A One Health Approach to Combatting Sporothrix brasiliensis: Narrative Review of an Emerging Zoonotic Fungal Pathogen in South America
Abstract
1. Introduction
2. Materials and Methods
3. Causal Agent
4. Geographic Distribution
5. Animal Hosts, Reservoirs, and Sources of Human S. brasiliensis Infection
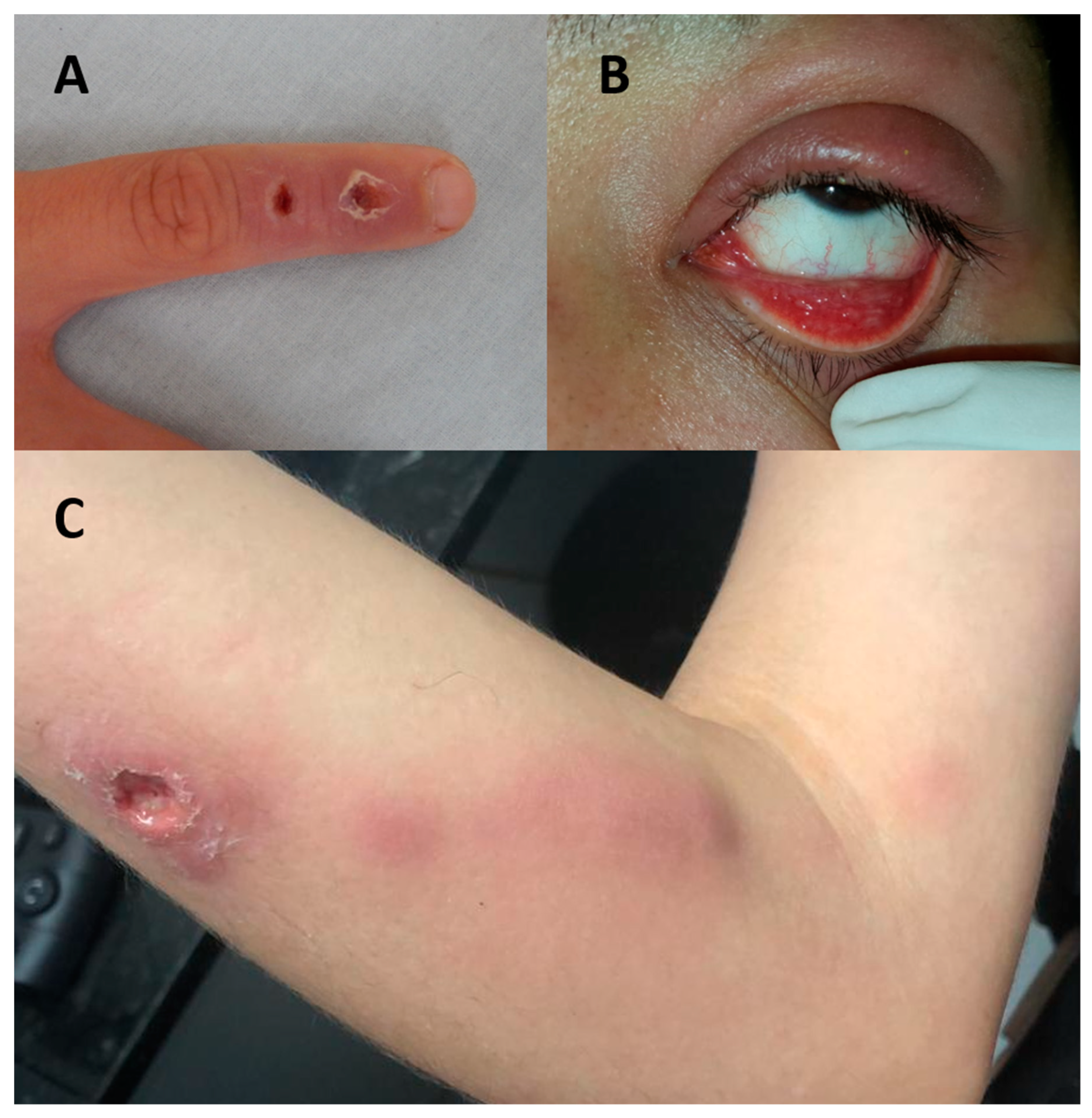
6. Human Sporotrichosis
6.1. Transmission
6.2. Clinical Presentation
6.3. Treatment
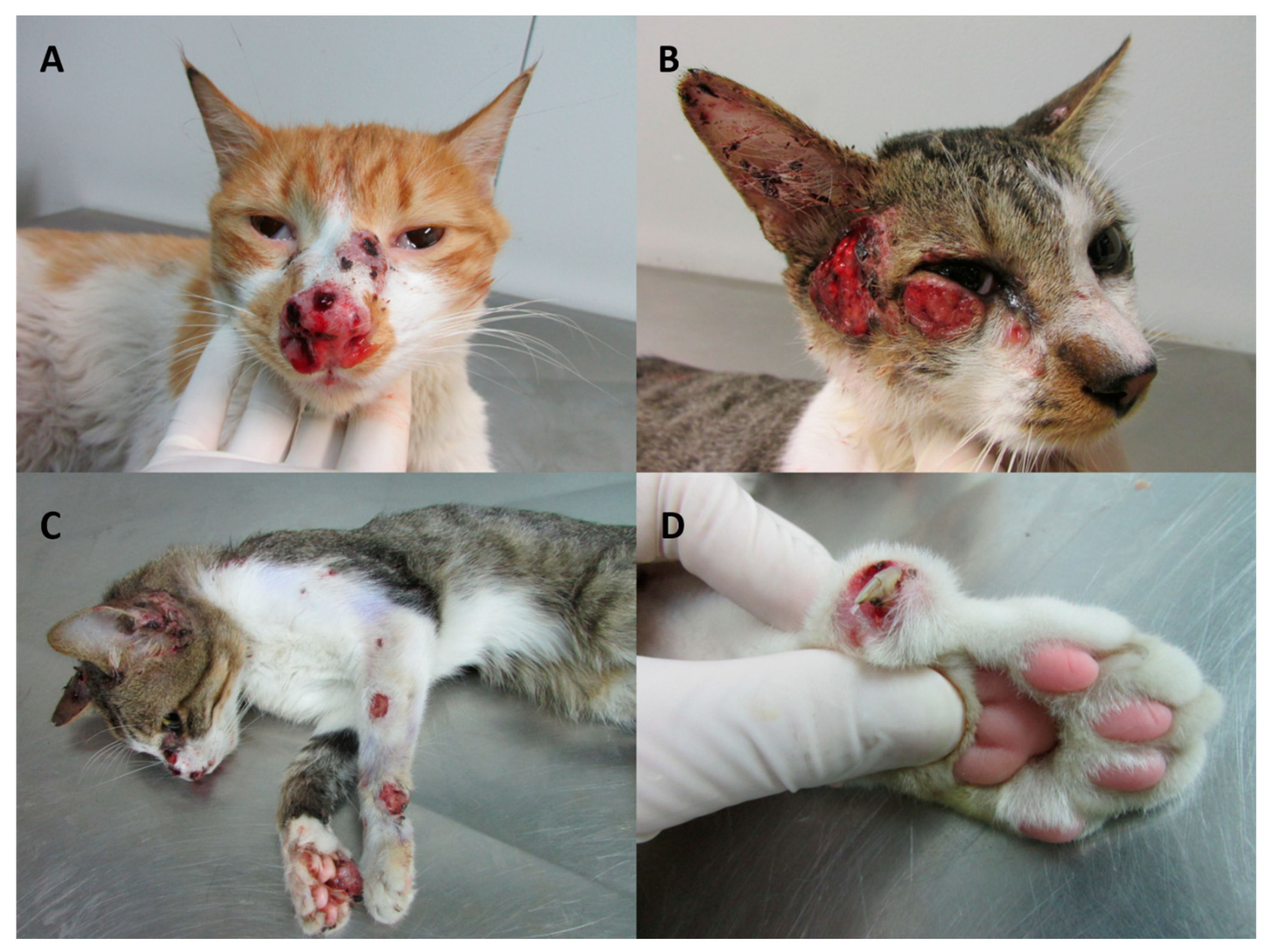
7. Feline Sporotrichosis
7.1. Feline Epidemiology
7.2. Clinical Presentation
7.3. Treatment
8. Diagnosis of Human and Feline Sporotrichosis
9. Prevention and Control
9.1. Preventing Transmission Among Cat Populations
9.2. Preventing Cat-to-Human Transmission
10. A One Health Approach to Cat-Transmitted Sporotrichosis Outbreak
11. Conclusions
Author Contributions
Funding
Conflicts of Interest
Disclaimer
Appendix A
| Database | Strategy | Run Date | Records |
|---|---|---|---|
| Medline (Ovid) 1946- | (sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR s brasiliensis OR rose thorn disease * OR rose gardener disease OR rose gardeners disease * OR rose handler disease * OR rose handlers disease * OR esporotricos *).mp. OR exp Sporothrix/ OR exp Sporotrichosis/ AND (brasiliensis OR brasilienses).mp. Limits: English, Spanish, Portuguese, 2008- | 4/18/19 | 179 articles |
| Embase (Ovid) 1947- | ( (sporothrix* OR sporothrices OR sporotrichum* OR sporotrichos* OR s brasiliensis OR rose thorn disease* OR rose gardener disease OR rose gardeners disease* OR rose handler disease* OR rose handlers disease* OR esporotricos*).mp. OR exp Sporothrix/ OR exp sporotrichosis/ AND (brasiliensis OR brasilienses).mp. ) OR exp Sporothrix brasiliensis/ Limits: English, Spanish, Portuguese, 2008- | 4/18/19 | 270 articles − 164 duplicates = 106 articles |
| CAB Abstracts (Ovid) 1910- | (sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR s brasiliensis OR rose thorn disease * OR rose gardener disease OR rose gardeners disease * OR rose handler disease * OR rose handlers disease * OR esporotricos *).mp. OR exp Sporothrix/ OR exp sporotrichosis/ AND (brasiliensis OR brasilienses).mp. Limits: English, Spanish, Portuguese, 2008- | 4/18/19 | 209 articles − 141 duplicates = 68 articles |
| Global Health (Ovid) 1910- | (sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR s brasiliensis OR rose thorn disease * OR rose gardener disease OR rose gardeners disease * OR rose handler disease * OR rose handlers disease * OR esporotricos *).mp. OR exp Sporothrix/ OR exp sporotrichosis/ AND (brasiliensis OR brasilienses).mp. Limits: English, Spanish, Portuguese, 2008- | 4/18/19 | 148 articles − 148 duplicates = 0 articles |
| Cochrane Library | # 1(sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR “s brasiliensis” OR “rose thorn disease” OR “rose gardener disease” OR “rose gardeners disease” OR “rose handler disease” OR “rose handlers disease” OR esporotricos *):ti,ab,kwLimits 17 # 2MeSH descriptor: [Sporothrix] explode all treesMeSH 2 # 3MeSH descriptor: [Sporotrichosis] explode all trees MeSH 9 # 4(brasiliensis OR brasilienses):ti,ab,kwLimits 13 # 5 # 1 OR # 2 OR # 3 Limits 17 # 6 # 4 AND # 5 Limits 0 Limits: English, Spanish, Portuguese, 2008- | 4/18/19 | 0 articles − 0 duplicates = 0 articles |
| Scopus 1960- | TITLE-ABS-KEY (sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR “s brasiliensis” OR “rose thorn disease” OR “rose gardener disease” OR “rose gardeners disease” OR “rose handler disease” OR “rose handlers disease” OR esporotricos *) AND TITLE-ABS-KEY(brasiliensis OR brasilienses) AND (LIMIT-TO (PUBYEAR, 2019) OR LIMIT-TO (PUBYEAR, 2018) OR LIMIT-TO (PUBYEAR, 2017) OR LIMIT-TO (PUBYEAR, 2016 ) OR LIMIT-TO (PUBYEAR, 2015) OR LIMIT-TO (PUBYEAR, 2014) OR LIMIT-TO (PUBYEAR, 2013) OR LIMIT-TO PUBYEAR, 2012) OR LIMIT-TO (PUBYEAR, 2011) OR LIMIT-TO (PUBYEAR, 2010) OR LIMIT-TO (PUBYEAR, 2009) OR LIMIT-TO (PUBYEAR, 2008)) AND (LIMIT-TO (LANGUAGE, “English”) OR LIMIT-TO ( LANGUAGE, “Spanish”) OR LIMIT-TO (LANGUAGE, “Portuguese”)) | 4/18/19 | 317 articles − 268 duplicates = 49 articles |
| Academic Search Complete (Ebsco) | TI,AB(sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR “s brasiliensis” OR “rose thorn disease” OR “rose gardener disease” OR “rose gardeners disease” OR “rose handler disease” OR “rose handlers disease” OR esporotricos *) OR DE “SPOROTRICHOSIS” OR DE “SPOROTRICHUM” AND (brasiliensis OR brasilienses) Limits: English, Spanish, Portuguese, 2008-, peer-reviewed | 4/18/19 | 120 articles − 110 duplicates = 10 articles |
| CINAHL (Ebsco) | TI,AB (sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR “s brasiliensis” OR “rose thorn disease” OR “rose gardener disease” OR “rose gardeners disease” OR “rose handler disease” OR “rose handlers disease” OR esporotricos *) OR (MH “Sporotrichosis”) AND (brasiliensis OR brasilienses) Limits: English, Spanish, Portuguese, 2008-, peer-reviewed | 4/18/19 | 9 articles − 9 duplicates = 0 articles |
| GreenFILE (Ebsco) | TI,AB(sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR “s brasiliensis” OR”rose thorn disease” OR “rose gardener disease” OR “rose gardeners disease” OR “rose handler disease” OR “rose handlers disease” OR esporotricos *) AND (brasiliensis OR brasilienses) Limits: English, Spanish, Portuguese, 2008-, peer-reviewed | 4/18/19 | 3 articles − 3 duplicates = 0 articles |
| Agricultural & Environmental Science Collection (Proquest) 1960- | TI,AB(sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR “s brasiliensis” OR “rose thorn disease” OR “rose gardener disease” OR “rose gardeners disease” OR “rose handler disease” OR “rose handlers disease” OR esporotricos*) OR MAINSUBJECT.EXACT (“Sporotrichosis”) AND (brasiliensis OR brasilienses) Limits: English, Spanish, Portuguese, 2008-, peer-reviewed | 4/19/19 | 203 articles − 160 duplicates = 43 articles |
| ProQuest Central (Proquest) 1960- | TI,AB(sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR “s brasiliensis” OR “rose thorn disease” OR “rose gardener disease” OR “rose gardeners disease” OR “rose handler disease” OR “rose handlers disease” OR esporotricos *) OR MESH.EXACT(“Sporotrichosis”) OR MESH.EXACT(“Sporothrix”) AND (brasiliensis OR brasilienses) Limits: English, Spanish, Portuguese, 2008-, peer-reviewed | 4/19/19 | 165 articles − 130 duplicates = 35 articles |
| Virtual Health Library (WHO) | (tw:(sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR “s brasiliensis” OR “rose thorn disease” OR “rose gardener disease” OR “rose gardeners disease” OR “rose handler disease” OR “rose handlers disease” OR esporotricos *)) AND (tw:(brasiliensis OR brasilienses)) Limits: English, Spanish, Portuguese, 2008- | 4/19/19 | 237 articles − 208 duplicates = 28 articles |
| CDC Stacks | Title,Subject,Description(sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR “s brasiliensis” OR “rose thorn disease” OR “rose gardener disease” OR “rose gardeners disease” OR “rose handler disease” OR “rose handlers disease” OR esporotricos*) AND (brasiliensis OR brasilienses) Limits: English, Spanish, Portuguese, 2008- | 4/19/19 | 0 articles |
| Homeland Security Digital Library | Title,Subject,Description(sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR “s brasiliensis” OR “rose thorn disease” OR “rose gardener disease” OR “rose gardeners disease” OR “rose handler disease” OR “rose handlers disease” OR esporotricos*) AND (brasiliensis OR brasilienses) Limits: English, Spanish, Portuguese, 2008- | 4/19/19 | 0 articles |
| PubMedCentral (NLM) | TI,AB(sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR “s brasiliensis” OR “rose thorn disease” OR “rose gardener disease” OR “rose gardeners disease” OR “rose handler disease” OR “rose handlers disease” OR esporotricos *) OR “sporothrix” [MeSH Terms] OR “sporotrichosis” [MeSH Terms] AND (brasiliensis OR brasilienses) Exclude: Medline Limits: English, Spanish, Portuguese, 2008- | 4/19/19 | 65 articles − 40 duplicates = 25 articles |
| InFocus Conference and Inform II Conference (ISHAM) | (sporothrix * OR sporothrices OR sporotrichum * OR sporotrichos * OR “s brasiliensis” OR “rose thorn disease” OR “rose gardener disease” OR “rose gardeners disease” OR “rose handler disease” OR “rose handlers disease” OR esporotricos *) AND (brasiliensis OR brasilienses) | 4/19/19 | 3 articles − 0 duplicates = 3 articles |
| Deduplicated in EndNote: | 546 articles | ||
Appendix B
Appendix B.1. Therapeutic Regimens and Other Recommendations for Feline Sporotrichosis
| Drug | Dose | Indications for Use |
|---|---|---|
| ITZ monotherapy (capsule) | Cats ≥ 3 kg: 100 mg/cat/day Cats ≥ 1 kg–3 kg: 50 mg/cat/day Cats < 1 kg: 25 mg/kg/day | -Cats with fixed cutaneous lesions and naïve to antifungal therapy |
| ITZ plus KI (capsules) | ITZ: same doses described above KI: 2.5–20 mg/kg/day— Half the dose of KI in the first week of treatment | -Cats presenting multiple skin lesions, mucosal lesions, or presence of respiratory signs -Cases refractory to ITZ monotherapy |
Appendix B.1.1. Criterion for Cure and Duration of Therapy
Appendix B.1.2. Use of Compounded Itraconazole and Potassium Iodide
Appendix B.1.3. Adverse Reactions and Contraindications
Appendix B.2. Other Therapeutic Options
Appendix B.2.1. Amphotericin B
Appendix B.2.2. Ketoconazole
Appendix B.2.3. Terbinafine
Appendix B.2.4. Fluconazole
Appendix B.2.5. Posaconazole
Appendix B.2.6. Other Alternative Therapies
References
- Rodrigues, A.M.; de Melo Teixeira, M.; de Hoog, G.S.; Schubach, T.M.; Pereira, S.A.; Fernandes, G.F.; Bezerra, L.M.; Felipe, M.S.; de Camargo, Z.P. Phylogenetic analysis reveals a high prevalence of Sporothrix brasiliensis in feline sporotrichosis outbreaks. PLoS Negl. Trop. Dis. 2013, 7, e2281. [Google Scholar] [CrossRef] [PubMed]
- de Beer, Z.W.; Duong, T.A.; Wingfield, M.J. The divorce of Sporothrix and Ophiostoma: Solution to a problematic relationship. Stud. Mycol. 2016, 83, 165–191. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.M.; Della Terra, P.P.; Gremiao, I.D.; Pereira, S.A.; Orofino-Costa, R.; de Camargo, Z.P. The threat of emerging and re-emerging pathogenic Sporothrix species. Mycopathologia 2020, 1–20. [Google Scholar] [CrossRef]
- Orofino-Costa, R.; de Macedo, P.M.; Bernardes-Engemann, A.R. Hyperendemia of Sporotrichosis in the Brazilian Southeast: Learning from Clinics and Therapeutics. Curr. Fungal Infect. Rep. 2015, 9, 220–228. [Google Scholar] [CrossRef]
- Barros, M.B.; Schubach, A.O.; Schubach, T.M.; Wanke, B.; Lambert-Passos, S.R. An epidemic of sporotrichosis in Rio de Janeiro, Brazil: Epidemiological aspects of a series of cases. Epidemiol. Infect. 2008, 136, 1192–1196. [Google Scholar] [CrossRef]
- Arrillaga-Moncrieff, I.; Capilla, J.; Mayayo, E.; Marimon, R.; Marine, M.; Gene, J.; Cano, J.; Guarro, J. Different virulence levels of the species of Sporothrix in a murine model. Clin. Microbiol. Infect. 2009, 15, 651–655. [Google Scholar] [CrossRef]
- Queiroz-Telles, F.; Buccheri, R.; Benard, G. Sporotrichosis in Immunocompromised Hosts. J. Fungi 2019, 5, 8. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.M.; Hoog, G.S.; Camargo, Z.P. Sporothrix Species Causing Outbreaks in Animals and Humans Driven by Animal-Animal Transmission. PLoS Pathog. 2016, 12, e1005638. [Google Scholar] [CrossRef]
- Gremião, I.D.; Miranda, L.H.; Reis, E.G.; Rodrigues, A.M.; Pereira, S.A. Zoonotic Epidemic of Sporotrichosis: Cat to Human Transmission. PLoS Pathog. 2017, 13, e1006077. [Google Scholar] [CrossRef] [PubMed]
- Ramos-e-Silva, M.; Vasconcelos, C.; Carneiro, S.; Cestari, T. Sporotrichosis. Clin. Dermatol. 2007, 25, 181–187. [Google Scholar] [CrossRef]
- Almeida-Paes, R.; de Oliveira, M.M.; Freitas, D.F.; do Valle, A.C.; Zancope-Oliveira, R.M.; Gutierrez-Galhardo, M.C. Sporotrichosis in Rio de Janeiro, Brazil: Sporothrix brasiliensis is associated with atypical clinical presentations. PLoS Negl. Trop. Dis. 2014, 8, e3094. [Google Scholar] [CrossRef] [PubMed]
- Cordoba, S.; Isla, G.; Szusz, W.; Vivot, W.; Hevia, A.; Davel, G.; Canteros, C.E. Molecular identification and susceptibility profile of Sporothrix schenckii sensu lato isolated in Argentina. Mycoses 2018, 61, 441–448. [Google Scholar] [CrossRef] [PubMed]
- García Duarte, J.M.; Wattiez Acosta, V.R.; Fornerón Viera, P.M.L.; Aldama Caballero, A.; Gorostiaga Matiauda, G.A.; Rivelli de Oddone, V.B.; Pereira Brunell, J.G. Esporotricosis trasmitida por gato doméstico. Reporte de un caso familiar. Rev. Nac. 2017, 9, 67–76. [Google Scholar]
- de Lima Barros, M.B.; Schubach, T.P.; Coll, J.O.; Gremião, I.D.; Wanke, B.; Schubach, A. Esporotricose: A evolução e os desafios de uma epidemia. Rev. Panam. Salud Publica 2010, 27, 455–460. [Google Scholar]
- Gremião, I.D.F.; Oliveira, M.M.E.; Monteiro de Miranda, L.H.; Saraiva Freitas, D.F.; Pereira, S.A. Geographic Expansion of Sporotrichosis, Brazil. Emerg. Infect. Dis. 2020, 26, 621–624. [Google Scholar] [CrossRef] [PubMed]
- Rios, M.E.; Suarez, J.M.D.; Moreno, J.; Vallee, J.; Moreno, J.P. Zoonotic Sporotrichosis Related to Cat Contact: First Case Report from Panama in Central America. Cureus 2018, 10, 10. [Google Scholar] [CrossRef]
- Gremiao, I.D.; Menezes, R.C.; Schubach, T.M.; Figueiredo, A.B.; Cavalcanti, M.C.; Pereira, S.A. Feline sporotrichosis: Epidemiological and clinical aspects. Med. Mycol. 2015, 53, 15–21. [Google Scholar] [CrossRef]
- de Silva, M.B.T.D.; de Mattos Costa, M.M.; da Silva Torres, C.C.; Galhardo, M.C.G.; do Valle, A.C.F.; de Avelar F. M. Magalhães, M.; Sabroza, P.C.; de Oliveira, R.M. Esporotricose urbana: Epidemia negligenciada no Rio de Janeiro, Brasil. Cad. Saude Publica 2012, 28, 1867–1880. [Google Scholar] [CrossRef]
- Marimon, R.; Cano, J.; Gene, J.; Sutton, D.A.; Kawasaki, M.; Guarro, J. Sporothrix brasiliensis, S. globosa, and S. mexicana, Three New Sporothrix Species of Clinical Interest. J. Clin. Microbiol. 2007, 45, 3198–3206. [Google Scholar] [CrossRef]
- Lutz, A.; Splendore, A. On a mycosis observed in men and mice: Contribution to the knowledge of the so-called sporotrichosis. Rev. Inst. Med. Trop. Sao Paolo 1907, 21, 443–450. [Google Scholar]
- Castro, R.A.; Kubitschek-Barreira, P.H.; Teixeira, P.A.; Sanches, G.F.; Teixeira, M.M.; Quintella, L.P.; Almeida, S.R.; Costa, R.O.; Camargo, Z.P.; Felipe, M.S.; et al. Differences in cell morphometry, cell wall topography and gp70 expression correlate with the virulence of Sporothrix brasiliensis clinical isolates. PLoS ONE 2013, 8, e75656. [Google Scholar] [CrossRef]
- Lopes-Bezerra, L.M.; Walker, L.A.; Nino-Vega, G.; Mora-Montes, H.M.; Neves, G.W.P.; Villalobos-Duno, H.; Barreto, L.; Garcia, K.; Franco, B.; Martinez-Alvarez, J.A.; et al. Cell walls of the dimorphic fungal pathogens Sporothrix schenckii and Sporothrix brasiliensis exhibit bilaminate structures and sloughing of extensive and intact layers. PLoS Negl. Trop. Dis. 2018, 12, e0006169. [Google Scholar] [CrossRef]
- Rees, R.K.; Swartzberg, J.E. Feline-transmitted sporotrichosis: A case study from California. Dermatol. Online J. 2011, 17, 2. [Google Scholar] [PubMed]
- Bove-Sevilla, P.M.; Mayorga-Rodríguez, J.; Hernández-Hernández, O. Esporotricosis transmitida por gato doméstico. Reporte de un caso. Med. Cután. Ibero. Lat. Am. 2008, 36, 33–35. [Google Scholar]
- Reed, K.D.; Moore, F.M.; Geiger, G.E.; Stemper, M.E. Zoonotic Transmission of Sporotrichosis: Case Report and Review. Clin. Infect. Dis. 1993, 16, 384–387. [Google Scholar] [CrossRef] [PubMed]
- Singer, J.I.; Muncie, J.E. Sporotrichosis; etiologic considerations and report of additional cases from New York. N. Y. State J. Med. 1952, 52, 52. [Google Scholar]
- Tang, M.M.; Tang, J.J.; Gill, P.; Chang, C.C.; Baba, R. Cutaneous sporotrichosis: A six-year review of 19 cases in a tertiary referral center in Malaysia. Int. J. Dermatol. 2012, 51, 702–708. [Google Scholar] [CrossRef]
- Zamri-Saad, M.; Salmiyah, T.S.; Jasni, S.; Cheng, B.Y.; Basri, K. Feline sporotrichosis: An increasingly important zoonotic disease in Malaysia. Veter. Rec. 1990, 127, 480. [Google Scholar]
- Yegneswaran, P.P.; Sripathi, H.; Bairy, I.; Lonikar, V.; Rao, R.; Prabhu, S. Zoonotic sporotrichosis of lymphocutaneous type in a man acquired from a domesticated feline source: Report of a first case in southern Karnataka, India. Int. J. Dermatol. 2009, 48, 1198–1200. [Google Scholar] [CrossRef]
- Zhang, Y.; Hagen, F.; Stielow, B.; Rodrigues, A.M.; Samerpitak, K.; Zhou, X.; Feng, P.; Yang, L.; Chen, M.; Deng, S.; et al. Phylogeography and evolutionary patterns in Sporothrix spanning more than 14 000 human and animal case reports. Persoonia 2015, 35, 1–20. [Google Scholar] [CrossRef]
- Schubach, A.; Barros, M.B.; Wanke, B. Epidemic sporotrichosis. Curr. Opin. Infect. Dis. 2008, 21, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Schubach, A.; Schubach, T.M.P.; de Lima Barros, M.B.; Wanke, B. Cat-transmitted Sporotrichosis, Rio de Janeiro, Brazil. Emerg. Infect. Dis. 2005, 11, 1952–1954. [Google Scholar] [CrossRef] [PubMed]
- Viana, P.G.; Gremiao, I.D.F.; Figueiredo, A.B.F.; Antonio, I.M.S.; Miranda, L.H.M.; Figueiredo, F.B.; Suarez-Mutis, M.; Pereira, S.A. Clinical and epidemiological aspects of the largest epidemic of sporotrichosis in dogs: 203 cases (2004–2014). Mycoses 2015, 4, 145. [Google Scholar] [CrossRef][Green Version]
- Schubach, T.M.P.; Schubach, A.; Okamoto, T.; Barros, M.B.L.; Figueiredo, F.B.; Cuzzi, T.; Pereira, S.A.; Dos Santos, I.B.; De Almeida Paes, R.; Rodrigo De Paes Leme, L.; et al. Canine sporotrichosis in Rio de Janeiro, Brazil: Clinical presentation, laboratory diagnosis and therapeutic response in 44 cases (1998–2003). Med. Mycol. 2006, 44, 87–92. [Google Scholar] [CrossRef]
- Montenegro, H.; Rodrigues, A.M.; Dias, M.A.; da Silva, E.A.; Bernardi, F.; de Camargo, Z.P. Feline sporotrichosis due to Sporothrix brasiliensis: An emerging animal infection in Sao Paulo, Brazil. BMC Vet. Res. 2014, 10, 269. [Google Scholar] [CrossRef]
- Fernández, N.; Iachini, R.; Farias, L.; Pozzi, N.; Tiraboschi, I. Esporotricosis; una zoonosis en alerta. In Proceedings of the InFocus Cordoba, Circulo Medico de Cordoba, Cordoba, Argentina, November 2015; p. 11. [Google Scholar]
- Etchecopaz, A.; Scarpa, M.; Mas, J.; Cuestas, M.L. Sporothrix brasiliensis: A growing hazard in the Northern area of Buenos Aires Province? Rev. Argent. Microbiol. 2020. [Google Scholar] [CrossRef]
- Kaadan, M.I.; Dennis, M.; Desai, N.; Yadavalli, G.; Lederer, P. One Health Education for Future Physicians: A Case Report of Cat-Transmitted Sporotrichosis. Open Forum Infect. Dis. 2020, 7, ofaa049. [Google Scholar] [CrossRef]
- Smith, L.M. Sporotrichosis: Report of four clinically atypical cases. South. Med. J. 1945, 38, 505–509. [Google Scholar] [CrossRef]
- Jin, X.-Z.; Zhang, H.-D.; Hiruma, M.; Yamamoto, I. Mother-and-Child Cases of Infection. Mycoses 1990, 33, 33–36. [Google Scholar] [CrossRef]
- de Lima Barros, M.B.; de Oliveira Schubach, A.; do Valle, A.C.F.; Galhardo, M.C.G.; Conceição-Silva, F.; Schubach, T.M.P.; Reis, R.S.; Wanke, B.; Marzochi, K.B.F.; Conceição, M.J. Cat-Transmitted Sporotrichosis Epidemic in Rio de Janeiro, Brazil: Description of a Series of Cases. Clin. Infect. Dis. 2004, 38, 529–535. [Google Scholar] [CrossRef]
- de Miranda, L.H.M.; Silva, J.N.; Gremião, I.D.F.; Menezes, R.C.; Almeida-Paes, R.; dos Reis, É.G.; de Oliveira, R.D.V.C.; de Araujo, D.S.D.A.; Ferreiro, L.; Pereira, S.A. Monitoring Fungal Burden and Viability of Sporothrix spp. in Skin Lesions of Cats for Predicting Antifungal Treatment Response. J. Fungi 2018, 4, 92. [Google Scholar] [CrossRef] [PubMed]
- Saravanakumar, P.S.; Eslami, P.; Zar, F.A. Lymphocutaneous Sporotrichosis Associated with a Squirrel Bite: Case Report and Review. Clin. Infect. Dis. 1996, 23, 647–648. [Google Scholar] [CrossRef]
- Moaven, L.D.; Altman, S.A.; Newnham, A.R. Sporotrichosis mimicking necrotising arachnidism. Med. J. Aust. 1999, 171, 685–686. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.D.; Keeling, J.H. Ant sting sporotrichosis. Cutis 2002, 69, 439–442. [Google Scholar] [PubMed]
- Alves, S.H.; Boettcher, C.S.; de Oliveira, D.C.; Tronco-Alves, G.R.; Sgaria, M.A.; Thadeu, P.; Oliveira, L.T.; Santurio, J.M. Sporothrix schenckii associated with armadillo hunting in Southern Brazil: Epidemiological and antifungal susceptibility profiles. Rev. Soc. Bras. Med. Trop. 2010, 43, 523–525. [Google Scholar] [CrossRef]
- Mayorga, R.; Cáceres, A.; Toriello, C.; Gutiérrez, G.; Alvarez, O.; Ramirez, M.E.; Mariat, F. An endemic area of sporotrichosis in Guatemala. Sabouraudia 1978, 16, 185–198. [Google Scholar] [CrossRef]
- Fichman, V.; Gremião, I.D.F.; Mendes-Júnior, A.; Sampaio, F.M.S.; Freitas, D.F.S.; Oliveira, M.M.E.; Almeida-Paes, R.; Valle, A.C.F.; Gutierrez-Galhardo, M.C. Sporotrichosis transmitted by a cockatiel (Nymphicus hollandicus). J. Eur. Acad. Dermatol. Venereol. 2018, 32, e157–e158. [Google Scholar] [CrossRef]
- de Beurmann, L.; Gougerot, R.W. Les sporotrichoses. Librarie Fe’lix 1912, 712–729. [Google Scholar]
- Madrid, I.M.; Mattei, A.S.; Fernandes, C.G.; Nobre Mde, O.; Meireles, M.C. Epidemiological findings and laboratory evaluation of sporotrichosis: A description of 103 cases in cats and dogs in southern Brazil. Mycopathologia 2012, 173, 265–273. [Google Scholar] [CrossRef]
- Ramirez-Soto, M.C.; Aguilar-Ancori, E.G.; Tirado-Sanchez, A.; Bonifaz, A. Ecological Determinants of Sporotrichosis Etiological Agents. J. Fungi 2018, 4, 95. [Google Scholar] [CrossRef]
- de Lima Barros, M.B.; de Almeida Paes, R.; Schubach, A.O. Sporothrix schenckii and Sporotrichosis. Clin. Microbiol. Rev. 2011, 24, 633–654. [Google Scholar] [CrossRef]
- Téllez, M.D.; Batista-Duharte, A.; Portuondo, D.; Quinello, C.; Bonne-Hernández, R.; Carlos, I.Z. Sporothrix schenckii complex biology: Environment and fungal pathogenicity. Microbiology 2014, 160, 2352–2365. [Google Scholar] [CrossRef] [PubMed]
- Aung, A.K.; Teh, B.M.; McGrath, C.; Thompson, P.J. Pulmonary sporotrichosis: Case series and systematic analysis of literature on clinico-radiological patterns and management outcomes. Med. Mycol. 2013, 51, 534–544. [Google Scholar] [CrossRef]
- Freitas, D.F.; Lima, I.A.; Curi, C.L.; Jordao, L.; Zancope-Oliveira, R.M.; Valle, A.C.; Galhardo, M.C.; Curi, A.L. Acute dacryocystitis: Another clinical manifestation of sporotrichosis. Mem. Inst. Oswaldo Cruz 2014, 109, 262–264. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Barros, M.; Pereira, S.; Schubach, A.; Wanke, B. Esporotricose. In Dinâmica das Doenças Infecciosas e Parasitárias; Coura, J., Ed.; Guanabara Koogan: Rio de Janeiro, Brazil, 2013; pp. 1196–1206. [Google Scholar]
- Mercurio, M.G.; Elewski, B.E. Therapy of sporotrichosis. Semin. Dermatol. 1993, 12, 285–289. [Google Scholar] [PubMed]
- Bonifaz, A. Sporotrichosis: An update. Mycoses 2012, 55, 27–28. [Google Scholar] [CrossRef]
- Bonifaz, A.; Vazquez-Gonzalez, D. Diagnosis and treatment of lymphocutaneous sporotrichosis: What are the options? Curr. Fungal Infect. Rep. 2013, 7, 252–259. [Google Scholar] [CrossRef]
- Silva-Vergara, M.L.; de Camargo, Z.P.; Silva, P.F.; Abdalla, M.R.; Sgarbieri, R.N.; Rodrigues, A.M.; dos Santos, K.C.; Barata, C.H.; Ferreira-Paim, K. Disseminated Sporothrix brasiliensis infection with endocardial and ocular involvement in an HIV-infected patient. Am. J. Trop. Med. Hyg. 2012, 86, 477–480. [Google Scholar] [CrossRef]
- Pappas, P.G.; Tellez, I.; Deep, A.E.; Nolasco, D.; Holgado, W.; Bustamante, B. Sporotrichosis in Peru: Description of an Area of Hyperendemicity. Clin. Infect. Dis. 2000, 30, 65–70. [Google Scholar] [CrossRef]
- Freitas, D.F.S.; de Siqueira Hoagland, B.; do Valle, A.C.F.; Fraga, B.B.; de Barros, M.B.; de Oliveira Schubach, A.; de Almeida-Paes, R.; Cuzzi, T.; Rosalino, C.M.V.; Zancopé-Oliveira, R.M.; et al. Sporotrichosis in HIV-infected patients: Report of 21 cases of endemic sporotrichosis in Rio de Janeiro, Brazil. Med. Mycol. 2012, 50, 170–178. [Google Scholar] [CrossRef]
- Song, Y.; Li, S.-S.; Zhong, S.-X.; Liu, Y.-Y.; Yao, L.; Huo, S.-S. Report of 457 sporotrichosis cases from Jilin province, northeast China, a serious endemic region. J. Eur. Acad. Dermatol. Venereol. 2013, 27, 313–318. [Google Scholar] [CrossRef]
- Papaiordanou, F.; da Silveira, B.R.; Abulafia, L.A. Hypersensitivity reaction to Sporothrix schenckii: Erythema nodosum associated with sporotrichosis. Rev. Soc. Bras. Med. Trop. 2015, 48, 504. [Google Scholar] [CrossRef] [PubMed]
- Arinelli, A.; do Couto Aleixo, A.L.; Freitas, D.F.S.; do Valle, A.C.F.D; Almeida-Paes, R.; Gutierrez-Galhardo, M.C.; Curi, A.L.L. Ocular Sporotrichosis: 26 Cases with Bulbar Involvement in a Hyperendemic Area of Zoonotic Transmission. Ocul. Immunol. Inflamm. 2019, 1–8. [Google Scholar] [CrossRef]
- de Lima Barros, M.B.; Schubach, A.O.; de Vasconcellos Carvalhaes de Oliveira, R.; Martins, E.B.; Teixeira, J.L.; Wanke, B. Treatment of Cutaneous Sporotrichosis With Itraconazole—Study of 645 Patients. Clin. Infect. Dis. 2011, 52, e200–e206. [Google Scholar] [CrossRef] [PubMed]
- Bernardes-Engemann, A.R.; de Lima Barros, M.; Zeitune, T.; Russi, D.C.; Orofino-Costa, R.; Lopes-Bezerra, L.M. Validation of a serodiagnostic test for sporotrichosis: A follow-up study of patients related to the Rio de Janeiro zoonotic outbreak. Med. Mycol. 2015, 53, 28–33. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; de Hoog, G.S.; de Cassia Pires, D.; Brihante, R.S.; Sidrim, J.J.; Gadelha, M.F.; Colombo, A.L.; de Camargo, Z.P. Genetic diversity and antifungal susceptibility profiles in causative agents of sporotrichosis. BMC Infect. Dis. 2014, 14, 219. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, C.A.; Bustamante, B.; Chapman, S.W.; Pappas, P.G. Clinical Practice Guidelines for the Management of Sporotrichosis: 2007 Update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2007, 45, 1255–1265. [Google Scholar] [CrossRef]
- Kauffman, C.A.; Hajjeh, R.; Chapman, S.W. Practice Guidelines for the Management of Patients with Sporotrichosis. Clin. Infect. Dis. 2000, 30, 684–687. [Google Scholar] [CrossRef]
- Cabezas, C.; Bustamante, B.; Holgado, W.; Begue, R.E. Treatment of cutaneous sporotrichosis with one daily dose of potassium iodide. Pediatr. Infect. Dis. J. 1996, 15, 352–354. [Google Scholar] [CrossRef]
- Chapman, S.W.; Pappas, P.; Kauffmann, C.; Smith, E.B.; Dietze, R.; Tiraboschi-Foss, N.; Restrepo, A.; Bustamante, A.B.; Opper, C.; Emady-Azar, S.; et al. Comparative evaluation of the efficacy and safety of two doses of terbinafine (500 and 1000 mg day(-1)) in the treatment of cutaneous or lymphocutaneous sporotrichosis. Mycoses 2004, 47, 62–68. [Google Scholar] [CrossRef]
- Itoh, M.; Okamoto, S.; Kariya, H. Survey of 200 Cases of Sporotrichosis. Dermatology 1986, 172, 209–213. [Google Scholar] [CrossRef]
- Costa, R.O.; Bernardes-Engemann, A.R.; Azulay-Abulafia, L.; Benvenuto, F.; de Lourdes Palermo Neves, M.; Lopes-Bezerra, L.M. Esporotricose na gestação: Relato de cinco casos numa epidemia zoonótica no Rio de Janeiro, Brasil. An. Bras. Dermatol 2011, 86, 995–998. [Google Scholar] [CrossRef] [PubMed]
- Fichman, V.; Valle, A.; de Macedo, P.M.; Freitas, D.F.S.; Oliveira, M.M.E.; Almeida-Paes, R.; Gutierrez-Galhardo, M.C. Cryosurgery for the treatment of cutaneous sporotrichosis in four pregnant women. PLoS Negl. Trop. Dis. 2018, 12, e0006434. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, C.P.; Galhardo, M.C.; Valle, A.C. Cryosurgery as adjuvant therapy in cutaneous sporotrichosis. Braz. J. Infect. Dis. 2011, 15, 181–183. [Google Scholar] [CrossRef][Green Version]
- Soeiro Sampaio, F.M.; Sguissardi de Oliveira, D.; Saraiva Freitas, D.F.; Francesconi do Valle, A.C. Electrosurgery as Adjuvant Therapy for Cutaneous Sporotrichosis. Dermatol. Surg. 2019, 17, 17. [Google Scholar] [CrossRef]
- Schubach, T.M.P.; Schubach, A.; Okamoto, T.; Barros, M.B.L.; Figueiredo, F.B.; Cuzzi, T.; Fialho-Monteiro, P.C.; Reis, R.S.; Perez, M.A.; Wanke, B. Evaluation of an epidemic of sporotrichosis in cats: 347 cases (1998–2001). J. Am. Veter. Med. Assoc. 2004, 224, 1623–1629. [Google Scholar] [CrossRef]
- de Souza, L.L.; da Silva Nascente, P.; Nobre, M.O.; Meinerz, A.R.M.; Meireles, M.C.A. Isolation of Sporothrix schenkii from the nails of healthy cats. Braz. J. Microbiol. 2006, 37, 372–374. [Google Scholar] [CrossRef]
- Sanchotene, K.O.; Madrid, I.M.; Klafke, G.B.; Bergamashi, M.; Della Terra, P.P.; Rodrigues, A.M.; de Camargo, Z.P.; Xavier, M.O. Sporothrix brasiliensis outbreaks and the rapid emergence of feline sporotrichosis. Mycoses 2015, 58, 652–658. [Google Scholar] [CrossRef]
- Boechat, J.S.; Oliveira, M.M.E.; Almeida-Paes, R.; Gremiao, I.D.F.; Machado, A.C.S.; Oliveira, R.V.C.; Figueiredo, A.B.F.; Rabello, V.B.S.; Silva, K.B.L.; Zancope-Oliveira, R.M.; et al. Feline sporotrichosis: Associations between clinical-epidemiological profiles and phenotypic-genotypic characteristics of the etiological agents in the Rio de Janeiro epizootic area. Mem. Inst. Oswaldo Cruz 2018, 113, 185–196. [Google Scholar] [CrossRef]
- Macedo-Sales, P.A.; Souto, S.; Destefani, C.A.; Lucena, R.P.; Machado, R.L.D.; Pinto, M.R.; Rodrigues, A.M.; Lopes-Bezerra, L.M.; Rocha, E.M.S.; Baptista, A.R.S. Domestic feline contribution in the transmission of Sporothrix in Rio de Janeiro State, Brazil: A comparison between infected and non-infected populations. BMC Vet. Res. 2018, 14, 19. [Google Scholar] [CrossRef]
- de Souza, E.W.; Borba, C.M.; Pereira, S.A.; Gremiao, I.D.F.; Langohr, I.M.; Oliveira, M.M.E.; de Oliveira, R.V.C.; da Cunha, C.R.; Zancope-Oliveira, R.M.; de Miranda, L.H.M.; et al. Clinical features, fungal load, coinfections, histological skin changes, and itraconazole treatment response of cats with sporotrichosis caused by Sporothrix brasiliensis. Sci. Rep. 2018, 8, 9074. [Google Scholar] [CrossRef]
- Pereira, S.A.; Gremiao, I.D.; Kitada, A.A.; Boechat, J.S.; Viana, P.G.; Schubach, T.M. The epidemiological scenario of feline sporotrichosis in Rio de Janeiro, State of Rio de Janeiro, Brazil. Rev. Soc. Bras. Med. Trop. 2014, 47, 392–393. [Google Scholar] [CrossRef]
- Pereira, S.A.; Passos, S.R.L.; Silva, J.N.; Gremião, I.D.F.; Figueiredo, F.B.; Teixeira, J.L.; Monteiro, P.C.F.; Schubach, T.M.P. Response to azolic antifungal agents for treating feline sporotrichosis. Veter. Rec. 2010, 166, 290–294. [Google Scholar] [CrossRef] [PubMed]
- Schubach, T.M.P.; Schubach, A.; Okamoto, T.; Pellon, I.V.; Fialho-Monteiro, P.C.; Reis, R.S.; Barros, M.B.L.; Andrade-Perez, M.; Wanke, B. Haematogenous spread of Sporothrix schenckii in cats with naturally acquired sporotrichosis. J. Small Anim. Pract. 2003, 44, 395–398. [Google Scholar] [CrossRef] [PubMed]
- de Miranda, L.H.M.; Meli, M.; Conceicao-Silva, F.; Novacco, M.; Menezes, R.C.; Pereira, S.A.; Sugiarto, S.; Dos Reis, E.G.; Gremiao, I.D.F.; Hofmann-Lehmann, R. Co-infection with feline retrovirus is related to changes in immunological parameters of cats with sporotrichosis. PLoS ONE 2018, 13, e0207644. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, J.; Huang, H.; Xue, R.; Hu, X.; Li, M.; Zhong, Y.; Yuan, L. Taenia taeniaeformis in Rat Favors Protracted Skin Lesions Caused by Sporothrix schenckii Infection: Dectin-1 and IL-17 Are Dispensable for Clearance of This Fungus. PLoS ONE 2012, 7, e52514. [Google Scholar] [CrossRef] [PubMed]
- Pereira, S.; Gremião, I.; Menezes, R. Sporotrichosis in Animals: Zoonotic Transmission. In Sporotrichosis: New Developments and Future Prospects; Carlos, I.Z., Ed.; Springer International Publishing: Manhattan, NY, USA, 2015; pp. 83–102. ISBN 978-3-319-11912-0. [Google Scholar]
- Werner, A.H.; Werner, B.E. Sporotrichosis in Man and Animal. Int. J. Dermatol. 1994, 33, 692–700. [Google Scholar] [CrossRef]
- Rosser, E.; Dunstan, R. Sporotrichosis. In Infectious Diseases of the Dog and Cat; Green, C., Ed.; Saunders Elsevier: Philadelphia, PA, USA, 2006; pp. 608–612. [Google Scholar]
- Reis, É.G.; Gremião, I.D.F.; Kitada, A.A.B.; Rocha, R.F.D.B.; Castro, V.S.P.; Barros, M.B.L.; Menezes, R.C.; Pereira, S.A.; Schubach, T.M.P. Potassium iodide capsule treatment of feline sporotrichosis. J. Feline Med. Surg. 2012, 14, 399–404. [Google Scholar] [CrossRef]
- Crothers, S.L.; White, S.D.; Ihrke, P.J.; Affolter, V.K. Sporotrichosis: A retrospective evaluation of 23 cases seen in northern California (1987–2007). Veter. Dermatol. 2009, 20, 249–259. [Google Scholar] [CrossRef]
- Madrid, I.M.; Mattei, A.; Martins, A.; Nobre, M.; Meireles, M. Feline Sporotrichosis in the Southern Region of Rio Grande Do Sul, Brazil: Clinical, Zoonotic and Therapeutic Aspects. Zoonoses Public Health 2010, 57, 151–154. [Google Scholar] [CrossRef]
- Rossi, C.N.; Odaguiri, J.; Larsson, C.E. Retrospective Assessment of the Treatment of Sporotrichosis in Cats and Dogs Using Itraconazole. Acta Sci. Vet. 2013, 41, 1–5. [Google Scholar]
- da Rocha, R.F.D.B.; Schubach, T.M.P.; Pereira, S.A.; dos Reis, E.G.; Carvalho, B.W.; Gremiao, I.D.F. Refractory feline sporotrichosis treated with itraconazole combined with potassium iodide. J. Small Anim. Pract. 2018, 59, 720–721. [Google Scholar] [CrossRef]
- Reis, E.G.; Schubach, T.M.; Pereira, S.A.; Silva, J.N.; Carvalho, B.W.; Quintana, M.S.; Gremiao, I.D. Association of itraconazole and potassium iodide in the treatment of feline sporotrichosis: A prospective study. Med. Mycol. 2016, 54, 684–690. [Google Scholar] [CrossRef]
- Brilhante, R.S.; Rodrigues, A.M.; Sidrim, J.J.; Rocha, M.F.; Pereira, S.A.; Gremiao, I.D.; Schubach, T.M.; de Camargo, Z.P. In vitro susceptibility of antifungal drugs against Sporothrix brasiliensis recovered from cats with sporotrichosis in Brazil. Med. Mycol. 2016, 54, 275–279. [Google Scholar] [CrossRef]
- Almeida-Paes, R.; Brito-Santos, F.; Figueiredo-Carvalho, M.H.G.; Machado, A.C.S.; Oliveira, M.M.E.; Pereira, S.A.; Gutierrez-Galhardo, M.C.; Zancope-Oliveira, R.M. Minimal inhibitory concentration distributions and epidemiological cutoff values of five antifungal agents against Sporothrix brasiliensis. Mem. Inst. Oswaldo Cruz 2017, 112, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Espinel-Ingroff, A.; Abreu, D.P.B.; Almeida-Paes, R.; Brilhante, R.S.N.; Chakrabarti, A.; Chowdhary, A.; Hagen, F.; Cordoba, S.; Gonzalez, G.M.; Govender, N.P.; et al. Multicenter, International Study of MIC/MEC Distributions for Definition of Epidemiological Cutoff Values for Sporothrix Species Identified by Molecular Methods. Antimicrob. Agents Chemother. 2017, 61, e01057-17. [Google Scholar] [CrossRef] [PubMed]
- Chaves, A.R.; de Campos, M.P.; Barros, M.B.L.; do Carmo, C.N.; Gremião, I.D.F.; Pereira, S.A.; Schubach, T.M.P. Treatment abandonment in feline sporotrichosis—Study of 147 cases. Zoonoses Public Health 2013, 60, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Gremião, I.D.F.; Schubach, T.M.P.; Pereira, S.A.; Rodrigues, A.M.; Honse, C.O.; Barros, M.B.L. Treatment of refractory feline sporotrichosis with a combination of intralesional amphotericin B and oral itraconazole. Aust. Veter. J. 2011, 89, 346–351. [Google Scholar] [CrossRef]
- Chakrabarti, A.; Bonifaz, A.; Gutierrez-Galhardo, M.C.; Mochizuki, T.; Li, S. Global epidemiology of sporotrichosis. Med. Mycol. 2015, 53, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, C. Sporotrichosis. Infectious Disease. In Diagnosis and Treatment of Fungal Infections; Hospenthal, D., Rinaldi, M., Eds.; Springer: Cham, Switzerland, 2015; pp. 237–244. ISBN 978-3-319-13090-3. [Google Scholar]
- Rex, J.H.; Okhuysen, P.C.; Bennet, J.E.; Dolin, R.; Blaser, M.J. Sporothrix schenckii. In Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases; Elsevier Health Sciences: Philadelphia, PA, USA, 2014; Volume 1, pp. 2920–2924. [Google Scholar]
- Orofino-Costa, R.; Macedo, P.M.; Rodrigues, A.M.; Bernardes-Engemann, A.R. Sporotrichosis: An update on epidemiology, etiopathogenesis, laboratory and clinical therapeutics. An. Bras. Dermatol. 2017, 92, 606–620. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.M.; De Hoog, G.S.; De Camargo, Z.P. Feline sporotrichosis. In Emerging and Epizootic Fungal Infections in Animals; Springer International Publishing: Manhattan, NY, USA, 2018; pp. 199–231. ISBN 9783319720937. [Google Scholar]
- Arenas, R.; Sánchez-Cardenas, C.D.; Ramirez-Hobak, L.; Leon Felipe Ruíz, A.; Ma Elisa Vega, M. Sporotrichosis: From KOH to Molecular Biology. J. Fungi 2018, 4, 62. [Google Scholar] [CrossRef]
- Pereira, S.A.; Menezes, R.C.; Gremião, I.D.F.; Silva, J.N.; Honse, C.D.O.; Figueiredo, F.B.; da Silva, D.T.; Kitada, A.A.B.; dos Reis, É.G.; Schubach, T.M.P. Sensitivity of cytopathological examination in the diagnosis of feline sporotrichosis. J. Feline Med. Surg. 2011, 13, 220–223. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.N.; Miranda, L.H.M.; Menezes, R.C.; Gremiao, I.D.F.; Oliveira, R.V.C.; Vieira, S.M.M.; Conceicao-Silva, F.; Ferreiro, L.; Pereira, S.A. Comparison of the Sensitivity of Three Methods for the Early Diagnosis of Sporotrichosis in Cats. J. Comp. Pathol. 2018, 160, 72–78. [Google Scholar] [CrossRef]
- Jessica, N.; Sonia, R.L.; Rodrigo, C.; Isabella, D.F.; Tania, M.P.; Jeferson, C.; Anna, B.F.; Sandro, A. Diagnostic accuracy assessment of cytopathological examination of feline sporotrichosis. Med. Mycol. 2015, 53, 880–884. [Google Scholar] [CrossRef]
- Sporothrix Antibody Latex Agglutination (LA) Test System. Available online: http://www.immy.com/products/latex-agglutination/sporothrix-antibody-latex-agglutination-la-test-system/ (accessed on 30 July 2019).
- Oliveira, M.M.; Franco-Duarte, R.; Romeo, O.; Pais, C.; Criseo, G.; Sampaio, P.; Zancope-Oliveira, R.M. Evaluation of T3B fingerprinting for identification of clinical and environmental Sporothrix species. FEMS Microbiol. Lett. 2015, 362. [Google Scholar] [CrossRef]
- Oliveira, M.M.; Almeida-Paes, R.; Gutierrez-Galhardo, M.C.; Zancope-Oliveira, R.M. Molecular identification of the Sporothrix schenckii complex. Rev. Iberoam. Micol. 2014, 31, 2–6. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.M.; de Hoog, G.S.; de Camargo, Z.P. Molecular Diagnosis of Pathogenic Sporothrix Species. PLoS Negl. Trop. Dis. 2015, 9, e0004190. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.M.; Najafzadeh, M.J.; de Hoog, G.S.; de Camargo, Z.P. Rapid Identification of Emerging Human-Pathogenic Sporothrix Species with Rolling Circle Amplification. Front. Microbiol. 2015, 6, 1385. [Google Scholar] [CrossRef]
- Rodriguez-Brito, S.; Camacho, E.; Mendoza, M.; Nino-Vega, G.A. Differential identification of Sporothrix spp. and Leishmania spp. by conventional PCR and qPCR in multiplex format. Med. Mycol. 2015, 53, 22–27. [Google Scholar] [CrossRef]
- Zhang, M.; Li, F.; Gong, J.; Yang, X.; Zhang, J.; Zhao, F. Development and evaluation of a real-time polymerase chain reaction for fast diagnosis of sporotrichosis caused by Sporothrix globosa. Med. Mycol. 2019, 30, 30. [Google Scholar] [CrossRef]
- Zhang, M.; Li, F.; Li, R.; Gong, J.; Zhao, F. Fast diagnosis of sporotrichosis caused by Sporothrix globosa, Sporothrix schenckii, and Sporothrix brasiliensis based on multiplex real-time PCR. PLoS Negl. Trop. Dis. 2019, 13, e0007219. [Google Scholar] [CrossRef]
- Hu, S.; Chung, W.-H.; Hung, S.-I.; Ho, H.-C.; Wang, Z.-W.; Chen, C.-H.; Lu, S.-C.; Kuo, T.; Hong, H.-S. Detection of Sporothrix schenckii in Clinical Samples by a Nested PCR Assay. J. Clin. Microbiol. 2003, 41, 1414–1418. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.M.; de Hoog, G.S.; de Camargo, Z.P. Genotyping species of the Sporothrix schenckii complex by PCR-RFLP of calmodulin. Diagn. Microbiol. Infect. Dis. 2014, 78, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.M.; Santos, C.; Sampaio, P.; Romeo, O.; Almeida-Paes, R.; Pais, C.; Lima, N.; Zancope-Oliveira, R.M. Development and optimization of a new MALDI-TOF protocol for identification of the Sporothrix species complex. Res. Microbiol. 2015, 166, 102–110. [Google Scholar] [CrossRef] [PubMed]
- M38-A2. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi, 3rd ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2017; Available online: https://clsi.org/standards/products/microbiology/documents/m38/ (accessed on 30 July 2019).
- Gremião, I.D.; da Rocha, E.; Montenegro, H.; Carneiro, A.; Xavier, M.; de Farias, M.; Monti, F.; Mansho, W.; Pereira, R.; Pereira, S. Guideline for the management of feline sporotrichosis caused by Sporothrix brasiliensis and literature revision. Braz. J. Microbiol. 2020, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Etchecopaz, A.N.; Lanza, N.; Toscanini, M.A.; Devoto, T.B.; Pola, S.J.; Daneri, G.L.; Iovannitti, C.A.; Cuestas, M.L. Sporotrichosis caused by Sporothrix brasiliensis in Argentina: Case report, molecular identification and in vitro susceptibility pattern to antifungal drugs. J. Mycol. Med. 2020, 30, 100908. [Google Scholar] [CrossRef] [PubMed]
- da Silva, D.T.; Menezes, R.C.; Ferreira Gremião, I.D.; Pacheco Schubach, T.M.; Boechat, J.S.; Pereira, S.A. Esporotricose zoonótica: Procedimentos de biossegurança. Acta Sci. Vet. 2012, 40, 1–10. [Google Scholar]
- Centers for Disease Control and Prevention Cats: How to Stay Healthy around Pet Cats. Available online: https://www.cdc.gov/healthypets/pets/cats.html#tabs-1-3 (accessed on 1 July 2020).
- Lloret, A.; Hartmann, K.; Pennisi, M.G.; Ferrer, L.; Addie, D.; Belák, S.; Boucraut-Baralon, C.; Egberink, H.; Frymus, T.; Gruffydd-Jones, T.; et al. Sporotrichosis in cats: ABCD guidelines on prevention and management. J. Feline Med. Surg. 2013, 15, 619–623. [Google Scholar] [CrossRef]
- Greene, C. Antifungal Chemotherapy. In Infectious Diseases of the Dog and Cat; Greene, C., Ed.; Saunders Elsevier: Philadelphia, PA, USA, 2012; pp. 579–588. [Google Scholar]
- Renschler, J.; Albers, A.; Sinclair-Mackling, H.; Wheat, L.J. Comparison of Compounded, Generic, and Innovator-Formulated Itraconazole in Dogs and Cats. J. Am. Anim. Hosp. Assoc. 2018, 54, 195–200. [Google Scholar] [CrossRef]
- Mawby, D.I.; Whittemore, J.C.; Genger, S.; Papich, M.G. Bioequivalence of Orally Administered Generic, Compounded, and Innovator-Formulated Itraconazole in Healthy Dogs. J. Vet. Intern. Med. 2014, 28, 72–77. [Google Scholar] [CrossRef]
- Webster, C.R.L.; Cooper, J. Therapeutic Use of Cytoprotective Agents in Canine and Feline Hepatobiliary Disease. Vet. Clin. Small Anim. Pract. 2009, 39, 631–652. [Google Scholar] [CrossRef]
- Avizeh, R.; Najafzadeh, H.; Jalali, M.R.; Shirali, S. Evaluation of prophylactic and therapeutic effects of silymarin and N-acetylcysteine in acetaminophen-induced hepatotoxicity in cats. J. Vet. Pharmacol. Ther. 2010, 33, 95–99. [Google Scholar] [CrossRef]
- Gremiao, I.D.; Schubach, T.M.; Pereira, S.A.; Rodrigues, A.M.; Chaves, A.R.; Barros, M.B. Intralesional amphotericin B in a cat with refractory localised sporotrichosis. J. Feline Med. Surg. 2009, 11, 720–723. [Google Scholar] [CrossRef] [PubMed]
- Dunstan, R.W.; Langham, R.F.; Reimann, K.A.; Wakenell, P.S. Feline sporotrichosis: A report of five cases with transmission to humans. J. Am. Acad. Dermatol. 1986, 15, 37–45. [Google Scholar] [CrossRef]
- Burke, M.; Grauer, G.; Macy, D. Successful treatment of cutaneolymphatic sporotrichosis in cat with ketoconazole and sodium iodine. J. Am. Anim. Hosp. Assoc. 1983, 19, 542–547. [Google Scholar]
- Raimer, S.; Ewert, A.; MacDonald, E.M. Ketoconazole therapy of experimentally induced sporotrichosis infections in cats. A preliminary study. Curr. Ther. Res. 1983, 33, 670–680. [Google Scholar]
- Davies, C.; Troy, G. Deep mycotic infections in cats. J. Am. Anim. Hosp. Assoc. 1996, 32, 380–391. [Google Scholar] [CrossRef]
- de-Oliverira-Nobre, M.; Pötter-de-Castro, A.; Caetano, D.; de Souza, L.L.; Meireles, M.C.A.; Ferreiro, L. Recurrence of sporotrichosis in cats with zoonotic involvement. Recurrencia de esporotricosis en gatos con implicaciones zoonóticas 2001. [Google Scholar]
- Han, H.S.; Kano, R.; Chen, C.; Noli, C. Comparison of two in vitro antifungal sensitivity tests and monitoring during therapy of Sporothrix schenckii sensu stricto in Malaysian cats. Vet. Dermatol. 2017, 28, 156-e32. [Google Scholar] [CrossRef]
- Batista-Duharte, A.; Pereira, S.A.; Freitas, D.F.S.; Fuentes, D.P.; Gutierrez-Galhardo, M.C.; Carlos, I.Z. Therapeutic and Prophylactic Tools for Sporotrichosis: Current Strategies and Future Tendencies. In Sporotrichosis; Zeppone Carlos, I., Ed.; Springer: Cham, Switzerland, 2015; p. 147. [Google Scholar]
- Francesconi, G.; Francesconi do Valle, A.C.; Passos, S.L.; de Lima Barros, M.B.; de Almeida Paes, R.; Curi, A.L.; Liporage, J.; Porto, C.F.; Galhardo, M.C. Comparative study of 250 mg/day terbinafine and 100 mg/day itraconazole for the treatment of cutaneous sporotrichosis. Mycopathologia 2011, 171, 349–354. [Google Scholar] [CrossRef]
- Francesconi, G.; Valle, A.C.; Passos, S.; Reis, R.; Galhardo, M.C. Terbinafine (250 mg/day): An effective and safe treatment of cutaneous sporotrichosis. J. Eur. Acad. Dermatol. Venereol. 2009, 23, 1273–1276. [Google Scholar] [CrossRef]
- Borba-Santos, L.P.; Rodrigues, A.M.; Gagini, T.B.; Fernandes, G.F.; Castro, R.; de Camargo, Z.P.; Nucci, M.; Lopes-Bezerra, L.M.; Ishida, K.; Rozental, S. Susceptibility of Sporothrix brasiliensis isolates to amphotericin B, azoles, and terbinafine. Med. Mycol. 2015, 53, 178–188. [Google Scholar] [CrossRef]
- Thomson, J.; Trott, D.J.; Malik, R.; Galgut, B.; McAllister, M.M.; Nimmo, J.; Renton, D.; Kidd, S.E. An atypical cause of sporotrichosis in a cat. Med. Mycol. Case Rep. 2019, 23, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Honse, C.O.; Rodrigues, A.M.; Gremião, I.D.F.; Pereira, S.A.; Schubach, T.M.P. Use of local hyperthermia to treat sporotrichosis in a cat. Vet. Rec. 2010, 166, 208–209. [Google Scholar] [CrossRef] [PubMed]
- Hirano, M.; Watanabe, K.; Murakami, M.; Kano, R.; Yanai, T.; Yamazoe, K.; Fukata, T.; Kudo, T. A case of feline sporotrichosis. J. Vet. Med Sci. 2006, 68, 283–284. [Google Scholar] [CrossRef] [PubMed]
- Gremião, I.D.F.; Pereira, S.A.; Rodrigues, A.M.; Figueiredo, F.B.; Nascimento Júnior, A.; dos Santos, I.B.; Schubach, T.M.P. Tratamento cirúrgico associado à terapia antifúngica convencional na esporotricose felina. Acta Sci. Vet. 2006, 34, 221. [Google Scholar] [CrossRef]
- de Souza, C.P.; Lucas, R.; Ramadinha, R.H.; Pires, T.B. Cryosurgery in association with itraconazole for the treatment of feline sporotrichosis. J. Feline Med. Surg. 2016, 18, 137–143. [Google Scholar] [CrossRef]
| Technique | Specimen Type | Characteristics |
|---|---|---|
| Culture | Affected tissue, body fluids, or blood |
|
| Microscopy | Cytological preparation or lesion biopsy |
|
| Immuno-diagnosis | Serum |
|
| DNA Detection | Affected tissue, fresh or formalin-fixed paraffin-embedded tissue, body fluids, or Sporothrix isolate |
|
| MALDI-TOF MS | Sporothrix isolate |
|
| Antifungal Susceptibility Testing | Sporothrix isolate |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rossow, J.A.; Queiroz-Telles, F.; Caceres, D.H.; Beer, K.D.; Jackson, B.R.; Pereira, J.G.; Ferreira Gremião, I.D.; Pereira, S.A. A One Health Approach to Combatting Sporothrix brasiliensis: Narrative Review of an Emerging Zoonotic Fungal Pathogen in South America. J. Fungi 2020, 6, 247. https://doi.org/10.3390/jof6040247
Rossow JA, Queiroz-Telles F, Caceres DH, Beer KD, Jackson BR, Pereira JG, Ferreira Gremião ID, Pereira SA. A One Health Approach to Combatting Sporothrix brasiliensis: Narrative Review of an Emerging Zoonotic Fungal Pathogen in South America. Journal of Fungi. 2020; 6(4):247. https://doi.org/10.3390/jof6040247
Chicago/Turabian StyleRossow, John A., Flavio Queiroz-Telles, Diego H. Caceres, Karlyn D. Beer, Brendan R. Jackson, Jose Guillermo Pereira, Isabella Dib Ferreira Gremião, and Sandro Antonio Pereira. 2020. "A One Health Approach to Combatting Sporothrix brasiliensis: Narrative Review of an Emerging Zoonotic Fungal Pathogen in South America" Journal of Fungi 6, no. 4: 247. https://doi.org/10.3390/jof6040247
APA StyleRossow, J. A., Queiroz-Telles, F., Caceres, D. H., Beer, K. D., Jackson, B. R., Pereira, J. G., Ferreira Gremião, I. D., & Pereira, S. A. (2020). A One Health Approach to Combatting Sporothrix brasiliensis: Narrative Review of an Emerging Zoonotic Fungal Pathogen in South America. Journal of Fungi, 6(4), 247. https://doi.org/10.3390/jof6040247







