Sphinganine-Analog Mycotoxins (SAMs): Chemical Structures, Bioactivities, and Genetic Controls
Abstract
1. Introduction
2. Chemical and Structural Properties
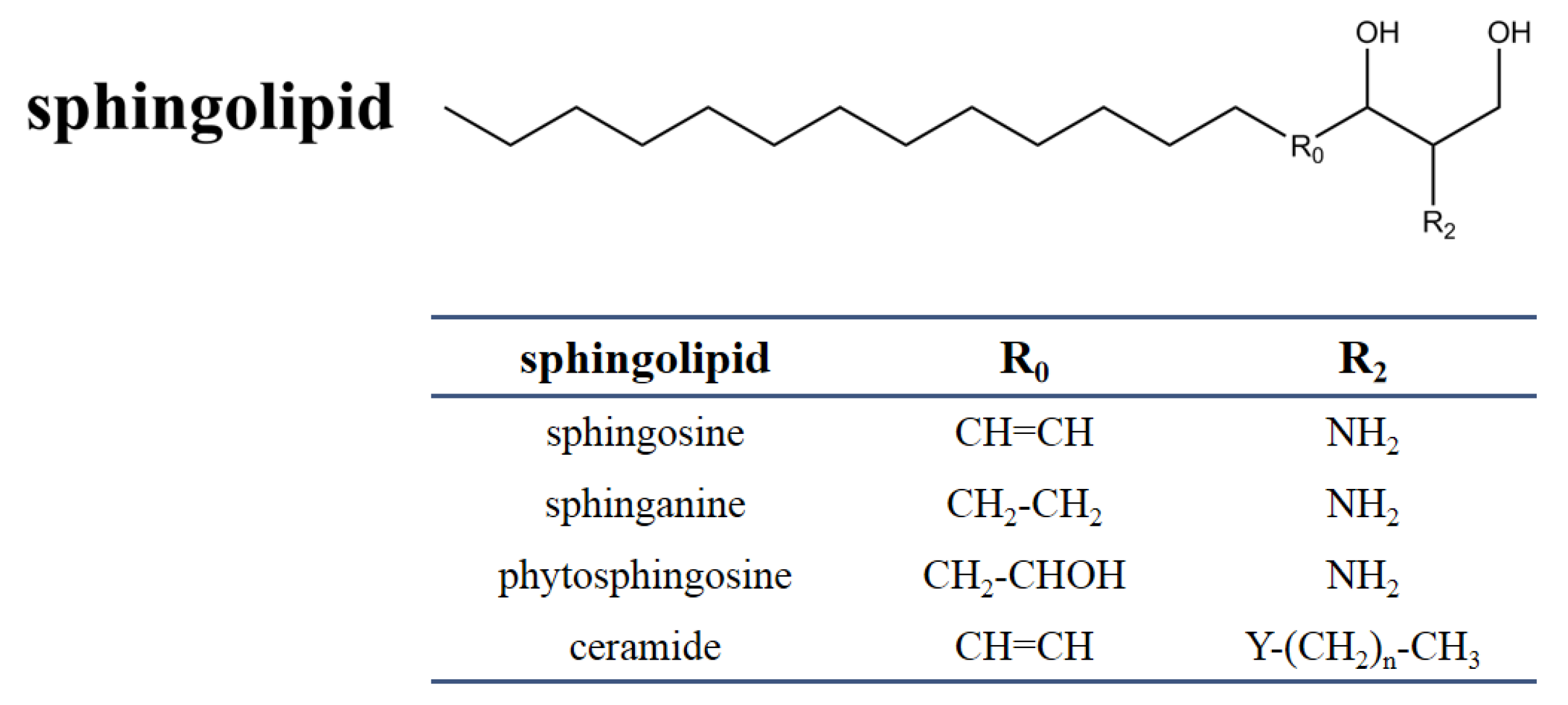
2.1. Chemical and Structural Properties of Sphingolipids
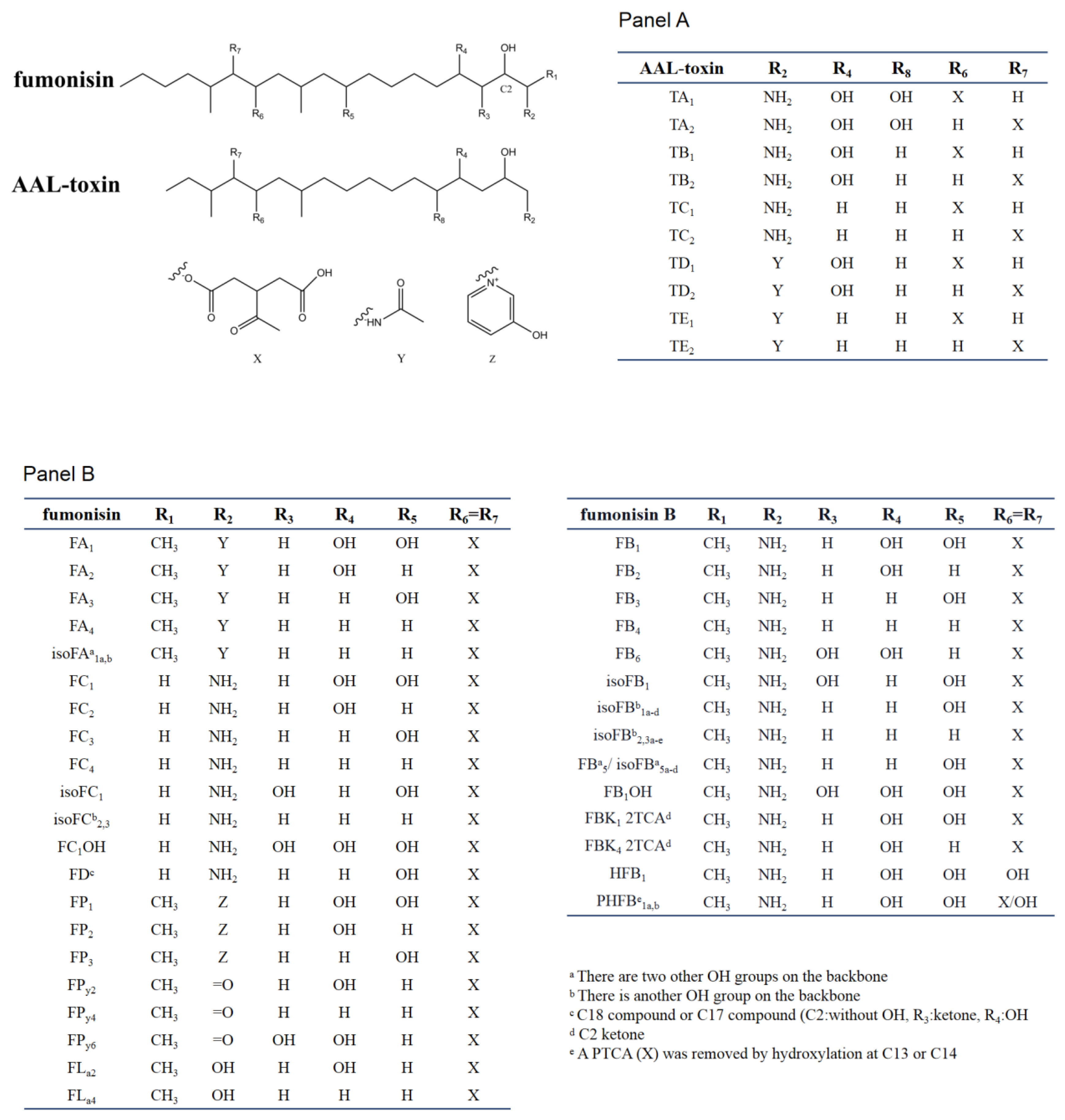
2.2. Chemical and Structural Properties of Fumonisins
2.3. Chemical and Structural Properties of AAL-Toxin
2.4. Chemical and Structural Properties of Analogs of SAMs
3. Relationships between SAMs’ Structure and Biological Activities
4. Detoxification of SAMs
5. Programmed Cell Death and Sphingolipids
6. SAMs Trigger PCD through Ceramide-Based Signaling Pathways
7. Plant Resistance to SAMs
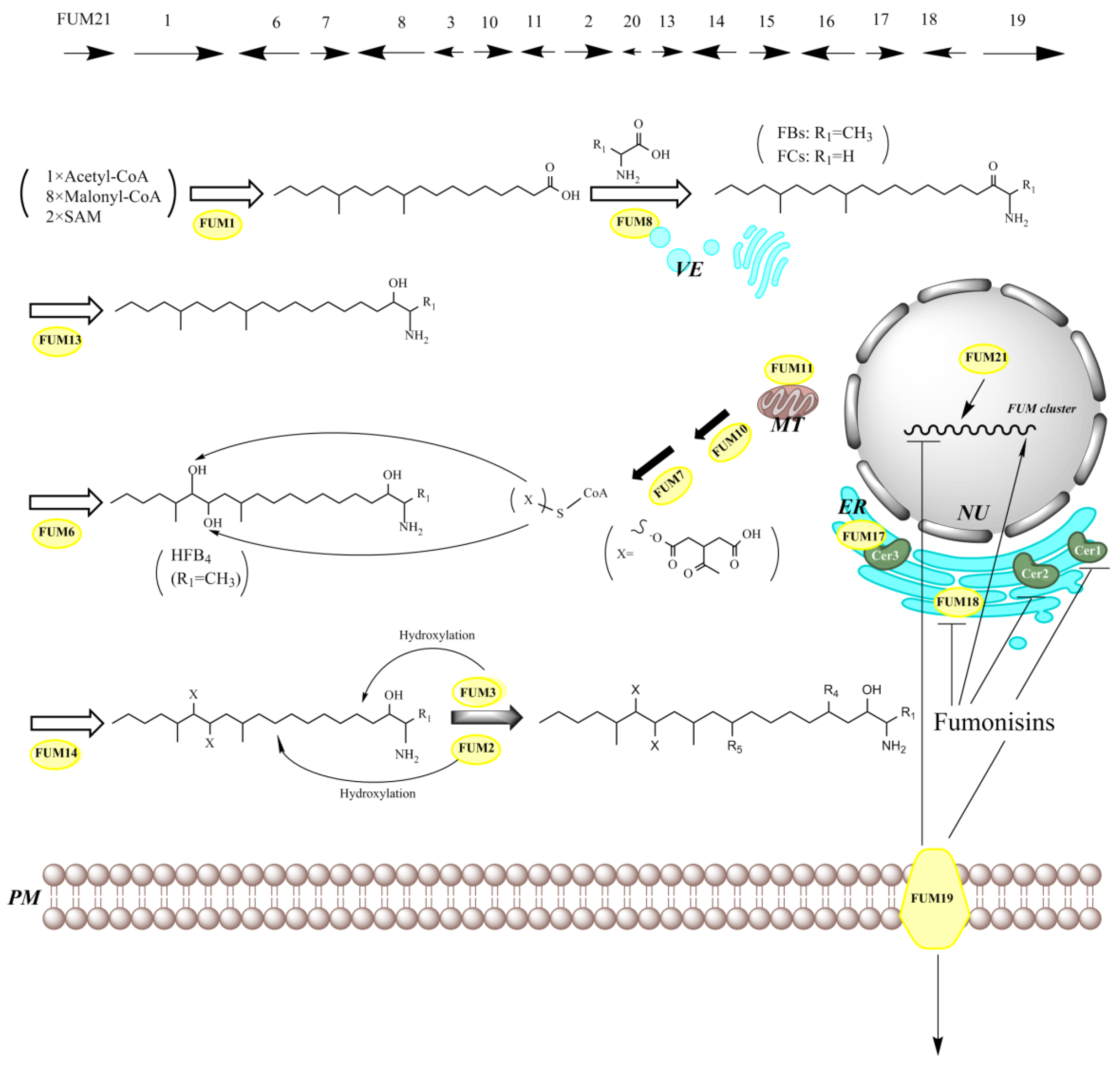
8. Genes Responsible for SAMs Production
| Homologue of Cluster Genes | Predict Gene Product and Function | Reference | ||
|---|---|---|---|---|
| fumonisin | AAL-toxin | |||
| Fusarium spp. | Aspergillus spp. | A. alternata | [169,224] | |
| FUM1 (fumonisin biosynthetic gene 1, previously designated as FUM5)) | fum1 | ALT1 (AAL-toxin biosynthetic gene 1) | polyketide synthase | [12,199,200] |
| FUM2 | absent | Dioxygenase for hydroxylation of C10 | [228] | |
| FUM3 | fum3 | Dioxygenase for hydroxylation of C5 (the same gene as FUM9) | [205,228,229] | |
| FUM4 | Not clear | [230] | ||
| FUM6 | fum6 | ALT2 | Cytochrome P450 monooxygenase–reductase fusion proteins for hydroxylation of C14/C15 | [192,231] |
| FUM7 | fum7 | ALT3 | Type III alcohol dehydrogenases for PTCA (propane-1,2,3-tricarboxylic acid) side chain formation | [192,194,232] |
| FUM8 | fum8 | α-oxoamine synthase and homologous for amino transfer and FBs/FCs production | [192,195] | |
| FUM10 | fum10 | Fatty acyl-CoA synthase for PTCA esterification | [193,194] | |
| FUM11 | mitochondrial transport protein for PTCA transport | [193,194] | ||
| FUM12 | cytochrome P450 monooxygenases | [193] | ||
| FUM13 | fum13 | ALT6 | Short-chain dehydrogenase/ketoreductase of C3 | [193,204,206] |
| FUM14 | fum14 | Non-ribosomal peptide synthetase for PTCA esterification | [193,194,233] | |
| FUM15 | fum15 | Cytochrome P450 monooxygenases | [193] | |
| FUM16 | fum16 | Fatty acyl-CoA synthetase | [193,194,224] | |
| FUM17 | CerS for self-protection against fumonisins | [193,197] | ||
| FUM18 | CerS for self-protection against fumonisins | [193,197] | ||
| FUM19 | fum19 | ABC transport protein as a repressor of FUM gene cluster | [193,197] | |
| FUM20 | Not clear | [234] | ||
| FUM21 | fum21 | ALT13 | Zn(II)2Cys6 transcription factor | [198] |
| absent | SDR1 | Short-chain dehydrogenase/reductase (SDR) | [7] | |
9. Evolution of SAMs Production
10. Detection Method of SAMs
11. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Gilchrist, D.G. Programmed cell death in plant disease: The purpose and promise of cellular suicide. Annu. Rev. Phytopathol. 1998, 36, 393–414. [Google Scholar] [CrossRef]
- Riley, R.T.; Wang, E.; Schroeder, J.J.; Smith, E.R.; Plattner, R.D.; Abbas, N.; Yoo, H.S.; Merrill, A.H., Jr. Evidence for disruption of sphingolipid metabolism as a contributing factor in the toxicity and carcinogenicity of fumonisins. Nat. Toxins 1996, 4, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Merrill, A.H., Jr.; Sullards, M.C.; Wang, E.; Voss, K.A.; Riley, R.T. Sphingolipid metabolism: Roles in signal transduction and disruption by fumonisins. Environ. Health Persp. 2001, 109, 283–289. [Google Scholar] [CrossRef]
- Gelderblom, W.C.A.; Jaskiewicz, K.; Marasas, W.F.O.; Thiel, P.G.; Horak, R.M.; Vleggaar, R.; Kriek, N.P.J. Fumonisins-novel mycotoxins with cancer-promoting activity produced by Fusarium moniliforme. Appl. Environ. Microb. 1988, 54, 1806–1811. [Google Scholar] [CrossRef] [PubMed]
- Rheeder, J.P.; Marasas, W.F.O.; Vismer, H.F. Production of fumonisin analogs by Fusarium species. Appl. Environ. Microb. 2002, 68, 2101–2105. [Google Scholar] [CrossRef]
- Frisvad, J.C.; Smedsgaard, J.; Samson, R.A.; Larsen, T.O.; Thrane, U. Fumonisin B2 production by Aspergillus niger. J. Agric. Food Chem. 2007, 55, 9727–9732. [Google Scholar] [CrossRef]
- Susca, A.; Proctor, R.H.; Butchko, R.A.E.; Haidukowski, M.; Stea, G.; Logrieco, A.; Moretti, A. Variation in the fumonisin biosynthetic gene cluster in fumonisin-producing and nonproducing black aspergilli. Fungal Genet. Biol. 2014, 73, 39–52. [Google Scholar] [CrossRef]
- Chen, J.; Mirocha, C.J.; Xie, W.; Hogge, L.; Olson, D. Production of the mycotoxin fumonisin B1 by Alternaria alternata f. sp. lycopersici. Appl. Environ. Microb. 1992, 58, 3928–3931. [Google Scholar] [CrossRef]
- Proctor, R.H.; Van Hove, F.; Susca, A.; Stea, G.; Busman, M.; van der Lee, T.; Waalwijk, C.; Moretti, A.; Ward, T.J. Birth, death and horizontal transfer of the fumonisin biosynthetic gene cluster during the evolutionary diversification of Fusarium. Mol. Microbiol. 2013, 90, 290–306. [Google Scholar] [CrossRef]
- Mogensen, J.M.; Møller, K.A.; Von Freiesleben, P.; Labuda, R.; Varga, E.; Sulyok, M.; Kubátová, A.; Thrane, U.; Andersen, B.; Nielsen, K.F. Production of fumonisins B2 and B4 in Tolypocladium species. J. Ind. Microbiol. Biot. 2011, 38, 1329–1335. [Google Scholar] [CrossRef]
- Alabouvette, C.; Lemanceau, P.; Steinberg, C. Recent advances in the biological control of fusarium wilts. Pestic. Sci. 1993, 37, 365–373. [Google Scholar] [CrossRef]
- Desjardins, A.E.; Plattner, R.D.; Nelsen, T.C.; Leslie, J.F. Genetic analysis of fumonisin production and virulence of Gibberella fujikuroi mating population A (Fusarium moniliforme) on maize (Zea mays) seedlings. Appl. Environ. Microb. 1995, 61, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Chulze, S.N. Strategies to reduce mycotoxin levels in maize during storage: A review. Food Addit. Contam. A 2010, 27, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Munkvold, G.P.; McGee, D.C.; Carlton, W.M. Importance of different pathways for maize kernel infection by Fusarium moniliforme. Phytopathology 1997, 87, 209–217. [Google Scholar] [CrossRef]
- Williams, L.D.; Glenn, A.E.; Bacon, C.W.; Smith, M.A.; Riley, R.T. Fumonisin production and bioavailability to maize seedlings grown from seeds inoculated with Fusarium verticillioides and grown in natural soils. J. Agric. Food Chem. 2006, 54, 5694–5700. [Google Scholar] [CrossRef]
- Kamle, M.; Mahato, D.K.; Devi, S.; Lee, K.E.; Kang, S.G.; Kumar, P. Fumonisins: Impact on agriculture, food, and human health and their management strategies. Toxins 2019, 11, 328. [Google Scholar] [CrossRef]
- Chu, F.S.; Li, G.Y. Simultaneous occurrence of fumonisin B1 and other mycotoxins in moldy corn collected from the People’s Republic of China in regions with high incidences of esophageal cancer. Appl. Environ. Microb. 1994, 60, 847–852. [Google Scholar] [CrossRef]
- Marasas, W.F.O.; Riley, R.T.; Hendricks, K.A.; Stevens, V.L.; Sadler, T.W.; Gelineau-Van Waes, J.; Missmer, S.A.; Cabrera, J.; Torres, O.; Gelderblom, W.C.A.; et al. Fumonisins Disrupt Sphingolipid Metabolism, Folate Transport, and Neural Tube Development in Embryo Culture and In Vivo: A Potential Risk Factor for Human Neural Tube Defects among Populations Consuming Fumonisin-Contaminated Maize. J. Nutr. 2004, 134, 711–716. [Google Scholar] [CrossRef]
- Gallo, A.; Giuberti, G.; Frisvad, J.C.; Bertuzzi, T.; Nielsen, K.F. Review on mycotoxin issues in ruminants: Occurrence in forages, effects of mycotoxin ingestion on health status and animal performance and practical strategies to counteract their negative effects. Toxins 2015, 7, 3057–3111. [Google Scholar] [CrossRef]
- Ross, P.F.; Rice, L.G.; Osweiler, G.D.; Nelson, P.E.; Richard, J.L.; Wilson, T.M. A review and update of animal toxicoses associated with fumonisin-contaminated feeds and production of fumonisins by Fusarium isolates. Mycopathologia 1992, 117, 109–114. [Google Scholar] [CrossRef]
- Harrison, L.R.; Colvin, B.M.; Greene, J.T.; Newman, L.E.; Cole, J.R., Jr. Pulmonary Edema and Hydrothorax in Swine Produced by Fumonisin B1, a Toxic Metabolite of Fusarium Moniliforme. J. Vet. Diagn. Investig. 1990, 2, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, D.G.; Grogan, R.G. Production and nature of a host-specific toxin from Alternaria alternata f. sp. lycopersici. Phytopathology 1976, 66, 165–171. [Google Scholar] [CrossRef]
- Meena, M.; Samal, S. Alternaria host-specific (HSTs) toxins: An overview of chemical characterization, target sites, regulation and their toxic effects. Toxicol. Rep. 2019, 6, 745–758. [Google Scholar] [CrossRef] [PubMed]
- Abbas, H.K.; Duke, S.O.; Paul, R.N.; Riley, R.T.; Tanaka, T. AAL-toxin, a potent natural herbicide which disrupts sphingolipid metabolism of plants. Pestic. Sci. 1995, 43, 181–187. [Google Scholar] [CrossRef]
- Mesbah, L.A.; Van Der Weerden, G.M.; Nijkamp, H.J.J.; Hille, J. Sensitivity among species of Solanaceae to AAL toxins produced by Alternaria alternata f.sp. lycopersici. Plant Pathol. 2000, 49, 734–741. [Google Scholar] [CrossRef]
- Renaud, J.B.; Kelman, M.J.; Qi, T.F.; Seifert, K.A.; Sumarah, M.W. Product ion filtering with rapid polarity switching for the detection of all fumonisins and AAL-toxins. Rapid Commun. Mass Sp. 2015, 29, 2131–2139. [Google Scholar] [CrossRef]
- Abbas, H.K.; Riley, R.T. The presence and phytotoxicity of fumonisins and AAL-toxin in Alternaria alternata. Toxicon 1996, 34, 133–136. [Google Scholar] [CrossRef]
- Yamagishi, D.; Akamatsu, H.; Otani, H.; Kodama, M. Pathological evaluation of host-specific AAL-toxins and fumonisin mycotoxins produced by Alternaria and Fusarium species. J. Gen. Plant Pathol. 2006, 72, 323–327. [Google Scholar] [CrossRef]
- Merrill, A.H. Sphingolipid Biosynthesis. In Encyclopedia of Biological Chemistry, 2nd ed.; Lennarz, W.J., Lane, M.D., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2013; pp. 281–286. [Google Scholar] [CrossRef]
- Hannun, Y.A.; Obeid, L.M. Sphingolipids and their metabolism in physiology and disease. Nat. Rev. Mol. Cell Biol. 2018, 19, 175–191. [Google Scholar] [CrossRef]
- Huby, E.; Napier, J.A.; Baillieul, F.; Michaelson, L.V.; Dhondt-Cordelier, S. Sphingolipids: Towards an integrated view of metabolism during the plant stress response. New Phytol. 2020, 225, 659–670. [Google Scholar] [CrossRef]
- Li, Q.; Fang, H.; Dang, E.; Wang, G. The role of ceramides in skin homeostasis and inflammatory skin diseases. J. Dermatol. Sci. 2020, 97, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Bartók, T.; Szécsi, Á.; Szekeres, A.; Mesterházy, Á.; Bartók, M. Detection of new fumonisin mycotoxins and fumonisin-like compounds by reversed-phase high-performance liquid chromatography/electrospray ionization ion trap mass spectrometry. Rapid Commun. Mass Sp. 2006, 20, 2447–2462. [Google Scholar] [CrossRef] [PubMed]
- Abbas, H.K.; Duke, S.O.; Tanaka, T. Phytotoxicity of fumonisins and related compounds. Toxin Rev. 1993, 12, 225–251. [Google Scholar] [CrossRef]
- Branham, B.E.; Plattner, R.D. Isolation and characterization of a new fumonisin from liquid cultures of Fusarium moniliforme. J. Nat. Prod. 1993, 56, 1630–1633. [Google Scholar] [CrossRef]
- Seo, J.A.; Kim, J.C.; Lee, Y.W. N-acetyl derivatives of type C fumonisins produced by Fusarium oxysporum. J. Nat. Prod. 1999, 62, 355–357. [Google Scholar] [CrossRef]
- Musser, S.M.; Gay, M.L.; Mazzola, E.P.; Plattner, R.D. Identification of a new series of fumonisins containing 3-hydroxypyridine. J. Nat. Prod. 1996, 59, 970–972. [Google Scholar] [CrossRef]
- Abbas, H.K.; Shier, W.T.; Seo, J.A.; Lee, Y.W.; Musser, S.M. Phytotoxicity and cytotoxicity of the fumonisin C and P series of mycotoxins from Fusarium spp. fungi. Toxicon 1998, 36, 2033–2037. [Google Scholar] [CrossRef]
- Bartók, T.; Tölgyesi, L.; Szekeres, A.; Varga, M.; Bartha, R.; Szécsi, A.; Bartók, M.; Mesterházy, A. Detection and characterization of twenty-eight isomers of fumonisin B1 (FB1) mycotoxin in a solid rice culture infected with Fusarium verticillioides by reversed-phase high-performance liquid chromatography/electrospray ionization time-of-flight and ion trap mass spectrometry. Rapid Commun. Mass Sp. 2010, 24, 35–42. [Google Scholar] [CrossRef]
- Bottini, A.T.; Bowen, J.R.; Gilchrist, D.G. Phytotoxins. II. Characterization of a phytotoxic fraction from Alternaria alternata f. sp. lycopersici. Tetrahedron Lett. 1981, 22, 2723–2726. [Google Scholar] [CrossRef]
- Bottini, A.T.; Gilchrist, D.G. Phytotoxins. I. A 1-amino dimethyl heptadecapentol from alternaria alternata f. sp. lycopersici. Tetrahedron Lett. 1981, 22, 2719–2722. [Google Scholar] [CrossRef]
- Caldas, E.D.; Jones, A.D.; Ward, B.; Winter, C.K.; Gilchrist, D.G. Structural Characterization of Three New AAL Toxins Produced by Alternaria alternata f. sp. lycopersici. J. Agric. Food Chem. 1994, 42, 327–333. [Google Scholar] [CrossRef]
- Saucedo-García, M.; Guevara-García, A.; González-Solís, A.; Cruz-García, F.; Vázquez-Santana, S.; Markham, J.E.; Lozano-Rosas, M.G.; Dietrich, C.R.; Ramos-Vega, M.; Cahoon, E.B.; et al. MPK6, sphinganine and the LCB2a gene from serine palmitoyltransferase are required in the signaling pathway that mediates cell death induced by long chain bases in Arabidopsis. New Phytol. 2011, 191, 943–957. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Bielawski, J.; Mu, J.; Dong, H.; Teng, C.; Zhang, J.; Yang, X.; Tomishige, N.; Hanada, K.; Hannun, Y.A.; et al. Involvement of sphingoid bases in mediating reactive oxygen intermediate production and programmed cell death in Arabidopsis. Cell Res. 2007, 17, 1030–1040. [Google Scholar] [CrossRef] [PubMed]
- Spassieva, S.D.; Markham, J.E.; Hille, J. The plant disease resistance gene Asc-1 prevents disruption of sphingolipid metabolism during AAL-toxin-induced programmed cell death. Plant J. 2002, 32, 561–572. [Google Scholar] [CrossRef]
- Kluepfel, D.; Bagli, J.; Baker, H.; Charest, M.P.; Kudelski, A.; Sehgal, S.N.; Vézina, C. Myriocin, a new antifungal antibiotic from Myriococcum albomyces. J. Antibiot. 1972, 25, 109–115. [Google Scholar] [CrossRef]
- Šašek, V.; Sailer, M.; Vokoun, J.; Musílek, V. Production of thermozymocidin (myriocin) by the pyrenomycete Melanconis flavovirens. J. Basic Microb. 1989, 29, 383–390. [Google Scholar] [CrossRef]
- Fujita, T.; Inoue, K.; Yamamoto, S.; Ikumoto, T.; Sasaki, S.; Toyama, R.; Chiba, K.; Hoshino, Y.; Okumoto, T. Fungal metabolites. Part 11. A potent immunosuppressive activity found in Isaria sinclairii metabolite. J. Antibiot. 1994, 47, 208–215. [Google Scholar] [CrossRef]
- Miyake, Y.; Kozutsumi, Y.; Nakamura, S.; Fujita, T.; Kawasaki, T. Serine palmitoyltransferase is the primary target of a sphingosine-like immunosuppressant, ISP-1/myriocin. Biochem. Bioph. Res. Co. 1995, 211, 396–403. [Google Scholar] [CrossRef]
- Tatematsu, K.; Tanaka, Y.; Sugiyama, M.; Sudoh, M.; Mizokami, M. Host sphingolipid biosynthesis is a promising therapeutic target for the inhibition of hepatitis B virus replication. J. Med. Virol. 2011, 83, 587–593. [Google Scholar] [CrossRef]
- Yu, S.; Jia, B.; Yang, Y.; Liu, N.; Wu, A. Involvement of PERK-CHOP pathway in fumonisin B1- induced cytotoxicity in human gastric epithelial cells. Food Chem. Toxicol. 2020, 136. [Google Scholar] [CrossRef]
- Liu, J.; Huang, X.; Withers, B.R.; Blalock, E.; Liu, K.; Dickson, R.C. Reducing sphingolipid synthesis orchestrates global changes to extend yeast lifespan. Aging Cell 2013, 12, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Reforgiato, M.R.; Milano, G.; Fabriàs, G.; Casas, J.; Gasco, P.; Paroni, R.; Samaja, M.; Ghidoni, R.; Caretti, A.; Signorelli, P. Inhibition of ceramide de novo synthesis as a postischemic strategy to reduce myocardial reperfusion injury. Basic Res. Cardiol. 2016, 111. [Google Scholar] [CrossRef] [PubMed]
- Horn, W.S.; Smith, J.L.; Bills, G.F.; Raghoobar, S.L.; Helms, G.L.; Kurtz, M.B.; Marrinan, J.A.; Frommer, B.R.; Thornton, R.A.; Mandala, S.M. Sphingofungins E and F: Novel serinepalmitoyl trans-ferase inhibitors from Paecilomyces variotii. J. Antibiot. 1992, 45, 1692–1696. [Google Scholar] [CrossRef] [PubMed]
- VanMiddlesworth, F.; Giacobbe, R.A.; Lopez, M.; Garrity, G.; Bland, J.A.; Bartizal, K.; Fromtling, R.A.; Polishook, J.; Zweerink, M.; Edison, A.M.; et al. Sphingofungins a, b, c, and d; a new family of antifungal agents: I. Fermentation, isolation, and biological activity. J. Antibiot. 1992, 45, 861–867. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Liu, Y.; Gao, J.; Hu, J.; He, H.; Dai, S.; Wang, L.; Dai, H.; Zhang, L.; Song, F. Antitubercular metabolites from the marine-derived fungus strain Aspergillus fumigatus MF029. Nat. Prod. Res. 2019. [Google Scholar] [CrossRef]
- Zhang, H.; Zhu, H.T.; Wang, D.; Yang, C.R.; Zhang, Y.J. Sphingofungins G and H: New five-membered lactones from Aspergillus penicilliodes Speg. Nat. Prod. Res. 2019, 33, 1284–1291. [Google Scholar] [CrossRef] [PubMed]
- Hanada, K.; Nishijima, M.; Fujita, T.; Kobayashi, S. Specificity of inhibitors of serine palmitoyltransferase (SPT), a key enzyme in sphingolipid biosynthesis, in intact cells. A novel evaluation system using an SPT-defective mammalian cell mutant. Biochem. Pharmacol. 2000, 59, 1211–1216. [Google Scholar] [CrossRef]
- Harris, G.H.; Turner Jones, E.T.; Meinz, M.S.; Nallin-Omstead, M.; Helms, G.L.; Bills, G.F.; Zink, D.; Wilson, K.E. Isolation and structure elucidation of viridiofungins A, B and C. Tetrahedron Lett. 1993, 34, 5235–5238. [Google Scholar] [CrossRef]
- Mandala, S.M.; Thornton, R.A.; Frommer, B.R.; Dreikorn, S.; Kurtz, M.B. Viridiofungins, novel inhibitors of sphingolipid synthesis. J. Antibiot 1997, 50, 339–343. [Google Scholar] [CrossRef]
- El-Hasan, A.; Walker, F.; Schöne, J.; Buchenauer, H. Detection of viridiofungin A and other antifungal metabolites excreted by Trichoderma harzianum active against different plant pathogens. Eur. J. Plant Pathol. 2009, 124, 457–470. [Google Scholar] [CrossRef]
- Abbas, H.K.; Duke, S.O.; Merrill, A.H., Jr.; Wang, E.; Shier, W.T. Phytotoxicity of Australifungin, AAL-toxins and fumonisin B1 to Lemna pausicostata. Phytochemistry 1998, 47, 1509–1514. [Google Scholar] [CrossRef]
- Mandala, S.M.; Thornton, R.A.; Frommer, B.R.; Curotto, J.E.; Rozdilsky, W.; Kurtz, M.B.; Giacobbe, R.A.; Bills, G.F.; Cabello, M.A. The Discovery of Australifungin, a Novel Inhibitor of Sphinganine N-Acyltransferase from Sporormiella australis Producing Organism, Fermentation, Isolation, and Biological Activity. J. Antibiot. 1995, 48, 349–356. [Google Scholar] [CrossRef]
- Uhlig, S.; Petersen, D.; Flåøyen, A.; Wilkins, A. 2-Amino-14,16-dimethyloctadecan-3-ol, a new sphingosine analogue toxin in the fungal genus Fusarium. Toxicon 2005, 46, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Marrouchi, R.; Benoit, E.; Le Caer, J.P.; Belayouni, N.; Belghith, H.; Molgó, J.; Kharrat, R. Toxic C17-sphinganine analogue mycotoxin, contaminating Tunisian mussels, causes flaccid paralysis in rodents. Mar. Drugs 2013, 11, 4724–4740. [Google Scholar] [CrossRef] [PubMed]
- Mirocha, C.J.; Gilchrist, D.G.; Shier, W.T.; Abbas, H.K.; Wen, Y.; Vesonder, R.F. AAL Toxins, funionisms (biology and chemistry) and host-specificity concepts. Mycopathologia 1992, 117, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Shier, W.T.; Abbas, H.K.; Mirocha, C.J. Toxicity of the mycotoxins fumonisins B1 and B2 and Alternaria alternata f. sp. lycopersici toxin (AAL) in cultured mammalian cells. Mycopathologia 1991, 116, 97–104. [Google Scholar] [CrossRef]
- Van Der Westhuizen, L.; Shephard, G.S.; Snyman, S.D.; Abel, S.; Swanevelder, S.; Gelderblom, W.C.A. Inhibition of sphingolipid biosynthesis in rat primary hepatocyte cultures by fumonisin B1 and other structurally related compounds. Food Chem. Toxicol. 1998, 36, 497–503. [Google Scholar] [CrossRef]
- Abbas, H.K.; Mulrooney, J.E. Effect of Some Phytopathogenic Fungi and Their Metabolites on Growth of Heliothis virescens (F.) and Its Host Plants. Biocontrol Sci. Techn. 1994, 4, 77–87. [Google Scholar] [CrossRef]
- Gelderblom, W.C.A.; Cawood, M.E.; Snyman, S.D.; Vleggaar, R.; Marasas, W.F.O. Structure-activity relationships of fumonisins in short-term carcinogenesis and cytotoxicity assays. Food Chem. Toxicol. 1993, 31, 407–414. [Google Scholar] [CrossRef]
- Tanaka, T.; Abbas, H.K.; Duke, S.O. Structure-dependent phytotoxicity of fumonisins and related compounds in a duckweed bioassay. Phytochemistry 1993, 33, 779–785. [Google Scholar] [CrossRef]
- Siler, D.J.; Gilchrist, D.G. Properties of host specific toxins produced by Alternaria alternata f. sp. lycopersici in culture and in tomato plants. Physiol. Plant Pathol. 1983, 23, 265–274. [Google Scholar] [CrossRef]
- Norred, W.P.; Riley, R.T.; Meredith, F.I.; Poling, S.M.; Plattner, R.D. Instability of N-acetylated fumonisin B1 (FA1) and the impact on inhibition of ceramide synthase in rat liver slices. Food Chem. Toxicol. 2001, 39, 1071–1078. [Google Scholar] [CrossRef]
- Shier, W.T.; Abbas, H.K. Current issues in research on fumonisins, mycotoxins which may cause nephropathy. J. Toxicol. Toxin Rev. 1999, 18, 323–335. [Google Scholar] [CrossRef]
- Shier, W.T.; Abbas, H.K.; Abou-Karam, M.; Badria, F.A.; Resch, P.A. Fumonisins: Abiogenic Conversions of an Environmental Tumor Promoter and Common Food Contaminant. J. Toxicol. Toxin Rev. 2003, 22, 591–616. [Google Scholar] [CrossRef]
- Savard, M.E.; Sinha, R.C.; Lau, R.; Séguin, C.; Buffam, S. Monoclonal antibodies for fumonisins B1, B2 and B3. Food Agric. Immunol. 2003, 15, 127–134. [Google Scholar] [CrossRef]
- Azaiez, I.; Meca, G.; Manyes, L.; Luciano, F.B.; Fernández-Franzón, M. Study of the chemical reduction of the fumonisins toxicity using allyl, benzyl and phenyl isothiocyanate in model solution and in food products. Toxicon 2013, 63, 137–146. [Google Scholar] [CrossRef]
- Lamprecht, S.C.; Marasas, W.F.O.; Alberts, J.F.; Cawood, M.E.; Gelderblom, W.C.A.; Shephard, G.S.; Thiel, P.G.; Calitz, F.J. Phytotoxicity of fumonisins and TA-toxin to corn and tomato. Phytopathology 1994, 84, 383–391. [Google Scholar] [CrossRef]
- Vesonder, R.F.; Peterson, R.E.; Labeda, D.; Abbas, H.K. Comparative phytotoxicity of the fumonisins, AAL-toxin and yeast sphingolipids in Lemna minor L. (duckweed). Arch. Environ. Con. Tox. 1992, 23, 464–467. [Google Scholar] [CrossRef]
- Voss, K.A.; Riley, R.T.; Snook, M.E.; Gelineau-van Waes, J. Reproductive and sphingolipid metabolic effects of fumonisin B1 and its alkaline hydrolysis product in LM/Bc mice: Hydrolyzed fumonisin B1 did not cause neural tube defects. Toxicol. Sci. 2009, 112, 459–467. [Google Scholar] [CrossRef]
- Grenier, B.; Bracarense, A.P.F.L.; Schwartz, H.E.; Trumel, C.; Cossalter, A.M.; Schatzmayr, G.; Kolf-Clauw, M.; Moll, W.D.; Oswald, I.P. The low intestinal and hepatic toxicity of hydrolyzed fumonisin B1 correlates with its inability to alter the metabolism of sphingolipids. Biochem. Pharmacol. 2012, 83, 1465–1473. [Google Scholar] [CrossRef]
- Gu, M.J.; Han, S.E.; Hwang, K.; Mayer, E.; Reisinger, N.; Schatzmayr, D.; Park, B.C.; Han, S.H.; Yun, C.H. Hydrolyzed fumonisin B1 induces less inflammatory responses than fumonisin B1 in the co-culture model of porcine intestinal epithelial and immune cells. Toxicol. Lett. 2019, 305, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Seiferlein, M.; Humpf, H.U.; Voss, K.A.; Sullards, M.C.; Allegood, J.C.; Wang, E.; Merrill, A.H., Jr. Hydrolyzed fumonisins HFB1 and HFB2 are acylated in vitro and in vivo by ceramide synthase to form cytotoxic N-acyl-metabolites. Mol. Nutr. Food Res. 2007, 51, 1120–1130. [Google Scholar] [CrossRef]
- Seefelder, W.; Knecht, A.; Humpf, H.U. Bound fumonisin B1: Analysis of fumonisin-B1 glyco and amino acid conjugates by liquid chromatography-electrospray ionization-tandem mass spectrometry. J. Agric. Food Chem. 2003, 51, 5567–5573. [Google Scholar] [CrossRef] [PubMed]
- Abbas, H.K.; Tanaka, T.; Duke, S.O.; Porter, J.K.; Wray, E.M.; Hodges, L.; Sessions, A.E.; Wang, E.; Merrill Jnr, A.H.; Riley, R.T. Fumonisin- and AAL-toxin-induced disruption of sphingolipid metabolism with accumulation of free sphingoid bases. Plant Physiol. 1994, 106, 1085–1093. [Google Scholar] [CrossRef] [PubMed]
- Henriques, J.; Lima, M.; Rosa, S.; Dias, A.S.; Dias, L.S. Allelopathic plants. XVIII. Solanum nigrum L. Allelopathy J. 2006, 17, 1–15. [Google Scholar]
- Duke, S.O.; Dayan, F.E. Modes of action of microbially-produced phytotoxins. Toxins 2011, 3, 1038–1064. [Google Scholar] [CrossRef]
- Abbas, H.K.; Tanaka, T.; Shier, W.T. Biological activities of synthetic analogues of Alternaria alternata toxin (AAL-toxin) and fumonisin in plant and mammalian cell cultures. Phytochemistry 1995, 40, 1681–1689. [Google Scholar] [CrossRef]
- Duke, S.O.; Dayan, F.E. Clues to new herbicide mechanisms of action from natural sources. ACS Symposium Series 2013, 1141, 203–215. [Google Scholar] [CrossRef]
- Luo, Y.; Liu, X.; Li, J. Updating techniques on controlling mycotoxins—A review. Food Control 2018, 89, 123–132. [Google Scholar] [CrossRef]
- McCormick, S.P. Microbial Detoxification of Mycotoxins. J. Chem. Ecol. 2013, 39, 907–918. [Google Scholar] [CrossRef]
- Agriopoulou, S.; Stamatelopoulou, E.; Varzakas, T. Advances in occurrence, importance, and mycotoxin control strategies: Prevention and detoxification in foods. Foods 2020, 9, 137. [Google Scholar] [CrossRef] [PubMed]
- Norred, W.P.; Voss, K.A.; Bacon, C.W.; Riley, R.T. Effectiveness of ammonia treatment in detoxification of fumonisin-contaminated corn. Food Chem. Toxicol. 1991, 29, 815–819. [Google Scholar] [CrossRef]
- Chourasia, H.K. Efficacy of ammonia in detoxification of fumonisin contaminated corn. Indian J. Exp. Biol. 2001, 39, 493–495. [Google Scholar]
- De Girolamo, A.; Lattanzio, V.M.T.; Schena, R.; Visconti, A.; Pascale, M. Effect of alkaline cooking of maize on the content of fumonisins B1 and B2 and their hydrolysed forms. Food Chem. 2016, 192, 1083–1089. [Google Scholar] [CrossRef] [PubMed]
- Chang, X.; Wang, J.; Sun, C.; Liu, H.; Wu, S.; Sun, J.; Wu, Z. Research on degradation of chlorine dioxide in primary mycotoxins. J. Chin. Cereals Oils Assoc. 2016, 31, 113–118. [Google Scholar]
- Voss, K.; Ryu, D.; Jackson, L.; Riley, R.; Gelineau-Van Waes, J. Reduction of Fumonisin Toxicity by Extrusion and Nixtamalization (Alkaline Cooking). J. Agric. Food Chem. 2017, 65, 7088–7096. [Google Scholar] [CrossRef] [PubMed]
- ten Bosch, L.; Pfohl, K.; Avramidis, G.; Wieneke, S.; Viöl, W.; Karlovsky, P. Plasma-based degradation of mycotoxins produced by Fusarium, Aspergillus and Alternaria species. Toxins 2017, 9, 97. [Google Scholar] [CrossRef] [PubMed]
- Wielogorska, E.; Ahmed, Y.; Meneely, J.; Graham, W.G.; Elliott, C.T.; Gilmore, B.F. A holistic study to understand the detoxification of mycotoxins in maize and impact on its molecular integrity using cold atmospheric plasma treatment. Food Chem. 2019, 301. [Google Scholar] [CrossRef] [PubMed]
- Afsah-Hejri, L.; Hajeb, P.; Ehsani, R.J. Application of ozone for degradation of mycotoxins in food: A review. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1777–1808. [Google Scholar] [CrossRef]
- Milani, J.; Maleki, G. Effects of processing on mycotoxin stability in cereals. J. Sci. Food Agric. 2014, 94, 2372–2375. [Google Scholar] [CrossRef] [PubMed]
- Jard, G.; Liboz, T.; Mathieu, F.; Guyonvarch, A.; Lebrihi, A. Review of mycotoxin reduction in food and feed: From prevention in the field to detoxification by adsorption or transformation. Food Addit. Contam. A 2011, 28, 1590–1609. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, R.; Nazzi, F.; Locci, R.; Firrao, G. Degradation of fumonisin B1 by a bacterial strain isolated from soil. Biodegradation 2006, 17, 31–38. [Google Scholar] [CrossRef]
- Heinl, S.; Hartinger, D.; Thamhesl, M.; Vekiru, E.; Krska, R.; Schatzmayr, G.; Moll, W.D.; Grabherr, R. Degradation of fumonisin B1 by the consecutive action of two bacterial enzymes. J. Biotechnol. 2010, 145, 120–129. [Google Scholar] [CrossRef]
- Chlebicz, A.; Śliżewska, K. In Vitro Detoxification of Aflatoxin B1, Deoxynivalenol, Fumonisins, T-2 Toxin and Zearalenone by Probiotic Bacteria from Genus Lactobacillus and Saccharomyces cerevisiae Yeast. Probiotics Antimicro. 2020, 12, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Burgess, K.M.N.; Renaud, J.B.; McDowell, T.; Sumarah, M.W. Mechanistic Insight into the Biosynthesis and Detoxification of Fumonisin Mycotoxins. ACS Chem. Biol. 2016, 11, 2618–2625. [Google Scholar] [CrossRef] [PubMed]
- Azam, M.S.; Yu, D.; Wu, A. Enzymes for degradation of Fusarium mycotoxins. In Food Safety & Mycotoxins; Wu, A., Ed.; Springer: Singapore, 2019; pp. 113–135. [Google Scholar] [CrossRef]
- Alberts, J.; Schatzmayr, G.; Moll, W.D.; Davids, I.; Rheeder, J.; Burger, H.M.; Shephard, G.; Gelderblom, W. Detoxification of the fumonisin mycotoxins in maize: An enzymatic approach. Toxins 2019, 11, 523. [Google Scholar] [CrossRef] [PubMed]
- Hartinger, D.; Schwartz, H.; Hametner, C.; Schatzmayr, G.; Haltrich, D.; Moll, W.D. Enzyme characteristics of aminotransferase FumI of Sphingopyxis sp. MTA144 for deamination of hydrolyzed fumonisin B1. Appl. Microbiol. Biot. 2011, 91, 757–768. [Google Scholar] [CrossRef]
- Masching, S.; Naehrer, K.; Schwartz-Zimmermann, H.E.; Sărăndan, M.; Schaumberger, S.; Dohnal, I.; Nagl, V.; Schatzmayr, D. Gastrointestinal degradation of fumonisin B1 by carboxylesterase FumD prevents fumonisin induced alteration of sphingolipid metabolism in Turkey and swine. Toxins 2016, 8, 84. [Google Scholar] [CrossRef]
- Schertz, H.; Kluess, J.; Frahm, J.; Schatzmayr, D.; Dohnal, I.; Bichl, G.; Schwartz-Zimmermann, H.; Breves, G.; Dänicke, S. Oral and intravenous fumonisin exposure in pigs—a single-dose treatment experiment evaluating toxicokinetics and detoxification. Toxins 2018, 10, 150. [Google Scholar] [CrossRef]
- Loi, M.; Fanelli, F.; Cimmarusti, M.T.; Mirabelli, V.; Haidukowski, M.; Logrieco, A.F.; Caliandro, R.; Mule, G. In vitro single and combined mycotoxins degradation by Ery4 laccase from Pleurotus eryngii and redox mediators. Food Control 2018, 90, 401–406. [Google Scholar] [CrossRef]
- Wang, X.; Qin, X.; Hao, Z.; Luo, H.; Yao, B.; Su, X. Degradation of four major mycotoxins by eight manganese peroxidases in presence of a dicarboxylic acid. Toxins 2019, 11, 566. [Google Scholar] [CrossRef] [PubMed]
- Avantaggaito, G.; Solfrizzo, M.; Visconti, A. Recent advances on the use of adsorbent materials for detoxification of Fusarium mycotoxins. Food Addit. Contam. 2005, 22, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.A.; Mays, T.; Romoser, A.; Marroquin-Cardona, A.; Mitchell, N.J.; Elmore, S.E.; Phillips, T.D. Modified hydra bioassay to evaluate the toxicity of multiple mycotoxins and predict the detoxification efficacy of a clay-based sorbent. J. Appl. Toxicol. 2014, 34, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Yuan, C.W.; Huang, J.T.; Chen, C.C.; Tang, P.C.; Huang, J.W.; Lin, J.J.; Huang, S.Y.; Chen, S.E. Evaluation of Efficacy and Toxicity of Exfoliated Silicate Nanoclays as a Feed Additive for Fumonisin Detoxification. J. Agric. Food Chem. 2017, 65, 6564–6571. [Google Scholar] [CrossRef] [PubMed]
- Beekrum, S.; Govinden, R.; Padayachee, T.; Odhav, B. Naturally occurring phenols: A detoxification strategy for fumonisin B1. Food Addit. Contam. 2003, 20, 490–493. [Google Scholar] [CrossRef]
- Siddoo-Atwal, C.; Atwal, A.S. A possible role for honey bee products in the detoxification of mycotoxins. Acta Hortic. 2012, 963, 237–245. [Google Scholar] [CrossRef]
- Xing, F.; Hua, H.; Selvaraj, J.N.; Yuan, Y.; Zhao, Y.; Zhou, L.; Liu, Y. Degradation of fumonisin B1 by cinnamon essential oil. Food Control 2014, 38, 37–40. [Google Scholar] [CrossRef]
- Gilchrist, D.G. Mycotoxins reveal connections between plants and animals in apoptosis and ceramide signaling. Cell Death Differ. 1997, 4, 689–698. [Google Scholar] [CrossRef]
- Jacobson, M.D.; Weil, M.; Raff, M.C. Programmed cell death in animal development. Cell 1997, 88, 347–354. [Google Scholar] [CrossRef]
- Green, D.R. The Coming Decade of Cell Death Research: Five Riddles. Cell 2019, 177, 1094–1107. [Google Scholar] [CrossRef]
- Das, A.; Kawai-Yamada, M.; Uchimiya, H. Programmed cell death in plants. In Abiotic Stress Adaptation in Plants: Physiological, Molecular and Genomic Foundation; Pareek, A., Sopory, S.K., Bohnert, H.J., Govindjee, Eds.; Springer: Amsterdam, The Netherlands, 2010; pp. 371–383. [Google Scholar] [CrossRef]
- Bedoui, S.; Herold, M.J.; Strasser, A. Emerging connectivity of programmed cell death pathways and its physiological implications. Nat. Rev. Mol. Cell Biol. 2020, 21, 678–695. [Google Scholar] [CrossRef] [PubMed]
- Rantong, G.; Gunawardena, A.H.L.A.N. Programmed cell death: Genes involved in signaling, regulation, and execution in plants and animals. Botany 2015, 93, 193–210. [Google Scholar] [CrossRef]
- Valandro, F.; Menguer, P.K.; Cabreira-Cagliari, C.; Margis-Pinheiro, M.; Cagliari, A. Programmed cell death (PCD) control in plants: New insights from the Arabidopsis thaliana deathosome. Plant Sci. 2020, 299. [Google Scholar] [CrossRef] [PubMed]
- Slotte, J.P. Biological functions of sphingomyelins. Prog. Lipid Res. 2013, 52, 424–437. [Google Scholar] [CrossRef]
- Luttgeharm, K.D.; Kimberlin, A.N.; Cahoon, E.B. Plant sphingolipid metabolism and function. Subcell. Biochem. 2016, 86, 249–286. [Google Scholar] [CrossRef]
- Chao, M.V. Ceramide: A potential second messenger in the nervous system. Mol. Cell. Neurosci. 1995, 6, 91–96. [Google Scholar] [CrossRef]
- Ng, C.K.Y.; Carr, K.; McAinsh, M.R.; Powell, B.; Hetherington, A.M. Drought-induced guard cell signal transduction involves sphingosine-1-phosphate. Nature 2001, 410, 596–599. [Google Scholar] [CrossRef]
- Lynch, D.V. Evidence that sphingolipid signaling is involved in responding to low temperature. New Phytol. 2012, 194, 7–9. [Google Scholar] [CrossRef]
- Dixon, S.C.; Soriano, B.J.; Lush, R.M.; Borner, M.M.; Figg, W.D. Apoptosis: Its role in the development of malignancies and its potential as a novel therapeutic target. Annu. Pharmacother. 1997, 31, 76–82. [Google Scholar] [CrossRef]
- Govindarajah, N.; Clifford, R.; Bowden, D.; Sutton, P.A.; Parsons, J.L.; Vimalachandran, D. Sphingolipids and acid ceramidase as therapeutic targets in cancer therapy. Crit. Rev. Oncol. Hemat. 2019, 138, 104–111. [Google Scholar] [CrossRef]
- Magnin-Robert, M.; Le Bourse, D.; Markham, J.; Dorey, S.; Clément, C.; Baillieul, F.; Dhondt-Cordelier, S. Modifications of sphingolipid content affect tolerance to hemibiotrophic and necrotrophic pathogens by modulating plant defense responses in Arabidopsis. Plant Physiol. 2015, 169, 2255–2274. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.R.; Eid, R.; Boucher, E.; Miller, K.A.; Mandato, C.A.; Greenwood, M.T. Stress is an agonist for the induction of programmed cell death: A review. BBA-Mol. Cell Res. 2019, 1866, 699–712. [Google Scholar] [CrossRef]
- Wong, K.; Li, X.B.; Hunchuk, N. N-acetylsphingosine (C2-ceramide) inhibited neutrophil superoxide formation and calcium influx. J. Biol. Chem. 1995, 270, 3056–3062. [Google Scholar] [CrossRef]
- Bi, F.C.; Liu, Z.; Wu, J.X.; Liang, H.; Xi, X.L.; Fang, C.; Sun, T.J.; Yin, J.; Dai, G.Y.; Rong, C.; et al. Loss of ceramide kinase in Arabidopsis impairs defenses and promotes ceramide accumulation and mitochondrial H2O2 burstsc w. Plant Cell 2014, 26, 3449–3469. [Google Scholar] [CrossRef]
- Townley, H.E.; McDonald, K.; Jenkins, G.I.; Knight, M.R.; Leaver, C.J. Ceramides induce programmed cell death in Arabidopsis cells in a calcium-dependent manner. Biol. Chem. 2005, 386, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Lachaud, C.; Prigent, E.; Thuleau, P.; Grat, S.; Da Silva, D.; Brière, C.; Mazars, C.; Cotelle, V. 14-3-3-Regulated Ca 2+ -dependent protein kinase CPK3 is required for sphingolipid-induced cell death in Arabidopsis. Cell Death Differ. 2013, 20, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, J.T.; Silverman, F.P.; Liang, H. Uncoupling salicylic acid-dependent cell death and defense-related responses from disease resistance in the Arabidopsis mutant acd5. Genetics 2000, 156, 341–350. [Google Scholar] [PubMed]
- Liang, H.; Yao, N.; Song, J.T.; Luo, S.; Lu, H.; Greenberg, J.T. Ceramides modulate programmed cell death in plants. Gene. Dev. 2003, 17, 2636–2641. [Google Scholar] [CrossRef]
- Simanshu, D.K.; Zhai, X.; Munch, D.; Hofius, D.; Markham, J.E.; Bielawski, J.; Bielawska, A.; Malinina, L.; Molotkovsky, J.G.; Mundy, J.W.; et al. Arabidopsis accelerated cell death 11, ACD11, Is a ceramide-1-phosphate transfer protein and intermediary regulator of phytoceramide levels. Cell Rep. 2014, 6, 388–399. [Google Scholar] [CrossRef]
- Kim, S.; Fyrst, H.; Saba, J. Accumulation of phosphorylated sphingoid long chain bases results in cell growth inhibition in Saccharomyces cerevisiae. Genetics 2000, 156, 1519–1529. [Google Scholar] [PubMed]
- Nagano, M.; Takahara, K.; Fujimoto, M.; Tsutsumi, N.; Uchimiya, H.; Kawai-Yamada, M. Arabidopsis sphingolipid fatty acid 2-hydroxylases (AtFAH1 and AtFAH2) are functionally differentiated in fatty acid 2-hydroxylation and stress responses. Plant Physiol. 2012, 159, 1138–1148. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Li, J.; Bostock, R.M.; Gilchrist, D.G. Apoptosis: A functional paradigm for programmed plant cell death induced by a host-selective phytotoxin and invoked during development. Plant Cell 1996, 8, 375–391. [Google Scholar] [CrossRef]
- Asai, T.; Stone, J.M.; Heard, J.E.; Kovtun, Y.; Yorgey, P.; Sheen, J.; Ausubel, F.M. Fumonisin B1-induced cell death in Arabidopsis protoplasts requires jasmonate-, ethylene-, and salicylate-dependent signaling pathways. Plant Cell 2000, 12, 1823–1835. [Google Scholar] [CrossRef]
- Stone, J.M.; Heard, J.E.; Asai, T.; Ausubel, F.M. Simulation of fungal-mediated cell death by fumonisin B1 and selection of fumonisin B1-resistant (fbr) Arabidopsis mutants. Plant Cell 2000, 12, 1811–1822. [Google Scholar] [CrossRef]
- Wang, H.; Jones, C.; Ciacci-Zanella, J.; Holt, T.; Gilchrist, D.G.; Dickman, M.B. Fumonisins and Alternaria alternata lycopersici toxins: Sphinganine analog mycotoxins induce apoptosis in monkey kidney cells. Proc. Natl. Acad. Sci. USA 1996, 93, 3461–3465. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.; Norred, W.P.; Bacon, C.W.; Riley, R.T.; Merrill, A.H., Jr. Inhibition of sphingolipid biosynthesis by fumonisins. Implications for diseases associated with Fusarium moniliforme. J. Biol. Chem. 1991, 266, 14486–14490. [Google Scholar] [PubMed]
- Merrill, A.H.; Wang, E.; Gilchrist, D.G.; Riley, R.T. Fumonisins and other inhibitors of de novo sphingolipid biosynthesis. Adv. Lipid Res. 1993, 26, 215–234. [Google Scholar] [PubMed]
- Gilchrist, D.G.; Wang, H.; Bostock, R.M. Sphingosine-related mycotoxins in plant and animal diseases. Can. J. Bot. 1995, 73, s459–s467. [Google Scholar] [CrossRef]
- Merrill, A.H., Jr.; Liotta, D.C.; Riley, R.I. Fumonisins: Fungal toxins that shed light on sphingolipid function. Trends Cell Biol. 1996, 6, 218–223. [Google Scholar] [CrossRef]
- Markham, J.E.; Molino, D.; Gissot, L.; Bellec, Y.; Hématy, K.; Marion, J.; Belcram, K.; Palauqui, J.C.; Satiat-Jeunemaître, B.; Faure, J.D. Sphingolipids containing very-long-chain fatty acids define a secretory pathway for specific polar plasma membrane protein targeting in Arabidopsis. Plant Cell 2011, 23, 2362–2378. [Google Scholar] [CrossRef]
- Ternes, P.; Feussner, K.; Werner, S.; Lerche, J.; Iven, T.; Heilmann, I.; Riezman, H.; Feussner, I. Disruption of the ceramide synthase LOH1 causes spontaneous cell death in Arabidopsis thaliana. New Phytol. 2011, 192, 841–854. [Google Scholar] [CrossRef]
- Moussatos, V.V.; Yang, S.F.; Ward, B.; Gilchrist, D.G. AAL-toxin induced physiological changes in lycopersicon esculentum mill: Roles for ethylene and pyrimidine intermediates in necrosis. Physiol. Mol. Plant Pathol. 1994, 44, 455–468. [Google Scholar] [CrossRef]
- Moore, T.; Martineau, B.; Bostock, R.M.; Lincoln, J.E.; Gilchrist, D.G. Molecular and genetic characterization of ethylene involvement in mycotoxin-induced plant cell death. Physiol. Mol. Plant Pathol. 1999, 54, 73–85. [Google Scholar] [CrossRef]
- Gechev, T.S.; Gadjev, I.Z.; Hille, J. An extensive microarray analysis of AAL-toxin-induced cell death in Arabidopsis thaliana brings new insights into the complexity of programmed cell death in plants. Cell. Mol. Life Sci. 2004, 61, 1185–1197. [Google Scholar] [CrossRef]
- Mase, K.I.; Mizuno, T.; Ishihama, N.; Fujii, T.; Mori, H.; Kodama, M.; Yoshioka, H. Ethylene signaling pathway and MAPK cascades are required for AAL toxin-induced Programmed cell death. Mol. Plant Microbe Interact. 2012, 25, 1015–1025. [Google Scholar] [CrossRef]
- Sultana, A.; Boro, P.; Mandal, K.; Chattopadhyay, S. AAL-toxin induced stress in Arabidopsis thaliana is alleviated through GSH-mediated salicylic acid and ethylene pathways. Plant Cell Tiss. Org. 2020, 141, 299–314. [Google Scholar] [CrossRef]
- Egusa, M.; Ozawa, R.; Takabayashi, J.; Otani, H.; Kodama, M. The jasmonate signaling pathway in tomato regulates susceptibility to a toxin-dependent necrotrophic pathogen. Planta 2009, 229, 965–976. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Jia, C.; Liu, L.; Zhang, Z.; Li, C.; Wang, Q. The involvement of jasmonates and ethylene in Alternaria alternata f. sp. lycopersici toxin-induced tomato cell death. J. Exp. Bot. 2011, 62, 5405–5418. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Koh, J.; Liu, L.; Shao, Z.; Liu, H.; Hu, S.; Zhu, N.; Dufresne, C.P.; Chen, S.; Wang, Q. Critical Role of COI1-Dependent Jasmonate Pathway in AAL toxin induced PCD in Tomato Revealed by Comparative Proteomics. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Witsenboer, H.M.A.; van de Griend, E.G.; Tiersma, J.B.; Nijkamp, H.J.J.; Hille, J. Tomato resistance to Alternaria stem canker: Localization in host genotypes and functional expression compared to non-host resistance. Theor. Appl. Genet. 1989, 78, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Van Der Biezen, E.A.; Nijkamp, H.J.J.; Hille, J. Mutations at the Asc locus of tomato confer resistance to the fungal pathogen Alternaria alternata f. sp. lycopersici. Theor. Appl. Genet. 1996, 92, 898–904. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Brandwagt, B.F.; Kneppers, T.J.A.; John, J.H.; Nijkamp, H.J.J.; Hille, J. Overexpression of the tomato Asc-1 gene mediates high insensitivity to AAL toxins and fumonisin B1 in tomato hairy roots and confers resistance to Alternaria alternata f. sp. lycopersici in Nicotiana umbratica plants. Mol. Plant Microbe Interact. 2002, 15, 35–42. [Google Scholar] [CrossRef]
- Brandwagt, B.F.; Mesbah, L.A.; Takken, F.L.W.; Laurent, P.L.; Kneppers, T.J.A.; Hille, J.; Nijkamp, H.J.J. A longevity assurance gene homolog of tomato mediates resistance to Altemaria alternata f. sp. lycopersici toxins and fumonisin B1. Proc. Natl. Acad. Sci. USA 2000, 97, 4961–4966. [Google Scholar] [CrossRef]
- Brandwagt, B.F.; Kneppers, T.J.A.; Van der Weerden, G.M.; Nijkamp, H.J.J.; Hille, J. Most AAL toxin-sensitive Nicotiana species are resistant to the tomato fungal pathogen Alternaria alternata f. sp. lycopersici. Mol. Plant Microbe Interact. 2001, 14, 460–470. [Google Scholar] [CrossRef]
- Egusa, M.; Miwa, T.; Kaminaka, H.; Takano, Y.; Kodama, M. Nonhost resistance of Arabidopsis thaliana against Alternaria alternata involves both pre- and postinvasive defenses but is collapsed by AAL-toxin in the absence of LOH2. Phytopathology 2013, 103, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Tsuge, T.; Harimoto, Y.; Akimitsu, K.; Ohtani, K.; Kodama, M.; Akagi, Y.; Egusa, M.; Yamamoto, M.; Otani, H. Host-selective toxins produced by the plant pathogenic fungus Alternaria alternata. FEMS Microbiol. Rev. 2012, 37, 44–66. [Google Scholar] [CrossRef] [PubMed]
- de Zélicourt, A.; Montiel, G.; Pouvreau, J.B.; Thoiron, S.; Delgrange, S.; Simier, P.; Delavault, P. Susceptibility of Phelipanche and Orobanche species to AAL-toxin. Planta 2009, 230, 1047–1055. [Google Scholar] [CrossRef]
- Lincoln, J.E.; Richael, C.; Overduin, B.; Smith, K.; Bostock, R.; Gilchrist, D.G. Expression of the antiapoptotic baculovirus p35 gene in tomato blocks programmed cell death and provides broad-spectrum resistance to disease. Proc. Natl. Acad. Sci. USA 2002, 99, 15217–15221. [Google Scholar] [CrossRef]
- Qureshi, M.K.; Sujeeth, N.; Gechev, T.S.; Hille, J. The zinc finger protein ZAT11 modulates paraquat-induced programmed cell death in Arabidopsis thaliana. Acta Physiol. Plant. 2013, 35, 1863–1871. [Google Scholar] [CrossRef]
- Shao, Z.; Zhao, Y.; Liu, L.; Chen, S.; Li, C.; Meng, F.; Liu, H.; Hu, S.; Wang, J.; Wang, Q. Overexpression of FBR41 enhances resistance to sphinganine analog mycotoxin-induced cell death and Alternaria stem canker in tomato. Plant Biotechnol. J. 2020, 18, 141–154. [Google Scholar] [CrossRef]
- Al-Jaal, B.A.; Jaganjac, M.; Barcaru, A.; Horvatovich, P.; Latiff, A. Aflatoxin, fumonisin, ochratoxin, zearalenone and deoxynivalenol biomarkers in human biological fluids: A systematic literature review, 2001–2018. Food Chem. Toxicol. 2019, 129, 211–228. [Google Scholar] [CrossRef] [PubMed]
- Alberts, J.; Rheeder, J.; Gelderblom, W.; Shephard, G.; Burger, H.M. Rural subsistence maize farming in South Africa: Risk assessment and intervention models for reduction of exposure to fumonisin mycotoxins. Toxins 2019, 11, 334. [Google Scholar] [CrossRef] [PubMed]
- Cendoya, E.; Chiotta, M.L.; Zachetti, V.; Chulze, S.N.; Ramirez, M.L. Fumonisins and fumonisin-producing Fusarium occurrence in wheat and wheat by products: A review. J. Cereal Sci. 2018, 80, 158–166. [Google Scholar] [CrossRef]
- Dall’Asta, C.; Battilani, P. Fumonisins and their modified forms, a matter of concern in future scenario? World Mycotoxin J. 2016, 9, 727–739. [Google Scholar] [CrossRef]
- Alberts, J.F.; van Zyl, W.H.; Gelderblom, W.C.A. Biologically based methods for control of fumonisin-producing Fusarium species and reduction of the fumonisins. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef]
- Gil-Serna, J.; Vázquez, C.; Patiño, B. Genetic regulation of aflatoxin, ochratoxin A, trichothecene, and fumonisin biosynthesis: A review. Int. Microbiol. 2020, 23, 89–96. [Google Scholar] [CrossRef]
- Santiago, R.; Cao, A.; Butrón, A. Genetic factors involved in fumonisin accumulation in maize kernels and their implications in maize agronomic management and breeding. Toxins 2015, 7, 3267–3296. [Google Scholar] [CrossRef]
- Chavez, R.A.; Cheng, X.; Stasiewicz, M.J. A review of the methodology of analyzing aflatoxin and fumonisin in single corn kernels and the potential impacts of these methods on food security. Foods 2020, 9, 297. [Google Scholar] [CrossRef]
- Deepa, N.; Sreenivasa, M.Y. Molecular methods and key genes targeted for the detection of fumonisin producing Fusarium verticillioides–An updated review. Food Biosci. 2019, 32. [Google Scholar] [CrossRef]
- Santiago, R.; Cao, A.; Malvar, R.A.; Butrón, A. Genomics of maize resistance to Fusarium ear rot and fumonisin contamination. Toxins 2020, 12, 431. [Google Scholar] [CrossRef]
- Diaz-Gomez, J.; Marin, S.; Capell, T.; Sanchis, V.; Ramos, A.J. The impact of Bacillus thuringiensis technology on the occurrence of fumonisins and other mycotoxins in maize. World Mycotoxin J. 2016, 9, 475–486. [Google Scholar] [CrossRef]
- Sheik Abdul, N.; Marnewick, J.L. Fumonisin B1-induced mitochondrial toxicity and hepatoprotective potential of rooibos: An update. J. Appl. Toxicol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Fan, L.; Yin, S.; Chen, H.; Hu, H. Molecular mechanisms of fumonisin B1-induced toxicities and its applications in the mechanism-based interventions. Toxicon 2019, 167, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Riley, R.T.; Merrill, A.H., Jr. Ceramide synthase inhibition by fumonisins: A perfect storm of perturbed sphingolipid metabolism, signaling, and disease. J. Lipid Res. 2019, 60, 1183–1189. [Google Scholar] [CrossRef]
- Chen, C.; Riley, R.T.; Wu, F. Dietary Fumonisin and Growth Impairment in Children and Animals: A Review. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1448–1464. [Google Scholar] [CrossRef]
- Pierron, A.; Alassane-Kpembi, I.; Oswald, I.P. Impact of two mycotoxins deoxynivalenol and fumonisin on pig intestinal health. Porcine Health Manag. 2016, 2. [Google Scholar] [CrossRef]
- Wang, X.; Wu, Q.; Wan, D.; Liu, Q.; Chen, D.; Liu, Z.; Martinez-Larrañaga, M.R.; Martínez, M.A.; Anadón, A.; Yuan, Z. Fumonisins: Oxidative stress-mediated toxicity and metabolism in vivo and in vitro. Arch. Toxicol. 2015, 90, 81–101. [Google Scholar] [CrossRef]
- Nützmann, H.W.; Scazzocchio, C.; Osbourn, A. Metabolic gene clusters in eukaryotes. Annu. Rev. Genet. 2018, 52, 159–183. [Google Scholar] [CrossRef]
- Seo, J.A.; Proctor, R.H.; Plattner, R.D. Characterization of four clustered and coregulated genes associated with fumonisin biosynthesis in Fusarium verticillioides. Fungal Genet. Biol. 2001, 34, 155–165. [Google Scholar] [CrossRef]
- Proctor, R.H.; Brown, D.W.; Plattner, R.D.; Desjardins, A.E. Co-expression of 15 contiguous genes delineates a fumonisin biosynthetic gene cluster in Gibberella moniliformis. Fungal Genet. Biol. 2003, 38, 237–249. [Google Scholar] [CrossRef]
- Butchko, R.A.E.; Plattner, R.D.; Proctor, R.H. Deletion analysis of FUM genes involved in tricarballylic ester formation during fumonisin biosynthesis. J. Agric. Food Chem. 2006, 54, 9398–9404. [Google Scholar] [CrossRef] [PubMed]
- Proctor, R.H.; Busman, M.; Seo, J.A.; Lee, Y.W.; Plattner, R.D. A fumonisin biosynthetic gene cluster in Fusarium oxysporum strain O-1890 and the genetic basis for B versus C fumonisin production. Fungal Genet. Biol. 2008, 45, 1016–1026. [Google Scholar] [CrossRef] [PubMed]
- Stępień, Ł.; Koczyk, G.; Waśkiewicz, A. Genetic and phenotypic variation of Fusarium proliferatum isolates from different host species. J. Appl. Genet. 2011, 52, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Janevska, S.; Ferling, I.; Jojić, K.; Rautschek, J.; Hoefgen, S.; Proctor, R.H.; Hillmann, F.; Valiante, V. Self-Protection against the Sphingolipid Biosynthesis Inhibitor Fumonisin B1 Is Conferred by a FUM Cluster-Encoded Ceramide Synthase. mBio 2020, 11. [Google Scholar] [CrossRef]
- Brown, D.W.; Butchko, R.A.E.; Busman, M.; Proctor, R.H. The Fusarium verticillioides FUM gene cluster encodes a Zn(II)2Cys6 protein that affects FUM gene expression and fumonisin production. Eukaryot. Cell 2007, 6, 1210–1218. [Google Scholar] [CrossRef]
- Desjardins, A.E.; Munkvold, G.P.; Plattner, R.D.; Proctor, R.H. FUM1-A gene required for fumonisin biosynthesis but not for maize ear rot and ear infection by Gibberella moniliformis in field tests. Mol. Plant Microbe Interact. 2002, 15, 1157–1164. [Google Scholar] [CrossRef]
- Proctor, R.H.; Desjardins, A.E.; Plattner, R.D.; Hohn, T.M. A polyketide synthase gene required for biosynthesis of fumonisin mycotoxins in Gibberella fujikuroi mating population A. Fungal Genet. Biol. 1999, 27, 100–112. [Google Scholar] [CrossRef]
- Bojja, R.S.; Cerny, R.L.; Proctor, R.H.; Du, L. Determining the Biosynthetic Sequence in the Early Steps of the Fumonisin Pathway by Use of Tree Gene-Disruption Mutants of Fusarium verticillioides. J. Agric. Food Chem. 2004, 52, 2855–2860. [Google Scholar] [CrossRef]
- Massi, F.P.; Sartori, D.; de Souza Ferranti, L.; Iamanaka, B.T.; Taniwaki, M.H.; Vieira, M.L.C.; Fungaro, M.H.P. Prospecting for the incidence of genes involved in ochratoxin and fumonisin biosynthesis in Brazilian strains of Aspergillus niger and Aspergillus welwitschiae. Int. J. Food Microbiol. 2016, 221, 19–28. [Google Scholar] [CrossRef]
- Desjardins, A.E.; Plattner, R.D.; Proctor, R.H. Genetic and biochemical aspects of fumonisin production. Adv. Exp. Med. Biol. 1996, 392, 165–173. [Google Scholar] [CrossRef]
- Butchko, R.A.E.; Plattner, R.D.; Proctor, R.H. FUM13 encodes a short chain dehydrogenase/reductase required for C-3 carbonyl reduction during fumonisin biosynthesis in Gibberella moniliformis. J. Agric. Food Chem. 2003, 51, 3000–3006. [Google Scholar] [CrossRef] [PubMed]
- Butchko, R.A.E.; Plattner, R.D.; Proctor, R.H. FUM9 Is Required for C-5 Hydroxylation of Fumonisins and Complements the Meitotically Defined Fum3 Locus in Gibberella moniliformis. Appl. Environ. Microb. 2003, 69, 6935–6937. [Google Scholar] [CrossRef] [PubMed]
- Yi, H.; Bojja, R.S.; Fu, J.; Du, L. Direct evidence for the function of FUM13 in 3-ketoreduction of mycotoxin fumonisins in Fusarium verticillioides. J. Agric. Food Chem. 2005, 53, 5456–5460. [Google Scholar] [CrossRef] [PubMed]
- Proctor, R.H.; Plattner, R.D.; Desjardins, A.E.; Busman, M.; Butchko, R.A.E. Fumonisin production in the maize pathogen Fusarium verticillioides: Genetic basis of naturally occurring chemical variation. J. Agric. Food Chem. 2006, 54, 2424–2430. [Google Scholar] [CrossRef]
- Pel, H.J.; De Winde, J.H.; Archer, D.B. Genome sequencing and analysis of the versatile cell factory Aspergillus niger CBS 513.88. Nat. Biotechnol. 2007, 25, 221–231. [Google Scholar] [CrossRef]
- Rösler, S.M.; Sieber, C.M.K.; Humpf, H.U.; Tudzynski, B. Interplay between pathway-specific and global regulation of the fumonisin gene cluster in the rice pathogen Fusarium fujikuroi. Appl. Microbiol. Biotechnol. 2016, 100, 5869–5882. [Google Scholar] [CrossRef]
- Myung, K.; Li, S.; Butchko, R.A.E.; Busman, M.; Proctor, R.H.; Abbas, H.K.; Calvo, A.M. FvVE1 regulates biosynthesis of the mycotoxins fumonisins and fusarins in Fusarium verticiliioides. J. Agric. Food Chem. 2009, 57, 5089–5094. [Google Scholar] [CrossRef]
- Kim, H.; Woloshuk, C.P. Functional characterization of fst1 in Fusarium verticillioides during colonization of maize kernels. Mol. Plant Microbe Interact. 2011, 24, 18–24. [Google Scholar] [CrossRef]
- Shin, J.H.; Kim, J.E.; Malapi-Wight, M.; Choi, Y.E.; Shaw, B.D.; Shim, W.B. Protein phosphatase 2A regulatory subunits perform distinct functional roles in the maize pathogen Fusarium verticillioides. Mol. Plant Pathol. 2013, 14, 518–529. [Google Scholar] [CrossRef]
- Ridenour, J.B.; Bluhm, B.H. The novel fungal-specific gene FUG1 has a role in pathogenicity and fumonisin biosynthesis in Fusarium verticillioides. Mol. Plant Pathol. 2017, 18, 513–528. [Google Scholar] [CrossRef]
- Niehaus, E.M.; Münsterkötter, M.; Proctor, R.H.; Brown, D.W.; Sharon, A.; Idan, Y.; Oren-Young, L.; Sieber, C.M.; Novák, O.; Pěnčík, A.; et al. Comparative “omics” of the Fusarium fujikuroi species complex highlights differences in genetic potential and metabolite synthesis. Genome Biol. Evol. 2016, 8, 3574–3599. [Google Scholar] [CrossRef] [PubMed]
- Montis, V.; Pasquali, M.; Visentin, I.; Karlovsky, P.; Cardinale, F. Identification of a cis-acting factor modulating the transcription of FUM1, a key fumonisin-biosynthetic gene in the fungal maize pathogen Fusarium verticillioides. Fungal Genet. Biol. 2013, 51, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Kohut, G.; Ádám, A.L.; Fazekas, B.; Hornok, L. N-starvation stress induced FUM gene expression and fumonisin production is mediated via the HOG-type MAPK pathway in Fusarium proliferatum. Int. J. Food Microbiol. 2009, 130, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Medina, A.; Schmidt-Heydt, M.; Cárdenas-Chávez, D.L.; Parra, R.; Geisen, R.; Magan, N. Integrating toxin gene expression, growth and fumonisin B1 and B2 production by a strain of Fusarium verticillioides under different environmental factors. J. R. Soc. Interface 2013, 10. [Google Scholar] [CrossRef]
- Stepień, Ł.; Waśkiewicz, A.; Wilman, K. Host extract modulates metabolism and fumonisin biosynthesis by the plant-pathogenic fungus Fusarium proliferatum. Int. J. Food Microbiol. 2015, 193, 74–81. [Google Scholar] [CrossRef]
- Wu, Y.; Li, T.; Gong, L.; Wang, Y.; Jiang, Y. Effects of different carbon sources on fumonisin production and fum gene expression by Fusarium proliferatum. Toxins 2019, 11, 289. [Google Scholar] [CrossRef] [PubMed]
- Witaszak, N.; Lalak-Kańczugowska, J.; Waśkiewicz, A.; Stȩpień, Ł. The impacts of asparagus extract fractions on growth and fumonisins biosynthesis in Fusarium proliferatum. Toxins 2020, 12, 95. [Google Scholar] [CrossRef]
- Johnson, L.; Harrison, J.H.; Hunt, C.; Shinners, K.; Doggett, C.G.; Sapienza, D. Nutritive value of corn silage as affected by maturity and mechanical processing: A contemporary review. J. Dairy Sci. 1999, 82, 2813–2825. [Google Scholar] [CrossRef]
- Mansfield, M.A.; Archibald, D.D.; Jones, A.D.; Kuldau, G.A. Relationship of sphinganine analog mycotoxin contamination in maize silage to seasonal weather conditions and to agronomic and ensiling practices. Phytopathology 2007, 97, 504–511. [Google Scholar] [CrossRef]
- Baker, S.E. Aspergillus niger genomics: Past, present and into the future. Med. Mycol. 2006, 44, 17–21. [Google Scholar] [CrossRef]
- Brown, D.W.; Baker, S.E. Mycotoxins: A fungal genomics perspective. Methods Mol. Biol. 2017, 1542, 367–379. [Google Scholar] [CrossRef] [PubMed]
- Noonim, P.; Mahakarnchanakul, W.; Nielsen, K.F.; Frisvad, J.C.; Samson, R.A. Fumonisin B2 production by Aspergillus niger in Thai coffee beans. Food Addit. Contam. A 2009, 26, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Månsson, M.; Klejnstrup, M.L.; Phipps, R.K.; Nielsen, K.F.; Frisvad, J.C.; Gotfredsen, C.H.; Larsen, T.O. Isolation and NMR characterization of fumonisin b2 and a new fumonisin B6 from aspergillus niger. J. Agric. Food Chem. 2010, 58, 949–953. [Google Scholar] [CrossRef] [PubMed]
- Palencia, E.R.; Mitchell, T.R.; Snook, M.E.; Glenn, A.E.; Gold, S.; Hinton, D.M.; Riley, R.T.; Bacon, C.W. Analyses of black Aspergillus species of peanut and maize for ochratoxins and fumonisins. J. Food Prot. 2014, 77, 805–813. [Google Scholar] [CrossRef]
- Desjardins, A.E.; Plattner, R.D.; Proctor, R.H. Linkage among genes responsible for fumonisin biosynthesis in Gibberella fujikuroi mating population A. Appl. Environ. Microb. 1996, 62, 2571–2576. [Google Scholar] [CrossRef]
- Ding, Y.; Bojja, R.S.; Du, L. Fum3p, a 2-Ketoglutarate-Dependent Dioxygenase Required for C-5 Hydroxylation of Fumonisins in Fusarium verticillioides. Appl. Environ. Microb. 2004, 70, 1931–1934. [Google Scholar] [CrossRef]
- Plattner, R.D.; Desjardins, A.E.; Leslie, J.F.; Nelson, P.E. Identification and characterization of strains of Gibberella fujikuroi mating population A with rare fumonisin production phenotypes. Mycologia 1996, 88, 416–424. [Google Scholar] [CrossRef]
- Uhlig, S.; Busman, M.; Shane, D.S.; Rønning, H.; Rise, F.; Proctor, R. Identification of early fumonisin biosynthetic intermediates by inactivation of the FUM6 gene in Fusarium verticillioides. J. Agric. Food Chem. 2012, 60, 10293–10301. [Google Scholar] [CrossRef]
- Li, Y.; Lou, L.; Cerny, R.L.; Butchko, R.A.E.; Proctor, R.H.; Shen, Y.; Du, L. Tricarballylic ester formation during biosynthesis of fumonisin mycotoxins in Fusarium verticillioides. Mycology 2013, 4, 179–186. [Google Scholar] [CrossRef][Green Version]
- Zaleta-Rivera, K.; Xu, C.; Yu, F.; Butchko, R.A.E.; Proctor, R.H.; Hidalgo-Lara, M.E.; Raza, A.; Dussault, P.H.; Du, L. A bidomain nonribosomal peptide synthetase encoded by FUM14 catalyzes the formation of tricarballylic esters in the biosynthesis of fumonisins. Biochemistry 2006, 45, 2561–2569. [Google Scholar] [CrossRef]
- Brown, D.W.; Cheung, F.; Proctor, R.H.; Butchko, R.A.E.; Zheng, L.; Lee, Y.; Utterback, T.; Smith, S.; Feldblyum, T.; Glenn, A.E.; et al. Comparative analysis of 87,000 expressed sequence tags from the fumonisin-producing fungus Fusarium verticillioides. Fungal Genet. Biol. 2005, 42, 848–861. [Google Scholar] [CrossRef] [PubMed]
- Susca, A.; Proctor, R.H.; Morelli, M.; Haidukowski, M.; Gallo, A.; Logrieco, A.F.; Moretti, A. Variation in fumonisin and ochratoxin production associated with differences in biosynthetic gene content in Aspergillus niger and A. welwitschiae isolates from multiple crop and geographic origins. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Yamada, O.; Machida, M.; Hosoyama, A.; Goto, M.; Takahashi, T.; Futagami, T.; Yamagata, Y.; Takeuchi, M.; Kobayashi, T.; Koike, H.; et al. Genome sequence of Aspergillus luchuensis NBRC 4314. DNA Res. 2016, 23, 507–515. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Akagi, Y.; Akamatsu, H.; Otani, H.; Kodama, M. Horizontal chromosome transfer, a mechanism for the evolution and differentiation of a plant-pathogenic fungus. Eukaryot. Cell 2009, 8, 1732–1738. [Google Scholar] [CrossRef]
- Akagi, Y.; Taga, M.; Yamamoto, M.; Tsuge, T.; Fukumasa-Nakai, Y.; Otani, H.; Kodama, M. Chromosome constitution of hybrid strains constructed by protoplast fusion between the tomato and strawberry pathotypes of Alternaria alternata. J. Gen. Plant Pathol. 2009, 75, 101–109. [Google Scholar] [CrossRef]
- Akamatsu, H.; Otani, H.; Kodama, M. Characterization of a gene cluster for host-specific AAL-toxin biosynthesis in the tomato pathotype of Alternaria alternata. Fungal Genet. Rep. 2003, 50, 355. [Google Scholar] [CrossRef]
- Zhu, X.; Vogeler, C.; Du, L. Functional complementation of fumonisin biosynthesis in FUM1-disrupted Fusarium verticillioides by the AAL-toxin Polyketide synthase gene ALT1 from Alternaria alternata f. sp. Lycopersici. J. Nat. Prod. 2008, 71, 957–960. [Google Scholar] [CrossRef]
- Takao, K.; Akagi, Y.; Tsuge, T.; Harimoto, Y.; Yamamoto, M.; Kodama, M. The global regulator LaeA controls biosynthesis of host-specific toxins, pathogenicity and development of Alternaria alternata pathotypes. J. Gen. Plant Pathol. 2016, 82, 121–131. [Google Scholar] [CrossRef]
- Vaquera, S.; Patriarca, A.; Cabrera, G.; Fernández Pinto, V. Temperature and water activity influence on simultaneous production of AAL toxins by Alternaria arborescens on tomato medium. Eur. J. Plant Pathol. 2017, 148, 1003–1009. [Google Scholar] [CrossRef]
- Kroken, S.; Glass, N.L.; Taylor, J.W.; Yoder, O.C.; Turgeon, B.G. Phylogenomic analysis of type I polyketide synthase genes in pathogenic and saprobic ascomycetes. Proc. Natl. Acad. Sci. USA 2003, 100, 15670–15675. [Google Scholar] [CrossRef]
- Condon, B.J.; Leng, Y.; Wu, D.; Bushley, K.E.; Ohm, R.A.; Otillar, R.; Martin, J.; Schackwitz, W.; Grimwood, J.; MohdZainudin, N.A.I.; et al. Comparative Genome Structure, Secondary Metabolite, and Effector Coding Capacity across Cochliobolus Pathogens. PLoS Genet. 2013, 9. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Lohmar, J.M.; Busman, M.; Brown, D.W.; Naumann, T.A.; Divon, H.H.; Uhlig, S.; Proctor, R.H. Identification and distribution of gene clusters required for synthesis of sphingolipid metabolism inhibitors in diverse species of the filamentous fungus Fusarium. BMC Genom. 2020, 21. [Google Scholar] [CrossRef]
- Sultana, S.; Kitajima, M.; Kobayashi, H.; Nakagawa, H.; Shimizu, M.; Kageyama, K.; Suga, H. A natural variation of fumonisin gene cluster associated with fumonisin production difference in Fusarium fujikuroi. Toxins 2019, 11, 200. [Google Scholar] [CrossRef] [PubMed]
- Fumero, M.V.; Villani, A.; Susca, A.; Haidukowski, M.; Cimmarusti, M.T.; Toomajian, C.; Leslie, J.F.; Chulze, S.N.; Moretti, A. Fumonisin and Beauvericin Chemotypes and Genotypes of the Sister Species Fusarium subglutinans and Fusarium temperatum. Appl. Environ. Microb. 2020, 86. [Google Scholar] [CrossRef] [PubMed]
- Glenn, A.E.; Zitomer, N.C.; Zimeri, A.M.; Williams, L.D.; Riley, R.T.; Proctor, R.H. Transformation-mediated complementation of a FUM gene cluster deletion in Fusarium verticillioides restores both fumonisin production and pathogenicity on maize seedlings. Mol. Plant Microbe Interact. 2008, 21, 87–97. [Google Scholar] [CrossRef]
- Geiser, D.M.; Jiménez-Gasco, M.D.M.; Kang, S.; Makalowska, I.; Veeraraghavan, N.; Ward, T.J.; Zhang, N.; Kuldau, G.A.; O’Donnell, K. FUSARIUM-ID v. 1.0: A DNA sequence database for identifying Fusarium. Eur. J. Plant Pathol. 2004, 110, 473–479. [Google Scholar] [CrossRef]
- Kristensen, R.; Torp, M.; Kosiak, B.; Holst-Jensen, A. Phylogeny and toxigenic potential is correlated in Fusarium species as revealed by partial translation elongation factor 1 alpha gene sequences. Mycol. Res. 2005, 109, 173–186. [Google Scholar] [CrossRef]
- Wang, J.; Wang, X.; Zhou, Y.; Du, L.; Wang, Q. Fumonisin detection and analysis of potential fumonisin-producing Fusarium spp. in Asparagus (Asparagus officinalis L.) in Zhejiang province of China. J. Sci. Food Agric. 2010, 90, 836–842. [Google Scholar] [CrossRef]
- Jurado, M.; Marín, P.; Callejas, C.; Moretti, A.; Vázquez, C.; González-Jaén, M.T. Genetic variability and Fumonisin production by Fusarium proliferatum. Food Microbiol. 2010, 27, 50–57. [Google Scholar] [CrossRef]
- Waśkiewicz, A.; Stepień, L.; Wilman, K.; Kachlicki, P. Diversity of pea-associated F. proliferatum and F. verticillioides populations revealed by FUM1 sequence analysis and fumonisin biosynthesis. Toxins 2013, 5, 488–503. [Google Scholar] [CrossRef]
- Stępień, Ł.; Koczyk, G.; Waśkiewicz, A. FUM cluster divergence in fumonisins-producing Fusarium species. Fungal Biol. 2011, 115, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Shephard, G.S.; Sydenham, E.W.; Thiel, P.G.; Gelderblom, W.C.A. Quantitative determination of fumonisins b1 and b2 by high-performance liquid chromatography with fluorescence detection. J. Liq. Chromatogr. 1990, 13, 2077–2087. [Google Scholar] [CrossRef]
- Rottinghaus, G.E.; Coatney, C.E.; Minor, H.C. A rapid, sensitive thin layer chromatography procedure for the detection of fumonisin B1 and B2. J. Vet. Diagn. Investig. 1992, 4, 326–329. [Google Scholar] [CrossRef] [PubMed]
- Schneider, E.; Usleber, E.; Märtlbauer, E. Rapid Detection of Fumonisin B1 in Corn-Based Food by Competitive Direct Dipstick Enzyme Immunoassay/Enzyme-Linked Immunofiltration Assay with Integrated Negative Control Reaction. J. Agric. Food Chem. 1995, 43, 2548–2552. [Google Scholar] [CrossRef]
- Plattner, R.D. Detection of fumonisins produced in Fusarium moniliforme cultures by HPLC with electrospray MS and evaporative light scattering detectors. Nat. Toxins 1995, 3, 294–298. [Google Scholar] [CrossRef]
- Caldas, E.D.; Winter, C.K.; Daniel Jones, A.; Ward, B.; Gilchrist, D.G. Electrospray Ionization Mass Spectrometry of Sphinganine Analog Mycotoxins. Anal. Chem. 1995, 67, 196–207. [Google Scholar] [CrossRef]
- Szurdoki, F.; Trousdale, E.; Ward, B.; Gee, S.J.; Hammock, B.D.; Gilchrist, D.G. Synthesis of protein conjugates and development of immunoassays for AAL toxins. J. Agric. Food Chem. 1996, 44, 1796–1803. [Google Scholar] [CrossRef]
- Yu, W.; Yu, F.Y.; Undersander, D.J.; Chu, F.S. Immunoassays of selected mycotoxins in hay, silage and mixed feed. Food Agric. Immunol. 1999, 11, 307–319. [Google Scholar] [CrossRef]
- Gaspardo, B.; Del Zotto, S.; Torelli, E.; Cividino, S.R.; Firrao, G.; Della Riccia, G.; Stefanon, B. A rapid method for detection of fumonisins B1 and B2 in corn meal using Fourier transform near infrared (FT-NIR) spectroscopy implemented with integrating sphere. Food Chem. 2012, 135, 1608–1612. [Google Scholar] [CrossRef]
- Man, Y.; Liang, G.; Li, A.; Pan, L. Analytical Methods for the Determination of Alternaria Mycotoxins. Chromatographia 2017, 80, 9–22. [Google Scholar] [CrossRef]
- Mirasoli, M.; Buragina, A.; Dolci, L.S.; Simoni, P.; Anfossi, L.; Giraudi, G.; Roda, A. Chemiluminescence-based biosensor for fumonisins quantitative detection in maize samples. Biosens. Bioelectron. 2012, 32, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Zangheri, M.; Di Nardo, F.; Anfossi, L.; Giovannoli, C.; Baggiani, C.; Roda, A.; Mirasoli, M. A multiplex chemiluminescent biosensor for type B-fumonisins and aflatoxin B1 quantitative detection in maize flour. Analyst 2015, 140, 358–365. [Google Scholar] [CrossRef]
- Shu, M.; Xu, Y.; Wang, D.; Liu, X.; Li, Y.; He, Q.; Tu, Z.; Qiu, Y.; Ji, Y.; Wang, X. Anti-idiotypic nanobody: A strategy for development of sensitive and green immunoassay for Fumonisin B1. Talanta 2015, 143, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Di Nardo, F.; Alladio, E.; Baggiani, C.; Cavalera, S.; Giovannoli, C.; Spano, G.; Anfossi, L. Colour-encoded lateral flow immunoassay for the simultaneous detection of aflatoxin B1 and type-B fumonisins in a single Test line. Talanta 2019, 192, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Hou, S.; Ma, J.; Cheng, Y.; Wang, H.; Sun, J.; Yan, Y. Quantum dot nanobead-based fluorescent immunochromatographic assay for simultaneous quantitative detection of fumonisin B1, dexyonivalenol, and zearalenone in grains. Food Control 2020, 117. [Google Scholar] [CrossRef]
- Ferrara, M.; Logrieco, A.F.; Moretti, A.; Susca, A. A loop-mediated isothermal amplification (LAMP) assay for rapid detection of fumonisin producing Aspergillus species. Food Microbiol. 2020, 90. [Google Scholar] [CrossRef]
- Wigmann, É.F.; Meyer, K.; Cendoya, E.; Maul, R.; Vogel, R.F.; Niessen, L. A loop-mediated isothermal amplification (LAMP) based assay for the rapid and sensitive group-specific detection of fumonisin producing Fusarium spp. Int. J. Food Microbiol. 2020, 325. [Google Scholar] [CrossRef] [PubMed]
- Bluhm, B.H.; Flaherty, J.E.; Cousin, M.A.; Woloshuk, C.P. Multiplex polymerase chain reaction assay for the differential detection of trichothecene- and fumonisin-producing species of Fusarium in cornmeal. J. Food Protect. 2002, 65, 1955–1961. [Google Scholar] [CrossRef]
- Kim, N.Y.; Lee, I.; Ji, G.E. Reliable and simple detection of ochratoxin and fumonisin production in black Aspergillus. J. Food Protect. 2014, 77, 653–658. [Google Scholar] [CrossRef] [PubMed]
- Nagaraj, D.; Adkar-Purushothama, C.R.; Marikunte Yanjarappa, S. Multiplex PCR for the early detection of fumonisin producing Fusarium verticillioides. Food Biosci. 2016, 13, 84–88. [Google Scholar] [CrossRef]
- Gareis, M.; Bauer, J.; Thiem, J.; Plank, G.; Grabley, S.; Gedek, B. Cleavage of Zearalenone-Glycoside, a “Masked” Mycotoxin, during Digestion in Swine. J. Vet. Med. Ser. B 1990, 37, 236–240. [Google Scholar] [CrossRef]
- Berthiller, F.; Maragos, C.M.; Dall’Asta, C. Introduction to masked mycotoxin. In Masked Mycotoxins in Food: Formation, Occurrence and Toxicological Relevance; Dall’Asta, C., Berthiller, F., Eds.; Royal Society of Chemistry: Cambridge, UK, 2015; pp. 1–8. [Google Scholar]
- Liu, J.; Applegate, T. Zearalenone (ZEN) in Livestock and Poultry: Dose, toxicokinetics, toxicity and estrogenicity. Toxins 2020, 12, 377. [Google Scholar] [CrossRef] [PubMed]
- Ekwomadu, T.I.; Dada, T.A.; Nleya, N.; Gopane, R.; Sulyok, M.; Mwanza, M. Variation of Fusarium free, masked, and emerging mycotoxin metabolites in maize from Agriculture Regions of South Africa. Toxins 2020, 12, 149. [Google Scholar] [CrossRef]
- Crews, C.; MacDonald, S.J. Natural occurrence of masked mycotoxins. In Issues in Toxicology; Berthiller, F., Dall’Asta, C., Eds.; Royal Society of Chemistry: Cambridge, UK, 2016; pp. 14–31. [Google Scholar] [CrossRef]
- Bryła, M.; Roszko, M.; Szymczyk, K.; Jedrzejczak, R.; Obiedziński, M.W. Fumonisins and their masked forms in maize products. Food Control 2016, 59, 619–627. [Google Scholar] [CrossRef]
- Falavigna, C.; Cirlini, M.; Galaverna, G.; Dall’Asta, C. Masked fumonisins in processed food: Co-occurrence of hidden and bound forms and their stability under digestive conditions. World Mycotoxin J. 2012, 5, 325–334. [Google Scholar] [CrossRef]
- Latorre, A.; Dagnac, T.; Lorenzo, B.F.; Llompart, M. Occurrence and stability of masked fumonisins in corn silage samples. Food Chem. 2015, 189, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Generotti, S.; Cirlini, M.; Dall’Asta, C.; Suman, M. Influence of the industrial process from caryopsis to cornmeal semolina on levels of fumonisins and their masked forms. Food Control 2015, 48, 170–174. [Google Scholar] [CrossRef]

| Analog of SAMs | Fungi/Origin | Activities | Scopus Citation (Review) | Reference | |
|---|---|---|---|---|---|
| Myriocins (thermozymocidin, ISP-I) | Myriococcum albomyces Melanconis flavovirens Isaria sinclairii | Antifungal activity Inhibitor of serine palmitoyltransferase (SPT) Immunosuppressive activity Protective effect on hepatotoxicity Relieve fumonisin B1 (FB1)-induced toxicity and cell death Multi-pharmacological function on human | 421(34) | [46,47,48,49,50,51,52,53] | |
| Sphingofungins | E/F | Paecilomyces variotii | Inhibitor of SPT Antifungal activity | 65(15) | [54,55,56,57,58] |
| A/B/C/D/I | Asp. fumigatus | ||||
| G/H | Asp. penicilliodes | ||||
| Viridiofungins | A/B/C | Trichoderma viride Pers | Inhibitors of SPT and squalene synthase Antifungal but lack antibacterial activity | 21(5) | [59,60,61] |
| Tri. harzianum | |||||
| Australifungin | Sporormiella australis | Inhibitors of sphinganine-N acyl transferase Antifungal activity, phytotoxicity | 26(7) | [62,63] | |
| 2-AOD-3-ol | F. avenaceum | Animal cell toxicity as fumonisin B | 5 | [64] | |
| C17-sphinganine analog mycotoxin | Contaminated mussels | Blocking skeletal muscle contraction | 1 | [65] | |
| Subject | Content | Reference |
|---|---|---|
| Assessment | Biomarkers, metabolism, and biomonitoring of fumonisins in human biological fluids | [174] |
| Assessment | Impact on agriculture, food, and human health and their management strategies | [16] |
| Assessment | Risk assessment and intervention models for fumonisin of maize in South Africa | [175] |
| Assessment | Fumonisins and related Fusarium occurrence in wheat and its by-products | [176] |
| Assessment | Fumonisins and their modified forms | [177] |
| Assessment | Biological methods for fumonisins reduction and related Fusarium species control | [178] |
| Assessment | Fumonisins and A. alternata f. sp. Lycopersici (AAL) toxins in ruminants and their forages | [19] |
| Biosynthesis | Genetic regulation of fumonisins biosynthesis by specific genes and global regulators | [179] |
| Biosynthesis | Impact of environmental variables and genetics of maize resistance on fumonisin accumulation | [180] |
| Detection | Analytical methods for fumonisins detection in single corn kernels | [181] |
| Detection | Molecular methods for early detection of fumonisin-producing F. verticillioides | [182] |
| Plant resistant | Genomic, genes, and pathways in maize resistance to Fusarium ear rot and fumonisin accumulation | [183] |
| Plant resistant | Relationship between Bt maize hybrids and fumonisins contamination level | [184] |
| Toxicity | Mitochondrial toxicity induced by FB1 | [185] |
| Toxicity | Molecular mechanisms underlying FB1-mediated toxicities and related interventions | [186] |
| Toxicity | CerS inhibition by fumonisins result in animal and plant disease | [187] |
| Toxicity | Dietary fumonisin and growth impairment in children and animals | [188] |
| Toxicity | Impact of fumonisin-contaminated feed on pig intestinal health | [189] |
| Toxicity | Oxidative stress-mediated toxicity and metabolism in vivo and in vitro | [190] |
| Fungi | Stains | fum Cluster | Reference |
|---|---|---|---|
| Asp. niger | fumonisin-producing strains | fum cluster | [208] |
| fumonisin-non-producing strains | Intact fum cluster | [7,235] | |
| Asp. welwitschiae | fumonisin-producing strains | fum cluster | [7] |
| fumonisin-non-producing strains | Three fum cluster types including an intact cluster | [7,235] | |
| Asp. tubingensis | fumonisin-producing strains | Not tested | [227] |
| fumonisin-non-producing strains | Multiple patterns of fum gene deletion | [7] | |
| Asp. brasiliensis | fumonisin-non-producing strains | Multiple patterns of fum gene deletion | [7] |
| Asp. luchuensis | fumonisin-non-producing strains | Multiple patterns of fum gene deletion | [7,236] |
| Asp. fumigatus | fumonisin-non-producing strains | Not detected | [208] |
| Asp. oryzae | fumonisin-non-producing strains | Not detected | [208] |
| Asp. nidulans | fumonisin-non-producing strains | Not detected | [208] |
| Asp. foetidus | fumonisin-producing strains | Not tested | [227] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, J.; Li, Z.; Cheng, Y.; Gao, C.; Guo, L.; Wang, T.; Xu, J. Sphinganine-Analog Mycotoxins (SAMs): Chemical Structures, Bioactivities, and Genetic Controls. J. Fungi 2020, 6, 312. https://doi.org/10.3390/jof6040312
Chen J, Li Z, Cheng Y, Gao C, Guo L, Wang T, Xu J. Sphinganine-Analog Mycotoxins (SAMs): Chemical Structures, Bioactivities, and Genetic Controls. Journal of Fungi. 2020; 6(4):312. https://doi.org/10.3390/jof6040312
Chicago/Turabian StyleChen, Jia, Zhimin Li, Yi Cheng, Chunsheng Gao, Litao Guo, Tuhong Wang, and Jianping Xu. 2020. "Sphinganine-Analog Mycotoxins (SAMs): Chemical Structures, Bioactivities, and Genetic Controls" Journal of Fungi 6, no. 4: 312. https://doi.org/10.3390/jof6040312
APA StyleChen, J., Li, Z., Cheng, Y., Gao, C., Guo, L., Wang, T., & Xu, J. (2020). Sphinganine-Analog Mycotoxins (SAMs): Chemical Structures, Bioactivities, and Genetic Controls. Journal of Fungi, 6(4), 312. https://doi.org/10.3390/jof6040312







