatible and incompatible tomato—Meloidogyne incognita interactions. Eur. J. Plant Pathol. 2011, 130, 489–502. [Google Scholar] [CrossRef]
- Radwan, D.E.M.; Fayez, K.A.; Mahmoud, S.Y.; Lu, G. Modifications of antioxidant activity and protein composition of bean leaf due to Bean yellow mosaic virus infection and salicylic acid treatments. Acta Physiol. Plant. 2010, 32, 891–904. [Google Scholar] [CrossRef]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Mohammad Pessarakli, M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. 2012, 26, 217037. [Google Scholar] [CrossRef]
- Małecka, A.; Derba-Maceluch, M.; Kaczorowska, K.; Piechalak, A.; Tomaszewska, B. Reactive oxygen species production and antioxidative defense system in pea root tissues treated with lead ions: Mitochondrial and peroxisomal level. Acta Physiol. Plant. 2009, 31, 1065–1075. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2000, 55, 373–399. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Noctor, G. Redox homeostasis and antioxidant signaling: A metabolic interface between stress perception and physiological responses. Plant Cell 2005, 17, 1866–1875. [Google Scholar] [CrossRef]
- Delaunois, B.; Jeandet, P.; Clément, C.; Baillieul, F.; Dorey, S.; Cordelier, S. Uncovering plant-pathogen crosstalk through apoplastic proteomic studies. Front. Plant Sci. 2014, 5, 249. [Google Scholar] [CrossRef]
- Nagesh, M.; Reddy, P. Biochemical changes in Glomus fasciculatum colonized roots of Lycopersicon esculentum in presence of Meloidogyne incognita. Indian J. Exp. Biol. 2004, 42, 721–727. [Google Scholar]
- Ashry, N.A.; Mohamed, H.I. Impact of secondary metabolites and related enzymes in flax resistance and/or susceptibility to powdery mildew. Afr. J. Biotechnol. 2012, 11, 1073–1077. [Google Scholar]
- Nagesh, M.; Reddy, P.; Ramchander, N. Integrated management of Meloidogyne incognita and Fusarium oxysporum f. sp. gladioli in gladiolus using antagonistic fungi and neem cake. In Proceedings of the 3rd International Symposium of Afro-Asian Nematologists, SBI, Coimbatore, India, 16–19 April 1998; pp. 263–266. [Google Scholar]
- Ezzat, A.S.; Badway, A.S.; Abdelkader, A.E. Sequenced Vermicompost, glycine Betaine, Proline Treatments Elevate Salinity Tolerance in Potatoes. Middle East J. Agric. Res. 2019, 8, 126–138. [Google Scholar]
- Lobato, M.C.; Machinandiarena, M.F.; Tambascio, C.; Dosio, G.A.; Caldiz, D.O.; Daleo, G.R.; Andreu, A.B.; Olivieri, F.P. Effect of foliar applications of phosphite on postharvest potato tubers. Eur. J. Plant Pathol. 2011, 130, 155–163. [Google Scholar] [CrossRef]
- Tikoria, R.; Kaur, A.; Ohri, P. Modulation of various phytoconstituents in tomato seedling growth and Meloidogyne incognita–Induced Stress Alleviation by Vermicompost Application. Front. Environ. Sci. 2022, 10, 891195. [Google Scholar] [CrossRef]
- El-Beltagi, H.S.; Farahat, A.A.; Alsayed, A.A.; Mahfoud, N.A. Response of antioxidant substances and enzymes activities as a defense mechanism against root-knot nematode infection. Not. Bot. Horti Agrobot. Cluj-Napoca 2012, 40, 132–142. [Google Scholar] [CrossRef]
- Afifi, A.M.R.; Al-Sayed, A.A.; Mahfoud, N.A.M.; Farahat, A.A. Enzymatic and non-enzymatic oxidants and antioxidants involved in defense mechanisms against root-knot, reniform and citrus nematodes in their hosts. Egypt. J. Agro. Nematol. 2014, 13, 172–188. [Google Scholar] [CrossRef]
- Arora, A.; Byrem, T.M.; Nair, M.G.; Strasburg, G.M. Modulation of liposomal membrane fluidity by flavonoids and isoflavonoids. Arch. Biochem. Biophys. 2000, 373, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Silva, H.; Anjo, S.I.; Manadas, B.; Abrantes, I.; Fonseca, L.; Cardoso, J.M.S. Comparative analysis of Bursaphelenchus xylophilus secretome under Pinus pinaster and P. pinea Stimuli. Front. Plant Sci. 2021, 12, 668064. [Google Scholar] [CrossRef]
- Grace, S.G.; Logan, B.A. Energy dissipation and radical scavenging by the plant phenylpropanoid pathway. Philos. Trans. R. Soc. B: Biol. Sci. 2000, 355, 1499–1510. [Google Scholar] [CrossRef]
- Michalak, A. Phenolic compounds and their antioxidant activity in plants growing under heavy metal stress. Pol. J. Environ. Stud. 2006, 15, 523–530. [Google Scholar]
- Galeng-Lawilao, J.; Kumar, A.; Cabasan, M.T.N.; De Waele, D. Comparison of the penetration, development and reproduction of Meloidogyne graminicola, and analysis of lignin and total phenolic content in partially resistant and resistant recombinant inbred lines of Oryza sativa. Trop. Plant Pathol. 2019, 44, 171–182. [Google Scholar] [CrossRef]
- Sharma, P.; Dubey, R.S. Drought induces oxidative stress and enhances the activities of antioxidant enzymes in growing rice seedlings. Plant Grow Reg. 2005, 46, 209–221. [Google Scholar] [CrossRef]
- Han, C.; Liu, Q.; Yang, Y. Short-term effects of experimental warming and enhanced Ultraviolet-B radiation on photosynthesis and antioxidant defense of Picea asperata seedlings. Plant Growth Regul. 2009, 58, 153–162. [Google Scholar] [CrossRef]
- Tanou, G.; Molassiotis, A.; Diamantidis, G. Induction of reactive oxygen species and necrotic death-like destruction in strawberry leaves by salinity. Environ. Exp. Bot. 2009, 65, 270–281. [Google Scholar] [CrossRef]
- Mishra, S.; Jha, A.B.; Dubey, R.S. Arsenite treatment induces oxidative stress, upregulates antioxidant system, and causes phytochelatin synthesis in rice seedlings. Protoplasma 2011, 248, 565–577. [Google Scholar] [CrossRef] [PubMed]
- Bali, S.; Kaur, P.; Jamwal, V.L.; Gandhi, S.G.; Sharma, A.; Ohri, P.; Bhardwaj, R.; Ali, M.A.; Ahmad, P. Seed priming with jasmonic acid counteracts root knot nematode infection in tomato by modulating the activity and expression of antioxidative enzymes. Biomolecules 2020, 10, 98. [Google Scholar] [CrossRef] [PubMed]
- Labudda, M.; Różańska, E.; Czarnocka, W.; Sobczak, M.; Dzik, J.M. Systemic changes in photosynthesis and reactive oxygen species homeostasis in shoots of Arabidopsis Thaliana infected with the beet cyst nematode Heterodera Schachtii. Mol. Plant Pathol. 2018, 19, 1690–1704. [Google Scholar] [CrossRef]
- Yamauchi, Y.; Furutera, A.; Seki, K.; Toyoda, Y.; Tanaka, K.; Sugimoto, Y. Malondialdehyde generated from peroxidized linolenic acid causes protein modification in heat-stressed plants. Plant Physiol. Biochem. 2008, 46, 786–793. [Google Scholar] [CrossRef] [PubMed]
- Oka, Y.; Chilt, I.; Spiegel, Y. Are pathogenesis-related proteins induced by Meloidogyne javanica or Hetrodera avenae invasion? J. Nematol. 1997, 29, 501–508. [Google Scholar]
- Maheshwari, R.; Dubey, R.S. Nickel-induced oxidative stress and the role of antioxidant defense in rice seedlings. Plant Growth Regul. 2009, 59, 37–49. [Google Scholar] [CrossRef]
- Romero-Puertas, M.C.; Palma, J.M.; Gomez, M.; Del’R’ıo, L.A.; Sandalio, L.M. Cadmium causes the oxidative modification of proteins in pea plants. Plant Cell Environ. 2002, 25, 677–686. [Google Scholar] [CrossRef]
- Waszczak, C.; Carmody, M.; Kangasjärvi, J. Reactive oxygen species in plant signaling. Ann. Rev. Plant Biol. 2018, 69, 209–236. [Google Scholar] [CrossRef]
- Sobotta, M.C.; Liou, W.; Stocker, S.; Talwar, D.; Oehler, M.; Ruppert, T.; Scharf, A.N.D.; Dick, T.P. Peroxiredoxin-2 and STAT3 form a redox relay for H2O2 signaling. Nat. Chem. Biol. 2014, 11, 64–70. [Google Scholar] [CrossRef]
- Sahebani, N.; Hadavi, H. Biological control of the root-knot nematode Meloidogyne javanica by Trichoderma harzianum. Soil Biol. Biochem. 2008, 40, 2016–2020. [Google Scholar] [CrossRef]
- Kesba, H.H.; El-Beltagi, H.E.S. Biochemical changes in grape rootstocks resulted from humic acid treatments in relation to nematode infection. Asian Pac. J. Trop. Biomed. 2012, 2, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Noureldeen, A.; Asif, M.; Ansari, T.; Khan, F.; Shariq, M.; Ahmad, F.; Mfarrej, M.F.B.; Khan, A.; Tariq, M.; Siddiqui, M.A.; et al. Effect of individual, simultaneous and sequential inoculation of Pseudomonas fluorescens and Meloidogyne incognita on growth, biochemical, enzymatic and nonenzymatic antioxidants of tomato (Solanum lycopersicum L.). Plants 2021, 10, 1145. [Google Scholar] [CrossRef] [PubMed]
- Vos, C.M.; Schouteden, N.; van Tuienen, D.; Chatagnier, O.; Elsen, A.; De Waele, D.; Panis, B.; Gianinazzi-Pearson, V. Mycorrhiza-induced resistance against the root-knot nematode Meloidogyne incognita involves priming of defense gene responses in tomato. Soil Biol. Biochem. 2013, 60, 45–54. [Google Scholar] [CrossRef]
- Prakash Sharma, I.; Sharma, A.K. Co-inoculation of tomato with an arbuscular mycorrhizal fungus improves plant immunity and reduces root-knot nematode infection. Rhizosphere 2017, 4, 25–28. [Google Scholar] [CrossRef]
- Martínez-Medina, A.; Fernandez, I.; Lok, G.B.; Pozo, M.J.; Pieterse, C.M.; Van Wees, S.C. Shifting from priming of salicylic acid-to jasmonic acid-regulated defenses by Trichoderma protects tomato against the root-knot nematode Meloidogyne incognita. New Phytol. 2017, 213, 1363–1377. [Google Scholar] [CrossRef] [PubMed]

| Fungal Isolate | Juveniles Mortality (%) | Egg Hatching Rate (%) | Egg Infection Rate (%) |
|---|---|---|---|
| Control | - | 95.69 ± 0.58 a | - |
| T. longibrachiatum | 87.43 ± 0.70 a | 41.20 ± 0.57 d | 76.50 ± 0.51 c |
| T. harzianum | 60.27 ± 0.76 b | 47.94 ± 0.62 c | 72.20 ± 1.11 d |
| T. asperellum | 44.13 ± 1.58 d | 55.21 ± 0.57 b | 66.29 ± 0.71 e |
| M. chlamydosporia | 62.28 ± 0.48 b | 32.40 ± 0.84 e | 86.84 ± 0.28 a |
| Lecanicillium spp. | 55.84 ± 0.69 c | 30.05 ± 0.34 f | 81.36 ± 0.33 b |
| Treatments | Shoot | Root | ||
|---|---|---|---|---|
| Weight (g) | Height (cm) | Weight (g) | Length (cm) | |
| Untreated Control | 19.80 ± 3.28 ab | 36.70 ± 3.35 b | 5.02 ± 1.69 cd | 20.30 ± 5.50 a |
| RKN only | 11.62 ± 1.24 d | 16.92 ± 7.95 c | 5.95 ± 0.96 c | 18.67 ± 2.34 b |
| T. longibrachiatum + RKN | 18.27 ± 2.70 b | 36.33 ± 3.71 b | 4.22 ± 0.97 d | 19.83 ± 4.37 a |
| T. harzianum + RKN | 18.65 ± 2.17 b | 40.00 ± 6.63 a | 6.16 ± 0.58 c | 22.50 ± 2.19 a |
| T. asperellum + RKN | 19.78 ± 2.50 ab | 32.08 ± 4.10 b | 4.32 ± 0.29 d | 19.83 ± 4.32 a |
| M. chlamydosporia + RKN | 15.57 ± 2.92 c | 30.50 ± 2.76 b | 6.98 ± 1.82 b | 18.83 ± 1.33 ab |
| Lecanicillium spp. + RKN | 21.32 ± 2.22 a | 35.25 ± 4.24 b | 7.00 ± 1.84 b | 18.08 ± 3.60 b |
| CHM + RKN | 17.00 ± 1.84 c | 31.33 ± 6.15 b | 7.72 ± 2.59 b | 18.50 ± 2.24 b |
| BIO + RKN | 16.03 ± 2.38 c | 28.00 ± 3.02 b | 9.05 ± 2.07 a | 18.08 ± 2.01 c |
| Treatments | APX | CAT | PPO | POD |
|---|---|---|---|---|
| U mn−1 g−1 FW | U mn−1 g−1 FW | U mn−1 g−1 FW | U mn−1 g−1 FW | |
| Untreated Control | 5.91 ± 1.02 c | 2.40 ± 0.51 d | 1.44 ± 0.37 c | 1.57 ± 0.34 d |
| RKN only | 12.02 ± 1.72 ab | 5.10 ± 0.80 d | 2.64 ± 0.34 c | 3.10 ± 0.12 c |
| T. longibrachiatum + RKN | 13.22 ± 0.32 a | 22.12 ± 0.61 a | 6.05 ± 0.41 a | 4.13 ± 0.32 b |
| T. harzianum + RKN | 11.41 ± 1.01 b | 18.64 ± 0.82 b | 6.37 ± 0.34 a | 3.91 ± 0.22 b |
| T. asperellum + RKN | 11.04 ± 0.21 b | 19.40 ± 0.34 ab | 5.51 ± 0.28 ab | 5.92 ± 0.46 a |
| M. chlamydosporia + RKN | 13.01 ± 0.20 a | 18.96 ± 0.10 b | 6.19 ± 0.50 a | 4.28 ± 0.40 b |
| Lecanicillium spp. + RKN | 10.61 ± 1.02 b | 18.82 ± 0.54 b | 4.56 ± 0.62 b | 3.64 ± 0.21 bc |
| CHM + RKN | 13.11 ± 0.32 a | 19.36 ± 0.90 ab | 6.31 ± 0.54 a | 3.10 ± 0.26 c |
| BIO + RKN | 11.18 ± 0.48 b | 10.60 ± 0.83 c | 4.32 ± 0.31 b | 3.64 ± 0.41 bc |
| Treatment | MDA | Total Protein Content | Total Phenol Content |
|---|---|---|---|
| µmol g−1 FW | mg g−1 FW | mg g−1 FW | |
| Untreated Control | 2.32 ± 0.99 c | 2.34 ± 0.18 c | 2.71 ± 0.14 bc |
| RKN (N) only | 12.44 ± 1.22 a | 3.36 ± 0.14 ab | 3.25 ± 0.23 a |
| T. longibrachiatum + RKN | 7.44 ± 1.01 b | 3.07 ± 0.08 abc | 2.01 ± 0.19 c |
| T. harzianum + RKN | 7.01 ± 0.92 b | 2.54 ± 0.26 abc | 2.52 ± 0.26 bc |
| T. asperellum + RKN | 6.97 ± 0.66 b | 3.12 ± 0.34 abc | 1.86 ± 0.43 c |
| M. chlamydosporia + RKN | 7.20 ± 0.46 b | 2.50 ± 0.43 bc | 2.32 ± 0.43 bc |
| Lecanicillium spp. + RKN | 10.76 ± 0.48 a | 2.93 ± 0.21 abc | 2.64 ± 0.32 abc |
| CHM + RKN | 7.09 ± 0.31 b | 2.62 ± 0.36 abc | 2.26 ± 0.43 bc |
| BIO + RKN | 7.34 ± 0.46 b | 3.49 ± 0.06 a | 2.84 ± 0.21 ab |
| Treatment | Gall Index | Galls Number/g of Root | Multiplication Rate (Pf/Pi) |
|---|---|---|---|
| RKN only | 2.55 ± 0.54 a | 86.00 ± 2.92 a | 4.62 ± 1.67 a |
| T. longibrachiatum + RKN | 1.16 ± 0.26 b | 36.00 ± 1.56 c | 0.53 ± 0.87 d |
| T. harzianum + RKN | 1.83 ± 0.54 ab | 71.00 ± 2.10 b | 2.18 ± 0.65 c |
| T. asperellum + RKN | 2.00 ± 0.50 ab | 70.66 ± 2.32 b | 3.87 ± 0.42 b |
| M. chlamydosporia + RKN | 1.33 ± 0.51 b | 36.00 ± 3.49 c | 2.68 ± 0.81 c |
| Lecanicillium spp. + RKN | 2.33 ± 0.34 a | 67.83 ± 3.41 b | 3.33 ± 0.33 b |
| CHM + RKN | 1.16 ± 0.40 b | 39.00 ± 2.45 c | 0.54 ± 0.19 d |
| BIO + RKN | 1.5 ± 0.54 b | 41.50 ± 2.30 c | 0.64 ± 0.21 d |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hajji-Hedfi, L.; Hlaoua, W.; Al-Judaibi, A.A.; Rhouma, A.; Horrigue-Raouani, N.; Abdel-Azeem, A.M. Comparative Effectiveness of Filamentous Fungi in Biocontrol of Meloidogyne javanica and Activated Defense Mechanisms on Tomato. J. Fungi 2023, 9, 37. https://doi.org/10.3390/jof9010037
Hajji-Hedfi L, Hlaoua W, Al-Judaibi AA, Rhouma A, Horrigue-Raouani N, Abdel-Azeem AM. Comparative Effectiveness of Filamentous Fungi in Biocontrol of Meloidogyne javanica and Activated Defense Mechanisms on Tomato. Journal of Fungi. 2023; 9(1):37. https://doi.org/10.3390/jof9010037
Chicago/Turabian StyleHajji-Hedfi, Lobna, Wassila Hlaoua, Awatif A. Al-Judaibi, Abdelhak Rhouma, Najet Horrigue-Raouani, and Ahmed M. Abdel-Azeem. 2023. "Comparative Effectiveness of Filamentous Fungi in Biocontrol of Meloidogyne javanica and Activated Defense Mechanisms on Tomato" Journal of Fungi 9, no. 1: 37. https://doi.org/10.3390/jof9010037
APA StyleHajji-Hedfi, L., Hlaoua, W., Al-Judaibi, A. A., Rhouma, A., Horrigue-Raouani, N., & Abdel-Azeem, A. M. (2023). Comparative Effectiveness of Filamentous Fungi in Biocontrol of Meloidogyne javanica and Activated Defense Mechanisms on Tomato. Journal of Fungi, 9(1), 37. https://doi.org/10.3390/jof9010037









