Lycosin-II Exhibits Antifungal Activity and Inhibits Dual-Species Biofilm by Candida albicans and Staphylococcus aureus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Peptide Synthesis, Purification, and Mass Analysis
2.3. Antifungal Activity
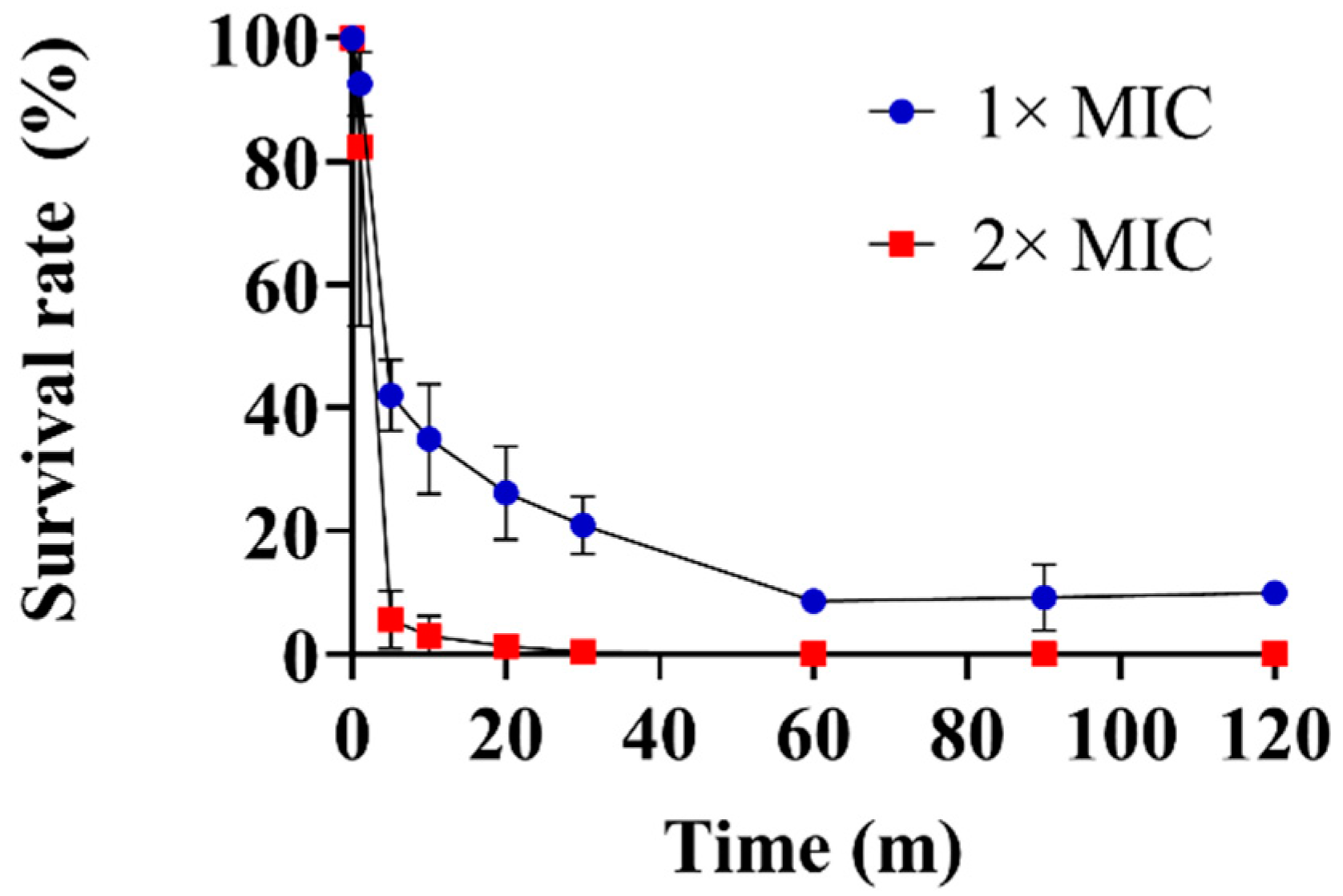
2.4. Time-Kill Kinetics Assay
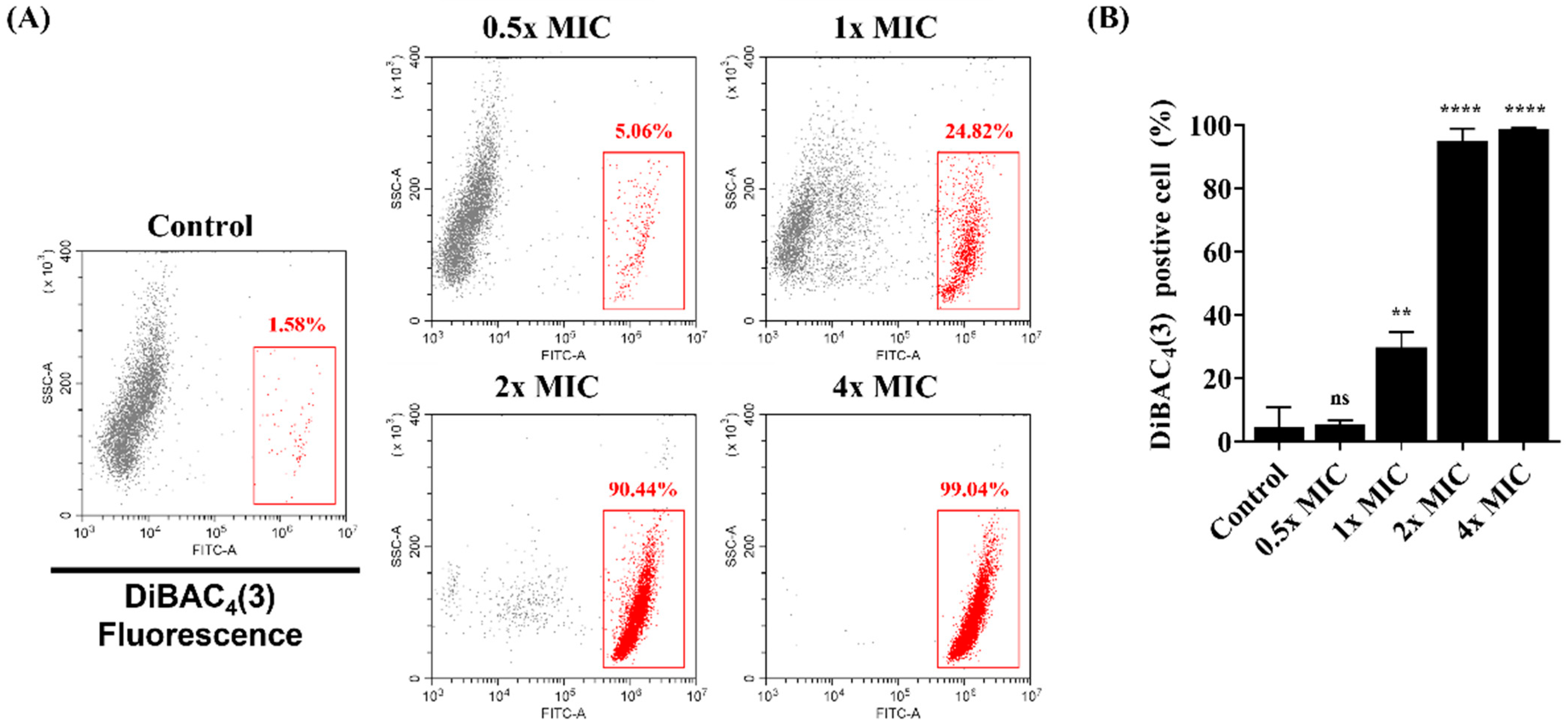
2.5. Membrane Depolarization Assay
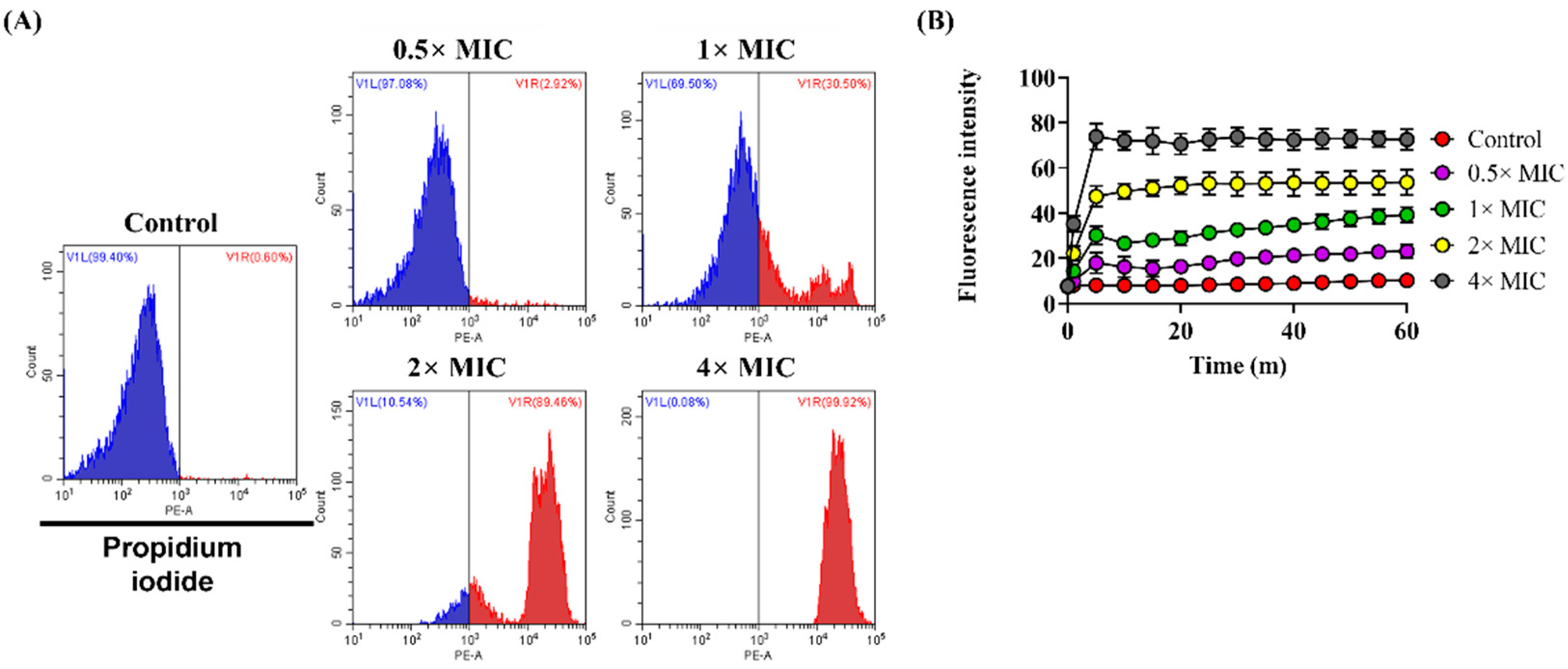
2.6. PI Uptake Assay
2.7. SYTOX Green Uptake Assay
2.8. Measurement of ROS
2.9. Analysis of the Effect of NAC on the Antifungal Activity of Lycosin-II
2.10. Biofilm Inhibition Assays
2.11. Determination of C. albicans and S. aureus Viability in a Dual-Species Biofilm
2.12. Visualization of the Biofilm
2.13. Statistics
3. Results
3.1. Antifungal Activity against C. albicans
3.2. Membrane Potential Levels of C. albicans
3.3. Effect of Lycosin-II on Membrane Integrity
3.4. Antifungal Activity against C. albicans
3.5. Inhibitory Effects of Lycosin-II on Biofilm
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Khezerlou, A.; Alizadeh-Sani, M.; Azizi-Lalabadi, M.; Ehsani, A. Nanoparticles and their antimicrobial properties against pathogens including bacteria, fungi, parasites and viruses. Microb. Pathog. 2018, 123, 505–526. [Google Scholar] [CrossRef] [PubMed]
- Bugli, F.; Massaro, F.; Buonocore, F.; Saraceni, P.R.; Borocci, S.; Ceccacci, F.; Bombelli, C.; Di Vito, M.; Marchitiello, R.; Mariotti, M. Design and characterization of myristoylated and non-myristoylated peptides effective against Candida spp. Clinical isolates. Int. J. Mol. Sci. 2022, 23, 2164. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Cheng, L.W.; Chan, K.L.; Tam, C.C.; Mahoney, N.; Friedman, M.; Shilman, M.M.; Land, K.M. Antifungal drug repurposing. Antibiotics 2020, 9, 812. [Google Scholar] [CrossRef] [PubMed]
- Burgain, A.; Tebbji, F.; Khemiri, I.; Sellam, A. Metabolic reprogramming in the opportunistic yeast candida albicans in response to hypoxia. Msphere 2020, 5, e00913–e00919. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Puumala, E.; Robbins, N.; Cowen, L.E. Antifungal drug resistance: Molecular mechanisms in candida albicans and beyond. Chem. Rev. 2020, 121, 3390–3411. [Google Scholar] [CrossRef]
- Chen, H.; Zhou, X.; Ren, B.; Cheng, L. The regulation of hyphae growth in candida albicans. Virulence 2020, 11, 337–348. [Google Scholar] [CrossRef]
- Nile, C.; Falleni, M.; Cirasola, D.; Alghamdi, A.; Anderson, O.F.; Delaney, C.; Ramage, G.; Ottaviano, E.; Tosi, D.; Bulfamante, G. Repurposing pilocarpine hydrochloride for treatment of candida albicans infections. Msphere 2019, 4, e00689-18. [Google Scholar] [CrossRef]
- Rafat, Z.; Hashemi, S.J.; Ashrafi, K.; Nikokar, I.; Jafari, A.; Foroushani, A.R.; Roohi, B.; Boroujeini, Z.B.; Rashidi, N.; Najar-Shahri, N. Fungal isolates of the respiratory tract in symptomatic patients hospitalized in pulmonary units: A mycological and molecular epidemiologic study. J. Multidiscip. Healthc. 2020, 13, 661. [Google Scholar] [CrossRef]
- Cavalheiro, M.; Teixeira, M.C. Candida biofilms: Threats, challenges, and promising strategies. Front. Med. 2018, 5, 28. [Google Scholar] [CrossRef]
- Lohse, M.B.; Gulati, M.; Johnson, A.D.; Nobile, C.J. Development and regulation of single-and multi-species candida albicans biofilms. Nat. Rev. Microbiol. 2018, 16, 19–31. [Google Scholar] [CrossRef]
- Carolus, H.; Van Dyck, K.; Van Dijck, P. Candida albicans and staphylococcus species: A threatening twosome. Front. Microbiol. 2019, 10, 2162. [Google Scholar] [CrossRef] [PubMed]
- Sovari, S.N.; Radakovic, N.; Roch, P.; Crochet, A.; Pavic, A.; Zobi, F. Combatting amr: A molecular approach to the discovery of potent and non-toxic rhenium complexes active against C. albicans-mrsa co-infection. Eur. J. Med. Chem. 2021, 226, 113858. [Google Scholar] [CrossRef] [PubMed]
- Mwangi, J.; Yin, Y.; Wang, G.; Yang, M.; Li, Y.; Zhang, Z.; Lai, R. The antimicrobial peptide zy4 combats multidrug-resistant pseudomonas aeruginosa and acinetobacter baumannii infection. Proc. Natl. Acad. Sci. USA 2019, 116, 26516–26522. [Google Scholar] [CrossRef] [PubMed]
- Elnagdy, S.; AlKhazindar, M. The potential of antimicrobial peptides as an antiviral therapy against COVID-19. ACS Pharmacol. Transl. Sci. 2020, 3, 780–782. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Yang, H.; Xiao, H.; Farooq, A.; Liu, Z.; Hu, M.; Shi, X. The spider venom peptide lycosin-ii has potent antimicrobial activity against clinically isolated bacteria. Toxins 2016, 8, 119. [Google Scholar] [CrossRef]
- Oh, J.H.; Park, J.; Park, Y. Anti-biofilm and anti-inflammatory effects of lycosin-ii isolated from spiders against multi-drug resistant bacteria. Biochim. Biophys. Acta-Biomembr. 2022, 1864, 183769. [Google Scholar] [CrossRef]
- Park, S.-C.; Kim, J.-Y.; Kim, E.-J.; Cheong, G.-W.; Lee, Y.; Choi, W.; Lee, J.R.; Jang, M.-K. Hydrophilic linear peptide with histidine and lysine residues as a key factor affecting antifungal activity. Int. J. Mol. Sci. 2018, 19, 3781. [Google Scholar] [CrossRef]
- Radhakrishnan, V.S.; Mudiam, M.K.R.; Kumar, M.; Dwivedi, S.P.; Singh, S.P.; Prasad, T. Silver nanoparticles induced alterations in multiple cellular targets, which are critical for drug susceptibilities and pathogenicity in fungal pathogen (candida albicans). Int. J. Nanomed. 2018, 13, 2647. [Google Scholar] [CrossRef]
- Hong, M.J.; Kim, M.K.; Park, Y. Comparative antimicrobial activity of hp404 peptide and its analogs against acinetobacter baumannii. Int. J. Mol. Sci. 2021, 22, 5540. [Google Scholar] [CrossRef]
- Dong, N.; Chou, S.; Li, J.; Xue, C.; Li, X.; Cheng, B.; Shan, A.; Xu, L. Short symmetric-end antimicrobial peptides centered on β-turn amino acids unit improve selectivity and stability. Front. Microbiol. 2018, 9, 2832. [Google Scholar] [CrossRef]
- Zorić, N.; Kosalec, I.; Tomić, S.; Bobnjarić, I.; Jug, M.; Vlainić, T.; Vlainić, J. Membrane of candida albicans as a target of berberine. BMC Complement. Altern. Med. 2017, 17, 268. [Google Scholar] [CrossRef] [PubMed]
- Seyedjavadi, S.S.; Khani, S.; Eslamifar, A.; Ajdary, S.; Goudarzi, M.; Halabian, R.; Akbari, R.; Zare-Zardini, H.; Imani Fooladi, A.A.; Amani, J. The antifungal peptide mch-amp1 derived from matricaria chamomilla inhibits candida albicans growth via inducing ros generation and altering fungal cell membrane permeability. Front. Microbiol. 2020, 10, 3150. [Google Scholar] [CrossRef] [PubMed]
- Ramamourthy, G.; Park, J.; Seo, C.; Vogel, H.J.; Park, Y. Antifungal and antibiofilm activities and the mechanism of action of repeating lysine-tryptophan peptides against candida albicans. Microorganisms 2020, 8, 758. [Google Scholar] [CrossRef]
- Wong, J.H.; Ng, T.B.; Legowska, A.; Rolka, K.; Hui, M.; Cho, C.H. Antifungal action of human cathelicidin fragment (ll13–37) on candida albicans. Peptides 2011, 32, 1996–2002. [Google Scholar] [CrossRef] [PubMed]
- Hwang, B.; Hwang, J.-S.; Lee, J.; Lee, D.G. The antimicrobial peptide, psacotheasin induces reactive oxygen species and triggers apoptosis in candida albicans. Biochem. Biophys. Res. Commun. 2011, 405, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chang, W.; Zhang, M.; Li, X.; Jiao, Y.; Lou, H. Diorcinol d exerts fungicidal action against candida albicans through cytoplasm membrane destruction and ros accumulation. PLoS ONE 2015, 10, e0128693. [Google Scholar]
- Chang, C.-K.; Kao, M.-C.; Lan, C.-Y. Antimicrobial activity of the peptide lfcinb15 against candida albicans. J. Fungi 2021, 7, 519. [Google Scholar] [CrossRef]
- Luo, Y.; McAuley, D.F.; Fulton, C.R.; Sá Pessoa, J.; McMullan, R.; Lundy, F.T. Targeting candida albicans in dual-species biofilms with antifungal treatment reduces staphylococcus aureus and mrsa in vitro. PLoS ONE 2021, 16, e0249547. [Google Scholar] [CrossRef]
- Kim, M.K.; Kang, N.H.; Ko, S.J.; Park, J.; Park, E.; Shin, D.W.; Kim, S.H.; Lee, S.A.; Lee, J.I.; Lee, S.H. Antibacterial and antibiofilm activity and mode of action of magainin 2 against drug-resistant acinetobacter baumannii. Int. J. Mol. Sci. 2018, 19, 3041. [Google Scholar] [CrossRef]
- Wang, Y.; Pei, Z.; Lou, Z.; Wang, H. Evaluation of anti-biofilm capability of cordycepin against candida albicans. Infect. Drug Resist. 2021, 14, 435. [Google Scholar] [CrossRef]
- Cruz, C.D.; Shah, S.; Tammela, P. Defining conditions for biofilm inhibition and eradication assays for gram-positive clinical reference strains. BMC Microbiol. 2018, 18, 173. [Google Scholar] [CrossRef] [PubMed]
- Galdiero, E.; Di Onofrio, V.; Maione, A.; Gambino, E.; Gesuele, R.; Menale, B.; Ciaravolo, M.; Carraturo, F.; Guida, M. Allium ursinum and allium oschaninii against klebsiella pneumoniae and candida albicans mono-and polymicrobic biofilms in in vitro static and dynamic models. Microorganisms 2020, 8, 336. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Dang, W.; Xie, J.; Zhu, R.; Sun, M.; Jia, F.; Zhao, Y.; An, X.; Qiu, S.; Li, X. Antimicrobial peptide protonectin disturbs the membrane integrity and induces ros production in yeast cells. Biochim. Biophys. Acta-Biomembr. 2015, 1848, 2365–2373. [Google Scholar] [CrossRef] [PubMed]
- Rasamiravaka, T.; Labtani, Q.; Duez, P.; El Jaziri, M. The formation of biofilms by pseudomonas aeruginosa: A review of the natural and synthetic compounds interfering with control mechanisms. BioMed Res. Int. 2015, 2015, 759348. [Google Scholar] [CrossRef]
- Chen, C.H.; Lu, T.K. Development and challenges of antimicrobial peptides for therapeutic applications. Antibiotics 2020, 9, 24. [Google Scholar] [CrossRef]
- Gao, J.; Wang, H.; Li, Z.; Wong, A.H.-H.; Wang, Y.-Z.; Guo, Y.; Lin, X.; Zeng, G.; Liu, H.; Wang, Y. Candida albicans gains azole resistance by altering sphingolipid composition. Nat. Commun. 2018, 9, 4495. [Google Scholar] [CrossRef]
- Aigner, M.; Lass-Flörl, C. Encochleated amphotericin b: Is the oral availability of amphotericin b finally reached? J. Fungi 2020, 6, 66. [Google Scholar] [CrossRef]
- Falanga, A.; Lombardi, L.; Franci, G.; Vitiello, M.; Iovene, M.R.; Morelli, G.; Galdiero, M.; Galdiero, S. Marine antimicrobial peptides: Nature provides templates for the design of novel compounds against pathogenic bacteria. Int. J. Mol. Sci. 2016, 17, 785. [Google Scholar] [CrossRef]
- Kumar, S.; Varela, M.F. Molecular mechanisms of bacterial resistance to antimicrobial agents. Chemotherapy 2013, 14, 522–534. [Google Scholar]
- Ma, H.; Zhao, X.; Yang, L.; Su, P.; Fu, P.; Peng, J.; Yang, N.; Guo, G. Antimicrobial peptide amp-17 affects candida albicans by disrupting its cell wall and cell membrane integrity. Infect. Drug Resist. 2020, 13, 2509. [Google Scholar] [CrossRef]
- Lee, H.; Hwang, J.-S.; Lee, J.; Kim, J.I.; Lee, D.G. Scolopendin 2, a cationic antimicrobial peptide from centipede, and its membrane-active mechanism. Biochim. Biophys. Acta-Biomembr. 2015, 1848, 634–642. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Lee, D.G. Fungicidal mechanisms of the antimicrobial peptide bac8c. Biochim. Biophys. Acta-Biomembr. 2015, 1848, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Dong, W.; Sun, L.; Ma, L.; Shang, D. Insights into the membrane interaction mechanism and antibacterial properties of chensinin-1b. Biomaterials 2015, 37, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Hwang, B.; Hwang, J.-S.; Lee, J.; Kim, J.-K.; Kim, S.R.; Kim, Y.; Lee, D.G. Induction of yeast apoptosis by an antimicrobial peptide, papiliocin. Biochem. Biophys. Res. Commun. 2011, 408, 89–93. [Google Scholar] [CrossRef]
- Dbouk, N.H.; Covington, M.B.; Nguyen, K.; Chandrasekaran, S. Increase of reactive oxygen species contributes to growth inhibition by fluconazole in cryptococcus neoformans. BMC Microbiol. 2019, 19, 243. [Google Scholar] [CrossRef]
- Kong, E.F.; Tsui, C.; Kucharíková, S.; Andes, D.; Van Dijck, P.; Jabra-Rizk, M.A. Commensal protection of staphylococcus aureus against antimicrobials by candida albicans biofilm matrix. MBio 2016, 7, e01365-16. [Google Scholar] [CrossRef]
- Kean, R.; Rajendran, R.; Haggarty, J.; Townsend, E.M.; Short, B.; Burgess, K.E.; Lang, S.; Millington, O.; Mackay, W.G.; Williams, C. Candida albicans mycofilms support staphylococcus aureus colonization and enhances miconazole resistance in dual-species interactions. Front. Microbiol. 2017, 8, 258. [Google Scholar] [CrossRef]
- Hu, Y.; Niu, Y.; Ye, X.; Zhu, C.; Tong, T.; Zhou, Y.; Zhou, X.; Cheng, L.; Ren, B. Staphylococcus aureus synergized with candida albicans to increase the pathogenesis and drug resistance in cutaneous abscess and peritonitis murine models. Pathogens 2021, 10, 1036. [Google Scholar] [CrossRef]


| Strains | MIC (μM) | |||
|---|---|---|---|---|
| Lycosin-II | Melittin | Fluconazole | Amphotericin B | |
| C. albicans (KCTC * 7270) | 2 | 2 | 16 | 1 |
| C. albicans (CCARM * 14001) | 2 | 2 | >32 | 0.5 |
| C. albicans (CCARM 14004) | 2 | 4 | >32 | 0.5 |
| C. albicans (CCARM 14020) | 2 | 2 | >32 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, J.; Kim, H.; Kang, H.-K.; Choi, M.-C.; Park, Y. Lycosin-II Exhibits Antifungal Activity and Inhibits Dual-Species Biofilm by Candida albicans and Staphylococcus aureus. J. Fungi 2022, 8, 901. https://doi.org/10.3390/jof8090901
Park J, Kim H, Kang H-K, Choi M-C, Park Y. Lycosin-II Exhibits Antifungal Activity and Inhibits Dual-Species Biofilm by Candida albicans and Staphylococcus aureus. Journal of Fungi. 2022; 8(9):901. https://doi.org/10.3390/jof8090901
Chicago/Turabian StylePark, Jonggwan, Hyeongsun Kim, Hee-Kyoung Kang, Moon-Chang Choi, and Yoonkyung Park. 2022. "Lycosin-II Exhibits Antifungal Activity and Inhibits Dual-Species Biofilm by Candida albicans and Staphylococcus aureus" Journal of Fungi 8, no. 9: 901. https://doi.org/10.3390/jof8090901
APA StylePark, J., Kim, H., Kang, H.-K., Choi, M.-C., & Park, Y. (2022). Lycosin-II Exhibits Antifungal Activity and Inhibits Dual-Species Biofilm by Candida albicans and Staphylococcus aureus. Journal of Fungi, 8(9), 901. https://doi.org/10.3390/jof8090901







