Bioactive Compounds from Mangrove Endophytic Fungus and Their Uses for Microorganism Control
Abstract
1. Introduction
2. Endophytic Fungi
| Endophytic Fungi | Mangrove Plant | Reference |
|---|---|---|
| Acremonium sp. and Acremonium strictum | Rhizophora apiculata | [32,33] |
| Alternaria longipe | Avicennia officinalis | [34] |
| Alternaria sp. | Myoporum bontioides, Rhizophora mucronata | [35,36] |
| Ascomycota sp. | Pluchea indica | [37] |
| Aspergillus clavatus | Myoporum bontioides | [38] |
| Aspergillus flavipes | Acanthus ilicifolius | [39] |
| Aspergillus flavus | Hibiscus tiliaceus, Sonneratia griffithii, Kandelia obovata | [40,41,42] |
| Aspergillus fumigatus | Acrostichum specioum, Sonneratia griffithii | [41,43] |
| Aspergillus nidulans | Rhizophora stylosa | [44,45] |
| Aspergillus niger | Sonneratia apetala,S. griffithii | [41,46,47,48] |
| Aspergillus sp. | Bruguiera gymnorrhiza, Avicennia africana, Xylocarpus moluccensis, Acanthus ilicifolius, Avicennia marina, Dalbergia ecastaphyllum | [31,49,50,51,52,53] |
| Aspergillus tubingensis | Pongamia pinnata | [54] |
| Aspergillus versicolor | Excoecaria agallocha | [55] |
| Campylocarpon sp. | Sonneratia caseolaris | [56] |
| Cladosporium sp. | Rhizophora apiculata, Aegiceras corniculatum, Kandelia candel, Rhizophora mucronata, Excoecaria agallocha | [36,52,57,58,59] |
| Colletotrichum gloeosporioides | Avicennia schaueriana and Laguncularia racemosa, Ceriops tagal and Sonneratia apetala | [29,47,60] |
| Colletotrichum sp. | Xylocarpus granatum, Avicennia schaueriana Laguncularia racemosa and Rhizophora mangle Aegiceras corniculatum, Avicennia africana, | [29,50,57] |
| Cytospora sp. | Ceriops tagal | [61] |
| Daldinia eschscholtzii | Bruguiera sexangula var. rhynchopetala | [62] |
| Diaporthe sp. | Avicennia schaueriana, Laguncularia racemosa, and Rhizophora mangle, Rhizophora stylosa | [30,63] |
| Dothiorella sp. | Aegiceras corniculatum | [64] |
| Emericella sp. | Aegiceras corniculatum | [65] |
| Epicoccum sp. | Avicennia africana | [50] |
| Eupenicillium sp. | Xylocarpus granatum | [66] |
| Eurotium chevalier | Rhizophora mucronata | [67] |
| Eurotium rubrum | Hibiscus tiliaceu | [68] |
| Fusarium equiseti | Sonneratia apetala | [47] |
| Fusarium lateritium | Rhizophora mucronata | [36] |
| Fusarium napiforme | Rhizophora mucronata | [69] |
| Fusarium phyllophilum | Avicennia africana | [50] |
| Fusarium sp. | Avicennia schaueriana, Laguncularia racemosa, Rhizophora mangle, Rhizophora mucronata, and Dalbergia ecastaphyllum | [30,36,69] |
| Glomerella cingulata and Guignardia sp. | Avicennia schaueriana, Laguncularia racemosa, Rhizophora mangle | [29] |
| Guignardia camelliae | Avicennia sp. | [70] |
| Guignardia sp. | Scyphiphora hydrophyllacea, Aegiceras corniculatum, Acanthus ilicifolius; | [59,71,72] |
| Glomerella sp. | Aegiceras corniculatum | [59] |
| Hypocrea virens | Premna serratifolia | [73] |
| Lasiodiplodia theobromae | Acanthus ilicifolius, Avicennia lanata | [74,75] |
| Leptosphaerulina sp. | Acanthus ilicifolius | [76] |
| Neosartorya hiratsukae | Avicennia sp. and Aricennia marina | [76,77] |
| Nodulisporium gregarium | Avicennia schaueriana | [29] |
| Nodulisporium sp. | Acanthus ilicifolius | [72] |
| Nigrospora sp. | Kandelia candel, Pongamia pinnata, and Rhizophora mucronata | [36,78,79] |
| Nigrospora sphaerica | Bruguiera gymnorrhyza | [79] |
| Phellinus noxius | Acanthus ilicifolius | [72] |
| Penicillium brocae | Avicennia marina | [80,81] |
| Penicillium chrysogenum | Porteresia coarctata, Myoporum bontioides | [82,83] |
| Penicillium citrinum | Bruguiera sexangula var. rhynchopetala | [84,85] |
| Penicillium coffeae and Penicillium herquei | Laguncularia racemosa | [86,87] |
| Penicillium simplicissimum | Bruguiera sexangula var. rhynchopetala | [88] |
| Penicillium spp. | Bruguiera sexangula var. Rhynchopetala, Bruguiera gymnorrhiza, K. candel, Avicennia africana, Dalbergia ecastaphyllum | [31,50,89,90,91] |
| Pestalotiopsis sp. | Aegiceras corniculatum, Rhizophora mucronata, Rhizophora stylosa | [36,92,93,94] |
| Pestalotiopsis vacinii | Kandelia candel | [7] |
| Phoma sp. | Thespesia populneoide, Myoporum bontioides, Rhizophora mucronata, Kandelia sp., Acanthus ilicifolius | [36,38,72,95] |
| Phomopsis archeri, P. diachenii | Avicennia schaueriana and Laguncularia racemosa | [29] |
| Phomopsis longicolla | Brguiera sexangula var. rhynchopetala | [38] |
| Phomopsis sp. | Rhizophora apiculata, Kandelia candel, Acanthus ilicifolius, Xylocarpus granatum, Avicennia africana | [50,96,97,98] |
| Phyllosticta capitalensis | Bruguiera sexangula | [94] |
| Phyllosticta sp. | Acanthus ilicifolius, Avicennia alba, Ceriops decandra, Lumnitzera littorea, Rhizophora apiculata, Rhizophora mucronata, Sonneratia alba, Xylocarpus moluccensis, Rhizophora mangle | [28,57] |
| Pleosporales sp. | Kandelia candel | [99] |
| Rhizopus sp. | Avicennia africana | [50] |
| Schizophyllum commune | Avicennia officinalis | [100] |
| Stemphylium sp. | Bruguiera sexangula var. rhynchopetala | [101,102] |
| Talaromyces sp. | Kandelia candel | [103] |
| Talaromyces stipitatus | Acanthus ilicifolius | [98] |
| Trichoderma sp. | Avicennia schaueriana Laguncularia racemosa, Rhizophora mangle, Clerodendrum inerme, Ceriops tagal, Bruguiera sp., Dalbergia ecastaphyllum | [30,70,103,104] |
| Xylaria psidii | Aegle marmelos | [104] |
| Xylaria sp. | Avicennia schaueriana, Laguncularia racemosa, Rhizophora mangle, Rhizophora mucronata Xylocarpus granatum, Acanthus ilicifolius | [30,36,51,72] |
| Zasmidium sp. | L. racemosa | [105,106] |
3. Bioactive Compounds from Mangrove Endophytic Fungus
3.1. Antiprotozoan
3.2. Antibacterial
3.3. Antiviral
4. Future Challenges
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Elshafie, H.S.; Caputo, L.; De Martino, L.; Grul’ová, D.; Zheljazkov, V.V.; De Feo, V.; Camele, I. Biological investigations of essential oils extracted from three Juniperus species and evaluation of their antimicrobial, antioxidant and cytotoxic activities. J. Appl. Micro 2020, 129, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- Camele, I.; Elshafie, H.S.; Caputo, L.; Sakr, S.H.; de Feo, V. Bacillus mojavensis: Biofilm formation and biochemical investigation of its bioactive metabolites. J. Bio. Res. 2019, 92, 39–45. [Google Scholar] [CrossRef]
- Cragg, G.M.; Newman, D.J. Biodiversity: A continuing source of novel drug leads*. Pure Appl. Chem. 2005, 77, 26–31. [Google Scholar] [CrossRef]
- Strohl, W.R. The role of natural products in a modern drug discovery program. Drug Discov. Today 2000. [Google Scholar] [CrossRef]
- Li, J.W.H.; Vederas, J.C. Drug discovery and natural products: End of an era or an endless frontier? Science 2009. [Google Scholar] [CrossRef]
- Dias, D.A.; Urban, S.; Roessner, U. A Historical Overview of Natural Products in Drug Discovery. Metabolites 2012, 2, 303–336. [Google Scholar] [CrossRef]
- Wang, K.-W.; Wang, S.-W.; Wu, B.; Wei, J.-G. Bioactive Natural Compounds from the Mangrove Endophytic Fungi. Mini Rev. Med. Chem. 2014, 14, 370–391. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Chaturvedi, P.; Kulkarni, M.G.; Van Staden, J. A critical review on exploiting the pharmaceutical potential of plant endophytic fungi. Biotechnol. Adv. 2020, 39, 107462. [Google Scholar] [CrossRef] [PubMed]
- Jakubczyk, D.; Dussart, F. Selected Fungal Natural Products with Antimicrobial Properties. Molecules 2020, 25, 911. [Google Scholar] [CrossRef] [PubMed]
- Elshafie, H.S.; Camele, I. An Overview of Metabolic Activity, Beneficial and Pathogenic Aspects of Burkholderia spp. Metabolites 2021, 11, 321. [Google Scholar] [CrossRef]
- Fleming, A. On the antibacterial action of cultures of a penicillium, with special reference to their use in the isolation of b. influenz? Br. J. Exp. Pathol. 1929, 10, 226–236. [Google Scholar] [CrossRef]
- Amit Koparde, A.; Chandrashekar Doijad, R.; Shripal Magdum, C. Natural Products in Drug Discovery. In Pharmacognosy-Medicinal Plants; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Ancheeva, E.; El-Neketi, M.; Daletos, G.; Ebrahim, W.; Song, W.; Lin, W.; Proksch, P. Anti-infective Compounds from Marine Organisms. Grand Chall. Mar. Biotechnol. 2018, 97–155. [Google Scholar] [CrossRef]
- Rafferty, J.P. Lakes and Wetlands South America. 2011. Available online: https://books.google.com.br/books?id=StCGBdKR4qgC&printsec=frontcover&hl=pt-BR&source=gbs_ge_summary_r&cad=0#v=onepage&q&f=false (accessed on 14 September 2020).
- Francisco, P.M.; Tambarussi, E.V.; de Alves, F.M.; Bajay, S.; Ciampi-Guillardi, M.; Souza, A.P. Genetic diversity and mating system of rhizophora mangle l. (rhizophoraceae) in Northern Brazil revealed by microsatellite analysis. Cerne 2018, 24, 295–302. [Google Scholar] [CrossRef]
- Silva, J.M.; Martins, M.B.G.; Cavalheiro, A.J. Caracterização anatômica e perfis químicos de folhas de Avicennia schaueriana Stapf. & Leech. ex Moldenke e Rhizophora mangle L. de manguezais impactados e não impactados do litoral paulista. Insul. Rev. Botânica 2010, 39, 14–33. [Google Scholar] [CrossRef][Green Version]
- Glauce Brasil, A. A efetividade da gestão das unidades de conservação na proteção dos sistemas de manguezais na ilha de santa Catarina. Doctoral Thesis, Universidade Federal de Santa Catarina, Florianópolis, Brazil, 2017. [Google Scholar]
- Patra, J.K.; Thatoi, H.N. Metabolic diversity and bioactivity screening of mangrove plants: A review. Acta Physiol. Plant. 2011. [Google Scholar] [CrossRef]
- Rajpar, M.N.; Zakaria, M. Mangrove fauna of Asia. In Mangrove Ecosystems of Asia; Springer: New York, NY, USA, 2014; pp. 153–197. [Google Scholar] [CrossRef]
- Cribb, J.W. WoRMS-World Register of Marine Species-Lulworthia cylindrica (Linder) Cribb & J.W. Cribb, 1955. Available online: http://www.marinespecies.org/aphia.php?p=taxdetails&id=438136 (accessed on 22 December 2020).
- Zhou, J.; Diao, X.; Wang, T.; Chen, G.; Lin, Q.; Yang, X.; Xu, J. Phylogenetic diversity and antioxidant activities of culturable fungal endophytes associated with the mangrove species Rhizophora stylosa and R. mucronata in the South China Sea. PLoS ONE 2018, 13, e0197359. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Guo, L.D. Endophytic fungal diversity: Review of traditional and molecular techniques. Mycology 2012, 3, 65–76. [Google Scholar] [CrossRef]
- Rajesh, R.W.; Rahul, M.S.; Ambalal, N.S. Trichoderma: A significant fungus for agriculture and environment. Afr. J. Agric. Res. 2016, 11, 1952–1965. [Google Scholar] [CrossRef]
- Adnan, M.; Islam, W.; Shabbir, A.; Khan, K.A.; Ghramh, H.A.; Huang, Z.; Chen, H.Y.H.; Lu, G.-G. Plant defense against fungal pathogens by antagonistic fungi with Trichoderma in focus. Microb. Pathog. 2019. [Google Scholar] [CrossRef]
- Luo, J.M.; Xiao, X.; Luo, S.L. Biosorption of cadmium(II) from aqueous solutions by industrial fungus Rhizopus cohnii. Trans. Nonferrous Met. Soc. China 2010, 20, 1104–1111. [Google Scholar] [CrossRef]
- Petruzzello, M. Rhizopus|Fungus Genus|Britannica. Encycl. Br. 2016. Available online: https://www.britannica.com/science/Rhizopus (accessed on 22 December 2020).
- Biasetto, C.R.; Somensi, A.; Figueiro, F.S.; De Moraes, L.A.B.; Silva, G.H.; Marx Young, M.C.; Da Silva Bolzani, V.; Araújo, A.R. Diketopiperazines and arylethylamides produced by Schizophyllum commune, an endophytic fungus in Alchornea glandulosa. Eclet. Quim. 2019, 44, 36–42. [Google Scholar] [CrossRef]
- Costa, I.P.M.W.; Maia, L.C.; Cavalcanti, M.A. Diversity of leaf endophytic fungi in mangrove plants of Northeast Brazil. Braz. J. Microbiol. 2012, 43, 1165–1173. [Google Scholar] [CrossRef]
- De Souza Sebastianes, F.L.; Romão-Dumaresq, A.S.; Lacava, P.T.; Harakava, R.; Azevedo, J.L.; De Melo, I.S.; Pizzirani-Kleiner, A.A. Species diversity of culturable endophytic fungi from Brazilian mangrove forests. Curr. Genet. 2013, 59, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Taub, L. Universidade Federal de Alfenas Rayra Annara da Fonseca Otacílio Pinto Bioprospecção e Caracterização de Fungos Endofíticos Produtores de Compostos Bioativos Isolados de Dalbergia; Universidade Federal de Alfenas: Alfenas, Brazil, 2019. [Google Scholar]
- Rajamanikyam, M.; Vadlapudi, V.; Amanchy, R.; Upadhyayula, S.M. Endophytic fungi as novel resources of natural therapeutics. Braz. Arch. Biol. Technol. 2017, 60, 17160542. [Google Scholar] [CrossRef]
- Hammerschmidt, L.; Debbab, A.; Ngoc, T.D.; Wray, V.; Hemphil, C.P.; Lin, W.; Broetz-Oesterhelt, H.; Kassack, M.U.; Proksch, P.; Aly, A.H. Polyketides from the mangrove-derived endophytic fungus Acremonium strictum. Tetrahedron Lett. 2014, 55, 3463–3468. [Google Scholar] [CrossRef]
- Rukachaisirikul, V.; Rodglin, A.; Sukpondma, Y.; Phongpaichit, S.; Buatong, J.; Sakayaroj, J. Phthalide and Isocoumarin Derivatives Produced by an Acremonium sp. Isolated from a Mangrove Rhizophora apiculata. J. Nat. Prod. 2012. [Google Scholar] [CrossRef]
- Ranganathan, N.; Mahalingam, G. Secondary metabolite as therapeutic agent from endophytic fungi Alternaria longipes strain VITN14G of mangrove plant Avicennia officinalis. J. Cell. Biochem. 2019, 120, 4021–4031. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ding, W.; Wang, R.; Du, Y.; Liu, H.; Kong, X.; Li, C. Identification and bioactivity of compounds from the mangrove endophytic fungus Alternaria sp. Mar. Drugs 2015, 13, 4492–4504. [Google Scholar] [CrossRef]
- Hamzah, T.N.T.; Lee, S.Y.; Hidayat, A.; Terhem, R.; Faridah-Hanum, I.; Mohamed, R. Diversity and characterization of endophytic fungi isolated from the tropical mangrove species, Rhizophora mucronata, and identification of potential antagonists against the soil-borne fungus, Fusarium solani. Front. Microbiol. 2018, 9, 1707. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, Z.; Liu, H.; Pan, Y.; Li, J.; Liu, L.; She, Z. Dichloroisocoumarins with Potential Anti-Inflammatory Activity from the Mangrove Endophytic Fungus Ascomycota sp. CYSK-4. Mar. Drugs 2018, 16, 54. [Google Scholar] [CrossRef]
- Li, W.; Xiong, P.; Zheng, W.; Zhu, X.; She, Z.; Ding, W.; Li, C. Identification and Antifungal Activity of Compounds from the Mangrove Endophytic Fungus Aspergillus clavatus R7. Mar. Drugs 2017, 15, 259. [Google Scholar] [CrossRef]
- Bai, Z.Q.; Wang, Y.; Lin, X.; Wang, Y.; Wang, J.; Zhou, X.; Yang, B.; Liu, J.; Yang, X.; Liu, Y. New phenyl derivatives from endophytic fungus Aspergillus flavipes AIL8 derived of mangrove plant Acanthus ilicifolius. Fitoterapia 2014, 95, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Lu, Z.; Qu, H.J.; Liu, P.; Miao, C.; Zhu, T.; Li, J.; Hong, K.; Zhu, W. Antimicrobial aflatoxins from the marine-derived fungus Aspergillus flavus 092008. Arch. Pharm. Res. 2012, 35, 1387–1392. [Google Scholar] [CrossRef]
- Handayani, D.; Rivai, H.; Hutabarat, M.; Rasyid, R. Antibacterial activity of endophytic fungi isolated from mangrove plant Sonneratia griffithii Kurz. J. Appl. Pharm. Sci. 2017, 7, 209–212. [Google Scholar] [CrossRef][Green Version]
- Wu, Y.; Chen, Y.; Huang, X.; Pan, Y.; Liu, Z.; Yan, T.; Cao, W.; She, Z. α-Glucosidase Inhibitors: Diphenyl Ethers and Phenolic Bisabolane Sesquiterpenoids from the Mangrove Endophytic Fungus Aspergillus flavus QQSG-3. Mar. Drugs 2018, 16, 307. [Google Scholar] [CrossRef]
- Guo, Z.; Gai, C.; Cai, C.; Chen, L.; Liu, S.; Zeng, Y.; Yuan, J.; Mei, W.; Dai, H. Metabolites with Insecticidal Activity from Aspergillus fumigatus JRJ111048 Isolated from Mangrove Plant Acrostichum specioum Endemic to Hainan Island. Mar. Drugs 2017, 15, 381. [Google Scholar] [CrossRef] [PubMed]
- An, C.Y.; Li, X.M.; Luo, H.; Li, C.S.; Wang, M.H.; Xu, G.M.; Wang, B.G. 4-Phenyl-3,4-dihydroquinolone derivatives from aspergillus nidulans MA-143, an endophytic fungus isolated from the mangrove plant rhizophora stylosa. J. Nat. Prod. 2013, 76, 1896–1901. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.Q.; Li, X.M.; Xu, G.M.; Li, X.; An, C.Y.; Wang, B.G. Antibacterial anthraquinone derivatives isolated from a mangrove-derived endophytic fungus Aspergillus nidulans by ethanol stress strategy. J. Antibiot. 2018, 71, 778–784. [Google Scholar] [CrossRef]
- Liu, D.; Li, X.M.; Li, C.S.; Wang, B.G. Nigerasterols A and B, antiproliferative sterols from the mangrove-derived endophytic fungus Aspergillus niger MA-132. Helv. Chim. Acta 2013, 96, 1055–1061. [Google Scholar] [CrossRef]
- Nurunnabi, T.R.; Sabrin, F.; Sharif, D.I.; Nahar, L.; Sohrab, M.H.; Sarker, S.D.; Rahman, S.M.M.; Billah, M.M. Antimicrobial activity of endophytic fungi isolated from the mangrove plant Sonneratia apetala (Buch.-Ham) from the Sundarbans mangrove forest. Adv. Tradit. Med. 2020, 20, 419–425. [Google Scholar] [CrossRef]
- Nurunnabi, T.R.; Nahar, L.; Al-Majmaie, S.; Rahman, S.M.M.; Sohrab, M.H.; Billah, M.M.; Ismail, F.M.D.; Rahman, M.M.; Sharples, G.P.; Sarker, S.D. Anti-MRSA activity of oxysporone and xylitol from the endophytic fungus Pestalotia sp. growing on the Sundarbans mangrove plant Heritiera fomes. Phyther. Res. 2018, 32, 348–354. [Google Scholar] [CrossRef]
- Li, S.; Wei, M.; Chen, G.; Lin, Y. Two new dihydroisocoumarins from the endophytic fungus Aspergillus sp. Collected from the South China Sea. Chem. Nat. Compd. 2012, 48, 371–373. [Google Scholar] [CrossRef]
- Akinduyite, A.E.; Ariole, C.N. Bioactive compounds and antibacterial activity of endophytic fungi isolated from Black Mangrove (Avicennia africana) leaves. Niger. J. Biotechnol. 2019, 35, 35. [Google Scholar] [CrossRef]
- Wang, L.; Han, X.; Zhu, G.; Wang, Y.; Chairoungdua, A.; Piyachaturawat, P.; Zhu, W. Polyketides from the Endophytic Fungus Cladosporium sp. Isolated From the Mangrove Plant Excoecaria agallocha. Front. Chem. 2018, 6, 344. [Google Scholar] [CrossRef]
- Wu, Y.; Chen, S.; Liu, H.; Huang, X.; Liu, Y.; Tao, Y.; She, Z. Cytotoxic isocoumarin derivatives from the mangrove endophytic fungus Aspergillus sp. HN15-5D. Arch. Pharm. Res. 2019, 42, 326–331. [Google Scholar] [CrossRef]
- Cai, R.; Jiang, H.; Zang, Z.; Li, C.; She, Z. New Benzofuranoids and Phenylpropanoids from the Mangrove Endophytic Fungus, Aspergillus sp. ZJ-68. Mar. Drugs 2019, 17, 478. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.B.; Feng, X.J.; Liu, L.; Chen, B.; Lu, Y.J.; Ma, L.; She, Z.G.; Lin, Y.C. Three dimeric naphtho-γ-pyrones from the mangrove endophytic fungus Aspergillus tubingensis isolated from Pongamia pinnata. Planta Med. 2010, 76, 1888–1891. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Liu, Y.; Ding, M.; Zhang, Z.; Liu, H.; Huang, X.; She, Z. New pyranonaphthazarin and 2-naphthoic acid derivatives from the mangrove endophytic fungus Leptosphaerulina sp. SKS032. Phytochem. Lett. 2017, 20, 214–217. [Google Scholar] [CrossRef]
- Zhu, M.; Zhang, X.; Feng, H.; Che, Q.; Zhu, T.; Gu, Q.; Li, D. Campyridones A–D, pyridone alkaloids from a mangrove endophytic fungus Campylocarpon sp. HDN13-307. Tetrahedron 2016, 72, 5679–5683. [Google Scholar] [CrossRef]
- Chaeprasert, S.; Piapukiew, J.; Whalley, A.J.S.; Sihanonth, P. Endophytic fungi from mangrove plant species of Thailand: Their antimicrobial and anticancer potentials. Bot. Mar. 2010, 53, 555–564. [Google Scholar] [CrossRef]
- Ai, W.; Lin, X.; Wang, Z.; Lu, X.; Mangaladoss, F.; Yang, X.; Zhou, X.; Tu, Z.; Liu, Y. Cladosporone A, a new dimeric tetralone from fungus Cladosporium sp. KcFL6′ derived of mangrove plant Kandelia candel. J. Antibiot. 2015, 68, 213–215. [Google Scholar] [CrossRef]
- Bin, G.; Chen, Y.; Zhang, H.; Zheng, X.; Zhang, Y.; Fang, H.; Zhong, Q.; Chenxiao, Z. Isolation, characterization and anti-multiple drug resistant (MDR) bacterial activity of endophytic fungi isolated from the mangrove plant, Aegiceras corniculatum. Trop. J. Pharm. Res. 2014, 13, 593–599. [Google Scholar] [CrossRef]
- Luo, X.W.; Chen, C.M.; Li, K.L.; Lin, X.P.; Gao, C.H.; Zhou, X.F.; Liu, Y.H. Sesquiterpenoids and meroterpenoids from a mangrove derived fungus Diaporthe sp. SCSIO 41011. Nat. Prod. Res. 2019, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Deng, Q.; Sun, M.; Xu, J. Cytospyrone and Cytospomarin: Two new polyketides isolated from mangrove endophytic fungus, Cytospora sp †. Molecules 2020, 25, 4224. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.X.; Shao, T.M.; Mei, R.Q.; Huang, G.L.; Zhou, X.M.; Zheng, C.J.; Wang, C.Y. Bioactive Secondary Metabolites from the Culture of the Mangrove-Derived Fungus Daldinia eschscholtzii HJ004. Mar. Drugs 2019, 17, 710. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Yang, J.; Chen, F.; Lin, X.; Chen, C.; Zhou, X.; Liu, S.; Liu, Y. Structurally diverse polyketides from the mangrove-derived fungus diaporthe sp. SCSIO 41011 with their anti-influenza A virus activities. Front. Chem. 2018, 6, 282. [Google Scholar] [CrossRef]
- Du, X.P.; Su, W.J. Two new polyketides from mangrove endophytic fungus dothiorella sp. Chem. Nat. Compd. 2014, 50, 214–216. [Google Scholar] [CrossRef]
- Zhang, G.; Sun, S.; Zhu, T.; Lin, Z.; Gu, J.; Li, D.; Gu, Q. Antiviral isoindolone derivatives from an endophytic fungus Emericella sp. associated with Aegiceras corniculatum. Phytochemistry 2011, 72, 1436–1442. [Google Scholar] [CrossRef] [PubMed]
- Mei, R.Q.; Nong, X.H.; Wang, B.; Sun, X.P.; Huang, G.L.; Luo, Y.P.; Zheng, C.J.; Chen, G.Y. A new phenol derivative isolated from mangrove-derived fungus Eupenicillium sp. HJ002. Nat. Prod. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- May Zin, W.W.; Buttachon, S.; Dethoup, T.; Pereira, J.A.; Gales, L.; Inácio, Â.; Costa, P.M.; Lee, M.; Sekeroglu, N.; Silva, A.M.S.; et al. Antibacterial and antibiofilm activities of the metabolites isolated from the culture of the mangrove-derived endophytic fungus Eurotium chevalieri KUFA 0006. Phytochemistry 2017, 141, 86–97. [Google Scholar] [CrossRef]
- Yan, H.J.; Li, X.M.; Li, C.S.; Wang, B.G. Alkaloid and anthraquinone derivatives produced by the marine-derived endophytic fungus Eurotium rubrum. Helv. Chim. Acta 2012, 95, 163–168. [Google Scholar] [CrossRef]
- Supratman, U.; Hirai, N.; Sato, S.; Watanabe, K.; Malik, A.; Annas, S.; Harneti, D.; Maharani, R.; Koseki, T.; Shiono, Y. New naphthoquinone derivatives from Fusarium napiforme of a mangrove plant. Nat. Prod. Res. 2019, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ling, O.M.; Teen, L.P.; Mujahid, A.; Proksch, P.; Müller, M. Initial screening of mangrove endophytic fungi for antimicrobial compounds and heavy metal biosorption potential. Sains Malays. 2016, 45, 1063–1071. [Google Scholar]
- Mei, W.L.; Zheng, B.; Zhao, Y.X.; Zhong, H.M.; Chen, X.L.W.; Zeng, Y.B.; Dong, W.H.; Huang, J.L.; Proksch, P.; Dai, H.F. Meroterpenes from endophytic fungus A1 of mangrove plant Scyphiphora hydrophyllacea. Mar. Drugs 2012, 10, 1993–2001. [Google Scholar] [CrossRef] [PubMed]
- Chi, W.C.; Pang, K.L.; Chen, W.L.; Wang, G.J.; Lee, T.H. Antimicrobial and iNOS inhibitory activities of the endophytic fungi isolated from the mangrove plant Acanthus ilicifolius var. xiamenensis. Bot. Stud. 2019, 60, 4. [Google Scholar] [CrossRef] [PubMed]
- Ratnaweera, P.B.; De Silva, E.D.; Wijesundera, R.L.; Andersen, R.J. Antimicrobial constituents of Hypocrea virens, an endophyte of the mangrove-associate plant Premna serratifolia L. J. Natl. Sci. Found. Sri Lanka 2016, 44, 43. [Google Scholar] [CrossRef]
- Chen, S.; Liu, Z.; Liu, H.; Long, Y.; Chen, D.; Lu, Y.; She, Z. Lasiodiplactone A, a novel lactone from the mangrove endophytic fungus Lasiodiplodia theobromae ZJ-HQ1. Org. Biomol. Chem. 2017, 15, 6338–6341. [Google Scholar] [CrossRef] [PubMed]
- Mazlan, N.W.; Tate, R.; Yusoff, Y.M.; Clements, C.; Edrada-Ebel, R. Metabolomics-Guided Isolation of Anti-Trypanosomal Compounds from Endophytic Fungi of the Mangrove plant Avicennia Lanata. Curr. Med. Chem. 2019, 27, 1815–1835. [Google Scholar] [CrossRef]
- Cui, H.; Liu, Y.; Li, T.; Zhang, Z.; Ding, M.; Long, Y.; She, Z. 3-Arylisoindolinone and sesquiterpene derivatives from the mangrove endophytic fungi Aspergillus versicolor SYSU-SKS025. Fitoterapia 2018, 124, 177–181. [Google Scholar] [CrossRef]
- Yu, G.; Zhou, G.; Zhu, M.; Wang, W.; Zhu, T.; Gu, Q.; Li, D. Neosartoryadins A and B, Fumiquinazoline Alkaloids from a Mangrove-Derived Fungus Neosartorya udagawae HDN13-313. Org. Lett. 2015. [Google Scholar] [CrossRef]
- Xia, X.; Li, Q.; Li, J.; Shao, C.; Zhang, J.; Zhang, Y.; Liu, X.; Lin, Y.; Liu, C.; She, Z. Two new derivatives of griseofulvin from the mangrove endophytic fungus nigrospora sp(Strain No.1403) from Kandelia candel (L.) Druce. Planta Med. 2011, 77, 1735–1738. [Google Scholar] [CrossRef]
- Ukwatta, K.M.; Lawrence, J.L.; Wijayarathna, C.D. The study of antimicrobial, anti-cancer, anti-inflammatory and α-glucosidase inhibitory activities of Nigronapthaphenyl, isolated from an extract of Nigrospora sphaerica. Mycol. Int. J. Fungal Biol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.H.; Zhang, P.; Li, X.M.; Wang, B.G. Penicibrocazines A-E, five new sulfide diketopiperazines from the marine-derived endophytic fungus Penicillium brocae. Mar. Drugs 2015, 13, 276–287. [Google Scholar] [CrossRef]
- Meng, L.H.; Li, X.M.; Liu, Y.; Xu, G.M.; Wang, B.G. Antimicrobial alkaloids produced by the mangrove endophyte Penicillium brocae MA-231 using the OSMAC approach. RSC Adv. 2017, 7, 55026–55033. [Google Scholar] [CrossRef]
- Devi, P.; Rodrigues, C.; Naik, C.G.; D’Souza, L. Isolation and Characterization of Antibacterial Compound from a Mangrove-Endophytic Fungus, Penicillium chrysogenum MTCC 5108. Indian J. Microbiol. 2012, 52, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Wu, Z.; Liang, F.; Gan, S.; Huang, Q.; Ding, W.; Li, C. A New L-alanine Derivative from the Mangrove Fungus Penicillium chrysogenum V11. Chem. Nat. Compd. 2018, 54, 520–522. [Google Scholar] [CrossRef]
- Huang, S.; Xu, J.; Li, F.; Zhou, D.; Xu, L.; Li, C. Identification and Antifungal Activity of Metabolites from the Mangrove Fungus Phoma sp. L28. Chem. Nat. Compd. 2017, 53, 237–240. [Google Scholar] [CrossRef]
- He, K.Y.; Zhang, C.; Duan, Y.R.; Huang, G.L.; Yang, C.Y.; Lu, X.R.; Zheng, C.J.; Chen, G.Y. New chlorinated xanthone and anthraquinone produced by a mangrove-derived fungus Penicillium citrinum HL-5126. J. Antibiot. 2017, 70, 823–827. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Li, X.M.; Li, X.; Li, H.L.; Meng, L.H.; Wang, B.G. New lactone and isocoumarin derivatives from the marine mangrove-derived endophytic fungus Penicillium coffeae MA-314. Phytochem. Lett. 2019, 32, 1–5. [Google Scholar] [CrossRef]
- Hayibor, K.; Kwain, S.; Osei, E.; Nartey, A.P.; Tetevi, G.M.; Owusu, K.B.-A.; Camas, M.; Camas, A.S.; Kyeremeh, K. Ghanaian mangrove wetland endophytic fungus, Penicillium herquei strain BRS2A-AR produces (9Z, 11E)-13-oxooctadeca-9,11-dienoic acid with activity against Trichomonas mobilensis. Int. J. Biol. Chem. Sci. 2019, 13, 1918. [Google Scholar] [CrossRef]
- Xu, R.; Li, X.M.; Wang, B.G. Penicisimpins A–C, three new dihydroisocoumarins from Penicillium simplicissimum MA-332, a marine fungus derived from the rhizosphere of the mangrove plant Bruguiera sexangula var. rhynchopetala. Phytochem. Lett. 2016, 17, 114–118. [Google Scholar] [CrossRef]
- Zheng, C.; Chen, Y.; Jiang, L.L.; Shi, X.M. Antiproliferative metabolites from the endophytic fungus Penicillium sp. FJ-1 isolated from a mangrove Avicennia marina. Phytochem. Lett. 2014, 10, 272–275. [Google Scholar] [CrossRef]
- Ding, B.; Wang, Z.; Huang, X.; Liu, Y.; Chen, W.; She, Z. Bioactive α-pyrone meroterpenoids from mangrove endophytic fungus Penicillium sp. Nat. Prod. Res. 2016, 30, 2805–2812. [Google Scholar] [CrossRef]
- Qi, X.; Li, X.; Zhao, J.; He, N.; Li, Y.; Zhang, T.; Wang, S.; Yu, L.; Xie, Y. GKK1032C, a new alkaloid compound from the endophytic fungus Penicillium sp. CPCC 400817 with activity against methicillin-resistant S. aureus. J. Antibiot. 2019, 72, 237–240. [Google Scholar] [CrossRef]
- Hemberger, Y.; Xu, J.; Wray, V.; Proksch, P.; Wu, J.; Bringmann, G. Pestalotiopens A and B: Stereochemically Challenging Flexible Sesquiterpene-Cyclopaldic Acid Hybrids from Pestalotiopsis sp. Chem. A Eur. J. 2013, 19, 15556–15564. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Xiong, B.; Xu, J. Chemical Investigation of Secondary Metabolites Produced by Mangrove Endophytic Fungus Phyllosticta Capitalensis. Nat. Prod. Res. 2019. [Google Scholar] [CrossRef]
- Xu, Z.; Wu, X.; Li, G.; Feng, Z.; Xu, J. Pestalotiopisorin B, a new isocoumarin derivative from the mangrove endophytic fungus Pestalotiopsis sp. HHL101. Nat. Prod. Res. 2020, 34, 1002–1007. [Google Scholar] [CrossRef]
- Huang, G.L.; Zhou, X.M.; Bai, M.; Liu, Y.X.; Zhao, Y.L.; Luo, Y.P.; Niu, Y.Y.; Zheng, C.J.; Chen, G.Y. Dihydroisocoumarins from the mangrove-derived fungus Penicillium citrinum. Mar. Drugs 2016, 14, 177. [Google Scholar] [CrossRef] [PubMed]
- Klaiklay, S.; Rukachaisirikul, V.; Phongpaichit, S.; Pakawatchai, C.; Saithong, S.; Buatong, J.; Preedanon, S.; Sakayaroj, J. Anthraquinone derivatives from the mangrove-derived fungus Phomopsis sp. PSU-MA214. Phytochem. Lett. 2012, 5, 738–742. [Google Scholar] [CrossRef]
- Zhang, D.; Tao, X.; Chen, R.; Liu, J.; Li, L.; Fang, X.; Yu, L.; Dai, J. Pericoannosin A, a Polyketide Synthase-Nonribosomal Peptide Synthetase Hybrid Metabolite with New Carbon Skeleton from the Endophytic Fungus Periconia sp. Org. Lett. 2015, 17, 4304–4307. [Google Scholar] [CrossRef] [PubMed]
- Cai, R.; Chen, S.; Liu, Z.; Tan, C.; Huang, X.; She, Z. A new α-pyrone from the mangrove endophytic fungus Phomopsis sp. HNY29-2B. Nat. Prod. Res. 2017, 2, 124–130. [Google Scholar] [CrossRef]
- Wen, S.; Fan, W.; Guo, H.; Huang, C.; Yan, Z.; Long, Y. Two new secondary metabolites from the mangrove endophytic fungus Pleosporales sp. SK7. Nat. Prod. Res. 2019. [Google Scholar] [CrossRef]
- Joel, E.L.; Bhimba, B.V. A secondary metabolite with antibacterial activity produced by mangrove foliar fungus Schizophyllum commune. Int. J. Chem. Env. Biol. Scn. 2013, 1, 2320–4087. [Google Scholar]
- Zhou, X.M.; Zheng, C.J.; Chen, G.Y.; Song, X.P.; Han, C.R.; Li, G.N.; Fu, Y.H.; Chen, W.H.; Niu, Z.G. Bioactive anthraquinone derivatives from the mangrove-derived fungus stemphylium sp. 33231. J. Nat. Prod. 2014, 77, 2021–2028. [Google Scholar] [CrossRef]
- Liu, F.; Cai, X.L.; Yang, H.; Xia, X.K.; Guo, Z.Y.; Yuan, J.; Li, M.F.; She, Z.G.; Lin, Y.C. The bioactive metabolites of the mangrove endophytic fungus talaromyces sp. ZH-154 isolated from kandelia candel (L.) Druce. Planta Med. 2010, 76, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Niaz, S.I.; Wang, Z.; Zhu, Y.; Lin, Y.; Li, J.; Liu, L. α-Glucosidase inhibitory and cytotoxic botryorhodines from mangrove endophytic fungus Trichoderma sp. 307. Nat. Prod. Res. 2018, 32, 2887–2892. [Google Scholar] [CrossRef]
- Arora, D.; Sharma, N.; Singamaneni, V.; Sharma, V.; Kushwaha, M.; Abrol, V.; Guru, S.; Sharma, S.; Gupta, A.P.; Bhushan, S.; et al. Isolation and characterization of bioactive metabolites from Xylaria psidii, an endophytic fungus of the medicinal plant Aegle marmelos and their role in mitochondrial dependent apoptosis against pancreatic cancer cells. Phytomedicine 2016, 23, 1312–1320. [Google Scholar] [CrossRef] [PubMed]
- Lopéz, D.; Cherigo, L.; Mejia, L.C.; Loza-Mejía, M.A.; Martínez-Luis, S. α-Glucosidase inhibitors from a mangrove associated fungus, Zasmidium sp. strain EM5-10. BMC Chem. 2019, 13, 22. [Google Scholar] [CrossRef] [PubMed]
- Strobel, G.; Daisy, B. Bioprospecting for Microbial Endophytes and Their Natural Products. Microbiol. Mol. Biol. Rev. 2003, 67, 491–502. [Google Scholar] [CrossRef]
- Saad, M.M.G.; Ghareeb, R.Y.; Saeed, A.A. The potential of endophytic fungi as bio-control agents against the cotton leafworm, Spodoptera littoralis (Boisd.) (Lepidoptera: Noctuidae). Egypt. J. Biol. Pest Control 2019, 29, 7. [Google Scholar] [CrossRef]
- Challis, G.L. Mining microbial genomes for new natural products and biosynthetic pathways. Microbiology 2008, 154, 1555–1569. [Google Scholar] [CrossRef] [PubMed]
- Cichewicz, R.H. Epigenome manipulation as a pathway to new natural product scaffolds and their congeners. Nat. Prod. Rep. 2010, 27, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Chiang, Y.M.; Chang, S.L.; Oakley, B.R.; Wang, C.C.C. Recent advances in awakening silent biosynthetic gene clusters and linking orphan clusters to natural products in microorganisms. Curr. Opin. Chem. Biol. 2011. [Google Scholar] [CrossRef] [PubMed]
- Beau, J.; Mahid, N.; Burda, W.N.; Harrington, L.; Shaw, L.N.; Mutka, T.; Kyle, D.E.; Barisic, B.; van Olphen, A.; Baker, B.J. Epigenetic Tailoring for the Production of Anti-Infective Cytosporones from the Marine Fungus Leucostoma persoonii. Mar. Drugs 2012, 10, 762–774. [Google Scholar] [CrossRef]
- Lim, F.Y.; Sanchez, J.F.; Wang, C.C.C.; Keller, N.P. Toward awakening cryptic secondary metabolite gene clusters in filamentous fungi. In Methods in Enzymology; Academic Press Inc.: Cambridge, MA, USA, 2012; pp. 303–324. [Google Scholar] [CrossRef]
- González-Menéndez, V.; Pérez-Bonilla, M.; Pérez-Victoria, I.; Martín, J.; Muñoz, F.; Reyes, F.; Tormo, J.; Genilloud, O. Multicomponent Analysis of the Differential Induction of Secondary Metabolite Profiles in Fungal Endophytes. Molecules 2016, 21, 234. [Google Scholar] [CrossRef]
- Demers, D.; Knestrick, M.; Fleeman, R.; Tawfik, R.; Azhari, A.; Souza, A.; Vesely, B.; Netherton, M.; Gupta, R.; Colon, B.; et al. Exploitation of Mangrove Endophytic Fungi for Infectious Disease Drug Discovery. Mar. Drugs 2018, 16, 376. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.Z.; Li, X.M.; Li, X.; Yang, S.Q.; Meng, L.H.; Wang, B.G. Polyketides from the mangrove-derived endophytic fungus Cladosporium cladosporioides. Mar. Drugs 2019, 17, 296. [Google Scholar] [CrossRef]
- World Health Organization. The World Malaria Report 2018; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Shang, Z.; Li, X.-M.; Li, C.-S.; Wang, B.-G. Diverse Secondary Metabolites Produced by Marine-Derived Fungus Nigrospora sp. MA75 on Various Culture Media. Chem. Biodivers. 2012, 9, 1338–1348. [Google Scholar] [CrossRef]
- Wang, J.; Wei, X.; Lu, X.; Xu, F.; Wan, J.; Lin, X.; Zhou, X.; Liao, S.; Yang, B.; Tu, Z.; et al. Eight new polyketide metabolites from the fungus Pestalotiopsis vaccinii endogenous with the mangrove plant Kandelia candel (L.) Druce. Tetrahedron 2014, 70, 9695–9701. [Google Scholar] [CrossRef]
- Campos, F.F.; Sales Junior, P.A.; Romanha, A.J.; Araújo, M.S.S.; Siqueira, E.P.; Resende, J.M.; Alves, T.M.A.; Martins-Filho, O.A.; Dos Santos, V.L.; Rosa, C.A.; et al. Bioactive endophytic fungi isolated from caesalpinia echinata Lam. (Brazilwood) and identification of beauvericin as a trypanocidal metabolite from fusarium sp. Mem. Inst. Oswaldo Cruz 2015, 110, 1–10. [Google Scholar] [CrossRef]
- Ferreira, M.C.; Vieira M de, L.A.; Zani, C.L.; Alves TM de, A.; Junior, P.A.S.; Murta, S.M.F.; Romanha, A.J.; Gil, L.H.V.G.; Carvalho AG de, O.; Zilli, J.E.; et al. Molecular phylogeny, diversity, symbiosis and discover of bioactive compounds of endophytic fungi associated with the medicinal Amazonian plant Carapa guianensis Aublet (Meliaceae). Biochem. Syst. Ecol. 2015, 59, 36–44. [Google Scholar] [CrossRef]
- Rosa, L.H.; Gonçalves, V.N.; Caligiorne, R.B.; Alves, T.M.A.; Rabello, A.; Sales, P.A.; Romanha, A.J.; Sobral, M.E.G.; Rosa, C.A.; Zani, C.L. Leishmanicidal, trypanocidal, and cytotoxic activities of endophytic fungi associated with bioactive plants in Brazil. Braz. J. Microbiol. 2010, 41, 420–430. [Google Scholar] [CrossRef]
- De Carvalho, C.R.; De Lourdes Almeida Vieira, M.; Cantrell, C.L.; Wedge, D.E.; Alves, T.M.A.; Zani, C.L.; Pimenta, R.S.; Sales, P.A.; Murta, S.M.F.; Romanha, A.J.; et al. Biological activities of ophiobolin K and 6-epi-ophiobolin K produced by the endophytic fungus Aspergillus calidoustus. Nat. Prod. Res. 2016, 30, 478–481. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Yin, R.; Zhou, Z.; Gu, G.; Dai, J.; Lai, D.; Zhou, L. Eremophilane-Type Sesquiterpenoids From the Endophytic Fungus Rhizopycnis vagum and Their Antibacterial, Cytotoxic, and Phytotoxic Activities. Front. Chem. 2020, 8, 596889. [Google Scholar] [CrossRef]
- Mansoldo, F.R.P.; Carta, F.; Angeli, A.; Cardoso V da, S.; Supuran, C.T.; Vermelho, A.B. Chagas Disease: Perspectives on the Past and Present and Challenges in Drug Discovery. Molecules 2020, 25, 5483. [Google Scholar] [CrossRef]
- Newell, D.G.; Koopmans, M.; Verhoef, L.; Duizer, E.; Aidara-Kane, A.; Sprong, H.; Opsteegh, M.; Langelaar, M.; Threfall, J.; Scheutz, F.; et al. Food-borne diseases-The challenges of 20years ago still persist while new ones continue to emerge. Int. J. Food Microbiol. 2010, 139, S3–S15. [Google Scholar] [CrossRef]
- Li, H.-Y.; Zhu, G.-J.; Zhang, Y.-Z.; Zhang, L.-B.; Hagan, E.A.; Martinez, S.; Chmura, A.A.; Francisco, L.; Tai, H.; Miller, M.; et al. A qualitative study of zoonotic risk factors among rural communities in southern China. Int. Health 2020, 12, 77–85. [Google Scholar] [CrossRef]
- Säde, E.; Murros, A.; Björkroth, J. Predominant enterobacteria on modified-atmosphere packaged meat and poultry. Food Microbiol. 2013, 34, 252–258. [Google Scholar] [CrossRef]
- Mouttotou, N.; Ahmad, S.; Kamran, Z.; Koutoulis, K.C. Prevalence, Risks and Antibiotic Resistance of Salmonella in Poultry Production Chain, in: Current Topics in Salmonella and Salmonellosis. InTech 2017. [Google Scholar] [CrossRef]
- Shang, K.; Wei, B.; Kang, M. Distribution and dissemination of antimicrobial-resistant Salmonella in broiler farms with or without enrofloxacin use. BMC Vet. Res. 2018, 14, 257. [Google Scholar] [CrossRef]
- Chen, J.; Xing, X.K.; Zhang, L.C.; Xing, Y.M.; Guo, S.X. Identification of Hortaea werneckii Isolated from Mangrove Plant Aegiceras comiculatum Based on Morphology and rDNA Sequences. Mycopathologia 2012, 174, 457–466. [Google Scholar] [CrossRef]
- Ai, W.; Wei, X.; Lin, S.; Wang, Z.; Tu, Z.; Yang, X.; Zhou, X.; Li, J.; Liu, Y. Guignardins AeF, spirodioxynaphthalenes from the endophytic fungus Guignardia sp. KcF8 as a new class of PTP1B and SIRT1 inhibitors. Tetrahedron 2014, 70, 5806–5814. [Google Scholar] [CrossRef]
- Bergelson, J.M.; Cunningham, J.A.; Droguett, G.; Kurt-Jones, E.A.; Krithivas, A.; Hong, J.S.; Horwitz, M.S.; Crowell, R.L.; Finberg, R.W. Isolation of a common receptor for coxsackie B viruses and adenoviruses 2 and 5. Science 1997, 275, 1320–1323. [Google Scholar] [CrossRef]
- Marjomäki, V.; Turkki, P.; Huttunen, M. Infectious Entry Pathway of Enterovirus B Species. Viruses 2015, 7, 6387–6399. [Google Scholar] [CrossRef] [PubMed]
- Linnakoski, R.; Reshamwala, D.; Veteli, P.; Cortina-Escribano, M.; Vanhanen, H.; Marjomäki, V. Antiviral agents from fungi: Diversity, mechanisms and potential applications. Front. Microbiol. 2018. [Google Scholar] [CrossRef]
- Chang, J.; Block, T.M.; Guo, J.T. Antiviral therapies targeting host ER alpha-glucosidases: Current status and future directions. Antivir. Res. 2013. [Google Scholar] [CrossRef]
- Ma, J.; Zhang, X.; Soloveva, V.; Warren, T.; Guo, F.; Wu, S.; Lu, H.; Guo, J.; Su, Q.; Shen, H.; et al. Enhancing the antiviral potency of ER α-glucosidase inhibitor IHVR-19029 against hemorrhagic fever viruses in vitro and in vivo. Antivir. Res. 2018, 150, 112–122. [Google Scholar] [CrossRef]
- González-Aldaco, K.; Torres-Reyes, L.A.; Ojeda-Granados, C.; José-Ábrego, A.; Fierro, N.A.; Román, S. Immunometabolic Effect of Cholesterol in Hepatitis C Infection: Implications in Clinical Management and Antiviral Therapy Concise Review. Ann. Hepatol. Off. J. Mex. Assoc. Hepatol. 2018, 17, 908–919. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, L.; Falanga, A.; Del Genio, V.; Palomba, L.; Galdiero, M.; Franci, G.; Galdiero, S. A boost to the antiviral activity: Cholesterol tagged peptides derived from glycoprotein B of Herpes Simplex virus type I. Int. J. Biol. Macromol. 2020, 162, 882–893. [Google Scholar] [CrossRef]
- Ashkenazi, A.; Fairbrother, W.J.; Leverson, J.D.; Souers, A.J. From basic apoptosis discoveries to advanced selective BCL-2 family inhibitors. Nat. Rev. Drug Discov. 2017. [Google Scholar] [CrossRef]
- Ebert, G.; Preston, S.; Allison, C.; Cooney, J.; Toe, J.G.; Stutz, M.D.; Ojaimi, S.; Scott, H.W.; Baschuk, N.; Nachbur, U.; et al. Cellular inhibitor of apoptosis proteins prevent clearance of hepatitis B virus. Proc. Natl. Acad. Sci. USA 2015, 112, 5797–5802. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.M.; Kim, J.; Tenson, T.; Min, J.Y.; Kainov, D.E. Influenza virus infection, interferon response, viral counter-response, and apoptosis. Viruses 2017, 9, 223. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.R.; Carté, B.K.; Sidebottom, P.J.; Yew, A.L.S.; Ng, S.B.; Huang, Y.; Butler, M.S. Circumdatin G, a new alkaloid from the fungus Aspergillus ochraceus. J. Nat. Prod. 2001, 64, 125–126. [Google Scholar] [CrossRef] [PubMed]
- Au, J.S.; Pockros, P.J. Novel Therapeutic Approaches for Hepatitis C. Clin. Pharmacol. Ther. 2013, 95, 78–88. [Google Scholar] [CrossRef]
- Wheeler, N.C.; Jech, K.; Masters, S.; Brobst, S.W.; Alvarado, A.B.; Hoover, A.J.; Snader, K.M. Effects of genetic, epigenetic, and environmental factors on taxol content in taxus brevifolia and related species. J. Nat. Prod. 1992, 55, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Roopa, G.; Madhusudhan, M.C.; Sunil, K.C.R.; Lisa, N.; Calvin, R.; Poornima, R.; Zeinab, N.; Kini, K.R.; Prakash, H.S.; Geetha, N. Identification of Taxol-producing endophytic fungi isolated from Salacia oblonga through genomic mining approach. J. Genet. Eng. Biotechnol. 2015, 13, 119–127. [Google Scholar] [CrossRef]
- Elavarasi, A.; Rathna, G.S.; Kalaiselvam, M. Taxol producing mangrove endophytic fungi Fusarium oxysporum from Rhizophora annamalayana. Asian Pac. J. Trop. Biomed. 2012, 2, S1081–S1085. [Google Scholar] [CrossRef]
- Ryang, J.; Yan, Y.; Song, Y.; Liu, F.; Ng, T.B. Anti-HIV, antitumor and immunomodulatory activities of paclitaxel from fermentation broth using molecular imprinting technique. AMB Express 2019, 9. [Google Scholar] [CrossRef]
- Rajamani, T.; Suryanarayanan, T.S.; Murali, T.S.; Thirunavukkarasu, N. Distribution and diversity of foliar endophytic fungi in the mangroves of Andaman Islands, India. Fungal Ecol. 2018, 36, 109–116. [Google Scholar] [CrossRef]
- Pamphile, J.A.; dos Santos Ribeiro, M.A.; Polonio, J.C. Secondary metabolites of endophyte fungi: Techniques and biotechnological approaches. In Diversity and Benefits of Microorganisms from the Tropics; Springer International Publishing: New York, NY, USA, 2017; pp. 185–206. [Google Scholar] [CrossRef]
- Deshmukh, S.K.; Society, M. Sunil Kumar Deshmukh. KAVAKA 2018, 13, 1–13. [Google Scholar]
- Marmann, A.; Aly, A.H.; Lin, W.; Wang, B.; Proksch, P. Co-cultivation-A powerful emerging tool for enhancing the chemical diversity of microorganisms. Mar. Drugs 2014, 12, 1043–1065. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.C.; Kauffman, C.A.; Jensen, P.R.; Fenical, W. Induced production of emericellamides A and B from the marine-derived fungus Emericella sp. in competing co-culture. J. Nat. Prod. 2007, 70, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.; Chen, G.; Chen, X.; Huang, M.; Wan, X. Aspergicin, a new antibacterial alkaloid produced by mixed fermentation of two marine-derived mangrove epiphytic fungi. Chem. Nat. Compd. 2011, 47, 767–769. [Google Scholar] [CrossRef]
| Chemical Class | Compounds Isolated | Fungus | Host Plant(s) | Localization | Biological Target | Reference |
|---|---|---|---|---|---|---|
Isocoumarin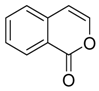 | trans and cis 4,8-dihydroxy-3-methylisochroman-1-one, 5-hydroxymellein and -mellein or 8-hydroxy-3-methylisochroman-1-one | Lasiodiplodia theobromae | Avicennia lanata | Terengganu, Malaysia | Trypanosoma brucei brucei | [75] |
Naphthoquinones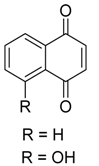 | Anhydrofusarubin, javanicin, dihydrojavanicin and solaniol | Fusarium sp. | Avicennia lanata | Terengganu, Malaysia | Trypanosoma brucei brucei | [75] |
Aflotoxin-derived mycotoxin | Aflatoxin B2b | Aspergillus flavus | Hibiscus tiliaceus | Hainan province, China | E. coli, B. subtilis and Enterobacter aerogenes | [40] |
Sesquiterpene | (7 S, 10 S)-7,10-ácido epoxysydonic; (7 R, 11 S) -7,12-epoxi s ácido ydonic; ácido 7-desoxi-7,14-didesidro-12-hydroxysydonic; (E) -7-desoxi-7,8-didesidro-12-ácido hydroxysydonic Pestalotiopen A | Aspergillus sp. | Xylocarpus moluccensis | Trang Province, Thailand | Staphylococcus aureus | [40,51] |
| Pestalotiopsis sp. | Rhizophora mucronata | Hainan Island, China | Enterococcus faecalis | [91] | ||
Polyketide-derived mycotoxin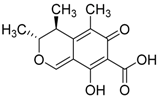 | 12α- acetoxy- 4,4-dimethyl-24-methylene-5α-cholesta-8-momoene-3β,11β-diol, 2α-acetoxy-4,4-dimethyl-24-methylene-5α-cholesta-8,14-diene-2α,3β,11β-triol | Penicillium sp. | Bruguiera sexangula var. Rhynchopetala | China | S. aureus, E. coli and Micrococcus tetragenu | [88] |
| Guignardone B and Guignardone I | Guignardia sp. | Scyphiphora hydrophyllacea | Hainan Province, China | Staphylococcus aureus (MRSA) and S. aureus. | [71] | |
| Guignardone A and guignardone J | Phyllosticta capitalensis | Bruguiera sexangula | Southern China | P. aeraeruginosa and S. aureus | [92] | |
Coumarin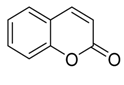 | Austinol | Penicillium citrinum | Bruguiera sexangula var. rhynchopetala | South China | S. aureus S. epidermidis | [85] |
| Bacillisporin A), bacillisporin B and Penicisimpins A–C | Penicillium simplicissimum | Hainan Island, China | Bacillus subtilis, Aeromonas hydrophilia, Escherichia coli, M. luteus, Pseudomonas aeruginosa, V. alginolyticus, V. harveyi and V. parahaemolyticus | [88] | ||
Isocoumarin | Dichlorodiaportintone, desmethyldichlorodiaportin, dichlorodiaportin | Ascomycota sp. | Pluchea indica | Guangxi Province, China | S. aureus, B. subtilis, E. coli, Klebsiella pneumoniae and Acinetobacter calcoaceticus | [37] |
| Spergillumarinas A and B | Aspergillus sp. | Bruguiera gymnorrhiza | SouthChina | S. aureus and B. subtilis | [49] | |
| Penicimarins G and H | Penicillium citrinum | Bruguiera sexangula var. rhynchopetala | S. aureus,S. epidermidis, Escherichia coli, Bacillus cereus and Vibrio alginolyticus | [85] | ||
| Pestalotiopisorin B | Pestalotiopsis sp. | Rhizophora stylosa | Hainan Island, China | E. coli and P. aeruginosa | [93] | |
4H-chromen-4-one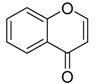 | 8-dihydroxy-chromone, bacillisporin A and bacillisporin B | Penicillium aculeatum | Kandelia candel | Yangjiang, Guangdong province, China | B. subtilis and Salmonella spp. | [94] |
Xanthones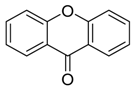 | 3,6,8-trihydroxy-1-methylxanthone | Nigrospora sp. | Pongamia pinnata | China | MRSA, E. coli and S. epidermidis | [117] |
Anthraquinone | Isoversicolorin C, versicolorin C | Aspergillus nidulans | Rhizophora stylosa | Twig, Chanthaburi Province, Eastern Thailand | E. coli, M. luteus, V. vulnificus, V. anguillarum, V. alginolyticus, Ed. ictaluri, V. parahaemolyticus S. aureus and E. Faecalis | [45] |
| Diaportheins B and Emodin | Eurotium chevalier | Rhizophora Mucronata | Hainan Island, China | E. coli | [67] | |
| 9-dehydroxyeurotinone | Eurotium rubrum | Hibiscus tiliaceu | South China | Staphylococcus aureus and Escherichia coli | [68] | |
| Bostrycin, and Deoxybostrycin | Nigrospora sp. | Kandelia candel | S.aureus, E.coli, P. aeruginosa, Sarcina ventriculi, B. subtilis | [78] | ||
| 2′-acetoxy-7-chlorocitreorosein | Penicillium citrinum | Bruguiera sexangula var. rhynchopetala | Vibrio parahaemolyticus | [84] | ||
| 2R,3S)-7-ethyl-1,2,3,4-tetrahydro-2,3,8-trihydroxy-6-methoxy-3-methyl-9,10-anthracenedione | Phomopsis sp. | Rhizophora apiculata | Songkhla province, Thailand | Staphylococcus aureus ATCC25923 and methicillin-resistant S. aureus SK1 | [96] | |
| 2-O-acetylaltersolanol B, Altersolanols A and B | Stemphylium sp. | Bruguiera sexangula var. rhynchopetala | South China | E. coli, S. aureus and B. subtilis | [96] | |
Naphthoquinones | 5-hydroxy-2-methoxy-6,7-dimethyl-1,4-naphthoquinone | Daldinia eschscholtzii | Bruguiera sexangula var. rhynchopetala | South China | B. cereus | [62] |
| 6-hydroxy-astropaquinone B, astropaquinone D and 3-O-methyl-9-O-methylfusarubin | Fusarium napiforme | Rhizophora mucronata | South Sulawesi Province, Indonesia | S. aureus and P.aeruginosa | [69] | |
Biphenyl | 5,50-dimethoxybiphenyl-2,20-diol | Phomopsis longicolla | Brguiera sexangula var. rhynchopetala | South China | Vibrio parahaemolyticus | [38] |
N-phenylnaphthalen-1-amine | Nigronapthaphenyl | Nigrospora sphaerica | Bruguiera gymnorrhyza | City of Galle, Sri Lanka | B, subtilis and Bacillus cereus | [115] |
Alkaloids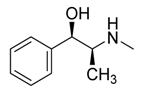 | GKK1032C | Penicillium sp. | Mangrove plant Chinese | Hainan province, China | methicillin-resistant S. aureus | [90] |
| Penicibrocazines B–E, Bbrocapyrrozins A and 4-hydroxy-3-phenyl-1H-pyrrol-2(5H)-one | Penicillium brocae | Avicennia marina | China | S. aureus, Micrococcus luteus | [80,81] | |
| Socromen-1-ona and 3, Ácido 4-dihidroxibenzóico | Phyllosticta capitalensis | Bruguiera sexangula | Southern China | P. aeruginosa, S. aureus, B. subtilis and E. coli | [92] | |
Sesquiterpene | Infectopyrones A and B | Stemphylium sp. | Bruguiera sexangula var. rhynchopetala | South China | B. subtilis Micrococcus tetragenus, Micrococcus luteus and S. albus | [101] |
p-quinone macrolactam | Cytosporone E | Acremonium Strictum | Rhizophora apiculata | Island of CatBa, Vietnam | S. aureus | [32] |
| Ent-cladospolide F | Cladosporium cladosporioides | Bruguiera gymnorrhiz | Hainan Island, China | S. aureus | [118] | |
| (2S)-2,3-dihydro-5,6-dihydroxy-2-methyl-4H-1-benzopyran-4-one and 4-ethyl-3-hydroxy-6-propenyl-2H-pyran-2-one | Colletotrichum gloeosporioides | Ceriops tagal | Hainan Province, China | Micrococcus tetragenus, S. aureus, Streptomyces albus, B. cereus and B. subtilis | [60] | |
| Cytospomarin | Cytospora sp. | Ceriops tagal | Hainan Island, China | E. coli and M. oryzae | [61] | |
| 8-O-methylnodulisporin F and nodulisporin H | Daldinia eschscholtzii | Brguiera sexangula var. rhynchopetala | South China | Staphylococcus aureus, methicillin-resistant S. aureus (MRSA) and Bacillus cereus | [62] | |
| Antiviral compounds | ||||||
Isoindolone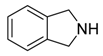 | Emerimidine A and B | Emericella sp. | Aegiceras corniculatum | HaiKou, China | H1N1 | [65] |
Alkaloid | Neosartoryadins A and B | Neosartorya hiratsukae | Aricennia marina | China | H1N1 | [78] |
p-quinone macrolactam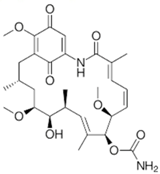 | Diaporthe sp. | Rhizophora stylosa | Hainan Province, China | H1N1 and H3N2 | [63] | |
| pestalotiopsone F, pestalotiopsone B,3,8-dihydroxy-6-methyl-9- oxo-9H-xanthene-1-carboxylate, and 5-chloroisorotiorin | Pestalotiopsis vacinii | Kandelia candel | Anti-enterovirus 71 (EV71) | [116] | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cadamuro, R.D.; da Silveira Bastos, I.M.A.; Silva, I.T.; da Cruz, A.C.C.; Robl, D.; Sandjo, L.P.; Alves, S., Jr.; Lorenzo, J.M.; Rodríguez-Lázaro, D.; Treichel, H.; et al. Bioactive Compounds from Mangrove Endophytic Fungus and Their Uses for Microorganism Control. J. Fungi 2021, 7, 455. https://doi.org/10.3390/jof7060455
Cadamuro RD, da Silveira Bastos IMA, Silva IT, da Cruz ACC, Robl D, Sandjo LP, Alves S Jr., Lorenzo JM, Rodríguez-Lázaro D, Treichel H, et al. Bioactive Compounds from Mangrove Endophytic Fungus and Their Uses for Microorganism Control. Journal of Fungi. 2021; 7(6):455. https://doi.org/10.3390/jof7060455
Chicago/Turabian StyleCadamuro, Rafael Dorighello, Isabela Maria Agustini da Silveira Bastos, Izabella Thais Silva, Ariadne Cristiane Cabral da Cruz, Diogo Robl, Louis Pergaud Sandjo, Sergio Alves, Jr., Jose M. Lorenzo, David Rodríguez-Lázaro, Helen Treichel, and et al. 2021. "Bioactive Compounds from Mangrove Endophytic Fungus and Their Uses for Microorganism Control" Journal of Fungi 7, no. 6: 455. https://doi.org/10.3390/jof7060455
APA StyleCadamuro, R. D., da Silveira Bastos, I. M. A., Silva, I. T., da Cruz, A. C. C., Robl, D., Sandjo, L. P., Alves, S., Jr., Lorenzo, J. M., Rodríguez-Lázaro, D., Treichel, H., Steindel, M., & Fongaro, G. (2021). Bioactive Compounds from Mangrove Endophytic Fungus and Their Uses for Microorganism Control. Journal of Fungi, 7(6), 455. https://doi.org/10.3390/jof7060455














