Histoplasmosis Diagnosed in Europe and Israel: A Case Report and Systematic Review of the Literature from 2005 to 2020
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Eligibility Criteria and Study Selection
2.3. Data Collection and Evidence Summary
3. Results
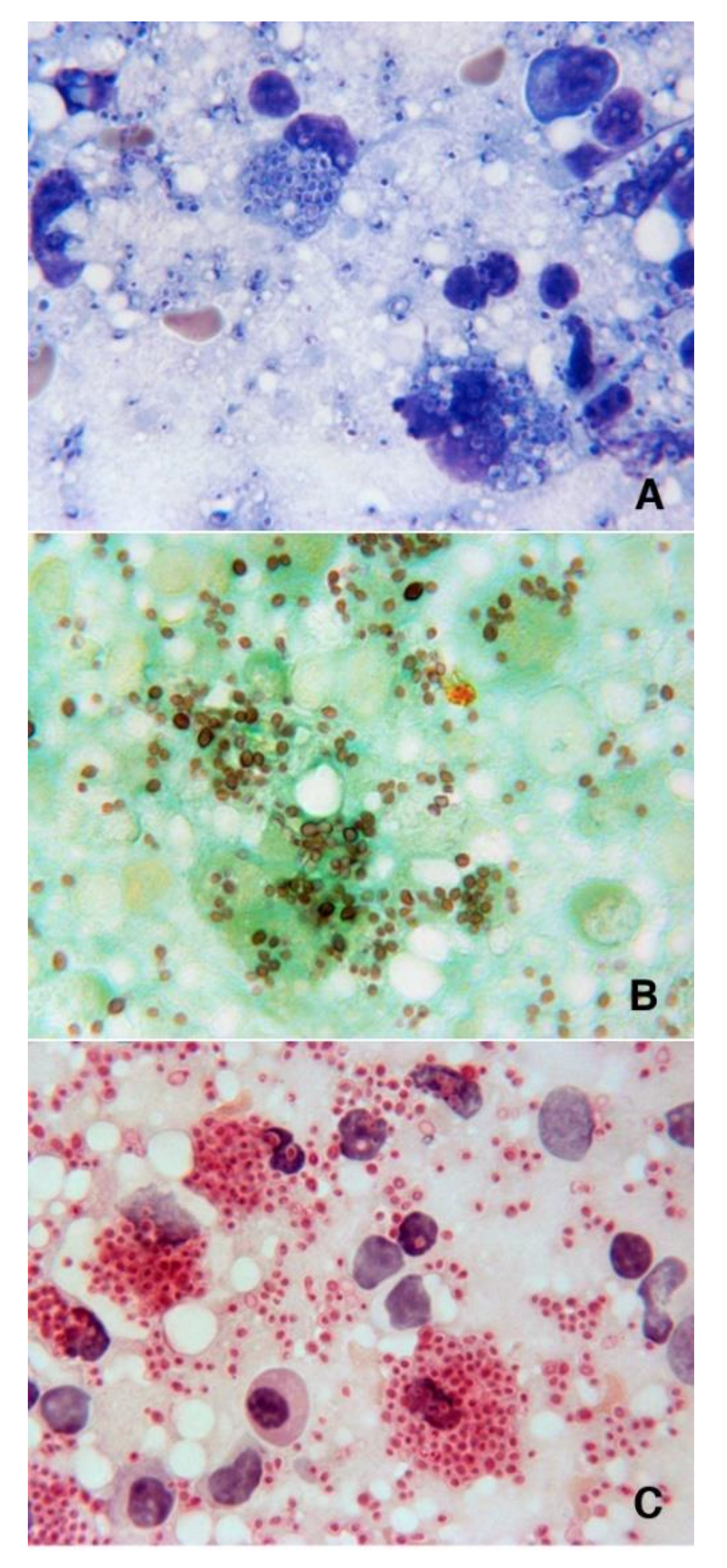
3.1. Case Report
3.2. Summary of the Literature
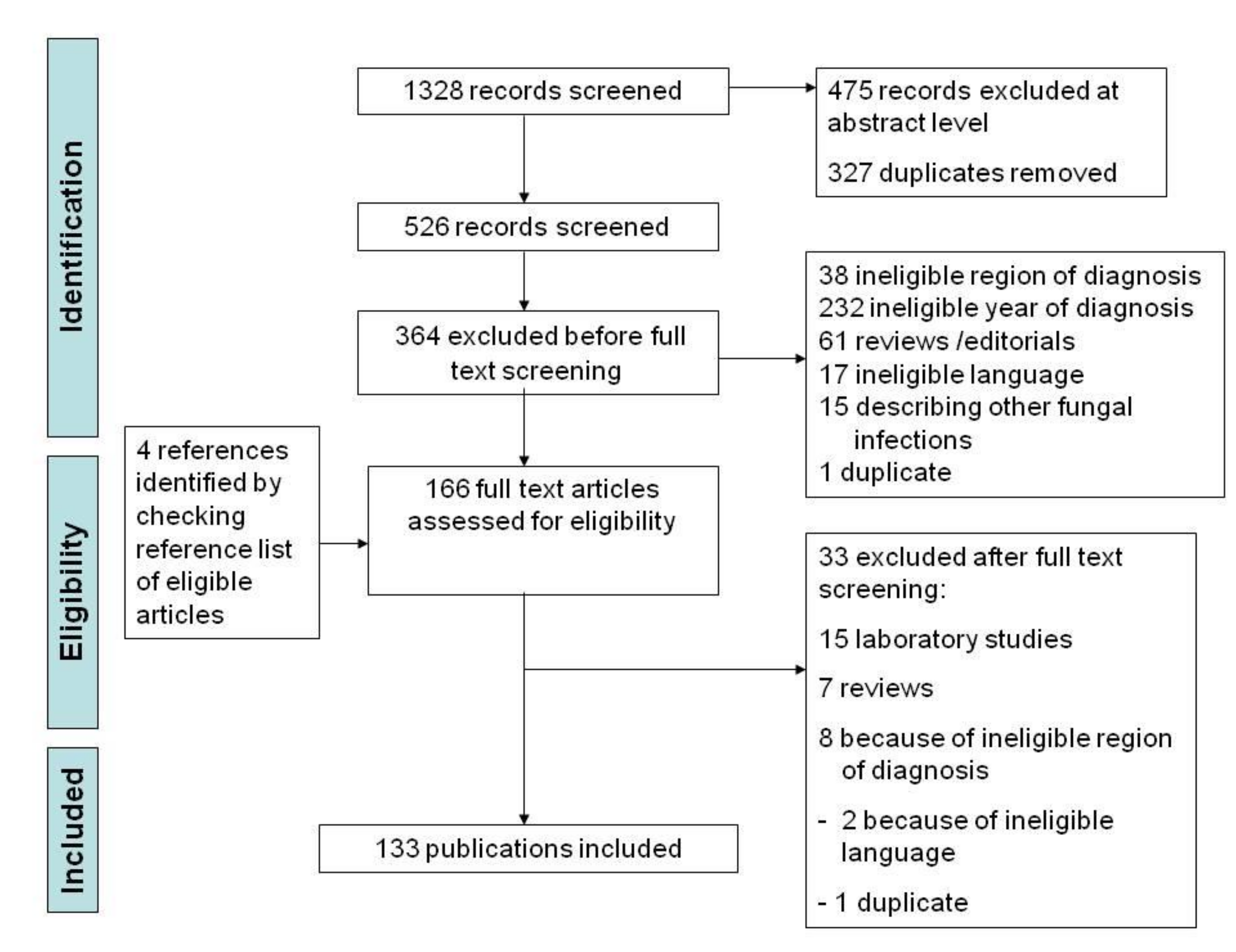
3.3. Study Selection and Characteristics
3.4. People Living with HIV (PLWH)
3.5. Patients with Other Immunocompromising Conditions (OIC)
3.6. Immunocompetent Patients
3.7. Histoplasma Capsulatum from Animals in Europe: The One Health Concept
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaufman, C.A. Histoplasmosis a clinical and laboratory update. Clin. Microbiol. Rev. 2007, 20, 115–132. [Google Scholar] [CrossRef] [PubMed]
- Drouhet, E. Les aspects cliniques de l’Histoplasmose Africaine. Ann. Soc. Belg. Med. Trop. 1972, 52, 391–406. [Google Scholar] [PubMed]
- Sepulveda, V.E.; Marquez, R.; Turissini, D.A.; Goldman, W.E.; Matute, D.R. Genome sequences reveal cryptic speciation in the human pathogen Histoplasma capsulatum. mBio 2017, 8, e01339-17. [Google Scholar] [CrossRef] [PubMed]
- Wheat, L.J.; Azar, M.M.; Bahr, N.C.; Spec, A.; Relich, R.F.; Hage, C. Histoplasmosis. Infect. Dis. Clin. N. Am. 2016, 30, 207–227. [Google Scholar] [CrossRef]
- Antinori, S. Histoplasma capsulatum: More widespread than previously thought. Am. J. Trop. Med. Hyg. 2014, 90, 982–983. [Google Scholar] [CrossRef]
- Bahr, N.C.; Antinori, S.; Wheat, L.J.; Sarosi, G. Histoplasmosis infections worldwide: Thinking outside the Ohio River Valley. Curr. Trop. Med. Rep. 2015, 2, 70–80. [Google Scholar] [CrossRef]
- Ashbee, H.R.; Evans, E.G.V.; Viviani, A.M.; Dupont, B.; Chryssanthou, E.; Surmont, I.; Tomosikova, A.; Vachkov, R.; Ener, B.; Zala, J.; et al. Histoplasmosis in Europe: Report on an epidemiological survey from the European Confederation of Medical Mycology Working Group. Med. Mycol. 2008, 46, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Antinori, S.; Magni, C.; Nebuloni, M.; Parravicini, C.; Corbellino, M.; Sollima, S.; Galimberti, L.; Ridolfo, A.L.; Wheat, J.L. Histoplasmosis among human immunodeficiency virus-infected people in Europe: Report of 4 cases and review of the literature. Medicine 2006, 85, 22–36. [Google Scholar] [CrossRef]
- Staffolani, S.; Buonfrate, D.; Angheben, A.; Gobbi, F.; Giorli, G.; Guerriero, M.; Bisoffi, Z.; Barchiesi, F. Acute histoplasmosis in immunocompetent travelers: A systematic review of the literature. BMC Infect. Dis. 2018, 18, 673. [Google Scholar] [CrossRef]
- Develoux, M.; Amona, F.M.; Hennequin, C. Histoplasmosis caused by Histoplasma capsulatum var. duboisii: A comprehensive review of cases from 1993 to 2019. Clin. Infect. Dis. 2020, ciaa1304. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic review and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, 97. [Google Scholar] [CrossRef]
- Molina-Morant, D.; Sanchez-Montalva, A.; Salvador, F.; Sao-Aviles, A.; Molina, I. Imported endemic mycoses in Spain: Evolution of hospitalized cases, clinical characteristics and correlation with migratory movements, 1997–2014. PLoS Negl. Trop. Dis. 2018, 12, e0006245. [Google Scholar] [CrossRef]
- Gascon, J.; Torres, J.M.; Jimenez, M.; Mejias, T.; Trivino, L.; Gobbi, F.; Quinto, L.; Puig, J.; Corachon, M. Histoplasmosis infection in Spanish travelers to Latin America. Eur. J. Clin. Microbiol. Infect. Dis. 2005, 24, 839–841. [Google Scholar] [CrossRef]
- Peigne, V.; Dromer, F.; Elie, C.; Lidove, O.; Lortholary, O.; French mycosis Study Group. Imported Acquired Immunodeficiency Syndrome-related histoplasmosis in metropolitan France: A comparison of pre-highly active anti-retroviral therapy and highly anti-retroviral therapy eras. Am. J. Trop. Med. Hyg. 2011, 85, 934–941. [Google Scholar] [CrossRef]
- Staffolani, S.; Riccardi, N.; Farina, C.; Lo Cascio, G.; Gulletta, M.; Gobbi, F.; Rodari, P.; Ursini, T.; Bertoli, G.; Ronzoni, N.; et al. Acute histoplasmosis in travelers: A retrospective study in an Italian referral center for tropical diseases. Pathog. Glob. Health 2020, 114, 40–45. [Google Scholar] [CrossRef]
- Segel, M.J.; Rozenman, J.; Lindsley, M.D.; Lachish, T.; Berkman, N.; Neuberger, A.; Schwartz, E. Histoplasmosis in Israeli travelers. Am. J. Trop. Med. Hyg. 2015, 92, 1168–1172. [Google Scholar] [CrossRef]
- Cosme, A.; Pardo, E.; Felipo, F.; Iribarren, J.A. Abdominal pain in a HIV-infected patient. Rev. Esp. Enferm. Dig. 2005, 97, 196–210. [Google Scholar] [CrossRef] [PubMed]
- Ferry, T.; Ponceau, B.; Gaultier, J.B.; Piens, M.A.; Biron, F.; Picot, S.; Chidiac, C.; Peyramond, D. Disseminated cryptococcosis and histoplasmosis co-infection in a HIV-infected woman in France. J. Infect. 2005, 51, e173–e176. [Google Scholar] [CrossRef] [PubMed]
- Matulionyte, R.; Garbino, J.; Uckay, I.; Lambert, J.F.; Hirschel, B. Disseminated histoplasmosis in an HIV-infected patient discovered by routine blood smear staining. Eur. J. Clin. Microbiol. Infect. Dis. 2005, 24, 361–363. [Google Scholar] [CrossRef] [PubMed]
- De Jose Gomez, M.I.; Orio Hernandez, M.; Bilbao Garagoitia, A.; Garcia Rodriguez, J.; Baquero Artigao, F. Histoplasmosis diseminada. An. Pediatr. 2005, 63, 249–252. [Google Scholar] [CrossRef]
- Martin Relloso, M.J.; Sanchez-Fayos, P.; Gonzalez Guirado, A.; Rico, L.; Porres, J.C. Colonic histoplasmosis in AIDS. Endoscopy 2005, 37, 1036. [Google Scholar] [CrossRef] [PubMed]
- Pistone, T.; Lacombe, K.; Poirot, J.L.; Girard, P.M.; Meynard, J.L. Imported concomitant coccidioidomycosis and histoplasmosis in an HIV-infected Colombian migrant in France. Trans. R. Soc. Trop. Med. Hyg. 2005, 99, 712–715. [Google Scholar] [CrossRef] [PubMed]
- Farina, C.; Rizzi, M.; Ricci, L.; Gobbi, E.; Caligaris, S.; Goglio, A. Imported and autochthonous histoplasmosis in Italy: New cases and old problems. Rev. Iberoam. Micol. 2005, 22, 169–171. [Google Scholar] [CrossRef]
- Buitrago, M.J.; Berenguer, J.; Mellado, E.; Rodriguez-Tudela, J.L.; Cuenca-Estrella, M. Detection of imported histoplasmosis in serum of HIV-infected patients using a real-time PCR-based assay. Eur. J. Clin. Microbiol. Infect. Dis. 2006, 25, 665–668. [Google Scholar] [CrossRef]
- Peters, E.J.G.; Kauffmann, R.H.; Blok, P. Fever and high lactate dehydrogenase in HIV-positive patients from the Antilles and Surinam: Histoplasmosis? Neth. J. Med. 2006, 64, 302–306. [Google Scholar]
- Breton, G.; Adle-Biassette, H.; Therby, A.; Ramanoelina, J.; Choudat, L.; Bissuel, F.; Huerre, M.; Dromer, F.; Dupont, B.; Lortholary, O. Immune reconstitution inflammatory syndrome in HIV-infected patients with disseminated histoplasmosis. Aids 2006, 20, 119–121. [Google Scholar] [CrossRef]
- Gil-Brusola, A.; Peman, J.; Santos, M.; Salavert, M.; Lacruz, J.; Gobernado, M. Disseminated histoplasmosis with hemophagocytic syndrome in a patient with AIDS: Description of one case and review of the Spanish literature. Rev. Iberoam. Micol. 2007, 24, 312–316. [Google Scholar] [CrossRef]
- Loulergue, P.; Bastides, F.; Baudouin, V.; Chandenier, J.; Mariani-Kurkdjian, P.; Dupont, B.; Viard, J.-P.; Dromer, F.; Lortholary, O. Literature review and case histories of Histoplasma capsulatum var. duboisii infections in HIV-infected patients. Emerg. Infect. Dis. 2007, 13, 1647–1652. [Google Scholar] [CrossRef]
- del Mar Lopez-Perezagua, M.; Martinez-Peinado, C.; Arjona-Zaragozi, F.J.; Pasquau-Liano, F. Linfadenitis cervical, pancytopenia y fiebre en paciente con infeccion por VIH. Enferm. Infecc. Microbiol. Clin. 2007, 25, 157–158. [Google Scholar] [CrossRef]
- Débat Zoguéreh, D.; Bigel, M.L.; Billy, C.; Perronne, V.; Richardin, F.; Granier, F. Disseminated histoplasmosis revealed by peripheral blood smear in an African immigrant with AIDS. Med. Malad. Infect. 2008, 38, 228–230. [Google Scholar] [CrossRef]
- Peppa, D.; Plumb, I.; du Parcq, J.; Taylor, S.; Miller, R.F. Gastrointestinal histoplasmosis in an HIV-infected patient living in a non-endemic area. Int. J. STD Aids 2008, 19, 864–865. [Google Scholar] [CrossRef] [PubMed]
- Pellaton, C.; Cavassini, M.; Jaton-Ogay, K.; Carron, P.-N.; Christen-Zaech, S.; Calandra, T.; Bille, J.; Hauser, P.M. Histoplasma capsulatum var. duboisii infection in a patient with AIDS: Rapid diagnosing using polymerase chain reaction-sequencing. Diagn. Microbiol. Infect. Dis. 2009, 64, 85–89. [Google Scholar] [CrossRef]
- Norman, F.F.; Martin-Davila, P.; Fortun, J.; Dronda, F.; Quereda, C.; Sanchez-Sousa, A.; Lopez-Velez, R. Imported histoplasmosis: Two distinct profiles in travelers and immigrants. J. Travel Med. 2009, 16, 258–262. [Google Scholar] [CrossRef]
- Nyffenegger, L.; Abbas, M.; Gex, G.; Boffi, E.; Schrenzel, J.; Bouchuiguir-Wafa, K.; Hirschel, B. Cas importés d’histoplasmose. Rev. Med. Suisse. 2009, 5, 2418–2423. [Google Scholar]
- Fritzsche, C.; Loebermann, M.; Aepinus, C.; Bolz, M.; Barten, M.; Reisinger, E.C. Vaginal ulceration and local lymphadenopathy in an African immigrant. Clin. Infect. Dis. 2009, 48, 493–494. [Google Scholar] [CrossRef]
- Pineau, S.; Talarmin, J.-P.; Morio, F.; Grossi, O.; Boutoille, D.; Léanté, F.; le Pape, P.; Gay-Andrieu, F.; Miegeville, M.; Raffi, F. Contribution of molecular biology and Aspergillus galactomannan antigen assay for the diagnosis of histoplasmosis. Med. Malad. Infect. 2010, 40, 541–543. [Google Scholar] [CrossRef]
- Ala-Kauhaluoma, M.; Aho, I.; Ristola, M.; Karma, A. Involvement of intraocular structures in disseminated histoplasmosis. Acta Ophthalmol. 2010, 88, 493–496. [Google Scholar] [CrossRef]
- Buitrago, M.J.; Bernal-Martinez, L.; Castelli, M.V.; Rodriguez-Tudela, J.L.; Cuenca-Estrella, M. Histoplasmosis and paracoccidioidomycosis in a non-endemic area: A review of cases and diagnosis. J. Travel Med. 2011, 18, 26–33. [Google Scholar] [CrossRef]
- Gomez-Moyano, E.; Crespo-Erchiga, V.; Vera-Casano, A. Probable primary cutaneous histoplasmosis in a patient infected with HIV. J. Mycol. Med. 2011, 21, 210–213. [Google Scholar] [CrossRef]
- Bourgeois, N.; Douard-Enault, C.; Reynes, J.; Lechiche, C.; Basset, D.; Rispall, P.; Lachaud, L. Seven imported histoplasmosis cases due to Histoplasma capsulatum var. capsulatum: From few weeks to more than three decades asymptomatic period. J. Mycol. Med. 2011, 21, 19–23. [Google Scholar] [CrossRef]
- Inojosa, W.; Rossi, M.C.; Laurino, L.; Giobbia, M.; Fuser, R.; Carniato, A.; Farina, F.; De Pieri, M.; Baldasso, F.; Forner, G.; et al. Progressive disseminated histoplasmosis among human immunodeficiency virus-infected patients from West-Africa. Report of four imported cases in Italy. Infez. Med. 2011, 7, 49–55. [Google Scholar]
- Borges-Costa, J.; Marques, T.; Soares-Almeida, L.; Sacramento-Marques, M. Progressive disseminated histoplasmosis as a presentation of AIDS in a patient from the Congo: The role of skin biopsy. Trop. Doct. 2011, 41, 251–252. [Google Scholar] [CrossRef]
- Sanmani, L.; Randall, C.J.; Palfrey, J.; Rowen, D. Hoarseness of voice in an AIDS patient: A rare presenting feature of disseminated histoplasmosis. Int. J. STD AIDS. 2011, 22, 115–116. [Google Scholar] [CrossRef] [PubMed]
- Navascués, A.; Rodriguez, I.; Reparaz, J.; Gil-Setas, A.; Martinez Penuela, J.M. Descripcion de quadro casos de histoplasmosis importada en Navarra. Rev. Iberoam. Micol. 2011, 28, 194–197. [Google Scholar] [CrossRef]
- Portillo, M.E.; Plasencia, V.; Nolla, J.; Segura, C. Severe acute respiratory failure in an HIV-infected patient from Panama. Enferm. Infecc. Microbiol. Clin. 2011, 29, 783–785. [Google Scholar]
- Scarlata, F.; Imburgia, C.; Trizzino, M.; Titone, L. Istoplasmosi a presentazione cutanea simil-lepromatosa in un paziente africano HIV+. Infez. Med. 2012, 3, 211–213. [Google Scholar]
- Riviere, S.; Denis, B.; Bougnoux, M.-E.; Lanternier, F.; Lecuit, M.; Lortholary, O. Serum Aspergillus galactomannan for the management of disseminated histoplasmosis in AIDS. Am. J. Trop. Med. Hyg. 2012, 87, 303–305. [Google Scholar] [CrossRef] [PubMed]
- Mambie, A.; Pasquet, A.; Melliez, H.; Bonne, S.; Blanc, A.-L.; Patoz, P.; Ajana, F. A case of immune reconstitution inflammatory syndrome related to a disseminated histoplasmosis in an HIV-1 infected patients. Aids 2013, 27, 2170–2171. [Google Scholar] [CrossRef]
- Tajan, J.; Espasa, M.; Sala, M.; Navarro, M.; Font, B.; Gonzalez-Martin, J.; Segura, F. Disseminated infection by Mycobacterium sherrisii and Histoplasma capsulatum in an African HIV-infected patient. Am. J. Trop. Med. Hyg. 2013, 88, 914–917. [Google Scholar] [CrossRef]
- Le Gal, S.; Damiani, C.; Virmaux, M.; Schmit, J.-L.; Totet, A.; Nevez, G. A 39-year-old man with Human Immunodeficiency virus infection presenting with an alveolo-interstitial pulmonary syndrome. J. Clin. Microbiol. 2013, 51, 3165. [Google Scholar] [CrossRef]
- Shah, N.; Owen, L.; Bhagani, S. “Occam Scissors”: Opportunistic infections in advanced HIV infection. BMJ Case Rep. 2013, 009544. [Google Scholar] [CrossRef]
- Martin-Iguacel, R.; Kurtzhals, J.; Jouvion, G.; Nielsen, S.D.; Llibre, J.M. Progressive disseminated histoplasmosis in the HIV population in Europe in the HAART era. Case report and literature review. Infection 2014, 42, 611–620. [Google Scholar] [CrossRef]
- Therby, A.; Polotzanu, O.; Khau, D.; Monnier, S.; Greder Belan, A.; Eloy, O. Interet du dosage de l’antigene galactomannane dans le diagnostic et le suivi de l’histoplasmose disséminée à Histoplasma capsulatum var. duboisii au cours du VIH: Enseignement à partir d’un cas Clinique. J. Mycol. Med. 2014, 24, 166–170. [Google Scholar]
- Stevenson, J.; Taylor, C. Posterior reversible encephalopathy syndrome in disseminated histoplasmosis and advanced HIV infection. Int. J. STD AIDS 2014, 25, 611–613. [Google Scholar] [CrossRef] [PubMed]
- De Hoog, S.H.; Blok, W.L.; van Ogtrop, M.L.; van den Berk, G.E.L. An unusual peripheral blood smear. Neth. J. Med. 2014, 72, 332–333. [Google Scholar] [PubMed]
- Amadori, F.; Doria, R.; Gemignani, G.; Flammini, S.; Leonildi, A.; Ciancia, E.M.; Sanguinetti, M.; Menichetti, F. Istoplasmosi: La multiforme faccia di una patologia. Infez. Med. 2015, 1, 63–68. [Google Scholar]
- Delfino, E.; Di Biagio, A.; Chandrapatham, K.; Viscoli, C.; Prinapori, R. Disseminated histoplasmosis with mucocutaneous immune reconstitution inflammatory syndrome in an HIV-infected patient. AIDS Res. Hum. Retrovir. 2015, 31, 274–275. [Google Scholar] [CrossRef]
- Almeida-Silva, F.; Serra Damasceno, L.; Buitrago Serna, M.J.; Valero, C.; Pereira Quintella, L.; Almeida-Paes, R.; de Medeiros Muniz, M.; Zancope-Oliveira, R.M. Multiple opportunistic fungal infections in an individual with severe HIV disease: A case report. Rev. Iberoam. Micol. 2016, 33, 118–121. [Google Scholar] [CrossRef]
- Gomez-Espejio, S.M.; Olalla-Sierra, J.; Marì-Jiménez, P.; Pereda-Salguero, T.; Pérez-Stachowski, J.; de la Torre Lima, J.; del Arco Jiménez, A.; Prada-Pardal, J.L. Reconstitution inflammatory syndrome like reactive hemophagocytic syndrome associated with disseminated histoplasmosis in a HIV patient. Mycopathologia 2017, 182, 767–770. [Google Scholar] [CrossRef]
- Lehur, A.C.; Zielenski, M.; Pluvy, J.; Grégoire, V.; Diamantis, S.; Bleibtreu, A.; Rioux, C.; Picard, A.; Vallois, D. Case of disseminated histoplasmosis in a HIV-infected patient revealed by nasal involvement with maxillary osteolysis. BMC Infect. Dis. 2017, 17, 328. [Google Scholar] [CrossRef]
- Zanotti, P.; Chirico, C.; Gulletta, M.; Ardighieri, L.; Casari, S.; Quiros Roldan, E.; Izzo, I.; Pinsi, G.; Lorenzin, G.; Facchetti, F.; et al. Disseminated histoplasmosis as AIDS-presentation. Case report and comprehensive review of current literature. Mediterr. J. Hematol. Infect. Dis. 2018, 10, e2018040. [Google Scholar] [CrossRef] [PubMed]
- Evrard, S.; Caprasse, P.; Gavage, P.; Vasbien, M.; Radermacher, J.; Hayette, M.-P.; Sacheli, R.; Van Esbroeck, M.; Cnops, L.; Firre, E.; et al. Disseminated histoplasmosis. Case report and review of the literature. Acta Clin. Bel. 2018, 73, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Valero, C.; Gago, S.; Monteiro, M.C.; Alastruey-Izquierdo, A.; Buitrago, M.J. African histoplasmosis: New clinical and microbiological insights. Med. Mycol. 2018, 56, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Le Meur, L.; Tantet, C.; Le, M.P.; Desselas, E.; Bonnal, C.; Lillo-Le-Louet, A.; Sonneville, R.; Massias, L.; Giraud, J.; Descamps, D.; et al. Serious neuropsychiatric adverse effects related to interaction between itraconazole and darunavir/ritonavir in an HIV-infected patient with cerebral histoplasmosis. J. Antimicrob. Chemother. 2018, 73, 1108–1110. [Google Scholar] [CrossRef]
- Papalini, C.; Belfiori, B.; Martino, G.; Papili, R.; Pitzurra, L.; Ascani, S.; Pasticci, M.B. An italian case of disseminated histoplasmosis associated with HIV. Case Rep. Infect. Dis. 2019, 2019. [Google Scholar] [CrossRef]
- Lebowitz, D.; Celi, L.; Nawej Tshikung, O.; Starobinski, M.; Chappuis, F.; Serratrice, J. Fever of un-Swiss origin. Presse Med. 2019, 48, 740–744. [Google Scholar] [CrossRef] [PubMed]
- Engelmann, E.W.M.; Posthuma, J.J.; Scholten, L.; Blankensteijn, L.L.; Boldewijn, M.B.; Gooszen, J.A.H. Gastrointestinal histoplasmosis mimicking peritonitis carcinomatosis. A rare case of an emergent surgical presentation of HIV de novo. J. Surg. Case Rep. 2019, 10, 1–4. [Google Scholar] [CrossRef]
- Bosch-Nicolau, P.; Salvador, F.; Sanchez-Montalva, A.; Sulleiro, E.; Burgos, J.; Molina, I. A case report of long treatment with itraconazole in a patient with chronic Chagas disease. BMC Infect. Dis. 2019, 19, 956. [Google Scholar] [CrossRef]
- Mussà, N.Y.; Ismail, S.; Carvalho, D. Disseminated histoplasmosis diagnosed in a bone marrow sample. Hematol. Transfus. Cell Ther. 2020. [Google Scholar] [CrossRef]
- Ara, J.; Matas, L.; Lauzurica, R.; Agraz, I.; Bayes, B.; Bonet, J.; Pérez, A.; Romero, R. Histoplasmosis en un transplantado renal. Nefrologia 2005, 25, 706–711. [Google Scholar]
- Meijer, J.A.A.; Sjogren, E.V.; Kuijper, E.; Verbist, B.M.; Visser, L.G. Necrotizing cervical lymphadenitis due to disseminated Histoplasma capsulatum infection. Eur. J. Clin. Microbiol. Infect. Dis. 2005, 24, 574–576. [Google Scholar] [CrossRef]
- Blanchard, C.; Nicolas, X.; Zagnoli, F.; Granier, H.; Talarmin, F.; Bellard, S. Miliaire cérébrale à Histoplasma capsulatum chez un patient séro-négatif pour le VIH. Rev. Neurol. 2007, 163, 740–742. [Google Scholar] [CrossRef]
- Braun-Falco, M.; Meiss, F. Cutaneous histoplasmosis. Lancet Infect. Dis. 2008, 8, 734. [Google Scholar] [CrossRef]
- Den Bakker, M.A.; Goemaere, N.N.T.; Severin, J.A.; Nouwen, J.L.; Verhagen, P.C.M.S. Histoplasma-associated inflammatory pseudotumor of the kidney mimicking renal carcinoma. Virchows Arch. 2009, 454, 229–232. [Google Scholar] [CrossRef][Green Version]
- Den Borst, M.H.; van Zeiji, J.H.; Grond, J.; Hoogendoorn, M. Histoplasmose bij een immuungecompromitteerde patient. Ned. Tijdschr. Geneeskd. 2010, 154, A1157. [Google Scholar]
- Van Koeveringe, M.P.; Brouwer, R.E. Histoplasma capsulatum reactivation with haemophagocytic syndrome in a patient with chronic lymphocytic leukaemia. Neth. J. Med. 2010, 68, 418–421. [Google Scholar]
- Letranchant, L.; Debourgogne, A.; Doco-Lecompte, T.; Contet-Audonneau, N.; May, T.; Machouart, M. Fatal Histoplasma capsulatum mitral endocarditis in a French patient treated for rheumatoid arthritis. Mycopathologia 2012, 173, 183–186. [Google Scholar] [CrossRef]
- Marques, N.; Lebre, A.; Marques, F.; Juliao, M.; Freitas, L.; Malcata, L.; Rabadao, E.; da Cunha, J.S. Isolated oral histoplasmosis presenting as fever of unknown origin in a Portuguese hemodialysis patient. Mycopathologia 2013, 176, 89–93. [Google Scholar] [CrossRef]
- Righi, E.; Lugano, M.; Assi, M.; Sartor, A.; De Carolis, E.; Crapis, M.; Cadeo, B.; Vella, A.; Sanguinetti, M.; Scarparo, C.; et al. Histoplasmosis in a lung transplant recipient from a nonendemic area. Transpl. Int. 2014, 27, e99–e101. [Google Scholar] [CrossRef]
- Ahumada, F.; Perez, D.; de Gorgolas, M.; Alvarez, B.; Rios, A.; Sanchez, A.; Villacampa, J.M. Subacute histoplasmosis with focal involvement of the epiglottis: Importance of differential diagnosis. Case Rep. Otoryngol. 2014, 2014, 235975. [Google Scholar] [CrossRef]
- Mota de Almeida, F.J.; Kivijarvi, K.; Roos, G.; Nylander, K. A case of disseminated histoplasmosis diagnosed after oral presentation in an old HIV-negative patient in Sweden. Gerodontology 2014, 32, 234–236. [Google Scholar] [CrossRef] [PubMed]
- Régnier-Rosencher, E.; Dupont, B.; Jacobelli, S.; Paugam, A.; Carlotti, A.; Boitier, F.; Gorin, I.; Dupin, N.; Avril, M.F. Late occurrence of Histoplasma duboisii cutaneous and pulmonary infection 18 years after exposure. J. Mycol. Med. 2014, 24, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Laurent, C.; Francois, A.; Le Roy, F.; Godin, M.; Guerrot, D. Souvenir de voyage. Nephrol. Ther. 2015, 11, 250–251. [Google Scholar] [CrossRef]
- Dussouil, A.S.; Allardet-Servent, J.; Dunogeant, L.; Grauer, J.L.; Ranque, S.; Nasser, V. Disseminated histoplasmosis partially mimicking a dermatomyositis in a patient with rheumatoid arthritis. Br. J. Dermatol. 2015, 173, 797–800. [Google Scholar] [CrossRef]
- Van Doom-Schepens, M.L.M.; Peters, E.J.; van Vugt, R.M.; van der Spoel, J.I.; van Dijk, K. Progressive disseminated histoplasmosis mimicking a flare of systemic lupus erythematosus: A European case report. JMM Case Rep. 2016, 3, e005035. [Google Scholar] [CrossRef]
- Van de Vosse, E.; van Wengen, A.; van der Meide, W.F.; Visser, L.G.; van Dissel, J.T. A 38-year-old woman with necrotising cervical lymphadenitis due to Histoplasma capsulatum. Infection 2017, 45, 917–920. [Google Scholar] [CrossRef]
- Lingscheid, T.; von Heinz, M.; Klages, B.; Rickerts, V.; Tintelnot, K.; Oestmann, J.-W.; Becker, M.; Temmesfeld-Wollbruck, B.; Suttorp, N.; Hubner, R.-H. A diagnostic predicament: Activated sarcoidosis or pulmonary histoplasmosis. A case report. Clin. Respir. J. 2017, 11, 374–377. [Google Scholar] [CrossRef]
- Schulze, A.B.; Heptner, B.; Kessler, T.; Baumgarten, B.; Stoica, V.; Mohr, M.; Wiewrodt, R.; Warneke, V.S.; Hartmann, W.; Wullenweber, J.; et al. Progressive histoplasmosis with hemophagocytic lymphohistiocytosis and epitheliod cell granulomatosis: A case report and review of the literature. Eur. J. Haematol. 2017, 99, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, N.; Jones, D.E.J.; Dyson, J.K.; Hoare, T.; Melmore, S.A.; Needham, S.; Thompson, N.P. Fatal gastrointestinal histoplasmosis 15 years after orthotopic liver transplantation. World J. Gastroenterol. 2017, 23, 7807–7812. [Google Scholar] [CrossRef]
- Gaume, M.; Marie-Hardy, L.; Larousserie, F.; Lavielle, M.; Roux, C.; Leclerc, P.; Paugam, A.; Archambeaus, D.; Eyrolle, L.; Gauzit, R.; et al. Histoplasmose ostéo-articulaire à Histoplasma capsulatum. Med. Malad. Infect. 2017, 47, 554–557. [Google Scholar] [CrossRef] [PubMed]
- Athanase, N.; Imbert, Y.; Ratrimoharilala, F. An unusual cause of perianal ulceration in an HIV-negative patient. Gastroenterology 2018, 155, 1701–1702. [Google Scholar] [CrossRef]
- Lucey, O.; Carroll, I.; Bjorn, T.; Millar, M. Reactivation of latent Histoplasma and disseminated cytomegalovirus in a returning traveller with ulcerative colitis. JMM Case Rep. 2018, 5, 1–3. [Google Scholar] [CrossRef]
- Oliveras, B.; Albert, M.; Lopez, C.; Fort, E.; Peries, L.; Gutierrez, L.; Busquets, D.; Uchima, H.; Aldeguer, X.; Pinol, V. A case report of gastrointestinal histoplasmosis in a patient treated with infliximab. Clin. J. Gastroenterol. 2021, 14, 690–692. [Google Scholar] [CrossRef]
- Carmans, L.; Van Craenenbroeck, A.; Lagrou, K.; Deroose, C.M.; Sagaert, X.; Wolthuis, A.; Van Wijngaerden, E.; Kuypers, D.R. Disseminated histoplasmosis in a kidney liver transplant patient from a non-endemic area: A diagnostic challenge. IDCases 2020, 22, e00971. [Google Scholar] [CrossRef]
- Margery, J.; Bonnichon, A.; Jeanbourquin, D.; Saint-Blancard, P.; Sane, M.; Dot, J.-M.; Grassin, F.; Vaylet, P.; L’Her, P.; Guigay, J. Présentation pseudo-tumorale de l’histoplasmose pulmonaire américaine. Rev. Pneumol. Clin. 2005, 67, 365–368. [Google Scholar] [CrossRef]
- De Vries, P.J.; Koolen, M.G.J.; Mulder, M.M.S.; Kortbeek, L.M. Acute pulmonary histoplasmosis from Ghana. Travel Med. Infect. Dis. 2006, 4, 286–289. [Google Scholar] [CrossRef]
- Galetta, D.; Pelosi, G.; Nebuloni, M.; Spaggiari, L. Challenging diagnosis of an unusual solitary pulmonary nodule. Thorac. Cardiov. Surg. 2007, 55, 120–129. [Google Scholar] [CrossRef]
- Alonso, D.; Munoz, J.; Letang, E.; Salvado, E.; Cuenca-Estrella, M.; Buitrago, M.J.; Torres, J.M.; Gascon, J. Imported acute histoplasmosis with rheumatologic manifestations in Spanish travelers. J. Travel Med. 2007, 14, 338–342. [Google Scholar] [CrossRef][Green Version]
- Hoenigl, M.; Schwetz, I.; Wurm, R.; Scheidl, S.; Olschewski, H.; Krause, R. Pulmonary histoplasmosis in three Austrian travelers after a journey to Mexico. Infection 2008, 36, 282–284. [Google Scholar] [CrossRef]
- Julg, B.; Elias, J.; Zahn, A.; Koppen, S.; Becker-Gaab, C.; Bogner, J.R. Bat-associated histoplasmosis can be transmitted at entrances of bat caves and not only inside the caves. J. Travel Med. 2008, 15, 133–136. [Google Scholar] [CrossRef]
- Garcia-Marron, M.; Garcia-Garcia, J.M.; Pajin-Collada, M.; Alvarez-Navascuée, F.; Martinez-Muniz, M.A.; Sanchez-Antuna, A.A. Histoplasmosis pulmonar cronica en un paciente no immunodeprimido, residente 10 anos antes en una zona endémica. Arc. Bronconeumol. 2008, 44, 567–570. [Google Scholar] [CrossRef]
- Torres-Rodriguez, J.M.; Segura-Roca, G.; Coll, J. Histoplasmosis en un varon immunocompetente manifestada 45 anos después de la infeccion. Rev. Iberoam. Micol. 2009, 26, 244–246. [Google Scholar] [CrossRef]
- Wobser, R.; Wilpert, J.; Kayser, G.; Walz, G.; Stubanus, M. Disseminated histoplasmosis with involvement of mediastinum and skin in an immunocompetent patient. Dtsch. Med. Wochenschr. 2009, 114, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Tay, Y.F.; Kullmann, D.M.; Howard, R.S.; Scott, G.M.; Hirsch, N.P.; Revesz, T.; Leary, S.M. Central nervous system histoplasmosis in an immunocompetent patient. J. Neurol. 2010, 257, 1931–1932. [Google Scholar] [CrossRef]
- Fortuna, G.; Mignogna, M.D. Oral histoplasmosis of a healthy man in a non-endemic area. Infection 2011, 39, 497–498. [Google Scholar] [CrossRef]
- Buitrago, M.J.; Gonzalo-Jimenez, N.; Navarro, M.; Rodriguez-Tudela, J.L.; Cuenca-Estrella, M. A case of primary cutaneous histoplasmosis acquired in the laboratory. Mycoses 2011, 54, e859–e861. [Google Scholar] [CrossRef] [PubMed]
- Locatelli Sanchez, M.; Mornex, J.F.; Philit, F.; Chumbi Floresa, W. Nodules pulmonaires au retour d’un voyage au Costa Rica. Rev. Malad. Respir. 2011, 28, 938–939. [Google Scholar] [CrossRef] [PubMed]
- Senechal, A.; Ferry, T.; Boibieux, A.; Brion, J.-P.; Epaulard, O.; Chidiac, C.; Peyramond, D. Imported pulmonary histoplasmosis in three French cavers after a trip to Cuba. J. Travel Med. 2012, 19, 64–65. [Google Scholar] [CrossRef]
- Kajfasfz, P.; Basiak, W. Outbreak of pulmonary histoplasmosis involving a group of four Polish travellers returning from Ecuador. Int. Marit. Health 2012, 63, 59–62. [Google Scholar]
- Pongratz, P.; Laferl, H.; Strau, G.; Stanek, G.; Wenisch, C. Coughing and fever after surfing in Central America. Wien Klin. Wochenschr. 2012, 124, 35–38. [Google Scholar] [CrossRef]
- Tsiodras, S.; Drogari-Apiranthitou, M.; Pilichos, K.; Leventakos, K.; Kelesidis, T.; Buitrago, M.J.; Petrikkos, G.; Panayiotides, I. An unusual cutaneous tumor: African histoplasmosis following mudbaths: Case report and review. Am. J. Trop. Med. Hyg. 2012, 86, 261–263. [Google Scholar] [CrossRef]
- Ehrhardt, J.; Tintelnot, K.; Kremsner, P.; Frank, M. Progressive malaise and joint pain after travel to Costa Rica. Dtsch. Med. Wochenschr. 2012, 137, 2260. [Google Scholar] [PubMed]
- Goncalves, D.; Ferraz, C.; Vaz, L. Posaconazole as rescue therapy in African histoplasmosis. Brazil. J. Infect. Dis. 2013, 17, 102–105. [Google Scholar] [CrossRef]
- Ardizzoni, A.; Baschieri, M.C.; Manca, L.; Salvadori, C.; Marinacci, G.; Farina, C.; Viale, P.L.; Blasi, E. The mycoarray as an aid for the diagnosis of an imported case of histoplasmosis in an Italian traveler retrurning from Brazil. J. Travel Med. 2013, 20, 336–339. [Google Scholar] [CrossRef] [PubMed]
- Cottle, L.E.; Gkrania-Klotsas, E.; Williams, H.J.; Brindle, H.E.; Carmichel, A.J.; Fry, G.; Beeching, N.J. A multinational outbreak of histoplasmosis following a biology field trip in the Ugandan rainforest. J. Travel Med. 2013, 20, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Bitterman, R.; Oren, I.; Geffen, Y.; Sprecher, H.; Schwartz, E.; Neuberger, A. Prolonged fever and splinter hemorrhages in an immunocompetent traveller with disseminated histoplasmosis. J. Travel Med. 2013, 20, 57–59. [Google Scholar] [CrossRef]
- Grancini, A.; Colosimo, M.; Mapelli, C.; Cogliati, M.; Pallotti, F.; Grande, R.; Torresani, E.; Tortorano, A.M. A case of Histoplasma capsulatum endophthalmitis diagnosed in Italy. Travel Med. Infect. Dis. 2013, 11, 256–258. [Google Scholar] [CrossRef]
- Raselli, C.; Reinhart, W.H.; Fleisch, F. Histoplasmosis- an unusual African souvenir. Dtsch. Med. Wochenschr. 2013, 138, 313–316. [Google Scholar]
- Muller-Stover, I.; Tintelnot, K.; Richter, J.; Haussinger, D. 55-jahrige patientin mit husten, fieber, gelenkschwellungen und exanthema nach einem urlaub in Ecuador. Internist 2014, 55, 976–980. [Google Scholar] [CrossRef]
- Richaud, C.; Chandesris, M.-O.; Lanternier, F.; Benzaquen-Forner, H.; Garcia-Hermoso, D.; Picard, C.; Catherinot, E.; Bougnoux, M.-E.; Lortholary, O. Imported African histoplasmosis in an immunocompetent patient 40 years after staying in a disease endemic area. Am. J. Trop. Med. Hyg. 2014, 91, 1011–1014. [Google Scholar] [CrossRef]
- Kooblall, M.; Keane, B.; Murray, G.; Moloney, E. Histoplasmosis mimicking primary lung neoplasm. BMJ Case Rep. 2014, bcr2013203335. [Google Scholar] [CrossRef] [PubMed]
- Labhardt, N.; Rickerts, V.; Popescu, S.; Neumayr, A. TB or not TB- persistent cough, fever, and night sweats in a 46-year-old traveller returning from South America. Travel Med. Infect. Dis. 2015, 13, 346–348. [Google Scholar] [CrossRef]
- Doleschal, B.; Rodhammer, T.; Tsybrovskyy, O.; Aichberger, K.J.; Lang, F. Disseminated histoplasmosis: A challenging differential diagnostic consideration for suspected malignant lesions in the digestive tract. Case Rep. Gastroenterol. 2016, 10, 653–660. [Google Scholar] [CrossRef]
- Ariaans, M.; Valladares, M.J.; Keuter, M.; Verweij, P.; van der Ven, A.J.; de Mast, Q. Fever and arthralgia after ‘volcano boarding’ in Nicaragua. Travel Med. Infect. Dis. 2017, 16, 68–69. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, L.; Silva, C.; Marques, N.; Verissimo, C. Tonsillar ulceration as manifestation of disseminated African histoplasmosis in an immunocompetent Portoguese host. Med. Mycol. Case Rep. 2017, 17, 14–16. [Google Scholar] [CrossRef] [PubMed]
- Bonsignore, A.; Fraternali Orcioni, G.; Barranco, R.; De Stefano, F.; Ravetti, J.L.; Ventura, F. Fatal disseminated histoplasmosis presentino as FUO in an immunocompetent Italian host. Legal Med. 2017, 25, 66–70. [Google Scholar] [CrossRef]
- Botsa, E.; Thanou, I.; Kabanarou, S.; Thanos, L. Rare case of pulmonary histoplasmosis complicated with bronchocentric granulomatosis in a non-endemic area. Resp. Med. Case Rep. 2017, 22, 1–3. [Google Scholar] [CrossRef]
- Denis, J.-B.; Coiffard, B.; Coltey, B.; Villeret, J.; Cassir, N.; Parroux, R.; Papazian, L. Granulomatose disséminée nécrosante liée à Histoplasma capsulatum. Med. Malad. Infect. 2017, 47, 61–64. [Google Scholar] [CrossRef]
- Lindner, A.K.; Rickerts, V.; Kurth, F.; Wilmes, D.; Richter, J. Chronic oral ulceration and lip swelling after a long term stay in Guatemala: A diagnostic challenge. Travel Med. Infect. Dis. 2018, 23, 103–104. [Google Scholar] [CrossRef]
- Hevroni, A.; Springer, C.; Wasser, O.; Avital, A.; Koplewitz, B.Z. Recurrent pneumonia due to fibrosing mediastinitis in a teenage girl: A case report with long-term follow-up. Case Rep. Pediatrics. 2018, 3246929. [Google Scholar] [CrossRef]
- Elias, A.; Abu Saleh, K.; Faranesh, N.; Dalal, R.; Geffen, Y.; Fisher, Y.; Neuberger, A.; Zaaroura, S. Histoplasmosis: First autochthonous case from Israel. Am. J. Trop. Med. Hyg. 2018, 98, 278–280. [Google Scholar] [CrossRef]
- Bissinger, A.L.; Biegner, T.; Frank, M.; Kremsner, P.G. Progressive ulcer on the tongue due to local histoplasmosis. Infection 2018, 46, 903–904. [Google Scholar] [CrossRef] [PubMed]
- Montagnac, C.; Eldin, C.; Thouret, A.; Ranque, S.; Brouqui, P. Histoplasmosis in an immunocompetent man returning from Brazil: A diagnostic challenge helped by 18 FDG PET CT. Travel Med. Infect. Dis. 2019, 27, 136–138. [Google Scholar] [CrossRef]
- Specialski, K.; Kita, K.; Kuziemski, K.; Tokarska, B.; Gorska, L.; Szade, J.; Sieminska, A.; Chelminska, M.; Jassem, E. Histoplasmosis in an elderly Polish tourist- a case report. BMC Pulm. Med. 2019, 19, 150. [Google Scholar]
- Paccoud, O.; Nervo, M.; Alanio, A.; Garcia-Hermoso, D.; Bougnoux, M.-E.; Mateo, J.; Mandonnet, E.; Lortholary, O.; Lanternier, F.; Adle-Biassette, H. Cerebral histoplasmosis caused by Histoplasma capsulatum var. duboisii in a patient with no known immunodeficiency. J. Travel Med. 2021, 28, taaa167. [Google Scholar]
- Mulec, J.; Simcic, S.; Kotar, T.; Kofol, R.; Stopinsck, S. Survey of Histoplasma capsulatum in bat guano and status of histoplasmosis in Slovenia, central Europe. Int. J. Speleol. 2020, 49, 1–10. [Google Scholar] [CrossRef]
- Farinas, F.; Flores, L.; Rodriguez, P.; Sabalete, T.; Quevedo, M.A. Histoplasmosis diseminada en una gacela dorca (Gazella dorcas neglecta) mantenida en condiciones de cautividad en Espana. Rev. Iberoam. Micol. 2009, 26, 152–154. [Google Scholar] [CrossRef]
- Mavropoulou, A.; Granda, G.; Calvi, L.; Passeri, B.; Volta, A.; Kramer, L.H.; Quintavalla, C. Disseminated histoplasmosis in a cat in Europe. J. Small Anim. Pract. 2010, 51, 176–180. [Google Scholar] [CrossRef]
- Fischer, N.M.; Favrot, C.; Monod, M.; Grest, P.; Rech, K.; Wilhelm, S. A case in Europe of feline histoplasmosis apparently limited to the skin. Vet. Dermatol. 2013, 24, 635–638. [Google Scholar] [CrossRef]
- Klang, A.; Loncaric, I.; Spergser, J.; Eigelsreiter, S.; Weissenbock, H. Disseminated histoplasmosis in a domestic cat imported from the USA to Austria. Med. Mycol. Case Rep. 2013, 2, 108–112. [Google Scholar] [CrossRef]
- Jacobsen, B.; Baumgartner, W.; Bialek, R. Disseminated histoplasmosis in a European hedgehog (Erinaceus europaeus) in Northern Germany. Mycoses 2010, 54, 538–541. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, T.; Seeger, H.; Kasuga, T.; Eskens, U.; Sauerwald, C.; Kaim, U. Detection and characterization of Histoplasma capsulatum in a German badger (Meles meles) by ITS sequencing and multilocus sequencing analysis. Med. Mycol. 2013, 511, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Reginato, A.; Giannuzzi, P.; Ricciardi, M.; De Simone, A.; Sanguinetti, M.; Porcellato, I.; Mandara, M.T. Extradural spinal cord lesion in a dog: First case study of canine neurological histoplasmosis in Italy. Vet. Microbiol. 2014, 170, 451–455. [Google Scholar] [CrossRef]
- Akdesir, E.; Origgi, F.C.; Wimmershoff, J.; Frey, J.; Frey, C.F.; Ryser-Degiorgis, M.-P. Causes of mortality and morbidity in free-ranging mustelids in Switzerland: Necropsy data from over 50 years of general health surveillance. BMC Vet. Res. 2018, 14, 195. [Google Scholar] [CrossRef]
- Mantovani, A.; Mazzoni, A.; Ajello, L. Histoplasmosis in Italy. I: Isolation of Histoplasma capsulatum from dogs in the province of Bologna. Sabourdia 1968, 6, 163–164. [Google Scholar] [CrossRef]
- Sotgiu, G.; Mazzoni, A.; Mantovani, A.; Ajello, L.; Palmer, J. Histoplasma capsulatum occurrence in soil from the Emilia-Romagna region of Italy. Science 1965, 5, 147. [Google Scholar] [CrossRef]
- Antinori, S.; Galimberti, L.; Bonaccorso, C.; Vago, L.; Nebuloni, M.; Esposito, R. A case of fatal disseminated histoplasmosis of autochthonous origin in an Italian AIDS patient. Eur. J. Clin. Microbiol. Infect. Dis. 1997, 16, 545–546. [Google Scholar] [CrossRef]
- Farina, C.; Gnecchi, F.; Michetti, G.; Parma, A.; Cavanna, C.; Nasta, P. Imported and autochthnous histoplasmosis in Bergamo province, Northern Italy. Scand. J. Infect. Dis. 2000, 32, 271–274. [Google Scholar]
- Poveda, F.; Garcia-Alegria, J.; Nieves, M.A.; Villar, E.; Montiel, N.; Del Arco, A. Disseminated histoplasmosis treated with liposomal amphotericin B following azathioprine therapy in a patient from a nonendemic area. Eur. J. Clin. Microbiol. Infect. Dis. 1998, 17, 357–359. [Google Scholar] [CrossRef]
- Ajello, L.; Kuttin, E.S.; Beemer, A.M.; Kaplan, W.; Padhye, A. Occurrence of Histoplasma capsulatum Darling, 1906 in Israel, with a review of the current status of histoplasmosis in the Middle East. Am. J. Trop. Med. Hyg. 1977, 26, 140–147. [Google Scholar] [CrossRef]
- Amona, F.M.; Denning, D.W.; Moukassa, D.; Develoux, M.; Hennequin, C. Histoplasmosis in the Republic of Congo dominated by African histoplasmosis, Histoplasma capsulatum var. duboisii. PLoS Negl. Trop. Dis. 2021, 15, e0009318. [Google Scholar]
- De Abreu Almeida, M.; Almeida-Silva, F.; Guimaraes, A.J.; Almeida-Paes, R.; Zancopé-Oliveira, R.M. The occurrence of histoplasmosis in Brazil: A systematic review. Int. J. Infect. Dis. 2019, 86, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control. Cave-associated histoplasmosis-Costa Rica. Morb. Mortal. Wkly Rep. 1988, 37, 312–313. [Google Scholar]
- Franklin, A.D.; Larson, L.; Rauseo, A.M.; Rutjanawech, S.; Hendrix, M.J.; Powderly, W.G.; Spec, A. A comparison of presentations and outcomes of histoplasmosis across patients with varying immune status. Med. Mycol. 2021, 59, 624–633. [Google Scholar] [CrossRef]
- Couppié, P.; Herceg, K.; Bourne-Watrin, M.; Thomas, V.; Blanchet, D.; Alsibai, K.D.; Louvel, D.; Djossou, F.; Demar, M.; Blaizot, R.; et al. The broad clinical spectrum of disseminated histoplasmosis in HIV-infected patients: A 30 years’ experience in French Guiana. J. Fungi 2019, 5, 115. [Google Scholar] [CrossRef]
- Nacher, M.; Valdes, A.; Adenis, A.; Blaizot, R.; Abboud, P.; Demar, M.; Djossou, F.; Epelboin, L.; Misslin, C.; Ntab, B.; et al. Gastrointestinal disseminated histoplasmosis in HIV-infected patients: A descriptive and comparative study. PLoS Negl. Trop. Dis. 2021, 15, e0009050. [Google Scholar] [CrossRef]
- Morote, S.; Nacher, M.; Blaizot, R.; Ntab, B.; Blanchet, D.; Alsibai, K.D.; Demar, M.; Djossou, F.; Couppié, P.; Adenis, A. Comparison of disseminated histoplasmosis with and without cutaneo-mucous lesions in persons living with HIV in French Guiana. J. Fungi 2020, 6, 133. [Google Scholar] [CrossRef]
- Nguyen, D.; Nacher, M.; Epelboin, L.; Melzani, A.; Demar, M.; Blanchet, D.; Blaizot, R.; Alsibai, K.D.; Abboud, P.; Djossou, F.; et al. Hemophagocytic lymphohistiocytosis during HIV infection in Cayenne Hospital 2021–2015: First think histoplasmosis. Front. Cell Infect. Microbiol. 2020, 10, 574584. [Google Scholar] [CrossRef] [PubMed]
- Wheat, L.J.; Hackett, E.; Durkin, M.; Connolly, P.; Petraitiene, R.; Walsh, T.J.; Knox, K.; Hage, C. Histoplasmosis-associated cross-reactivity in the BioRad Platelia Aspergillus enzyme immunoassay. Clin. Vaccine Immunol. 2007, 14, 638–640. [Google Scholar] [CrossRef]
- Buitrago, M.J.; Martin-Gomez, M.T. Timely diagnosis of histoplasmosis in non-endemic countries: A laboratory challenge. Front. Microbiol. 2020, 11, 467. [Google Scholar] [CrossRef]
- Alanio, A.; Gits-Muselli, M.; Lanternier, F.; Sturny-Leclère, A.; Benazra, M.; Hamane, S.; Rodrigues, A.M.; Garcia-Hermoso, D.; Lortholary, O.; Dromer, F.; et al. Evaluation of a new Histoplasma spp. quantitative RT-PCR assay. J. Mol. Diagn. 2021, 23, 698–709. [Google Scholar] [CrossRef]
- Wheat, L.J.; Freifeld, A.G.; Kleiman, M.B.; Baddley, J.W.; McKinsey, D.S.; Loyd, J.E.; Kauffman, C.A. Clinical practice guidelines for the management of patients with histoplasmosis: 2007 update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2007, 45, 807–825. [Google Scholar] [CrossRef]
| Author, Year, Reference | Type of Study | Country | Period of Study | No. of Patients Diagnosed | Age Median Years (Range) | Male Gender | Risk Factors | Disease | Identified Histoplasma Species | Outcomes |
|---|---|---|---|---|---|---|---|---|---|---|
| Gascon, 2005, [13] | Prospective, single centre | Spain | Mar. 2001– Apr. 2003 | 69/342 (20.2%) 13/69 (19%) symptomatic | NR | NR | Travellers to Central/South America | Acute pulmonary disease | NR | NR |
| Molina-Morants, 2018, [12] | Retrospective, patients admitted to Spanish hospitals | Spain | Jan. 1997– Dec. 2014 | 286 (incidence: 0.53/100,000) | 37 (30–50) | 193 (67.5%) | 188 immunodeficiency (65.7%) :151 HIV (80.7%); 14 solid neoplasia (7.5%); 10 solid organ transplant (5.3%); 9 haematological malignancy (4.8%);5 systemic autoimmune disease (2.7%); 5 end-stage renal disease (2.7%); 3 cirrhosis (1.6%); 2 primary immune deficiency (1.1%) | 72 isolated pulmonary disease (25.2%); 36 extra- pulmonary disease (12.6%); 168 unspecified disease (58.7%) | 10 H. duboisii | 44 deaths (15.4%) |
| Peigne, 2011, [14] | Retrospective, NRCMA | France | 1985– 2006 | 104 | 38 * 40 ** | 31 (78%) * 37 (58%) ** | HIV/AIDS | 73 PDH (70%); blood (36.5%); bone marrow (61.5%); skin (38.5%); lymph nodes (24%); respiratory samples (30.8%); CNS (1.9%) | NR | 7 IRIS (11%)41 deaths (39.4%) (median follow-up: 31.5 months) |
| Segel, 2015, [16] | Retrospective, single centre | Israel | 2000– 2011 | 23 | 31 | 17 (73.9%) | Travellers to Central/South America (95.6%); 14 exposure to bat habitats (61%) | Symptomatic pulmonary disease 60.9% | 2 H. capsulatum | NR |
| Staffolani, 2020, [15] | Retrospective study, single centre | Italy | Jan. 2005– Dec. 2015 | 23 | 38.5 Ecuador cluster; 46.7 others | 17 (73.9%) | 17 Scientific expedition (Ecuador): bat excreta; 3 speleologist; 2 tourism, 1 work (Panama, Bolivia, Mexico, Cuba) | 2 PDH (1 immunocompetent, 1 immunocompromised) 21 pulmonary disease | 2 H. capsulatum | All alive |
| Characteristic | No. (%) |
|---|---|
| Median age, years (range) | 37 (2–63) |
| Males | 73 (64) |
| Previously known HIV infection | 37 (32.4) |
| Histoplasmosis indicating HIV infection | 40 (35.1) |
| Time of HIV diagnosis unknown | 37 (32.4) |
| Median latency, months (range) | 66 (1–420) |
| Median CD4+ cells/μL | 17 (0–594) |
| Area of suspected Histoplasma exposure | |
| Central/South America § | 55 (50) |
| Sub-Saharan Africa ^ | 47 (42.7) |
| Asia # | 6 (5.5) |
| United States/Mexico | 1 (0.9) |
| Europe | 1 (0.9) |
| Not reported | 4 (3.5) |
| Country of diagnosis | |
| Spain | 56 (49.1) |
| France | 22 (19.3) |
| Italy | 14 (12.3) |
| Switzerland, The Netherlands | 6 (5.2) |
| United Kingdom | 4 (3.5) |
| Portugal | 2 (1.7) |
| Belgium, Denmark, Finland, Germany | 1 (0.9) |
| Histoplasmaspecies | |
| H. capsulatum | 80 (70.2) |
| H. duboisii | 20 (17.5) |
| Histoplasma spp. | 14 (12.3) |
| Concomitant infections/neoplasia | 33 (28.9) |
| Fungal | |
| Oral candidiasis | 8 (24.4) |
| Oesophageal candidiasis | 2 (6.1) |
| PJP | 5 (15.1) |
| Cryptococcosis | 2 (6.1) |
| Coccidioidomycosis | 1 (3.0) |
| Bacterial | |
| Tuberculosis | 2 (6.1) |
| MAC | 3 (9.1) |
| Salmonella bacteremia | 1 (3.0) |
| Protozoal | |
| Chagas disease | 1 (3.0) |
| Isopsoriasis | 1 (3.0) |
| Helminthic | |
| Strongyloides stercoralis | 3 (9.1) |
| Neoplasia | |
| Kaposi’s sarcoma | 3 (9.1) |
| Multiple | 8 (7.0) |
| Methods of diagnosis | |
| Histology + culture | 32 (28.1) |
| Culture + PCR | 19 (16.7) |
| Histology | 16 (14.0) |
| Culture | 14 (12.3) |
| Histology + PCR | 13 (11.4) |
| Histology + culture + PCR | 7 (6.1) |
| Histology + culture + blood smear | 5 (4.4) |
| Histology + PCR | 2 (1.7) |
| PCR | 3 (2.6) |
| Histology + PCR + blood smear | 1 (0.9) |
| Autopsy | 2 (1.7) |
| Treatment | |
| L-AMB, itraconazole | 30 (38) |
| d-AMB, itraconazole | 12 (15.2) |
| Itraconazole | 14 (12.7) |
| L-AMB | 8 (10.1) |
| L-AMB, voriconazole or posaconazole | 3 (3.8) |
| ABLC, itraconazole, fluconazole | 3 (3.8) |
| Fluconazole | 3 (3.8) |
| ABLC | 2 (2.5) |
| d-AMB | 2 (2.5) |
| Not treated | 2 (2.5) |
| Not reported | 35 |
| Outcome | |
| Survived | 60 (75.9) |
| Died | 19 (24.1) |
| Not reported | 35 |
| People Living with HIV | Other Immunocompromising Conditions | ||
|---|---|---|---|
| Signs and Symptoms, No. (%) | Organs in which Histoplasma was Demonstrated, No. | Signs and Symptoms, No. (%) | Organs in which Histoplasma was Demonstrated, No. |
| Fever, 57/78 (73.1) Splenomegaly, 22/32 (65.6) Hepatomegaly, 21/32 (65.6) Lymph node enlargement, 40/78 (51.3) Weight loss, 38/78 (48.7) Skin lesions, 25/78 (32.0) Cough, 20/78 (25.6) Dyspnoea, 17/78 (21.8) Diarrhoea, 14/78 (17.9) Abdominal pain, 10/78 (12.8) Nausea, 5/78 (6.4) | Bone marrow, 38 Blood, 29 Lymph nodes, 29 Lung, 24 Skin, 20 Intestine, 12 Liver, 8 Peripheral blood smear, 5 Oropharyngeal mucosa, 5 Cerebrospinal fluid, 2 Larynges, 2 Tonsils, 2 Pleural fluid, 1 Vagina, 1 Brain biopsy, 1 Oesophagus, 1 | Fever, 17/26 (65.4) Weight loss, 11/26 (42.3) Cough, 9/26 (34.6) Dyspnoea, 7/26 (26.9) Diarrhoea, 6/26 (23.1) Skin lesions, 5/26 (19.2) Hepatomegaly, 4/26 (15.4)Lymph node enlargement, 4/26 (15.4) Abdominal pain, 3/26 (11.5) Splenomegaly, 1/26 (3.8) Nausea, 0/26 (0) | Lung, 8 Bone marrow, 6 Oropharyngeal mucosa, 6 Lymph nodes, 6 Skin, 5 Intestine, 4 Liver, 3 Kidney, 2 Brain biopsy, 1 Muscle, 1 Spleen, 1 Synovial membrane, 1 Blood, 1 Mitral valve, 1 |
| Characteristics | No. (%) |
|---|---|
| Median age, years (range) | 59 (6–86) |
| Males | 16 (57.1) |
| Latency interval | 3 months–42 years |
| Geographical area of suspected Histoplasma exposure | |
| Central/South America * | 14 (50.0) |
| Sub-Saharan Africa § | 5 (12.9) |
| Asia ^ | 2 (7.1) |
| Sub-Saharan Africa/Central America ° | 1 (3.6) |
| South America/Asia ** | 1 (3.6) |
| Asia/Central America # | 1 (3.6) |
| Asia (Malaysia)/United States | 1 (3.6) |
| South America (Suriname)/United States | 1 (3.6) |
| Europe (Italy, Spain) | 2 (7.1) |
| Country of diagnosis | |
| France | 8 (28.6) |
| Spain | 6 (21.4) |
| Netherlands | 5 (17.9) |
| Germany | 3 (10.7) |
| United Kingdom | 2 (7.1) |
| Belgium, Italy, Portugal, Sweden | 1 (3.6) |
| Underlying diseases/immunosuppressive treatment | |
| SOT (kidney 2, kidney/liver 1, liver 2, lung 1, NR 1) | 7 (25.0) |
| Autoimmune diseases (4 RA, 1 SLE, 1 dermatomyositis, | 7 (25.0) |
| 1 myasthenia gravis) | |
| Primary immunodeficiency (IgA deficit 1, idiopathic CD4 lymphopenia 2, autoantibodies against IFN-γ 1) | 4 (14.3) |
| Sarcoidosis | |
| Chronic lymphocytic leukaemia | 2 (7.1) |
| Cancer | 2 (7.1) |
| Ulcerative colitis | 2 (7.1) |
| Haemodialysis | 1 (3.6) |
| Steroids | 1 (3.6) |
| 2 (7.1) | |
| Methods of diagnosis | |
| Histology + culture | 8 (28.6) |
| Histology + PCR | 5 (17.8) |
| Histology + culture + PCR | 5 (17.8) |
| Histology | 4 (14.3) |
| Culture | 3 (10.7) |
| Culture + PCR | 1 (3.6) |
| Autopsy | 2 (7.1) |
| Histoplasmaspecies | |
| H. capsulatum | 25 (89.3) |
| H. duboisii | 2 (7.1) |
| Histoplasma spp. | 1 (3.6) |
| Treatment | |
| d-AMB, itraconazole | 5 (19.2) |
| L-AMB, itraconazole | 5 (19.2) |
| Itraconazole | 5 (19.2) |
| L-AMB | 3 (11.5) |
| ABLC, itraconazole | 1 (3.8) |
| L-AMB, posaconazole | 1 (3.8) |
| Voriconazole, itraconazole | 1 (3.8) |
| Fluconazole, micafungin, L-AMB | 1 (3.8) |
| Fluconazole | 1 (3.8) |
| Not treated | 1 |
| Not reported | 1 |
| Outcome | |
| Survived | 17 (68.0) |
| Died | 8 (32.0) |
| Not reported | 3 |
| Blood Test | PLWH | OIC |
|---|---|---|
| Haemoglobin, No. | 38 | 12 |
| Anaemia (<12 g/dL), No. (%) | 36 (94.7) | 12 (100) |
| Median Haemoglobin value, g/dL (range) | 7.8 (3.9–12.3) | 9.8 (5.3–11.6) |
| White blood cells, No. | 37 | 14 |
| Leukopenia (<4000/μL), No. (%) | 26 (70.3) | 6 (42.8) |
| Median white blood cells value, cells/L (range) | 3800 (960–13,600) | 5545 (1400–16,760) |
| Platelets, No. | 27 | 8 |
| Thrombocytopenia (<150,000/μL) | 19 (70.4) | 3 (37.5) |
| Median platelets value/μL | 80,000 (20,000–272,000) | 170,500 (17,000–579,000) |
| AST, No. | 16 | 7 |
| Median AST value, UL (range) | 97.5 (50–610) | 108 (9–1046) |
| ALT, No. | 14 | 9 |
| Median ALT value, UL (range) | 63 (27–301) | 58 (8–487) |
| Characteristics | No. (%) or Median (Range) |
|---|---|
| Age, years | 43 (17–78) |
| Males | 56 (69.1) |
| Latency | |
| Travellers | 14 days (2–120) |
| Expatriates, migrants | 6.5 years (3–46) |
| Geographical areas of suspected Histoplasma exposure | |
| Central/South America ^ | 50 (61.7) |
| Sub-Saharan Africa § | 20 (24.7) |
| Sub-Saharan Africa and Central/South America # | 3 (3.7) |
| United States * | 2 (2.5) |
| India | 1 (1.2) |
| Europe (2 Italy, 2 Spain) | 4 (4.9) |
| Israel | 1 (1.2) |
| Country of diagnosis | |
| Spain | 19 (23.5) |
| France | 12 (14.8) |
| Germany | 8 (9.9) |
| Italy, Poland | 6 (7.4) |
| Austria, Slovenia | 5 (6.2) |
| Netherlands | 4 (4.9) |
| Israel, United Kingdom, Switzerland | 3 (3.7) |
| Greece, Ireland, Portugal | 2 (2.5) |
| Sweden | 1 (1.2) |
| Patient categories | |
| Travellers | 40 (49.4) |
| Expatriates | 5 (12.3) |
| Workers | 26 (32.1) |
| Migrants | 2 (2.5) |
| Military service people | 2 (2.5) |
| Autochthonous | 5 (7.4) |
| Method of diagnosis | |
| Serology | 36 (44.4) |
| Histology | 10 (12.3) |
| Histology + PCR | 9 (11.1) |
| Histoplasma antigen | 4 (4.9) |
| Histology + culture + PCR | 3 (3.7) |
| Culture | 3 (3.7) |
| Culture + PCR | 3 (3.7) |
| Histology + culture | 2 (2.5) |
| PCR | 2 (2.5) |
| Serology + PCR | 2 (2.5) |
| Clinical | 6 (7.4) |
| Autopsy | 1 (1.2) |
| Histoplasmaspecies | |
| H. capsulatum | 29 (78.4) |
| H. duboisii | 6 (16.2) |
| Histoplasma spp. | 2 (2.5) |
| Treatment | |
| Itraconazole | 45 (60.8) |
| L-AMB | 6 (8.1) |
| Ketoconazole, itraconazole | 2 (2.7) |
| d-AMB, itraconazole | 2 (2.7) |
| No treatment | 19 (25.7) |
| Not reported | 7 |
| Outcome | |
| Survived | 68 (94.4) |
| Died | 4 (5.6) |
| Not reported | 9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antinori, S.; Giacomelli, A.; Corbellino, M.; Torre, A.; Schiuma, M.; Casalini, G.; Parravicini, C.; Milazzo, L.; Gervasoni, C.; Ridolfo, A.L. Histoplasmosis Diagnosed in Europe and Israel: A Case Report and Systematic Review of the Literature from 2005 to 2020. J. Fungi 2021, 7, 481. https://doi.org/10.3390/jof7060481
Antinori S, Giacomelli A, Corbellino M, Torre A, Schiuma M, Casalini G, Parravicini C, Milazzo L, Gervasoni C, Ridolfo AL. Histoplasmosis Diagnosed in Europe and Israel: A Case Report and Systematic Review of the Literature from 2005 to 2020. Journal of Fungi. 2021; 7(6):481. https://doi.org/10.3390/jof7060481
Chicago/Turabian StyleAntinori, Spinello, Andrea Giacomelli, Mario Corbellino, Alessandro Torre, Marco Schiuma, Giacomo Casalini, Carlo Parravicini, Laura Milazzo, Cristina Gervasoni, and Anna Lisa Ridolfo. 2021. "Histoplasmosis Diagnosed in Europe and Israel: A Case Report and Systematic Review of the Literature from 2005 to 2020" Journal of Fungi 7, no. 6: 481. https://doi.org/10.3390/jof7060481
APA StyleAntinori, S., Giacomelli, A., Corbellino, M., Torre, A., Schiuma, M., Casalini, G., Parravicini, C., Milazzo, L., Gervasoni, C., & Ridolfo, A. L. (2021). Histoplasmosis Diagnosed in Europe and Israel: A Case Report and Systematic Review of the Literature from 2005 to 2020. Journal of Fungi, 7(6), 481. https://doi.org/10.3390/jof7060481







