Treating (Recurrent) Vulvovaginal Candidiasis with Medical-Grade Honey—Concepts and Practical Considerations
Abstract
1. Introduction
2. Diagnosis of RVVC
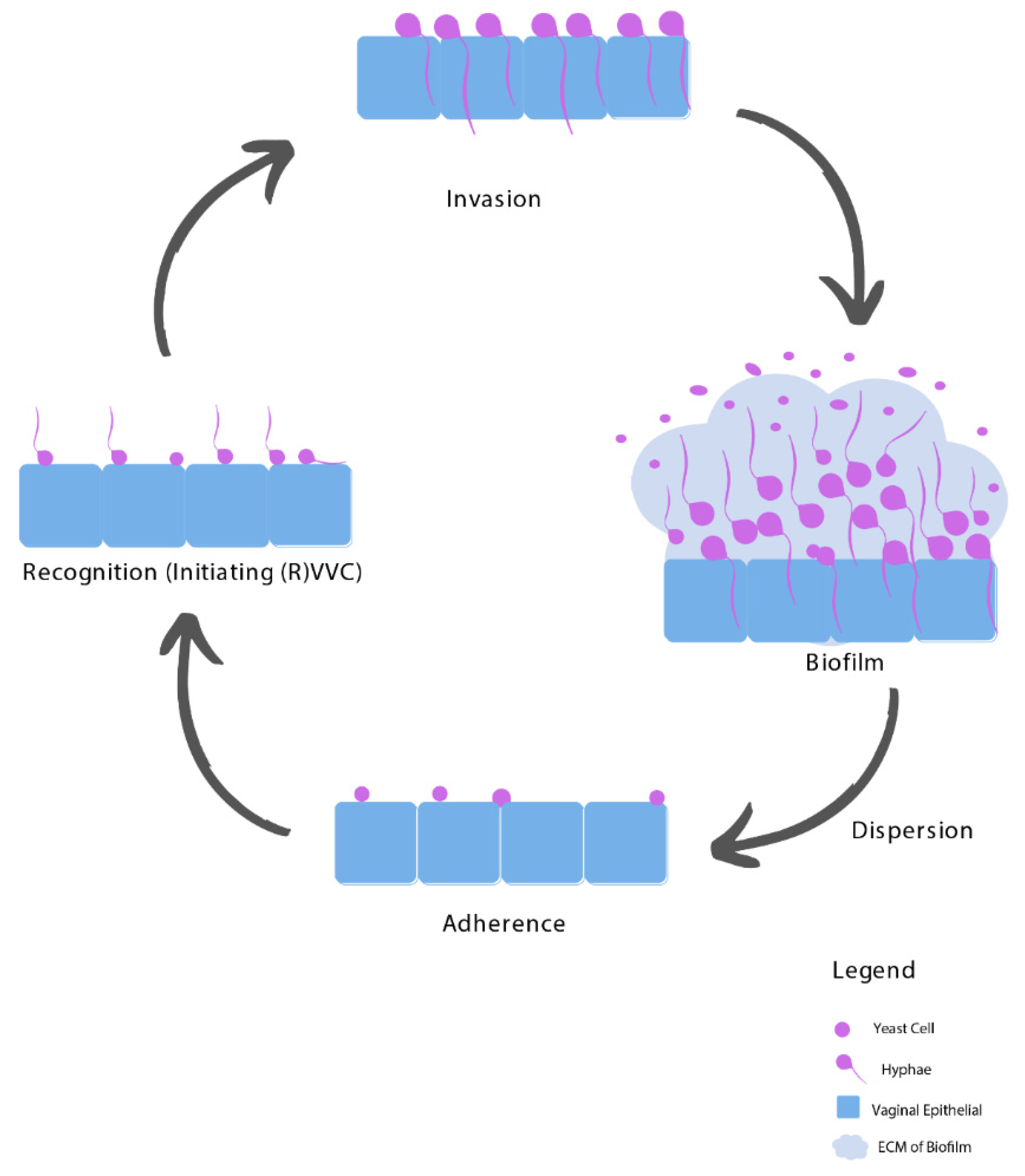
3. Pathogenesis of RVVC
3.1. Adhesion
3.2. Recognition
3.3. Invasion
3.4. Biofilms
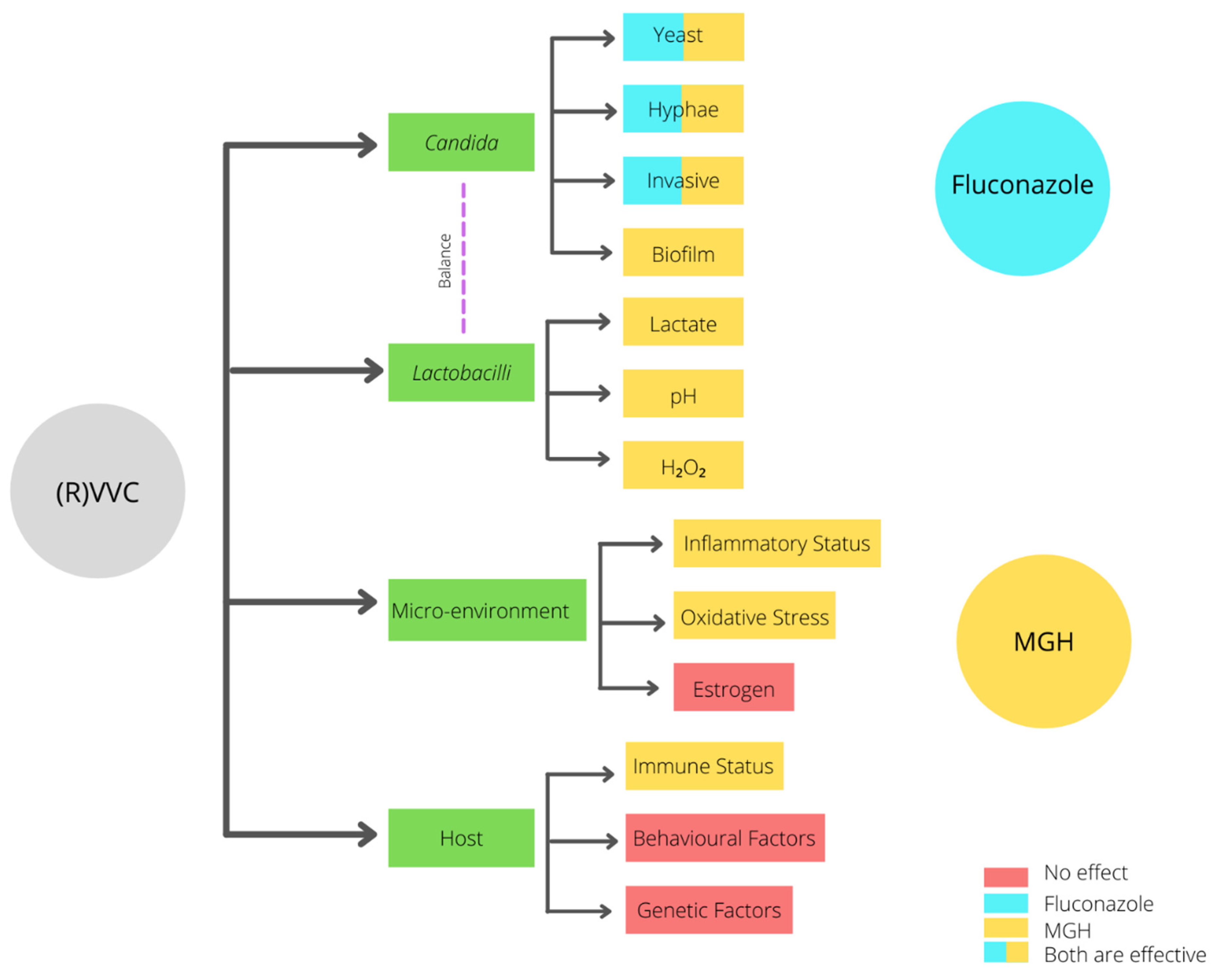
4. Risk Factors of RVVC
4.1. Imbalanced Vaginal Microbiota Composition
4.2. Host-Related Predisposing Factors
4.3. Idiopathic RVVC
5. Treatment of RVVC and Its Efficacy
5.1. Resistance towards Fluconazole
5.2. Unnecessary and Inappropriate Use of Fluconazole
5.3. Non-Albicans Species
5.4. Biofilms Complicate RVVC Treatment
6. Medical-Grade Honey as an Alternative Treatment Option
6.1. The Antimicrobial Activity of MGH against Candida Species
6.2. MGH Resolves Non-Albicans Candida Species
6.3. The Effect of MGH on Biofilms
6.4. Lactobacilli Are Not Affected by MGH
6.5. MGH Modulates the Vaginal Microenvironment
6.6. Effect of MGH on the Immune Status
6.7. Clinical Studies Supporting a Role for MGH in the Treatment of (R)VVC
6.8. Considerations for MGH Application
7. Study Design of a New Prospective Randomized Controlled Trial
8. Rationale for Selecting the MGH-Based Formulation
9. Conclusions
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
References
- Sobel, J.D. Recurrent vulvovaginal candidiasis. Am. J. Obstet. Gynecol. 2016, 214, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Denning, D.W.; Kneale, M.; Sobel, J.D.; Rautemaa-Richardson, R. Global burden of recurrent vulvovaginal candidiasis: A systematic review. Lancet Infect. Dis. 2018, 18, e339–e347. [Google Scholar] [CrossRef]
- Sobel, J.D.; Wiesenfeld, H.C.; Martens, M.; Danna, P.; Hooton, T.M.; Rompalo, A.; Sperling, M.; Livengood, C., 3rd; Horowitz, B.; Von Thron, J.; et al. Maintenance fluconazole therapy for recurrent vulvovaginal candidiasis. N. Engl. J. Med. 2004, 351, 876–883. [Google Scholar] [CrossRef]
- Rosati, D.; Bruno, M.; Jaeger, M.; Ten Oever, J.; Netea, M.G. Recurrent Vulvovaginal Candidiasis: An Immunological Perspective. Microorganisms 2020, 8, 144. [Google Scholar] [CrossRef] [PubMed]
- Pappas, P.G.; Kauffman, C.A.; Andes, D.R.; Clancy, C.J.; Marr, K.A.; Ostrosky-Zeichner, L.; Reboli, A.C.; Schuster, M.G.; Vazquez, J.A.; Walsh, T.J.; et al. Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2016, 62, e1–e50. [Google Scholar] [CrossRef]
- Bauters, T.G.; Dhont, M.A.; Temmerman, M.I.; Nelis, H.J. Prevalence of vulvovaginal candidiasis and susceptibility to fluconazole in women. Am. J. Obstet. Gynecol. 2002, 187, 569–574. [Google Scholar] [CrossRef]
- Banaeian-Borujeni, S.; Mobini, G.R.; Pourgheysari, B.; Validi, M. Comparison of the effect of honey and miconazole against Candida albicans in vitro. Adv. Biomed. Res. 2013, 2, 57. [Google Scholar] [CrossRef]
- Gardella, C.E.L.; Lentz, G.M. Comprehensive Gynecology; Lentz, G.L.R., Gershenson, D., Valea, F.A., Eds.; Elsevier: Philadelphia, PA, USA, 2012; Volume 7, pp. 524–565. [Google Scholar]
- Workowski, K.A.; Bolan, G.A. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm. Rep. Morb. Mortal. Wkly. Rep. Recomm. Rep. 2015, 64, 1–137. [Google Scholar]
- Sobel, J.D.; Faro, S.; Force, R.W.; Foxman, B.; Ledger, W.J.; Nyirjesy, P.R.; Reed, B.D.; Summers, P.R. Vulvovaginal candidiasis: Epidemiologic, diagnostic, and therapeutic considerations. Am. J. Obstet. Gynecol. 1998, 178, 203–211. [Google Scholar] [CrossRef]
- Lema, V.M. Recurrent Vulvo-Vaginal Candidiasis: Diagnostic and Management Challenges in a Developing Country Context. Obstet. Gynecol. Int. J. 2017, 7, 260. [Google Scholar] [CrossRef]
- Schoch, C.L.; Seifert, K.A.; Huhndorf, S.; Robert, V.; Spouge, J.L.; Levesque, C.A.; Chen, W.; Fungal Barcoding, C.; Fungal Barcoding Consortium. Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc. Natl. Acad. Sci. USA 2012, 109, 6241–6246. [Google Scholar] [CrossRef] [PubMed]
- Patel, R. A Moldy Application of MALDI: MALDI-ToF Mass Spectrometry for Fungal Identification. J. Fungi 2019, 5, 4. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, M.A.; Sobel, J.D. Vulvovaginal Candidiasis Caused by Non-albicans Candida Species: New Insights. Curr. Infect. Dis. Rep. 2010, 12, 465–470. [Google Scholar] [CrossRef]
- Makanjuola, O.; Bongomin, F.; Fayemiwo, S.A. An Update on the Roles of Non-albicans Candida Species in Vulvovaginitis. J. Fungi 2018, 4, 121. [Google Scholar] [CrossRef]
- Donders, G.G.; Sobel, J.D. Candida vulvovaginitis: A store with a buttery and a show window. Mycoses 2017, 60, 70–72. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.P.; Willems, H.M.E.; Moyes, D.L.; Shoaie, S.; Barker, K.S.; Tan, S.L.; Palmer, G.E.; Hube, B.; Naglik, J.R.; Peters, B.M. Candidalysin Drives Epithelial Signaling, Neutrophil Recruitment, and Immunopathology at the Vaginal Mucosa. Infect. Immun. 2018, 86, e00645-17. [Google Scholar] [CrossRef] [PubMed]
- Ardizzoni, A.; Wheeler, R.T.; Pericolini, E. It Takes Two to Tango: How a Dysregulation of the Innate Immunity, Coupled With Candida Virulence, Triggers VVC Onset. Front. Microbiol. 2021, 12, 692491. [Google Scholar] [CrossRef]
- Willems, H.M.E.; Ahmed, S.S.; Liu, J.; Xu, Z.; Peters, B.M. Vulvovaginal Candidiasis: A Current Understanding and Burning Questions. J. Fungi 2020, 6, 27. [Google Scholar] [CrossRef]
- Cauchie, M.; Desmet, S.; Lagrou, K. Candida and its dual lifestyle as a commensal and a pathogen. Res. Microbiol. 2017, 168, 802–810. [Google Scholar] [CrossRef]
- Mukaremera, L.; Lee, K.K.; Mora-Montes, H.M.; Gow, N.A.R. Candida albicans Yeast, Pseudohyphal, and Hyphal Morphogenesis Differentially Affects Immune Recognition. Front. Immunol. 2017, 8, 629. [Google Scholar] [CrossRef]
- Calderone, R.A.; Fonzi, W.A. Virulence factors of Candida albicans. Trends Microbiol. 2001, 9, 327–335. [Google Scholar] [CrossRef]
- Mayer, F.L.; Wilson, D.; Hube, B. Candida albicans pathogenicity mechanisms. Virulence 2013, 4, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Zangl, I.; Pap, I.J.; Aspock, C.; Schuller, C. The role of Lactobacillus species in the control of Candida via biotrophic interactions. Microb. Cell 2019, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Moyes, D.L.; Wilson, D.; Richardson, J.P.; Mogavero, S.; Tang, S.X.; Wernecke, J.; Hofs, S.; Gratacap, R.L.; Robbins, J.; Runglall, M.; et al. Candidalysin is a fungal peptide toxin critical for mucosal infection. Nature 2016, 532, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Moyes, D.L.; Richardson, J.P.; Naglik, J.R. Candida albicans-epithelial interactions and pathogenicity mechanisms: Scratching the surface. Virulence 2015, 6, 338–346. [Google Scholar] [CrossRef] [PubMed]
- Kalia, N.; Singh, J.; Kaur, M. Microbiota in vaginal health and pathogenesis of recurrent vulvovaginal infections: A critical review. Ann. Clin. Microbiol. Antimicrob. 2020, 19, 5. [Google Scholar] [CrossRef]
- Fidel, P.L., Jr.; Barousse, M.; Espinosa, T.; Ficarra, M.; Sturtevant, J.; Martin, D.H.; Quayle, A.J.; Dunlap, K. An intravaginal live Candida challenge in humans leads to new hypotheses for the immunopathogenesis of vulvovaginal candidiasis. Infect. Immun. 2004, 72, 2939–2946. [Google Scholar] [CrossRef]
- Wachtler, B.; Citiulo, F.; Jablonowski, N.; Forster, S.; Dalle, F.; Schaller, M.; Wilson, D.; Hube, B. Candida albicans-epithelial interactions: Dissecting the roles of active penetration, induced endocytosis and host factors on the infection process. PLoS ONE 2012, 7, e36952. [Google Scholar] [CrossRef]
- Peters, B.M.; Palmer, G.E.; Nash, A.K.; Lilly, E.A.; Fidel, P.L., Jr.; Noverr, M.C. Fungal morphogenetic pathways are required for the hallmark inflammatory response during Candida albicans vaginitis. Infect. Immun. 2014, 82, 532–543. [Google Scholar] [CrossRef]
- Re, A.C.S.; Martins, J.F.; Cunha-Filho, M.; Gelfuso, G.M.; Aires, C.P.; Gratieri, T. New perspectives on the topical management of recurrent candidiasis. Drug Deliv. Transl. Res. 2021, 11, 1568–1585. [Google Scholar] [CrossRef]
- Sherry, L.; Kean, R.; McKloud, E.; O’Donnell, L.E.; Metcalfe, R.; Jones, B.L.; Ramage, G. Biofilms Formed by Isolates from Recurrent Vulvovaginal Candidiasis Patients Are Heterogeneous and Insensitive to Fluconazole. Antimicrob. Agents Chemother. 2017, 61, e01065-17. [Google Scholar] [CrossRef]
- Muzny, C.A.; Schwebke, J.R. Biofilms: An Underappreciated Mechanism of Treatment Failure and Recurrence in Vaginal Infections. Clin. Infect. Dis. 2015, 61, 601–606. [Google Scholar] [CrossRef]
- Gulati, M.; Nobile, C.J. Candida albicans biofilms: Development, regulation, and molecular mechanisms. Microbes Infect. 2016, 18, 310–321. [Google Scholar] [CrossRef]
- Paiva, L.C.; Vidigal, P.G.; Donatti, L.; Svidzinski, T.I.; Consolaro, M.E. Assessment of in vitro biofilm formation by Candida species isolates from vulvovaginal candidiasis and ultrastructural characteristics. Micron 2012, 43, 497–502. [Google Scholar] [CrossRef]
- Harriott, M.M.; Lilly, E.A.; Rodriguez, T.E.; Fidel, P.L.; Noverr, M.C. Candida albicans forms biofilms on the vaginal mucosa. Microbiology 2010, 156, 3635–3644. [Google Scholar] [CrossRef] [PubMed]
- Alves, C.T.; Silva, S.; Pereira, L.; Williams, D.W.; Azeredo, J.; Henriques, M. Effect of progesterone on Candida albicans vaginal pathogenicity. Int. J. Med. Microbiol. 2014, 304, 1011–1017. [Google Scholar] [CrossRef] [PubMed]
- Sobel, J.D. Editorial Commentary: Vaginal Biofilm: Much Ado About Nothing, or a New Therapeutic Challenge? Clin. Infect. Dis. 2015, 61, 607–608. [Google Scholar] [CrossRef] [PubMed]
- Valenti, P.; Rosa, L.; Capobianco, D.; Lepanto, M.S.; Schiavi, E.; Cutone, A.; Paesano, R.; Mastromarino, P. Role of Lactobacilli and Lactoferrin in the Mucosal Cervicovaginal Defense. Front. Immunol. 2018, 9, 376. [Google Scholar] [CrossRef] [PubMed]
- Lykke, M.R.; Becher, N.; Haahr, T.; Boedtkjer, E.; Jensen, J.S.; Uldbjerg, N. Vaginal, Cervical and Uterine pH in Women with Normal and Abnormal Vaginal Microbiota. Pathogens 2021, 10, 90. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.A.; Beasley, D.E.; Dunn, R.R.; Archie, E.A. Lactobacilli Dominance and Vaginal pH: Why Is the Human Vaginal Microbiome Unique? Front. Microbiol. 2016, 7, 1936. [Google Scholar] [CrossRef] [PubMed]
- Fluconazole 150mg Capsules. Available online: https://www.medicines.org.uk/emc/product/6086/smpc#gref (accessed on 8 July 2021).
- Whaley, S.G.; Berkow, E.L.; Rybak, J.M.; Nishimoto, A.T.; Barker, K.S.; Rogers, P.D. Azole Antifungal Resistance in Candida albicans and Emerging Non-albicans Candida Species. Front. Microbiol. 2016, 7, 2173. [Google Scholar] [CrossRef]
- Zhang, Y.Q.; Gamarra, S.; Garcia-Effron, G.; Park, S.; Perlin, D.S.; Rao, R. Requirement for ergosterol in V-ATPase function underlies antifungal activity of azole drugs. PLoS Pathog. 2010, 6, e1000939. [Google Scholar] [CrossRef]
- Rodrigues, M.L. The Multifunctional Fungal Ergosterol. mBio 2018, 9, e01755-18. [Google Scholar] [CrossRef]
- Berkow, E.L.; Lockhart, S.R. Fluconazole resistance in Candida species: A current perspective. Infect. Drug Resist. 2017, 10, 237–245. [Google Scholar] [CrossRef]
- Heimark, L.; Shipkova, P.; Greene, J.; Munayyer, H.; Yarosh-Tomaine, T.; DiDomenico, B.; Hare, R.; Pramanik, B.N. Mechanism of azole antifungal activity as determined by liquid chromatographic/mass spectrometric monitoring of ergosterol biosynthesis. J. Mass Spectrom. 2002, 37, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Marchaim, D.; Lemanek, L.; Bheemreddy, S.; Kaye, K.S.; Sobel, J.D. Fluconazole-resistant Candida albicans vulvovaginitis. Obstet. Gynecol. 2012, 120, 1407–1414. [Google Scholar] [CrossRef]
- Badiee, P.; Alborzi, A. Susceptibility of clinical Candida species isolates to antifungal agents by E-test, Southern Iran: A five year study. Iran. J. Microbiol. 2011, 3, 183–188. [Google Scholar]
- Citak, S.; Ozcelik, B.; Cesur, S.; Abbasoglu, U. In vitro susceptibility of Candida species isolated from blood culture to some antifungal agents. Jpn. J. Infect. Dis. 2005, 58, 44–46. [Google Scholar] [PubMed]
- Saporiti, A.M.; Gomez, D.; Levalle, S.; Galeano, M.; Davel, G.; Vivot, W.; Rodero, L. Vaginal candidiasis: Etiology and sensitivity profile to antifungal agents in clinical use. Rev. Argent. Microbiol. 2001, 33, 217–222. [Google Scholar] [PubMed]
- Tseng, Y.H.; Lee, W.T.; Kuo, T.C. In-Vitro Susceptibility of Fluconazole and Amphotericin B against Candida Isolates from Women with Vaginal Candidiasis in Taiwan. J. Food Drug Anal. 2005, 13, 12–16. [Google Scholar] [CrossRef]
- Zhang, J.Y.; Liu, J.H.; Liu, F.D.; Xia, Y.H.; Wang, J.; Liu, X.; Zhang, Z.Q.; Zhu, N.; Yan, Y.; Ying, Y.; et al. Vulvovaginal candidiasis: Species distribution, fluconazole resistance and drug efflux pump gene overexpression. Mycoses 2014, 57, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Jackson, S.T.; Mullings, A.M.; Rainford, L.; Miller, A. The epidemiology of mycotic vulvovaginitis and the use of antifungal agents in suspected mycotic vulvovaginitis and its implications for clinical practice. West. Indian Med. J. 2005, 54, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Cerdeira, C.; Martinez-Herrera, E.; Carnero-Gregorio, M.; Lopez-Barcenas, A.; Fabbrocini, G.; Fida, M.; El-Samahy, M.; Gonzalez-Cespon, J.L. Pathogenesis and Clinical Relevance of Candida Biofilms in Vulvovaginal Candidiasis. Front. Microbiol. 2020, 11, 544480. [Google Scholar] [CrossRef] [PubMed]
- Taff, H.T.; Mitchell, K.F.; Edward, J.A.; Andes, D.R. Mechanisms of Candida biofilm drug resistance. Future Microbiol. 2013, 8, 1325–1337. [Google Scholar] [CrossRef]
- Vediyappan, G.; Rossignol, T.; d’Enfert, C. Interaction of Candida albicans biofilms with antifungals: Transcriptional response and binding of antifungals to beta-glucans. Antimicrob. Agents Chemother. 2010, 54, 2096–2111. [Google Scholar] [CrossRef]
- Hermanns, R.; Mateescu, C.; Thrasyvoulou, A.; Tananaki, C.; Wagener, F.A.D.T.G.; Cremers, N.A.J. Defining the standards for medical grade honey. J. Apic. Res. 2020, 59, 125–135. [Google Scholar] [CrossRef]
- Yilmaz, A.C.; Aygin, D. Honey Dressing in Wound Treatment: A Systematic Review. Complement. Ther. Med. 2020, 51, 102388. [Google Scholar] [CrossRef] [PubMed]
- Emineke, S.; Cooper, A.J.; Fouch, S.; Birch, B.R.; Lwaleed, B.A. Diluted honey inhibits biofilm formation: Potential application in urinary catheter management? J. Clin. Pathol. 2017, 70, 140–144. [Google Scholar] [CrossRef]
- Aissat, S.; Ahmed, M.; Djebli, N. Propolis-Sahara honeys preparation exhibits antibacterial and anti-biofilm activity against bacterial biofilms formed on urinary catheters. Asian Pac. J. Trop. Dis. 2016, 6, 873–877. [Google Scholar] [CrossRef]
- Fernandes, L.; Ribeiro, H.; Oliveira, A.; Sanches Silva, A.; Freitas, A.; Henriques, M.; Rodrigues, M.E. Portuguese honeys as antimicrobial agents against Candida species. J. Tradit. Complement. Med. 2021, 11, 130–136. [Google Scholar] [CrossRef]
- Boukraa, L.; Benbarek, H.; Moussa, A. Synergistic action of starch and honey against Candida albicans in correlation with diastase number. Braz. J. Microbiol. 2008, 39, 40–43. [Google Scholar] [CrossRef]
- Mandal, M.D.; Mandal, S. Honey: Its medicinal property and antibacterial activity. Asian Pac. J. Trop. Biomed. 2011, 1, 154–160. [Google Scholar] [CrossRef]
- Nair, H.K.R.; Tatavilis, N.; Pospisilova, I.; Kucerova, J.; Cremers, N.A.J. Medical-Grade Honey Kills Antibiotic-Resistant Bacteria and Prevents Amputation in Diabetics with Infected Ulcers: A Prospective Case Series. Antibiotics 2020, 9, 529. [Google Scholar] [CrossRef]
- Blair, S.E.; Cokcetin, N.N.; Harry, E.J.; Carter, D.A. The unusual antibacterial activity of medical-grade Leptospermum honey: Antibacterial spectrum, resistance and transcriptome analysis. Eur. J. Clin. Microbiol. Infect. Dis. 2009, 28, 1199–1208. [Google Scholar] [CrossRef]
- Koc, A.N.; Silici, S.; Ercal, B.D.; Kasap, F.; Hormet-Oz, H.T.; Mavus-Buldu, H. Antifungal activity of Turkish honey against Candida spp. and Trichosporon spp: An in vitro evaluation. Med. Mycol. 2009, 47, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Smaropoulos, E.; Cremers, N.A. Medical grade honey for the treatment of paediatric abdominal wounds: A case series. J. Wound Care 2020, 29, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Smaropoulos, E.; Cremers, N.A.J. The pro-healing effects of medical grade honey supported by a pediatric case series. Complement. Ther. Med. 2019, 45, 14–18. [Google Scholar] [CrossRef]
- Smaropoulos, E.; Cremers, N.A.J. Medical grade honey for the treatment of extravasation-induced injuries in preterm neonates—A case series. Adv. Neonatal Care 2020, 21, 122–132. [Google Scholar] [CrossRef]
- Smaropoulos, E.; Cremers, N.A.J. Treating severe wounds in pediatrics with medical grade honey: A case series. Clin. Case Rep. 2020, 8, 469–476. [Google Scholar] [CrossRef]
- Fluconazole is a Triazole Antifungal Used to Treat Various Fungal Infections Including Candidiasis. Available online: https://www.drugbank.ca/drugs/DB00196 (accessed on 26 April 2021).
- Doke, S.K.; Raut, J.S.; Dhawale, S.; Karuppayil, S.M. Sensitization of Candida albicans biofilms to fluconazole by terpenoids of plant origin. J. Gen. Appl. Microbiol. 2014, 60, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Cavalheiro, M.; Teixeira, M.C. Candida Biofilms: Threats, Challenges, and Promising Strategies. Front. Med. 2018, 5, 28. [Google Scholar] [CrossRef]
- Irish, J.; Carter, D.A.; Shokohi, T.; Blair, S.E. Honey has an antifungal effect against Candida species. Med. Mycol. 2006, 44, 289–291. [Google Scholar] [CrossRef] [PubMed]
- Shokri, H.; Sharifzadeh, A. Fungicidal efficacy of various honeys against fluconazole-resistant Candida species isolated from HIV(+) patients with candidiasis. J. Mycol. Med. 2017, 27, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.J.; Al-Ghamdi, A.; Usmani, S.; Al-Waili, N.S.; Sharma, D.; Nuru, A.; Al-Attal, Y. Effect of jujube honey on Candida albicans growth and biofilm formation. Arch. Med. Res. 2013, 44, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Estevinho, M.L.; Afonso, S.E.; Feas, X. Antifungal effect of lavender honey against Candida albicans, Candida krusei and Cryptococcus neoformans. J. Food Sci. Technol. 2011, 48, 640–643. [Google Scholar] [CrossRef] [PubMed]
- Hermanns, R.; Cremers, N.A.J.; Leeming, J.P.; van der Werf, E.T. Sweet Relief: Determining the Antimicrobial Activity of Medical Grade Honey Against Vaginal Isolates of Candida albicans. J. Fungi 2019, 5, 85. [Google Scholar] [CrossRef]
- Al-Waili, N.S. Mixture of honey, beeswax and olive oil inhibits growth of Staphylococcus aureus and Candida albicans. Arch. Med. Res. 2005, 36, 10–13. [Google Scholar] [CrossRef]
- Khosravi, A.R.; Shokri, H.; Katiraee, F.; Ziglari, T.; Forsi, M. Fungicidal potential of different Iranian honeys against some pathogenic Candida species. J. Apic. Res. 2008, 47, 256–260. [Google Scholar] [CrossRef]
- Deorukhkar, S.C.; Saini, S.; Mathew, S. Non-albicans Candida Infection: An Emerging Threat. Interdiscip. Perspect. Infect. Dis. 2014, 2014, 615958. [Google Scholar] [CrossRef]
- Shamala, T.R.; Shri Jyothi, Y.; Saibaba, P. Stimulatory effect of honey on multiplication of lactic acid bacteria under in vitro and in vivo conditions. Lett. Appl. Microbiol. 2000, 30, 453–455. [Google Scholar] [CrossRef]
- Samarghandian, S.; Farkhondeh, T.; Samini, F. Honey and Health: A Review of Recent Clinical Research. Pharmacogn. Res. 2017, 9, 121–127. [Google Scholar] [CrossRef]
- Vallianou, N.; Gounari, P.; Skourtis, A.; Panagos, J.; Kazazis, C. Honey and its Anti-Inflammatory, Anti-Bacterial and Anti-Oxidant Properties. Gen. Med. Open Access 2014, 2, 132–137. [Google Scholar] [CrossRef]
- Yaghoobi, R.; Kazerouni, A.; Kazerouni, O. Evidence for Clinical Use of Honey in Wound Healing as an Anti-bacterial, Anti-inflammatory Anti-oxidant and Anti-viral Agent: A Review. Jundishapur J. Nat. Pharm. Prod. 2013, 8, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Bogdanov, S.; Jurendic, T.; Sieber, R.; Gallmann, P. Honey for nutrition and health: A review. J. Am. Coll. Nutr. 2008, 27, 677–689. [Google Scholar] [CrossRef] [PubMed]
- Vaday, G.G.; Franitza, S.; Schor, H.; Hecht, I.; Brill, A.; Cahalon, L.; Hershkoviz, R.; Lider, O. Combinatorial signals by inflammatory cytokines and chemokines mediate leukocyte interactions with extracellular matrix. J. Leukoc. Biol. 2001, 69, 885–892. [Google Scholar] [PubMed]
- Sokol, C.L.; Luster, A.D. The chemokine system in innate immunity. Cold Spring Harb. Perspect. Biol. 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Miguel, M.G.; Antunes, M.D.; Faleiro, M.L. Honey as a Complementary Medicine. Integr. Med. Insights 2017, 12, 1178633717702869. [Google Scholar] [CrossRef] [PubMed]
- Gasparrini, M.; Afrin, S.; Forbes-Hernandez, T.Y.; Cianciosi, D.; Reboredo-Rodriguez, P.; Amici, A.; Battino, M.; Giampieri, F. Protective effects of Manuka honey on LPS-treated RAW 264.7 macrophages. Part 2: Control of oxidative stress induced damage, increase of antioxidant enzyme activities and attenuation of inflammation. Food Chem. Toxicol. 2018, 120, 578–587. [Google Scholar] [CrossRef]
- Ahmad, A.; Khan, R.A.; Mesaik, M.A. Anti inflammatory effect of natural honey on bovine thrombin-induced oxidative burst in phagocytes. Phytother. Res. 2009, 23, 801–808. [Google Scholar] [CrossRef]
- Gustafsson, K.; Tatz, A.M.; Slavin, R.A.; Dahan, R.; Ahmad, W.A.; Sutton, G.A.; Kelmer, G. Will local intraoperative application of Medical Grade Honey in the incision protect against incisional infection in horses undergoing colic surgery? AAEP Proc. 2019, 65, 387–388. [Google Scholar]
- Mandel, H.H.; Sutton, G.A.; Abu, E.; Kelmer, G. Intralesional application of medical grade honey improves healing of surgically treated lacerations in horses. Equine Vet. J. 2020, 52, 41–45. [Google Scholar] [CrossRef]
- Banaeian, S.; Sereshti, M.; Rafieian, M.; Farahbod, F.; Kheiri, S. Comparison of vaginal ointment of honey and clotrimazole for treatment of vulvovaginal candidiasis: A random clinical trial. J. Mycol. Med. 2017, 27, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Seifinadergoli, Z.; Nahidi, F.; Safaiyan, A.; Javadzadeh, Y.; Eteraf-Oskouei, T. Comparison of the efficacy of honey gel and clotrimazole cream in the treatment of vaginal candidiasis symptoms: A randomized clinical trial. Electron. Physician 2018, 10, 6904–6911. [Google Scholar] [CrossRef] [PubMed]
- Darvishi, M.; Jahdi, F.; Hamzegardeshi, Z.; Goodarzi, S.; Vahedi, M. The Comparison of vaginal cream of mixing yogurt, honey and clotrimazole on symptoms of vaginal candidiasis. Glob. J. Health Sci. 2015, 7, 108–116. [Google Scholar] [CrossRef]
- Abdelmonem, A.M.; Rasheed, S.M.; Mohamed, A. Bee-honey and yogurt: A novel mixture for treating patients with vulvovaginal candidiasis during pregnancy. Arch. Gynecol. Obstet. 2012, 286, 109–114. [Google Scholar] [CrossRef]
- Aboushady, R.M.N.; Eswi, A.; Mostafa, F.A.M. Sider Honey vs. Fluconazole and the Cure Rate of Vulvovaginal Candidiasis. IOSR J. Nurs. Health Sci. 2015, 4, 5–14. [Google Scholar] [CrossRef]
- Jahdi, F.; Hamzehgardeshi, Z.; Kuolaei, M.D.; Vahedi, M.; Goodarzi, S. Vulvovaginal Candidiasis Symptom Reduction—Honey, Yogurt-and-Honey and Clotrimazole Vaginal Cream—A Triple Blind Randomized Control Trial. J. Evol. Med. Dent. Sci. 2021, 10, 515–521. [Google Scholar] [CrossRef]
- Rasooli, T.; Nahidi, F.; Mojab, F.; Nasiri, M.; Parsapour, H. Effect of Honey Cinnamon Vaginal Cream and Clotrimazole Vaginal Cream on Improvement of Candida Vaginitis Symptoms in Women: Randomized Clinical Trial. Iran. J. Obstet. Gynecol. Infertil. 2019, 22, 59–67. [Google Scholar] [CrossRef]
- Fazel, N.; Hashemian, M.; Ramezani, M.; Akaberi, A. Comparative Effect alone Honey and Mix with Chlotrimazol on Vaginitis Candidacies. Iran. J. Obstet. Gynecol. Infertil. 2012, 14, 48–54. [Google Scholar] [CrossRef]
- Cremers, N.; Belas, A.; Santos Costa, S.; Couto, I.; de Rooster, H.; Pomba, C. In vitro antimicrobial efficacy of two medical grade honey formulations against common high-risk meticillin-resistant staphylococci and Pseudomonas spp. pathogens. Vet. Dermatol. 2020, 31, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Pleeging, C.C.F.; Coenye, T.; Mossialos, D.; De Rooster, H.; Chrysostomou, D.; Wagener, F.A.D.T.G.; Cremers, N.A.J. Synergistic Antimicrobial Activity of Supplemented Medical-Grade Honey against Pseudomonas aeruginosa Biofilm Formation and Eradication. Antibiotics 2020, 9, 866. [Google Scholar] [CrossRef] [PubMed]
- Munstedt, K.; Momm, F.; Hubner, J. Honey in the management of side effects of radiotherapy- or radio/chemotherapy-induced oral mucositis. A systematic review. Complement. Ther. Clin. Pract. 2019, 34, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.M.; Luo, Y.W.; Tam, K.W.; Lin, C.C.; Huang, T.W. Prophylactic and therapeutic effects of honey on radiochemotherapy-induced mucositis: A meta-analysis of randomized controlled trials. Support. Care Cancer 2019, 27, 2361–2370. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Gong, G.; Jin, E.; Han, X.; Zhuo, Y.; Yang, S.; Song, B.; Zhang, Y.; Piao, C. Topical application of honey in the management of chemo/radiotherapy-induced oral mucositis: A systematic review and network meta-analysis. Int. J. Nurs. Stud. 2019, 89, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Boon, M.E. Honey proves attractive for vaginal use. Jaarversl. LCPL 2003, 32–39. Available online: http://docplayer.net/43938553-A4-honey-cream-proves-attractive-for-vaginal-use-the-vaginal-smear-as-a-test-3.html (accessed on 15 August 2004).
- De Groot, T.; Janssen, T.; Faro, D.; Cremers, N.A.J.; Chowdhary, A.; Meis, J.F. Antifungal Activity of a Medical-Grade Honey Formulation against Candida auris. J. Fungi 2021, 7, 50. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.M.P.; Devesa, J.S.P.; Hill, P.B. In vitro efficacy of a honey-based gel against canine clinical isolates of Staphylococcus pseudintermedius and Malassezia pachydermatis. Vet. Dermatol. 2018, 29, 180-e65. [Google Scholar] [CrossRef]
- Holubová, A.; Chlupácová, L.; Cetlová, L.; Cremers, N.A.J.; Pokorná, A. Medical-Grade Honey as an Alternative Treatment for Antibiotics in Non-Healing Wounds—A Prospective Case Series. Antibiotics 2021, 10, 918. [Google Scholar] [CrossRef]
- Zbuchea, A. Honey, Food and Medicine: Scientific Rationale and Practical Efficiency in External Administration of Medicinal Honey for Wound Healing. J. Agric. Sci. Technol. B 2017, 7, 206–219. [Google Scholar] [CrossRef][Green Version]
| Characteristic | Fluconazole | MGH |
|---|---|---|
| Candida albicans | + | + |
| (Increased raise in VVC caused by) NAC species | − | + |
| Biofilms | − | + |
| Increased resistance | − | + |
| Microenvironment/vaginal mucosal response | − | + |
| Lactobacilli | − | +− |
| pH | − | + |
| Osmotic effect | − | + |
| Antimicrobial | + | + |
| Anti-inflammatory | − | + |
| Antioxidative | − | + |
| Selected Studies | Methods | Results |
|---|---|---|
| Irish et al. 2006 [75] | Clinical isolates of Candida albicans, Candida glabrata, and Candida dubliensis were tested against four different honeys: -Jarrrah honey with hydrogen peroxide activity -Medihoney -Comvita wound care 18+, a pure Leptospermum honey -Artificial honey used to simulate the high sugar levels found in honey | -Jarrah honey has a MIC of 18.5% compared to 38.2% for Medihoney, 39.9% for Comvita honey, and 42.6% for artificial honey against Candida albicans. Jarrah honey was also significantly more active against Candida Glabrata and Candida dubliensis. -Honey was effective against isolates who are resistant to itraconazole and/or fluconazole. |
| Shokri et al. 2017 [76] | Fungicidal efficacy of 3 Iranian honey samples against fluconazole-resistant Candida species (including Candida albicans, Candida glabrata, Candida krusei, and Candida tropicalis) isolated from HIV+ patients with candidiasis | All tested honeys had antifungal activity against FLU-resistant Candida species, ranging from 20% to 56.25% (v/v) and 25% to 56.25% (v/v) for MICs and minimum fungicidal concentrations (MFCs), respectively. No statistically significant differences were observed between the honey samples. |
| Ansari et al. 2013 [77] | Effect of jujube honey on Candida albicans growth and biofilm formation | 40% w/v Jujube honey interferes with formation of Candida albicans biofilms and disrupts established biofilms. |
| Banaean-Boroujeni et al. 2013 [7] | Effect of Iranian honey and miconazol against Candida albicans, in vitro | Honey prevented growth of Candida albicans greatly only at an 80% concentration, whereas miconazol inhibited it completely. Honey did not interfere with the Lactobacillus. |
| Estevinho et al. 2011 [78] | Monofloral lavender honey samples were analyzed to test antifungal effect against Candida albicans, Candida krusei, and Cryptococcus neoformans | The honey concentration that inhibited 10% of the yeasts’ growth ranged from 31% (Candida albicans) and 16.8% (Candida krusei). |
| Koc et al. 2009 [67] | Antifungal activity of four Turkish honey samples against 40 yeast species (Candida albicans, Candida Krusei, Candida Glabrata, and Trichosporon spp.) | The honeys had antifungal activity for Candida albicans at a mean MIC of 45.56%, for Candida Glabrata at 64.09%, and for Candida krusei at 56.88%. |
| Hermanns et al. 2019 [79] | Five clinical Candida albicans isolates and a control strain were tested against unprocessed Mexican Yucatan honey with hydrogen peroxide activity and L-Mesitran soft | -No effect of 40% Mexican Yucatan MGH alone. -L-mesitran has fungistatic (25–50% MIC) and fungicidal (50% MFC) activity, corresponding to honey concentrations of 10% and 20%, respectively. -The supplements in L-Mesitran enhanced the antimicrobial activity of the honey formulation. |
| Al-Waili et al. 2005 [80] | Effects of honey, olive oil, and beeswax and the mixture on growth of Staphylococcus aureus and Candida albicans isolates | The amount of honey present in the honey mixture (50% w/v) completely inhibited the growth of Candida albicans. |
| Khosravi et al. 2008 [81] | Anti-candidal activity of 28 locally produced honeys from Iran against Candida albicans, Candida parapsilosis, Candida tropicalis, Candida kefyr, Candida glabrata, and Candida dubliniensis | The MIC and MFC means of different honeys were 24–47% and 29–56% against different Candida species, respectively. |
| Fernandes et al. 2021 [62] | Antifungal activity of five Portuguese honeys and Manuka honey in planktonic and biofilm models of Candida albicans, Candida tropicalis, Candida glabrata, and Candida parapsilosis | -All honeys had a potent activity against Candida species (MIC 25–50% (w/v)). -Biofilms can be reduced at a concentration of 50–75% honey. |
| Selected Studies | Treatment | Trial Design | Purpose/Objective/Content | Results/Conclusion |
|---|---|---|---|---|
| Banaeian et al. 2017 [95] | Clotrimazole 1% cream (n = 36) versus honey cream (honey and neutral crème in 70:30 ratio) (n = 44), both 5 g with applicator for 7 nights | RCT | -Inflammation -Vaginal discharge -Irritation/itching At baseline in the fourth and eight days of treatment -Recurrence after 3 months | -Significant decrease in discharge, inflammation and itching in both groups. -Inflammation and discharge scores were significantly lower on the eight day in the clotrimazole group, compared to the honey group. -No significant difference in the itching score on the eighth day between the honey and clotrimazole groups. -Greater efficacy of honey for RVVC. After 3 months, 5 of 17 patients from the honey group had a recurrent infection, and 8 of 15 patients of the clotrimazole group had a recurrent infection. -Honey decreased VVC symptoms without affecting lactobacillus. |
| Seifinadergoli et al. 2018 [96] | Clotrimazole 1% cream (n = 53) versus 50% honey gel (n = 53), both 5 g with applicator for 8 nights | RCT | -Vaginal discharge -Itching -Burning -Dyspareunia -Urinary problem At baseline, in the fourth and eight days after treatment | -Vaginal discharge for 68% in honey group versus 72% in clotrimazole group after 8 days (p < 0.001). -Itching for 4% in honey group versus 10% in clotrimazole group after 8 days (p < 0.001). -Burning for 4% in honey group versus 0% in clotrimazole group after 8 days (p < 0.001). -Culture was 50% positive in honey group versus 20% in clotrimazole group after eight days, and there was no statistical difference. -Equally effective in making the culture and wet smear results negative. -Honey can be used as an alternative for other antifungal drugs. |
| Darvishi et al. 2015 [97] | Clotrimazole 1% cream (n = 35) versus cream mixed of yogurt and honey (n = 35), both 5 g with applicator for 7 days | RCT | -Itching -Irritation -Dysuria -Dyspareunia -Discharge -Cultures At baseline, 7 days after treatment, 14 days after treatment | -Itching for 2.9% in yogurt and honey group versus 25.7% in clotrimazole group 14 days after treatment (p < 0.02). -Irritation for 2.9% in yogurt and honey group versus 22.9% in clotrimazole group 14 days after treatment (p < 0.04). -Discharge for 2.9% in yogurt and honey group versus 22.9% in clotrimazole group 14 days after treatment (p < 0.04). -Significant improvement in symptoms in yogurt and honey group compared to clotrimazole (p < 0.05). -No significant differences in mycological cure rate between yoghurt and honey (71.4%) and clotrimazole (85.7%) groups at 14 days after treatment. |
| Abdelmonem et al. 2012 [98] | Tioconazole 100 mg vaginal tablet (n = 47) once daily for 7 days versus bee honey and yogurt mixture vaginally with an applicator 30 g 62.5% honey twice a day for 7 days (n = 82) by pregnant women | Prospective comparative study | -Clinical cure -Mycological cure Before treatment and after treatment | -Clinical cure rate was significantly higher in the honey and yogurt group (87.8%) compared to the tioconazole group (72.3%). -The mycological cure rate was significantly higher in the tiozonazole group (91.5%) compared to the honey and yogurt group (76.9%). -Itching was 3.7% in the honey and yogurt group versus 14.9% in the tioconazole group. -Discharge was 4.9% in the honey and yogurt group versus 12.8% in the tioconazole group. -Vulvo-vaginal redness was 3.7% in the honey and yogurt group versus 8.5% in the tioconazole group. |
| Aboushady et al. 2015 [99] | Fluconazole (n = 30) versus Sider honey 5 mL (80%) (n = 30) applied vaginally twice/day for 7 days | Quasi-experimental design | -Clinical cure -Mycological cure Before treatment and after treatment | -Clinical cure rate from sider honey (86.6%) compared to fluconazole (40%). -Mycological cure rate from sider honey (76.7%) compared to fluconazole (43.3%). -Vaginal discharge after sider honey (10%) compared to fluconazole (50%). -Burning sensation after sider honey (0%) compared to fluconazole (33.3%). -Itching after sider honey (0%) compared to fluconazole (10%). |
| Jahdi et al. 2021 [100] | -Mixture of honey and yogurt cream (n = 35) -Honey cream (n = 35) -Clotrimazole cream (n = 35) versus mixture of honey and yogurt cream (n = 35) versus honey cream (n = 35); each group was treated for 7 nights, all 5 g with applicator intravaginally | RCT | -Leucorrhea -Itch -Irritation -Dysuria -Dyspareunia Before, at 7 and 14 days after treatment -Mycological cure rate 7 and 14 days after treatment | -Itching after 14 days of treatment with clotrimazole (25.7%) versus yogurt and honey (2.9%) versus honey (14%). -Irritation after 14 days of treatment with clotrimazole (22.9%) versus yogurt and honey (2.9%) versus honey (5.7%). -Significant difference between yogurt and honey group and honey vaginal cream group versus clotrimazole group in improving symptoms. -Negative culture 14 days after treatment was found in yogurt and honey group (71.4%), honey group (68.6%), and clotrimazole group (85.7%) with no significant differences (p > 0.05). |
| Rasooli et al. 2019 [101] | -Honey cinnamon vaginal cream (n = 50) -Clotrimazole vaginal cream (n = 50) | RCT | -Clinical effect -Microscopic effect | -In both groups, symptoms improved after treatment (p < 0.001). -In the honey cinnamon vaginal cream group, burning was significantly less than the clotrimazole group (p = 0.008). -Culture results were similar after treatment (p = 0.461). -Honey cinnamon vaginal cream can be advisable to use it as an alternative to the clotrimazole vaginal cream. |
| Fazel et al. 2017 [102] | -Honey 5 mL intravaginally (n = 15) -Honey 5 mL and clotrimazole 100 mg (n = 30) -Clotrimazole 100 mg intravaginally (n = 32) -Treated daily for 7 days | Double blind clinical trial | -Clinical effect -Microscopic effect | -Alleviations occurred in symptoms and signs of vaginitis after treatment with honey and clotrimazole. -The success rate was 100% in the honey group and in the honey and clotrimazole group. -Use of honey alone represents a novel and effective formulation for the treatment of vaginitis. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
van Riel, S.J.J.M.; Lardenoije, C.M.J.G.; Oudhuis, G.J.; Cremers, N.A.J. Treating (Recurrent) Vulvovaginal Candidiasis with Medical-Grade Honey—Concepts and Practical Considerations. J. Fungi 2021, 7, 664. https://doi.org/10.3390/jof7080664
van Riel SJJM, Lardenoije CMJG, Oudhuis GJ, Cremers NAJ. Treating (Recurrent) Vulvovaginal Candidiasis with Medical-Grade Honey—Concepts and Practical Considerations. Journal of Fungi. 2021; 7(8):664. https://doi.org/10.3390/jof7080664
Chicago/Turabian Stylevan Riel, Senna J. J. M., Celine M. J. G. Lardenoije, Guy J. Oudhuis, and Niels A. J. Cremers. 2021. "Treating (Recurrent) Vulvovaginal Candidiasis with Medical-Grade Honey—Concepts and Practical Considerations" Journal of Fungi 7, no. 8: 664. https://doi.org/10.3390/jof7080664
APA Stylevan Riel, S. J. J. M., Lardenoije, C. M. J. G., Oudhuis, G. J., & Cremers, N. A. J. (2021). Treating (Recurrent) Vulvovaginal Candidiasis with Medical-Grade Honey—Concepts and Practical Considerations. Journal of Fungi, 7(8), 664. https://doi.org/10.3390/jof7080664







