Lobomycosis Epidemiology and Management: The Quest for a Cure for the Most Neglected of Neglected Tropical Diseases
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Case Reports, New Cases, and the Hidden Prevalence of Lobomycosis
3.2. Lobomycosis in Dolphins and Zoonotic Potential
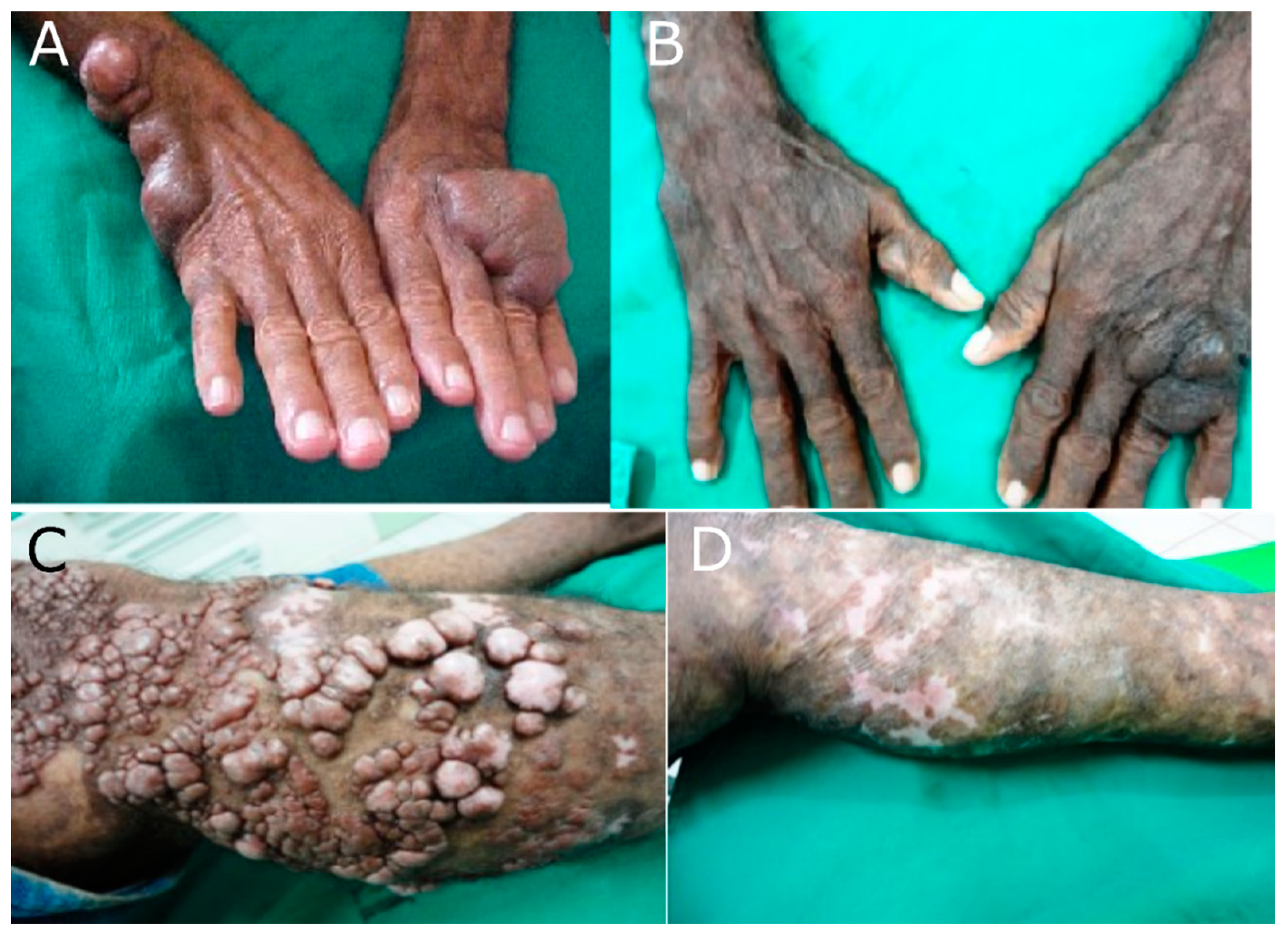
3.3. Clinical Presentation and Diagnosis of Lobomycosis
3.4. The Quest for a Cure for Lobomycosis
| Drug | Dosing (Daily) * | Duration @ | n Patient | Follow-Up Time % | Outcome # | Surgery Used | Side Effects $ | % of cure | Ref |
|---|---|---|---|---|---|---|---|---|---|
| Sulfadimethoxine | 0.5–2 g | 11 d | 1 | Not done | 2 | No | No | - | [64] |
| Sulfadimethoxine | 0.25–0.5 g | 18 d | 2 | Not done | 1 | No | No | - | [65] |
| Ketoconazole | 0.2–0.4 g | 90 d | 1 | Not done | 2B | No | No | - | [66] |
| Ketoconazole | 0.2 g | 180 d | 1 | Not done | 1 | No | No | - | [67] |
| Clofazimine Itraconazole | 0.1 g 0.1 g | 1 y | 1 | 2 years | 3C | No | Yes1 | - | [68] |
| Clofazimine Dapsone Itraconazole | 0.05 g 0.1 g 0.2 g | 1 y | 1 | Not available | 3C | Yes | No | - | [46] |
| Posaconazole | 0.8 g | 27 m | 1 | 4 years | 3B | No | Yes 2 | - | [70] |
| Itraconazole Clofazimine Cryotherapy with liquid nitrogen | 0.2 g 0.1 g every 3 months | 2 y | 1 | Not available | 3C | Yes | No | - | [34] |
| Clofazimine Dapsone Clofazimine Rifampin Dapsone Itraconazole | 0.05 g 0.1 g 0.3 g/m 0.6 g/m 0.3 g/m 0.2 g | 4 y | 103 | 2 years | 3C | Yes | Yes 1 | 25% | [71] |
| Posaconazole | 0.8 g | 30 m | 1 | Not available | 2 | No | No | - | [72] |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- de Brito, A.C.; Quaresma, J.A.S. Lacaziose (doença de Jorge Lobo): Revisão e atualização. An. Bras. Dermatol. 2007, 82, 461–474. [Google Scholar] [CrossRef] [Green Version]
- Lacaz, C.S.; Baruzzi, R.G.; Rosa, M.C.B. Doença de Jorge Lobo; EDUSP: São Paulo, Brazil, 1986; pp. 1–92. [Google Scholar]
- Lobo, J. Um caso de blastomicose, produzido por uma espécie nova, encontrada em Recife. Rev. Med. Pernamb. 1931, 1, 763–775. [Google Scholar]
- Guedes, A.C.M. Pele e anexos. In Bogliolo Patologia, 7th ed.; Brasileiro Filho, G., Ed.; Guanabara Koogan: Rio de Janeiro, Brazil, 2006; Volume 1, pp. 1200–1287. [Google Scholar]
- Gompertz, O.F.; Gambale, W.; Paula, C.R.; Correa, B. Micoses subcutâneas. In Microbiologia, 5th ed.; Trabulsi, L.R., Alterthum, F., Eds.; Atheneu: São Paulo, Brazil, 2008; pp. 511–515. [Google Scholar]
- Symeres, W.S. A possible case of Lobo’s diseases acquired in Europe from a bottle-nosed dolphin (Tursiops truncates). Bull. Société Pathol. Exot. Fil. 1983, 76, 777–784. [Google Scholar]
- Simões-Lopes, P.C.; Paula, G.S. First case of lobomycosis in a bottlenose dolphin from southern Brazil. Mar. Mamm. Sci. 1993, 9, 329–331. [Google Scholar] [CrossRef]
- Silva, A.R.; Pinheiro, G.S.; Matos, W.B.; Couto, M.J.A.; Gonçalves, E.G.R. Doença de Jorge Lobo: Primeiro caso registrado no Estado do Maranhão. Cad. Pesqui. 2013, 20, 64–67. [Google Scholar] [CrossRef] [Green Version]
- Francesconi, V.A.; Klein, A.P.; Santos, A.P.B.G.; Ramasawmy, R.; Francesconi, F. Lobomycosis: Epidemiology, clinical presentation, and management options. Ther. Clin. Risk Manag. 2014, 10, 851–860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woods, W.J.; Belone, A.F.; Carneiro, L.B.; Rosa, P.S. Ten years’ experience with Jorge Lobo’s disease in the state of Acre, Amazon region, Brazil. Rev. Inst. Med. Trop. São Paulo 2010, 52, 273–278. [Google Scholar] [CrossRef] [Green Version]
- Floriano, M.C. Doença de Jorge Lobo Entre os Índios Caiabu: Epidemiologia, Análise Histopatológica e Imuno-Histoquímica e Imuno-Histoquímica Nas Diferentes Apresentações Clínicas. Ph.D. Thesis, Universidade Federal de São Paulo, São Paulo, Brazil, 2014. [Google Scholar]
- Borelli, D. The reservoir area of lobomycosis. Comments on the work of Dr Carlos Peña on 2 Colombian cases. Mycopathol. Mycol. Appl. 1969, 37, 145–149. [Google Scholar] [CrossRef]
- Guimaraes, F.N.; Macedo, D.G. Blastomycosis in the Amazon Valley (keloidian and South American). Hosp. (Rio J.) 1950, 38, 223–253. [Google Scholar]
- Rodriquez-Toro, G. Lobomycosis. Int. J. Dermatol. 1993, 32, 324–332. [Google Scholar] [CrossRef]
- Ramos-E-Silva, M.; Aguiar-Santos-Vilela, F.; Cardoso-de-Brito, A.; Coelho-Carneiro, S. Lobomycosis. Literature review and future perspectives. Actas Dermo-Sifiliogr. 2009, 100 (Suppl. 1), 92–100. [Google Scholar] [CrossRef]
- Lupi, O.; Tyring, S.K.; McGinnis, M.R. Tropical dermatology: Fungal tropical diseases. J. Am. Acad. Dermatol. 2005, 53, 931–951. [Google Scholar] [CrossRef] [PubMed]
- Paniz-Mondolfi, A.E.; Jaimes, O.R.; Jones, L.D. Lobomycosis in Venezuela. Int. J. Dermatol. 2007, 46, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Saint-Blancard, P.; Maccari, F.; Le Guyadec, T.; Lanternier, G.; Le Vagueresse, R. Lobomycosis: A mycosis seldom observed in metropolitan France. Ann. Pathol. 2000, 20, 241–244. [Google Scholar]
- Beltrame, A.; Danesi, P.; Farina, C.; Orza, P.; Perandin, F.; Zanardello, C.; Rodari, P.; Staffolani, S.; Bisoffi, Z. Molecular confirmation of lobomycosis in an Italian traveler acquires in the Amazon Region of Venezuela. Am. J. Trop. Med. Hyg. 2017, 97, 1757–1760. [Google Scholar] [CrossRef] [Green Version]
- Fialho, A.S.; Lacaz, C.S. Jorge Lobo blastomycosis. Folha Med. 1950, 31, 36–40. [Google Scholar]
- Burns, R.A.; Roy, J.S.; Woods, C.; Padhye, A.A.; Warnock, D.W. Report of the first human case of Lobomycosis in United States. J. Clin. Microb. 2000, 38, 1283–1285. [Google Scholar] [CrossRef] [Green Version]
- Elsayed, S.; Kuhn, S.M.; Barber, S.; Church, D.L.; Adams, S.; Kasper, R. Human case of lobomycosis. Emerg. Infect. Dis. 2004, 10, 715–718. [Google Scholar] [CrossRef]
- Al-Daraji, W.I.; Husain, E.; Robson, A. Lobomycosis in African patients. Br. J. Dermatol. 2008, 159, 231–266. [Google Scholar] [CrossRef]
- Nery-Guimarães, F. Inoculações em hamsters da blastomicose sul-americana (doença de Lutz), da blastomicose queloidiforme (doença de Lobo) e da blastomicose dos índios do Tapajós-Xingu. Hosp. (Rio J.) 1964, 66, 581–593. [Google Scholar]
- Baruzzi, R.G.; D’Andretta, C., Jr.; Carvalhal, S.; Ramos, O.L.; Pontes, P.L. Ocorrência de blastomicose queloidiana entre índios Caiabi. Rev. Inst. Med. Trop. São Paulo 1967, 9, 135–142. [Google Scholar] [PubMed]
- Machado, P.A.; Silveira, D.F. Piraip, a falsa lepra dos Caiabis. Rev. Bras. Leprol. 1966, 34, 60. [Google Scholar]
- Baruzzi, R.G.; Castro, R.M.; D’Andretta, C., Jr.; Carvalhal, S.; Ramos, O.L.; Pontes, P.L. Occurrence of Lobo’s blastomycosis among “Caiabi” brazilian Indians. Int. J. Dermatol. 1973, 12, 95–98. [Google Scholar] [CrossRef]
- Baruzzi, R.G.; Marcopito, L.F.; Vicente, L.S.; Michalany, N.S. Jorge Lobo’s disease (keloidal blastomycosis) and tinea imbricata in Indians from the Xingu National Park, Central Brazil. Trop. Dr. 1982, 12, 13–15. [Google Scholar] [CrossRef] [PubMed]
- Baruzzi, R.G.; Rodrigues, D.A.; Michalany, N.S.; Salomão, R. Squamous-cell carcinoma and lobomycosis (Jorge Lobo’s disease). Int. J. Dermatol. 1989, 28, 183–185. [Google Scholar] [CrossRef]
- Baruzzi, R.G.; Marcopito, L.F. Doença de Jorge Lobo. In Tratado de Infectologia; Veronesi, R., Focaccia, R., Eds.; Atheneu: São Paulo, Brazil, 1996; pp. 1116–1119. [Google Scholar]
- Pradinaud, R. Loboa loboi. In Topley and Wilson’s Microbiology and Microbial Infections; Collier, L., Balows, A., Sussman, M., Eds.; Oxford University Press: New York, NY, USA, 1998; pp. 585–594. [Google Scholar]
- Opromolla, D.V.A.; Taborda, P.R.O.; Taborda, V.B.A.; Viana, S.; Furtado, J.F. Lobomicose: Relato de 40 casos novos. An. Bras. Dermatol. 1999, 74, 135–141. [Google Scholar]
- Lacaz, C.S.; Porto, E.; Martins, J.E.C.; Heins-Vaccari, E.M.; Melo, N.T. Tratado de Micologia Médica, 9th ed.; Savier: São Paulo, Brazil, 2002; pp. 462–478. [Google Scholar]
- Araújo, M.G.; Santos, S.N.M.B.; Guedes, A.C.M.; Cirilo, N.S.; Aguiar, C.R. Lobomycosis: A therapeutic challenge. An. Bras. Dermatol. 2018, 93, 279–281. [Google Scholar] [CrossRef]
- Arenas, C.M.; Rodriguez-Toro, G.; Ortiz-Florez, A.; Serrato, I. Lobomycosis in soldiers, Colombia. Emerg. Infect. Dis. 2019, 25, 654–660. [Google Scholar] [CrossRef]
- Pech-Ortiz, L.; Maya-Aranda, S.; Hernández-Castro, R.; Xicohtencatl-Cortes, J.; Tirado-Sánchez, A.; Bonifaz, A. Lacaziosis (lobomycosis) from Southern Mexico: A case confirmed by molecular biology. Mycopathologia 2020, 185, 737–739. [Google Scholar] [CrossRef]
- Papadavid, E.; Dalamaga, M.; Kapniari, I.; Pantelidaki, E.; Papageorgiou, S.; Pappa, V.; Tsirigotis, P.; Dervenoulas, I.; Stavrianeas, N.; Rigopoulos, D. Lobomycosis: A case from Southeastern Europe and review of the literature. J. Dermatol. Case Rep. 2012, 6, 65–69. [Google Scholar] [CrossRef]
- Florian, M.C.; Rodrigues, A.; de Mendonça, S.B.M.; Colombo, A.L.; Tomimori, J. Epidemiologic and clinical progression of lobomycosis among Kaiabi Indians, Brazil, 1965–2019. Emerg. Infect. Dis. 2020, 26, 930–936. [Google Scholar] [CrossRef]
- Oliveira, F.D.S.; Lopes, N.P.; Talhari, C.; Schettini, A. What is your diagnosis? Keloidal cord-like lesion on the leg. An. Bras. Dermatol. 2020, 95, 386–389. [Google Scholar] [CrossRef]
- Gonçalves, F.G. (Serviço Estadual de Dermatologia do Acre, Programa Estadual de Dermatologia do Acre, Programa Estadual de Controle da Hanseníase, SESACRE/FUNDHACRE, Rio Branco, Acre, Brazil). Personal communication, 2022.
- Migaki, G.; Valerio, M.G.; Irvine, B.; Garner, F.M. Lobos disease in an Atlantic bottle-nosed dolphin. J. Am. Vet. Med. Assoc. 1971, 159, 578–582. [Google Scholar]
- Caldwell, D.K.; Caldwell, M.C.; Woodard, J.C.; Ajello, L.; Kaplan, W.; McLure, H.M. Lobomycosis as a disease of the Atlantic bottlenosed dolphin (Tursiops truncatus Montagu, 1821). Am. J. Trop. Med. Hyg. 1975, 24, 105–114. [Google Scholar] [CrossRef]
- Wiersema, J.P.; Niemel, P.L. Lobo’s disease in Surinam patients. Trop. Geogr. Med. 1965, 17, 89–111. [Google Scholar]
- De Vries, G.A.; Laarman, J.J. A case of Lobo’s disease in the dolphin Sotalia guianensis. Aquat. Mamm. 1973, 1, 26–33. [Google Scholar]
- Norton, S.A. Dolphin-to-human transmission of lobomycosis. J. Am. Acad. Dermatol. 2006, 55, 723–724. [Google Scholar] [CrossRef]
- Rosa, P.S.; Soares, C.T.; Belone, A.; Vilela, R.; Ura, S.; Filho, M.C.; Mendoza, L. Accidental Jorge Lobo’s disease in a worker dealing with Lacazia loboi infected mice: A case report. J. Med. Case Rep. 2009, 3, 67. [Google Scholar] [CrossRef] [Green Version]
- Moreno, I.B.; Ott, P.H.; Tavares, M.; Oliveira, L.R.; Borba, M.R.; Driemeier, D.; Nakashima, S.B.; Heinzelmann, L.S.; Siciliano, S.; Van Bressen, M.F. Mycotic dermatitis in common bottlenose Dolphins (Tursiops truncatus) from Southern Brazil, with confirmed record of lobomycosis disease. In Proceedings of the Annual Meeting of the International Whaling Commission (IWC), IWC Scientific Committee, Santiago, Chile, 23–26 June 2008. Paper SC/60/DW1. [Google Scholar]
- Paniz-Mondolfi, A.E.; Sander-Hoffmann, L. Lobomycosis in inshore and estuarine dolphins. Emerg. Infect. Dis. 2009, 15, 672–673. [Google Scholar] [CrossRef]
- Reif, J.S.; Mazzoil, M.S.; McCulloch, S.D.; Varela, R.A.; Goldstein, J.D.; Fair, P.A.; Bossart, G.D. Lobomycosis in Atlantic bottlenose dolphins from the Indian River Lagoon, Florida. J. Am. Vet. Med. Assoc. 2006, 228, 104–108. [Google Scholar] [CrossRef]
- Murdoch, M.E.; Reif, J.S.; Mazzoil, M.; McCulloch, S.D.; Fair, P.A.; Bossart, G.D. Lobomycosis in bottlenose dolphins (Tursiops truncatus) from the Indian River Lagoon, Florida: Estimation of prevalence, temporal trends, and spatial distribution. EcoHealth 2008, 5, 289–297. [Google Scholar] [CrossRef]
- Durden, W.N.; St Leger, J.; Stolen, M.; Mazza, T.; Londono, C. Lacaziosis in bottlenose dolphins (Tursiops truncatus) in the Indian River Lagoon, Florida, USA. J. Wildl. Dis. 2009, 45, 849–856. [Google Scholar] [CrossRef] [Green Version]
- Bressem, M.F.V.; Santos, M.C.O.; Oshima, J.E.F. Skin diseases in Guiana dolphins (Sotalia guianensis) from the Paranaguá estuary, Brazil: A possible indicator of a compromised marine environment. Mar. Environ. Res. 2009, 67, 63–68. [Google Scholar] [CrossRef]
- Kiszka, J.; Van Bressem, M.F.; Pusineri, C. Lobomycosis-like disease and other skin conditions in Indo-Pacific bottlenose dolphins Tursiops aduncus from the Indian Ocean. Dis. Aquat. Org. 2009, 84, 151–157. [Google Scholar] [CrossRef] [Green Version]
- Paniz-Mondolfi, A.; Talhari, C.; Hoffman, L.S.; Connor, D.L.; Talhari, S.; Bermudez-Villapol, L.; Hernadez-Perez, M.; Van Bressem, M.F. Lobomycosis: An emerging disease in humans and delphinidae. Mycoses 2012, 55, 298–309. [Google Scholar] [CrossRef]
- Brito, A.C. Lacaziose (Doença de Jorge Lobo). In Compêndio de Micologia Médica, 2nd ed.; Zaitz, C., Campbell, I., Marques, A.S., Ruiz, L.R.B., Framil, V.M.S., Eds.; Editora Guanabara Koogan: Rio de Janeiro, Brazil, 2012; pp. 219–231. [Google Scholar]
- Bessesen, B.L.; Oviedo, L.; Hart, L.B.; Herra-Miranda, D.; Pacheco-Polanco, J.D.; Baker, L.; Saborío-Rodriguez, G.; Bermúdez-Villapol, L.; Aceved-Gutiérrez, A. Lacaziosis-like disease among bottlenose dolphins Tursiops truncatus photographed in Golf Dulce, Costa Rica. Dis. Aquat. Org. 2014, 107, 173–180. [Google Scholar] [CrossRef]
- Ramos, E.A.; Castelblanco-Martínez, D.N.; Garcia, J.; Arias, J.R.; Foley, J.R.; Audley, K.; Van Waerebeek, K.; Van Bressem, M.F. Lobomycosis-like disease in common bottlenose dolphins Tursiops truncatus from Belize and Mexico: Bridging the gap between the Americas. Dis. Aquat. Org. 2018, 128, 1–12. [Google Scholar] [CrossRef]
- Rotstein, D.S.; Burdett, L.G.; McLellan, W.; Schwacke, L.; Rowles, T.; Terio, K.A.; Schultz, S.; Pabst, A. Lobomycosis in offshore bottlenose dolphins (Tursiops truncatus), North Carolina. Emerg. Infect. Dis. 2009, 15, 588–590. [Google Scholar] [CrossRef]
- Pradinaud, R. Entre le Yucatan, la Floride et la Guyane Française, la lobomycose existe-t-elle aux Antilles. Bull. Société Pathol. Exot. 1984, 77, 392–400. [Google Scholar]
- Vilela, R.; Mendoza, L.; Rosa, P.S.; Belone, A.F.; Madeira, S.; Opromolla, D.V.; Resende, M.A. Molecular model for studying the uncultivated fungal pathogen Lacazia loboi. J. Clin. Microbiol. 2005, 43, 3657–3661. [Google Scholar] [CrossRef] [Green Version]
- Nogueira, L.; Mendes, L.; Rodrigues, C.A.; Santos, M.; Talhari, S.; Talhari, C. Lobomycosis and squamous cell carcinoma. An. Bras. Dermatol. 2013, 88, 293–295. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.F.R.; Silva, A.J.G. Vinyl adhesive tape effective for direct microscopy diagnosis of chromomycosis, lobomycosis, and paracoccidiodomycosis. Diagn. Microbiol. Infect. Dis. 2005, 52, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Opromolla, D.V.A.; Belone, A.F.F.; Taborda, P.R.O.; Taborda, V.B.A. Correlação clínico-patológica em 40 casos novos de lobomicose. An. Bras. Dermatol. 2000, 75, 425–434. [Google Scholar]
- Reyes, O.; Goihman, M.; Goldstein, C. Blastomicosis queloidiana o enfermidad de Jorge Lobo. Dermatol. Venez. 1960, 2, 245–255. [Google Scholar]
- Silverie, R.; Ravisse, P.; Vilar, J.P.; Moulins, C. La blastomycose chéloidienne ou maladie de Jorge Lobo en Guyane Française. Bull. Société Pathol. Exot. 1963, 56, 29–35. [Google Scholar]
- Cucê, L.C.; Wroclawski, E.L.; Sampaio, S.A. Treatment of paracoccidioidomycosis, candidiasis, chromomycosis, lobomycosis and mycetoma with ketoconazole. Int. J. Dermatol. 1980, 19, 405–408. [Google Scholar] [CrossRef]
- Lawrence, D.N.; Ajello, L. Lobomycosis in Western Brazil: Report of a clinical trial with ketoconazole. Am. J. Trop. Med. Hyg. 1986, 35, 162–166. [Google Scholar] [CrossRef]
- Fischer, M.; Chrusciak-Talhari, A.; Reinel, D.; Talhari, S. Successful treatment with clofazimine and itraconazole in a 46-year-old patient after 32 years duration of disease. Hautarzt 2002, 53, 677–681. [Google Scholar] [CrossRef]
- Talhari, S.; Talhari, C. Lobomycosis. Clin. Dermatol. 2012, 30, 420–424. [Google Scholar] [CrossRef]
- Bustamante, B.; Seas, C.; Salomon, M.; Bravo, F. Lobomycosis successfully treated with posaconazole. Am. J. Trop. Med. Hyg. 2013, 88, 1207–1208. [Google Scholar] [CrossRef] [Green Version]
- Gonçalves, F.G.; Rosa, P.S.; Belone, A.F.F.; Carneiro, L.B.; Barros, V.L.Q.; Bispo, R.F.; Sbardellot, Y.A.S.; Neves, S.A.V.M.; Vittor, A.Y.; Woods, W.J.; et al. Multidrug therapy for leprosy can cure patientes with lobomycosis in Acre State, Brazil: A proof of therapy study. Am. J. Trop. Med. Hyg. 2021, 104, 634–639. [Google Scholar] [CrossRef] [PubMed]
- Pasqualotto, A.C.; Jaskulski Filho, S.D.; Sena, M.G.; Santos, A.; Severo, M. Posaconazole for lobomycosis. Braz. J. Infect. Dis. 2021, 25, 101576. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonçalves, F.G.; Rosa, P.S.; Belone, A.d.F.F.; Carneiro, L.B.; de Barros, V.L.Q.; Bispo, R.F.; Sbardelott, Y.A.d.S.; Neves, S.A.V.M.; Vittor, A.Y.; Woods, W.J.; et al. Lobomycosis Epidemiology and Management: The Quest for a Cure for the Most Neglected of Neglected Tropical Diseases. J. Fungi 2022, 8, 494. https://doi.org/10.3390/jof8050494
Gonçalves FG, Rosa PS, Belone AdFF, Carneiro LB, de Barros VLQ, Bispo RF, Sbardelott YAdS, Neves SAVM, Vittor AY, Woods WJ, et al. Lobomycosis Epidemiology and Management: The Quest for a Cure for the Most Neglected of Neglected Tropical Diseases. Journal of Fungi. 2022; 8(5):494. https://doi.org/10.3390/jof8050494
Chicago/Turabian StyleGonçalves, Franciely G., Patrícia S. Rosa, Andrea de F. F. Belone, Léia B. Carneiro, Vania L. Q. de Barros, Rosineide F. Bispo, Yally A. da S. Sbardelott, Sebastião A. V. M. Neves, Amy Y. Vittor, William J. Woods, and et al. 2022. "Lobomycosis Epidemiology and Management: The Quest for a Cure for the Most Neglected of Neglected Tropical Diseases" Journal of Fungi 8, no. 5: 494. https://doi.org/10.3390/jof8050494
APA StyleGonçalves, F. G., Rosa, P. S., Belone, A. d. F. F., Carneiro, L. B., de Barros, V. L. Q., Bispo, R. F., Sbardelott, Y. A. d. S., Neves, S. A. V. M., Vittor, A. Y., Woods, W. J., & Laporta, G. Z. (2022). Lobomycosis Epidemiology and Management: The Quest for a Cure for the Most Neglected of Neglected Tropical Diseases. Journal of Fungi, 8(5), 494. https://doi.org/10.3390/jof8050494







