Lack of Linkages among Fruiting Depth, Weight, and Maturity in Irrigated Truffle Fungi Marks the Complexity of Relationships among Morphogenetic Stages
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site
2.2. Experimental Design and Data Collection
2.3. Statistical Analysis
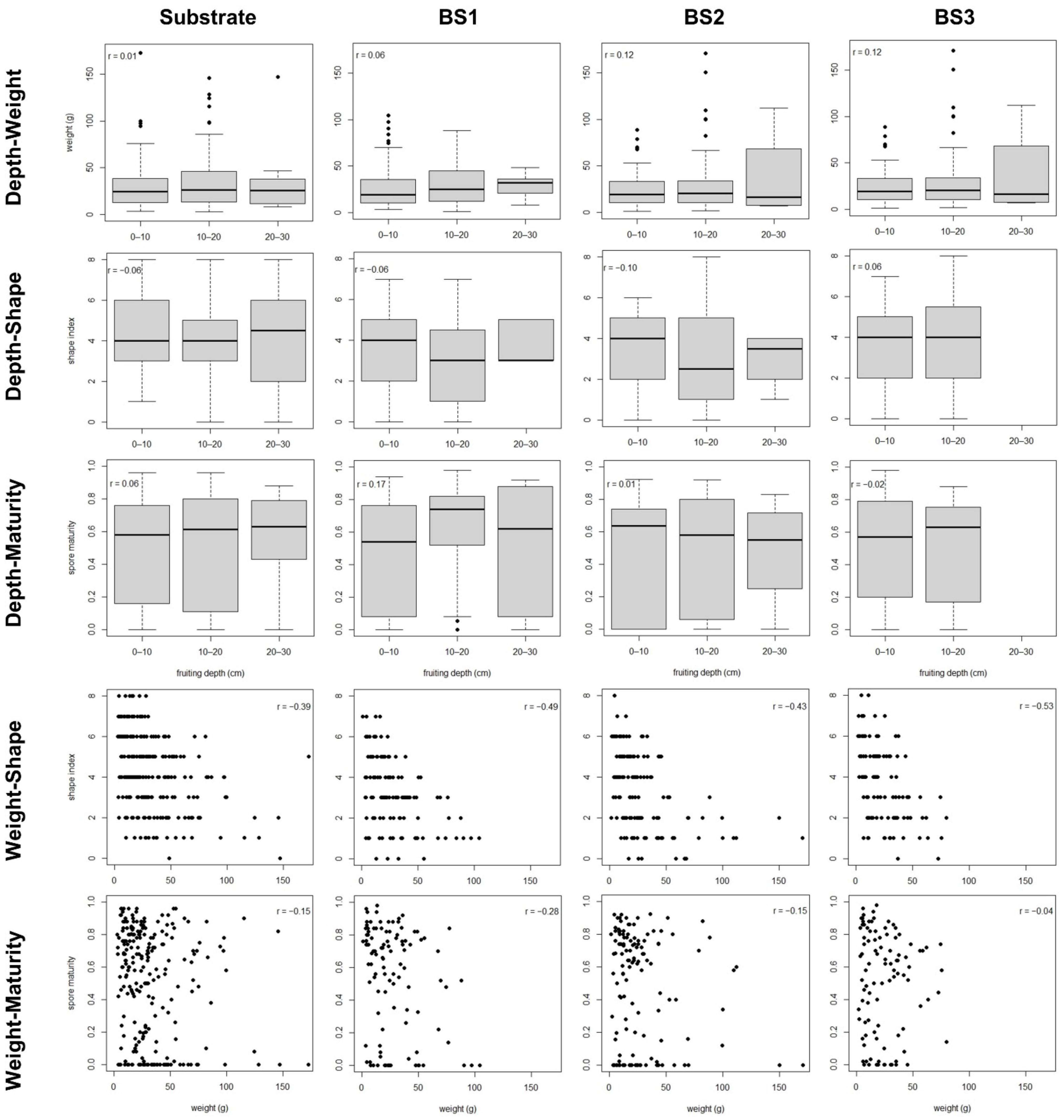
3. Results
3.1. Single Fruitbodies
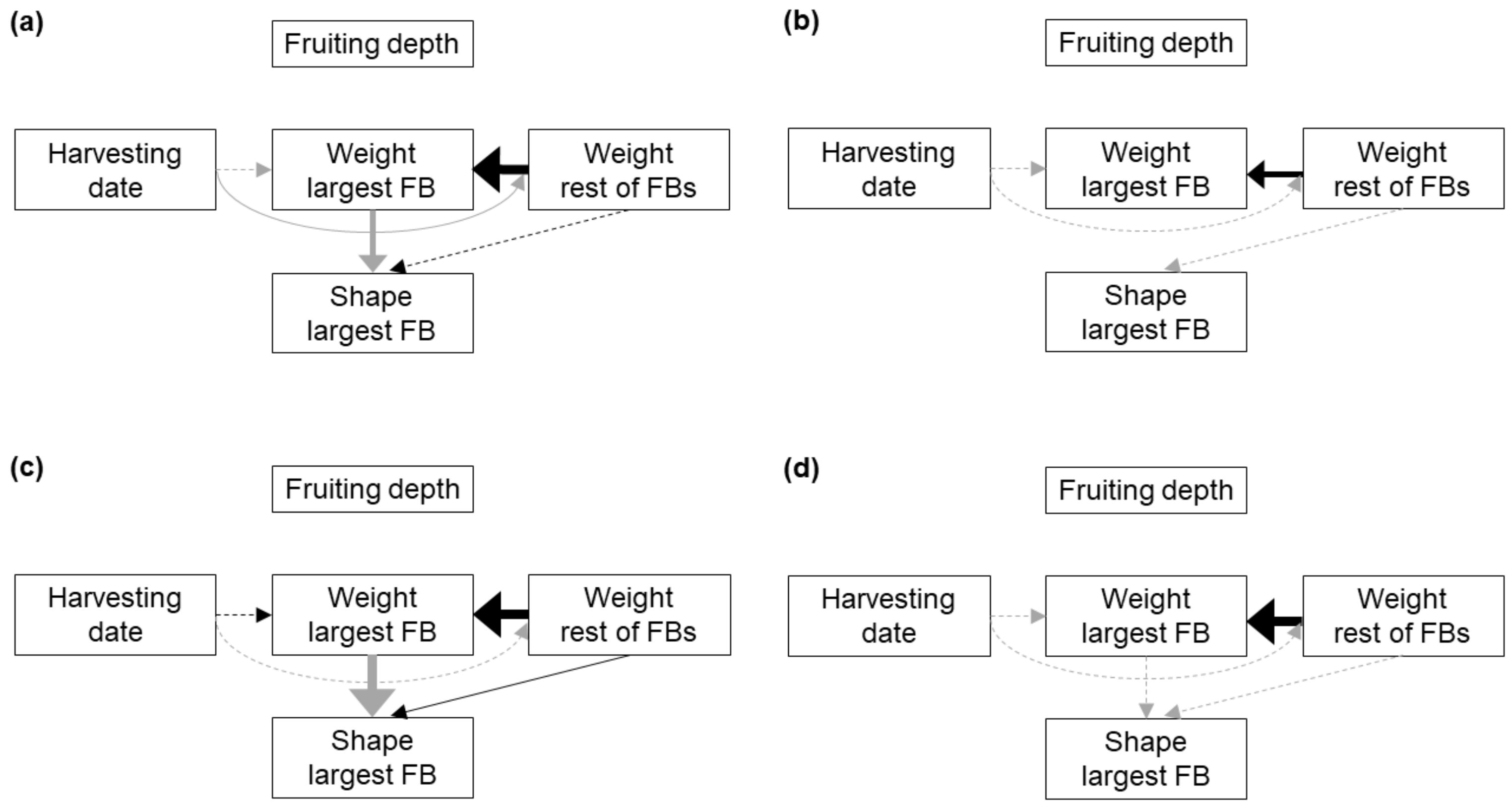
3.2. Fruitbody Clusters
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pacioni, G.; Leonardi, M.; Di Carlo, P.; Ranalli, D.; Zinni, A.; De Laurentiis, G. Instrumental monitoring of the birth and development of truffles in a Tuber melanosporum orchard. Mycorrhiza 2014, 24, 65–72. [Google Scholar] [CrossRef]
- Taschen, E.; Rousset, F.; Sauve, M.; Benoit, L.; Dubois, M.-P.; Richard, F.; Selosse, M.-A. How the truffle got its mate: Insights from genetic structure in spontaneous and planted Mediterranean populations of Tuber melanosporum. Mol. Ecol. 2016, 25, 5611–5627. [Google Scholar] [CrossRef]
- Martin, F.; Kohler, A.; Murat, C.; Balestrini, R.; Coutinho, P.M.; Jaillon, O.; Montanini, B.; Morin, E.; Noel, B.; Percudani, R.; et al. Périgord black truffle genome uncovers evolutionary origins and mechanisms of symbiosis. Nature 2010, 464, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Le Tacon, F.; Zeller, B.; Plain, C.; Hossann, C.; Bréchet, C.; Robin, C. Carbon Transfer from the Host to Tuber melanosporum Mycorrhizas and Ascocarps Followed Using a 13C Pulse-Labeling Technique. PLoS ONE 2013, 8, e64626. [Google Scholar] [CrossRef] [PubMed]
- Splivallo, R.; Ottonello, S.; Mello, A.; Karlovsky, P. Truffle volatiles: From chemical ecology to aroma biosynthesis. New Phytol. 2011, 189, 688–699. [Google Scholar] [CrossRef] [PubMed]
- Parguey-Leduc, A.; Montant, C.; Kulifaj, M. Structure et évolution de l’ascocarpe du Tuber melanosporum Vitt. (Truffe noire du Périgord). C. R. Acad. Sci. III 1984, 299, 315–320. [Google Scholar]
- Zarivi, O.; Cesare, P.; Ragnelli, A.M.; Aimola, P.; Leonardi, M.; Bonfigli, A.; Colafarina, S.; Poma, A.M.; Miranda, M.; Pacioni, G. Validation of reference genes for quantitative real-time PCR in Périgord black truffle (Tuber melanosporum) developmental stages. Phytochemistry 2015, 116, 78–86. [Google Scholar] [CrossRef]
- Montant, C.; Kulifaj, M.; Gleize, R. Note sur la recolte de jeunes ascocarpes du Tuber melanosporum Vitt. (Truffe noire de perigord) et leur evolution. C. R. Acad. Sci. III 1983, 296, 463–468. [Google Scholar]
- Montant, C.; Kulifaj, M. L’ascocarpe du Tuber melanosporum Vitt. Contrôle des facteurs externes agissant sur sa formation, sa croissance et sa maturation. C. R. Acad. Sci. III 1990, 311, 123–126. [Google Scholar]
- Zarivi, O.; Bonfigli, A.; Colafarina, S.; Aimola, P.; Ragnelli, A.M.; Pacioni, G.; Miranda, M. Tyrosinase expression during black truffle development: From free living mycelium to ripe fruit body. Phytochemistry 2011, 72, 2317–2324. [Google Scholar] [CrossRef]
- Rubini, A.; Belfiori, B.; Riccioni, C.; Arcioni, S.; Martin, F.; Paolocci, F. Tuber melanosporum: Mating type distribution in a natural plantation and dynamics of strains of different mating types on the roots of nursery-inoculated host plants. New Phytol. 2011, 189, 723–735. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.; Zarivi, O.; Bonfigli, A.; Amicarelli, F.; Aimola, P.; Ragnelli, A.M.; Pacioni, G. Melanogenesis, Tyrosinase Expression, and Reproductive Differentiation in Black and White Truffles (Ascomycotina). Pigment Cell Res. 1997, 10, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Caboni, P.; Scano, P.; Sanchez, S.; Garcia-Barreda, S.; Corrias, F.; Marco, P. Multi-platform metabolomic approach to discriminate ripening markers of black truffles (Tuber melanosporum). Food Chem. 2020, 319, 126573. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.E.; Molina, R.; Huso, M.M.; Luoma, D.L.; McKay, D.; Castellano, M.A.; Lebel, T.; Valachovic, Y. Species richness, abundance, and composition of hypogeous and epigeous ectomycorrhizal fungal sporocarps in young, rotation-age, and old-growth stands of Douglas-fir (Pseudotsuga menziesii) in the Cascade Range of Oregon, USA. Can. J. Bot. 2002, 80, 186–204. [Google Scholar] [CrossRef]
- Murat, C.; Bonneau, L.; De La Varga, H.; Olivier, J.-M.; Fizzala, S.; Le Tacon, F. Le Trapping truffle production in holes: A promising technique for improving production and unravelling truffle life cycle. Ital. J. Mycol. 2016, 45, 47–53. [Google Scholar]
- Zarivi, O.; Bonfigli, A.; Cesare, P.; Amicarelli, F.; Pacioni, G.; Miranda, M. Truffle thio-flavours reversibly inhibit truffle tyrosinase. FEMS Microbiol. Lett. 2003, 220, 81–88. [Google Scholar] [CrossRef][Green Version]
- UNECE. UNECE Standard FFV-53 Concerning the Marketing and Commercial Quality Control of Truffles: 2017 Edition; UNECE: New York, NY, USA, 2017. [Google Scholar]
- Pregitzer, K.S.; King, J.S.; Burton, A.J.; Brown, S.E. Responses of tree fine roots to temperature. New Phytol. 2000, 147, 105–115. [Google Scholar] [CrossRef]
- Tromp-van Meerveld, H.J.; McDonnell, J.J. On the interrelations between topography, soil depth, soil moisture, transpiration rates and species distribution at the hillslope scale. Adv. Water Resour. 2006, 29, 293–310. [Google Scholar] [CrossRef]
- Moore, D.; Gange, A.C.; Gange, E.G. Fruit bodies: Their production and development in relation to environment. In Ecology of Saprotrophic Basidiomycetes; Boddy, L., Frankland, J.C., van West, P., Eds.; Academic Press: London, UK, 2008; pp. 79–103. ISBN 9780123741851. [Google Scholar]
- Garcia-Barreda, S.; Marco, P.; Martín-Santafé, M.; Tejedor-Calvo, E.; Sánchez, S. Edaphic and temporal patterns of Tuber melanosporum fruitbody traits and effect of localised peat-based amendment. Sci. Rep. 2020, 10, 4422. [Google Scholar] [CrossRef]
- Garcia-Barreda, S.; Sánchez, S.; Marco, P.; Serrano-Notivoli, R. Agro-climatic zoning of Spanish forests naturally producing black truffle. Agric. For. Meteorol. 2019, 269–270, 231–238. [Google Scholar] [CrossRef]
- Jaillard, B.; Barry-Etienne, D.; Colinas, C.; De Miguel, A.M.; Genola, L.; Libre, A.; Neveu, P.; Oliach, D.; Saenz, W.; Saez, M.; et al. Alkalinity and structure of soils determine the truffle production in the Pyrenean Regions. For. Syst. 2014, 23, 364–377. [Google Scholar] [CrossRef]
- Zeppa, S.; Guidi, C.; Zambonelli, A.; Potenza, L.; Vallorani, L.; Pierleoni, R.; Sacconi, C.; Stocchi, V. Identification of putative genes involved in the development of Tuber borchii fruit body by mRNA differential display in agarose gel. Curr. Genet. 2002, 42, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Shipley, B. Cause and Correlation in Biology: A User’s Guide to Path Analysis, Structural Equations and Causal Inference with R, 2nd ed.; Cambridge University Press: Cambridge, UK, 2016; ISBN 0511017723. [Google Scholar]
- Marchetti, G.M.; Drton, M.; Sadeghi, K. ggm: Functions for Graphical Markov Models (version 2.5). 2015. Available online: https://CRAN.R-project.org/package=ggm (accessed on 31 January 2021).
- Mądry, W.; Studnicki, M.; Rozbicki, J.; Golba, J.; Gozdowski, D.; Pecio, A.; Oleksy, A. Ontogenetic-based sequential path analysis of grain yield and its related traits in several winter wheat cultivars. Acta Agric. Scand. Sect. B Soil Plant Sci. 2015, 65, 605–618. [Google Scholar] [CrossRef]
- Shipley, B. The AIC model selection method applied to path analytic models compared using a d-separation test. Ecology 2013, 94, 560–564. [Google Scholar] [CrossRef] [PubMed]
- Barrett, G.E.; Alexander, P.D.; Robinson, J.S.; Bragg, N.C. Achieving environmentally sustainable growing media for soilless plant cultivation systems—A review. Sci. Hortic. 2016, 212, 220–234. [Google Scholar] [CrossRef]
- Wood, S.N. Generalized Additive Models: An Introduction with R; Chapman and Hall/CRC: Boca Raton, FL, USA, 2017. [Google Scholar]
- Wood, S.N. Fast stable restricted maximum likelihood and marginal likelihood estimation of semiparametric generalized linear models. J. R. Stat. Soc. 2011, 73, 3–36. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2019. [Google Scholar]
- Eslick, H.; Dell, B. The role of irrigation in the rot of truffles in Western Australia. In Proceedings of the Abstract Book of the 1st International Congress of Trufficulture of Teruel Tuber 2013, Teruel, Spain, 5–8 March 2013; Gobierno de Aragón—CITA: Zaragoza, Spain, 2013; p. 37. [Google Scholar]
- Sourzat, P.; Kulifaj, M.; Montant, C. Résultats Techniques Sur la Trufficulture à Partir D’expérimentations Conduites Dans le Lot Entre 1985 et 1992; Station d’Expérimentations sur la Truffe: Cahors, France, 1993. [Google Scholar]
- Luoma, D.L. Biomass and Community Structure of Sporocarps Formed by Hypogeous Ectomycorrhizal Fungi within Selected Forest Habitats of the H. J. Andrews Experimental Forest, Oregon. Ph.D. Thesis, Oregon State University, Corvallis, OR, USA, 1988. [Google Scholar]
- Luoma, D.L.; Frenkel, R.E.; Trappe, J.M. Fruiting of Hypogeous Fungi in Oregon Douglas-Fir Forests: Seasonal and Habitat Variation. Mycologia 1991, 83, 335–353. [Google Scholar] [CrossRef]
- Hacquard, S.; Tisserant, E.; Brun, A.; Legué, V.; Martin, F.; Kohler, A. Laser microdissection and microarray analysis of Tuber melanosporum ectomycorrhizas reveal functional heterogeneity between mantle and Hartig net compartments. Environ. Microbiol. 2013, 15, 1853–1869. [Google Scholar] [CrossRef]
- Le Tacon, F.; Zeller, B.; Plain, C.; Hossann, C.; Bréchet, C.; Martin, F.; Kohler, A.; Villerd, J.; Robin, C. Study of nitrogen and carbon transfer from soil organic matter to Tuber melanosporum mycorrhizas and ascocarps using 15N and 13C soil labelling and whole-genome oligoarrays. Plant Soil 2015, 395, 351–373. [Google Scholar] [CrossRef]
- Montanini, B.; Levati, E.; Bolchi, A.; Kohler, A.; Morin, E.; Tisserant, E.; Martin, F.; Ottonello, S. Genome-wide search and functional identification of transcription factors in the mycorrhizal fungus Tuber melanosporum. New Phytol. 2011, 189, 736–750. [Google Scholar] [CrossRef]
- Mandrile, L.; Mello, A.; Vizzini, A.; Balestrini, R.; Rossi, A.M. Near-infrared spectroscopy as a new method for post-harvest monitoring of white truffles. Mycol. Prog. 2020, 19, 329–337. [Google Scholar] [CrossRef]
- Garcia-Barreda, S.; Camarero, J.J.; Vicente-Serrano, S.M.; Serrano-Notivoli, R. Variability and trends of black truffle production in Spain (1970–2017): Linkages to climate, host growth, and human factors. Agric. For. Meteorol. 2020, 287, 107951. [Google Scholar] [CrossRef]
- Olivier, J.-M.; Savignac, J.-C.; Sourzat, P. Truffe et Trufficulture; Editions Fanlac: Périgueux, France, 1996; ISBN 2-86577-180-6. [Google Scholar]
- Hunt, G.A.; Trappe, J.M. Seasonal hypogeous sporocarp production in a western Oregon Douglas-fir stand. Can. J. Bot. 1987, 65, 438–445. [Google Scholar] [CrossRef]
- Frey-Klett, P.; Burlinson, P.; Deveau, A.; Barret, M.; Tarkka, M.; Sarniguet, A. Bacterial-fungal interactions: Hyphens between agricultural, clinical, environmental, and food microbiologists. Microbiol. Mol. Biol. Rev. 2011, 75, 583–609. [Google Scholar] [CrossRef] [PubMed]
- Künzler, M. How fungi defend themselves against microbial competitors and animal predators. PLoS Pathog. 2018, 14, e1007184. [Google Scholar] [CrossRef]
- Kues, U.; Liu, Y. Fruiting body production in basidiomycetes. Appl. Microbiol. Biotechnol. 2000, 54, 141–152. [Google Scholar] [CrossRef]
- De Groot, P.W.J.; Visser, J.; Van Griensven, L.J.L.D.; Schaap, P.J. Biochemical and molecular aspects of growth and fruiting of the edible mushroom Agaricus bisporus. Mycol. Res. 1998, 102, 1297–1308. [Google Scholar] [CrossRef]
- Antony-Babu, S.; Deveau, A.; Van Nostrand, J.D.; Zhou, J.; Le Tacon, F.; Robin, C.; Frey-Klett, P.; Uroz, S. Black truffle-associated bacterial communities during the development and maturation of Tuber melanosporum ascocarps and putative functional roles. Environ. Microbiol. 2014, 16, 2831–2847. [Google Scholar] [CrossRef]
- Benucci, G.M.N.; Bonito, G.M. The Truffle Microbiome: Species and Geography Effects on Bacteria Associated with Fruiting Bodies of Hypogeous Pezizales. Microb. Ecol. 2016, 72, 4–8. [Google Scholar] [CrossRef]
- Pacioni, G.; Leonardi, M. Truffle-Inhabiting Fungi. In True Truffle (Tuber spp.) in the World; Zambonelli, A., Iotti, M., Murat, C., Eds.; Springer: Cham, Switzerland, 2016; pp. 283–299. [Google Scholar]


| Stage of Fruitbody Morphogenesis | Associated Development Characters | Period |
|---|---|---|
| Formation of mating structures (gametes), mating, and early stage of fruitbody differentiation (hyphal stage) | Fruiting depth | May–June |
| Fruitbody growth: development and swelling (peridial, veined, ascal and sporal stages) | Weight, shape | July–early November |
| Maturation (pigmented stage). Ripening (aroma development) | Spore maturity, harvesting date | November–March |
| Model | C (df, P) | K | AICc | W |
|---|---|---|---|---|
| Substrate | ||||
| A | 12.5 (14, 0.56) | 11.6 | 36.9 | 0.80 |
| B | 9.7 (8, 0.28) | 14.5 | 40.7 | 0.12 |
| C | 8.2 (6, 0.23) | 15.5 | 41.4 | 0.08 |
| Bulk soil of block 1 | ||||
| A | 22.3 (14, 0.07) | 10.5 | 45.6 | 0.05 |
| B | 9.9 (8, 0.27) | 13.5 | 40.8 | 0.50 |
| C | 7.5 (6, 0.28) | 14.5 | 41.1 | 0.45 |
| Bulk soil of block 2 | ||||
| A | 7.9 (14, 0.90) | 10.6 | 31.0 | 0.83 |
| B | 5.2 (8, 0.74) | 13.6 | 35.6 | 0.08 |
| C | 3.3 (6, 0.77) | 14.3 | 35.7 | 0.08 |
| Bulk soil of block 3 | ||||
| A | 10.9 (14, 0.69) | 10.5 | 34.6 | 0.89 |
| B | 9.0 (8, 0.35) | 13.5 | 40.3 | 0.05 |
| C | 6.7 (6, 0.35) | 14.2 | 40.1 | 0.06 |
| Response | Predictor | Substrate | BS2 | BS3 | |||
|---|---|---|---|---|---|---|---|
| P | D2 | P | D2 | P | D2 | ||
| Weight 1 | Harvesting date | <0.001 | 5.8 | 0.03 | 3.6 | 0.04 | 4.1 |
| Shape | Weight | <0.001 | 16.4 | <0.001 | 21.9 | <0.001 | 28.7 |
| Maturity | Harvesting date | <0.001 | 40.8 | <0.001 | 35.5 | <0.001 | 34.2 |
| Response | Predictor | P | D2 |
|---|---|---|---|
| Weight 1 | Harvesting date | 0.13 | - |
| Fruiting depth | 0.20 | - | |
| Shape | Weight | <0.001 | 23.6 |
| Maturity | Harvesting date | <0.001 | 22.7 |
| Weight | 0.01 | 4.2 | |
| Fruiting depth | 0.47 | - |
| Model | C (df, P) | K | AICc | W |
|---|---|---|---|---|
| Substrate | ||||
| A | 74.9 (14, <0.001) | - | - | - |
| B | 60.0 (10, <0.001) | - | - | - |
| C | 61.7 (8, <0.001) | - | - | - |
| D | 6.6 (10, 0.76) | 13.3 | 35.2 | 0.85 |
| E | 2.6 (6, 0.86) | 16.4 | 38.6 | 0.15 |
| Bulk soil of block 1 | ||||
| A | 22.3 (14, 0.07) | 8.4 | 43.6 | 0.11 |
| B | 18.0 (10, 0.06) | 10.5 | 46.1 | 0.03 |
| C | 17.1 (8, 0.03) | - | - | - |
| D | 10.4 (10, 0.41) | 10.8 | 39.7 | 0.81 |
| E | 7.4 (6, 0.29) | 13.2 | 45.8 | 0.04 |
| Bulk soil of block 2 | ||||
| A | 29.4 (14, 0.009) | - | - | - |
| B | 26.7 (10, 0.003) | - | - | - |
| C | 20.7 (8, 0.008) | - | - | - |
| D | 7.6 (10, 0.67) | 11.2 | 39.9 | 0.99 |
| E | 6.9 (6, 0.33) | 13.7 | 50.0 | 0.01 |
| Bulk soil of block 3 | ||||
| A | 19.3 (14, 0.15) | 8.2 | 87.7 | <0.01 |
| B | 12.7 (10, 0.24) | 10.2 | 59.4 | 0.39 |
| C | 12.2 (8, 0.14) | 11.2 | 70.1 | <0.01 |
| D | 9.5 (10, 0.49) | 10.5 | 58.6 | 0.60 |
| E | 0.8 (6, 0.99) | 12.6 | 78.9 | <0.01 |
| Response | Predictor | Substrate | BS1 | BS2 | BS3 | ||||
|---|---|---|---|---|---|---|---|---|---|
| P | D2 | P | D2 | P | D2 | P | D2 | ||
| We.rest | Harvesting date | 0.04 | 3.2 | 0.80 | - | 0.054 | - | 0.50 | - |
| We.largest | Harvesting date | 0.72 | - | 0.053 | - | 0.38 | - | 0.19 | - |
| We.rest | <0.001 | 26.1 | 0.004 | 18.9 | <0.001 | 29.2 | 0.014 | 28.6 | |
| Shape | We.largest | <0.001 | 18.3 | 0.13 | - | 0.002 | 23.0 | 0.70 | - |
| We.rest | 0.93 | - | 0.92 | - | 0.049 | 2.2 | 0.58 | - | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia-Barreda, S.; Sánchez, S.; Marco, P.; Benucci, G.M.N.; González, V. Lack of Linkages among Fruiting Depth, Weight, and Maturity in Irrigated Truffle Fungi Marks the Complexity of Relationships among Morphogenetic Stages. J. Fungi 2021, 7, 102. https://doi.org/10.3390/jof7020102
Garcia-Barreda S, Sánchez S, Marco P, Benucci GMN, González V. Lack of Linkages among Fruiting Depth, Weight, and Maturity in Irrigated Truffle Fungi Marks the Complexity of Relationships among Morphogenetic Stages. Journal of Fungi. 2021; 7(2):102. https://doi.org/10.3390/jof7020102
Chicago/Turabian StyleGarcia-Barreda, Sergi, Sergio Sánchez, Pedro Marco, Gian Maria Niccolò Benucci, and Vicente González. 2021. "Lack of Linkages among Fruiting Depth, Weight, and Maturity in Irrigated Truffle Fungi Marks the Complexity of Relationships among Morphogenetic Stages" Journal of Fungi 7, no. 2: 102. https://doi.org/10.3390/jof7020102
APA StyleGarcia-Barreda, S., Sánchez, S., Marco, P., Benucci, G. M. N., & González, V. (2021). Lack of Linkages among Fruiting Depth, Weight, and Maturity in Irrigated Truffle Fungi Marks the Complexity of Relationships among Morphogenetic Stages. Journal of Fungi, 7(2), 102. https://doi.org/10.3390/jof7020102






