β-Glucan Metabolic and Immunomodulatory Properties and Potential for Clinical Application
Abstract
1. Introduction
2. Discovery and Characterization of β-glucans
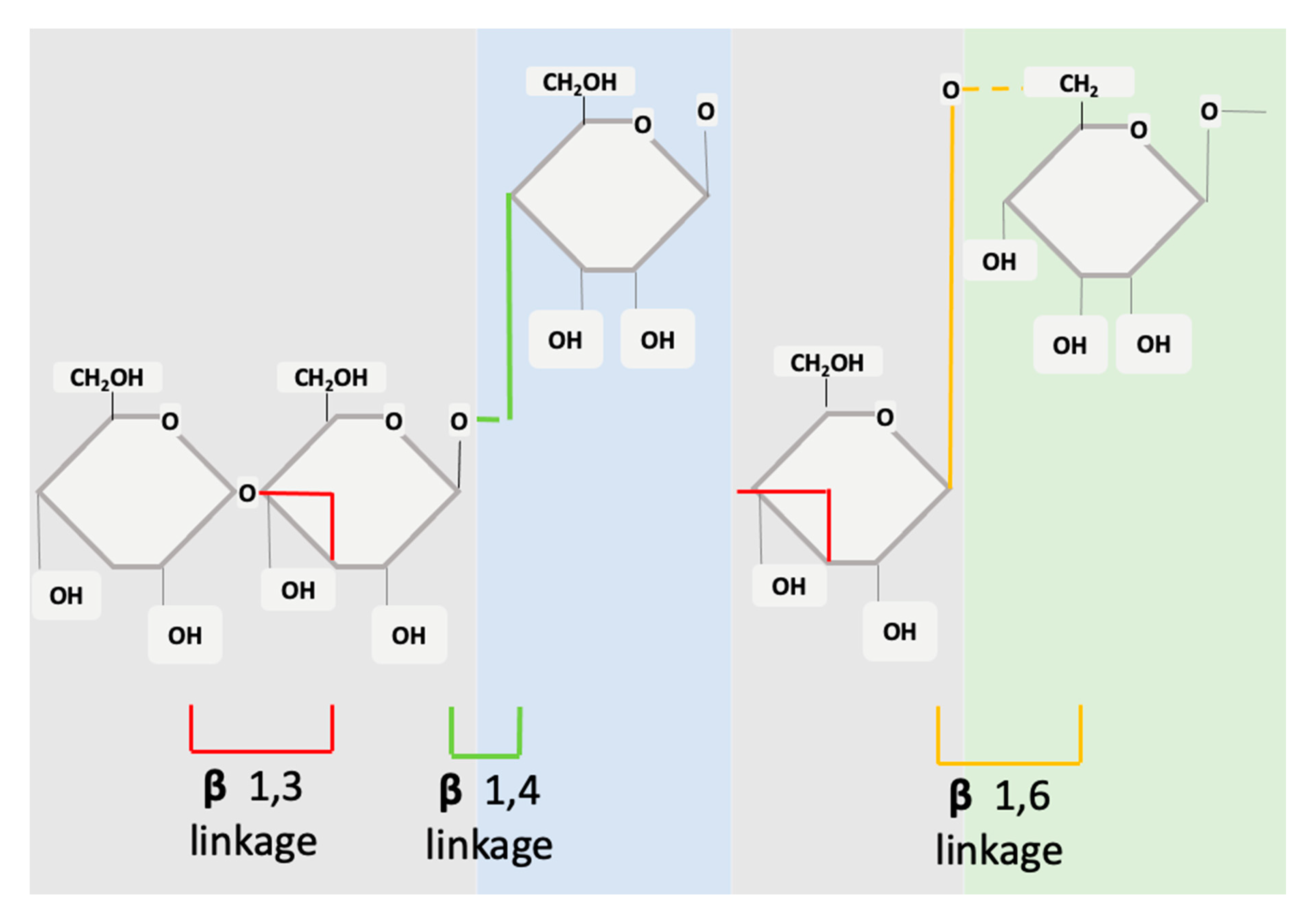
3. β-glucan Structure
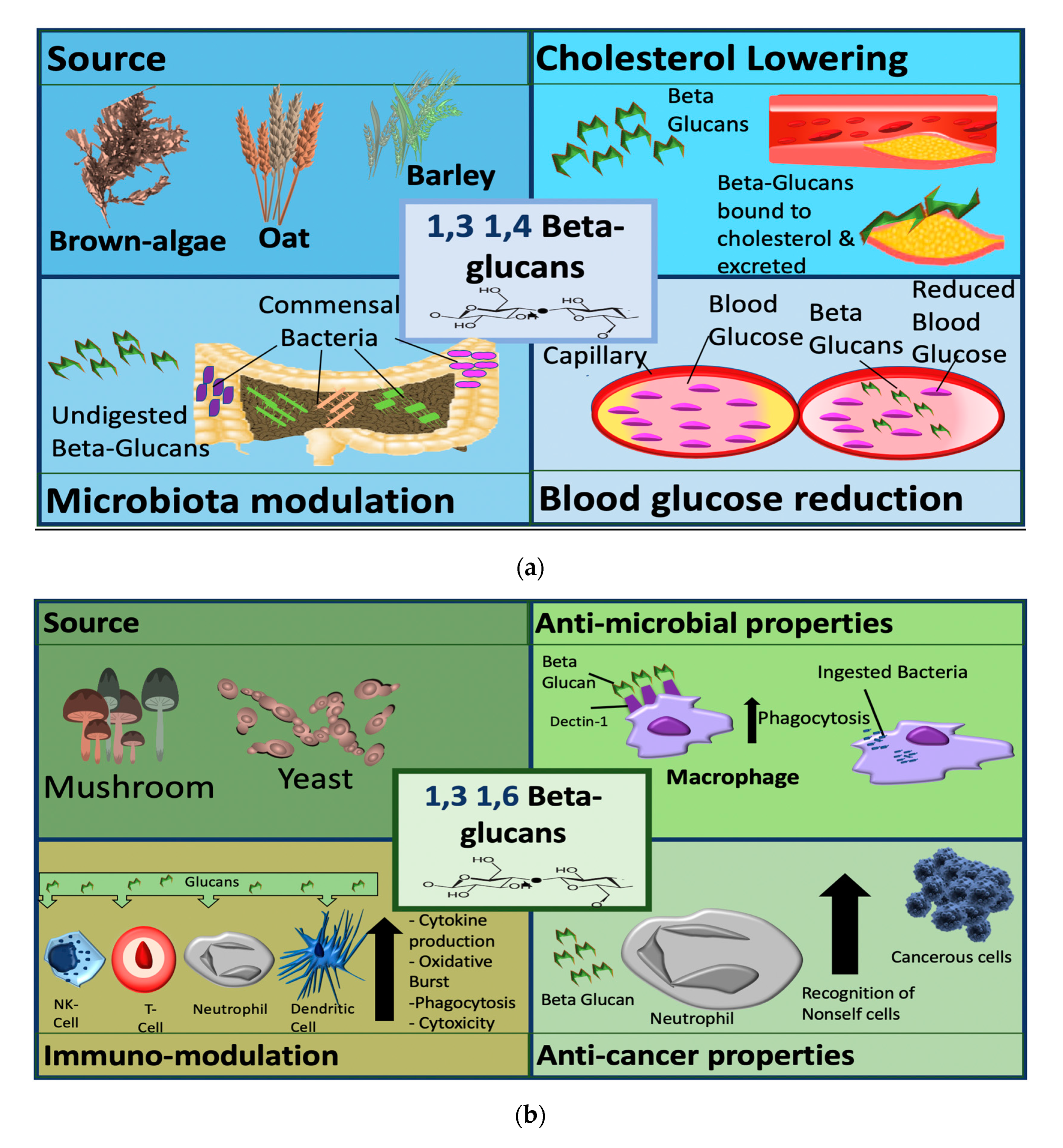
4. Sources of β-glucans
5. Mechanisms of Action
5.1. Role of β-glucans Structure
5.2. Role of β-glucan Source
5.3. Cholesterol-Lowering Effects
5.4. Enhancement of Glycaemic Control
5.5. Effects on Gut and Microbiota
5.6. Key Knowledge Deficits
6. Fungal β-glucans (1,3 and 1,6 Linked)
7. Insights into β-Glucan Effects from Pre-Clinical Models
7.1. Bacterial Sepsis
7.2. Lung Injury
7.3. Cancer Therapy
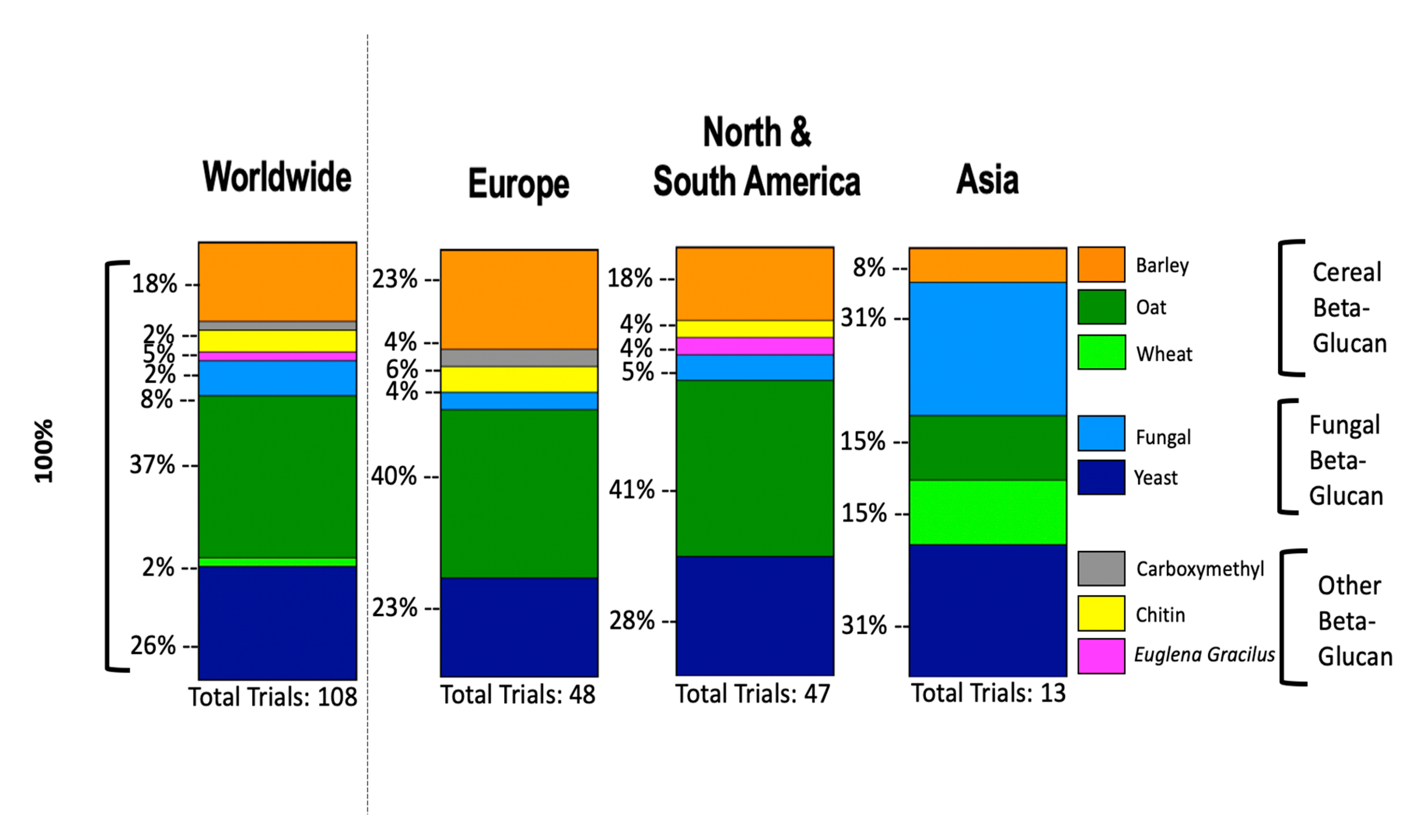
8. Clinical Trials of β-glucans
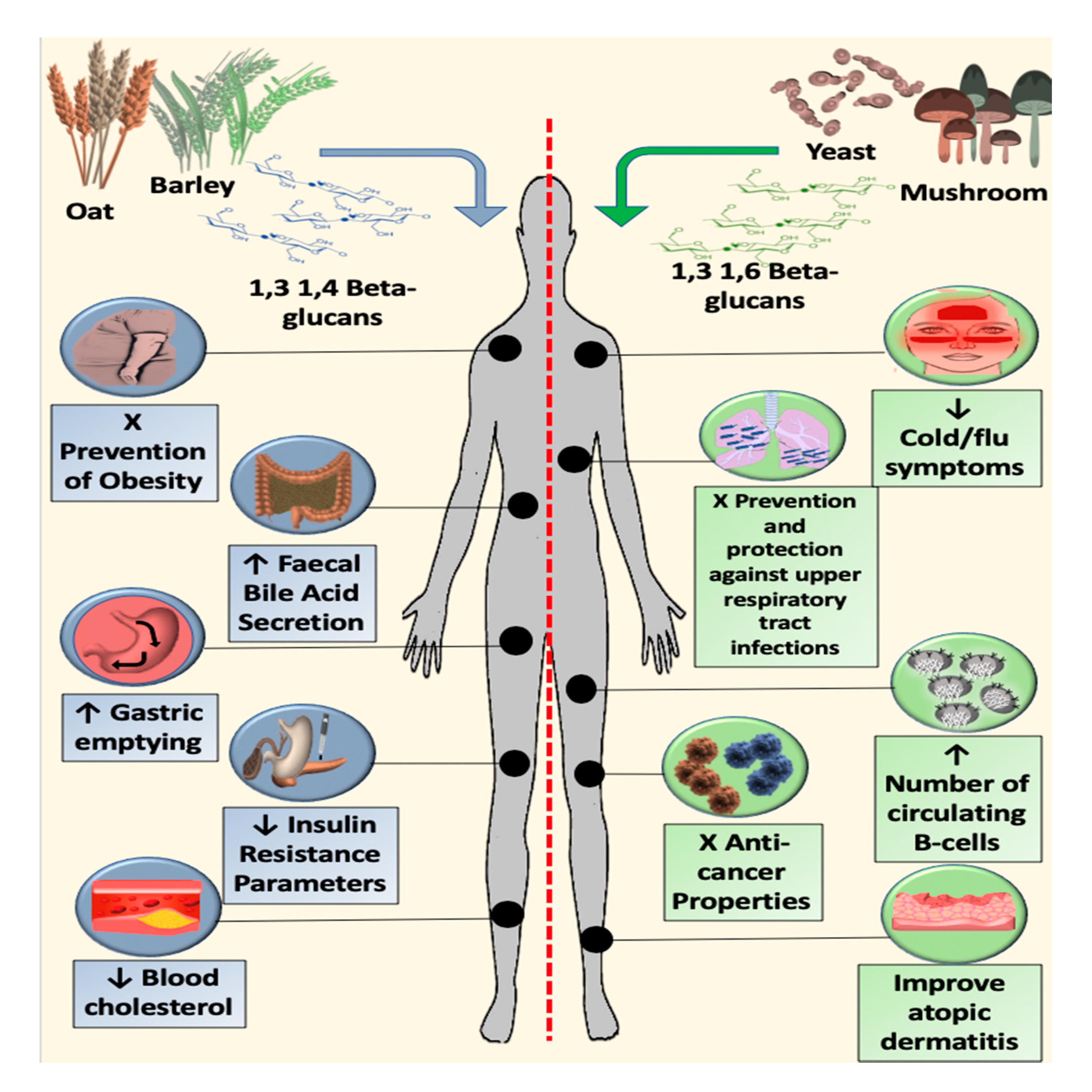
9. Metabolic Effects of Cereal β-glucans
10. Immunomodulatory Effects of Fungal β-glucans
11. CTs Cancer Therapy
12. Translational Challenges and Opportunities
13. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Vetvicka, V.; Vannucci, L.; Sima, P.; Richter, J. Beta Glucan: Supplement or Drug? From Laboratory to Clinical Trials. Molecules 2019, 24, 1251. [Google Scholar] [CrossRef] [PubMed]
- Wasser, S.P. Medicinal mushrooms as a source of antitumor and immunomodulating polysaccharides. Appl. Microbiol. Biotechnol. 2002, 60, 258–274. [Google Scholar] [CrossRef] [PubMed]
- Gil-Ramírez, A.; Soler-Rivas, C. The use of edible mushroom extracts as bioactive ingredients to design novel functional foods with hypocholesterolemic activities. In Mushrooms: Cultivation, Antioxidant Properties and Health Benefits; Nova Science Publishers: Harpak, NY, USA, 2014; pp. 43–74. [Google Scholar]
- Reshetnikov, S.V.; Tan, K.-K. Higher Basidiomycota as a Source of Antitumor and Immunostimulating Polysaccharides (Review). Int. J. Med. Mushrooms 2001, 3, 34. [Google Scholar] [CrossRef]
- Ahmad, A.; Kaleem, M. β-Glucan as a Food Ingredient. In Biopolymers for Food Design; Academic Press: Cambridge, MA, USA, 2018; pp. 351–381. [Google Scholar]
- Nie, S.; Cui, S.W.; Jiang, L. Beta-Glucans and Their Derivatives. In Bioactive Polysaccharides; Academic Press: Cambridge, MA, USA, 2018; pp. 99–141. [Google Scholar] [CrossRef]
- Bernstein, A.M.; Titgemeier, B.; Kirkpatrick, K.; Golubic, M.; Roizen, M.F. Major Cereal Grain Fibers and Psyllium in Relation to Cardiovascular Health. Nutrients 2013, 5, 1471–1487. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Sheng, X.; Shi, A.; Hu, H.; Yang, Y.; Liu, L.; Fei, L.; Liu, H. β-Glucans: Relationships between Modification, Conformation and Functional Activities. Molecules 2017, 22, 257. [Google Scholar] [CrossRef]
- Novak, M.; Vetvicka, V. β-Glucans, History, and the Present: Immunomodulatory Aspects and Mechanisms of Action. J. Immunotoxicol. 2008, 5, 47–57. [Google Scholar] [CrossRef]
- Yang, D.; Zhou, Z.; Zhang, L.-J. An overview of fungal glycan-based therapeutics. In Progress in Molecular Biology and Translational Science; Elsevier BV: Hoboken, NJ, USA, 2019; Volume 163, pp. 135–163. [Google Scholar]
- Vetvicka, V.; Větvičková, J. Glucans and Cancer: Comparison of Commercially Available β-glucans–Part IV. Anticancer Res. 2018, 38, 1327–1333. [Google Scholar] [CrossRef]
- Kerckhoffs, D.A.J.M.; Hornstra, G.; Mensink, R.P. Cholesterol-lowering effect of β-glucan from oat bran in mildly hypercholesterolemic subjects may decrease when β-glucan is incorporated into bread and cookies. Am. J. Clin. Nutr. 2003, 78, 221–227. [Google Scholar] [CrossRef]
- Vetvicka, V.; Vannucci, L.; Sima, P. β-glucan as a new tool in vaccine development. Scand. J. Immunol. 2019, 91, e12833. [Google Scholar] [CrossRef]
- Pillemer, L.; Blum, L.; Lepow, I.H.; Ross, O.A.; Todd, E.W.; Wardlaw, A.C. The Properdin System and Immunity: I. Demonstration and Isolation of a New Serum Protein, Properdin, and Its Role in Immune Phenomena. Science 1954, 120, 279–285. [Google Scholar] [CrossRef]
- Riggi, S.J.; Di Luzio, N.R. Identification of a reticuloendothelial stimulating agent in zymosan. Am. J. Physiol. Content 1961, 200, 297–300. [Google Scholar] [CrossRef] [PubMed]
- Ross, O.A. The properdin system in relation to fatal bacteremia following total-body irradiation of laboratory animals. Ann. N. Y. Acad. Sci. 1956, 66, 274–279. [Google Scholar] [CrossRef]
- Kiser, J.S.; Lindh, H.; De Mello, G.C. THE EFFECT OF VARIOUS SUBSTANCES ON RESISTANCE TO EXPERIMENTAL INFECTIONS. Ann. N. Y. Acad. Sci. 1956, 66, 312–328. [Google Scholar] [CrossRef]
- Old, L.J.; Clarke, D.A.; Benacerraf, B.; Goldsmith, M. The reticuloendothelial system and the neoplastic process*. Ann. N. Y. Acad. Sci. 2006, 88, 264–280. [Google Scholar] [CrossRef]
- Di Carlo, F.J.; Fiore, J.V. On the Composition of Zymosan. Science 1958, 127, 756–757. [Google Scholar] [CrossRef]
- Heller, J.H. NONTOXIC RES STIMULATORY LIPIDS*. Ann. N. Y. Acad. Sci. 2006, 88, 116–121. [Google Scholar] [CrossRef]
- Chihara, G.; Maeda, Y.Y.; Hamuro, J.; Sasaki, T.; Fukuoka, F. Inhibition of Mouse Sarcoma 180 by Polysaccharides from Lentinus edodes (Berk.) Sing. Nat. Cell Biol. 1969, 222, 687–688. [Google Scholar] [CrossRef]
- Yang, J.; Tu, J.; Liu, H.; Wen, L.; Jiang, G.; Yang, B. Identification of an immunostimulatory polysaccharide in banana. Food Chem. 2019, 277, 46–53. [Google Scholar] [CrossRef]
- Kaur, R.; Sharma, M.; Ji, D.; Xu, M.; Agyei, D. Structural Features, Modification, and Functionalities of Beta-Glucan. Fibers 2019, 8, 1. [Google Scholar] [CrossRef]
- Bae, I.Y.; Kim, H.W.; Yoo, H.J.; Kim, E.S.; Lee, S.; Park, D.Y.; Lee, H.G. Correlation of branching structure of mushroom β-glucan with its physiological activities. Food Res. Int. 2013, 51, 195–200. [Google Scholar] [CrossRef]
- Bashir, K.M.I.; Choi, J.-S. Clinical and Physiological Perspectives of β-Glucans: The Past, Present, and Future. Int. J. Mol. Sci. 2017, 18, 1906. [Google Scholar] [CrossRef] [PubMed]
- Borchani, C.; Fonteyn, F.; Jamin, G.; Destain, J.; Willems, L.; Paquot, M.; Blecker, C.; Thonart, P.; Chema, B.; Fabienne, F.; et al. Structural Characterization, Technological Functionality and Physiological Aspects of Fungal ?-D-Glucans: A Review. Crit. Rev. Food Sci. Nutr. 2015, 56, 1746–1752. [Google Scholar] [CrossRef] [PubMed]
- Johansson, L.; Virkki, L.; Maunu, S.; Lehto, M.; Ekholm, P.; Varo, P. Structural characterization of water soluble β-glucan of oat bran. Carbohydr. Polym. 2000, 42, 143–148. [Google Scholar] [CrossRef]
- Tohamy, A.A.; El-Ghor, A.A.; El Nahas, S.; Noshy, M.M. β-Glucan inhibits the genotoxicity of cyclophosphamide, adriamycin and cisplatin. Mutat. Res. Toxicol. Environ. Mutagen. 2003, 541, 45–53. [Google Scholar] [CrossRef]
- Gupta, M.; Abu-Ghannam, N.; Gallaghar, E. Barley for Brewing: Characteristic Changes during Malting, Brewing and Applications of its By-Products. Compr. Rev. Food Sci. Food Saf. 2010, 9, 318–328. [Google Scholar] [CrossRef]
- Sikora, P.; Tosh, S.M.; Brummer, Y.; Olsson, O. Identification of high β-glucan oat lines and localization and chemical characterization of their seed kernel β-glucans. Food Chem. 2013, 137, 83–91. [Google Scholar] [CrossRef]
- Izydorczyk, M.; Dexter, J. Barley β-glucans and arabinoxylans: Molecular structure, physicochemical properties, and uses in food products–a Review. Food Res. Int. 2008, 41, 850–868. [Google Scholar] [CrossRef]
- Benito-Román, Ó.; Alonso, E.; Gairola, K.; Cocero, M. Fixed-bed extraction of β-glucan from cereals by means of pressurized hot water. J. Supercrit. Fluids 2013, 82, 122–128. [Google Scholar] [CrossRef]
- Mejía, S.M.V.; De Francisco, A.; Bohrer, B. A comprehensive review on cereal β-glucan: Extraction, characterization, causes of degradation, and food application. Crit. Rev. Food Sci. Nutr. 2020, 60, 3693–3704. [Google Scholar] [CrossRef]
- Zhu, F.; Du, B.; Xu, B. A critical review on production and industrial applications of beta-glucans. Food Hydrocoll. 2016, 52, 275–288. [Google Scholar] [CrossRef]
- Man, D.H.; Yeon, S.H.; Sung, H.H.; Hyun, W.S. Solubilization of water-insoluble β-glucan isolated from Ganoderma lucidum. J. Environ. Biol. 2008, 29, 237–242. [Google Scholar]
- Nakashima, A.; Yamada, K.; Iwata, O.; Sugimoto, R.; Atsuji, K.; Ogawa, T.; Ishibashi-Ohgo, N.; Suzuki, K. β-Glucan in Foods and Its Physiological Functions. J. Nutr. Sci. Vitaminol. 2018, 64, 8–17. [Google Scholar] [CrossRef]
- Zhan, X.; Lin, C.-C.; Zhang, H.-T. Recent advances in curdlan biosynthesis, biotechnological production, and applications. Appl. Microbiol. Biotechnol. 2012, 93, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Burkus, Z.; Temelli, F. Rheological properties of barley β-glucan. Carbohydr. Polym. 2005, 59, 459–465. [Google Scholar] [CrossRef]
- Novak, M. Glucans as Biological Response Modifiers. Endocr. Metab. Immune Disord. -Drug Targets 2009, 9, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Henrion, M.; Francey, C.; Lê, K.-A.; Lamothe, L. Cereal B-Glucans: The Impact of Processing and How It Affects Physiological Responses. Nutrients 2019, 11, 1729. [Google Scholar] [CrossRef]
- Banchathanakij, R.; Suphantharika, M. Effect of different β-glucans on the gelatinisation and retrogradation of rice starch. Food Chem. 2009, 114, 5–14. [Google Scholar] [CrossRef]
- Ulmius, M.; Önning, G.; Nilsson, L. Solution behavior of barley β-glucan as studied with asymmetrical flow field-flow fractionation. Food Hydrocoll. 2012, 26, 175–180. [Google Scholar] [CrossRef]
- Surenjav, U.; Zhang, L.; Xu, X.; Zhang, X.; Zeng, F. Effects of molecular structure on antitumor activities of (1→3)-β-d-glucans from different Lentinus Edodes. Carbohydr. Polym. 2006, 63, 97–104. [Google Scholar] [CrossRef]
- Rieder, A.; Grimmer, S.; Kolset, S.O.; Michaelsen, T.E.; Knutsen, S.H. Cereal β-glucan preparations of different weight average molecular weights induce variable cytokine secretion in human intestinal epithelial cell lines. Food Chem. 2011, 128, 1037–1043. [Google Scholar] [CrossRef]
- Zeković, D.B.; Kwiatkowski, S.; Vrvić, M.M.; JakovljeviĆ, D.; Moran, C.A. Natural and Modified (1→3)-β-D-Glucans in Health Promotion and Disease Alleviation. Crit. Rev. Biotechnol. 2005, 25, 205–230. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Seviour, R.J. Medicinal importance of fungal β-(1→3), (1→6)-glucans. Mycol. Res. 2007, 111, 635–652. [Google Scholar] [CrossRef] [PubMed]
- Driscoll, M.; Hansen, R.; Ding, C.; Cramer, D.E.; Yan, J. Therapeutic potential of various β-glucan sources in conjunction with anti-tumor monoclonal antibody in cancer therapy. Cancer Biol. Ther. 2009, 8, 218–225. [Google Scholar] [CrossRef] [PubMed]
- Soltanian, S.; Stuyven, E.; Cox, E.; Sorgeloos, P.; Bossier, P. Beta-glucans as immunostimulant in vertebrates and invertebrates. Crit. Rev. Microbiol. 2009, 35, 109–138. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Baruah, K.; Cox, E.; Vanrompay, D.; Bossier, P. Structure-Functional Activity Relationship of β-Glucans From the Perspective of Immunomodulation: A Mini-Review. Front. Immunol. 2020, 11, 658. [Google Scholar] [CrossRef] [PubMed]
- Raa, J. Immune modulation by non-digestible and non-absorbable beta-1,3/1,6-glucan. Microb. Ecol. Health Dis. 2015, 26, 27824. [Google Scholar] [CrossRef] [PubMed]
- Sletmoen, M.; Stokke, B.T. Higher order structure of (1,3)-β-D-glucans and its influence on their biological activities and complexation abilities. Biopolym. Orig.Res. Biomol. 2008, 89, 310–321. [Google Scholar] [CrossRef]
- Brown, G.D.; Gordon, S. A new receptor for β-glucans. Nat. Cell Biol. 2001, 413, 36–37. [Google Scholar] [CrossRef]
- Chan, G.C.-F.; Chan, W.K.; Sze, D.M.-Y. The effects of β-glucan on human immune and cancer cells. J. Hematol. Oncol. 2009, 2, 25. [Google Scholar] [CrossRef]
- Taylor, P.R.; Tsoni, S.V.; Willment, J.A.; Dennehy, K.M.; Rosas, M.; Findon, H.; Haynes, K.; Steele, C.; Botto, M.; Gordon, S.; et al. Dectin-1 is required for β-glucan recognition and control of fungal infection. Nat. Immunol. 2007, 8, 31–38. [Google Scholar] [CrossRef]
- Brown, G.D.; Gordon, S. Immune recognition of fungal β-glucans. Cell. Microbiol. 2005, 7, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.; Masterson, C.; Rezoagli, E.; O’Toole, D.; Major, I.; Stack, G.D.; Lynch, M.; Laffey, J.G.; Rowan, N.J. β-Glucan extracts from the same edible shiitake mushroom Lentinus edodes produce differential in-vitro immunomodulatory and pulmonary cytoprotective effects—Implications for coronavirus disease (COVID-19) immunotherapies. Sci. Total Environ. 2020, 732, 139330. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S.; Giles, W.H.; Mokdad, A.H. Increasing Prevalence of the Metabolic Syndrome Among U.S. Adults. Diabetes Care 2004, 27, 2444–2449. [Google Scholar] [CrossRef] [PubMed]
- Detection, E.E.P.O. Executive Summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001, 285, 2486–2497. [Google Scholar] [CrossRef]
- Dhewantara, F.X. Kusmiati Cholesterol-Lowering Effect of Beta Glucan Extracted from Saccharomyces cerevisiae in Rats. Sci. Pharm. 2016, 84, 153–165. [Google Scholar] [CrossRef]
- Lefebvre, P.; Cariou, B.; Lien, F.; Kuipers, F.; Staels, B. Role of Bile Acids and Bile Acid Receptors in Metabolic Regulation. Physiol. Rev. 2009, 89, 147–191. [Google Scholar] [CrossRef]
- Staels, B.; Fonseca, V.A. Bile Acids and Metabolic Regulation: Mechanisms and clinical responses to bile acid sequestration. Diabetes Care 2009, 32, S237–S245. [Google Scholar] [CrossRef]
- Pullinger, C.R.; Eng, C.; Salen, G.; Shefer, S.; Batta, A.K.; Erickson, S.K.; Verhagen, A.; Rivera, C.R.; Mulvihill, S.J.; Malloy, M.J.; et al. Human cholesterol 7α-hydroxylase (CYP7A1) deficiency has a hypercholesterolemic phenotype. J. Clin. Investig. 2002, 110, 109–117. [Google Scholar] [CrossRef]
- Sato, K.; Ohuchi, A.; Sook, S.H.; Toyomizu, M.; Akiba, Y. Changes in mRNA expression of 3-hydroxy-3-methylglutaryl coenzyme A reductase and cholesterol 7 alpha-hydroxylase in chickens. Biochim. et Biophys. Acta (BBA)-Bioenerg. 2003, 1630, 96–102. [Google Scholar] [CrossRef]
- Tong, L.-T.; Zhong, K.; Liu, L.; Qiu, J.; Guo, L.; Zhou, X.; Cao, L.; Zhou, S. Effects of dietary wheat bran arabinoxylans on cholesterol metabolism of hypercholesterolemic hamsters. Carbohydr. Polym. 2014, 112, 1–5. [Google Scholar] [CrossRef]
- Tong, L.-T.; Zhong, K.; Liu, L.; Zhou, X.; Qiu, J.; Zhou, S. Effects of dietary hull-less barley β-glucan on the cholesterol metabolism of hypercholesterolemic hamsters. Food Chem. 2015, 169, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Houten, S.M.; Watanabe, M.; Auwerx, J. Endocrine functions of bile acids. EMBO J. 2006, 25, 1419–1425. [Google Scholar] [CrossRef] [PubMed]
- Joyce, S.A.; Kamil, A.; Fleige, L.; Gahan, C.G. The Cholesterol-Lowering Effect of Oats and Oat Beta Glucan: Modes of Action and Potential Role of Bile Acids and the Microbiome. Front. Nutr. 2019, 6, 171. [Google Scholar] [CrossRef] [PubMed]
- Zhou, A.L.; Hergert, N.; Rompato, G.; Lefevre, M. Whole Grain Oats Improve Insulin Sensitivity and Plasma Cholesterol Profile and Modify Gut Microbiota Composition in C57BL/6J Mice. J. Nutr. 2014, 145, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Drzikova, B.; Dongowski, G.; Gebhardt, E.; Habel, A. The composition of dietary fibre-rich extrudates from oat affects bile acid binding and fermentation in vitro. Food Chem. 2005, 90, 181–192. [Google Scholar] [CrossRef]
- Andersson, K.; Svedberg, K.; Lindholm, M.W.; Oste, R.; Hellstrand, P. Oats (Avena sativa) reduce atherogenesis in LDL-receptor-deficient mice. Atherosclerosis 2010, 212, 93–99. [Google Scholar] [CrossRef]
- Gunness, P.; Michiels, J.; Vanhaecke, L.; De Smet, S.; Kravchuk, O.; Van De Meene, A.; Gidley, M.J. Reduction in circulating bile acid and restricted diffusion across the intestinal epithelium are associated with a decrease in blood cholesterol in the presence of oat β-glucan. FASEB J. 2016, 30, 4227–4238. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.-H.; Lai, M.-H. Fermentation of Resistant Rice Starch Produces Propionate Reducing Serum and Hepatic Cholesterol in Rats. J. Nutr. 2000, 130, 1991–1995. [Google Scholar] [CrossRef]
- Tiwari, U.; Cummins, E. Meta-analysis of the effect of β-glucan intake on blood cholesterol and glucose levels. Nutrition 2011, 27, 1008–1016. [Google Scholar] [CrossRef]
- McRorie, J.; McKeown, N.M. Understanding the Physics of Functional Fibers in the Gastrointestinal Tract: An Evidence-Based Approach to Resolving Enduring Misconceptions about Insoluble and Soluble Fiber. J. Acad. Nutr. Diet. 2017, 117, 251–264. [Google Scholar] [CrossRef]
- Wolever, T.M.; Tosh, S.M.; Gibbs, A.L.; Brand-Miller, J.; Duncan, A.M.; Hart, V.; Lamarche, B.; Thomson, B.A.; Duss, R.; Wood, P.J. Physicochemical properties of oat β-glucan influence its ability to reduce serum LDL cholesterol in humans: A randomized clinical trial. Am. J. Clin. Nutr. 2010, 92, 723–732. [Google Scholar] [CrossRef] [PubMed]
- Kahlon, T.; Smith, G.; Shao, Q. In vitro binding of bile acids by kidney bean (Phaseolus vulgaris), black gram (Vigna mungo), bengal gram (Cicer arietinum) and moth bean (Phaseolus aconitifolins). Food Chem. 2005, 90, 241–246. [Google Scholar] [CrossRef]
- Ellegård, L.; Andersson, H. Oat bran rapidly increases bile acid excretion and bile acid synthesis: An ileostomy study. Eur. J. Clin. Nutr. 2007, 61, 938–945. [Google Scholar] [CrossRef] [PubMed]
- Theuwissen, E.; Mensink, R.P. Water-soluble dietary fibers and cardiovascular disease. Physiol. Behav. 2008, 94, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Maki, K.C.; Shinnick, F.; Seeley, M.A.; Veith, P.E.; Quinn, L.C.; Hallissey, P.J.; Temer, A.; Davidson, M. Food Products Containing Free Tall Oil-Based Phytosterols and Oat β-Glucan Lower Serum Total and LDL Cholesterol in Hypercholesterolemic Adults. J. Nutr. 2003, 133, 808–813. [Google Scholar] [CrossRef] [PubMed]
- Keenan, J.M.; Pins, J.J.; Frazel, C.; Moran, A.; Turnquist, L. Oat ingestion reduces systolic and diastolic blood pressure in patients with mild or borderline hypertension: A pilot trial. J. Fam. Pr. 2002, 51, 369. [Google Scholar]
- Biörklund, M.; Van Rees, A.; Mensink, R.P.; Önning, G.; Bi, M. Changes in serum lipids and postprandial glucose and insulin concentrations after consumption of beverages with β-glucans from oats or barley: A randomised dose-controlled trial. Eur. J. Clin. Nutr. 2005, 59, 1272–1281. [Google Scholar] [CrossRef]
- Streppel, M.T.; Arends, L.R.; Veer, P.V.T.; Grobbee, D.E.; Geleijnse, J.M. Dietary Fiber and Blood Pressure. Arch. Intern. Med. 2005, 165, 150–156. [Google Scholar] [CrossRef]
- El Khoury, D.; Cuda, C.; Luhovyy, B.L.; Anderson, G.H. Beta Glucan: Health Benefits in Obesity and Metabolic Syndrome. J. Nutr. Metab. 2012, 2012, 851362. [Google Scholar] [CrossRef]
- Behall, K.M.; Scholfield, D.J.; Hallfrisch, J.G.; Liljeberg-Elmståhl, H.G. Consumption of Both Resistant Starch and -Glucan Improves Postprandial Plasma Glucose and Insulin in Women. Diabetes Care 2006, 29, 976–981. [Google Scholar] [CrossRef]
- Selph, S.; Ginsburg, A.D.; Chou, R. Impact of contacting study authors to obtain additional data for systematic reviews: Diagnostic accuracy studies for hepatic fibrosis. Syst. Rev. 2014, 3, 107. [Google Scholar] [CrossRef] [PubMed]
- Wood, P.J.; Beer, M.U.; Butler, G.B. Evaluation of role of concentration and molecular weight of oat β-glucan in determining effect of viscosity on plasma glucose and insulin following an oral glucose load. Br. J. Nutr. 2000, 84, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Hooda, S.; Matte, J.; Vasanthan, T.; Zijlstra, R.T. Dietary purified oat β-glucan reduces peak glucose absorption and portal insulin release in portal-vein catheterized grower pigs. Livest. Sci. 2010, 134, 15–17. [Google Scholar] [CrossRef]
- Regand, A.; Chowdhury, Z.; Tosh, S.M.; Wolever, T.M.; Wood, P. The molecular weight, solubility and viscosity of oat beta-glucan affect human glycemic response by modifying starch digestibility. Food Chem. 2011, 129, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Cai, F.; Shen, R.-L.; Liu, Y. Hypoglycaemic effects and inhibitory effect on intestinal disaccharidases of oat beta-glucan in streptozotocin-induced diabetic mice. Food Chem. 2011, 129, 1066–1071. [Google Scholar] [CrossRef]
- Granfeldt, Y.; Nyberg, L.; Björck, I. Muesli with 4 g oat β-glucans lowers glucose and insulin responses after a bread meal in healthy subjects. Eur. J. Clin. Nutr. 2007, 62, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Mäkeläinen, H.; Anttila, H.; Sihvonen, J.; Hietanen, R.-M.; Tahvonen, R.; Salminen, E.; Mikola, M.; Sontag-Strohm, T. The effect of β-glucan on the glycemic and insulin index. Eur. J. Clin. Nutr. 2006, 61, 779–785. [Google Scholar] [CrossRef]
- Jenkins, A.L.; Jenkins, D.J.A.; Zdravkovic, U.; Würsch, P.; Vuksan, V. Depression of the glycemic index by high levels of β-glucan fiber in two functional foods tested in type 2 diabetes. Eur. J. Clin. Nutr. 2002, 56, 622–628. [Google Scholar] [CrossRef]
- Tapola, N.; Karvonen, H.; Niskanen, L.; Mikola, M.; Sarkkinen, E. Glycemic responses of oat bran products in type 2 diabetic patients. Nutr. Metab. Cardiovasc. Dis. 2005, 15, 255–261. [Google Scholar] [CrossRef]
- Wolever, T.M.S.; Tosh, S.M.; Spruill, S.E.; Jenkins, A.L.; Ezatagha, A.; Duss, R.; Johnson, J.; Chu, Y.; Steinert, R.E. Increasing oat β-glucan viscosity in a breakfast meal slows gastric emptying and reduces glycemic and insulinemic responses but has no effect on appetite, food intake, or plasma ghrelin and PYY responses in healthy humans: A randomized, placebo-controlled, crossover trial. Am. J. Clin. Nutr. 2019, 111, 319–328. [Google Scholar] [CrossRef]
- Gamel, T.H.; Abdel-Aal, E.-S.M.; Ames, N.P.; Duss, R.; Tosh, S.M. Enzymatic extraction of beta-glucan from oat bran cereals and oat crackers and optimization of viscosity measurement. J. Cereal Sci. 2014, 59, 33–40. [Google Scholar] [CrossRef]
- Miyamoto, J.; Watanabe, K.; Taira, S.; Kasubuchi, M.; Li, X.; Irie, J.; Itoh, H.; Kimura, I. Barley β-glucan improves metabolic condition via short-chain fatty acids produced by gut microbial fermentation in high fat diet fed mice. PLoS ONE 2018, 13, e0196579. [Google Scholar] [CrossRef] [PubMed]
- Hooda, S.; Matte, J.J.; Vasanthan, T.; Zijlstra, R.T. Dietary Oat β-Glucan Reduces Peak Net Glucose Flux and Insulin Production and Modulates Plasma Incretin in Portal-Vein Catheterized Grower Pigs. J. Nutr. 2010, 140, 1564–1569. [Google Scholar] [CrossRef] [PubMed]
- King, D.E. Dietary fiber, inflammation, and cardiovascular disease. Mol. Nutr. Food Res. 2005, 49, 594–600. [Google Scholar] [CrossRef] [PubMed]
- Liatis, S.; Tsapogas, P.; Chala, E.; Dimosthenopoulos, C.; Kyriakopoulos, K.; Kapantais, E.; Katsilambros, N. The consumption of bread enriched with betaglucan reduces LDL-cholesterol and improves insulin resistance in patients with type2 diabetes. Diabetes Metab. 2009, 35, 115–120. [Google Scholar] [CrossRef]
- Lobato, R.V.; Silva, V.D.O.; Andrade, E.F.; Orlando, D.R.; Zangeronimo, M.G.; De Sousa, R.V.; Pereira, L.J. Metabolic effects of ?-glucans (saccharomyces cerevisae) per os administration in rats with streptozotocin-induced diabetes. Nutrición Hospitalaria 2015, 32, 256–264. [Google Scholar]
- Reyna-Villasmil, N.; Cano, C.; Bermúdez, V.J.; Medina, M.T.; Souki, A.J.; Ambard, M.; Nuñez, M.; Ferrer, M.A.; Inglett, G.E. Sweeteners and Beta-Glucans Improve Metabolic and Anthropometrics Variables in Well Controlled Type 2 Diabetic Patients. Am. J. Ther. 2003, 10, 438–443. [Google Scholar] [CrossRef]
- Battilana, P.; Ornstein, K.; Minehira, K.; Schwarz, J.M.; Acheson, K.; Schneiter, P.; Burri, J.; Jéquier, E.; Tappy, L. Mechanisms of action of β-glucan in postprandial glucose metabolism in healthy men. Eur. J. Clin. Nutr. 2001, 55, 327–333. [Google Scholar] [CrossRef]
- Choi, J.S.; Kim, H.; Jung, M.H.; Hong, S.; Song, J. Consumption of barley β-glucan ameliorates fatty liver and insulin resistance in mice fed a high-fat diet. Mol. Nutr. Food Res. 2010, 54, 1004–1013. [Google Scholar] [CrossRef]
- Brockman, D.A.; Chen, X.; Gallaher, D.D. Consumption of a high β-glucan barley flour improves glucose control and fatty liver and increases muscle acylcarnitines in the Zucker diabetic fatty rat. Eur. J. Nutr. 2012, 52, 1743–1753. [Google Scholar] [CrossRef]
- Belobrajdic, D.P.; Jobling, S.A.; Morell, M.K.; Taketa, S.; Bird, A.R. Wholegrain barley β-glucan fermentation does not improve glucose tolerance in rats fed a high-fat diet. Nutr. Res. 2015, 35, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Mälkki, Y.; Virtanen, E. Gastrointestinal Effects of Oat Bran and Oat Gum: A Review. LWT 2001, 34, 337–347. [Google Scholar] [CrossRef]
- Atanasov, J.; Schloermann, W.; Trautvetter, U.; Glei, M. The effects of β-glucans on intestinal health. Ernahrungs Umschau 2020, 67, 52–59. [Google Scholar] [CrossRef]
- Shen, X.J.; Rawls, J.F.; Randall, T.A.; Burcall, L.; Mpande, C.N.; Jenkins, N.; Jovov, B.; Abdo, Z.; Sandler, R.S.; Keku, T.O. Molecular characterization of mucosal adherent bacteria and associations with colorectal adenomas. Gut Microbes 2010, 1, 138–147. [Google Scholar] [CrossRef]
- Drzikova, B.; Dongowski, G.; Gebhardt, E. Dietary fibre-rich oat-based products affect serum lipids, microbiota, formation of short-chain fatty acids and steroids in rats. Br. J. Nutr. 2005, 94, 1012–1025. [Google Scholar] [CrossRef]
- Jayachandran, M.; Chen, J.; Chung, S.S.M.; Xu, B. A critical review on the impacts of β-glucans on gut microbiota and human health. J. Nutr. Biochem. 2018, 61, 101–110. [Google Scholar] [CrossRef]
- Ryan, P.M.; London, L.E.E.; Bjorndahl, T.C.; Mandal, R.; Murphy, K.; Fitzgerald, G.F.; Shanahan, F.; Ross, R.P.; Wishart, D.S.; Caplice, N.M.; et al. Microbiome and metabolome modifying effects of several cardiovascular disease interventions in apo-E-/- mice. Microbiome 2017, 5, 30. [Google Scholar] [CrossRef]
- Swennen, K.; Courtin, C.M.; Delcour, J.A. Non-digestible Oligosaccharides with Prebiotic Properties. Crit. Rev. Food Sci. Nutr. 2006, 46, 459–471. [Google Scholar] [CrossRef]
- Chambers, E.S.; Viardot, A.; Psichas, A.; Morrison, D.J.; Murphy, K.G.; Zac-Varghese, S.E.K.; MacDougall, K.; Preston, T.; Tedford, C.; Finlayson, G.S.; et al. Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults. Gut 2014, 64, 1744–1754. [Google Scholar] [CrossRef]
- Synytsya, A.; Novak, M. Structural analysis of glucans. Ann. Transl. Med. 2014, 2, 17. [Google Scholar]
- Synytsya, A.; Novák, M. Structural diversity of fungal glucans. Carbohydr. Polym. 2013, 92, 792–809. [Google Scholar] [CrossRef] [PubMed]
- C.Ooi, V.; Liu, V.E.C.A.F. Immunomodulation and Anti-Cancer Activity of Polysaccharide-Protein Complexes. Curr. Med. Chem. 2000, 7, 715–729. [Google Scholar] [CrossRef]
- Jesenak, M.; Majtan, J.; Rennerova, Z.; Kyselovic, J.; Banovcin, P.; Hrubisko, M. Immunomodulatory effect of pleuran (β-glucan from Pleurotus ostreatus) in children with recurrent respiratory tract infections. Int. Immunopharmacol. 2013, 15, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, E.; Shoji, S.; Nishihara, M.; Shimoda, T.; Nishima, S. Contribution of Lung Fibroblast Migration in the Fibrotic Process of Airway Remodeling in Asthma. Allergol. Int. 2008, 57, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Fuller, R.; Butt, H.; Noakes, P.S.; Kenyon, J.; Yam, T.S.; Calder, P.C. Influence of yeast-derived 1,3/1,6 glucopolysaccharide on circulating cytokines and chemokines with respect to upper respiratory tract infections. Nutrition 2012, 28, 665–669. [Google Scholar] [CrossRef] [PubMed]
- Kofuji, K.; Aoki, A.; Tsubaki, K.; Konishi, M.; Isobe, T.; Murata, Y. Antioxidant Activity ofβ-Glucan. ISRN Pharm. 2012, 2012, 125864. [Google Scholar] [CrossRef]
- Yamada, J.; Hamuro, J.; Hatanaka, H.; Hamabata, K.; Kinoshita, S. Alleviation of seasonal allergic symptoms with superfine β-1,3-glucan: A randomized study. J. Allergy Clin. Immunol. 2007, 119, 1119–1126. [Google Scholar] [CrossRef]
- Vaclav, V.; Josef, R.; Vladimir, S.; Lucie, R.D.; Vlastimil, K. Placebo-driven clinical trials of transfer point glucan #300 in children with chronic respiratory problems: III. clinical findings. Am. J. Immunol. 2013, 9, 88–93. [Google Scholar] [CrossRef]
- Fang, J.; Wang, Y.; Lv, X.; Shen, X.; Ni, X.; Ding, K. Structure of a β-glucan from Grifola frondosa and its antitumor effect by activating Dectin-1/Syk/NF-κB signaling. Glycoconj. J. 2012, 29, 365–377. [Google Scholar] [CrossRef]
- Dalonso, N.; Goldman, G.H.; Gern, R.M.M. β-(1→3),(1→6)-Glucans: Medicinal activities, characterization, biosynthesis and new horizons. Appl. Microbiol. Biotechnol. 2015, 99, 7893–7906. [Google Scholar] [CrossRef]
- Baert, K.; Sonck, E.; Goddeeris, B.M.; Devriendt, B.; Cox, E. Cell type-specific differences in β-glucan recognition and signalling in porcine innate immune cells. Dev. Comp. Immunol. 2015, 48, 192–203. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Fukuda, T.; Donai, K.; Kuroda, K.; Masuda, M.; Nakamura, S.; Yoneyama, H.; Isogai, E. Leptospiral lipopolysaccharide stimulates the expression of toll-like receptor 2 and cytokines in pig fibroblasts. Anim. Sci. J. 2014, 86, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Kanjan, P.; Sahasrabudhe, N.M.; De Haan, B.J.; De Vos, P. Immune effects of β-glucan are determined by combined effects on Dectin-1, TLR2, 4 and 5. J. Funct. Foods 2017, 37, 433–440. [Google Scholar] [CrossRef]
- Xu, X.; Chen, P.; Zhang, L.; Ashida, H. Chain structures of glucans from Lentinus edodes and their effects on NO production from RAW 264.7 macrophages. Carbohydr. Polym. 2012, 87, 1855–1862. [Google Scholar] [CrossRef]
- Jedinak, A.; Dudhgaonkar, S.; Wu, Q.; Simon, J.E.; Sliva, D. Anti-inflammatory activity of edible oyster mushroom is mediated through the inhibition of NF-κB and AP-1 signaling. Nutr. J. 2011, 10, 52. [Google Scholar] [CrossRef]
- Soltys, J.; Quinn, M.T. Modulation of Endotoxin- and Enterotoxin-Induced Cytokine Release by In Vivo Treatment with β-(1,6)-Branched β-(1,3)-Glucan. Infect. Immun. 1999, 67, 244–252. [Google Scholar] [CrossRef]
- Johnson, E.; Førland, D.T.; Saetre, L.; Bernardshaw, S.V.; Lyberg, T.; Hetland, G.; Sætre, L. Effect of an Extract Based on the Medicinal MushroomAgaricus blazeiMurill on Release of Cytokines, Chemokines and Leukocyte Growth Factors in Human BloodEx VivoandIn Vivo. Scand. J. Immunol. 2009, 69, 242–250. [Google Scholar] [CrossRef]
- Brown, G.D.; Herre, J.; Williams, D.L.; Willment, J.A.; Marshall, A.S.J.; Gordon, S. Dectin-1 Mediates the Biological Effects of β-Glucans. J. Exp. Med. 2003, 197, 1119–1124. [Google Scholar] [CrossRef]
- Gantner, B.N.; Simmons, R.M.; Canavera, S.J.; Akira, S.; Underhill, D.M. Collaborative Induction of Inflammatory Responses by Dectin-1 and Toll-like Receptor. J. Exp. Med. 2003, 197, 1107–1117. [Google Scholar] [CrossRef]
- Herre, J. Dectin-1 uses novel mechanisms for yeast phagocytosis in macrophages. Blood 2004, 104, 4038–4045. [Google Scholar] [CrossRef]
- Smeekens, S.P.; Gresnigt, M.S.; Becker, K.L.; Cheng, S.-C.; Netea, S.A.; Jacobs, L.; Jansen, T.; Van De Veerdonk, F.L.; Williams, D.L.; Joosten, L.A.; et al. An anti-inflammatory property of Candida albicans β-glucan: Induction of high levels of interleukin-1 receptor antagonist via a Dectin-1/CR3 independent mechanism. Cytokine 2015, 71, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.; Masterson, C.; Rezoagli, E.; O’Toole, D.; Laffey, J.; Major, I.; Stack, G.; Rowan, N. Immunomodulation Properties of a Novel β-Glucan Extract from the Mushroom Lentinus Edodes in an In-Vitro Lung Injury Model. In ALUNG INJURY, SEPSIS, AND ARDS; American Thoracic Society: New York, NY, USA, 2019; p. A2114. [Google Scholar]
- Vetvicka, V.; Vetvickova, J. Glucan supplementation enhances the immune response against an influenza challenge in mice. Ann. Transl. Med. 2015, 3, 22. [Google Scholar] [PubMed]
- Rasmussen, L.-T.; Fandrem, J.; Seljelid, R. Dynamics of Blood Components and Peritoneal Fluid during Treatment of Murine E. coli Sepsis with beta-1,3-D-polyglucose Derivatives. Scand. J. Immunol. 1990, 32, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Onderdonk, A.B.; Cisneros, R.L.; Hinkson, P.; Ostroff, G. Anti-infective effect of poly-beta 1-6-glucotriosyl-beta 1-3-glucopyranose glucan in vivo. Infect. Immun. 1992, 60, 1642–1647. [Google Scholar] [CrossRef] [PubMed]
- Cisneros, R.L.; Gibson, F.C.; Tzianabos, A.O. Passive transfer of poly-(1-6)-beta-glucotriosyl-(1-3)-beta-glucopyranose glucan protection against lethal infection in an animal model of intra-abdominal sepsis. Infect. Immun. 1996, 64, 2201–2205. [Google Scholar] [CrossRef] [PubMed]
- Tzianabos, A.O.; Gibson, F.C.; Cisneros, R.L.; Kasper, D.L. Protection against experimental intraabdominal sepsis by two polysaccharide immunomodulators. J. Infect. Dis. 1998, 178, 200–206. [Google Scholar] [CrossRef][Green Version]
- Liang, J.; Melican, D.; Cafro, L.; Palace, G.; Fisette, L.; Armstrong, R.; Patchen, M.L. Enhanced clearance of a multiple antibiotic resistant Staphylococcus aureus in rats treated with PGG-glucan is associated with increased leukocyte counts and increased neutrophil oxidative burst activity. Int. J. Immunopharmacol. 1998, 20, 595–614. [Google Scholar] [CrossRef]
- Senoglu, N.; Yuzbasioglu, M.F.; Aral, M.; Ezberci, M.; Kurutas, E.B.; Bulbuloglu, E.; Oksuz, H.; Ciragil, P.; Ezberci, F. Protective Effects ofN-Acetylcysteine and β -Glucan Pretreatment on Oxidative Stress in Cecal Ligation and Puncture Model of Sepsis. J. Investig. Surg. 2008, 21, 237–243. [Google Scholar] [CrossRef]
- Gülmen, Ş.; Kiris, I.; Kocyigit, A.; Dogus, D.K.; Ceylan, B.G.; Meteoglu, I. β-Glucan Protects against Lung Injury Induced by Abdominal Aortic Ischemia-Reperfusion in Rats. J. Surg. Res. 2010, 164, e325–e332. [Google Scholar] [CrossRef]
- Babayigit, H.; Kucuk, C.; Sozuer, E.; Yazici, C.; Kose, K.; Akgun, H. Protective effect of β-glucan on lung injury after cecal ligation and puncture in rats. Intensive Care Med. 2005, 31, 865–870. [Google Scholar] [CrossRef]
- Bedirli, A.; Kerem, M.; Pasaoglu, H.; Akyurek, N.; Tezcaner, T.; Elbeg, S.; Memis, L.; Sakrak, O. Beta-glucan attenuates inflammatory cytokine release and prevents acute lung injury in an experimental model of sepsis. Shock 2007, 27, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.S.H.; Jonas, A.B.; Qiu, X.; Ottoson, N.R.; Walsh, R.M.; Gorden, K.B.; Harrison, B.; Maimonis, P.J.; Leonardo, S.M.; Ertelt, K.E.; et al. Imprime PGG-Mediated Anti-Cancer Immune Activation Requires Immune Complex Formation. PLoS ONE 2016, 11, e0165909. [Google Scholar] [CrossRef] [PubMed]
- Bose, N.; Chan, A.S.H.; Eguerrero, F.; Maristany, C.M.; Eqiu, X.; Walsh, R.; Ertelt, K.E.; Jonas, A.B.; Gorden, K.B.; Dudney, C.M.; et al. Binding of Soluble Yeast β-Glucan to Human Neutrophils and Monocytes is Complement-Dependent. Front. Immunol. 2013, 4, 230. [Google Scholar] [CrossRef] [PubMed]
- Hong, F.; Hansen, R.D.; Yan, J.; Allendorf, D.J.; Baran, J.T.; Ostroff, G.R.; Ross, G.D. β-Glucan Functions as an Adjuvant for Monoclonal Antibody Immunotherapy by Recruiting Tumoricidal Granulocytes as Killer Cells. Cancer Res. 2003, 63, 9023–9031. [Google Scholar] [PubMed]
- Vetvicka, V.; Dvořák, B.; Vetvickova, J.; Richter, J.; Krizan, J.; Sima, P.; Yvin, J.-C. Orally administered marine (1→3)-β-d-glucan Phycarine stimulates both humoral and cellular immunity. Int. J. Biol. Macromol. 2007, 40, 291–298. [Google Scholar] [CrossRef]
- Thandapilly, S.J.; Ndou, S.P.; Wang, Y.; Nyachoti, C.M.; Ames, N. Barley β-glucan increases fecal bile acid excretion and short chain fatty acid levels in mildly hypercholesterolemic individuals. Food Funct. 2018, 9, 3092–3096. [Google Scholar] [CrossRef]
- Wang, Y.; Harding, S.V.; Thandapilly, S.J.; Tosh, S.M.; Jones, P.J.H.; Ames, N.P. Barley β-glucan reduces blood cholesterol levels via interrupting bile acid metabolism. Br. J. Nutr. 2017, 118, 822–829. [Google Scholar] [CrossRef]
- Aoe, S.; Ikenaga, T.; Noguchi, H.; Kohashi, C.; Kakumoto, K.; Kohda, N. Effect of cooked white rice with high β-glucan barley on appetite and energy intake in healthy Japanese subjects: A randomized controlled trial. Plant Foods Hum. Nutr. 2014, 69, 325–330. [Google Scholar] [CrossRef]
- Aoe, S.; Ichinose, Y.; Kohyama, N.; Komae, K.; Takahashi, A.; Abe, D.; Yoshioka, T.; Yanagisawa, T. Effects of high β-glucan barley on visceral fat obesity in Japanese individuals: A randomized, double-blind study. Nutrition 2017, 42, 1–6. [Google Scholar] [CrossRef]
- Thondre, P.S.; Shafat, A.; Clegg, M.E. Molecular weight of barley β-glucan influences energy expenditure, gastric emptying and glycaemic response in human subjects. Br. J. Nutr. 2013, 110, 2173–2179. [Google Scholar] [CrossRef]
- Zaremba, S.M.; Gow, I.F.; Drummond, S.; McCluskey, J.T.; Steinert, R.E. Effects of oat β-glucan consumption at breakfast on ad libitum eating, appetite, glycemia, insulinemia and GLP-1 concentrations in healthy subjects. Appetite 2018, 128, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Pentikäinen, S.P.; Karhunen, L.; Flander, L.; Katina, K.; Meynier, A.; Aymard, P.; Vinoy, S.; Poutanen, K. Enrichment of biscuits and juice with oat β-glucan enhances postprandial satiety. Appetite 2014, 75, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Rebello, C.J.; Chu, Y.; Johnson, W.D.; Martin, C.K.; Han, H.; Bordenave, N.; Shi, Y.; O’Shea, M.; Greenway, F.L. The role of meal viscosity and oat β-glucan characteristics in human appetite control: A randomized crossover trial. Nutr. J. 2014, 13, 49. [Google Scholar] [CrossRef] [PubMed]
- Thongoun, P.; Pavadhgul, P.; Bumrungpert, A.; Satitvipawee, P.; Harjani, Y.; Kurilich, A. Effect of oat consumption on lipid profiles in hypercholesterolemic adults. J. Med. Assoc. Thail. Chotmaihet Thangphaet 2013, 96, 25. [Google Scholar]
- Queenan, K.M.; Stewart, M.L.; Smith, K.N.; Thomas, W.; Fulcher, R.G.; Slavin, J.L. Concentrated oat β-glucan, a fermentable fiber, lowers serum cholesterol in hypercholesterolemic adults in a randomized controlled trial. Nutr. J. 2007, 6, 6–8. [Google Scholar] [CrossRef]
- Pavadhgul, P.; Bumrungpert, A.; Harjani, Y.; Kurilich, A. Oat porridge consumption alleviates markers of inflammation and oxidative stress in hypercholesterolemic adults. ASIA Pac. J. Clin. Nutr. 2019, 28, 260–265. [Google Scholar]
- De Souza, S.R.; De Oliveira, G.M.M.; Luiz, R.R.; Rosa, G. Effects of oat bran and nutrition counseling on the lipid and glucose profile and anthropometric parameters of hypercholesterolemia patients. Nutrición Hospitalaria 2016, 33, 123–130. [Google Scholar] [CrossRef]
- Aarsæther, E.; Rydningen, M.; Engstad, R.E.; Busund, R. Cardioprotective effect of pretreatment with β-glucan in coronary artery bypass grafting. Scand. Cardiovasc. J. 2006, 40, 298–304. [Google Scholar] [CrossRef]
- Davidson, M.H.; Dugan, L.D.; Burns, J.H.; Bova, J.; Story, K.; Drennan, K.B. The Hypocholesterolemic Effects of β-Glucan in Oatmeal and Oat Bran. JAMA 1991, 265, 1833–1839. [Google Scholar] [CrossRef]
- Lia, A.; Hallmans, G.; Sandberg, A.S.; Sundberg, B.; Aman, P.; Andersson, H. Oat beta-glucan increases bile acid excretion and a fiber-rich barley fraction increases cholesterol excretion in ileostomy subjects. Am. J. Clin. Nutr. 1995, 62, 1245–1251. [Google Scholar] [CrossRef]
- Lovegrove, J.A.; Clohessy, A.; Milon, H.; Williams, C.M. Modest doses of β-glucan do not reduce concentrations of potentially atherogenic lipoproteins. Am. J. Clin. Nutr. 2000, 72, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Velikonja, A.; Lipoglavšek, L.; Zorec, M.; Orel, R.; Avguštin, G. Alterations in gut microbiota composition and metabolic parameters after dietary intervention with barley beta glucans in patients with high risk for metabolic syndrome development. Anaerobe 2019, 55, 67–77. [Google Scholar] [CrossRef]
- Wang, Y.; Harding, S.V.; Eck, P.; Thandapilly, S.J.; Gamel, T.H.; Abdel-Aal, E.-S.M.; Crow, G.H.; Tosh, S.M.; Jones, P.J.H.; Ames, N. High-Molecular-Weight β-Glucan Decreases Serum Cholesterol Differentially Based on the CYP7A1 rs3808607 Polymorphism in Mildly Hypercholesterolemic Adults. J. Nutr. 2015, 146, 720–727. [Google Scholar] [CrossRef] [PubMed]
- Abrams, D.I.; Couey, P.; Shade, S.B.; Kelly, M.E.; Kamanu-Elias, N.; Stamets, P. Antihyperlipidemic effects of Pleurotus ostreatus (oyster mushrooms) in HIV-infected individuals taking antiretroviral therapy. BMC Complement. Altern. Med. 2011, 11, 60. [Google Scholar] [CrossRef]
- Varricchio, A.M.; Capasso, M.; Della Volpe, A.; Malafronte, L.; Mansi, N.; Varricchio, A.; Ciprandi, G. Resveratrol plus carboxymethyl-β-glucan in children with recurrent respiratory infections: A preliminary and real-life experience. Ital. J. Pediatr. 2014, 40, 93. [Google Scholar] [CrossRef] [PubMed]
- Gaullier, J.-M.; Sleboda, J.; Ofjord, E.S.; Ulvestad, E.; Nurminiemi, M.; Moe, C.; Albrektsen, T.; Gudmundsen, O. Supplementation with a Soluble Beta-Glucan Exported from Shiitake Medicinal Mushroom, Lentinus edodes (Berk.) Singer Mycelium: A Crossover, Placebo-Controlled Study in Healthy Elderly. Int. J. Med. Mushrooms 2011, 13, 319–326. [Google Scholar] [CrossRef]
- Browder, W.; Williams, D.; Pretus, H.; Olivero, G.; Enrichens, F.; Mao, P.; Franchello, A. Beneficial effect of enhanced macrophage function in the trauma patient. Ann. Surg. 1990, 211, 605–613. [Google Scholar]
- Júnior, J.D.F.; Júnior, M.D.R.E.S.; Maciel, F.M.; Soares, A.D.M.; Mendes, N.F. Infection prevention in patients with severe multiple trauma with the immunomodulator beta 1-3 polyglucose (glucan). Surg. Gynecol. Obstet. 1993, 177, 383–388. [Google Scholar]
- Babineau, T.J.; Marcello, P.; Swails, W.; Kenler, A.; Bistrian, B.; Forse, R.A. Randomized Phase I/II Trial of a Macrophage-Specific Immunomodulator (PGG-Glucan) in High-Risk Surgical Patients. Ann. Surg. 1994, 220, 601–609. [Google Scholar] [CrossRef]
- Babineau, T.J.; Hackford, A.; Kenler, A.; Bistrian, B.; Forse, R.A.; Fairchild, P.G.; Heard, S.; Keroack, M.; Caushaj, P.; Benotti, P.N. A Phase II Multicenter, Double-blind, Randomized, Placebo-Controlled Study of Three Dosages of an Immunomodulator (PGG-Glucan) in High-Risk Surgical Patients. Arch. Surg. 1994, 129, 1204–1210. [Google Scholar] [CrossRef]
- Dellinger, E.P.; Babineau, T.J.; Bleicher, P.; Kaiser, A.B.; Seibert, G.B.; Postier, R.G.; Vogel, S.B.; Norman, J.; Kaufman, D.; Galandiuk, S.; et al. Effect of PGG-glucan on the Rate of Serious Postoperative Infection or Death Observed After High-Risk Gastrointestinal Operations. Arch. Surg. 1999, 134, 977–983. [Google Scholar] [CrossRef] [PubMed]
- Leentjens, J.; Quintin, J.; Gerretsen, J.; Kox, M.; Pickkers, P.; Moorlag, S.J. The Effects of Orally Administered Beta-Glucan on Innate Immune Responses in Humans, a Randomized Open-Label Intervention Pilot-Study. PLoS ONE 2014, 9, e108794. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, T. Effects of lentinan on the patients with advanced or recurrent gastric, colorectal and breast cancer. Jpn. J. Cancer Chemother. 1983, 10, 387–393. [Google Scholar]
- Wesa, K.M.; Cunningham-Rundles, S.; Klimek, V.M.; Vertosick, E.; Coleton, M.I.; Yeung, K.S.; Lin, H.; Nimer, S.; Cassileth, B.R. Maitake mushroom extract in myelodysplastic syndromes (MDS): A phase II study. Cancer Immunol. Immunother. 2015, 64, 237–247. [Google Scholar] [CrossRef]
- Weitberg, A.B. A phase I/II trial of beta-(1,3)/(1,6) D-glucan in the treatment of patients with advanced malignancies receiving chemotherapy. J. Exp. Clin. Cancer Res. 2008, 27, 40. [Google Scholar] [CrossRef]
- Zent, C.S.; Call, T.G.; Bowen, D.A.; Conte, M.J.; LaPlant, B.R.; Witzig, T.E.; Ansell, S.M.; Weiner, G.J. Early treatment of high risk chronic lymphocytic leukemia with alemtuzumab, rituximab and poly-(1-6)-beta-glucotriosyl-(1-3)- beta-glucopyranose beta-glucan is well tolerated and achieves high complete remission rates. Leuk. Lymphoma 2015, 56, 2373–2378. [Google Scholar] [CrossRef]
- Thomas, M.; Sadjadian, P.; Kollmeier, J.; Lowe, J.; Mattson, P.; Trout, J.R.; Gargano, M.; Patchen, M.L.; Walsh, R.; Beliveau, M.; et al. A randomized, open-label, multicenter, phase II study evaluating the efficacy and safety of BTH1677 (1,3–1,6 beta glucan; Imprime PGG) in combination with cetuximab and chemotherapy in patients with advanced non-small cell lung cancer. Investig. New Drugs 2017, 35, 345–358. [Google Scholar] [CrossRef]
- Engel-Riedel, W.; Schneller, F.; Wolf, M.; Schuette, W.; Lowe, J.; Mattson, P.; Gargano, M.; Patchen, M.L.; Huhn, R.; Braun, A. Imprime Pgg, a Novel Immune Modulator, in the 1St-Line Treatment of Stage Iv Nsclc: Results from a Randomized, Controlled, Multicenter Phase 2 Study. Ann. Oncol. 2014, 25, v1. [Google Scholar] [CrossRef]
- Du, B.; Meenu, M.; Liu, H.; Xu, B. A Concise Review on the Molecular Structure and Function Relationship of β-Glucan. Int. J. Mol. Sci. 2019, 20, 4032. [Google Scholar] [CrossRef]
- Cui, W.; Wood, P. Relationships between structural features, molecular weight and rheological properties of cereal β-D-glucans. Hydrocolloids 2000, 159–168. [Google Scholar] [CrossRef]
- Lazaridou, A.; Biliaderis, C. Molecular aspects of cereal β-glucan functionality: Physical properties, technological applications and physiological effects. J. Cereal Sci. 2007, 46, 101–118. [Google Scholar] [CrossRef]
- Bohn, J.A.; BeMiller, J.N. (1→3)-β-d-Glucans as biological response modifiers: A review of structure-functional activity relationships. Carbohydr. Polym. 1995, 28, 3–14. [Google Scholar] [CrossRef]
- Yuan, H.; Lan, P.; He, Y.; Li, C.; Ma, X. Effect of the Modifications on the Physicochemical and Biological Properties of β-Glucan—A Critical Review. Molecules 2019, 25, 57. [Google Scholar] [CrossRef] [PubMed]
- Castro, E.D.M.; Calder, P.C.; Roche, H.M. β-1,3/1,6-Glucans and Immunity: State of the Art and Future Directions. Mol. Nutr. Food Res. 2020, 1901071. [Google Scholar] [CrossRef]
| Areas of Research | Study Title | NCT | Design | β-glucan Type | Dose | Population |
|---|---|---|---|---|---|---|
| Microbiota and gastrointestinal health | Characterization of the Gut Microbiota Composition and Activity After Three Weeks of Chitin-glucan Supplementation | NCT03505177 | n/a | Chitin-glucan | 4.5 g/day | Healthy |
| Characterization of Chitin-glucan Fibre Fermentation in Human After a Single Administration | NCT03494491 | n/a | Chitin-glucan | 4.5 g/day | Healthy | |
| Effect of 6 Weeks Daily Consumption of a Cereal-based Juice Beverage on Gastrointestinal Health (NEWDRINK) | NCT03046667 | n/a | Barley | Drink = 1 dose per day—dose not stated | Irritable Bowel | |
| Characterization of Gut Microbiota Composition and Activity After a Daily Supplementation of 4.5 g/Day of Chitin Glucan Fibre during 3 Weeks in At-cardiometabolic Risk Volunteers (FITACHITIN) | NCT03773900 | n/a | Chitin | 1.5 g/3 times daily | Cardiometabolic Risk Abdominal Obesity | |
| β-glucan on Faecal Microflora in Polypectomised Patients | NCT00893659 | n/a | Unknown | 3 g/day | Polypectomised Patients | |
| Synbiotics and Gastrointestinal Function Related Quality of Life After Colectomy for Cancer | NCT01479907 | n/a | Unknown | 2.5 g/sachet | Colorectal Neoplasms | |
| The Effect of Oats Containing 1.4g β Glucan on Faecal Bacterial Population(s) and Plasma Cholesterol in Healthy Adults with Elevated Cholesterol Levels | NCT03450395 | n/a | Oats | 40 g of crude oats/day | Microbiome Plasma Cholesterol Prebiotic | |
| Beta-glucan Effects on Lipid Profile, Glycemia and intestinal Health (BELT) (BELT) | NCT03313713 | n/a | Unknown | 3 g/day | Atherosclerosis | |
| Healthy Effects of an Innovative Probiotic Pasta (SFLABPASTA) | NCT02236533 | n/a | Barley | Pasta once a day | Obesity, Inflammation, Dyslipidaemia, Constipation | |
| The Effectiveness of Pleuran in Treatment of Acute Gastroenteritis in Children (EPTAGE) | NCT03988257 | Phase 2 | Mushroom | 10 mg Pleuran | Diarrhoea; Acute | |
| Impact of Consumption of Beta-glucans on the Intestinal Microbiota and Glucose and Lipid Metabolism | NCT02041104 | n/a | Barley | 6 g/day | Metabolic Syndrome, Dyslipidaemia, Obesity, Abdominal, Hyperglycaemia, Hypertension | |
| Combined Nutritional Therapies for the Treatment of Ulcerative Colitis | NCT03444311 | n/a | Oat | Unknown | Colitis, Ulcerative | |
| Prebiotic Supplementation and Intestinal Barrier Function in Elderly: an RCT | NCT03336385 | n/a | Oat | Daily—dose not known | Prebiotics, Aged | |
| Chronic Cardiovascular and Gut-bacteria Effects of Phenolic Rich Oats in Adults with Above Average Blood Pressure | NCT02847312 | n/a | Oat | 60 g of Oat cake | Healthy | |
| The Effect of Hot Cereal on Digestive Health in Children | NCT02868515 | n/a | Oat | 3 g/day | Subjective Measures of Digestive, Health Post Consumption | |
| Dietary Fibres Effect on the Gut Microbiota Composition | NCT04114513 | n/a | Unknown Tate & Lyle powder | 2 increasing to 8 g per day | Microbiome, Metabolic Syndrome, Cardiovascular Risk Factor, Inflammation, Dyslipidaemias | |
| β -1,3/1,6-D-Glucan Ganoderma Lucidum on Ulcerative Colitis | NCT04029649 | Phase 2, Phase 3 | Fungal | Capsule containing 180 mg/three times daily | Ulcerative Colitis | |
| Glycemic control; Diabetes | β glucan and Acetate Production | NCT03714646 | n/a | Unknown | 12 g once | Pre-Diabetes, Obesity |
| Inulin and Acetate Production and Human Substrate Metabolism | NCT03711383 | n/a | Unknown | Unknown | Obesity, Pre-Diabetes | |
| Efficacy and Safety Study of Soluble Beta-1,3/1,6-glucan (SBG) Versus Placebo in Chronic Diabetic Foot Ulcers | NCT00804414 | Phase 3 | SBG—Yeast | Topical Application | Diabetes, Diabetic Ulcer | |
| The Glycaemic Response Elicited by β-glucans of Different Physical Properties and Form | NCT01610518 | n/a | Oat | 4 g | Type 2 Diabetes | |
| Effect of Serving Size and Addition of Sugar on the Glycaemic Response Elicited by Oatmeal (Panther) | NCT02506972 | n/a | Oat | 30 g of oats | Diabetes Mellitus | |
| Effect of Viscous Soluble Fibres on Body Weight | NCT03257449 | n/a | Oat, Barley | Unknown | Overweight and Obesity, T2DM (Type 2 Diabetes Mellitus), General Population | |
| Impact of DHA/Oat on Metabolic Health in Gestational Diabetes Mellitus | NCT03569501 | n/a | Oat | 4.05 mg/day | Gestational Diabetes Mellitus in Pregnancy | |
| Efficacy of Soluble β-1,3/1,6-Glucan Compared to Placebo on Chronic Leg Ulcers in Diabetes Patients | NCT00288392 | Phase 2 | SBG—Yeast | Unknown | Foot Ulcer | |
| Food Modification to Alter Glycaemia and Insulinaemia | NCT03706378 | n/a | Yellow Noodle—wheat | 50 g of β-glucan in 230.4 g of yellow noodle per day | Diabetes | |
| The Glycaemic Response of Local Foods Using the Continuous Glucose Monitoring System | NCT03703544 | n/a | Yellow Noodle—wheat | Unknown | Diabetes | |
| Oat β-glucan as a Supplement in Chilean Type 2 Diabetics | NCT04299763 | Phase 2 | Oat | 5 g daily with breakfast | Type2 Diabetes | |
| The Effect of Insoluble Yeast β-glucan Intake on Pre-diabetic Patients | NCT03495362 | n/a | Yeast | 500 mg capsule twice a day | Pre-diabetic | |
| Effects of (1,3), (1,6)- β-D-glucan on Insulin Sensitivity and Inflammatory Markers of the Metabolic Syndrome | NCT00403689 | n/a | Yeast | 1.5 g/daily | Overweight | |
| Intake of Beta-glucan and Postprandial Regulation of Blood Glucose Metabolism in Healthy Subjects | NCT03293693 | n/a | unknown | 0.5 g–8 g | Post Prandial Blood Glucose, Gut Microbiota, Satiety | |
| The Effect of Content of Barley Beta-glucans in Bread on Postprandial Blood Sugar (ARRS-bGL-01) | NCT03878576 | n/a | Barley | 25 g | Glycaemic Index | |
| Evaluation of Woulgan in Diabetic Foot Ulcer | NCT02631512 | Phase 4 | Woulgan-contains SBG—yeast | Gel Application | Diabetic Foot Ulcers | |
| A Study of the Effect of Oats on Post Prandial Glucose Response | NCT02651597 | n/a | Oat | Unknown | Normoglycemic, Normal Body Weight | |
| Effects of Barley on Glucose Control | NCT02367989 | n/a | Barley | 2–6 g/day | Healthy | |
| Barley and Rice Mixture Effects on Blood Glucose | NCT03387345 | n/a | Barley | Unknown | Blood Glucose, Dietary Fibre, Hunger | |
| Lipid Regulation | Effect of the Molecular Weight of Oat β-glucan on Its Ability to Lower Serum Cholesterol (Bluebird) | NCT00981981 | Phase 2 | Oat | 3–4 g/day | Hypercholesterolemia |
| Effect of Beta-glucan on Cholesterol Lowering | NCT01408719 | n/a | Barley | 3–5 g β glucan | Hypercholesterolemia | |
| The Effect of Viscous Dietary Fibres on LDL-cholesterol | NCT04133805 | n/a | Barley, Oat | Unknown | Cardiovascular Risk Factor, Hypercholesterolemia | |
| Oat and Cholesterol | NCT03911427 | n/a | Oat | Powdered sachets three times daily | Elevated LDL Cholesterol | |
| Impact of Consumption of Oats in Lipid Profile of Children and Adolescents with Dyslipidaemia | NCT01581697 | Phase 1, Phase 2 | Oat bran | 3 g with 3 meals a day | Atherosclerosis, Hypercholesterolemia | |
| Nutritional Counselling Associated with the Ingestion of Oat Bran in Hypercholesterolemic Subjects | NCT02189200 | n/a | Oat | 40 g oat bran per day | Dietary Modification | |
| Effects of Lentinula Edodes Bars on Dyslipidaemia and Oxidative Stress in Cholesterol Individuals: Randomized Study | NCT04186780 | n/a | Fungal | 2 cereal bars of Shiitake per day | Dyslipidaemias | |
| Effects of Chitin-glucan on Oxidized Low-Density Lipoprotein (LDL) | NCT01232309 | n/a | Chitin | 1.5 g–4.5 g of glucan | Cardiovascular | |
| Obesity and diet regulation | Effects of Oligofructose and Barley on Satiety and Energy Intake | NCT00776256 | n/a | Oats, Barley | 1 g/serving | Appetite, Regulation |
| β-glucan and Insulin Sensitivity in Obese Humans | NCT01393210 | n/a | Unknown | Unknown | Obesity | |
| Diet for the Maintenance of Weight Loss and Metabolic Health in Obese Postmenopausal Women (WELCOME) | NCT04136093 | n/a | Oat, Barley | 50 g of oatmeal and barley groats | Metabolic Syndrome, Diet Modification, Postmenopause | |
| Efficacy of Hydroxycinnamates and Beta-glucans as a Dietary Tool Against Obesity Pilot Study (OBHEALTH_PS) (OBHEALTH_PS) | NCT04321590 | n/a | Oat | 3 g or 5 g/day | Overweight, Obesity | |
| Dietary Fibres and Satiety in Bariatric Patients (FIBAR) | NCT03573258 | Early phase 1 | Oat | 6 g | Bariatric Surgery Candidate | |
| SATIN: Satiety Innovation. Study 2-University of Aberdeen (SATIN) | NCT02604316 | n/a | Viscofibre, Oat and Barley | 6 g for 10 days | Overweight, Obesity | |
| Effects of β-glucan on Energy Intake and Satiety | NCT02637388 | n/a | Oatwell, Powder—Oats | 4 g as part of breakfast | Obesity | |
| A Trial Comparing a Diet Including Products Aimed at Targeting Satiety (SATIN) | NCT02485743 | n/a | Unknown | Unknown | Weight, Appetite | |
| The Effect of a Breakfast Meal Containing Oat β-glucan on Food Intake at a Subsequent Meal in Normal-weight and Overweight Subjects | NCT03490851 | n/a | Oat | 2–4 g | Satiety | |
| Efficacy and Safety of Fermented Barley on Decrement of Body Fat in Obese Subjects | NCT01402128 | Phase 2, Phase 3 | Barley | 3 g/day | Overweight; Hyperlipidaemia | |
| The Effect of Dietary Fibre on Food Liking | NCT03241238 | n/a | Unknown | Unknown | Different Fermentable Fibre, Satiation | |
| Metabolic Syndrome | β-glucans and the Metabolic Syndrome—a Human Intervention Study Under BEST | NCT01317264 | n/a | Oat, Barley, Mutant Barley | 5 g/day | healthy |
| Effect of Dietary Fibre and Whole Grain on the Metabolic Syndrome | NCT01316354 | n/a | Unconfirmed | Bread with 50 g available carbohydrate | Metabolic Syndrome | |
| Pivotal Assessment of the Effects of Bioactive on Health and Wellbeing. From Human Genome to Food Industry (PATHWAY-27) | NCT02702713 | n/a | Oat (N.C), Pathway-27 website http://www.pathway27.eu/ | 3 g beta-glucan—not stated frequency | Metabolic Syndrome | |
| Metabolic Effect of New Foods Through Gut-brain Axis (CHECKMATE) | NCT01851304 | n/a | Barley | 3 g/100 g bread portion | Obesity, Overweight | |
| Effects of β-glucan From Barley and Oats on Glucose and Lipid Metabolism, and satiety (glucan) | NCT03648112 | n/a | Barley, Oats | 80 g crude flakes/day (oat or barley) | Lipid Metabolism, Glucose Metabolism, Satiety | |
| Effects of Dried Bilberry, Liquid Oats, or Their Combination After AMI (BIOAMI) | NCT03620266 | n/a | Glucanova®—Oat | Shakes containing oats 3 times daily, | Myocardial Infarction | |
| Influence of Dietary Fibre-rich Meals on Gene Expression and Postprandial Glucose and Lipid Response | NCT01005342 | n/a | Oat | 62–82 g—single intake | Hypoglycaemia, Hyperglycaemia | |
| The Effects of β-glucan Enriched Oatcake Consumption on Metabolic Disease Risk Factors | NCT02615444 | n/a | Oat | 4 g/day | Metabolic Syndrome X, Cardiovascular Diseases | |
| Canola Oil, Fibre and DHA Enhanced Clinical Trial | NCT02091583 | n/a | Barley | 3 g/day | Metabolic Syndrome | |
| Glycaemic Impact of Oatmeal Plus OatWellXF28 | NCT02818452 | n/a | Oat | 27 g–32.72 g of Oatmeal containing β-glucan | Glycaemic Responses | |
| Four-hour Glycaemic Kinetic Response Following 13C-enriched Oatmeal Breakfast Compared to Hot Corn Grits | NCT03165773 | n/a | Oat | 87 g oatmeal | Glycaemic and Insulinemic Response | |
| Portfolio 5—Multicentre Dietary Advice on Serum Lipids in Hyperlipidaemia | NCT00438425 | n/a | Oat, Barley | 9.8 g/1000 kcal | Hyperlipidaemia, Cardiovascular Disease | |
| Magnetic Resonance Imaging-Portfolio Diet Study #7 (MRIPD#7) | NCT02078635 | n/a | Oat, Barley | 9.8 g/1000 kcal | Cardiovascular Diseases, Hypercholesterolemia, Diabetes, Metabolic Syndrome, Obesity | |
| Effects of Chitin-glucan on Oxidized Low-Density Lipoprotein (LDL) | NCT01232309 | n/a | Chitin | 1.5 g–4.5 g daily | Cardiovascular | |
| Compare the Efficacy and Safety of β-glucan as Add-On to Statin in Subjects with Hyperlipidaemia. (BetAvena) | NCT03857256 | Phase 2 | CP105F, Oat β-glucan | 1.5 g, 3 g or 6 g daily, Administered 3 times daily | Hyperlipidaemias | |
| The Effect of Oral β-glucan Supplement on Appetite and Insulin Resistance in non-Alcoholic Fatty Liver Disease | NCT02178839 | n/a | Oat | 3 g daily | Non-Alcoholic Fatty Liver Disease | |
| ProAliFun_6.5_Health Effects of a Functional Pasta Enriched with Barley Beta-glucans on Healthy Subjects (ProAliFun65) | NCT02710513 | n/a | Barley | 100 g of β-glucan pasta/day | Healthy | |
| Clinical Trial to Evaluate the Addition to an Antiretroviral Treatment of a Probiotic (RECOVER) | NCT03542786 | n/a | Oat | As part of pro-biotic once a day for 6 months | HIV, Premature Aging |
| Areas of Research | Study Title: | NCT | Design | β-glucan Type | Dose | Population |
|---|---|---|---|---|---|---|
| Solid cancer and Haematological malignancy | Efficacy and Safety Study of SBG vs Placebo in Head and Neck Cancer Patients Undergoing Radiation Therapy | NCT00790322 | Phase III | Soluble- β-glucan SBG, Yeast derived | Not-stated | Head and Neck Cancer |
| The Protective Effect of Soluble Beta-1,3/1,6-glucan Compared to Placebo in Oral Mucositis in Head and Neck Cancer Patients | NCT00289003 | Phase II | SBG—Yeast | Unknown | Oral Mucositis | |
| Safety of Soluble β-glucan (SBG) in Treatment of Patients with Non-Hodgkin’s Lymphoma | NCT00533728 | Phase 1 | SBG, Yeast | Unknown | Non-Hodgkin’s Lymphoma | |
| Effect of SBG in Patients with Breast Cancer | NCT00533364 | Phase 1, Phase 2 | SBG, Yeast | Unknown | Breast Cancer | |
| The Effect of β-glucan in Non-Small Cell Lung Cancer | NCT00682032 | n/a | Imucell WGP-Yeast | 1 (one) 250 mg β-glucan capsule 3 times a day for 14 days | Non-Small Cell Lung Cancer | |
| Bivalent Vaccine with Escalating Doses of the Immunological Adjuvant OPT-821, in Combination with Oral β-glucan for High-Risk Neuroblastoma | NCT00911560 | Phase 1, Phase 2 | Yeast | Oral β-glucan (40 mg/kg/day) in conjunction with vaccine | Neuroblastoma | |
| β-glucan and Monoclonal Antibody 3F8 in Treating Patients with Metastatic Neuroblastoma | NCT00492167 | Phase 1 | Yeast | In conjunction with monoclonal antibody—dose-escalation study of β-glucan | Neuroblastoma | |
| Lung Cancer Vaccine Plus Oral Dietary Supplement | NCT01829373 | Phase 1 | Vaccine plus oral beta-glucan-Yeast | Unknown | Lung Cancer | |
| β-glucan and Rituximab in Treating Young Patients with Relapsed or Progressive Lymphoma or Leukaemia, or Lymphoproliferative Disorder Related to Donor Stem Cell Transplantation | NCT00087009 | Phase 1 | Unknown | Oral β-glucan in conjunction with IV Rituximab | Leukaemia, Lymphoma, Lymphoproliferative Disorder | |
| Rituximab Plus β-glucan in Chronic Lymphocytic Leukaemia (CLL)/Small Lymphocytic Lymphoma (SLL) | NCT00290407 | Phase 2 | Imucell WGP, Yeast | 250 mg, orally (tablet), three times a day for 9 weeks | Leukaemia, Lymphocytic, Chronic, Lymphoma, Small Lymphocytic | |
| β-glucan and Monoclonal Antibody in Treating Patients with Metastatic Neuroblastoma | NCT00037011 | Phase 1 | Unknown | Oral β-glucan in conjunction with IV antibody | Neuroblastoma | |
| β-glucan in Treating Patients with Locally Advanced or Metastatic non-Small Cell Lung Cancer | NCT00857025 | Phase 1 | glucan MM-10-001, Fungal | Oral β-glucan once daily | Lung Cancer | |
| Imprime PGG, Alemtuzumab, and Rituximab in Treating Patients with High-Risk Chronic Lymphocytic Leukaemia | NCT01269385 | Phase 1, Phase 2 | PGG β-glucan, Imprime PGG, Yeast | Dose escalation study IV administration | B-cell Chronic Lymphocytic Leukaemia, Refractory Chronic Lymphocytic Leukaemia, Stage 0 Chronic Lymphocytic Leukaemia, Stage I Chronic Lymphocytic Leukaemia, Stage II Chronic Lymphocytic Leukaemia | |
| (PM-01) IMPRIME PGG® With BTH1704 and Gemcitabine for Advanced Pancreatic Cancer (PM-01) | NCT02132403 | Phase 1 | IMPRIME PGG, Yeast | Assigned doses | Pancreatic Cancer | |
| A Phase 2 Clinical Trial of Rituxan and B-glucan PGG in Relapsed Indolent Non-Hodgkin Lymphoma | NCT02086175 | Phase 2 | IMPRIME PGG, Yeast | I.V 4 mg/kg weekly for 4 weeks. | Relapsed/Refractory Indolent B Cell Non-Hodgkin Lymphoma | |
| Biological Therapy in Treating Patients with Neuroblastoma That Has Not Responded to Previous Treatment | NCT00089258 | Phase 2 | Unknown | Unknown | Neuroblastoma | |
| MucoLox Formulation to Mitigate Mucositis Symptoms in Head/Neck Cancer | NCT03461354 | Phase 2 | Unknown | Unknown | Mucositis Oral, Head and Neck Cancer | |
| Phase 2 Study of Imprime PGG & Pembrolizumab in Subjects with Adv SCCHN Who Failed Pembro Monotherapy or Experiencing SD | NCT03246685 | Phase 2 | Imprime PGG, Yeast | 4 mg/kg IV over a 2-h infusion time on Days 1, 8 and 15 of each 3-week treatment cycle. | Squamous Cell Carcinoma of the Head and Neck | |
| Study of Imprime PGG and Pembrolizumab in Advanced Melanoma and Triple-Negative Breast Cancer | NCT02981303 | Phase 2 | Imprime, PGG, Yeast | 4 mg/kg IV over a 2-h infusion time | Advanced Melanoma, Triple-Negative Breast Cancer | |
| Phase I, Dose-Escalation Study of Soluble Beta-glucan (SBG) in Patients with Advanced Solid Tumours | NCT01910597 | Phase 1 | SBG -yeast | Unknown | Advanced Solid Tumours | |
| Immunomodulation | Efficacy and Safety of Resveratrol and Carbossimetyl Beta-glucan in Treatment of Upper Airways Disease in Infancy (VIRNEO) | NCT03683108 | Phase 3, Phase 3 | Carbossimetyl β glucan, Carbossimetyl β glucan | 3 drops, 4 times a day for 1 week. | Common Cold |
| Nebulized Resveratrol Plus Carboxymethyl-β-glucan for Reducing IL-5 in Children with Allergic Rhinitis (RENIM) | NCT03349619 | Phase 4 | Carboxymethyl-β-glucan | Two sprays (100 uL/spray) three times a day for 4 weeks | Allergic Rhinitis | |
| Effects of Orally Administered Beta-glucan on Leukocyte Function in Humans (BG) | NCT01727895 | n/a | Glucan, #300, Yeast | 500 mg/day | Immunologic Deficiency Syndromes | |
| Safety and Efficacy Study of Oral XIGO Tablets to Treat Common Cold | NCT01092039 | n/a | Unknown | Unknown | Common Cold | |
| Efficacy and Safety of Imuneks 10 mg Capsules in the Prophylaxis of Cold | NCT02807220 | Phase 4 | Micro ionized β-glucan -source unknown | 10 mg—2 capsules every morning | Cold Symptoms | |
| Alleviation of Cedar Pollen Induced Allergic Symptoms by Orally Taken Superfine β-1,3-glucan | NCT00276445 | Phase 4 | Fungal | Unknown | Allergic Conjunctivitis |
| Areas of Research | Study Title | NCT | Design | β-glucan Type | Dose | Population |
|---|---|---|---|---|---|---|
| Wound Healing | Efficacy and Safety Study of Soluble β-1,3/1,6-glucan in Thermal Burns | NCT00283426 | Phase 1 | SBG, Yeast | Unknown | Burns |
| A Randomized Comparison Study of Aquacel Ag and Glucan II as Donor Site Dressings | NCT00581217 | n/a | Glucan II, Oat | unknown | Burns | |
| Multi-Centre, Prospective, Randomized, Comparison of AWBAT™-D vs. Xeroform™ or Glucan II™ for Treatment of Donor Sites in Burn Surgery (AWBAT-D) | NCT00964470 | n/a | Glucan II, Oat | Unknown | Treatment of Donor Site Burns | |
| Efficacy of TR 987, β-1,3-1,6-D-glucan, in the Treatment of Chronic Venous Insufficiency Ulcers | NCT03154619 | Phase 2 | Glucoprime, Yeast | Gel application twice weekly | Venous Leg Ulcer | |
| Preadmission Skin Wipe Use for Surgical Site Infection Prophylaxis in Adult Orthopaedic Surgery Patients | NCT03401749 | Phase 4 | Unknown | Ingredient in wipes before surgery | Surgical Site Infection | |
| To Study the Effect of β-glucans on Wound Healing | NCT02078128 | n/a | Unknown | 30 mg/kg daily | Burns | |
| Beta-1,3/1,6-D-glucan Ganoderma Lucidum on Non-infectious and Idiopathic Uveitis | NCT04162314 | Phase 2, Phase 3 | Fungal | Capsule containing 180 mg/three times daily | Uveitis | |
| Soluble β-glucan (SBG) as Treatment for Diabetic Foot Ulcers | NCT00632008 | Phase 3 | SBG—Yeast | Topical Application twice a week | Chronic Diabetic Foot Ulcers | |
| Clinical Trial to Evaluate Papilocare® Gel Efficacy into Repairment of Cervical Lesions Caused by HPV (PAPILOCAN) | NCT04210336 | Phase 3 | Unknown | Topical application | HPV Infection, Lesion Cervix | |
| Treatment of Chronic Anal Fissure (TOCA) | NCT02158013 | n/a | Yeast | Gel application/ twice daily two weeks | Chronic anal fissure | |
| Irritation and Anal Bleeding in Patients Affected by Haemorrhoids. | NCT03569930 | Phase 4 | Unknown | Anal application Frequency, Unknown | Haemorrhoids | |
| Cognitive Performance | A Follow-up Trial of Proglucamune® in the Treatment of Protective Qi Insufficiency, a TCM Condition | NCT03782974 | n/a | Yeast | 2 tablets of proglucamune/day—200 mg of β-glucan for 8 weeks | Protective Qi Insufficiency (a Condition Term From TCM) |
| An Evaluation of Proglucamune in the Treatment of Protective Qi Insufficiency | NCT03829228 | n/a | Yeast, Fungal, Proglucamune tablet | 2–100 mg tablets per day | Protective Qi Insufficiency | |
| Investigation of How Morning Nutrition Influences Cognitive Performance | NCT03169283 | n/a | Oat (NC) | Cereal β-glucan in morning | Cognitive Performance | |
| Safety Studies | Dose Escalation Safety Study of MM-10-001 in Healthy Subjects | NCT00677027 | Phase 1 | Lentinan—Fungal | Unknown | Healthy |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murphy, E.J.; Rezoagli, E.; Major, I.; Rowan, N.J.; Laffey, J.G. β-Glucan Metabolic and Immunomodulatory Properties and Potential for Clinical Application. J. Fungi 2020, 6, 356. https://doi.org/10.3390/jof6040356
Murphy EJ, Rezoagli E, Major I, Rowan NJ, Laffey JG. β-Glucan Metabolic and Immunomodulatory Properties and Potential for Clinical Application. Journal of Fungi. 2020; 6(4):356. https://doi.org/10.3390/jof6040356
Chicago/Turabian StyleMurphy, Emma J., Emanuele Rezoagli, Ian Major, Neil J. Rowan, and John G. Laffey. 2020. "β-Glucan Metabolic and Immunomodulatory Properties and Potential for Clinical Application" Journal of Fungi 6, no. 4: 356. https://doi.org/10.3390/jof6040356
APA StyleMurphy, E. J., Rezoagli, E., Major, I., Rowan, N. J., & Laffey, J. G. (2020). β-Glucan Metabolic and Immunomodulatory Properties and Potential for Clinical Application. Journal of Fungi, 6(4), 356. https://doi.org/10.3390/jof6040356








