Quantifying the Role of Ground Beetles for the Dispersal of Fusarium and Alternaria Fungi in Agricultural Landscapes
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Sampling Design
2.3. Estimation of Exogenous Fungi (Culture-Dependent Method)
2.3.1. Quantity of Fungal Load
2.3.2. Quantity of Fusarium and Alternaria Species
2.4. Estimation of the Quantity of Endogenous and Exogenous Fungal DNA (qPCR)
2.4.1. Sample Preparation
2.4.2. Genomic DNA Extraction and qPCR
2.5. Statistical Analysis
3. Results
3.1. Exogenous Fungi (Culture-Dependent Method)
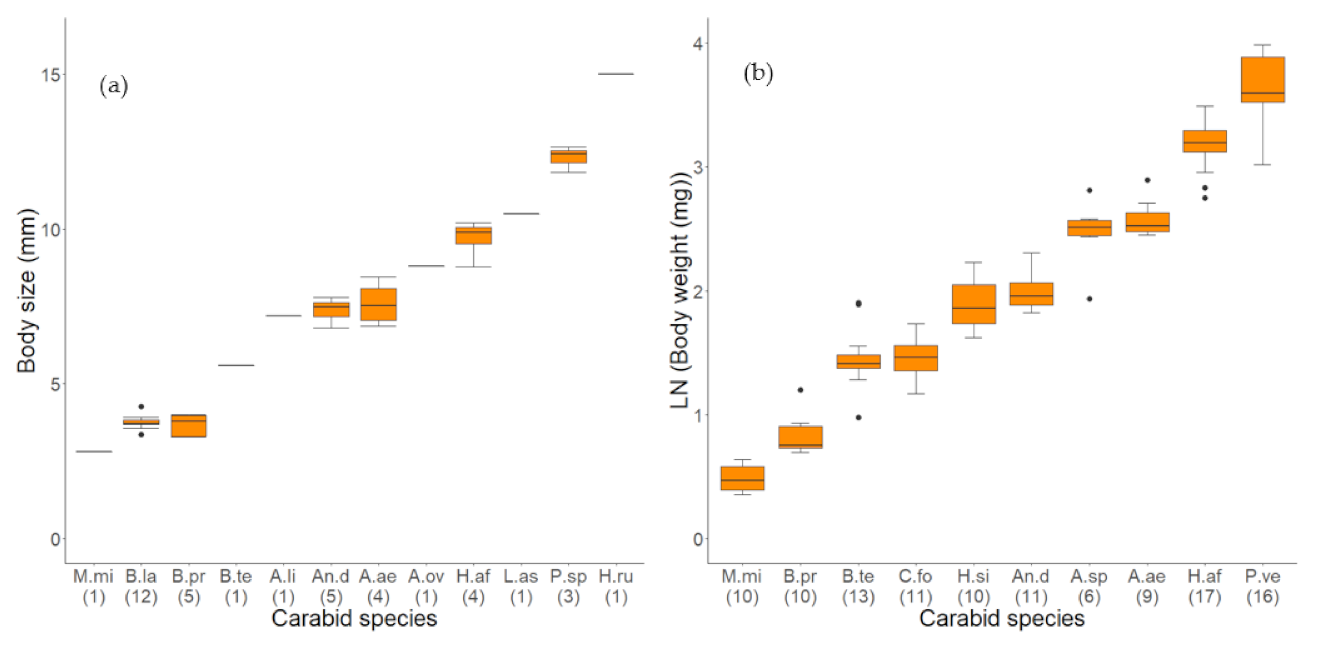
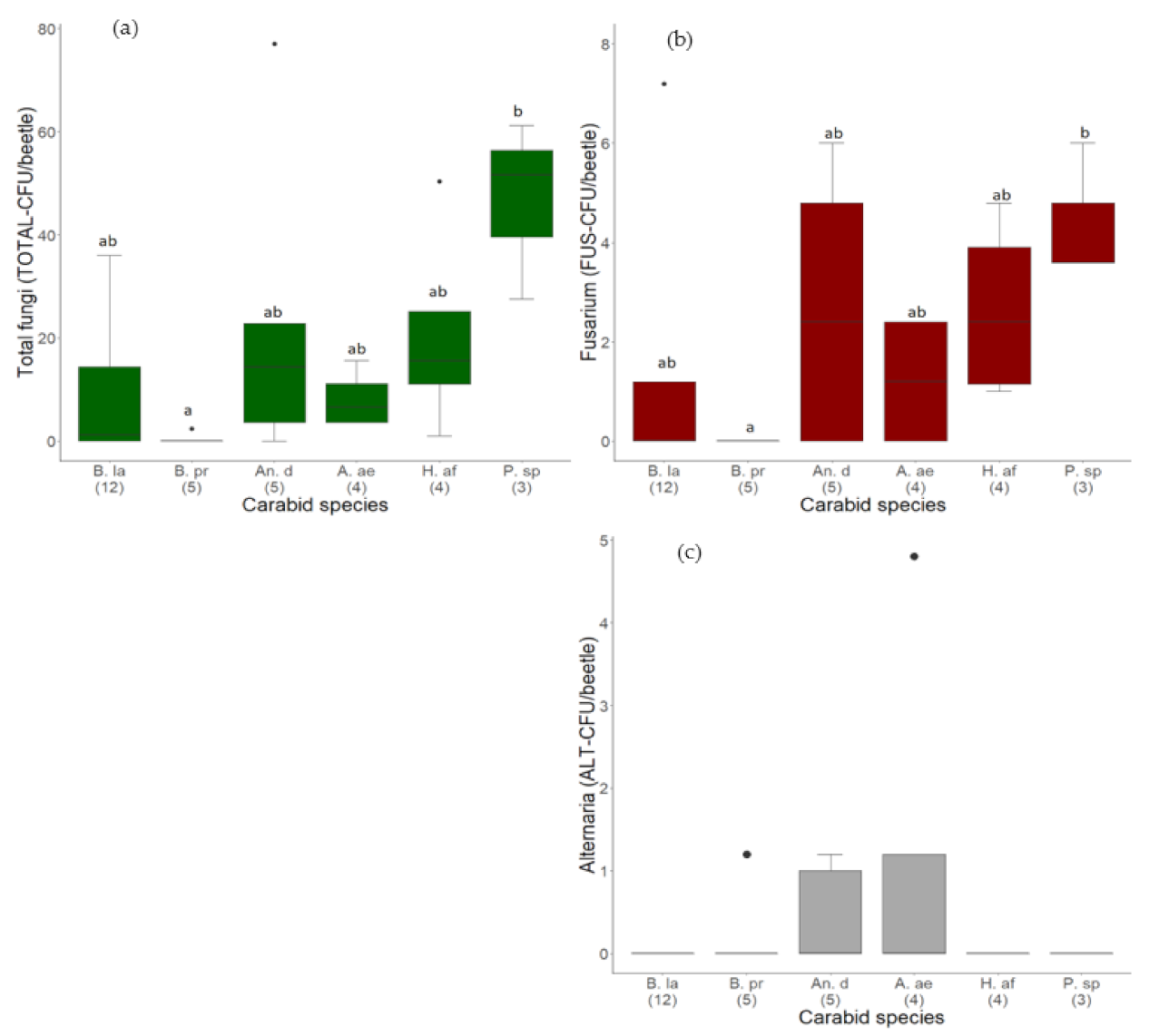
3.1.1. Quantity of Fungal Load on Different Carabid Species
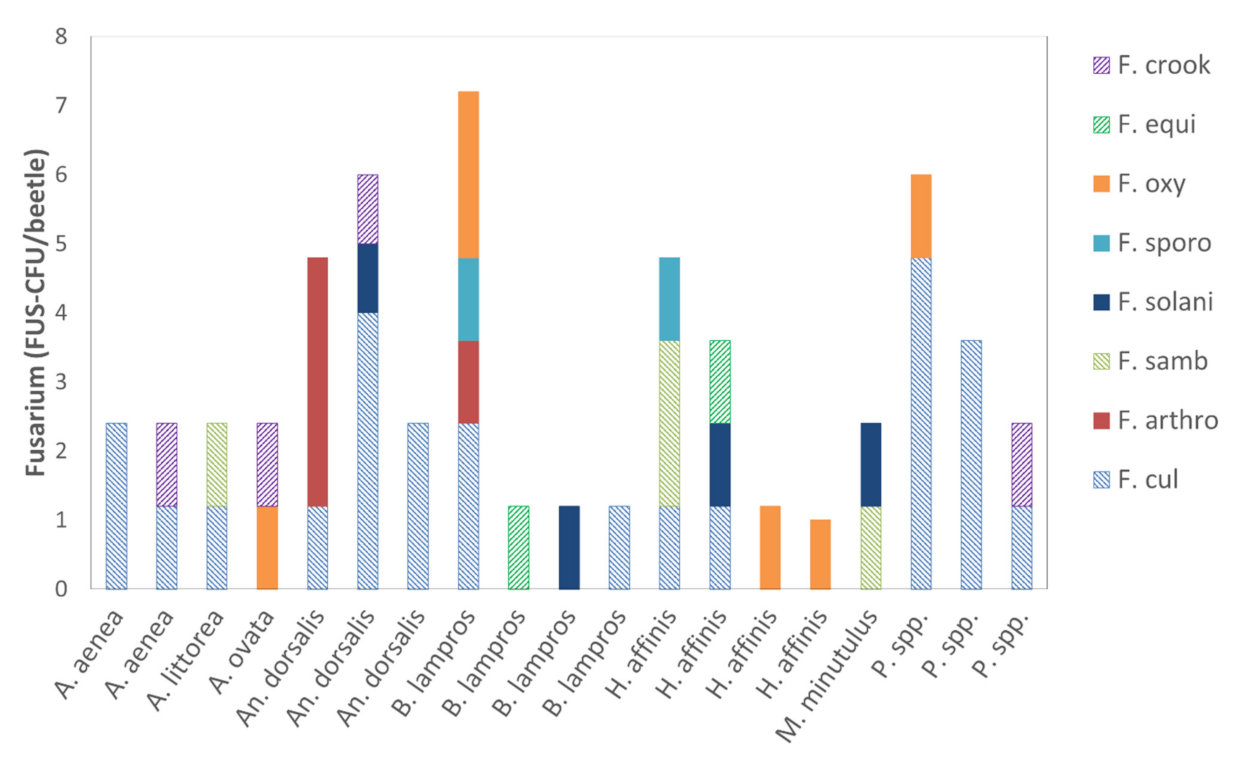
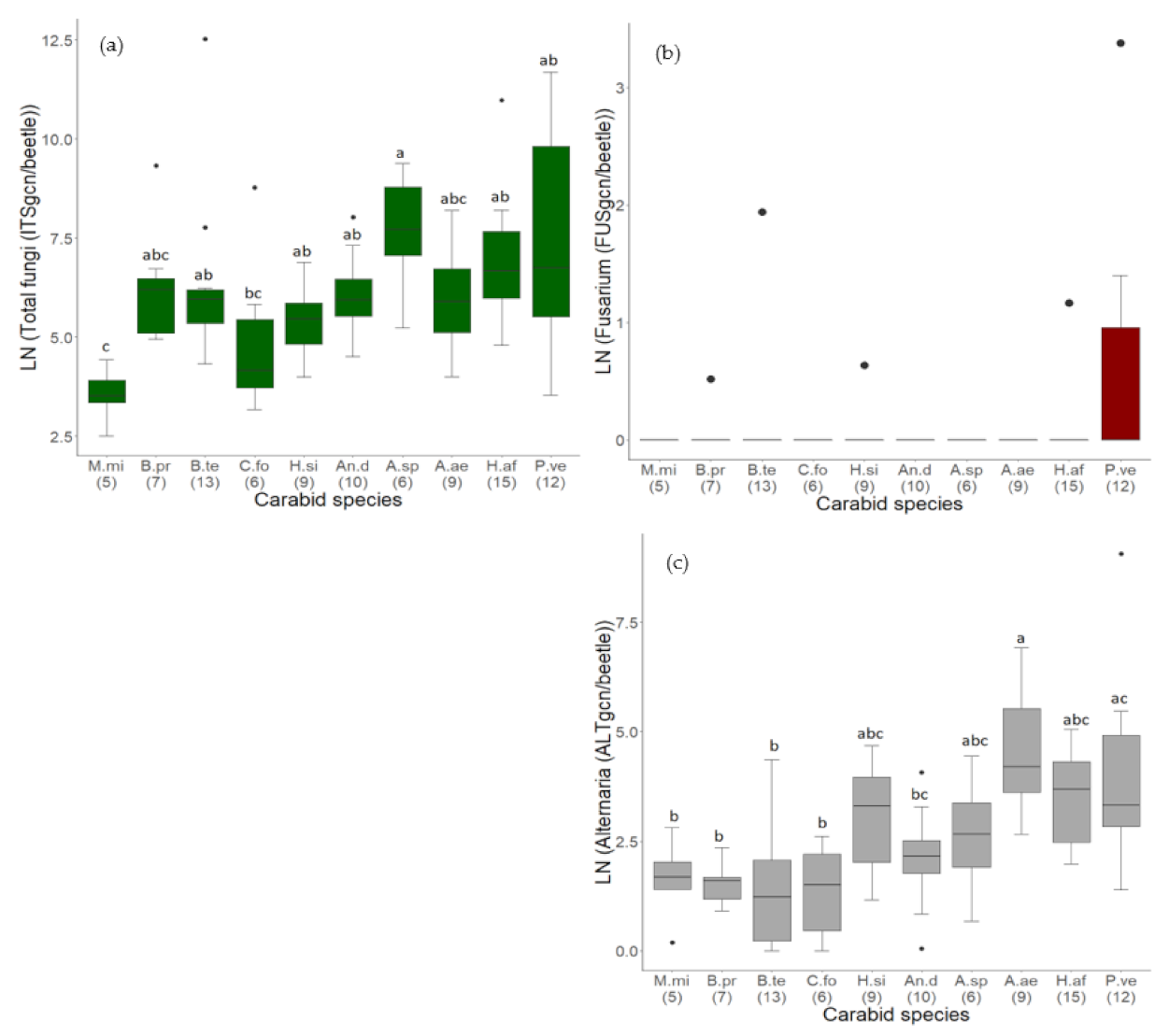
3.1.2. Quantity of Alternaria and Fusarium Species
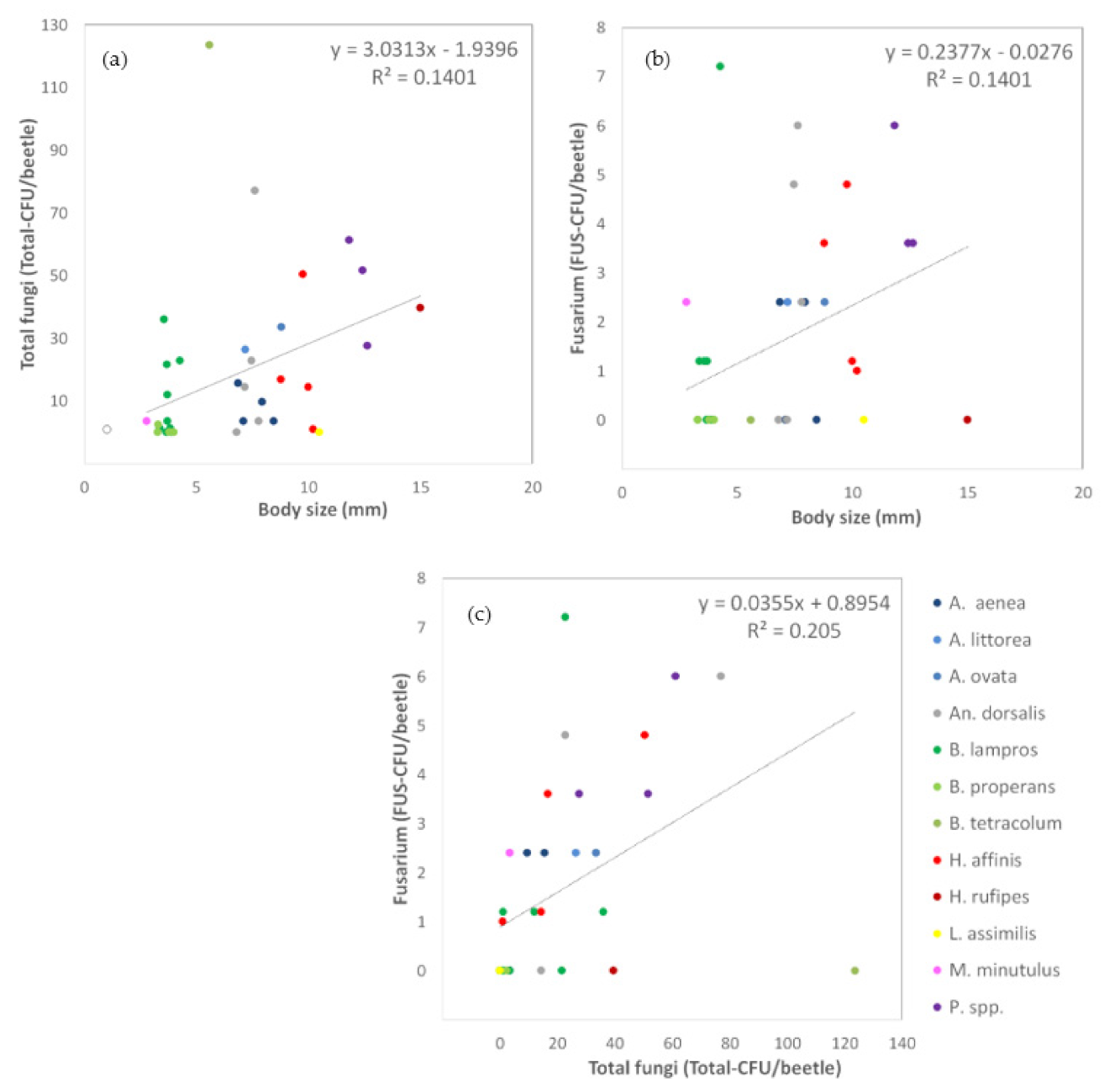
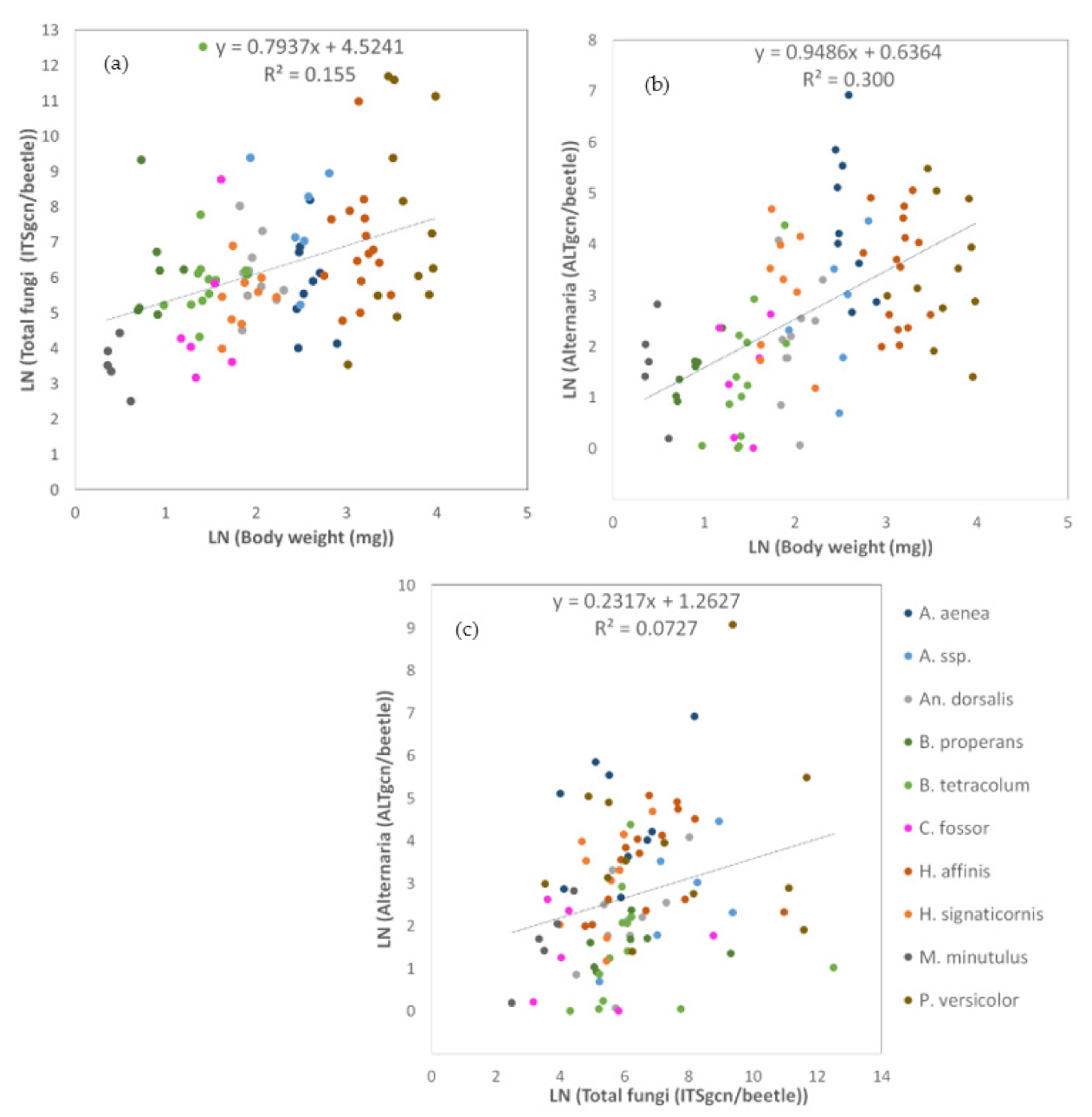
3.1.3. Relationships between Carabid Body Size and Number of Fungal Propagules
3.2. Quantification of Endogenous and Exogenous Fungal DNA (qPCR)
3.2.1. Comparison of Fungal Contamination between Carabid Species
3.2.2. Relationship between Carabid Body Weight and the Load of Endogenous and Exogenous Fungal DNA
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations and Definitions
| Al. | Alternaria |
| ALT-CFU/beetle | Load of Alternaria fungi per carabid beetle |
| ALTgcn/beetle | Alternaria DNA genome copy number per carabid beetle |
| CFU | Colony Forming Unit |
| F. | Fusarium |
| FUS-CFU/beetle | Load of Fusarium fungi per carabid beetle |
| FUSgcn/beetle | Fusarium DNA genome copy number per carabid beetle |
| gcn | genome copy number |
| ITS | Internal Transcriber Spacer |
| ITSgcn/beetle | Total fungal DNA genome copy number per carabid beetle |
| PDA | Potato dextrose agar |
| qPCR | real-time polymerase chain reaction |
| SNA | Synthetic Nutrient-Poor Agar |
| TOTAL-CFU/beetle | Total fungal number of colony forming units per carabid beetle |
| Fungal load | number of adhered or ingested fungal propagules or genomes, per individual or sample |
| Frequency | probability or number of times of the occurrence of a certain fungal genera or species |
| Abundance | Number of propagules or genomes of fungal genera or species per sample |
Carabid Species
| A. ae | Amara aenea |
| A. li | Amara littorea |
| A. ov | Amara ovata |
| A. sp | Amara spp. |
| A. | Amara |
| An. | Anchomenus |
| An. d | Anchomenus dorsalis |
| B. | Bembidion |
| B. la | Bembidion lampros |
| B. pr | Bembidion properans |
| B. te | Bembidion tetracolum |
| C. | Clivina |
| C. fo | Clivina fossor |
| Ca. | Carabus |
| Ca. a | Carabus auratus |
| H. | Harpalus |
| H. af | Harpalus affinis |
| H. ru | Harpalus rufipes |
| H. si | Harpalus signaticornis |
| L. | Limodromus |
| L. as | Limodromus assimilis |
| M. | Microlestes |
| M. mi | Microlestes minutulus |
| P. | Poecilus |
| P. sp | Poecilus spp. |
| P. ve | Poecilus versicolor |
References
- Sahin, B.; Kar, S.; Safak, O.N. Spitting type seed dispersal by domestic goat in the Zoochorial process of blackthorn plum. Ann. Bot. 2021, 11, 43–53. [Google Scholar] [CrossRef]
- Zona, S. Fruit and Seed Dispersal of Salvia L. (Lamiaceae): A Review of the Evidence. Bot. Rev. 2017, 83, 195–212. [Google Scholar] [CrossRef]
- Vašutová, M.; Mleczko, P.; López-García, A.; Maček, I.; Boros, G.; Ševčík, J.; Fujii, S.; Hackenberger, D.; Tuf, I.H.; Hornung, E.; et al. Taxi drivers: The role of animals in transporting mycorrhizal fungi. Mycorrhiza 2019, 29, 413–434. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.; Moberg, F. Mobile link organisms and ecosystem functioning: Implications for ecosystem resilience and management. Ecosystems 2003, 6, 87–98. [Google Scholar] [CrossRef]
- Moyo, P.; Allsopp, E.; Roets, F.; Mostert, L.; Halleen, F. Arthropods Vector Grapevine Trunk Disease Pathogens. Phytopathology 2014, 104, 1063–1069. [Google Scholar] [CrossRef] [PubMed]
- Feldman, T.S.; O’Brien, H.E.; Arnold, A.E. Moths that vector a plant pathogen also transport endophytic fungi and mycoparasitic antagonists. Microb. Ecol. 2008, 56, 742–750. [Google Scholar] [CrossRef] [PubMed]
- Heck, D.W.; Alves, G.; Mizubuti, E.S.G. Weevil Borers Affect the Spatio-Temporal Dynamics of Banana Fusarium Wilt. J. Fungi 2021, 7, 329. [Google Scholar] [CrossRef]
- Mayfield, A.E.; Peña, J.E.; Crane, J.H.; Smith, J.A.; Branch, C.L.; Ottoson, E.D.; Hughes, M. Ability of the redbay ambrosia beetle (Coleoptera: Curculionidae: Scolytinae) to bore into young avocado (Lauraceae) plants and transmit the laurel wilt pathogen (Raffaelea sp.). Fla. Entomol. 2008, 91, 485–487. [Google Scholar] [CrossRef]
- Kurtz, B.; Karlovsky, P.; Vidal, S. Interaction Between Western Corn Rootworm (Coleoptera: Chrysomelidae) Larvae and Root-Infecting Fusarium verticillioides. Environ. Entomol. 2010, 39, 1532–1538. [Google Scholar] [CrossRef]
- Sobek, E.A.; Munkvold, G.P. European corn borer (Lepidoptera: Pyralidae) larvae as vectors of Fusarium moniliforme, causing kernel rot and symptomless infection of maize kernels. J. Econ. Entomol. 1999, 92, 503–509. [Google Scholar] [CrossRef]
- Anslan, S.; Bahram, M.; Tedersoo, L. Temporal changes in fungal communities associated with guts and appendages of Collembola as based on culturing and high-throughput sequencing. Soil Biol. Biochem. 2016, 96, 152–159. [Google Scholar] [CrossRef]
- Mannino, M.C.; Huarte-Bonnet, C.; Davyt-Colo, B.; Pedrini, N. Is the Insect Cuticle the only Entry Gate for Fungal Infection? Insights into Alternative Modes of Action of Entomopathogenic Fungi. J. Fungi 2019, 5, 33. [Google Scholar] [CrossRef] [PubMed]
- Lilleskov, E.A.; Bruns, T.D. Spore dispersal of a resupinate ectomycorrhizal fungus, Tomentella sublilacina, via soil food webs. Mycologia 2005, 97, 762–769. [Google Scholar] [CrossRef] [PubMed]
- Dromph, K.M. Dispersal of entomopathogenic fungi by collembolans. Soil Biol. Biochem. 2001, 33, 2047–2051. [Google Scholar] [CrossRef]
- Guillen Sánchez, C.; Tixier, P.; Tapia Fernández, A.; Conejo Barboza, A.M.; Sandoval Fernández, J.A.; de Lapeyre de Bellaire, L. Can the banana weevil Cosmopolites sordidus be a vector of Fusarium oxysporum f.sp. cubense race 1? Unravelling the internal and external acquisition of effective inoculum. Pest Manag. Sci. 2021, 77, 3002–3012. [Google Scholar] [CrossRef]
- Jevtić, R.; Župunski, V.; Lalošević, M.; Živanov, S.T. Colonization of winter wheat grain with Fusarium and Alternaria species and influence on pest control management. J. Gen. Plant Pathol. 2019, 85, 273–281. [Google Scholar] [CrossRef]
- Bottalico, A.; Perrone, G. Toxigenic Fusarium species and mycotoxins associated with head blight in small-grain cereals in Europe. Eur. J. Plant Pathol. 2002, 108, 611–624. [Google Scholar] [CrossRef]
- Thomma, B.P.H.J. Alternaria spp.: From general saprophyte to specific parasite. Mol. Plant Pathol. 2003, 4, 225–236. [Google Scholar] [CrossRef]
- Leslie, J.F.; Summerell, B.A. The Fusarium Laboratory Manual, 1st ed.; Blackwell Publishing: Ames, IA, USA, 2016; ISBN 9780813819198. [Google Scholar]
- O’Donnell, K.; Humber, R.A.; Geiser, D.M.; Kang, S.; Park, B.; Robert, V.A.R.G.; Crous, P.W.; Johnston, P.R.; Aoki, T.; Rooney, A.P.; et al. Phylogenetic diversity of insecticolous fusaria inferred from multilocus DNA sequence data and their molecular identification via FUSARIUM-ID and Fusarium MLST. Mycologia 2012, 104, 427–445. [Google Scholar] [CrossRef] [PubMed]
- Sharma, L.; Marques, G. Fusarium, an Entomopathogen—A Myth or Reality? Pathogens 2018, 7, 93. [Google Scholar] [CrossRef]
- El-Hamalawi, Z.A.; Stanghellini, M.E. Disease Development on Lisianthus Following Aerial Transmission of Fusarium avenaceum by Adult Shore Flies, Fungus Gnats, and Moth Flies. Plant Dis. 2005, 89, 619–623. [Google Scholar] [CrossRef]
- Yun, T.S.; Park, S.Y.; Yu, J.; Hwang, Y.; Hong, K.J. Isolation and identification of fungal species from the insect pest Tribolium castaneum in rice processing complexes in Korea. Plant Pathol. J. 2018, 34, 356–366. [Google Scholar] [CrossRef]
- Guedes, F.L.A.; Attili-Angelis, D.; Pagnocca, F.C. Selective isolation of dematiaceous fungi from the workers of Atta laevigata (Formicidae: Attini). Folia Microbiol. 2012, 57, 21–26. [Google Scholar] [CrossRef]
- Hubert, J.; Stejskal, V.; Munzbergová, Z.; Kubátová, A.; Váňová, M.; Žd’árková, E. Mites and fungi in heavily infested stores in the Czech Republic. J. Econ. Entomol. 2004, 97, 2144–2153. [Google Scholar] [CrossRef] [PubMed]
- Suproniene, S.; Kadziene, G.; Irzykowski, W.; Sneideris, D.; Ivanauskas, A.; Sakalauskas, S.; Serbiak, P.; Svegzda, P.; Auskalniene, O.; Jedryczka, M. Weed species within cereal crop rotations can serve as alternative hosts for Fusarium graminearum causing Fusarium head blight of wheat. Fungal Ecol. 2019, 37, 30–37. [Google Scholar] [CrossRef]
- Landschoot, S.; Audenaert, K.; Waegeman, W.; Pycke, B.; Bekaert, B.; De Baets, B.; Haesaert, G. Connection between primary Fusarium inoculum on gramineous weeds, crop residues and soil samples and the final population on wheat ears in Flanders, Belgium. Crop Prot. 2011, 30, 1297–1305. [Google Scholar] [CrossRef]
- Postic, J.; Cosic, J.; Vrandecic, K.; Jurkovic, D.; Saleh, A.A.; Leslie, J.F. Diversity of Fusarium Species Isolated from Weeds and Plant Debris in Croatia. J. Phytopathol. 2012, 81, 76–81. [Google Scholar] [CrossRef]
- Kulkarni, S.S.; Dosdall, L.M.; Willenborg, C.J. The Role of Ground Beetles (Coleoptera: Carabidae) in Weed Seed Consumption: A Review. Weed Sci. 2015, 63, 355–376. [Google Scholar] [CrossRef]
- Lövei, G.L.; Sunderland, K.D. Ecology and behavior of ground beetles (Coleoptera: Carabidae). Annu. Rev. Entomol. 1996, 41, 231–256. [Google Scholar] [CrossRef]
- Wolters, V. Spiders, Carabids, and Staphylinids: The Ecological Potential of Predatory Macroarthropods. Fauna Soil Ecosyst. 2020, 321–376. [Google Scholar] [CrossRef]
- Honek, A.; Martinkova, Z.; Jarosik, V. Ground beetles (Carabidae) as seed predators. Eur. J. Entomol. 2003, 100, 531–544. [Google Scholar] [CrossRef]
- Kotze, J.; Brandmayr, P.; Casale, A.; Dauffy-Richard, E.; Dekoninck, W.; Koivula, M.J.; Lövei, G.L.; Mossakowski, D.; Noordijk, J.; Paarmann, W.; et al. Forty years of carabid beetle research in Europe—From taxonomy, biology, ecology and population studies to bioindication, habitat assessment and conservation. Zookeys 2011, 100, 55–148. [Google Scholar] [CrossRef] [PubMed]
- Holland, J.M. Carabid beetles: Their Ecology, Survival and use in Agroecosystems. In The Agroecology of Carabid Beetles; Holland, J.M., Ed.; Intercept Limited: Andover, UK, 2002; Volume 62, ISBN 1898298769. [Google Scholar]
- Schöpke, B.; Heinze, J.; Pätzig, M.; Heinken, T. Do dispersal traits of wetland plant species explain tolerance against isolation effects in naturally fragmented habitats? Plant Ecol. 2019, 8, 801–815. [Google Scholar] [CrossRef]
- Kalettka, T.; Rudat, C. Hydrogeomorphic types of glacially created kettle holes in North-East Germany. Limnologica 2006, 36, 54–64. [Google Scholar] [CrossRef]
- The Leibniz Centre for Agricultural Landscape Research (ZALF). AgroScapeLab Quillow (ASLQ). Available online: https://www.zalf.de/de/struktur/eip/Seiten/AgroScapeLab.aspx (accessed on 1 July 2021).
- Platen, R.; Kalettka, T.; Ulrichs, C. Kettle Holes in the Agrarian Landscape: Isolated and Ecological Unique Habitats for Carabid Beetles (Col.: Carabidae) and Spiders (Arach.: Araneae). J. Landsc. Ecol. 2016, 9, 29–60. [Google Scholar] [CrossRef]
- Lozada-Gobilard, S.; Landivar Albis, C.M.; Rupik, K.B.; Pätzig, M.; Hausmann, S.; Tiedemann, R.; Joshi, J. Habitat quality and connectivity in kettle holes enhance bee diversity in agricultural landscapes. Agric. Ecosyst. Environ. 2021, 319, 107525. [Google Scholar] [CrossRef]
- Müller-Motzfeld, G. Adephaga 1; Carbidae (Laufkäfer). Die Käfer Mitteleuropas, 2nd ed.; Spektrum Verlag: Berlin/Heidelberg, Germany, 2004; ISBN 3827415519. [Google Scholar]
- Nierenberg, H. Untersuchungen über die Morphologische und Biologische Differenzierung in der Fusarium-Sektion Liseola; Kommisionsverlag Paul Parey: Berlin, Germany, 1976. [Google Scholar]
- Yli-Mattila, T.; Hussien, T.; Gavrilova, O.; Gagkaeva, T. Morphological and molecular variation between Fusarium avenaceum, Fusarium arthrosporioides and Fusarium anguioides strains. Pathogens 2018, 7, 94. [Google Scholar] [CrossRef]
- Simmons, G.E. Alternaria Taxonomy: Current Status, Viewpoint, Challenge; Elsevier: Amsterdam, The Netherlands, 1992. [Google Scholar]
- Kahl, S.M.; Ulrich, A.; Kirichenko, A.A.; Müller, M.E.H. Phenotypic and phylogenetic segregation of Alternaria infectoria from small-spored Alternaria species isolated from wheat in Germany and Russia. J. Appl. Microbiol. 2015, 119, 1637–1650. [Google Scholar] [CrossRef]
- Gardes, M.; Bruns, T.D. ITS primers with enhanced specificity for basidiomycetes—Application to the identification of mycorrhizae and rusts. Mol. Ecol. 1993, 2, 113–118. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.D.; Lee, S.J.W.T.; Taylor, J.W. Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. In PCR Protocols—A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Müller, T.; Ruppel, S.; Behrendt, U.; Lentzsch, P.; Müller, M.E.H. Antagonistic Potential of Fluorescent Pseudomonads Colonizing Wheat Heads Against Mycotoxin Producing Alternaria and Fusaria. Front. Microbiol. 2018, 9, 1–14. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Zar, J.H. Biostatistical Analysis; Prentice Hall: Upper Saddle River, NJ, USA, 1989. [Google Scholar]
- Engel, P.; Moran, N.A. The gut microbiota of insects—Diversity in structure and function. FEMS Microbiol. Rev. 2013, 37, 699–735. [Google Scholar] [CrossRef] [PubMed]
- Višňovska, D.; Pyszko, P.; Šigut, M.; Kostovčik, M.; Kolařik, M.; Kotaskova, N.; Drozd, P. Caterpillar gut and host plant phylloplane mycobiomes differ: A new perspective on fungal involvement in insect guts. FEMS Microbiol. Ecol. 2020, 96. [Google Scholar] [CrossRef] [PubMed]
- Platen, R. Database for the ecology of ground beetles of Germany. Leibniz-Center for Agricultural Landscape Research (ZALF), Working group: Provisioning Biodiversity in Agricultural Systems. 2021; unpublished. [Google Scholar]
- Stefanini, I. Yeast-insect associations: It takes guts. Yeast 2018, 35, 315–330. [Google Scholar] [CrossRef]
- Moubasher, A.H.; Abdel-Sater, M.A.; Soliman, Z. Yeasts and filamentous fungi inhabiting guts of three insect species in Assiut, Egypt. Mycosphere 2017, 8, 1297–1316. [Google Scholar] [CrossRef]
- Guo, Z.; Pfohl, K.; Karlovsky, P.; Dehne, H.-W.; Altincicek, B. Dissemination of Fusarium proliferatum by mealworm beetle Tenebrio molitor. PLoS ONE 2018, 13, e0204602. [Google Scholar] [CrossRef]
- Bezos, D.; Martínez-Álvarez, P.; Sanz-Ros, A.V.; Martín-García, J.; Fernandez, M.M.; Diez, J.J. Fungal communities associated with bark beetles in Pinus radiata plantations in Northern Spain affected by pine pitch canker, with special focus on Fusarium species. Forests 2018, 9, 698. [Google Scholar] [CrossRef]
- Blodgett, J.T.; Swart, W.J.; Louw, S.V.M. Identification of fungi and fungal pathogens associated with Hypolixus haerens and decayed and cankered stems of Amaranthus hybridus. Plant Dis. 2004, 88, 333–337. [Google Scholar] [CrossRef] [PubMed][Green Version]
- El-Hamalawi, Z.A. Acquisition, retention and dispersal of soilborne plant pathogenic fungi by fungus gnats and moth flies. Ann. Appl. Biol. 2008, 153, 195–203. [Google Scholar] [CrossRef]
- Dillard, H.R.; Cobb, A.C.; Lamboy, J.S. Transmission of Alternaria brassicicola to Cabbage by Flea Beetles (Phyllotreta cruciferae). Plant Dis. 1998, 82, 153–157. [Google Scholar] [CrossRef]
- Meldrum, R.A.; Daly, A.M.; Tran-Nguyen, L.T.T.; Aitken, E.A.B. Are banana weevil borers a vector in spreading Fusarium oxysporum f. sp. cubense tropical race 4 in banana plantations? Australas. Plant Pathol. 2013, 42, 543–549. [Google Scholar] [CrossRef]
- Holland, J.M.; Thomas, C.F.G.; Birkett, T.; Southway, S. Spatio-temporal distribution and emergence of beetles in arable fields in relation to soil moisture. Bull. Entomol. Res. 2007, 97, 89. [Google Scholar] [CrossRef]
- Baars, A.M.A. Patterns of Movement of Radioactive Carabid Beetles. Oecologia 1979, 44, 125–140. [Google Scholar] [CrossRef]
- Ranjha, M.H.; Irmler, U. Movement of carabids from grassy strips to crop land in organic agriculture. J. Insect Conserv. 2014, 18, 457–467. [Google Scholar] [CrossRef]
- De Lucca, A.J. Harmful fungi in both Agriculture and Medicine. Rev. Iberoam. Micol. 2007, 24, 3–13. [Google Scholar] [CrossRef]
- Kluth, S.; Kruess, A.; Tscharntke, T. Insects as vectors of plant pathogens: Mutualistic and antagonistic interactions. Oecologia 2002, 133, 193–199. [Google Scholar] [CrossRef]
- Drakulic, J.; Caulfield, J.; Woodcock, C.; Jones, S.P.T.; Linforth, R.; Bruce, T.J.A.; Ray, V. Sharing a Host Plant (Wheat [Triticum aestivum]) Increases the Fitness of Fusarium graminearum and the Severity of Fusarium Head Blight but Reduces the Fitness of Grain Aphids (Sitobion avenae). Appl. Environ. Microbiol. 2015, 81, 3492–3501. [Google Scholar] [CrossRef] [PubMed]
- Vaquera, S.; Patriarca, A.; Fernández Pinto, V. Water activity and temperature effects on growth of Alternaria arborescens on tomato medium. Int. J. Food Microbiol. 2014, 185, 136–139. [Google Scholar] [CrossRef]
- Fernandes, C.; Mota, M.; Barros, L.; Dias, M.I.; Ferreira, I.C.F.R.; Piedade, A.P.; Casadevall, A.; Gonçalves, T. Pyomelanin Synthesis in Alternaria alternata Inhibits DHN-Melanin Synthesis and Decreases Cell Wall Chitin Content and Thickness. Front. Microbiol. 2021, 12, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Boucias, D.G.; Pendland, J.C. Attachment of Mycopathogens to Cuticle The Initial Event of Mycoses in Arthropod Hosts. In The Fungal Spore and Disease Initiation in Plants and Animals; Cole, G.T., Hoch, H.C., Eds.; Springer Science + Buisness Media, LLC: New York, NY, USA, 1991; pp. 101–103. ISBN 9781489926371. [Google Scholar]
- Bartelt, R.J.; Wicklow, D.T. Volatiles from Fusarium verticillioides (Sacc.) Nirenb. and Their Attractiveness to Nitidulid Beetles. J. Agric. Food Chem. 1999, 47, 2447–2454. [Google Scholar] [CrossRef] [PubMed]
- Oster, M.; Smith, L.; Beck, J.J.; Howard, A.; Field, C.B. Orientation behavior of predaceous ground beetle species in response to volatile emissions identified from yellow starthistle damaged by an invasive slug. Arthropod. Plant. Interact. 2014, 8, 429–437. [Google Scholar] [CrossRef]
- Parry, D.W.; Jenkinson, P.; McLeod, L. Fusarium ear blight (scab) in small grain cereals?a review. Plant Pathol. 1995, 44, 207–238. [Google Scholar] [CrossRef]
- Champeil, A.; Doré, T.; Fourbet, J. Fusarium head blight: Epidemiological origin of the effects of cultural practices on head blight attacks and the production of mycotoxins by Fusarium in wheat grains. Plant Sci. 2004, 166, 1389–1415. [Google Scholar] [CrossRef]
- Logrieco, A.; Moretti, A.; Solfrizzo, M. Alternaria toxins and plant diseases: An overview of origin, occurrence and risks. World Mycotoxin J. 2009, 2, 129–140. [Google Scholar] [CrossRef]
- Liggitt, J.; Jenkinson, P.; Parry, D.W. The role of saprophytic microflora in the development of Fusarium ear blight of winter wheat caused by Fusarium culmorum. Crop Prot. 1997, 16, 679–685. [Google Scholar] [CrossRef]
- Lee, H.B.; Patriarca, A.; Magan, N. Alternaria in Food: Ecophysiology, Mycotoxin Production and Toxicology. Mycobiology 2015, 43, 93–106. [Google Scholar] [CrossRef] [PubMed]
- Schiro, G.; Verch, G.; Grimm, V.; Müller, M.E.H. Alternaria and Fusarium Fungi: Differences in Distribution and Spore Deposition in a Topographically Heterogeneous Wheat Field. J. Fungi 2018, 4, 63. [Google Scholar] [CrossRef]
- Leplat, J.; Friberg, H.; Abid, M.; Steinberg, C. Survival of Fusarium graminearum, the causal agent of Fusarium head blight. A review. Agron. Sustain. Dev. 2013, 33, 97–111. [Google Scholar] [CrossRef]
- Müller, M.E.H.; Steier, I.; Köppen, R.; Siegel, D.; Proske, M.; Korn, U.; Koch, M. Cocultivation of phytopathogenic Fusarium and Alternaria strains affects fungal growth and mycotoxin production. J. Appl. Microbiol. 2012, 113, 874–887. [Google Scholar] [CrossRef]
- Hoffmann, A.; Lischeid, G.; Koch, M.; Lentzsch, P.; Sommerfeld, T.; Müller, M.E.H. Co-Cultivation of Fusarium, Alternaria, and Pseudomonas on Wheat-Ears Affects Microbial Growth and Mycotoxin Production. Microorganisms 2021, 9, 443. [Google Scholar] [CrossRef]
- Yamoah, E.; Jones, E.E.; Suckling, D.M.; Bourdôt, G.W.; Walter, M.; Stewart, A. Using insects as potential vectors of Fusarium tumidum to control gorse. N. Z. Entomol. 2011, 34, 5–11. [Google Scholar] [CrossRef]
- Perelló, A.; Moreno, M.; Sisterna, M. Alternaria infectoria species-group associated with black point of wheat in Argentina. Plant Pathol. 2008, 57, 379. [Google Scholar] [CrossRef]
| Abbreviation | Abbreviation | N | Median | |
|---|---|---|---|---|
| Carabid Species | Long | Short | Indiv. | (IQR) |
| Amara aenea | A. aenea | A. ae | 4 | 7.52 (1.03) |
| Amara littorea | A. littorea | A. li | 1 | 7.37 (NA) |
| Amara ovata | A. ovata | A. ov | 1 | 7.19 (NA) |
| Anchomenus dorsalis | An. dorsalis | An. d | 5 | 7.47 (0.46) |
| Bembidion lampros | B. lampros | B. la | 12 | 3.72 (0.14) |
| Bembidion properans | B. properans | B. pr | 5 | 3.79 (0.67) |
| Harpalus affinis | H. affinis | H. af | 4 | 9.89 (0.53) |
| Harpalus rufipes | H. rufipes | H. ru | 1 | 15.00 (NA) |
| Limodromus assimilis | L. assimilis | L. as | 1 | 10.49 (NA) |
| Microlestes minutulus | M. minutulus | M. mi | 1 | 2.80 (NA) |
| Poecilusspp. | P.spp. | P. sp | 3 | 12.42 (0.41) |
| Carabid. | Abbreviation | Abbreviation | Extracted | Weight (mg) | Extracted Samp. |
|---|---|---|---|---|---|
| Species | Long | Short | Indiv. | Median (IQR) | (Ø Indiv./Samp.) |
| Amara aenea | A. aenea | A. ae | 10 | 11.50 (1.96) | 9 (1.11) |
| Amara spp. | A. spp. | A. sp | 6 | 11.32 (1.49) | 6 (1.0) |
| Anchomenus dorsalis | An. dorsalis | An. D | 11 | 5.96 (1.40) | 11 (1.0) |
| Bembidion properans | B. properans | B. pr | 43 | 1.47 (0.46) | 10 (4.3) |
| Bembidion tetracolum | B. tetracolum | B. te | 30 | 3.11 (0.80) | 13 (2.31) |
| Clivina fossor | C. fossor | C. fo | 13 | 3.24 (1.29) | 11 (1.18) |
| Harpalus affinis | H. affinis | H. af | 17 | 22.62 (3.54) | 17 (1.0) |
| Harpalus signaticornis | H. signaticornis | H. si | 11 | 5.31 (1.91) | 10 (1.1) |
| Microlestes minutulus | M. minutulus | M. mi | 21 | 0.49 (0.20) | 10 (2.1) |
| Poecilus versicolor | P. versicolor | P. ve | 16 | 35.34 (17.33) | 16 (1.0) |
| Carabid Species | DNA Con. in µg per Extracted Indiv. (Median (IQR) | A260/280 per Extracted Indiv. (Median (IQR) | Samples Included in qPCR (Samples Excluded) |
|---|---|---|---|
| A. aenea | 3152.25 (2269.13) | 1.9 (0.04) | 9 (0) |
| A. spp. | 3513.37 (858.94) | 1.87 (0.02) | 6 (0) |
| An. dorsalis | 2241.00 (924.75) | 1.85 (0.04) | 10 (1) |
| B. properans | 509.63 (438.41) | 1.85 (0.11) | 7 (3) |
| B. tetracolum | 1205.25 (626.63) | 1.85 (0.02) | 13 (0) |
| C. fossor | 518.63 (333.00) | 1.83 (0.18) | 6 (5) |
| H. affinis | 3924.00 (2580.75) | 1.87 (0.05) | 15 (2) |
| H. signaticornis | 2012.63 (1426.5) | 184 (0.07) | 9 (1) |
| M. minutulus | 358.69 (312.47) | 1.79 (0.11) | 5 (5) |
| P. versicolor | 2948.63 (2817.00) | 1.87 (0.13) | 12 (4) |
| Criteria | Culture (N = 39) | qPCR (N = 92) |
|---|---|---|
| % of Positive Carabids * | ||
| Total fungi | 74.4% | 100% |
| Fusarium | 48.7% | 8.7% |
| Alternaria | 12.8% | 97.8% |
| Relationships (rs) between size/weight and | ||
| Total fungi (range) | 0.44 | 0.47 |
| Fusarium | 0.43 | n.s. |
| Alternaria | n.s. | 0.58 |
| Pattern of carabid species-specific differences Large/heavy > small/light | N = 33 | N = 92 |
| Total fungi | 2 groups, largest species has highest fungal load | 3 groups, slightly increased fungal load in smaller and midsized species, large intraspecific variance |
| Fusarium | 2 groups, midsized species slightly increased fungal load | N. s., very low frequency, |
| Alternaria | N.s., very low frequency | 3 groups, a medium-sized species with very high fungal load |
| Fungi Species | Number of CFU | Conidia Type |
|---|---|---|
| F. culmorum | 28.0 | Macro |
| F. sambucinum | 5.8 | Macro |
| F. crookwellense | 4.6 | Macro |
| F. equiseti | 2.4 | Macro |
| F. oxysporum | 7.0 | Micro |
| F. arthrosporioides | 4.8 | Micro |
| F. solani | 4.6 | Micro |
| F. sporotrichioides | 2.4 | Micro |
| Sum | 40.8 | Macro |
| Sum | 18.8 | Micro |
| Toxin amount | ||
| Al. infectoria | 4.6 | low |
| Al. tenuissima | 2.4 | high |
| Al. alternata | 1.2 | high |
| Al. arborescence | 1.2 | high |
| Sum | 4.6 | Low |
| Sum | 4.8 | high |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heitmann, N.; Glemnitz, M.; Lentzsch, P.; Platen, R.; Müller, M.E.H. Quantifying the Role of Ground Beetles for the Dispersal of Fusarium and Alternaria Fungi in Agricultural Landscapes. J. Fungi 2021, 7, 863. https://doi.org/10.3390/jof7100863
Heitmann N, Glemnitz M, Lentzsch P, Platen R, Müller MEH. Quantifying the Role of Ground Beetles for the Dispersal of Fusarium and Alternaria Fungi in Agricultural Landscapes. Journal of Fungi. 2021; 7(10):863. https://doi.org/10.3390/jof7100863
Chicago/Turabian StyleHeitmann, Nadja, Michael Glemnitz, Peter Lentzsch, Ralph Platen, and Marina E. H. Müller. 2021. "Quantifying the Role of Ground Beetles for the Dispersal of Fusarium and Alternaria Fungi in Agricultural Landscapes" Journal of Fungi 7, no. 10: 863. https://doi.org/10.3390/jof7100863
APA StyleHeitmann, N., Glemnitz, M., Lentzsch, P., Platen, R., & Müller, M. E. H. (2021). Quantifying the Role of Ground Beetles for the Dispersal of Fusarium and Alternaria Fungi in Agricultural Landscapes. Journal of Fungi, 7(10), 863. https://doi.org/10.3390/jof7100863






