Reinforcing the Immunocompromised Host Defense against Fungi: Progress beyond the Current State of the Art
Abstract
1. Introduction
2. Treatment of Invasive Fungal Infections: The Unmet Clinical Need
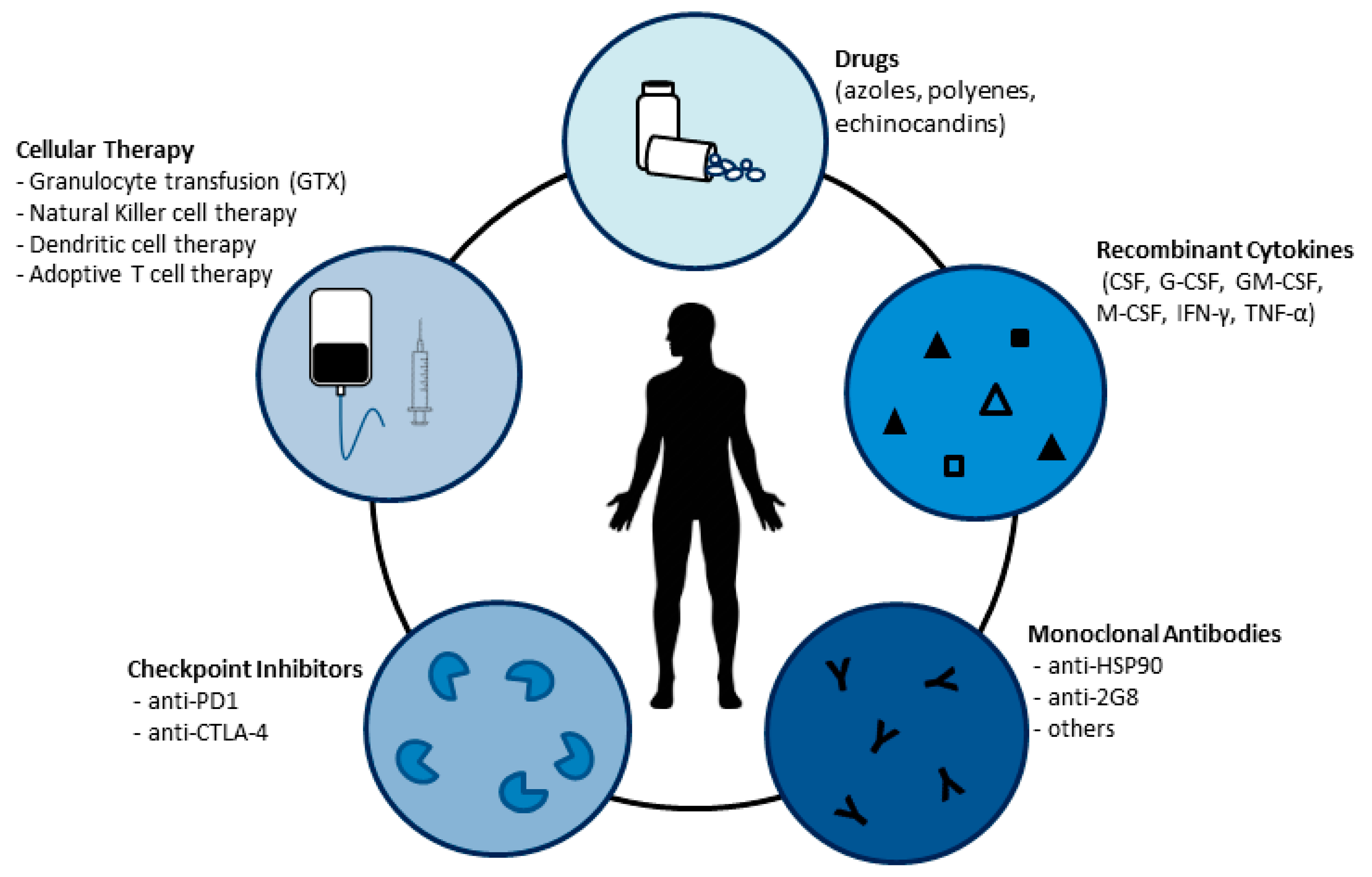
3. Strategies to Reinforce the Host Defense against Fungi
3.1. Recombinant Cytokines
3.1.1. Colony-Stimulating Factors (CSFs)
3.1.2. Interferon-Gamma (IFN-γ)
3.2. Antibodies
3.3. Checkpoint Inhibitors
3.4. Transfer of Innate Immune Cells to Control Fungal Infections
3.4.1. Granulocyte Transfusion (GTX)
3.4.2. Natural Killer and Dendritic Cell Therapy
4. Harnessing T Cells to Control Fungal Infections
4.1. Adoptive Immunotherapy with FSTs: The Pros
4.1.1. High Specificity and Potential Efficacy
4.1.2. Broad Protection against Multiple Pathogens
4.1.3. Long-Lasting Immunity
4.1.4. Off-the-Shelf Therapy
4.1.5. Triggering of Epitope Spreading
4.2. Adoptive Immunotherapy with FSTs: The Cons
4.2.1. Selection of the Antigenic Target
4.2.2. Antigenic Competition
4.2.3. FSTs Outside the Allogeneic Transplantation Context?
4.2.4. “Off-Target” or/and “On-Target Toxicity”
4.2.5. Sustainability of the Clinical Benefit?
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fausto, A.; Rodrigues, M.L.; Coelho, C. The still underestimated problem of fungal diseases worldwide. Front. Microbiol. 2019, 10, 214. [Google Scholar]
- Denning, D.W.; Bromley, M.J. How to bolster the antifungal pipeline: Few drugs are coming to market, but opportunities for drug development exist. Science 2015, 347, 1414–1416. [Google Scholar] [CrossRef] [PubMed]
- Maertens, J.; Vrebos, M.; Boogaerts, M. Assessing risk factors for systemic fungal infections. Eur. J. Cancer Care 2001, 10, 56–62. [Google Scholar] [CrossRef]
- Schmiedel, Y.; Zimmerli, S. Common invasive fungal diseases: An overview of invasive candidiasis, aspergillosis, cryptococcosis, and Pneumocystis pneumonia. Swiss Med. Wkly. 2016, 146, w14281. [Google Scholar] [CrossRef] [PubMed]
- Lamoth, F.; Kontoyiannis, D.P. The Candida auris alert: Facts and perspectives. J. Infect. Dis. 2018, 217, 516–520. [Google Scholar] [CrossRef] [PubMed]
- Bendall, L.J.; Bradstock, K.F. G-CSF: From granulopoietic stimulant to bone marrow stem cell mobilizing agent. Cytokine Growth Factor Rev. 2014, 25, 355–367. [Google Scholar] [CrossRef]
- Pursell, K.; Verral, S.; Daraiesh, F.; Shrestha, N.; Skariah, A.; Hasan, E.; Pitrak, D. Impaired phagocyte respiratory burst responses to opportunistic fungal pathogens in transplant recipients: In vitro effect of r-metHuG-CSF (Filgrastim). Transpl. Infect. Dis. 2003, 5, 29–37. [Google Scholar] [CrossRef]
- Liles, W.C.; Huang, J.E.; Van Burik, J.A.H.; Bowden, R.A.; Dale, D.C. Granulocyte colony-stimulating factor administered in vivo augments neutrophil-mediated activity against opportunistic fungal pathogens. J. Infect. Dis. 1997, 175, 1012–1015. [Google Scholar] [CrossRef]
- Roilides, E.; Farmaki, E. Granulocyte colony-stimulating factor and other cytokines in antifungal therapy. Clin. Microbiol. Infect. 2001, 7, 62–67. [Google Scholar] [CrossRef]
- Hazel, D.L.; Newland, A.C.; Kelsey, S.M. Granulocyte colony stimulating factor increases the efficacy of conventional amphotericin in the treatment of presumed deep-seated fungal infection in neutropenic patients following intensive chemotherapy or bone marrow transplantation for haematological malignancies. Hematology 1999, 4, 305–311. [Google Scholar] [CrossRef]
- Farkas, F.; Mistrik, M.; Batorova, A. The use of granulocyte colony stimulating factor after autologous hematopoietic stem cell transplantation. Bratisl. Lek. Listy 2019, 120. [Google Scholar] [CrossRef]
- Celmeli, F.; Oztoprak, N.; Turkkahraman, D.; Seyman, D.; Mutlu, E.; Frede, N.; Köksoy, S.; Grimbacher, B. Successful granulocyte colony-stimulating factor treatment of relapsing Candida Albicans meningoencephalitis caused by CARD9 deficiency. Pediatr. Infect. Dis. J. 2016, 35, 428–431. [Google Scholar] [CrossRef]
- Du, B.; Shen, N.; Hu, J.; Tao, Y.; Mo, X.; Cao, Q. Complete clinical remission of invasive Candida infection with CARD9 deficiency after G-CSF treatment. Comp. Immunol. Microbiol. Infect. Dis. 2020, 70. [Google Scholar] [CrossRef] [PubMed]
- Mhaskar, R.; Clark, O.A.C.; Lyman, G.; Engel Ayer Botrel, T.; Morganti Paladini, L.; Djulbegovic, B. Colony-stimulating factors for chemotherapy-induced febrile neutropenia. Cochrane Database Syst. Rev. 2014, 2014. [Google Scholar] [CrossRef]
- Wan, L.; Zhang, Y.; Lai, Y.; Jiang, M.; Song, Y.; Zhou, J.; Zhang, Z.; Duan, X.; Fu, Y.; Liao, L.; et al. Effect of granulocyte-macrophage colony-stimulating factor on prevention and treatment of invasIVe fungal disease in recipients of allogeneic stem-cell transplantation: A prospectIVe multicenter randomized phase IV trial. J. Clin. Oncol. 2015, 33, 3999–4006. [Google Scholar] [CrossRef] [PubMed]
- Schroder, K.; Hertzog, P.J.; Ravasi, T.; Hume, D.A. Interferon-γ: An overview of signals, mechanisms and functions. J. Leukoc. Biol. 2004, 75, 163–189. [Google Scholar] [CrossRef]
- Scriven, J.E.; Tenforde, M.W.; Levitz, S.M.; Jarvis, J.N. Modulating host immune responses to fight invasive fungal infections. Curr. Opin. Microbiol. 2017, 40, 95–103. [Google Scholar] [CrossRef]
- Delsing, C.E.; Gresnigt, M.S.; Leentjens, J.; Preijers, F.; Frager, F.A.; Kox, M.; Monneret, G.; Venet, F.; Bleeker-Rovers, C.P.; van de Veerdonk, F.L.; et al. Interferon-gamma as adjunctive immunotherapy for invasive fungal infections: A case series. BMC Infect. Dis. 2014, 14, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, J.N.; Meintjes, G.; Rebe, K.; Williams, G.N.; Bicanic, T.; Williams, A.; Schutz, C.; Bekker, L.G.; Wood, R.; Harrison, T.S. Adjunctive interferon-γ immunotherapy for the treatment of HIV-associated cryptococcal meningitis: A randomized controlled trial. AIDS 2012, 26, 1105–1113. [Google Scholar] [CrossRef] [PubMed]
- Danion, F.; Aimanianda, V.; Bayry, J.; Duréault, A.; Wong, S.S.W.; Bougnoux, M.E.; Tcherakian, C.; Alyanakian, M.A.; Guegan, H.; Puel, A.; et al. Aspergillus fumigatus Infection in Humans With STAT3-Deficiency Is Associated With Defective Interferon-Gamma and Th17 Responses. Front. Immunol. 2020, 11. [Google Scholar] [CrossRef]
- Garraffo, A.; Pilmis, B.; Toubiana, J.; Puel, A.; Mahlaoui, N.; Blanche, S.; Lortholary, O.; Lanternier, F. Invasive Fungal Infection in Primary Immunodeficiencies Other Than Chronic Granulomatous Disease. Curr. Fungal Infect. Rep. 2017, 11, 25–34. [Google Scholar] [CrossRef]
- Pappas, P.G.; Bustamante, B.; Ticona, E.; Hamill, R.J.; Johnson, P.C.; Reboli, A.; Aberg, J.; Hasbun, R.; Hsu, H.H. Recombinant interferon-γ1b as adjunctive therapy for AIDS-related acute cryptococcal meningitis. J. Infect. Dis. 2004, 189, 2185–2191. [Google Scholar] [CrossRef] [PubMed]
- Pachl, J.; Svoboda, P.; Dé Rique Jacobs, F.; Vandewoude, K.; Van Der Hoven, B.; Spronk, P.; Masterson, G.; Malbrain, M.; Aoun, M.; Garbino, J.; et al. A Randomized, Blinded, Multicenter Trial of Lipid-Associated Amphotericin B Alone versus in Combination with an Antibody-Based Inhibitor of Heat Shock Protein 90 in Patients with Invasive Candidiasis. Clin. Infect. Dis. 2006, 42, 1404–1413. [Google Scholar] [CrossRef]
- Mycograb|European Medicines Agency. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/mycograb (accessed on 25 May 2021).
- Rachini, A.; Pietrella, D.; Lupo, P.; Torosantucci, A.; Chiani, P.; Bromuro, C.; Proietti, C.; Bistoni, F.; Cassone, A.; Vecchiarelli, A. An anti-β-glucan monoclonal antibody inhibits growth and capsule formation of Cryptococcus neoformans in vitro and exerts therapeutic, anticryptococcal activity in vivo. Infect. Immun. 2007, 75, 5085–5094. [Google Scholar] [CrossRef] [PubMed]
- Torosantucci, A.; Chiani, P.; Bromuro, C.; De Bernardis, F.; Palma, A.S.; Liu, Y.; Minogna, G.; Maras, B.; Colone, M.; Stringaro, A.; et al. Protection by anti-β-glucan antibodies is associated with restricted β-1,3 glucan binding specificity and inhibition of fungal growth and adherence. PLoS ONE 2009, 4, e5392. [Google Scholar] [CrossRef]
- Larsen, R.A.; Pappas, P.G.; Perfect, J.; Aberg, J.A.; Casadevall, A.; Cloud, G.A.; James, R.; Filler, S.; Dismukes, W.E. Phase I evaluation of the safety and pharmacokinetics of murine-derived anticryptococcal antibody 18B7 in subjects with treated cryptococcal meningitis. Antimicrob. Agents Chemother. 2005, 49, 952–958. [Google Scholar] [CrossRef] [PubMed]
- Ostrosky-Zeichner, L.; Casadevall, A.; Galgiani, J.N.; Odds, F.C.; Rex, J.H. An insight into the antifungal pipeline: Selected new molecules and beyond. Nat. Rev. Drug Discov. 2010, 9, 719–727. [Google Scholar] [CrossRef]
- Nosanchuk, J.D.; Dadachova, E. Radioimmunotherapy of fungal diseases: The therapeutic potential of cytocidal radiation delivered by antibody targeting fungal cell surface antigens. Front. Microbiol. 2012, 2, 283. [Google Scholar] [CrossRef]
- Loreto, É.S.; Tondolo, J.S.M.; Alves, S.H.; Santurio, J.M. Immunotherapy for Fungal Infections. In Immunotherapy—Myths, Reality, Ideas, Future; InTech: Londou, UK, 2017. [Google Scholar]
- Mellinghoff, S.C.; Von Bergwelt-Baildon, M.; Schößer, H.A.; Schößer, S.; Cornely, O.A. A novel approach to candidemia? The potential role of checkpoint inhibition. Med. Mycol. 2019, 57, 151–154. [Google Scholar] [CrossRef]
- Chang, K.C.; Burnham, C.-A.; Compton, S.M.; Rasche, D.P.; Mazuski, R.; SMcDonough, J.; Unsinger, J.; Korman, A.J.; Green, J.M.; Hotchkiss, R.S. Blockade ofthe negative co-stimulatory molecules PD-1 and CTLA-4 improves survival in primary and secondary fungal sepsis. Crit. Care 2013, 17, R85. [Google Scholar] [CrossRef]
- Grimaldi, D.; Pradier, O.; Hotchkiss, R.S.; Vincent, J.L. Nivolumab plus interferon-γ in the treatment of intractable mucormycosis. Lancet Infect. Dis. 2017, 17, 18. [Google Scholar] [CrossRef]
- West, K.A.; Gea-Banacloche, J.; Stroncek, D.; Kadri, S.S. Granulocyte transfusions in the management of invasive fungal infections. Br. J. Haematol. 2017, 177, 357–374. [Google Scholar] [CrossRef]
- Schmidt, S.; Tramsen, L.; Hanisch, M.; Latgé, J.P.; Huenecke, S.; Koehl, U.; Lehrnbecher, T. Human natural killer cells exhibit direct activity against Aspergillus fumigatus hyphae, but not against resting conidia. J. Infect. Dis. 2011, 203, 430–435. [Google Scholar] [CrossRef]
- Schmidt, S.; Luckowitsch, M.; Hogardt, M.; Lehrnbecher, T. Natural killer cell line nk-92-mediated damage of medically important fungi. J. Fungi 2021, 7, 144. [Google Scholar] [CrossRef]
- Soe, W.M.; Lim, J.H.J.; Williams, D.L.; Goh, J.G.; Tan, Z.; Sam, Q.H.; Chotirmall, S.H.; Ali, N.A.B.M.; Lee, S.C.; Seet, J.E.; et al. Using expanded natural killer cells as therapy for invasive aspergillosis. J. Fungi 2020, 6, 231. [Google Scholar] [CrossRef]
- Bozza, S.; Perruccio, K.; Montagnoli, C.; Gaziano, R.; Bellocchio, S.; Burchielli, E.; Nkwanyuo, G.; Pitzurra, L.; Velardi, A.; Romani, L. A dendritic cell vaccine against invasive aspergillosis in allogeneic hematopoietic transplantation. Blood 2003, 102, 3807–3814. [Google Scholar] [CrossRef]
- Ogonek, J.; Kralj Juric, M.; Ghimire, S.; Varanasi, P.R.; Holler, E.; Greinix, H.; Weissinger, E. Immune Reconstitution after Allogeneic Hematopoietic Stem Cell Transplantation. Front. Immunol. 2016, 7, 507. [Google Scholar] [CrossRef]
- Stuehler, C.; Kuenzli, E.; Jaeger, V.K.; Baettig, V.; Ferracin, F.; Rajacic, Z.; Kaiser, D.; Bernardini, C.; Forrer, P.; Weisser, M.; et al. Immune Reconstitution After Allogeneic Hematopoietic Stem Cell Transplantation and Association with Occurrence and Outcome of Invasive Aspergillosis. J. Infect. Dis. 2015, 212, 959–967. [Google Scholar] [CrossRef] [PubMed]
- Perruccio, K.; Tosti, A.; Burchielli, E.; Topini, F.; Ruggeri, L.; Carotti, A.; Capanni, M.; Urbani, E.; Mancusi, A.; Aversa, F.; et al. Transferring functional immune responses to pathogens after haploidentical hematopoietic transplantation. Blood 2005, 106, 4397–4406. [Google Scholar] [CrossRef] [PubMed]
- Balduzzi, A.; Lucchini, G.; Hirsch, H.H.; Basso, S.; Cioni, M.; Rovelli, A.; Zincone, A.; Grimaldi, M.; Corti, P.; Bonanomi, S.; et al. Polyomavirus JC-targeted T-cell therapy for progressive multiple leukoencephalopathy in a hematopoietic cell transplantation recipient. Bone Marrow Transplant. 2011, 46, 987–992. [Google Scholar] [CrossRef] [PubMed]
- Blyth, E.; Clancy, L.; Simms, R.; Ma, C.K.K.; Burgess, J.; Deo, S.; Byth, K.; Dubosq, M.-C.; Shaw, P.J.; Micklethwaite, K.P.; et al. Donor-derived CMV-specific T cells reduce the requirement for CMV-directed pharmacotherapy after allogeneic stem cell transplantation. Blood 2013, 121, 3745–3758. [Google Scholar] [CrossRef] [PubMed]
- Comoli, P.; Basso, S.; Zecca, M.; Pagliara, D.; Baldanti, F.; Bernardo, M.E.; Barberi, W.; Moretta, A.; Labirio, M.; Paulli, M.; et al. Preemptive Therapy of EBV-Related Lymphoproliferative Disease after Pediatric Haploidentical Stem Cell Transplantation. Am. J. Transplant. 2007, 7, 1648–1655. [Google Scholar] [CrossRef] [PubMed]
- Doubrovina, E.; Oflaz-Sozmen, B.; Prockop, S.E.; Kernan, N.A.; Abramson, S.; Teruya-Feldstein, J.; Hedvat, C.; Chou, J.F.; Heller, G.; Barker, J.N.; et al. Adoptive immunotherapy with unselected or EBV-specific T cells for biopsy-proven EBV+ lymphomas after allogeneic hematopoietic cell transplantation. Blood 2012, 119, 2644–2656. [Google Scholar] [CrossRef]
- Einsele, H.; Roosnek, E.; Rufer, N.; Sinzger, C.; Riegler, S.; Löffler, J.; Grigoleit, U.; Moris, A.; Rammensee, H.-G.; Kanz, L.; et al. Infusion of cytomegalovirus (CMV)-specific T cells for the treatment of CMV infection not responding to antiviral chemotherapy. Blood 2002, 99, 3916–3922. [Google Scholar] [CrossRef] [PubMed]
- Feuchtinger, T.; Matthes-Martin, S.; Richard, C.; Lion, T.; Fuhrer, M.; Hamprecht, K.; Handgretinger, R.; Peters, C.; Schuster, F.R.; Beck, R.; et al. Safe adoptive transfer of virus-specific T-cell immunity for the treatment of systemic adenovirus infection after allogeneic stem cell transplantation. Br. J. Haematol. 2006, 134, 64–76. [Google Scholar] [CrossRef]
- Gerdemann, U.; Katari, U.L.; Papadopoulou, A.; Keirnan, J.M.; Craddock, J.A.; Liu, H.; Martinez, C.A.; Kennedy-Nasser, A.; Leung, K.S.; Gottschalk, S.M.; et al. Safety and clinical efficacy of rapidly-generated trivirus-directed T cells as treatment for adenovirus, EBV, and CMV infections after allogeneic hematopoietic stem cell transplant. Mol. Ther. 2013, 21, 2113–2121. [Google Scholar] [CrossRef] [PubMed]
- Heslop, H.E.; Slobod, K.S.; Pule, M.A.; Hale, G.A.; Rousseau, A.; Smith, C.A.; Bollard, C.M.; Liu, H.; Wu, M.-F.; Rochester, R.J.; et al. Long-term outcome of EBV-specific T-cell infusions to prevent or treat EBV-related lymphoproliferative disease in transplant recipients. Blood 2010, 115, 925–935. [Google Scholar] [CrossRef]
- Haynes, M.P.; Chong, P.L.-G.; Buckley, H.R.; Pieringer, R.A. Fluorescence Studies on the Molecular Action of Amphotericin B on Susceptible and Resistant Fungal Cells †. Biochemistry 1996, 35, 7983–7992. [Google Scholar] [CrossRef]
- Leen, A.M.; Myers, G.D.; Sili, U.; Huls, M.H.; Weiss, H.; Leung, K.S.; Carrum, G.; Krance, R.A.; Chang, C.-C.; Molldrem, J.J.; et al. Monoculture-derived T lymphocytes specific for multiple viruses expand and produce clinically relevant effects in immunocompromised individuals. Nat. Med. 2006, 12, 1160–1166. [Google Scholar] [CrossRef]
- Moosmann, A.; Bigalke, I.; Tischer, J.; Schirrmann, L.; Kasten, J.; Tippmer, S.; Leeping, M.; Prevalsek, D.; Jaeger, G.; Ledderose, G.; et al. Effective and long-term control of EBV PTLD after transfer of peptide-selected T cells. Blood 2010, 115, 2960–2970. [Google Scholar] [CrossRef]
- Papadopoulou, A.; Gerdemann, U.; Katari, U.L.; Tzannou, I.; Liu, H.; Martinez, C.; Leung, K.; Carrum, G.; Gee, A.P.; Vera, J.F.; et al. Activity of Broad-Spectrum T Cells as Treatment for AdV, EBV, CMV, BKV, and HHV6 Infections after HSCT. Sci. Transl. Med. 2014, 6, 242ra83. [Google Scholar] [CrossRef]
- Rooney, C.M.; Smith, C.A.; Ng, C.Y.; Loftin, S.K.; Sixbey, J.W.; Gan, Y.; Srivastava, D.K.; Bowman, L.C.; Krance, R.A.; Brenner, M.K.; et al. Infusion of cytotoxic T cells for the prevention and treatment of Epstein-Barr virus-induced lymphoma in allogeneic transplant recipients. Blood 1998, 92, 1549–1555. [Google Scholar] [CrossRef] [PubMed]
- Rooney, C.M.; Smith, C.A.; Ng, C.Y.; Loftin, S.; Li, C.; Krance, R.A.; Brenner, M.K.; Heslop, H.E. Use of gene-modified virus-specific T lymphocytes to control Epstein-Barr-virus-related lymphoproliferation. Lancet 1995, 345, 9–13. [Google Scholar] [CrossRef]
- Walter, E.A.; Greenberg, P.D.; Gilbert, M.J.; Finch, R.J.; Watanabe, K.S.; Thomas, E.D.; Riddell, S.R. Reconstitution of cellular immunity against cytomegalovirus in recipients of allogeneic bone marrow by transfer of T-cell clones from the donor. N. Engl. J. Med. 1995, 333, 1038–1044. [Google Scholar] [CrossRef] [PubMed]
- Peggs, K.S.; Verfuerth, S.; Pizzey, A.; Khan, N.; Guiver, M.; Moss, P.A.; Mackinnon, S. Adoptive cellular therapy for early cytomegalovirus infection after allogeneic stem-cell transplantation with virus-specific T-cell lines. Lancet 2003, 362, 1375–1377. [Google Scholar] [CrossRef]
- Gustafsson, A.; Levitsky, V.; Zou, J.Z.; Frisan, T.; Dalianis, T.; Ljungman, P.; Ringden, O.; Winiarski, J.; Ernberg, I.; Masucci, M.G. Epstein-Barr virus (EBV) load in bone marrow transplant recipients at risk to develop posttransplant lymphoproliferative disease: Prophylactic infusion of EBV-specific cytotoxic T cells. Blood 2000, 95, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Castellano-González, G.; McGuire, H.M.; Luciani, F.; Clancy, L.E.; Li, Z.; Avdic, S.; Hughes, B.; Singh, M.; De St Groth, B.F.; Renga, G.; et al. Rapidly expanded partially HLA DRB1-matched fungus-specific T cells mediate in vitro and in vivo antifungal activity. Blood Adv. 2020, 4, 3443–3456. [Google Scholar] [CrossRef] [PubMed]
- Cenci, E.; Mencacci, A.; Bacci, A.; Bistoni, F.; Kurup, V.P.; Romani, L. T Cell Vaccination in Mice with Invasive Pulmonary Aspergillosis. J. Immunol. 2000, 165, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Zhu, P.; Li, L.; Wan, Z.; Zhao, Z.; Li, R. Adoptive immunity mediated by HLA-A*0201 restricted Asp f16 peptides-specific CD8+ T cells against Aspergillus fumigatus infection. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 3089–3096. [Google Scholar] [CrossRef]
- Gottlieb, D.J.; Clancy, L.E.; Withers, B.; McGuire, H.M.; Luciani, F.; Singh, M.; Hughes, B.; Gloss, B.; Kliman, D.; Ma, C.K.K.; et al. Prophylactic antigen-specific T-cells targeting seven viral and fungal pathogens after allogeneic haemopoietic stem cell transplant. Clin. Transl. Immunol. 2021, 10. [Google Scholar] [CrossRef]
- Papadopoulou, A.; Kaloyannidis, P.; Yannaki, E.; Cruz, C.R. Adoptive transfer of Aspergillus-specific T cells as a novel anti-fungal therapy for hematopoietic stem cell transplant recipients: Progress and challenges. Crit. Rev. Oncol. Hematol. 2016, 98, 62–72. [Google Scholar] [CrossRef]
- Deo, S.S.; Gottlieb, D.J. Adoptive T-cell therapy for fungal infections in haematology patients. Clin. Transl. Immunol. 2015, 4, e40. [Google Scholar] [CrossRef]
- Ramadan, G.; Konings, S.; Kurup, V.P.; Keever-Taylor, C.A. Generation of Aspergillus- and CMV- specific T-cell responses using autologous fast DC. Cytotherapy 2004, 6, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Beck, O.; Topp, M.S.; Koehl, U.; Roilides, E.; Simitsopoulou, M.; Hanisch, M.; Sarfati, J.; Latgé, J.P.; Klingebiel, T.; Einsele, H.; et al. Generation of highly purified and functionally active human TH1 cells against Aspergillus fumigatus. Blood 2006, 107, 2562–2569. [Google Scholar] [CrossRef] [PubMed]
- Tramsen, L.; Beck, O.; Schuster, F.R.; Hunfeld, K.P.; Latgé, J.P.; Sarfati, J.; Röger, F.; Klingebiel, T.; Koehl, U.; Lehrnbecher, T. Generation and characterization of anti-Candida T cells as potential immunotherapy in patients with Candida infection after allogeneic hematopoietic stem-cell transplant. J. Infect. Dis. 2007, 196, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.; Ramadan, G.; Davies, B.; Margolis, D.A.; Keever-Taylor, C.A. Stimulation by means of dendritic cells followed by Epstein-Barr virus-transformed B cells as antigen-presenting cells is more efficient than dendritic cells alone in inducing Aspergillus f16-specific cytotoxic T cell responses. Clin. Exp. Immunol. 2008, 151, 284–296. [Google Scholar] [CrossRef] [PubMed]
- Tramsen, L.; Koehl, U.; Tonn, T.; Latgé, J.-P.; Schuster, F.R.; Borkhardt, A.; Uharek, L.; Quaritsch, R.; Beck, O.; Seifried, E.; et al. Clinical-scale generation of human anti-Aspergillus T cells for adoptive immunotherapy. Bone Marrow Transplant. 2009, 43, 13–19. [Google Scholar] [CrossRef]
- Gaundar, S.S.; Clancy, L.; Blyth, E.; Meyer, W.; Gottlieb, D.J. Robust polyfunctional T-helper 1 responses to multiple fungal antigens from a cell population generated using an environmental strain of Aspergillus fumigatus. Cytotherapy 2012, 14, 1119–1130. [Google Scholar] [CrossRef]
- Schmidt, S.; Tramsen, L.; Perkhofer, S.; Lass-Flörl, C.; Röger, F.; Schubert, R.; Lehrnbecher, T. Characterization of the cellular immune responses to rhizopus oryzae with potential impact on immunotherapeutic strategies in hematopoietic stem cell transplantation. J. Infect. Dis. 2012, 206, 135–139. [Google Scholar] [CrossRef]
- Jolink, H.; Meijssen, I.C.; Hagedoorn, R.S.; Arentshorst, M.; Drijfhout, J.W.; Mulder, A.; Claas, F.H.J.; van Dissel, J.T.; Falkenburg, J.H.F.; Heemskerk, M.H.M. Characterization of the T-cell-mediated immune response against the Aspergillus fumigatus proteins Crf1 and catalase 1 in healthy individuals. J. Infect. Dis. 2013, 208, 847–856. [Google Scholar] [CrossRef]
- Kumaresan, P.R.; Manuri, P.R.; Albert, N.D.; Maiti, S.; Singh, H.; Mi, T.; Roszik, J.; Rabinovich, B.; Olivares, S.; Krishnamurthy, J.; et al. Bioengineering T cells to target carbohydrate to treat opportunistic fungal infection. Proc. Natl. Acad. Sci. USA 2014, 111, 10660–10665. [Google Scholar] [CrossRef]
- Bacher, P.; Jochheim-Richter, A.; Mockel-Tenbrink, N.; Kniemeyer, O.; Wingenfeld, E.; Alex, R.; Ortigao, A.; Karpova, D.; Lehrnbecher, T.; Ullmann, A.J.; et al. Clinical-scale isolation of the total Aspergillus fumigatus-reactive T-helper cell repertoire for adoptive transfer. Cytotherapy 2015, 17, 1396–1405. [Google Scholar] [CrossRef]
- Stuehler, C.; Nowakowska, J.; Bernardini, C.; Topp, M.S.; Battegay, M.; Passweg, J.; Khanna, N. Multispecific Aspergillus T cells selected by CD137 or CD154 induce protective immune responses against the most relevant mold infections. J. Infect. Dis. 2015, 211, 1251–1261. [Google Scholar] [CrossRef]
- Deo, S.S.; Virassamy, B.; Halliday, C.; Clancy, L.; Chen, S.; Meyer, W.; Sorrell, T.C.; Gottlieb, D.J. Stimulation with lysates of Aspergillus terreus, Candida krusei and Rhizopus oryzae maximizes cross-reactivity of anti-fungal T cells. Cytotherapy 2016, 18, 65–79. [Google Scholar] [CrossRef]
- Castillo, P.; Wright, K.E.; Kontoyiannis, D.P.; Walsh, T.; Patel, S.; Chorvinsky, E.; Bose, S.; Hazrat, Y.; Omer, B.; Albert, N.; et al. A New Method for Reactivating and Expanding T Cells Specific for Rhizopus oryzae. Mol. Ther. Methods Clin. Dev. 2018, 9, 305–312. [Google Scholar] [CrossRef]
- Papadopoulou, A.; Alvanou, M.; Koukoulias, K.; Athanasiou, E.; Lazaridou, A.; Savvopoulos, N.; Kaloyannidis, P.; Markantonatou, A.-M.; Vyzantiadis, T.-A.; Yiangou, M.; et al. Clinical-scale production of Aspergillus-specific T cells for the treatment of invasive aspergillosis in the immunocompromised host. Bone Marrow Transplant. 2019, 54, 1963–1972. [Google Scholar] [CrossRef]
- Tramsen, L.; Schmidt, S.; Boenig, H.; Latgé, J.-P.; Lass-Flörl, C.; Roeger, F.; Seifried, E.; Klingebiel, T.; Lehrnbecher, T. Clinical-scale generation of multi-specific anti-fungal T cells targeting Candida, Aspergillus and mucormycetes. Cytotherapy 2013, 15, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Khanna, N.; Stuehler, C.; Conrad, B.; Lurati, S.; Krappmann, S.; Einsele, H.; Berges, C.; Topp, M.S. Generation of a multipathogen-specific T-cell product for adoptive immunotherapy based on activation-dependent expression of CD154. Blood 2011, 118, 1121–1131. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou, A.; Koukoulias, K.; Alvanou, M.; Kouimtzidis, A.; Athanasiou, E.; Savvopoulos, N.; Markantonatou, A.M.; Siotou, E.; Vyzantiadis, T.A.; Anagnostopoulos, A.; et al. Multipathogen-specific T cells against viral and fungal infections. Bone Marrow Transplant. 2021. [Google Scholar] [CrossRef] [PubMed]
- Koukoulias, K.; Papayanni, P.-G.; Georgakopoulou, A.; Alvanou, M.; Laidou, S.; Kouimtzidis, A.; Pantazi, C.; Gkoliou, G.; Vyzantiadis, T.-A.; Spyridonidis, A.; et al. “Cerberus” T Cells: A Glucocorticoid-Resistant, Multi-Pathogen Specific T Cell Product to Fight Infections in Severely Immunocompromised Patients. Front. Immunol. 2021, 11, 608701. [Google Scholar] [CrossRef]
- Houghtelin, A.; Bollard, C.M. Virus-specific T cells for the immunocompromised patient. Front. Immunol. 2017, 8, 1272. [Google Scholar] [CrossRef]
- Kaeuferle, T.; Krauss, R.; Blaeschke, F.; Willier, S.; Feuchtinger, T. Strategies of adoptive T -cell transfer to treat refractory viral infections post allogeneic stem cell transplantation. J. Hematol. Oncol. 2019, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Roemhild, A.; Reinke, P. Virus-specific T-cell therapy in solid organ transplantation. Transpl. Int. 2016, 29, 515–526. [Google Scholar] [CrossRef] [PubMed]
- Tzannou, I.; Papadopoulou, A.; Naik, S.; Leung, K.; Martinez, C.A.; Ramos, C.A.; Carrum, G.; Sasa, G.; Lulla, P.; Watanabe, A.; et al. Off-the-shelf virus-specific T cells to treat BK virus, human herpesvirus 6, cytomegalovirus, Epstein-Barr virus, and adenovirus infections after allogeneic hematopoietic stem-cell transplantation. J. Clin. Oncol. 2017, 35, 3547. [Google Scholar] [CrossRef] [PubMed]
- Leen, A.M.; Bollard, C.M.; Mendizabal, A.M.; Shpall, E.J.; Szabolcs, P.; Antin, J.H.; Kapoor, N.; Pai, S.-Y.; Rowley, S.D.; Kebriaei, P.; et al. Multicenter study of banked third-party virus-specific T cells to treat severe viral infections after hematopoietic stem cell transplantation. Blood 2013, 121, 5113–5123. [Google Scholar] [CrossRef] [PubMed]
- Uhlin, M.; Gertow, J.; Uzunel, M.; Okas, M.; Berglund, S.; Watz, E.; Brune, M.; Ljungman, P.; Maeurer, M.; Mattsson, J. Rapid Salvage Treatment with Virus-Specific T Cells for Therapy-Resistant Disease. Clin. Infect. Dis. 2012, 55, 1064–1073. [Google Scholar] [CrossRef]
- Haque, T.; Wilkie, G.M.; Jones, M.M.; Higgins, C.D.; Urquhart, G.; Wingate, P.; Burns, D.; McAulay, K.; Turner, M.; Bellamy, C.; et al. Allogeneic cytotoxic T-cell therapy for EBV-positive posttransplantation lymphoproliferative disease: Results of a phase 2 multicenter clinical trial. Blood 2007, 110, 1123–1131. [Google Scholar] [CrossRef]
- Haque, T.; Wilkie, G.M.; Taylor, C.; Amlot, P.L.; Murad, P.; Iley, A.; Dombagoda, D.; Britton, K.M.; Swerdlow, A.J.; Crawford, D.H. Treatment of Epstein-Barr-virus-positive post-transplantation lymphoproliferative disease with partly HLA-matched allogeneic cytotoxic T cells. Lancet 2002, 360, 436–442. [Google Scholar] [CrossRef]
- Prockop, S.; Doubrovina, E.; Suser, S.; Heller, G.; Barker, J.; Dahi, P.; Perales, M.A.; Papadopoulos, E.; Sauter, C.; Castro-Malaspina, H.; et al. Off-the-shelf EBV-specific T cell immunotherapy for rituximab-refractory EBV-associated lymphoma following transplantation. J. Clin. Investig. 2020, 130, 733–747. [Google Scholar] [CrossRef]
- Gallot, G.; Vollant, S.; Saïagh, S.; Clémenceau, B.; Vivien, R.; Cerato, E.; Bignon, J.D.; Ferrand, C.; Jaccard, A.; Vigouroux, S.; et al. T-cell therapy using a bank of EBV-specific Cytotoxic T cells: Lessons from a phase I/II feasibility and safety study. J. Immunother. 2014, 37, 170–179. [Google Scholar] [CrossRef]
- Barker, J.N.; Doubrovina, E.; Sauter, C.; Jaroscak, J.J.; Perales, M.A.; Doubrovin, M.; Prockop, S.E.; Koehne, G.; O’Reilly, R.J. Successful treatment of EBV-associated posttransplantation lymphoma after cord blood transplantation using third-party EBV-specific cytotoxic T lymphocytes. Blood 2010, 116, 5045–5049. [Google Scholar] [CrossRef]
- Romani, L. Immunity to fungal infections. Nat. Rev. Immunol. 2011, 11, 275–288. [Google Scholar] [CrossRef]
- Guglani, L.; Khader, S.A. Th17 cytokines in mucosal immunity and inflammation. Curr. Opin. HIV AIDS 2010, 5, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Conti, H.R.; Gaffen, S.L. IL-17–Mediated Immunity to the Opportunistic Fungal Pathogen Candida albicans. J. Immunol. 2015, 195, 780–788. [Google Scholar] [CrossRef]
- Hernández-Santos, N.; Gaffen, S.L. Th17 cells in immunity to Candida albicans. Cell Host Microbe 2012, 11, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Tomblyn, M.; Chiller, T.; Einsele, H.; Gress, R.; Sepkowitz, K.; Storek, J.; Wingard, J.R.; Young, J.-A.H.; Boeckh, M.J.; Boeckh, M.A. Guidelines for preventing infectious complications among hematopoietic cell transplantation recipients: A global perspective. Biol. Blood Marrow Transplant. 2009, 15, 1143–1238. [Google Scholar] [CrossRef]
- Ma, C.K.K.; Blyth, E.; Clancy, L.; Simms, R.; Burgess, J.; Brown, R.; Deo, S.; Micklethwaite, K.P.; Gottlieb, D.J. Addition of varicella zoster virus-specific T cells to cytomegalovirus, Epstein-Barr virus and adenovirus tri-specific T cells as adoptive immunotherapy in patients undergoing allogeneic hematopoietic stem cell transplantation. Cytotherapy 2015, 17, 1406–1420. [Google Scholar] [CrossRef] [PubMed]
- Gerdemann, U.; Keirnan, J.M.; Katari, U.L.; Yanagisawa, R.; Christin, A.S.; Huye, L.E.; Perna, S.K.; Ennamuri, S.; Gottschalk, S.; Brenner, M.K.; et al. Rapidly generated multivirus-specific cytotoxic T lymphocytes for the prophylaxis and treatment of viral infections. Mol. Ther. 2012, 20, 1622–1632. [Google Scholar] [CrossRef] [PubMed]
- Vasileiou, S.; Turney, A.M.; Kuvalekar, M.; Mukhi, S.S.; Watanabe, A.; Lulla, P.; Ramos, C.A.; Naik, S.; Vera, J.F.; Tzannou, I.; et al. Rapid generation of multivirus-specific T lymphocytes for the prevention and treatment of respiratory viral infections. Haematologica 2020, 105, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Stuehler, C.; Khanna, N.; Bozza, S.; Zelante, T.; Moretti, S.; Kruhm, M.; Lurati, S.; Conrad, B.; Worschech, E.; Stevanović, S.; et al. Cross-protective TH1 immunity against Aspergillus fumigatus and Candida albicans. Blood 2011, 117, 5881–5891. [Google Scholar] [CrossRef]
- Bacher, P.; Hohnstein, T.; Beerbaum, E.; Röcker, M.; Blango, M.G.; Kaufmann, S.; Röhmel, J.; Eschenhagen, P.; Grehn, C.; Seidel, K.; et al. Human Anti-fungal Th17 Immunity and Pathology Rely on Cross-Reactivity against Candida albicans. Cell 2019, 176, 1340–1355. [Google Scholar] [CrossRef] [PubMed]
- Low, C.Y.; Rotstein, C. Emerging fungal infections in immunocompromised patients. F1000 Med. Rep. 2011, 3, 14. [Google Scholar] [CrossRef]
- Kontoyiannis, D.P.; Marr, K.A.; Park, B.J.; Alexander, B.D.; Anaissie, E.J.; Walsh, T.J.; Ito, J.; Andes, D.R.; Baddley, J.W.; Brown, J.M.; et al. Prospective surveillance for invasive fungal infections in hematopoietic stem cell transplant recipients, 2001–2006: Overview of the Transplant-Associated Infection Surveillance Network (TRANSNET) Database. Clin. Infect. Dis. 2010, 50, 1091–1100. [Google Scholar] [CrossRef]
- Pappas, P.G.; Alexander, B.D.; Andes, D.R.; Hadley, S.; Kauffman, C.A.; Freifeld, A.; Anaissie, E.J.; Brumble, L.M.; Herwaldt, L.; Lto, J.; et al. Invasive fungal infections among organ transplant recipients: Results of the transplant-associated infection surveillance network (Transnet). Clin. Infect. Dis. 2010, 50, 1101–1111. [Google Scholar] [CrossRef]
- Seder, R.A.; Ahmed, R. Similarities and differences in CD4+ and CD8+ effector and memory T cell generation. Nat. Immunol. 2003, 4, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Riddell, S.R.; Watanabe, K.S.; Goodrich, J.M.; Li, C.R.; Agha, M.E.; Greenberg, P.D. Restoration of viral immunity in immunodeficient humans by the adoptive transfer of T cell clones. Science 1992, 257, 238–241. [Google Scholar] [CrossRef] [PubMed]
- Abraham, A.A.; John, T.D.; Keller, M.D.; Cruz, C.R.N.; Salem, B.; Roesch, L.; Liu, H.; Hoq, F.; Grilley, B.J.; Gee, A.P.; et al. Safety and feasibility of virus-specific T cells derived from umbilical cord blood in cord blood transplant recipients. Blood Adv. 2019, 3, 2057–2068. [Google Scholar] [CrossRef] [PubMed]
- Haque, T.; McAulay, K.A.; Kelly, D.; Crawford, D.H. Allogeneic t-cell therapy for epstein-barr virus-positive posttransplant lymphoproliferative disease: Long-term follow-up. Transplantation 2010, 90, 93–94. [Google Scholar] [CrossRef]
- Withers, B.; Blyth, E.; Clancy, L.E.; Yong, A.; Fraser, C.; Burgess, J.; Simms, R.; Brown, R.; Kliman, D.; Dubosq, M.C.; et al. Long-term control of recurrent or refractory viral infections after allogeneic HSCT with third-party virus-specific T cells. Blood Adv. 2017, 1, 2193–2205. [Google Scholar] [CrossRef]
- Koenig, S.; Conley, A.J.; Brewah, Y.A.; Jones, G.M.; Leath, S.; Boots, L.J.; Davey, V.; Pantaleo, G.; Demarest, J.F.; Carter, C.; et al. Transfer of HIV-1-specific cytotoxic T lymphocytes to an AIDS patient leads to selection for mutant HIV variants and subsequent disease progression. Nat. Med. 1995, 1, 330–336. [Google Scholar] [CrossRef]
- Gottschalk, S.; Ng, C.Y.C.; Perez, M.; Smith, C.A.; Sample, C.; Brenner, M.K.; Heslop, H.E.; Rooney, C.M. An Epstein-Barr virus deletion mutant associated with fatal lymphoproliferative disease unresponsive to therapy with vires-specific CTLs. Blood 2001, 97, 835–843. [Google Scholar] [CrossRef]
- Vanderlugt, C.L.; Miller, S.D. Epitope spreading in immune-mediated diseases: Implications for immunotherapy. Nat. Rev. Immunol. 2002, 2, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.; Lineburg, K.E.; Martins, J.P.; Ambalathingal, G.R.; Neller, M.A.; Morrison, B.; Matthews, K.K.; Rehan, S.; Crooks, P.; Panikkar, A.; et al. Autologous CMV-specific T cells are a safe adjuvant immunotherapy for primary glioblastoma multiforme. J. Clin. Investig. 2020, 130, 6041–6053. [Google Scholar] [CrossRef]
- Bollard, C.M.; Gottschalk, S.; Torrano, V.; Diouf, O.; Ku, S.; Hazrat, Y.; Carrum, G.; Ramos, C.; Fayad, L.; Shpall, E.J.; et al. Sustained complete responses in patients with lymphoma receiving autologous cytotoxic T lymphocytes targeting Epstein-Barr virus latent membrane proteins. J. Clin. Oncol. 2014, 32, 798–808. [Google Scholar] [CrossRef] [PubMed]
- Hunder, N.N.; Wallen, H.; Cao, J.; Hendricks, D.W.; Reilly, J.Z.; Rodmyre, R.; Jungbluth, A.; Gnjatic, S.; Thompson, J.A.; Yee, C. Treatment of Metastatic Melanoma with Autologous CD4+ T Cells against NY-ESO-1. N. Engl. J. Med. 2008, 358, 2698–2703. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Chávez, M.J.; Pérez-García, L.A.; Niño-Vega, G.A.; Mora-Montes, H.M. Fungal strategies to evade the host immune recognition. J. Fungi 2017, 3, 51. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, A.-R.; Shin, E.-C. Cytomegalovirus Infection and Memory T Cell Inflation. Immune Netw. 2015, 15, 186. [Google Scholar] [CrossRef] [PubMed]
- Kedl, R.M.; Rees, W.A.; Hildeman, D.A.; Schaefer, B.; Mitchell, T.; Kappler, J.; Marrack, P. T cells compete for access to antigen-bearing antigen-presenting cells. J. Exp. Med. 2000, 192, 1105–1113. [Google Scholar] [CrossRef] [PubMed]
- Kedl, R.M.; Schaefer, B.C.; Kappler, J.W.; Marrack, P. T cells down-modulate peptide-MHC complexes on APCs in vivo. Nat. Immunol. 2002, 3, 27–32. [Google Scholar] [CrossRef]
- Kazi, S.; Mathur, A.; Wilkie, G.; Cheal, K.; Battle, R.; McGowan, N.; Fraser, N.; Hargreaves, E.; Turner, D.; Campbell, J.D.M.; et al. Long-term follow up after third-party viral-specific cytotoxic lymphocytes for immunosuppression-and epstein-barr virus-associated lymphoproliferative disease. Haematologica 2019, 104, e356–e359. [Google Scholar] [CrossRef]
- Muftuoglu, M.; Olson, A.; Marin, D.; Ahmed, S.; Mulanovich, V.; Tummala, S.; Chi, T.L.; Ferrajoli, A.; Kaur, I.; Li, L.; et al. Allogeneic BK Virus–Specific T Cells for Progressive Multifocal Leukoencephalopathy. N. Engl. J. Med. 2018, 379, 1443–1451. [Google Scholar] [CrossRef]
- Haque, T.; Amlot, P.L.; Helling, N.; Thomas, J.A.; Sweny, P.; Rolles, K.; Burroughs, A.K.; Prentice, H.G.; Crawford, D.H. Reconstitution of EBV-specific T cell immunity in solid organ transplant recipients. J. Immunol. 1998, 160, 6204–6209. [Google Scholar]
- Smith, C.; Beagley, L.; Rehan, S.; Neller, M.A.; Crooks, P.; Solomon, M.; Holmes-Liew, C.-L.; Holmes, M.; McKenzie, S.C.; Hopkins, P.; et al. Autologous Adoptive T-cell Therapy for Recurrent or Drug-resistant Cytomegalovirus Complications in Solid Organ Transplant Recipients: A Single-arm Open-label Phase I Clinical Trial. Clin. Infect. Dis. 2018, 68, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Spec, A.; Shindo, Y.; Burnham, C.-A.D.; Wilson, S.; Ablordeppey, E.A.; Beiter, E.R.; Chang, K.; Drewry, A.M.; Hotchkiss, R.S. T cells from patients with Candida sepsis display a suppressive immunophenotype. Crit. Care 2015, 20, 15. [Google Scholar] [CrossRef] [PubMed]
- Campanelli, A.P.; Martins, G.A.; Souto, J.T.; Pereira, M.S.F.; Livonesi, M.C.; Martinez, R.; Silva, J.S. Fas-Fas Ligand (CD95-CD95L) and Cytotoxic T Lymphocyte Antigen–4 Engagement Mediate T Cell Unresponsiveness in Patients with Paracoccidioidomycosis. J. Infect. Dis. 2003, 187, 1496–1505. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Withers, B.; Sutrave, G.; Clancy, L.E.; Yong, M.I.; Blyth, E. Pathogen-Specific T Cells Beyond CMV, EBV and Adenovirus. Curr. Hematol. Malig. Rep. 2019, 14, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Bollard, C.M.; Heslop, H.E. T cells for viral infections after allogeneic hematopoietic stem cell transplant. Blood 2016, 127, 3331–3340. [Google Scholar] [CrossRef]
- Qian, C.; Wang, Y.; Reppel, L.; D’Aveni, M.; Campidelli, A.; Decot, V.; Bensoussan, D. Viral-specific T-cell transfer from HSCT donor for the treatment of viral infections or diseases after HSCT. Bone Marrow Transplant. 2018, 53, 114–122. [Google Scholar] [CrossRef]
- Ottaviano, G.; Chiesa, R.; Feuchtinger, T.; Vickers, M.; Dickinson, A.; Gennery, A.; Veys, P.; Todryk, S. Adoptive T Cell Therapy Strategies for Viral Infections in Patients Receiving Haematopoietic Stem Cell Transplantation. Cells 2019, 8, 47. [Google Scholar] [CrossRef]
- Gottlieb, D. Antifungal T cells-progress in manufacture and prospects for the clinic. Cytotherapy 2015, 17, 1329–1331. [Google Scholar] [CrossRef] [PubMed]
- Santomasso, B.; Bachier, C.; Westin, J.; Rezvani, K.; Shpall, E.J. The Other Side of CAR T-Cell Therapy: Cytokine Release Syndrome, Neurologic Toxicity, and Financial Burden. Am. Soc. Clin. Oncol. Educ. B 2019, 433–444. [Google Scholar] [CrossRef]
- Dellière, S.; Guery, R.; Candon, S.; Rammaert, B.; Aguilar, C.; Lanternier, F.; Chatenoud, L.; Lortholary, O. Understanding pathogenesis and care challenges of immune reconstitution inflammatory syndrome in fungal infections. J. Fungi 2018, 4, 139. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou, A.; Krance, R.A.; Allen, C.E.; Lee, D.; Rooney, C.M.; Brenner, M.K.; Leen, A.M.; Heslop, H.E. Systemic inflammatory response syndrome after administration of unmodified T lymphocytes. Mol. Ther. 2014, 22, 1134–1138. [Google Scholar] [CrossRef]
- Simmons, H.Z.; Bazzell, A.F.; Dains, J.E. Adverse Effects of Virus-Specific T-Cell Therapy: An Integrative Review. J. Adv. Pract. Oncol. 2019, 10, 120–131. [Google Scholar]
- Yu, A.I.; Zhao, L.; Eaton, K.A.; Ho, S.; Chen, J.; Poe, S.; Becker, J.; Gonzalez, A.; McKinstry, D.; Hasso, M.; et al. Gut Microbiota Modulate CD8 T Cell Responses to Influence Colitis-Associated Tumorigenesis. Cell Rep. 2020, 31. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Xia, Y.; He, F.; Zhu, C.; Ren, W. Intestinal mycobiota in health and diseases: From a disrupted equilibrium to clinical opportunities. Microbiome 2021, 9, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Iliev, I.D.; Leonardi, I. Fungal dysbiosis: Immunity and interactions at mucosal barriers. Nat. Rev. Immunol. 2017, 17, 635–646. [Google Scholar] [CrossRef]
- Hakki, M.; Riddell, S.R.; Storek, J.; Carter, R.A.; Stevens-Ayers, T.; Sudour, P.; White, K.; Corey, L.; Boeckh, M. Immune reconstitution to cytomegalovirus after allogeneic hematopoietic stem cell transplantation: Impact of host factors, drug therapy, and subclinical reactivation. Blood 2003, 102, 3060–3067. [Google Scholar] [CrossRef]
- Savoldo, B.; Goss, J.; Liu, Z.; Huls, M.H.; Doster, S.; Gee, A.P.; Brenner, M.K.; Heslop, H.E.; Rooney, C.M. Generation of autologous Epstein-Barr virus-specific cytotoxic T cells for adoptive immunotherapy in solid organ transplant recipients. Transplantation 2001, 72, 1078–1086. [Google Scholar] [CrossRef]
- Vafadari, R.; Kraaijeveld, R.; Weimar, W.; Baan, C.C. Tacrolimus inhibits NF-κB activation in peripheral human T cells. PLoS ONE 2013, 8, e60784. [Google Scholar] [CrossRef]
- Widmann, T.; Sester, U.; Gärtner, B.C.; Schubert, J.; Pfreundschuh, M.; Köhler, H.; Sester, M. Levels of CMV Specific CD4 T Cells Are Dynamic and Correlate with CMV Viremia after Allogeneic Stem Cell Transplantation. PLoS ONE 2008, 3, e3634. [Google Scholar] [CrossRef] [PubMed]
- Zhan, X.; Brown, B.; Slobod, K.S.; Hurwitz, J.L. Inhibition of ex vivo-expanded cytotoxic T-lymphocyte function by high-dose cyclosporine. Transplantation 2003, 76, 739–740. [Google Scholar] [CrossRef]
- Menger, L.; Gouble, A.; Marzolini, M.A.V.; Pachnio, A.; Bergerhoff, K.; Henry, J.Y.; Smith, J.; Pule, M.; Moss, P.; Riddell, S.R.; et al. TALEN-mediated genetic inactivation of the glucocorticoid receptor in cytomegalovirus-specific T cells. Blood 2015, 126, 2781–2789. [Google Scholar] [CrossRef] [PubMed]
- Kaeuferle, T.; Deisenberger, L.; Jablonowski, L.; Stief, T.A.; Blaeschke, F.; Willier, S.; Feuchtinger, T. CRISPR-Cas9-Mediated Glucocorticoid Resistance in Virus-Specific T Cells for Adoptive T Cell Therapy Posttransplantation. Mol. Ther. 2020, 28, 1965–1973. [Google Scholar] [CrossRef] [PubMed]
- Basar, R.; Daher, M.; Uprety, N.; Gokdemir, E.; Alsuliman, A.; Ensley, E.; Ozcan, G.; Mendt, M.; Sanabria, M.H.; Kerbauy, L.N.; et al. Large-scale GMP-compliant CRISPR-Cas9—Mediated deletion of the glucocorticoid receptor in multivirus-speci fi c T cells. Blood Adv. 2020, 4. [Google Scholar] [CrossRef] [PubMed]
- Mo, F.; Watanabe, N.; McKenna, M.K.; Hicks, M.J.; Srinivasan, M.; Gomes-Silva, D.; Atilla, E.; Smith, T.; Atilla, P.A.; Ma, R.; et al. Engineered off-the-shelf therapeutic T cells resist host immune rejection. Nat. Biotechnol. 2021, 39, 56–63. [Google Scholar] [CrossRef] [PubMed]
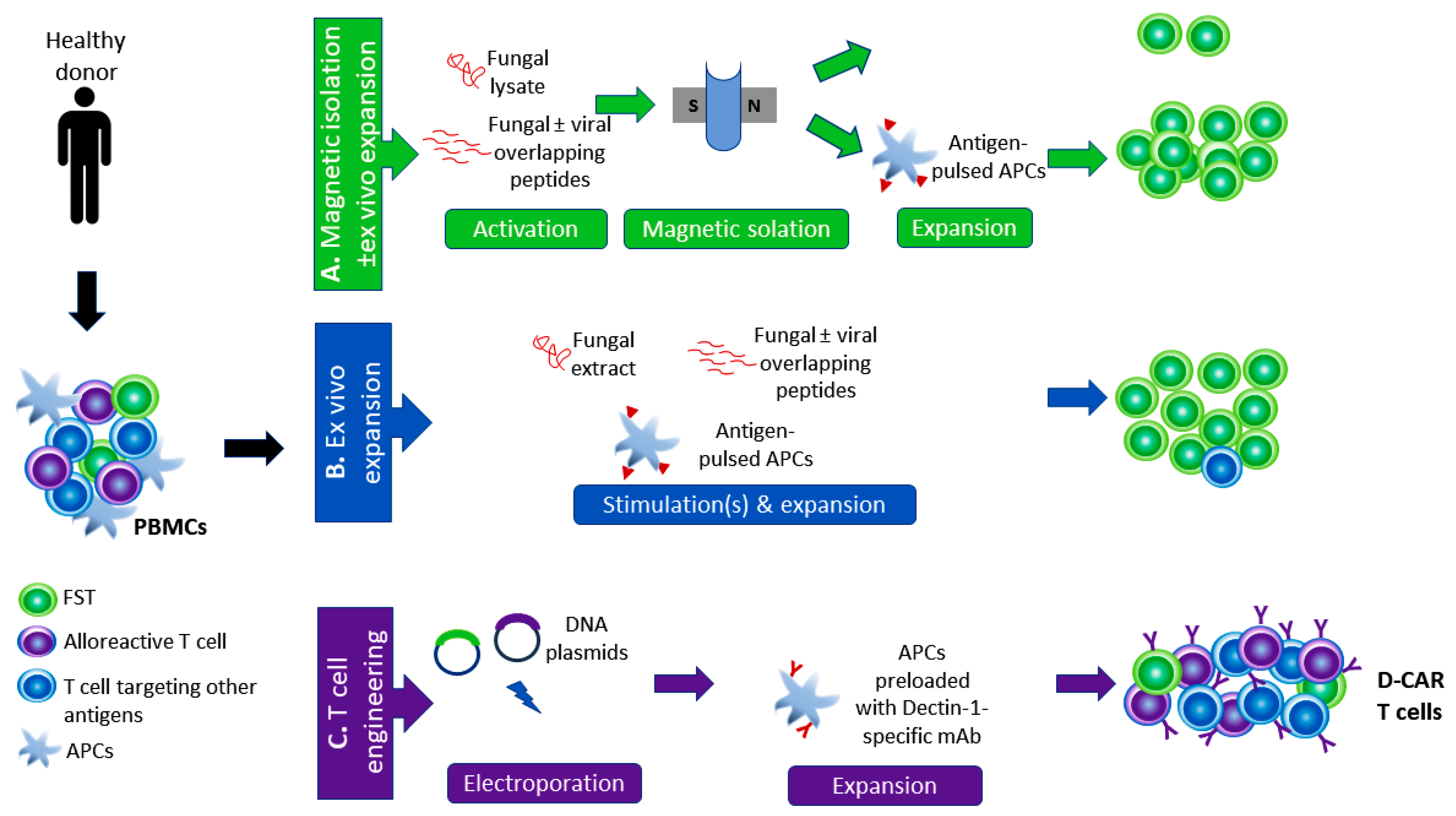
| Targeted Pathogen(s) | Manufacturing Protocol | Group | Antigen Competition | Cross-Reactivity | Clinically Tested | |
|---|---|---|---|---|---|---|
| Monofungus-specific T cell products | A. fumigatus | Ex vivo expansion post stimulation of PBMCs with A. fumigatus extract | Ramadan et al., 2004 [65] | n/a | Not tested | No |
| A. fumigatus | Ex vivo expansion post stimulation of PBMCs with A. fumigatus extract | Perrucio et al., 2005 [41] | n/a | Not tested | Yes | |
| A. fumigatus | IFN-γ selection post stimulation of PBMCs with Aspergillus extract | Beck et al., 2006 [66] | n/a | Against A. flavus, A. niger, and P. chrysogenum-not against A. alternata and C. albicans | No | |
| C. albicans | IFN-γ selection and ex vivo expansion post stimulation of PBMCs with C. albicans extract | Tramsen et al., 2007 [67] | n/a | Against C. tropicalis but not C. glabrata, P. chrysogenum, A. alternata, and A. fumigatus | No | |
| A. fumigatus | Ex vivo expansion post stimulation of PBMCs with A. fumigatus Af16 | Zhu et al., 2008 [68] | n/a | Not tested | No | |
| A. fumigatus | IFN-γ selection and ex vivo expansion post stimulation of WBC with A. fumigatus extract | Tramsen et al., 2009 [69] | n/a | Limited cross-reactivity against other filamentous fungi (data not shown), but not Candida species | No | |
| A. fumigatus | Ex vivo expansion post stimulation of PBMCs with A. fumigatus lysate | Gaundar et al., 2012 [70] | n/a | Against A. niger and A. flavus, C. albicans, S. apiospermum and Penicillium species—not against A. terreus, C. glabrata, Fusarium species and Mucor species | No | |
| R. oryzae | CD154 selection, ex vivo expansion and IFN-γ enrichment post stimulation of PBMCs with R. oryzae cell extract | Schmidt et al., 2012 [71] | n/a | Against R. microsporus, R. pusillus, M. circinelloides, A. fumigatus, P. chrysogenum and C. albicans. No response against A. flavus, A. alternata and M. racemosus | No | |
| A. fumigatus | CD137 selection and ex vivo expansion post stimulation of PBMCs with Crf1 and catalase-1 (A. fumigatus) | Jolink et al., 2013 [72] | n/a | Not tested | No | |
| Aspergillus species | Dectin-1 CAR T cells specific for β-glucans and bispecific T cells co-expressing Dectin-1-CAR and CD19-CAR | Kumaresan et al., 2014 [73] | n/a | Not tested | No | |
| A. fumigatus | CD137 selection post stimulation of PBMCs with A. fumigatus lysate | Bacher et al., 2015 [74] | n/a | Against other Aspergillus spp. and C. albicans | No | |
| A. fumigatus | CD154 or CD137 selection and ex vivo expansion post stimulation of PBMCs with fungal extract or Crf1 and/or Pmp20 and/or Gel1 | Stuehler et al., 2015 [75] | n/a | Against A. flavus, A. terreus, and A. niger, S. apiospermum, S. prolificans, F. solani, R. microsporus, L. corymbifera, and C. albicans | No | |
| A. fumigatus or A. flavus or A. terreus or C. albicans or C. krusei or F. solani or F. oxysporum or R. oryzae or L. prolificans | Εx vivo expansion post stimulation of PBMCs with fungal extract | Deo et al., 2016 [76] | n/a | A terreus, F oxysporum and L prolificans T cell cultures cross-reacted with one another and against A. fumigatus, A. flavus and F. solani. C. krusei T cell cultures cross-reacted against C. albicans, or with Aspergillus and Fusarium species. Cross-reactivity with R. oryzae was observed in a subset of the T cell cultures | No | |
| R. oryzae | Ex vivo expansion post stimulation of PBMCs with R. oryzae lysate | Castillo et al., 2018 [77] | n/a | Against A. fumigatus | No | |
| A. fumigatus | Ex vivo expansion post stimulation of PBMCs with A. fumigatus lysate or Crf1, Gel1 and SHMT | Papadopoulou et al., 2019 [78] | n/a | Against A. flavus, A. niger, F. solani, F. oxysporum, C. tropicalis and C. albicans | No | |
| Multifungus specific T cell products | A. fumigatus, C. albicans and R. oryzae | IFN-γ selection and ex vivo expansion post stimulation of PBMCs with fungal extract | Tramsen et al., 2013 [79] | Lower numbers of T cells responding to A. fumigatus were detected in the multifungus T cell product | Against A. niger, P. chrysogenum, C. tropicalis, M. circinelloides, R. pusillus, R. microsporus and R. microsporus-oligosporus | No |
| A. terreus, C. krusei and R. oryzae | Εx vivo expansion ±TNF-α selection post stimulation of PBMCs with fungal extract | Deo et al., 2016 [76] | Not illustrated | Against A. fumigatus, C. albicans, C. krusei and Lomentospora, A. flavus and R. oryzae | No | |
| C. krusei and A. terreus | CD137 selection and ex vivo expansion post stimulation of PBMCs with fungal extract | Castellano-Gonzalez et al., 2020 [59] | Not illustrated | Higher cross-reactivity against A. fumigatus, A. flavus, A. terreus, C. albicans and C. krusei and lower against F. solani, F. oxysporum and S. prolificans | No | |
| Multi-pathogen-specific T cell products | AdV, EBV, CMV, C. albicans, and/or A. fumigatus or AdV, EBV, C. albicans, and A. fumigatus | CD154 selection and ex vivo expansion post stimulation of PBMCs with hexon (AdV), LMP2 (EBV), pp65 (CMV), mannose protein 65 (MP65, C. albicans) and Crf1 (A. fumigatus) | Khanna et al., 2011 [80] | Notable decrease in the frequencies of antigen-specific T cells in the multipathogen-specific cultures vs. single lines | Not tested | No |
| EBV, CMV, BKV, and A. fumigatus | Ex vivo expansion post stimulation of PBMCs with IE1 and pp65 (CMV), EBNA1, LMP2 and BZLF1 (EBV), Large T and VP1 (BKV) and Crf1, Gel1 and SHMT (A. fumigatus) | Papadopoulou et al., 2021 [81] | No | Cross-reactivity against C. albicans, C. tropicalis, F. solani, F. oxysporum | No | |
| AdV, EBV, CMV, BKV, VzV, influenza and A. fumigatus | Ex vivo expansion post stimulation of PBMCs with pp65 (CMV), Hexon (AdV), EBNA1, LMP2A and BZLF1 (EBV), Large T and VP1 (BKV), Vzv vaccine, Influenza vaccine and lysate (A. fumigatus). Cultures from individually stimulated products were combined and restimulated for further expansion. | Gottllieb et al., 2021 [62] | n/a | Not tested | Yes | |
| AdV, EBV, CMV, BKV, and A. fumigatus—steroid resistant | Ex vivo expansion post stimulation of PBMCs with hexon and penton (AdV), IE1 and pp65 (CMV), EBNA1, LMP2 and BZLF1 (EBV), Large T and VP1 (BKV) and Crf1, Gel1 and SHMT (A. fumigatus) and genetical modification to inactivate the glucocorticoid receptor | Koukoulias et al., 2021 [82] | No | Cross-reactivity against A. flavus, A. niger, F. solani, F. oxysporum | No |
| Pros | Cons |
|---|---|
| High specificity and potential efficacy | Selection of the appropriate antigenic target |
| Broad coverage against multiple pathogens | Antigenic competition |
| Long-lasting immunity with graft donor-derived FSTs | “Off-target” or/and “on-target” toxicity |
| Immediate availability as an off-the-shelf therapy | No long-term persistence of anti-fungus immunity with third-party, off-the shelf FSTs |
| Triggering of epitope spreading | Sustainability of the clinical benefit? |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karavalakis, G.; Yannaki, E.; Papadopoulou, A. Reinforcing the Immunocompromised Host Defense against Fungi: Progress beyond the Current State of the Art. J. Fungi 2021, 7, 451. https://doi.org/10.3390/jof7060451
Karavalakis G, Yannaki E, Papadopoulou A. Reinforcing the Immunocompromised Host Defense against Fungi: Progress beyond the Current State of the Art. Journal of Fungi. 2021; 7(6):451. https://doi.org/10.3390/jof7060451
Chicago/Turabian StyleKaravalakis, Georgios, Evangelia Yannaki, and Anastasia Papadopoulou. 2021. "Reinforcing the Immunocompromised Host Defense against Fungi: Progress beyond the Current State of the Art" Journal of Fungi 7, no. 6: 451. https://doi.org/10.3390/jof7060451
APA StyleKaravalakis, G., Yannaki, E., & Papadopoulou, A. (2021). Reinforcing the Immunocompromised Host Defense against Fungi: Progress beyond the Current State of the Art. Journal of Fungi, 7(6), 451. https://doi.org/10.3390/jof7060451






