H2O2 Induces Calcium and ERMES Complex-Dependent Mitochondrial Constriction and Division as Well as Mitochondrial Outer Membrane Remodeling in Aspergillus nidulans
Abstract
:1. Introduction
2. Materials and Methods
2.1. Strains, Media, and Growth Conditions
2.2. Gene Deletion and Organelle Labelling
2.3. Confocal and Airyscan Microscopy
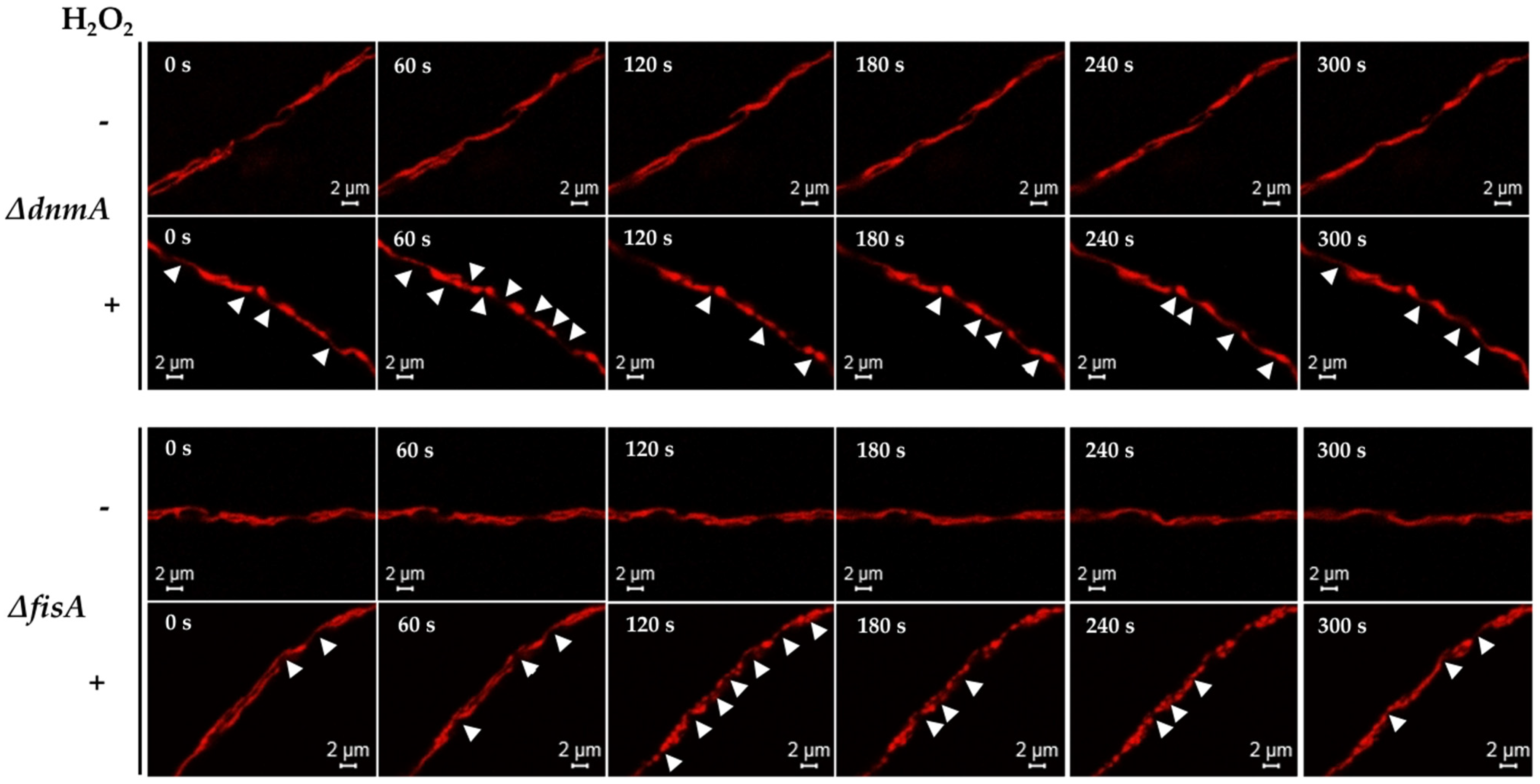
3. Results
3.1. H2O2 and CCCP Induce Mitochondrial Constriction and Membrane Depolarization
3.2. H2O2-Induced Mitochondrial Constriction Sites Are in Contact with the Endoplasmic Reticulum
3.3. Mitochondrial Constrictions and ER-Dotted Structures Are Formed during Asexual Development
3.4. Calcium Is Required for Mitochondrial Constrictions Induced by H2O2 or CCCP but Not for the Remodeling of the Mitochondrial Outer Membrane
3.5. MdmB, a Subunit of the ERMES Complex, Is Required for Mitochondrial Constriction and Division
4. Discussion
4.1. H2O2 Induces Mitochondrial Depolarization, Constriction and Rearrangements of the Outer Mitochondrial Membrane
4.2. Intracellular Ca2+ and MdmB, a Subunit of the ERMES Complex, Are Required for H2O2-Induced Mitochondrial Constriction and Division
4.3. H2O2 Differential Roles in Mitochondrial Dynamics
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aguirre, J.; Rios-Momberg, M.; Hewitt, D.; Hansberg, W. Reactive oxygen species and development in microbial eukaryotes. Trends Microbiol. 2005, 13, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Hansberg, W.; Aguirre, J. Hyperoxidant states cause microbial cell differentiation by cell isolation from dioxygen. J. Theor. Biol. 1990, 142, 201–221. [Google Scholar] [CrossRef]
- Cano-Dominguez, N.; Alvarez-Delfin, K.; Hansberg, W.; Aguirre, J. NADPH oxidases NOX-1 and NOX-2 require the regulatory subunit NOR-1 to control cell differentiation and growth in Neurospora crassa. Eukaryot. Cell 2008, 7, 1352–1361. [Google Scholar] [CrossRef] [Green Version]
- Cano-Dominguez, N.; Bowman, B.; Peraza-Reyes, L.; Aguirre, J. Neurospora crassa NADPH Oxidase NOX-1 Is Localized in the Vacuolar System and the Plasma Membrane. Front. Microbiol. 2019, 10, 1825. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dirschnabel, D.E.; Nowrousian, M.; Cano-Dominguez, N.; Aguirre, J.; Teichert, I.; Kuck, U. New Insights Into the Roles of NADPH Oxidases in Sexual Development and Ascospore Germination in Sordaria macrospora. Genetics 2014, 196, 729–744. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lara-Ortiz, T.; Riveros-Rosas, H.; Aguirre, J. Reactive oxygen species generated by microbial NADPH oxidase NoxA regulate sexual development in Aspergillus nidulans. Mol. Microbiol. 2003, 50, 1241–1255. [Google Scholar] [CrossRef] [Green Version]
- Malagnac, F.; Lalucque, H.; Lepere, G.; Silar, P. Two NADPH oxidase isoforms are required for sexual reproduction and ascospore germination in the filamentous fungus Podospora anserina. Fungal Genet. Biol. 2004, 41, 982–997. [Google Scholar] [CrossRef]
- Kawasaki, L.; Sanchez, O.; Shiozaki, K.; Aguirre, J. SakA MAP kinase is involved in stress signal transduction, sexual development and spore viability in Aspergillus nidulans. Mol. Microbiol. 2002, 45, 1153–1163. [Google Scholar] [CrossRef]
- Mendoza-Martínez, A.E.; Cano-Domínguez, N.; Aguirre, J. Yap1 homologs mediate more than the redox regulation of the antioxidant response in filamentous fungi. Fungal Biol. 2019, 24, 253–262. [Google Scholar] [CrossRef]
- Rhee, S.G. Redox signaling: Hydrogen peroxide as intracellular messenger. Exp. Mol. Med. 1999, 31, 53–59. [Google Scholar] [CrossRef] [Green Version]
- Garrido-Bazan, V.; Jaimes-Arroyo, R.; Sanchez, O.; Lara-Rojas, F.; Aguirre, J. SakA and MpkC Stress MAPKs Show Opposite and Common Functions During Stress Responses and Development in Aspergillus nidulans. Front. Microbiol. 2018, 9, 2581. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lara-Rojas, F.; Sanchez, O.; Kawasaki, L.; Aguirre, J. Aspergillus nidulans transcription factor AtfA interacts with the MAPK SakA to regulate general stress responses, development and spore functions. Mol. Microbiol. 2011, 80, 436–454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carrasco-Navarro, U.; Aguirre, J. H2O2 Induces Major Phosphorylation Changes in Critical Regulators of Signal Transduction, Gene Expression, Metabolism and Developmental Networks in Aspergillus nidulans. J. Fungi 2021, 7, 624. [Google Scholar] [CrossRef] [PubMed]
- Jaimes-Arroyo, R.; Lara-Rojas, F.; Bayram, O.; Valerius, O.; Braus, G.H.; Aguirre, J. The SrkA Kinase Is Part of the SakA Mitogen-Activated Protein Kinase Interactome and Regulates Stress Responses and Development in Aspergillus nidulans. Eukaryot Cell 2015, 14, 495–510. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salazar-Roa, M.; Malumbres, M. Fueling the Cell Division Cycle. Trends Cell Biol. 2017, 27, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Kleele, T.; Rey, T.; Winter, J.; Zaganelli, S.; Mahecic, D.; Perreten Lambert, H.; Ruberto, F.P.; Nemir, M.; Wai, T.; Pedrazzini, T.; et al. Distinct fission signatures predict mitochondrial degradation or biogenesis. Nature 2021, 593, 435–439. [Google Scholar] [CrossRef]
- Friedman, J.R.; Lackner, L.L.; West, M.; DiBenedetto, J.R.; Nunnari, J.; Voeltz, G.K. ER tubules mark sites of mitochondrial division. Science 2011, 334, 358–362. [Google Scholar] [CrossRef] [Green Version]
- Bleazard, W.; McCaffery, J.M.; King, E.J.; Bale, S.; Mozdy, A.; Tieu, Q.; Nunnari, J.; Shaw, J.M. The dynamin-related GTPase Dnm1 regulates mitochondrial fission in yeast. Nat. Cell Biol. 1999, 1, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Kraus, F.; Roy, K.; Pucadyil, T.J.; Ryan, M.T. Function and regulation of the divisome for mitochondrial fission. Nature 2021, 590, 57–66. [Google Scholar] [CrossRef]
- Tabara, L.C.; Morris, J.L.; Prudent, J. The Complex Dance of Organelles during Mitochondrial Division. Trends Cell Biol. 2021, 31, 241–253. [Google Scholar] [CrossRef]
- Wakabayashi, J.; Zhang, Z.Y.; Wakabayashi, N.; Tamura, Y.; Fukaya, M.; Kensler, T.W.; Iijima, M.; Sesaki, H. The dynamin-related GTPase Drp1 is required for embryonic and brain development in mice. J. Cell Biol. 2009, 186, 805–816. [Google Scholar] [CrossRef] [Green Version]
- Garrido-Bazan, V.; Pardo, J.P.; Aguirre, J. DnmA and FisA Mediate Mitochondria and Peroxisome Fission, and Regulate Mitochondrial Function, ROS Production and Development in Aspergillus nidulans. Front. Microbiol. 2020, 11, 837. [Google Scholar] [CrossRef] [PubMed]
- Serasinghe, M.N.; Chipuk, J.E. Mitochondrial Fission in Human Diseases. Handb. Exp. Pharmacol. 2017, 240, 159–188. [Google Scholar] [CrossRef] [PubMed]
- Griffin, E.E.; Graumann, J.; Chan, D.C. The WD40 protein Caf4p is a component of the mitochondrial fission machinery and recruits Dnm1p to mitochondria. J. Cell Biol. 2005, 170, 237–248. [Google Scholar] [CrossRef]
- Tieu, Q.; Nunnari, J. Mdv1p is a WD repeat protein that interacts with the dynamin-related GTPase, Dnm1p, to trigger mitochondrial division. J. Cell Biol. 2000, 151, 353–365. [Google Scholar] [CrossRef] [PubMed]
- Tieu, Q.; Okreglak, V.; Naylor, K.; Nunnari, J. The WD repeat protein, Mdv1p, functions as a molecular adaptor by interacting with Dnm1p and Fis1p during mitochondrial fission. J. Cell Biol. 2002, 158, 445–452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Timberlake, W.E.; Clutterbuck, A.J. Genetic regulation of conidiation. In Aspergillus: 50 Years On; Martinelli, S.D., Kinghorn, J.R., Eds.; Elsevier: Amsterdam, The Netherlands, 1994; Volume 29, pp. 283–427. [Google Scholar]
- Hill, T.W.; Käfer, E. Improved protocols for Aspergillus minimal medium: Trace element and minimal medium stock solution. Fungal Genet. Newsl. 2001, 48, 20–21. [Google Scholar] [CrossRef] [Green Version]
- Yu, J.H.; Hamari, Z.; Han, K.H.; Seo, J.A.; Reyes-Dominguez, Y.; Scazzocchio, C. Double-joint PCR: A PCR-based molecular tool for gene manipulations in filamentous fungi. Fungal Genet. Biol. 2004, 41, 973–981. [Google Scholar] [CrossRef] [PubMed]
- Nayak, T.; Szewczyk, E.; Oakley, C.E.; Osmani, A.; Ukil, L.; Murray, S.L.; Hynes, M.J.; Osmani, S.A.; Oakley, B.R. A versatile and efficient gene-targeting system for Aspergillus nidulans. Genetics 2006, 172, 1557–1566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanchez, O.; Aguirre, J. Efficient transformation of Aspergillus nidulans by electroporation of germinated conidia. Fungal Genet. Newslett. 1996, 43, 48–51. [Google Scholar] [CrossRef] [Green Version]
- Sanchez, O.; Navarro, R.E.; Aguirre, J. Increased transformation frequency and tagging of developmental genes in Aspergillus nidulans by restriction enzyme-mediated integration (REMI). Mol. Gen. Genet. 1998, 258, 89–94. [Google Scholar] [CrossRef]
- Lopez-Fuentes, A.D.; Meizoso-Huesca, A.; Peraza-Reyes, L. An endoplasmic reticulum domain is associated with the polarized growing cells of Podospora anserina hyphae. Fungal Genet. Biol. 2020, 137, 103338. [Google Scholar] [CrossRef] [PubMed]
- Pfanner, N.; Douglas, M.G.; Endo, T.; Hoogenraad, N.J.; Jensen, R.E.; Meijer, M.; Neupert, W.; Schatz, G.; Schmitz, U.K.; Shore, G.C. Uniform nomenclature for the protein transport machinery of the mitochondrial membranes. Trends Biochem. Sci. 1996, 21, 51–52. [Google Scholar] [CrossRef]
- Campiño-Arias, D. La Proteína PxdA en el Transporte y la Función Peroxisomal Durante el Crecimiento y la Diferenciación Celular en el Hongo Aspergillus nidulans. Masters degree Thesis, Universidad Nacional Autónoma de México, México City, Mexico, 2022. [Google Scholar]
- Hernandez-Juarez, C.; Flores-Cruz, R.; Jimenez-Sanchez, A. Fluorescent probe for early mitochondrial voltage dynamics. Chem Commun. 2021, 57, 5526–5529. [Google Scholar] [CrossRef] [PubMed]
- Demine, S.; Renard, P.; Arnould, T. Mitochondrial Uncoupling: A Key Controller of Biological Processes in Physiology and Diseases. Cells 2019, 8, 795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eisner, V.; Csordas, G.; Hajnoczky, G. Interactions between sarco-endoplasmic reticulum and mitochondria in cardiac and skeletal muscle—pivotal roles in Ca2+ and reactive oxygen species signaling. J. Cell Sci. 2013, 126, 2965–2978. [Google Scholar] [CrossRef] [Green Version]
- Lang, A.B.; Peter, A.T.J.; Walter, P.; Kornmann, B. ER-mitochondrial junctions can be bypassed by dominant mutations in the endosomal protein Vps13. J. Cell Biol. 2015, 210, 883–890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Csordas, G.; Weaver, D.; Hajnoczky, G. Endoplasmic Reticulum-Mitochondrial Contactology: Structure and Signaling Functions. Trends Cell Biol. 2018, 28, 523–540. [Google Scholar] [CrossRef] [PubMed]
- Kornmann, B.; Currie, E.; Collins, S.R.; Schuldiner, M.; Nunnari, J.; Weissman, J.S.; Walter, P. An ER-mitochondria tethering complex revealed by a synthetic biology screen. Science 2009, 325, 477–481. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koch, K.V.; Suelmann, R.; Fischer, R. Deletion of mdmB impairs mitochondrial distribution and morphology in Aspergillus nidulans. Cell Motil. Cytoskelet. 2003, 55, 114–124. [Google Scholar] [CrossRef]
- Lee, H.; Yoon, Y. Transient Contraction of Mitochondria Induces Depolarization through the Inner Membrane Dynamin OPA1 Protein. J. Biol. Chem 2014, 289, 11862–11872. [Google Scholar] [CrossRef] [Green Version]
- Wolf, D.M.; Segawa, M.; Kondadi, A.K.; Anand, R.; Bailey, S.T.; Reichert, A.S.; van der Bliek, A.M.; Shackelford, D.B.; Liesa, M.; Shirihai, O.S. Individual cristae within the same mitochondrion display different membrane potentials and are functionally independent. Embo. J. 2019, 38, e101056. [Google Scholar] [CrossRef] [PubMed]
- Cho, B.; Cho, H.M.; Jo, Y.; Kim, H.D.; Song, M.; Moon, C.; Kim, H.; Kim, K.; Sesaki, H.; Rhyu, I.J.; et al. Constriction of the mitochondrial inner compartment is a priming event for mitochondrial division. Nat. Commun. 2017, 8, 15754. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hernández-Becerril, A. Evaluación de NoxR y RacA Como Posibles Reguladores de la Actividad de NADPH Oxidasa NoxA de Aspergillus nidulans. Masters degree Thesis, Universidad Nacional Autónoma de México, México city, Mexico, 2010. [Google Scholar]
- Geissel, B.; Penka, M.; Neubauer, M.; Wagener, J. The ER-mitochondria encounter structure contributes to hyphal growth, mitochondrial morphology and virulence of the pathogenic mold Aspergillus fumigatus. Int. J. Med. Microbiol. 2017, 307, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Lang, A.; Peter, A.T.J.; Kornmann, B. ER-mitochondria contact sites in yeast: Beyond the myths of ERMES. Curr. Opin. Cell Biol. 2015, 35, 7–12. [Google Scholar] [CrossRef]
- Debattisti, V.; Gerencser, A.A.; Saotome, M.; Das, S.; Hajnoczky, G. ROS Control Mitochondrial Motility through p38 and the Motor Adaptor Miro/Trak. Cell Rep. 2017, 21, 1667–1680. [Google Scholar] [CrossRef] [Green Version]
- Hung, C.H.L.; Cheng, S.S.Y.; Cheung, Y.T.; Wuwongse, S.; Zhang, N.Q.; Ho, Y.S.; Lee, S.M.Y.; Chang, R.C.C. A reciprocal relationship between reactive oxygen species and mitochondrial dynamics in neurodegeneration. Redox. Biol. 2018, 14, 7–19. [Google Scholar] [CrossRef] [PubMed]
- Miyazono, Y.; Hirashima, S.; Ishihara, N.; Kusukawa, J.; Nakamura, K.; Ohta, K. Uncoupled mitochondria quickly shorten along their long axis to form indented spheroids, instead of rings, in a fission-independent manner. Sci. Rep. 2018, 8, 350. [Google Scholar] [CrossRef] [Green Version]
- Aklima, J.; Onojima, T.; Kimura, S.; Umiuchi, K.; Shibata, T.; Kuraoka, Y.; Oie, Y.; Suganuma, Y.; Ohta, Y. Effects of Matrix pH on Spontaneous Transient Depolarization and Reactive Oxygen Species Production in Mitochondria. Front. Cell Dev. Biol. 2021, 9, 1582. [Google Scholar] [CrossRef] [PubMed]
- Bonora, M.; Giorgi, C.; Pinton, P. Molecular mechanisms and consequences of mitochondrial permeability transition. Nat. Rev. Mol. Cell Bio 2022, 23, 266–285. [Google Scholar] [CrossRef]
- Booth, D.M.; Varnai, P.; Joseph, S.K.; Hajnoczky, G. Oxidative bursts of single mitochondria mediate retrograde signaling toward the ER. Mol. Cell 2021, 81, 3866–3876.e2. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.S.; Chakrabarti, R.; Kollasser, J.; Rottner, K.; Stradal, T.E.B.; Kage, F.; Higgs, H.N. Parallel kinase pathways stimulate actin polymerization at depolarized mitochondria. Curr. Biol. 2022, 32, 1577–1592.e8. [Google Scholar] [CrossRef] [PubMed]
- Mattie, S.; Krols, M.; McBride, H.M. The enigma of an interconnected mitochondrial reticulum: New insights into mitochondrial fusion. Curr. Opin. Cell Biol. 2019, 59, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Mottie, S.; Riemer, J.; Wideman, J.G.; McBride, H.M. A new mitofusin topology places the redox-regulated C terminus in the mitochondrial intermembrane space. J. Cell Biol. 2018, 217, 507–515. [Google Scholar] [CrossRef] [Green Version]
- Shutt, T.; Geoffrion, M.; Milne, R.; McBride, H.M. The intracellular redox state is a core determinant of mitochondrial fusion. Embo. Rep. 2012, 13, 909–915. [Google Scholar] [CrossRef] [Green Version]
- Kuroiwa, T.; Nishida, K.; Yoshida, Y.; Fujiwara, T.; Mori, T.; Kuroiwa, H.; Misumi, O. Structure, function and evolution of the mitochondrial division apparatus. BBA Mol. Cell Res. 2006, 1763, 510–521. [Google Scholar] [CrossRef] [Green Version]










Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garrido-Bazán, V.; Aguirre, J. H2O2 Induces Calcium and ERMES Complex-Dependent Mitochondrial Constriction and Division as Well as Mitochondrial Outer Membrane Remodeling in Aspergillus nidulans. J. Fungi 2022, 8, 829. https://doi.org/10.3390/jof8080829
Garrido-Bazán V, Aguirre J. H2O2 Induces Calcium and ERMES Complex-Dependent Mitochondrial Constriction and Division as Well as Mitochondrial Outer Membrane Remodeling in Aspergillus nidulans. Journal of Fungi. 2022; 8(8):829. https://doi.org/10.3390/jof8080829
Chicago/Turabian StyleGarrido-Bazán, Verónica, and Jesús Aguirre. 2022. "H2O2 Induces Calcium and ERMES Complex-Dependent Mitochondrial Constriction and Division as Well as Mitochondrial Outer Membrane Remodeling in Aspergillus nidulans" Journal of Fungi 8, no. 8: 829. https://doi.org/10.3390/jof8080829
APA StyleGarrido-Bazán, V., & Aguirre, J. (2022). H2O2 Induces Calcium and ERMES Complex-Dependent Mitochondrial Constriction and Division as Well as Mitochondrial Outer Membrane Remodeling in Aspergillus nidulans. Journal of Fungi, 8(8), 829. https://doi.org/10.3390/jof8080829





