Species Distribution, Antifungal Susceptibility, and Molecular Epidemiology of Candida Species Causing Candidemia in a Tertiary Care Hospital in Bangkok, Thailand
Abstract
:1. Introduction
2. Materials and Methods
2.1. Yeast Isolates, Identification, and Ethical Statement
2.2. Antifungal Susceptibility Testing
2.3. DNA Extraction
2.4. Genotyping
3. Results
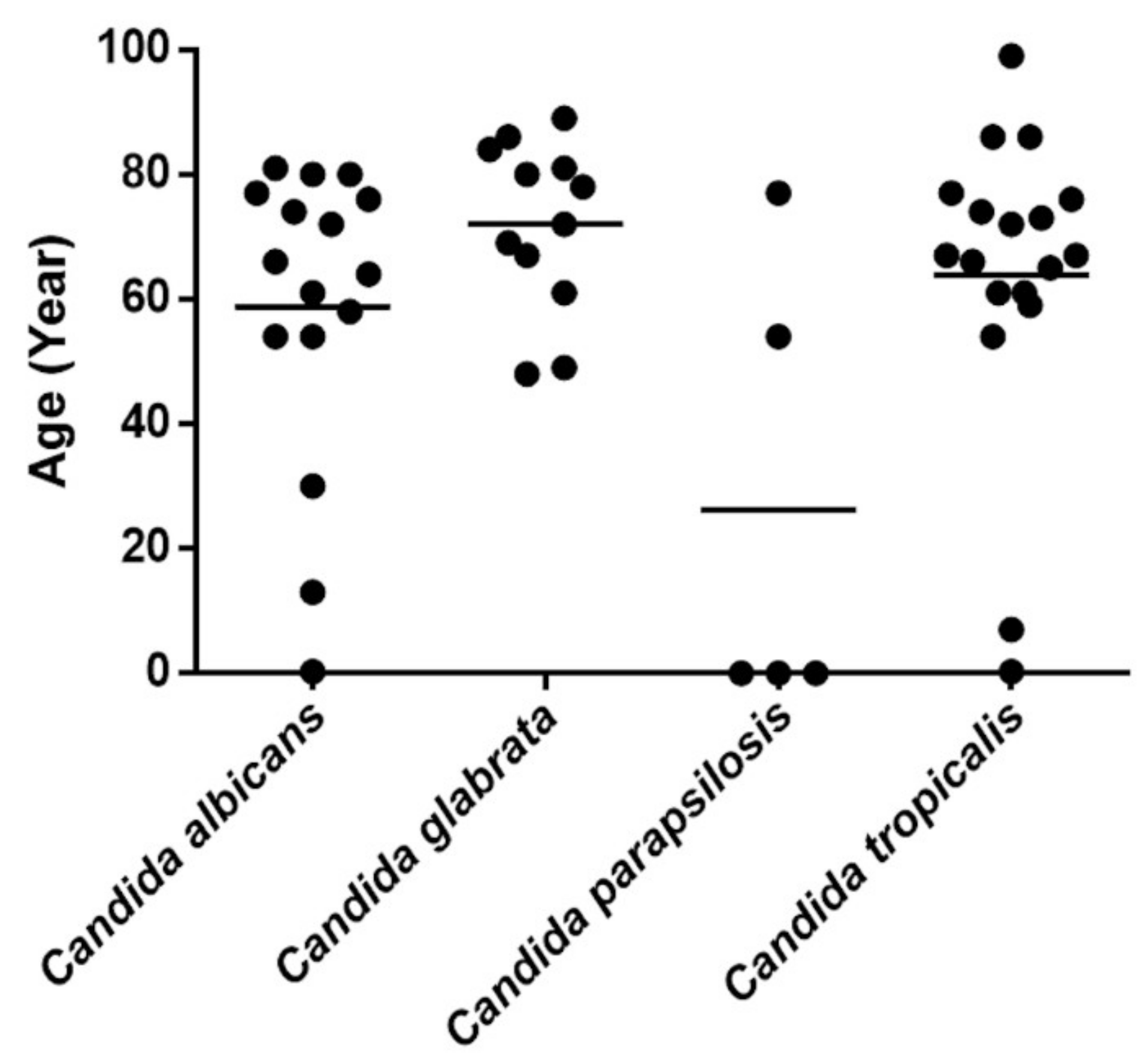
3.1. Candida Species Distribution among Patients with Candidemia
3.2. Antifungal Susceptibility Testing
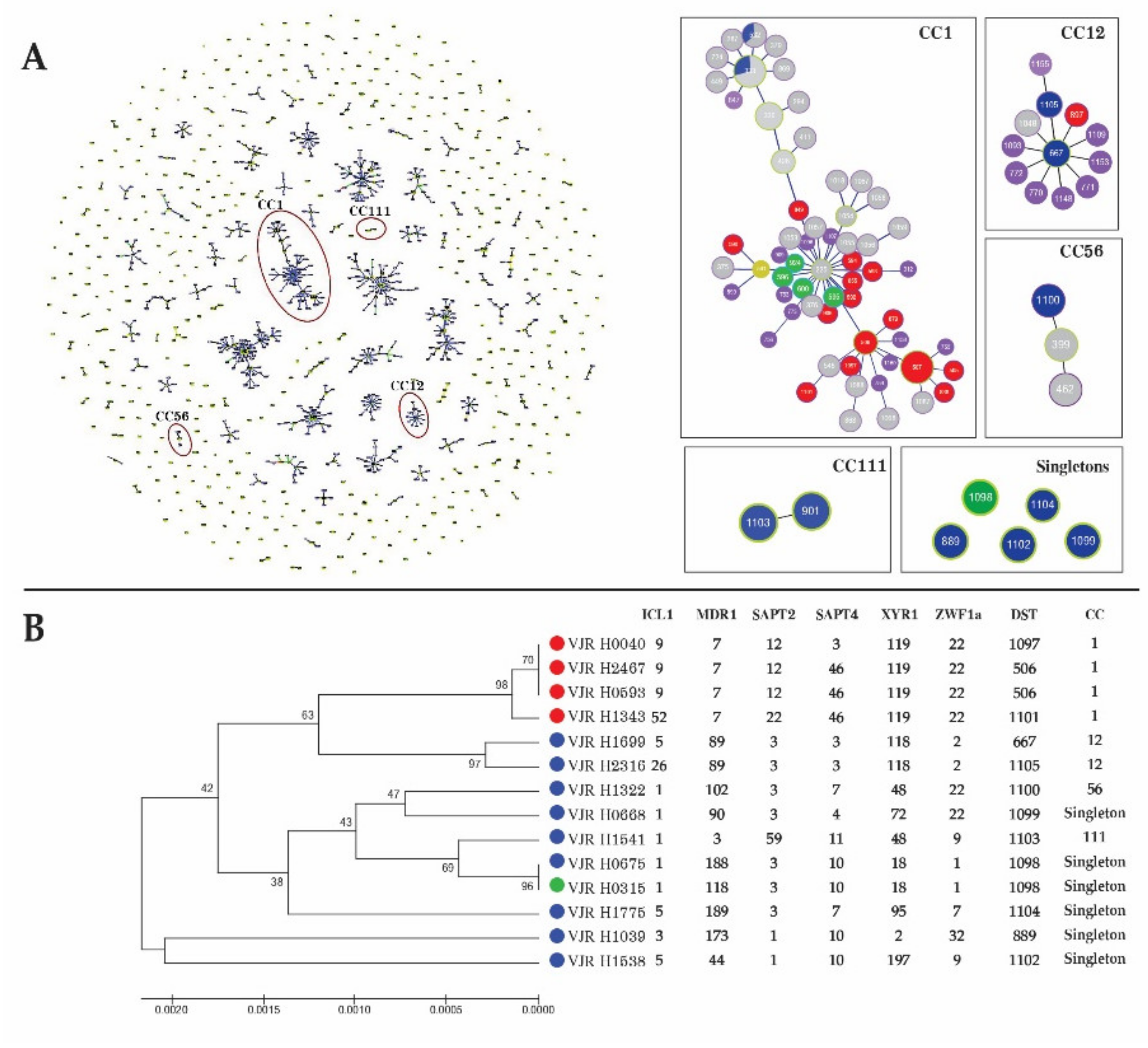
3.3. MLST of C. tropicalis
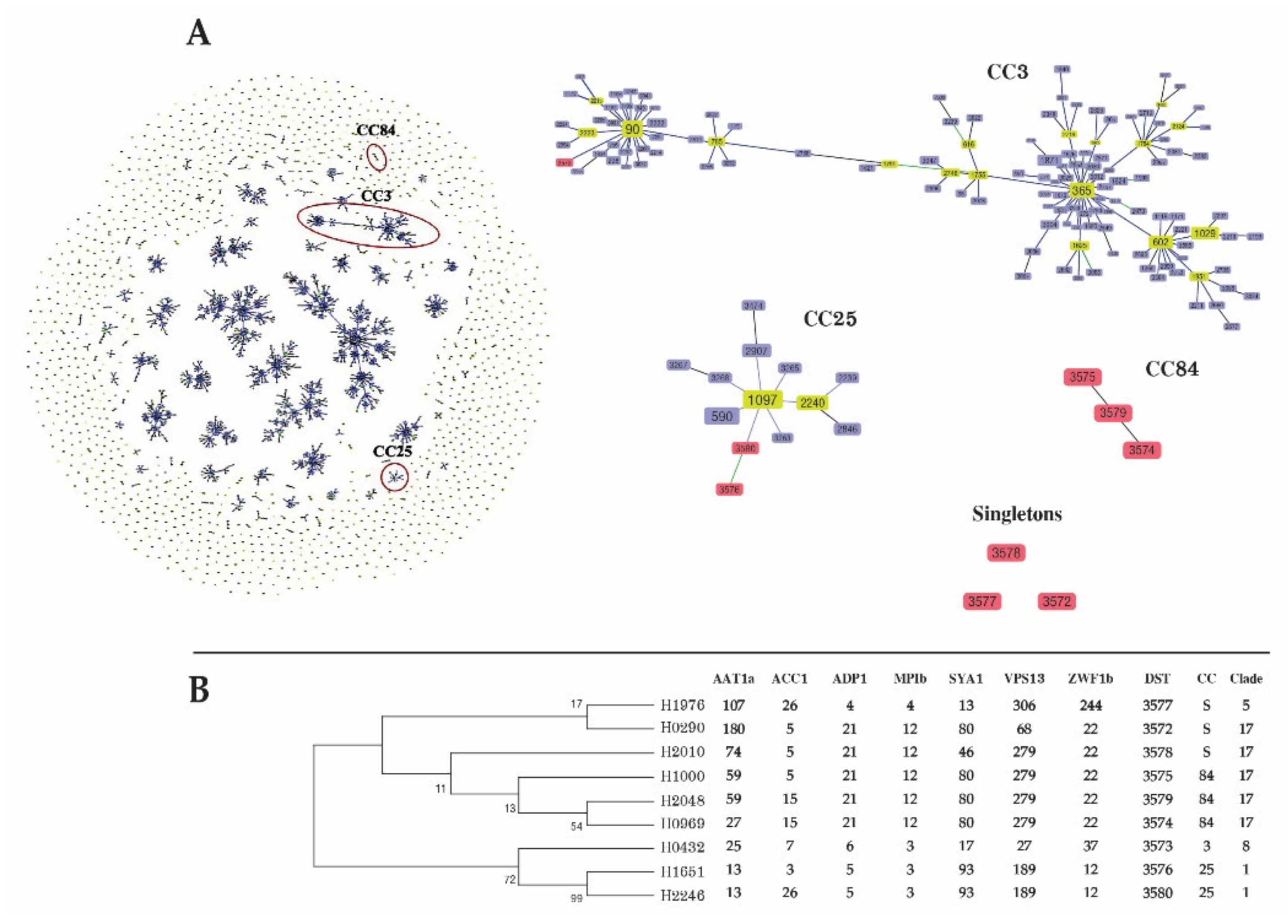
3.4. MLST of C. albicans
3.5. MLST of C. glabrata
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brown, G.D.; Denning, D.W.; Gow, N.A.R.; Levitz, S.M.; Netea, M.G.; White, T.C. Hidden Killers: Human Fungal Infections. Sci. Transl. Med. 2012, 4, 165rv13. [Google Scholar] [CrossRef] [Green Version]
- Méan, M.; Marchetti, O.; Calandra, T. Bench-to-bedside review: Candida infections in the intensive care unit. Crit. Care 2008, 12, 204. [Google Scholar] [CrossRef] [Green Version]
- Wisplinghoff, H.; Bischoff, T.; Tallent, S.M.; Seifert, H.; Wenzel, R.P.; Edmond, M.B. Nosocomial Bloodstream Infections in US Hospitals: Analysis of 24,179 Cases from a Prospective Nationwide Surveillance Study. Clin. Infect. Dis. 2004, 39, 309–317. [Google Scholar] [CrossRef] [Green Version]
- Yapar, N. Epidemiology and risk factors for invasive candidiasis. Ther. Clin. Risk Manag. 2014, 10, 95–105. [Google Scholar] [CrossRef] [Green Version]
- Sardi, J.C.O.; Scorzoni, L.; Bernardi, T.; Fusco-Almeida, A.M.; Mendes Giannini, M.J.S. Candida species: Current epidemiology, pathogenicity, biofilm formation, natural antifungal products and new therapeutic options. J. Med. Microbiol. 2013, 62 Pt 1, 10–24. [Google Scholar] [CrossRef]
- Richter, S.S.; Galask, R.P.; Messer, S.A.; Hollis, R.J.; Diekema, D.; Pfaller, M.A. Antifungal Susceptibilities of Candida Species Causing Vulvovaginitis and Epidemiology of Recurrent Cases. J. Clin. Microbiol. 2005, 43, 2155–2162. [Google Scholar] [CrossRef] [Green Version]
- Krcmery, V.; Barnes, A. Non-albicans Candida spp. causing fungaemia: Pathogenicity and antifungal resistance. J. Hosp. Infect. 2002, 50, 243–260. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols, a Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Bougnoux, M.-E.; Tavanti, A.; Bouchier, C.; Gow, N.A.R.; Magnier, A.; Davidson, A.D.; Maiden, M.C.J.; D’Enfert, C.; Odds, F.C. Collaborative Consensus for Optimized Multilocus Sequence Typing of Candida albicans. J. Clin. Microbiol. 2003, 41, 5265–5266. [Google Scholar] [CrossRef] [Green Version]
- Tavanti, A.; Davidson, A.D.; Johnson, E.M.; Maiden, M.C.J.; Shaw, D.J.; Gow, N.; Odds, F.C. Multilocus Sequence Typing for Differentiation of Strains of Candida tropicalis. J. Clin. Microbiol. 2005, 43, 5593–5600. [Google Scholar] [CrossRef] [Green Version]
- Dodgson, A.R.; Pujol, C.; Denning, D.W.; Soll, D.R.; Fox, A.J. Multilocus sequence typing of Candida glabrata reveals geographically enriched clades. J. Clin. Microbiol. 2003, 41, 5709–5717. [Google Scholar] [CrossRef] [Green Version]
- Tulyaprawat, O.; Pharkjaksu, S.; Chongtrakool, P.; Ngamskulrungroj, P. An Association of an eBURST Group with Triazole Resistance of Candida tropicalis Blood Isolates. Front. Microbiol. 2020, 11, 934. [Google Scholar] [CrossRef]
- Chai, Y.A.; Wang, Y.; Khoo, A.L.; Chan, F.Y.; Chow, C.; Kumarasinghe, G.; Singh, K.; Tambyah, P.A. Predominance of Candida tropicalis bloodstream infections in a Singapore teaching hospital. Med. Mycol. 2007, 45, 435–439. [Google Scholar] [CrossRef] [Green Version]
- Kang, S.J.; Kim, S.E.; Kim, U.J.; Jang, H.-C.; Park, K.-H.; Shin, J.H.; Jung, S.I. Clinical characteristics and risk factors for mortality in adult patients with persistent candidemia. J. Infect. 2017, 75, 246–253. [Google Scholar] [CrossRef]
- NaRanong, C.; Anunnatsiri, S.; Srigulbutr, S. Epidemiology and Antifungal Susceptibility in Patients with Candidemia in a University Hospital, Thailand. J. Med. Assoc. Thai. 2020, 103, 1048–1056. [Google Scholar] [CrossRef]
- Kothalawala, M.; Jayaweera, J.; Arunan, S.; Jayathilake, A. The emergence of non-albicans candidemia and evaluation of HiChrome Candida differential agar and VITEK2 YST(R) platform for differentiation of Candida bloodstream isolates in teaching hospital Kandy, Sri Lanka. BMC Microbiol. 2019, 19, 136. [Google Scholar] [CrossRef]
- Xiao, Z.; Wang, Q.; Zhu, F.; An, Y. Epidemiology, species distribution, antifungal susceptibility and mortality risk factors of candidemia among critically ill patients: A retrospective study from 2011 to 2017 in a teaching hospital in China. Antimicrob. Resist. Infect. Control 2019, 8, 89. [Google Scholar] [CrossRef]
- Lamoth, F.; Lockhart, S.R.; Berkow, E.L.; Calandra, T. Changes in the epidemiological landscape of invasive candidiasis. J. Antimicrob. Chemother. 2018, 73, i4–i13. [Google Scholar] [CrossRef] [Green Version]
- Morita, K.; Honda, A.; Koya, J.; Toyama, K.; Ikeda, M.; Misawa, Y.; Okugawa, S.; Nakamura, F.; Moriya, K.; Kurokawa, M. Three cases of Candida fermentati fungemia following hematopoietic stem cell transplantation. J. Infect. Chemother. 2018, 24, 576–578. [Google Scholar] [CrossRef] [PubMed]
- Borman, A.M.; Petch, R.; Linton, C.J.; Palmer, M.; Bridge, P.D.; Johnson, E.M. Candida nivariensis, an Emerging Pathogenic Fungus with Multidrug Resistance to Antifungal Agents. J. Clin. Microbiol. 2008, 46, 933–938. [Google Scholar] [CrossRef] [Green Version]
- Güler, N.C.; Tosun, I.; Aydin, F. The identification of Meyerozyma guilliermondii from blood cultures and surveillance samples in a university hospital in Northeast Turkey: A ten-year survey. J. Mycol. Méd. 2017, 27, 506–513. [Google Scholar] [CrossRef]
- Kett, D.H.; Azoulay, E.; Echeverria, P.M.; Vincent, J.-L. Candida bloodstream infections in intensive care units: Analysis of the extended prevalence of infection in intensive care unit study. Crit. Care Med. 2011, 39, 665–670. [Google Scholar] [CrossRef] [Green Version]
- Dimopoulos, G.; Flevari, A.; Theodorakopoulou, M.; Velegraki, A.; Armaganidis, A. Treatment of invasive candidiasis in the elderly: A review. Clin. Interv. Aging 2013, 8, 1199–1208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dimopoulos, G.; Paiva, J.-A.; Meersseman, W.; Pachl, J.; Grigoras, I.; Sganga, G.; Montravers, P.; Auzinger, G.; Sá, M.B.; Miller, P.J.; et al. Efficacy and safety of anidulafungin in elderly, critically ill patients with invasive Candida infections: A post hoc analysis. Int. J. Antimicrob. Agents 2012, 40, 521–526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muñoz, P.; Giannella, M.; Fanciulli, C.; Guinea, J.; Valerio, M.; Rojas, L.; Rodríguez-Créixems, M.; Bouza, E. Candida tropicalis fungaemia: Incidence, risk factors and mortality in a general hospital. Clin. Microbiol. Infect. 2011, 17, 1538–1545. [Google Scholar] [CrossRef] [Green Version]
- Leroy, O.; Gangneux, J.-P.; Montravers, P.; Mira, J.-P.; Gouin, F.; Sollet, J.-P.; Carlet, J.; Reynes, J.; Rosenheim, M.; Regnier, B.; et al. Epidemiology, management, and risk factors for death of invasive Candida infections in critical care: A multicenter, prospective, observational study in France (2005–2006). Crit. Care Med. 2009, 37, 1612–1618. [Google Scholar] [CrossRef]
- Deorukhkar, S.C.; Saini, S.; Mathew, S. Virulence factors contributing to pathogenicity of Candida tropicalis and its antifungal susceptibility profile. Int. J. Microbiol. 2014, 2014, 456878. [Google Scholar] [CrossRef] [Green Version]
- Gupta, A.; Gupta, A.; Varma, A. Candida glabrata candidemia: An emerging threat in critically ill patients. Indian J. Crit. Care Med. 2015, 19, 151–154. [Google Scholar] [CrossRef] [PubMed]
- Baronia, A.K.; Ahmed, A. Comments on “Candida glabrata candidemia, an emerging threat in critically ill patients”. Indian J. Crit. Care Med. 2015, 19, 294–295. [Google Scholar] [CrossRef] [Green Version]
- Hedderwick, S.A.; Wan, J.Y.; Bradley, S.F.; Sangeorzan, J.A.; Terpenning, M.S.; Kauffman, C.A. Risk Factors for Colonization with Yeast Species in a Veterans Affairs Long-term Care Facility. J. Am. Geriatr. Soc. 1998, 46, 849–853. [Google Scholar] [CrossRef] [Green Version]
- Pappas, P.G.; Rex, J.; Lee, J.; Hamill, R.J.; Larsen, R.A.; Powderly, W.; Kauffman, C.A.; Hyslop, N.; Mangino, J.E.; Chapman, S.; et al. A Prospective Observational Study of Candidemia: Epidemiology, Therapy, and Influences on Mortality in Hospitalized Adult and Pediatric Patients. Clin. Infect. Dis. 2003, 37, 634–643. [Google Scholar] [CrossRef] [Green Version]
- Tan, T.Y.; Hsu, L.Y.; Alejandria, M.M.; Chaiwarith, R.; Chinniah, T.; Chayakulkeeree, M.; Choudhury, S.; Chen, Y.H.; Shin, J.H.; Kiratisin, K.; et al. Antifungal susceptibility of invasive Candida bloodstream isolates from the Asia-Pacific region. Med. Mycol. 2016, 54, 471–477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Metin, D.Y.; Hilmioglu-Polat, S.; Samlioglu, P.; Doganay-Oflazoglu, B.; Inci, R.; Tumbay, E. Evaluation of Antifungal Susceptibility Testing with Microdilution and Etest Methods of Candida Blood Isolates. Mycopathologia 2011, 172, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Taj-Aldeen, S.J.; Kolecka, A.; Boesten, R.; Alolaqi, A.; Almaslamani, M.; Chandra, P.; Meis, J.F.; Boekhout, T. Epidemiology of candidemia in Qatar, the Middle East: Performance of MALDI-TOF MS for the identification of Candida species, species distribution, outcome, and susceptibility pattern. Infection 2013, 42, 393–404. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.P.; Miranda, I.; Lisboa, C.; Pina-Vaz, C.; Rodrigues, A.G. Prevalence, Distribution, and Antifungal Susceptibility Profiles of Candida parapsilosis, C. orthopsilosis, and C. metapsilosis in a Tertiary Care Hospital. J. Clin. Microbiol. 2009, 47, 2392–2397. [Google Scholar] [CrossRef] [Green Version]
- Miyazaki, H.; Miyazaki, Y.; Geber, A.; Parkinson, T.; Hitchcock, C.; Falconer, D.J.; DWard, D.J.; Marsden, K.; Bennett, J.B. Fluconazole resistance associated with drug efflux and increased transcription of a drug transporter gene, PDH1, in Candida glabrata. Antimicrob. Agents Chemother. 1998, 42, 1695–1701. [Google Scholar] [CrossRef] [Green Version]
- Barchiesi, F.; Di Francesco, L.F.; Arzeni, D.; Caselli, F.; Gallo, D.; Scalise, G. Electrophoretic karyotyping and triazole susceptibility of Candida glabrata clinical isolates. Eur. J. Clin. Microbiol. Infect. Dis. 1999, 18, 184–187. [Google Scholar] [CrossRef]
- You, L.; Qian, W.; Yang, Q.; Mao, L.; Zhu, L.; Huang, X.; Jin, J.; Meng, H. Affiliations expand ERG11 gene mutations and MDR1 upregulation confer pan-azole resistance in Candida tropicalis causing disseminated candidiasis in an acute lymphoblastic leukemia patient on posaconazole prophylaxis. Antimicrob. Agents Chemother. 2017, 61, e02496-16. [Google Scholar] [CrossRef] [Green Version]
- Jin, L.; Cao, Z.; Wang, Q.; Wang, Y.; Wang, X.; Chen, H.; Wang, H. MDR1 overexpression combined with ERG11 mutations induce high-level fluconazole resistance in Candida tropicalis clinical isolates. BMC Infect. Dis. 2018, 18, 162. [Google Scholar] [CrossRef] [Green Version]
- Oxman, D.A.; Chow, J.K.; Frendl, G.; Hadley, S.; Hershkovitz, S.; Ireland, P.; McDermott, L.A.; Tsai, K.; Marty, F.; Kontoyiannis, D.P.; et al. Candidaemia associated with decreased in vitro fluconazole susceptibility: Is Candida speciation predictive of the susceptibility pattern? J. Antimicrob. Chemother. 2010, 65, 1460–1465. [Google Scholar] [CrossRef] [Green Version]
- Pfaller, M.A.; Jones, R.N.; Castanheira, M. Regional data analysis of Candida non-albicans strains collected in United States medical sites over a 6-year period, 2006–2011. Mycoses 2014, 57, 602–611. [Google Scholar] [CrossRef]
- Tortorano, A.M.; Prigitano, A.; Lazzarini, C.; Passera, M.; Deiana, M.L.; Cavinato, S.; De Luca, C.; Grancini, A.; Cascio, G.L.; Ossi, C.; et al. A 1-year prospective survey of candidemia in Italy and changing epidemiology over one decade. Infection 2013, 41, 655–662. [Google Scholar] [CrossRef]
- Faria-Ramos, I.; Neves-Maia, J.; Ricardo, E.; Santos-Antunes, J.; Silva, A.T.; Costa-De-Oliveira, S.; Cantón, E.; Rodrigues, A.G.; Pina-Vaz, C. Species distribution and in vitro antifungal susceptibility profiles of yeast isolates from invasive infections during a Portuguese multicenter survey. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 2241–2247. [Google Scholar] [CrossRef]
- Guinea, J.; Zaragoza, O.; Escribano, P.; Mazuelos, E.M.; Pemán, J.; Sanchez-Reus, F.; Cuenca-Estrella, M. Molecular Identification and Antifungal Susceptibility of Yeast Isolates Causing Fungemia Collected in a Population-Based Study in Spain in 2010 and 2011. Antimicrob. Agents Chemother. 2013, 58, 1529–1537. [Google Scholar] [CrossRef] [Green Version]
- Wiederhold, N.P. Echinocandin Resistance in Candida Species: A Review of Recent Developments. Curr. Infect. Dis. Rep. 2016, 18, 42. [Google Scholar] [CrossRef] [PubMed]
- Chou, H.-H.; Lo, H.-J.; Chen, K.-W.; Liao, M.-H.; Li, S.-Y. Multilocus sequence typing of Candida tropicalis shows clonal cluster enriched in isolates with resistance or trailing growth of fluconazole. Diagn. Microbiol. Infect. Dis. 2007, 58, 427–433. [Google Scholar] [CrossRef]
- Wang, Q.; Li, C.; Tang, D.; Tang, K. Molecular epidemiology of Candida tropicalis isolated from urogenital tract infections. MicrobiologyOpen 2020, 9, 1121. [Google Scholar] [CrossRef]
- Fan, X.; Xiao, M.; Liao, K.; Kudinha, T.; Wang, H.; Zhang, L.; Hou, X.; Kong, F.; Xu, Y.-C. Notable Increasing Trend in Azole Non-susceptible Candida tropicalis Causing Invasive Candidiasis in China (August 2009 to July 2014): Molecular Epidemiology and Clinical Azole Consumption. Front. Microbiol. 2017, 8, 464. [Google Scholar] [CrossRef] [PubMed]
- Lyon, G.M.; Karatela, S.; Sunay, S.; Adiri, Y. Antifungal Susceptibility Testing of Candida Isolates from the Candida Surveillance Study. J. Clin. Microbiol. 2010, 48, 1270–1275. [Google Scholar] [CrossRef] [Green Version]
- Odds, F.C.; Bougnoux, M.-E.; Shaw, D.J.; Bain, J.M.; Davidson, A.D.; Diogo, D.; Jacobsen, M.D.; Lecomte, M.; Li, S.-Y.; Tavanti, A.; et al. Molecular Phylogenetics of Candida albicans. Eukaryot. Cell 2007, 6, 1041–1052. [Google Scholar] [CrossRef] [Green Version]
- Shin, J.H.; Bougnoux, M.-E.; D’Enfert, C.; Kim, S.H.; Moon, C.-J.; Joo, M.Y.; Lee, K.; Kim, M.-N.; Lee, H.S.; Shin, M.G.; et al. Genetic Diversity among Korean Candida albicans Bloodstream Isolates: Assessment by Multilocus Sequence Typing and Restriction Endonuclease Analysis of Genomic DNA by Use of BssHII. J. Clin. Microbiol. 2011, 49, 2572–2577. [Google Scholar] [CrossRef] [Green Version]
- Ropars, J.; Maufrais, C.; Diogo, D.; Marcet-Houben, M.; Perin, A.; Sertour, N.; Mosca, K.; Permal, E.; Laval, G.; Bouchier, C.; et al. Gene flow contributes to diversification of the major fungal pathogen Candida albicans. Nat. Commun. 2018, 9, 2253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, A.C.; Hickma, M.A. Host-induced genome instability rapidly generates phenotypic variation across Candida albicans strains and ploidy states. mSphere 2020, 5, e00433-20. [Google Scholar] [CrossRef] [PubMed]
- Odds, F.C. Molecular phylogenetics and epidemiology of Candida albicans. Futur. Microbiol. 2010, 5, 67–79. [Google Scholar] [CrossRef]
- Pham, L.; Pharkjaksu, S.; Chongtrakool, P.; Suwannakarn, K.; Ngamskulrungroj, P. A Predominance of Clade 17 Candida albicans Isolated from Hemocultures in a Tertiary Care Hospital in Thailand. Front. Microbiol. 2019, 10, 1194. [Google Scholar] [CrossRef] [Green Version]
- Furlaneto, M.C.; Goes, H.P.; Perini, H.F.; Dos Santos, R.C.; Furlaneto-Maia, L. How much do we know about hemolytic capability of pathogenic Candida species? Folia Microbiol. 2018, 63, 405–412. [Google Scholar] [CrossRef]
- Hou, X.; Xiao, M.; Chen, S.C.; Kong, F.; Wang, H.; Chu, Y.Z.; Kang, M.; Sun, Z.Y.; Zhi-Hu, H.; Li, R.Y.; et al. Molecular epidemiology and antifungal susceptibility of Candida glabrata in China (August 2009 to July 2014): A multi-center study. Front. Microbiol. 2017, 8, 880. [Google Scholar] [CrossRef]
- Byun, S.A.; Won, E.J.; Kim, M.-N.; Lee, W.G.; Lee, K.; Lee, H.S.; Uh, Y.; Healey, K.; Perlin, D.S.; Choi, M.J.; et al. Multilocus Sequence Typing (MLST) Genotypes of Candida glabrata Bloodstream Isolates in Korea: Association with Antifungal Resistance, Mutations in Mismatch Repair Gene (Msh2), and Clinical Outcomes. Front. Microbiol. 2018, 9, 1523. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carreté, L.; Ksiezopolska, E.; Pegueroles, C.; Gómez-Molero, E.; Saus, E.; Iraola-Guzmán, S.; Loska, D.; Bader, O.; Fairhead, C.; Gabaldón, T. Patterns of Genomic Variation in the Opportunistic Pathogen Candida glabrata Suggest the Existence of Mating and a Secondary Association with Humans. Curr. Biol. 2018, 28, 15–27.e7. [Google Scholar] [CrossRef] [Green Version]
- Amanloo, S.; Shams-Ghahfarokhi, P.M.; Ghahri, M.; Razzaghi-Abyaneh, M. Genotyping of clinical isolates of Candida glabrata from Iran by multilocus sequence typing and determination of population structure and drug resistance profile. Med. Mycol. 2017, 56, 207–215. [Google Scholar] [CrossRef]

| Yeast Isolate | No. of Isolates (%) | Age Mean ± SD | % Mortality |
|---|---|---|---|
| Candida albicans | 16 (29.63) | 58.77 ± 24.37 | 56.25 |
| Candida tropicalis | 18 (33.33) | 63.90 ± 24.53 | 61.11 |
| Candida glabrata | 12 (22.22) | 72 ± 13.717 | 33.33 |
| Candida parapsilosis | 5 (9.26) | 26.23 ± 36.76 | 40 |
| Candida nivariensis | 1 (1.85) | 54 | 0 |
| Candida guilliermondii | 1 (1.85) | 0.03 | 0 |
| Candida caribbica | 1 (1.85) | 59 | 0 |
| Candida Species | Antifungal Agent | MIC (ug/mL) | Isolates (%) | Isolates (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Range | MIC50 | MIC90 | S | SDD | I | R | wt | Non-wt | ||
| C. albicans | Anidulafungin | 0.015–0.12 | 0.12 | 0.12 | 100 | N/A | 0 | 0 | 0 | 0 |
| Micafungin | 0.008–0.015 | 0.008 | 0.015 | 100 | N/A | 0 | 0 | 0 | 0 | |
| Caspofungin | 0.03–0.12 | 0.12 | 0.12 | 100 | N/A | 0 | 0 | 0 | 0 | |
| Flucyctosine | 0.06–0.12 | 0.06 | 0.12 | N/A | N/A | N/A | N/A | N/A | N/A | |
| Posaconazole | 0.03 | 0.03 | 0.03 | N/A | N/A | N/A | N/A | 100 | 0 | |
| Voriconazole | 0.008–0.015 | 0.008 | 0.015 | 100 | N/A | 0 | 0 | N/A | N/A | |
| Itaconazole | 0.06–0.25 | 0.12 | 0.12 | N/A | N/A | N/A | N/A | N/A | N/A | |
| Fluconazole | 0.25–1 | 0.5 | 1 | 100 | N/A | 0 | 0 | N/A | N/A | |
| AmphotericinB | 0.25–0.5 | 0.5 | 0.5 | N/A | N/A | N/A | N/A | 100 | 0 | |
| C. glabrata | Anidulafungin | 0.03–0.25 | 0.12 | 0.25 | 80 | N/A | 20 | 0 | N/A | N/A |
| Micafungin | 0.15–0.03 | 0.015 | 0.03 | 100 | N/A | 0 | 0 | N/A | N/A | |
| Caspofungin | 0.12–0.25 | 0.25 | 0.25 | 20 | N/A | 80 | 0 | N/A | N/A | |
| Flucyctosine | 0.06–0.06 | 0.06 | 0.06 | N/A | N/A | N/A | N/A | N/A | N/A | |
| Posaconazole | 1.0–2.0 | 2.0 | 2 | N/A | N/A | N/A | N/A | 10 | 90 | |
| Voriconazole | 0.25–2 | 1 | 2 | N/A | N/A | N/A | N/A | 10 | 90 | |
| Itaconazole | 0.5–2 | 1 | 2 | N/A | N/A | N/A | N/A | 100 | 0 | |
| Fluconazole | 8–64 | 32 | 64 | N/A | 80 | N/A | 20 | N/A | N/A | |
| AmphotericinB | 0.5–1 | 0.5 | 1.0 | N/A | N/A | N/A | N/A | 100 | 0 | |
| C. tropicalis | Anidulafungin | 0.12–0.25 | 0.12 | 0.12 | 100 | N/A | 0 | 0 | N/A | N/A |
| Micafungin | 0.03–0.06 | 0.03 | 0.03 | 100 | N/A | 0 | 0 | N/A | N/A | |
| Caspofungin | 0.06–0.25 | 0.12 | 0.25 | 100 | N/A | 0 | 0 | N/A | N/A | |
| Flucyctosine | 0.06–0.12 | 0.06 | 0.06 | N/A | N/A | N/A | N/A | N/A | N/A | |
| Posaconazole | 0.12–2 | 0.25 | 2 | N/A | N/A | N/A | N/A | 14.29 | 85.71 | |
| Voriconazole | 0.12–8 | 0.12 | 8 | 57.14 | N/A | 14.29 | 28.5 | N/A | N/A | |
| Itaconazole | 0.25–4 | 0.25 | 2 | N/A | N/A | N/A | N/A | 71.43 | 28.57 | |
| Fluconazole | 2–256 | 2 | 256 | 64.29 | N/A | 7.14 | 28.57 | N/A | N/A | |
| AmphotericinB | 0.5–1 | 1 | 1 | N/A | N/A | N/A | N/A | 100 | 0 | |
| C. parapsilosis | Anidulafungin | 1–2 | 1 | 2 | 100 | N/A | 0 | 0 | N/A | N/A |
| Micafungin | 1–2 | 1 | 2 | 100 | N/A | 0 | 0 | N/A | N/A | |
| Caspofungin | 0.5 | 0.5 | 0.5 | 100 | N/A | 0 | 0 | N/A | N/A | |
| Flucyctosine | 0.06–0.12 | 0.12 | 0.12 | N/A | N/A | N/A | N/A | N/A | N/A | |
| Posaconazole | 0.03–0.12 | 0.06 | 0.12 | N/A | N/A | N/A | N/A | 100 | 0 | |
| Voriconazole | 0.015–0.06 | 0.015 | 0.12 | 100 | N/A | 0 | 0 | N/A | N/A | |
| Itaconazole | 0.06–0.25 | 0.06 | 0.25 | N/A | N/A | N/A | N/A | N/A | N/A | |
| Fluconazole | 0.5–4 | 1 | 2 | 50 | N/A | 50 | 0 | N/A | N/A | |
| AmphotericinB | 0.5 | 0.5 | 0.5 | N/A | N/A | N/A | N/A | 100 | 0 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boonsilp, S.; Homkaew, A.; Phumisantiphong, U.; Nutalai, D.; Wongsuk, T. Species Distribution, Antifungal Susceptibility, and Molecular Epidemiology of Candida Species Causing Candidemia in a Tertiary Care Hospital in Bangkok, Thailand. J. Fungi 2021, 7, 577. https://doi.org/10.3390/jof7070577
Boonsilp S, Homkaew A, Phumisantiphong U, Nutalai D, Wongsuk T. Species Distribution, Antifungal Susceptibility, and Molecular Epidemiology of Candida Species Causing Candidemia in a Tertiary Care Hospital in Bangkok, Thailand. Journal of Fungi. 2021; 7(7):577. https://doi.org/10.3390/jof7070577
Chicago/Turabian StyleBoonsilp, Siriphan, Anchalee Homkaew, Uraporn Phumisantiphong, Daranee Nutalai, and Thanwa Wongsuk. 2021. "Species Distribution, Antifungal Susceptibility, and Molecular Epidemiology of Candida Species Causing Candidemia in a Tertiary Care Hospital in Bangkok, Thailand" Journal of Fungi 7, no. 7: 577. https://doi.org/10.3390/jof7070577
APA StyleBoonsilp, S., Homkaew, A., Phumisantiphong, U., Nutalai, D., & Wongsuk, T. (2021). Species Distribution, Antifungal Susceptibility, and Molecular Epidemiology of Candida Species Causing Candidemia in a Tertiary Care Hospital in Bangkok, Thailand. Journal of Fungi, 7(7), 577. https://doi.org/10.3390/jof7070577






