On the Fly: Tritrophic Associations of Bats, Bat Flies, and Fungi
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Association Web
3.2. Structural Specificity
3.3. Ecomorphology
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bowler, D.E.; Bjorkman, A.D.; Dornelas, M.; Myers-Smith, I.H.; Navarro, L.M.; Niamir, A.; Supp, S.R.; Waldock, C.; Vellend, M.; Blowes, S.A.; et al. Mapping human pressures across the planet uncovers anthropogenic threat complexes. People Nat. 2020, 2, 380–394. [Google Scholar] [CrossRef]
- Marcogliese, D.J. Parasites of the superorganism: Are they indicators of ecosystem health? Int. J. Parasitol. 2005, 35, 705–716. [Google Scholar] [CrossRef]
- Carlson, C.J.; Hopkins, S.; Bell, K.C.; Doña, J.; Godfrey, S.S.; Kwak, M.L.; Lafferty, K.D.; Moir, M.L.; Speer, K.A.; Strona, G.; et al. A global parasite conservation plan. Biol. Conserv. 2020, 250, 108596. [Google Scholar] [CrossRef]
- Windsor, D.A. Most of the species on Earth are parasites. Int. J. Parasitol. 1998, 28, 1939–1941. [Google Scholar] [CrossRef]
- Carlson, C.J.; Burgio, K.R.; Dougherty, E.R.; Phillips, A.J.; Bueno, V.M.; Clements, C.F.; Castaldo, G.; Dallas, T.A.; Cizauskas, C.A.; Cumming, G.S.; et al. Parasite biodiversity faces extinction and redistribution in a changing climate. Sci. Adv. 2017, 3, e1602422. [Google Scholar] [CrossRef] [PubMed]
- Poulin, R.; Morand, S. The diversity of parasites. Q. Rev. Biol. 2000, 75, 277–293. [Google Scholar] [CrossRef] [PubMed]
- Poulin, R.; Randhawa, H.S. Evolution of parasitism along convergent lines: From ecology to genomics. Parasitology 2015, 142, S6–S15. [Google Scholar] [CrossRef] [PubMed]
- de Baets, K.; Littlewood, D.T.J. The importance of fossils in understanding the evolution of parasites and their vectors. Adv. Parasitol. 2015, 90, 1–51. [Google Scholar] [CrossRef]
- Lafferty, K.D.; Allesina, S.; Arim, M.; Briggs, C.J.; de Leo, G.; Dobson, A.P.; Dunne, J.A.; Johnson, P.T.J.; Kuris, A.M.; Marcogliese, D.J.; et al. Parasites in food webs: The ultimate missing links. Ecol. Lett. 2008, 11, 533–546. [Google Scholar] [CrossRef]
- Sato, T.; Egusa, T.; Fukushima, K.; Oda, T.; Ohte, N.; Tokuchi, N.; Watanabe, K.; Kanaiwa, M.; Murakami, I.; Lafferty, K.D. Nematomorph parasites indirectly alter the food web and ecosystem function of streams through behavioural manipulation of their cricket hosts. Ecol. Lett. 2012, 15, 786–793. [Google Scholar] [CrossRef]
- Brearley, G.; Rhodes, J.; Bradley, A.; Baxter, G.; Seabrook, L.; Lunney, D.; Liu, Y.; Mcalpine, C. Wildlife disease prevalence in human-modified landscapes. Biol. Rev. 2013, 88, 427–442. [Google Scholar] [CrossRef] [PubMed]
- Colwell, R.K.; Dunn, R.R.; Harris, N.C. Coextinction and persistence of dependent species in a changing world. Ann. Rev. Ecol. Evol. Syst. 2012, 43, 183–203. [Google Scholar] [CrossRef]
- Dunn, R.R.; Harris, N.C.; Colwell, R.K.; Koh, L.P.; Sodhi, N.S. The sixth mass coextinction: Are most endangered species parasites and mutualists? Proc. R. Soc. B 2009, 276, 3037–3045. [Google Scholar] [CrossRef] [PubMed]
- Strona, G. Past, present and future of host–parasite co-extinctions. Int. J. Parasitol Parasites Wildl. 2015, 4, 431–441. [Google Scholar] [CrossRef]
- Poulin, R.; Krasnov, B.R.; Morand, S. Patterns of host specificity in parasites exploiting small mammals. In Micromammals and Macroparasites—from Evolutionary Ecology to Management; Morand, S., Krasnov, B.R., Poulin, R., Eds.; Springer: Tokyo, Japan, 2006; pp. 233–256. [Google Scholar]
- Esser, H.J.; Herre, E.A.; Blüthgen, N.; Loaiza, J.R.; Bermúdez, S.E.; Jansen, P.A. Host specificity in a diverse Neotropical tick community: An assessment using quantitative network analysis and host phylogeny. Parasites Vectors 2016, 9, 372. [Google Scholar] [CrossRef]
- Holt, R.D.; Dobson, A.P.; Begon, M.; Bowers, R.G.; Schauber, E.M. Parasite establishment in host communities. Ecol. Lett. 2003, 6, 837–842. [Google Scholar] [CrossRef]
- Koh, L.P.; Dunn, R.R.; Sodhi, N.S.; Colwell, R.K.; Proctor, H.C.; Smith, V.S. Species coextinctions and the biodiversity crisis. Science 2004, 305, 1632–1634. [Google Scholar] [CrossRef]
- Poulin, R.; Krasnov, B.R.; Mouillot, D. Host specificity in phylogenetic and geographic space. Trends Parasitol. 2011, 27, 355–361. [Google Scholar] [CrossRef]
- Parratt, S.R.; Laine, A.-L. The role of hyperparasitism in microbial pathogen ecology and evolution. ISME J. 2016, 10, 1815–1822. [Google Scholar] [CrossRef]
- Parratt, S.R.; Barrès, B.; Penczykowski, R.M.; Laine, A.L. Local adaptation at higher trophic levels: Contrasting hyperparasite–pathogen infection dynamics in the field and laboratory. Mol. Ecol. 2017, 26, 1964–1979. [Google Scholar] [CrossRef]
- Haelewaters, D.; Page, R.A.; Pfister, D.H. Laboulbeniales hyperparasites (Fungi, Ascomycota) of bat flies: Independent origins and host associations. Ecol. Evol. 2018, 8, 8396–8418. [Google Scholar] [CrossRef] [PubMed]
- Gleason, F.H.; Lilje, O.; Marano, A.V.; Sime-Ngando, T.; Sullivan, B.K.; Kirchmair, M.; Neuhauser, S. Ecological functions of zoosporic hyperparasites. Front. Microbiol. 2014, 5, 244. [Google Scholar] [CrossRef] [PubMed]
- Holt, R.D.; Hochberg, M.E. The coexistence of competing parasites. Part II - Hyperparasitism and food chain dynamics. J. Theor. Biol. 1998, 193, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Simmons, N.B.; Cirranello, A.L. Bat Species of the World: A taxonomic and geographic database. Available online: https://batnames.org (accessed on 6 November 2020).
- Patterson, B.D.; Dick, C.W.; Dittmar, K. Roosting habits of bats affect their parasitism by bat flies (Diptera: Streblidae). J. Trop. Ecol. 2007, 23, 177–189. [Google Scholar] [CrossRef]
- Gannon, M.R.; Bovard, B.N. The value of bats: Keystone species in the Keystone State. In Conservation and Ecology of Pennsylvania’s Bats; Butchkoski, C.M., Reeder, D.M., Turner, G.G., Whidden, H.P., Eds.; The Pennsylvania Academy of Science: Allentown, PA, USA, 2016; pp. 5–31. [Google Scholar]
- Maine, J.J.; Boyles, J.G. Bats initiate vital agroecological interactions in corn. Proc. Natl. Acad. Sci. USA 2015, 112, 12438–12443. [Google Scholar] [CrossRef]
- International Union for Conservation of Nature. The IUCN Red List of Threatened Species. Version 2020-2. Available online: https://www.iucnredlist.org (accessed on 28 October 2020).
- Frick, W.F.; Kingston, T.; Flanders, J. A review of the major threats and challenges to global bat conservation. Ann. N. Y. Acad. Sci. 2019, 5–25. [Google Scholar] [CrossRef]
- Naegle, M.A.; Mugleston, J.D.; Bybee, S.M.; Whiting, M.F. Reassessing the phylogenetic position of the epizoic earwigs (Insecta: Dermaptera). Mol. Phylogenet. Evol. 2016, 100, 382–390. [Google Scholar] [CrossRef]
- Reeves, W.K.; Beck, J.; Orlova, M.V.; Daly, J.L.; Pippin, K.; Revan, F.; Loftis, A.D. Ecology of bats, their ectoparasites, and associated pathogens on Saint Kitts Island. J. Med. Entomol. 2016, 53, 1218–1225. [Google Scholar] [CrossRef]
- Haelewaters, D.; Hiller, T.; Dick, C.W. Bats, bat flies, and fungi: A case of hyperparasitism. Trends Parasitol. 2018, 34, 784–799. [Google Scholar] [CrossRef]
- Dick, C.W.; Patterson, B.D. Against all odds: Explaining high host specificity in dispersal-prone parasites. Int. J. Parasitol. 2007, 37, 871–876. [Google Scholar] [CrossRef]
- Dittmar, K.; Porter, M.L.; Murray, S.; Whiting, M.F. Molecular phylogenetic analysis of nycteribiid and streblid bat flies (Diptera: Brachycera, Calyptratae): Implications for host associations and phylogeographic origins. Mol. Phylogenet. Evol. 2006, 38, 155–170. [Google Scholar] [CrossRef] [PubMed]
- Dittmar, K.; Morse, S.F.; Dick, C.W.; Patterson, B.D. Bat fly evolution from the Eocene to the present (Hippoboscoidea, Streblidae and Nycteribiidae). In Parasite Diversity and Diversification—Evolutionary Ecology Meets Phylogenetics; Morand, S., Krasnov, B.R., Timothy, D., Littlewood, J., Eds.; Cambridge University Press: Cambridge, UK, 2015; pp. 246–264. [Google Scholar]
- Dick, C.W.; Pospischil, R. Nycteribiidae (bat flies). In Encyclopedia of Parasitology; Mehlhorn, H., Ed.; Springer: Berlin, Germany, 2015. [Google Scholar] [CrossRef]
- Dick, C.W. Ecology and Host Specificity of Neotropical Bat Flies (Diptera: Streblidae) and Their Chiropteran Hosts. Ph.D. Thesis, Texas Tech University, Lubbock, TX, USA, 2005. [Google Scholar]
- Dick, C.W.; Patterson, B.D. Bat flies: Obligate ectoparasites of bats. In Micromammals and Macroparasites: From Evolutionary Ecology to Management; Morand, S., Krasnov, B., Poulin, R., Eds.; Springer: Tokyo, Japan, 2006; pp. 179–194. [Google Scholar]
- Dick, C.W.; Miller, J.A. Streblidae. In Manual of Central American Diptera; Brown, B.V., Borkent, A., Cumming, J.M., Wood, D.M., Woodley, N.E., Zumbado, M., Eds.; National Research Council Press: Ottawa, ON, Canada, 2010; Volume II, pp. 1249–1260. [Google Scholar]
- Hiller, T.; Honner, B.; Page, R.A.; Tschapka, M. Leg structure explains host site preference in bat flies (Diptera: Streblidae) parasitizing neotropical bats (Chiroptera: Phyllostomidae). Parasitology 2018, 145, 1475–1482. [Google Scholar] [CrossRef] [PubMed]
- Szentiványi, T.; Christe, P.; Glaizot, O. Bat flies and their microparasites: Current knowledge and distribution. Front. Vet. Sci. 2019, 6, 115. [Google Scholar] [CrossRef] [PubMed]
- Blackwell, M.; Haelewaters, D.; Pfister, D.H. Laboulbeniomycetes: Evolution, natural history, and Thaxter’s final word. Mycologia 2020, 112, in press. [Google Scholar] [CrossRef]
- De Kesel, A. Host specificity and habitat preference of Laboulbenia slackensis. Mycologia 1996, 88, 565–573. [Google Scholar] [CrossRef]
- Haelewaters, D.; De Kesel, A.; Pfister, D.H. Integrative taxonomy reveals hidden species within a common fungal parasite of ladybirds. Sci. Rep. 2018, 8, 15966. [Google Scholar] [CrossRef]
- De Kesel, A. Relative importance of direct and indirect infection in the transmission of Laboulbenia slackensis (Ascomycetes, Laboulbeniales). Belg. J. Bot. 1995, 128, 124–130. [Google Scholar]
- Roskov, Y.; Ower, G.; Orrell, T.; Nicolson, D.; Bailly, N.; Kirk, P.M.; Bourgoin, T.; DeWalt, R.E.; Decock, W.; van Nieukerken, E.; et al. Species 2000 & ITIS Catalogue of Life, 2019 Annual Checklist. Available online: https://www.catalogueoflife.org/annual-checklist/2019 (accessed on 15 November 2020).
- Haelewaters, D.; Dima, B.; Abdel-Hafiz, B.I.I.; Abdel-Wahab, M.A.; Abul-Ezz, S.R.; Acar, I.; Aguirre-Acosta, E.; Aime, M.C.; Aldemir, S.; Ali, M.; et al. Fungal Systematics and Evolution 6. Sydowia 2020, 72, 231–356. [Google Scholar] [CrossRef]
- Haelewaters, D.; Blackwell, M.; Pfister, D.H. Laboulbeniomycetes: Intimate fungal associates of arthropods. Ann. Rev. Entomol. 2021, 66, 13.1–13.20. [Google Scholar] [CrossRef]
- Dogonniuck, A.E.; Squires, T.J.; Weir, A. Studies on Dimorphomyceteae: I. New species of Nycteromyces and Dimeromyces (Laboulbeniales) on bat flies (Streblidae). Mycologia 2019, 111, 118–126. [Google Scholar] [CrossRef]
- Haelewaters, D.; Pfliegler, W.P.; Szentiványi, T.; Földvári, M.; Sándor, A.D.; Barti, L.; Camacho, J.J.; Gort, G.; Estók, P.; Hiller, T.; et al. Parasites of parasites of bats: Laboulbeniales (Fungi: Ascomycota) on bat flies (Diptera: Nycteribiidae) in Central Europe. Parasites Vectors 2017, 10, 96. [Google Scholar] [CrossRef] [PubMed]
- Handley, C.O., Jr. Checklist of the mammals of Panama. In Ectoparasites of Panama; Wenzel, R.L., Tipton, V.J., Eds.; Field Museum of Natural History: Chicago, IL, USA, 1966; pp. 753–795. [Google Scholar]
- Timm, R.M.; LaVal, R.K. A field key to the bats of Costa Rica. In Occasional Publication Series; University of Kansas Center of Latin American Studies: Lawrence, KS, USA, 1998; Volume 22, pp. 1–30. [Google Scholar]
- Dietz, C.; Kiefer, A. Bats of Britain and Europe; Bloomsbury Publishing: London, UK, 2016. [Google Scholar]
- Murariu, D.; Chişamera, G.; Măntoiu, D.Ş.; Pocora, I. Romanian Fauna. Mammalia. Volume XVI, Fascicle 3: Chiroptera; Editura Academiei Române: Bucharest, Romania, 2016. [Google Scholar]
- Williamson, K. The Fair Isle apparatus for collecting bird ectoparasites. Brit. Birds 1954, 47, 235–236. [Google Scholar]
- Theodor, O.; Moscona, A. On bat parasites in Palestine. I. Nycteribiidae, Streblidae, Hemiptera, Siphonaptera. Parasitology 1954, 44, 157–245. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, R.L.; Tipton, V.J. Ectoparasites of Panama; Field Museum of Natural History: Chicago, IL, USA, 1966. [Google Scholar]
- Theodor, O. An Illustrated Catalogue of the Rothschild Collection of Nycteribiidae in the British Museum (Natural History), with Keys and Short Descriptions for the Identification of Subfamilies, Genera, Species and Subspecies; British Museum (Natural History) Publication: London, UK, 1967. [Google Scholar]
- Wenzel, R.L. The streblid batflies of Venezuela (Diptera: Streblidae). Brigh. Young Univ. Sci. Bull. 1976, 20, 1–177. [Google Scholar] [CrossRef]
- Guerrero, R. Catalogo de los Streblidae (Diptera: Pupipara) parasitos de murcielagos (Mammalia: Chiroptera) del Nuevo Mundo I. Clave para los géneros y Nycterophiliinae. Acta Biol. Venez. 1993, 14, 61–75. [Google Scholar]
- Guerrero, R. Catalogo de los Streblidae (Diptera: Pupipara) parasitos de murcielagos (Mammalia: Chiroptera) del Nuevo Mundo II. Los grupos: Pallidus, caecus, major, uniformis y longipes del género Trichobius (Gervais, 1844). Acta Biol. Venez. 1994, 15, 1–18. [Google Scholar]
- Guerrero, R. Catalogo de los Streblidae (Diptera: Pupipara) parasitos de murcielagos (Mammalia: Chiroptera) del Nuevo Mundo IV. Trichobiinae con alas desarrolladas. Bol. Entomol. Venez. 1994, 9, 161–192. [Google Scholar]
- Guerrero, R. Catalogo de los Streblidae (Diptera: Pupipara) parasitos de murcielagos (Mammalia: Chiroptera) del Nuevo Mundo III. Los grupos: Dugesii, dunni y phyllostomae del género Trichobius (Gervais, 1844). Acta Biol. Venez. 1995, 15, 1–27. [Google Scholar]
- Guerrero, R. Catalogo de los Streblidae (Diptera: Pupipara) parasitos de murcielagos (Mammalia: Chiroptera) del Nuevo Mundo V. Trichobiinae con alas reducidas o ausentes y miscelaneos. Bol. Entomol. Venez. 1995, 10, 135–160. [Google Scholar]
- Guerrero, R. Catálogo de los Streblidae (Diptera: Pupipara) parásitos de murciélagos (Mammalia: Chiroptera) del Nuevo Mundo VI. Streblinae. Acta Biol. Venez. 1996, 16, 1–26. [Google Scholar]
- Guerrero, R. Catálogo de los Streblidae (Diptera: Pupipara) parásitos de murciélagos (Mammalia: Chiroptera) del Nuevo Mundo VII. Lista de especies, hospedadores y países. Acta Biol. Venez. 1997, 17, 9–24. [Google Scholar]
- Guerrero, R. Notes on Neotropical batflies (Diptera, Streblidae). I. The genus Trichobius, with description of two new species and one new subspecies from Venezuela. Acta Parasitol. 1998, 43, 86–93. [Google Scholar]
- Guerrero, R. Notes on Neotropical bat flies (Diptera: Streblidae): II. Review of the genus Xenotrichobius. Acta Parasitol. 1998, 43, 142–147. [Google Scholar]
- Miller, J.; Tschapka, M. The Bat Flies of La Selva (Diptera: Nycteribiidae, Streblidae). Available online: http://www.biologie.uni-ulm.de/bio3/Batfly/index.html (accessed on 14 November 2020).
- Dick, C.W. Review of the bat flies of Honduras, Central America (Diptera: Streblidae). J. Parasitol. Res. 2013, 437696. [Google Scholar] [CrossRef]
- Liu, J.; Haelewaters, D.; Pfliegler, W.P.; Page, R.A.; Dick, C.W.; Aime, M.C. A new species of Gloeandromyces from Ecuador and Panama revealed by morphology and phylogenetic reconstruction, with a discussion of secondary barcodes in Laboulbeniomycetes taxonomy. Mycologia 2020, 112, in press. [Google Scholar] [CrossRef]
- Thaxter, R. Contribution toward a monograph of the Laboulbeniaceae. Part II. Mem. Am. Acad. Arts Sci. 1908, 13, 217–469. [Google Scholar] [CrossRef]
- Thaxter, R. Contribution towards a monograph of the Laboulbeniaceae. Part III. Mem. Am. Acad. Arts Sci. 1924, 14, 309–426. [Google Scholar]
- Haelewaters, D.; Pfister, D.H. Morphological species of Gloeandromyces (Ascomycota, Laboulbeniales) evaluated using single-locus species delimitation methods. Fungal Syst. Evol. 2019, 3, 19–33. [Google Scholar] [CrossRef]
- Estrada-Villegas, S.; Halczok, T.K.; Tschapka, M.; Page, R.A.; Brändel, S.D.; Hiller, T. Bats and their bat flies: Community composition and host specificity on a Pacific island archipelago. Acta Chiropt. 2018, 20, 161–176. [Google Scholar] [CrossRef]
- Szentiványi, T.; Haelewaters, D.; Pfliegler, W.P.; Clément, L.; Christe, P.; Glaizot, O. Laboulbeniales (Fungi: Ascomycota) infection of bat flies (Diptera: Nycteribiidae) from Miniopterus schreibersii across Europe. Parasites Vectors 2018, 11, 395. [Google Scholar] [CrossRef]
- Szentiványi, T.; Haelewaters, D.; Rádai, Z.; Mizsei, E.; Pfliegler, W.P.; Báthori, F.; Tartally, A.; Christe, P.; Glaizot, O. Climatic effects on the distribution of ant- and bat fly-associated fungal ectoparasites (Ascomycota, Laboulbeniales). Fungal Ecol. 2019, 39, 371–379. [Google Scholar] [CrossRef]
- Walker, M.J.; Dorrestein, A.; Camacho, J.J.; Meckler, L.A.; Silas, K.A.; Hiller, T.; Haelewaters, D. A tripartite survey of hyperparasitic fungi associated with ectoparasitic flies on bats (Mammalia: Chiroptera) in a neotropical cloud forest in Panama. Parasite 2018, 25, 19. [Google Scholar] [CrossRef] [PubMed]
- RStudio Team. RStudio: Integrated Development for R. Available online: https://www.rstudio.com/ (accessed on 16 November 2020).
- Dormann, C.F.; Gruber, B.; Fründ, J. Introducing the bipartite package: Analysing ecological networks. R News 2008, 8, 8–11. [Google Scholar]
- Blüthgen, N.; Menzel, F.; Blüthgen, N. Measuring specialization in species interaction networks. BMC Ecol. 2006, 6, 9. [Google Scholar] [CrossRef]
- Dittmar, K.; Dick, C.W.; Patterson, B.D.; Whiting, M.F.; Gruwell, M.E. Pupal deposition and ecology of bat flies (Diptera: Streblidae): Trichobius sp. (caecus group) in a Mexican cave habitat. J. Parasitol. 2009, 95, 308–314. [Google Scholar] [CrossRef]
- Fründ, J.; Dormann, C.F.; Holzschuh, A.; Tscharntke, T. Bee diversity effects on pollination depend on functional complementarity and niche shifts. Ecology 2013, 94, 2042–2054. [Google Scholar] [CrossRef]
- Moleón, M.; Sánchez-Zapata, J.A.; Sebastián-González, E.; Owen-Smith, N. Carcass size shapes the structure and functioning of an African scavenging assemblage. Oikos 2015, 124, 1391–1403. [Google Scholar] [CrossRef]
- Cumming, G.S. Host distributions do not limit the species ranges of most African ticks (Acari: Ixodida). Bull. Entomol. Res. 1999, 89, 303–327. [Google Scholar] [CrossRef]
- Cumming, G.S.; van Vuuren, D.P. Will climate change affect ectoparasite species ranges? Glob. Ecol. Biogeogr. 2006, 15, 486–497. [Google Scholar] [CrossRef]
- Pilosof, S.; Dick, C.W.; Korine, C.; Patterson, B.D.; Krasnov, B.R. Effects of Anthropogenic disturbance and climate on patterns of bat fly parasitism. PLoS ONE 2012, 7, e41487. [Google Scholar] [CrossRef]
- Haelewaters, D.; Hiller, T.; Kemp, E.A.; van Wielink, P.S.; Shapiro-Ilan, D.I.; Aime, M.C.; Nedvěd, O.; Pfister, D.H.; Cottrell, T.E. Mortality of native and invasive ladybirds co-infected by ectoparasitic and entomopathogenic fungi. PeerJ 2020, 8, e10110. [Google Scholar] [CrossRef] [PubMed]
- Szentiványi, T.; Kravchenko, K.; Vlaschenko, A.; Estók, P. First record of Laboulbeniales (Fungi: Ascomycota) infection on bat flies (Diptera: Nycteribiidae) from the Caucasus region. Folia Entomol. Hung. 2018, 79, 195–201. [Google Scholar] [CrossRef]

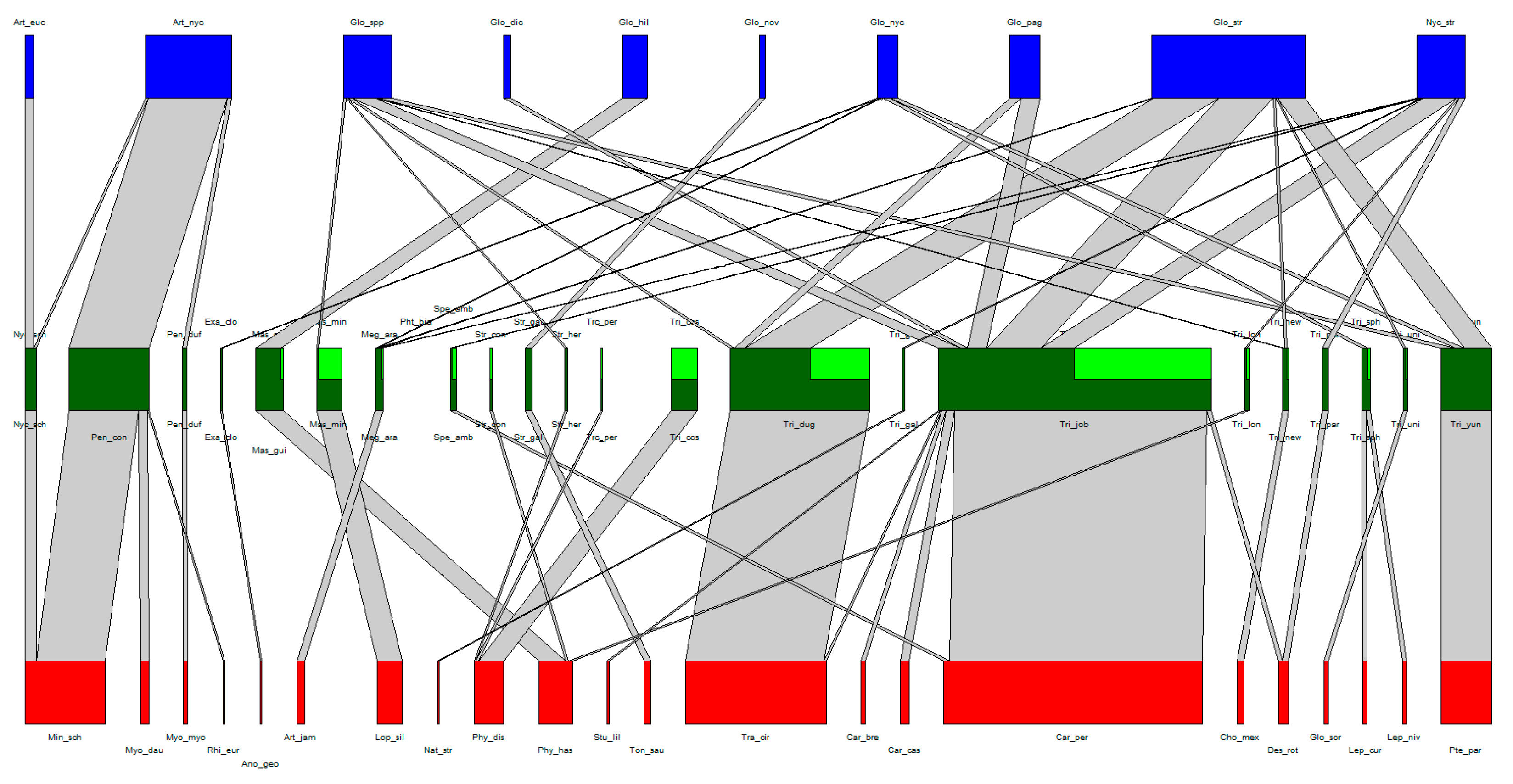
| Bat Fly Species | N | d’i | Bat Species |
|---|---|---|---|
| Nycteribia schmidlii | 5 | 0.47 | Miniopterus schreibersii |
| Penicillidia conspicua | 38 | 0.94 | Miniopterus schreibersii Myotis daubentonii Rhinolophus euryale |
| Penicillidia dufouri | 2 | 1 | Myotis myotis |
| Exastinion clovisi | 1 | 1 | Anoura geoffroyi |
| Mastroptera guimaraesi | 13 | 0.93 | Phyllostomus hastatus |
| Mastoptera minuta | 12 | 1 | Lophostoma silvicolum |
| Megistopoda aranea | 4 | 1 | Artibeus jamaicensis |
| Speisera ambigua | 2 | 0.12 | Carollia perspicillata |
| Strebla consocia | 1 | 0.42 | Phyllostomus hastatus |
| Strebla galindoi | 3 | 1 | Tonatia saurophila |
| Strebla hertigi | 1 | 0.45 | Phyllostomus discolor |
| Trichobioides perspicillatus | 1 | 0.45 | Phyllostomus discolor |
| Trichobius costalimai | 13 | 0.95 | Phyllostomus discolor |
| Trichobius dugesioides | 66 | 0.99 | Trachops cirrhosus |
| Trichobius galei | 1 | 1 | Natalus stramineus |
| Trichobius joblingi | 132 | 0.92 | Carollia brevicauda Carollia castanea Carollia perspicillata Desmodus rotundus Sturnira lilium Trachops cirrhosus |
| Trichobius longipes | 2 | 0.54 | Phyllostomus hastatus |
| Trichobius sp. nov. | 3 | 1 | Choeronycteris mexicana |
| Trichobius parasiticus | 3 | 0.88 | Desmodus rotundus |
| Trichobius sphaeronotus | 3 | 1 | Leptonycteris curasoae Leptonycteris nivalis |
| Trichobius uniformis | 2 | 1 | Glossophaga soricina |
| Trichobius yunkeri | 24 | 1 | Pteronotus parnellii |
| Laboulbeniales Species | N | d’i | Bat Fly Species |
|---|---|---|---|
| Arthrorhynchus eucampsipodae | 4 | 0.94 | Nycteribia schmidlii |
| Arthrorhynchus nycteribiae | 41 | 0.97 | Nycteribia schmidlii Penicillidia dufouri Penicillidia conspicua |
| Gloeandromyces dickii | 3 | 0.19 | Trichobius joblingi |
| Gloeandromyces hilleri | 12 | 1 | Mastoptera guimaraesi |
| Gloeandromyces sp. nov. | 3 | 1 | Strebla galindoi |
| Gloeandromyces nycteribiidarum | 10 | 0.69 | Exastinion clovisi Megistopoda aranea Trichobius caecus Trichobius sphaeronotus Trichobius yunkeri |
| Gloeandromyces pageanus | 14 | 0.21 | Trichobius dugesioides Trichobius joblingi |
| Gloeandromyces streblae | 73 | 0.45 | Megistopoda aranea Trichobius dugesioides Trichobius joblingi Trichobius sp. nov. Trichobius uniformis Trichobius yunkeri |
| Nycteromyces streblidinus | 23 | 0.5 | Megistopoda aranea Speisera ambigua Trichobius galei Trichobius joblingi Trichobius longipes Trichobius parasiticus |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Groot, M.D.; Dumolein, I.; Hiller, T.; Sándor, A.D.; Szentiványi, T.; Schilthuizen, M.; Aime, M.C.; Verbeken, A.; Haelewaters, D. On the Fly: Tritrophic Associations of Bats, Bat Flies, and Fungi. J. Fungi 2020, 6, 361. https://doi.org/10.3390/jof6040361
de Groot MD, Dumolein I, Hiller T, Sándor AD, Szentiványi T, Schilthuizen M, Aime MC, Verbeken A, Haelewaters D. On the Fly: Tritrophic Associations of Bats, Bat Flies, and Fungi. Journal of Fungi. 2020; 6(4):361. https://doi.org/10.3390/jof6040361
Chicago/Turabian Stylede Groot, Michiel D., Iris Dumolein, Thomas Hiller, Attila D. Sándor, Tamara Szentiványi, Menno Schilthuizen, M. Catherine Aime, Annemieke Verbeken, and Danny Haelewaters. 2020. "On the Fly: Tritrophic Associations of Bats, Bat Flies, and Fungi" Journal of Fungi 6, no. 4: 361. https://doi.org/10.3390/jof6040361
APA Stylede Groot, M. D., Dumolein, I., Hiller, T., Sándor, A. D., Szentiványi, T., Schilthuizen, M., Aime, M. C., Verbeken, A., & Haelewaters, D. (2020). On the Fly: Tritrophic Associations of Bats, Bat Flies, and Fungi. Journal of Fungi, 6(4), 361. https://doi.org/10.3390/jof6040361






