Fungi–Nematode Interactions: Diversity, Ecology, and Biocontrol Prospects in Agriculture
Abstract
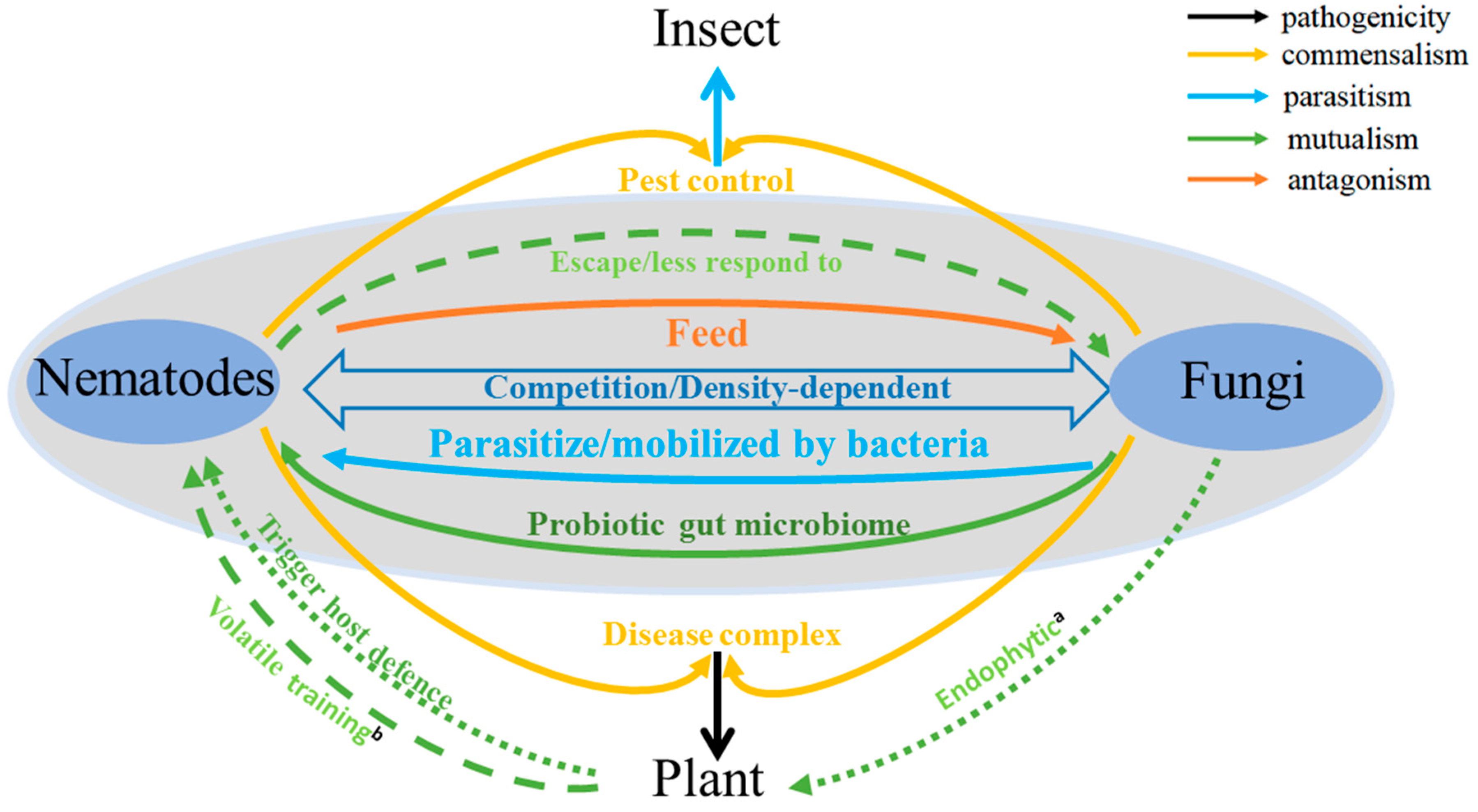
1. Introduction
2. Antagonistic Interactions
2.1. Nematodes Feeding on and Antagonizing Fungi
2.2. Fungi Antagonizing Nematodes
2.2.1. Diversity and Evolution of Fungal Predation Structures
2.2.2. Host Recognition, Adhesion, Host Specificity, and Infection Process
2.2.3. Competition between Nematode-Trapping Fungi and Nematodes
Innate Immune Defense Responses in Nematodes
Competition between Different Fungal Species and Nematodes
3. Synergistic Interactions between Phytophagous Nematodes and Phytopathogenic Fungi against Host Plants
3.1. Interactions between Phytophagous Nematodes and Soil-borne Fungal Pathogens
3.2. Factors Influencing Interactions between Phytophagous Nematodes and Phytopathogenic Fungi
4. Fungi and Nematodes Interact through a Third Party
4.1. Induced Resistance
4.2. Alteration of Root Exudates
4.3. Chemical Signals
4.4. Microbiome
5. Applications of Our Understanding in Fungi–Nematode Interactions in Agriculture: The Control of Phytophagous Nematodes and Soilborne Fungal Pathogens
5.1. Nematodes as Biocontrol Agents against Plant Pathogenic Fungi
5.2. Biocontrol of Nematodes with Nematophagous Fungi
5.2.1. Potential for the Discovery of Novel Candidates
5.2.2. Development and Integration of New Methods
Funding
Acknowledgments
Conflicts of Interest
References
- Topalovic, O.; Heuer, H. Plant-nematode interactions assisted by microbes in the rhizosphere. Curr. Issues Mol. Biol. 2019, 30, 75–88. [Google Scholar]
- Bongers, T.; Bongers, M. Functional diversity of nematodes. Appl. Soil Ecol. 1998, 10, 239–251. [Google Scholar]
- Burros, L. The Nature and Properties of Soils, 11th ed.; Nyle, C.B., Ray, R.W., Eds.; Prentice Hall Inc.: Upper Saddle River, NJ, USA, 1996; p. 740. [Google Scholar]
- Hassani, M.A.; Durán, P.; Hacquard, S. Microbial interactions within the plant holobiont. Microbiome 2018, 6, 58. [Google Scholar] [PubMed]
- Van Megen, H.; Elsen, S.V.D.; Holterman, M.; Karssen, G.; Mooyman, P.; Bongers, T.; Holovachov, O.; Bakker, J.; Helder, J. A phylogenetic tree of nematode based on about 1200 full-length small subunit ribosomal DNA sequences. Nematology 2009, 11, 927–950. [Google Scholar]
- Ragozzino, A.; D’Errico, G. Interactions between nematodes and fungi: A concise review. Redia 2011, 94, 123–125. [Google Scholar]
- Lamondia, J.; Timper, P. Interactions of microfungi and plant-parasitic nematodes. In Biology of Microfungi; Springer International Publishing: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Su, H.; Zhao, Y.; Zhou, J.; Feng, H.; Jiang, D.; Zhang, K.Q.; Yang, J. Trapping devices of nematode-trapping fungi: Formation, evolution, and genomic perspectives. Biol. Rev. 2017, 92, 357–368. [Google Scholar]
- Hasna, M.K.; Insunza, V.; Lagerlöf, J.; Rämert, B. Food attraction and population growth of fungivorous nematodes with different fungi. Ann. Appl. Biol. 2007, 151, 175–182. [Google Scholar]
- Wall, D.; Caswell, E. The ecology of nematodes in agroecosystems. Ann. Rev. Phytopathol. 2003, 23, 275–296. [Google Scholar]
- Giannakis, N. Interactions between Mycophagous Nematodes, Mycorrhizal and Other Soil Fungi. Ph.D. Thesis, University of Leeds, Leeds, UK, 1990. [Google Scholar]
- Maboreke, H.; Graf, M.; Grams, T.; Herrmann, S.; Scheu, S.; Ruess, L. Multitrophic interactions in the rhizosphere of a temperate forest tree affect plant carbon flow into the belowground food web. Soil Biol. Biochem. 2017, 115, 526–536. [Google Scholar]
- Hua, J.; Jiang, Q.; Bai, J.; Ding, F.; Lin, X.; Yin, Y. Interactions between arbuscular mycorrhizal fungi and fungivorous nematodes on the growth and arsenic uptake of tobacco in arsenic-contaminated soils. Appl. Soil Ecol. 2014, 84, 176–184. [Google Scholar]
- Lagerlöf, J.; Insunza, V.; Lundegårdh, B.; Rämert, B. Interaction between a fungal plant disease, fungivorous nematodes and compost suppressiveness. Acta Agric. Scand. Sect. B Plant Soil Sci. 2011, 61, 372–377. [Google Scholar]
- Haraguchi, S.; Yoshiga, T. Potential of the fungal feeding nematode Aphelenchus avenae to control fungi and the plant parasitic nematode Ditylenchus destructor associated with garlic. Biol. Control 2020, 143, 104203. [Google Scholar]
- Karim, N.; Jones, J.; Okada, H.; Kikuchi, T. Analysis of expressed sequence tags and identification of genes encoding cell-wall-degrading enzymes from the fungivorous nematode Aphelenchus avenae. BMC Genom. 2009, 10, 525. [Google Scholar]
- Kikuchi, T.; Jones, J.; Aikawa, T.; Kosaka, H.; Ogura, N. A family of glycosyl hydrolase family 45 cellulases from the pine wood nematode Bursaphelenchus xylophilus. FEBS Lett. 2004, 572, 201–205. [Google Scholar]
- Tayyrov, A.; Schmieder, S.; Bleuler-Martinez, S.; Plaza, D.; Künzler, M. Toxicity of potential fungal defense proteins towards the fungivorous nematodes Aphelenchus avenae and Bursaphelenchus okinawaensis. Appl. Environ. Microbiol. 2018, 84, e02051-18. [Google Scholar]
- Schmieder, S.; Stanley, C.; Rzepiela, A.; van Swaay, D.; Sabotič, J.; Nørrelykke, S.; deMello, A.; Aebi, M.; Künzler, M. Bidirectional propagation of signals and nutrients in fungal networks via specialized hyphae. Curr. Biol. 2019, 29, 217–228. [Google Scholar]
- Tayyrov, A.; Stanley, C.; Azevedo, S.; Künzler, M. Combining microfluidics and RNA-sequencing to assess the inducible defensome of a mushroom against nematodes. BMC Genom. 2019, 20, 243. [Google Scholar]
- Soares, F.E.d.F.; Sufiate, B.L.; de Queiroz, J.H. Nematophagous fungi: Far beyond the endoparasite, predator and ovicidal groups. Agric. Nat. Res. 2018, 52, 1–8. [Google Scholar]
- Persmark, L.; Jansson, H.B. Nematophagous fungi in the rhizosphere of agricultural crops. FEMS Microbiol. Ecol. 1997, 22, 303–312. [Google Scholar]
- Yang, E.; Xu, L.; Yang, Y.; Zhang, X.; Xiang, M.; Wang, C.; An, Z.; Liu, X. Origin and evolution of carnivorism in the Ascomycota (fungi). Proc. Natl. Acad. Sci. USA 2012, 109, 10960–10965. [Google Scholar]
- Hsueh, Y.P.; Mahanti, P.; Schroeder, F.C.; Sternberg, P.W. Nematode-trapping fungi eavesdrop on nematode pheromones. Curr. Biol. 2013, 23, 83–86. [Google Scholar] [CrossRef]
- Meerupati, T.; Andersson, K.M.; Friman, E.; Kumar, D.; Tunlid, A.; Ahren, D. Genomic mechanisms accounting for the adaptation to parasitism in nematode-trapping fungi. PLoS Genet. 2013, 9, e1003909. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, G.H.; Zou, C.G.; Ji, X.L.; Liu, T.; Zhao, P.J.; Liang, L.M.; Xu, J.P.; An, Z.Q.; Zheng, X.; et al. Bacteria can mobilize nematode-trapping fungi to kill nematodes. Nat. Commun. 2014, 5, 5776. [Google Scholar] [CrossRef]
- Ji, X.; Yu, Z.; Yang, J.; Xu, J.; Zhang, Y.; Liu, S.; Zou, C.; Li, J.; Liang, L.; Zhang, K.Q. Expansion of adhesion genes drives pathogenic adaptation of nematode-trapping fungi. iScience 2020, 23, 101057. [Google Scholar] [CrossRef]
- Yang, C.-T.; De Ulzurrun, G.V.-D.; Gonçalves, A.P.; Lin, H.-C.; Chang, C.-W.; Huang, T.-Y.; Chen, S.-A.; Lai, C.-K.; Tsai, I.J.; Schroeder, F.C.; et al. Natural diversity in the predatory behavior facilitates the establishment of a robust model strain for nematode-trapping fungi. Proc. Natl. Acad. Sci. USA 2020, 117, 6762–6770. [Google Scholar] [CrossRef] [PubMed]
- Hsueh, Y.P.; Gronquist, M.R.; Schwarz, E.M.; Nath, R.D.; Lee, C.H.; Gharib, S.; Schroeder, F.C.; Sternberg, P.W. Nematophagous fungus Arthrobotrys oligospora mimics olfactory cues of sex and food to lure its nematode prey. eLife 2017, 6, 79. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xiang, M.; Che, Y. The living strategy of nematophagous fungi. Mycoscience 2009, 50, 20–25. [Google Scholar] [CrossRef]
- Li, J.; Zou, C.; Xu, J.; Ji, X.; Niu, X.; Yang, J.; Huang, X.; Zhang, K.Q. Molecular mechanisms of nematode-nematophagous microbe interactions: Basis for biological control of plant-parasitic nematodes. Annu. Rev. Phytopathol. 2015, 53, 67–95. [Google Scholar] [CrossRef]
- Jiang, X.; Xiang, M.; Liu, X. Nematode-trapping fungi. In The Fungai Kingdom; ASM Press: Washington, DC, USA, 2017; Volume 5. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, E.; An, Z.; Liu, X. Evolution of nematode-trapping cells of predatory fungi of the Orbiliaceae based on evidence from rRNA-encoding DNA and multiprotein sequences. Proc. Natl. Acad. Sci. USA 2007, 104, 8379–8384. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.Q.; Hyde, K.D. Nematode-Trapping Fungi; Zhang, K.Q., Hyde, K.D., Eds.; Springer Science & Business: Dordrecht, The Netherlands, 2014; Volume 23. [Google Scholar]
- Persmark, L.; Nordbring-Hertz, B. Conidial trap formation of nematode-trapping fungi in soil and soil extracts. FEMS Microbiol. Ecol. 1997, 22, 313–323. [Google Scholar] [CrossRef]
- Li, L.; Yang, M.; Qu, Q.; Chen, Y.; Luo, J.; Liang, L.; Zhang, K. Nematode-trapping fungi and fungus-associated bacteria interactions: The role of bacterial diketopiperazines and biofilms on Arthrobotrys oligospora surface in hyphal morphogenesis. Environ. Microbiol. 2016, 18, 3827–3839. [Google Scholar] [CrossRef] [PubMed]
- Barron, G.; Dierkes, Y. Nematophagous fungi: Hohenbuehelia the perfect state of nematoctonus. Can. J. Bot. 1977, 55, 3054–3062. [Google Scholar] [CrossRef]
- Drechsler, C. Four phycomycetes destructive to nematodes and rhizopods. Mycologia 1941, 33, 248–269. [Google Scholar] [CrossRef]
- Gray, N. Nematophagous fungi with particular reference to their ecology. Biol. Rev. 1987, 62, 245–304. [Google Scholar] [CrossRef]
- Tzean, S. Nematophagous resupinate basidiomycetous fungi. Phytopathology 1993, 83, 1015–1020. [Google Scholar] [CrossRef]
- Luo, H.; Liu, Y.; Fang, L.; Li, X.; Tang, N.; Zhang, K. Coprinus comatus damages nematode cuticles mechanically with spiny balls and produces potent toxins to immobilize nematodes. Appl. Environ. Microbiol. 2007, 73, 3916–3923. [Google Scholar] [CrossRef]
- Luo, H.; Li, X.; Li, G.; Pan, Y.; Zhang, K. Acanthocytes of Stropharia rugosoannulata function as a nematode-attacking device. Appl. Environ. Microbiol. 2006, 72, 2982–2987. [Google Scholar] [CrossRef]
- Beakes, G.; Glockling, S. Injection tube differentiation in gun cells of a Haptoglossa species which infects nematodes. Fung. Genet. Biol. 1998, 24, 45–68. [Google Scholar] [CrossRef]
- Yang, J.; Wang, L.; Ji, X.; Feng, Y.; Li, X.; Zou, C.-G.; Xu, J.; Ren, Y.; Mi, Q.; Wu, J.; et al. Genomic and proteomic analyses of the fungus Arthrobotrys oligospora provide insights into nematode-trap formation. PLoS Pathog. 2011, 7, e1002179. [Google Scholar] [CrossRef]
- Zhang, W.; Cheng, X.; Liu, X.; Xiang, M. Genome studies on nematophagous and entomogenous fungi in China. J. Fungi 2016, 2, 9. [Google Scholar] [CrossRef]
- Larriba, E.; Jaime, M.; Carbonell-Caballero, J.; Conesa, A.; Dopazo, J.; Nislow, C.; Martín-Nieto, J.; Lopez-Llorca, L. Sequencing and functional analysis of the genome of a nematode egg-parasitic fungus, Pochonia chlamydosporia. Fung. Genet. Biol. 2014, 65, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Zhang, W.; Lai, Y.; Xiang, M.; Wang, X.; Zhang, X.; Liu, X. Drechslerella stenobrocha genome illustrates the mechanism of constricting rings and the origin of nematode predation in fungi. BMC Genom. 2014, 15, 114. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.; Liu, K.; Zhang, X.; Xiaoling, Z.; Li, K.; Wang, N.; Shu, C.; Yunpeng, W.; Wang, C.; Bushley, K.; et al. Comparative genomics and transcriptomics analyses reveal divergent lifestyle features of nematode endoparasitic fungus Hirsutella minnesotensis. Genome Biol. Evol. 2014, 11, 3077–3093. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, G.Z.; Fang, M.L.; Deng, C.; Zhang, K.Q.; Yu, Z.F.; Xu, J.P. Comparative analyses of mitochondrial genomes provide evolutionary insights into nematode-trapping fungi. Front. Microbiol. 2020, 11, 617. [Google Scholar] [CrossRef] [PubMed]
- Nordbring-Hertz, B. Scanning electron microscopy of the nematode-trapping organs in Arthrobotrys oligospora. Physiol. Plant. 1972, 26, 279–284. [Google Scholar] [CrossRef]
- Veenhuis, D.M.; van Wijk, C.; Wyss, U.; Nordbring-Hertz, B.; Harder, W. Significance of electron dense microbodies in trap cells of the nematophagous fungus Arthrobotrys oligospora. Anton. Leeuw. 1989, 56, 251–261. [Google Scholar] [CrossRef]
- Xie, M.H.; Bai, N.; Yang, J.L.; Jiang, K.X.; Zhou, D.X.; Zhao, Y.N.; Li, D.N.; Niu, X.M.; Zhang, K.Q.; Yang, J.K. Protein kinase Ime2 is required for mycelial growth, conidiation, osmoregulation, and pathogenicity in nematode-trapping fungus Arthrobotrys oligospora. Front. Microbiol. 2020, 10, 3065. [Google Scholar] [CrossRef]
- Tunlid, A.; Johansson, T.; Nordbring-Hertz, B. Surface polymers of the nematode-trapping fungus Arthrobotrys oligospora. J. Gen. Microbiol. 1991, 137, 1231–1240. [Google Scholar] [CrossRef]
- Johnstone, I.L. The cuticle of the nematode Caenorhabditis elegans: A complex collagen structure. Bioessays 1994, 16, 171–178. [Google Scholar] [CrossRef]
- Yang, J.K.; Tian, B.Y.; Liang, L.M.; Zhang, K.Q. Extracellular enzymes and the pathogenesis of nematophagous fungi. Appl. Microbiol. Biotechnol. 2007, 75, 21–31. [Google Scholar] [CrossRef]
- Li, J.; Yu, L.; Yang, J.K.; Dong, L.Q.; Tian, B.Y.; Yu, Z.F.; Liang, L.M.; Zhang, Y.; Wang, X.; Zhang, K.Q. New insights into the evolution of subtilisin-like serine protease genes in Pezizomycotina. BMC Evol. Biol. 2010, 10, 68. [Google Scholar] [CrossRef] [PubMed]
- Nordbring-Hertz, B.; Mattiasson, B. Action of a nematode-trapping fungus shows lectin-mediated host-microorganism interaction. Nature 1979, 281, 477–479. [Google Scholar] [CrossRef]
- Rosen, S.; Ek, B.; Rask, L.; Tunlid, A. Purification and Characterization of a Surface Lectin from the Nematode-Trapping Fungus Arthrobotrys oligospora. J. Gen. Microbiol. 1992, 138, 2663–2672. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Cheng, X.; Wang, J.; Tian, D.; Tang, K.; Xu, T.; Zhang, M.; Wang, Y.; Wang, M. Structural insights into the fungi-nematodes interaction mediated by fucose-specific lectin AofleA from Arthrobotrys oligospora. Int. J. Biol. Macromol. 2020, 164, 783–793. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.M.; Shen, R.F.; Mo, Y.Y.; Yang, J.K.; Ji, X.L.; Zhang, K.Q. A proposed adhesin AoMad1 helps nematode-trapping fungus Arthrobotrys oligospora recognizing host signals for life-style switching. Fung. Genet. Biol. 2015, 81, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Youssar, L.; Wernet, V.; Hensel, N.; Yu, X.; Hildebrand, H.G.; Schreckenberger, B.; Kriegler, M.; Hetzer, B.; Frankino, P.; Dillin, A.; et al. Intercellular communication is required for trap formation in the nematode-trapping fungus Duddingtonia flagrans. PLoS Genet. 2019, 15, e1008029. [Google Scholar] [CrossRef]
- Wang, B.L.; Chen, Y.H.; He, J.N.; Xue, H.X.; Yan, N.; Zeng, Z.J.; Bennett, J.W.; Zhang, K.Q.; Niu, X.M. Integrated metabolomics and morphogenesis reveal volatile signaling of the nematode-trapping fungus Arthrobotrys oligospora. Appl. Environ. Microbiol. 2018, 84. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Liu, Z.; Liu, L.; Li, J.; Gao, H.; Yang, J.; Zhang, K.-Q. The nitrate assimilation pathway is involved in the trap formation of Arthrobotrys oligospora, a nematode-trapping fungus. Fung. Genet. Biol. 2016, 92, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.L.; Gao, Y.; Zhang, K.Q.; Zou, C.G. Autophagy is required for trap formation in the nematode-trapping fungus Arthrobotrys oligospora. Environ. Microbiol. Rep. 2013, 5, 511–517. [Google Scholar] [CrossRef]
- Tunlid, A.; Rosén, S.; Ek, B.; Rask, L. Purification and characterization of an extracellular serine protease from the nematode-trapping fungus Arthrobotrys oligospora. Microbiology 1994, 140, 1687–1695. [Google Scholar] [CrossRef]
- Yang, J.; Le, C.; Liang, L.; Tian, B.; Zhang, Y.; Cheng, C.; Zhang, K.-Q. Cloning and characterization of an extracellular serine protease from the nematode-trapping fungus Arthrobotrys conoides. Arch. Microbiol. 2007, 188, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.B.; Yang, J.; Lin, C.; Zhang, Y.; Zhang, K.Q. Purification and characterization of an extracellular serine protease from the nematode-trapping fungus Dactylella shizishanna. Lett. Appl. Microbiol. 2006, 42, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Liang, L.; Zhang, Y.; Le, C.; Zhang, L.; Ye, F.; Gan, Z.; Zhang, K.-Q. Purification and cloning of a novel serine protease from the nematode-trapping fungus Dactylellina varietas and its potential roles in infection against nematodes. Appl. Microbiol. Biotechnol. 2007, 75, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Yang, J.; Zhang, K.-Q. Characterization of an extracellular protease and its cDNA from the nematode-trapping fungus Monacrosporium microscaphoides. Can. J. Microbiol. 2006, 52, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.-K.; Ye, F.-P.; Mi, Q.-L.; Tang, S.-Q.; Le, C.; Yang, J. Purification and cloning of an extracellular serine protease from the nematode-trapping fungus Monacrosporium cystosporium. J. Microbiol. Biotechnol. 2008, 18, 852–858. [Google Scholar] [PubMed]
- Lebrigand, K.; He, L.D.; Thakur, N.; Arguel, M.J.; Polanowska, J.; Henrissat, B.; Record, E.; Magdelenat, G.; Barbe, V.; Raffaele, S.; et al. Comparative genomic analysis of Drechmeria coniospora reveals core and specific genetic requirements for fungal endoparasitism of nematodes. PLoS Genet. 2016, 12, e1006017. [Google Scholar] [CrossRef] [PubMed]
- Jansson, H.-B.; Jeyaprakash, A.; Zuckerman, B. Differential adhesion and infection of nematodes by the endo-parasitic fungus Meria coniospora (Deuteromycetes). Appl. Environ. Microbiol. 1985, 49, 552–555. [Google Scholar] [CrossRef]
- Zuckerman, B.; Dicklow, M.B.; Coles, G.; Jansson, H.-B. Cryopreservation studies on the nematophagous fungus Drechmeria coniospora. Revue Nematol. 1988, 11, 327–331. [Google Scholar]
- Zhang, L.; Zhou, Z.; Guo, Q.; Fokkens, L.; Miskei, M.; Pócsi, I.; Zhang, W.; Chen, M.; Wang, L.; Sun, Y.; et al. Insights into adaptations to a near-obligate nematode endoparasitic lifestyle from the finished genome of Drechmeria coniospora. Sci. Rep. 2016, 6, 23122. [Google Scholar] [CrossRef]
- Wang, R.; Dong, L.; He, R.; Wang, Q.; Chen, Y.; Liangjian, Q.; Zhang, Y.-A. Comparative genomic analyses reveal the features for adaptation to nematodes in fungi. DNA Res. 2018, 25, 245–256. [Google Scholar] [CrossRef]
- Wang, Y.-L.; Li, L.-F.; Li, D.-X.; Wang, B.; Zhang, K.; Niu, X. Yellow pigment aurovertins mediate interactions between the pathogenic fungus Pochonia chlamydosporia and its nematode host. J. Agric. Food Chem. 2015, 63, 6577–6587. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Llorca, L.; Olivares-Bernabeu, C.; Salinas, J.; Jansson, H.-B.; Kolattukudy, P. Pre-penetration events in fungal parasitism of nematode eggs. Mycol. Res. 2002, 106, 499–506. [Google Scholar] [CrossRef]
- Huang, X.; Zhao, N.; Zhang, K. Extracellular enzymes serving as virulence factors in nematophagous fungi involved in infection of the host. Res. Microbiol. 2005, 155, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-H.; Chang, H.-W.; Yang, C.-T.; Wali, N.; Shie, J.-J.; Hsueh, Y.-P. Sensory cilia as the Achilles heel of nematodes when attacked by carnivorous mushrooms. Proc. Natl. Acad. Sci. USA 2020, 117, 6014–6022. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, K.-Q.; Hyde, K. The ecology of nematophagous fungi in natural environments. In Nematode-Trapping Fungi; Zhang, K.-Q., Hyde, K.D., Eds.; Springer: Dordrecht, The Netherlands, 2014; pp. 211–229. [Google Scholar]
- Jansson, H.-B. Predacity by nematophagous fungi and its relation to the attraction of nematodes. Microb. Ecol. 1982, 8, 233–240. [Google Scholar] [CrossRef]
- McCallum, K.; Garsin, D. The role of reactive oxygen species in modulating the Caenorhabditis elegans immune response. PLoS Pathog. 2016, 12, e1005923. [Google Scholar] [CrossRef] [PubMed]
- Couillault, C.; Pujol, N.; Reboul, J.; Ehret-Sabatier, L.; Guichou, J.-F.; Kohara, Y.; Ewbank, J. TLR-independent control of innate immunity in Caenorhabditis elegans by the TIR domain adaptor protein TIR-1, an ortholog of human SARM. Nat. Immunol. 2004, 5, 488–494. [Google Scholar] [CrossRef]
- Pujol, N.; Zugasti, O.; Wong, D.; Couillault, C.; Kurz, C.; Schulenburg, H.; Ewbank, J. Anti-fungal innate immunity in C. elegans is enhanced by evolutionary diversification of antimicrobial peptides. PLoS Pathog. 2008, 4, e1000105. [Google Scholar] [CrossRef]
- Zou, C.-G.; Tu, Q.; Niu, J.; Ji, X.-L.; Zhang, K.-Q. The DAF-16/FOXO transcription factor functions as a regulator of epidermal innate immunity. PLoS Pathog. 2013, 9, e1003660. [Google Scholar] [CrossRef]
- Nag, P.; Aggarwal, P.; Ghosh, S.; Narula, K.; Tayal, R.; Maheshwari, N.; Chakraborty, N.; Chakraborty, S. Interplay of neuronal and non-neuronal genes regulates intestinal DAF-16-mediated immune response during Fusarium infection of Caenorhabditis elegans. Cell Death Discov. 2017, 3, 17073. [Google Scholar] [CrossRef]
- Singh, U.B.; Sahu, A.; Sahu, N.; Singh, B.P.; Singh, R.K.; Renu; Singh, D.P.; Jaiswal, R.K.; Sarma, B.K.; Singh, H.B.; et al. Can endophytic Arthrobotrys oligospora modulate accumulation of defence related biomolecules and induced systemic resistance in tomato (Lycopersicon esculentum Mill.) against root knot disease caused by Meloidogyne incognita. Appl. Soil Ecol. 2014, 63, 45–56. [Google Scholar]
- Dijksterhuis, J.; Sjollema, K.; Veenhuis, D.M.; Harder, W. Competitive interactions between two nematophagous fungi during infection and digestion of the nematode Panagrellus redivivus. Mycol. Res. 1994, 98, 1458–1462. [Google Scholar] [CrossRef]
- El-Borai Kora, F.; Stuart, R.; Campos-Herrera, R.; Pathak, E.; Duncan, L. Entomopathogenic nematodes, root weevil larvae, and dynamic interactions among soil texture, plant growth, herbivory, and predation. J. Invertebr. Pathol. 2011, 109, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Alexander, M. Why microbial predators and parasites do not eliminate their prey and hosts. Ann. Rev. Microbiol. 1981, 35, 113–133. [Google Scholar] [CrossRef] [PubMed]
- Phillips, A.J.; Mangel, M. Density-dependent host-pathogen dynamics in soil microcosms. Ecology 1992, 73, 495–506. [Google Scholar] [CrossRef]
- Jaffee, B.; Muldoon, A. Numerical responses of the nematophagous fungi Hirsutella rhossiliensis, Monacrosporium cionopagum, and M. ellipsosporum. Mycologia 1995, 87, 643–650. [Google Scholar] [CrossRef]
- Jaffee, B.A.; Innis, T.M. Sampling strategies for detection of density-dependent parasitism of soil-borne nematodes by nematophagous fungi. Rev. Nematol. 1991, 14, 147–150. [Google Scholar]
- Jaffee, B.; Gaspard, J.; Ferris, H.; Muldoon, A. Quantification of parasitism of the soil-borne nematode Criconemella xenoplax by the nematophagous fungus Hirsutella rhossiliensis. Soil Biol. Biochem. 1988, 20, 631–636. [Google Scholar] [CrossRef]
- Pathak, E.; Campos-Herrera, R.; El-Borai, F.; Duncan, L. Spatial relationships between entomopathogenic nematodes and nematophagous fungi in Florida citrus orchards. J. Invertebr. Pathol. 2017, 144, 37–46. [Google Scholar] [CrossRef]
- Costa, S.; Kerry, B.; Bardgett, R.; Davies, K. Interactions between nematodes and their microbial enemies in coastal sand dunes. Oecologia 2012, 170, 1053–1066. [Google Scholar] [CrossRef]
- Pathak, E.; El-Borai Kora, F.; Campos-Herrera, R.; Johnson, E.; Stuart, R.; Graham, J.; Duncan, L. Use of real-time PCR to discriminate predatory and saprophagous behavior by nematophagous fungi. Fung. Biol. 2012, 116, 563–573. [Google Scholar] [CrossRef] [PubMed]
- Jaffuel, G.; Mäder, P.; Blanco-Pérez, R.; Chiriboga Morales, X.; Fliessbach, A.; Turlings, T.; Campos-Herrera, R. Prevalence and activity of entomopathogenic nematodes and their antagonists in soils that are subject to different agricultural practices. Agr. Ecosys. Environ. 2016, 230, 329–340. [Google Scholar] [CrossRef]
- Duncan, L.W.; Stuart, R.; El-Borai Kora, F.; Campos-Herrera, R.; Pathak, E.; Giurcanu, M.; Graham, J.H. Modifying orchard planting sites conserves entomopathogenic nematodes, reduces weevil herbivory and increases citrus tree growth, survival and fruit yield. Biol. Control 2013, 64, 26–36. [Google Scholar] [CrossRef]
- Atkinson, G.F. Some diseases of cotton. Ala. Agric. Exp. Stn. Bull. 1892, 41, 61–65. [Google Scholar]
- Lacey, L.; Grzywacz, D.; Shapiro-Ilan, D.; Frutos, R.; Brownbridge, M.; Goettel, M. Insect pathogens as biological control agents: Back to the future. J. Invertebr. Pathol. 2015, 132, 1–41. [Google Scholar] [CrossRef]
- Ansari, M.; Shah, F.; Tirry, L.; Moens, M. Field trials against Hoplia philanthus (Coleoptera: Scarabaeidae) with a combination of an entomopathogenic nematode and the fungus Metarhizium anisopliae CLO 53. Biol. Control 2006, 39, 453–459. [Google Scholar] [CrossRef]
- Wu, S.-Y.; El-Borai, F.; Graham, J.; Duncan, L. The saprophytic fungus Fusarium solani increases the insecticidal efficacy of the entomopathogenic nematode Steinernema diaprepesi. J. Invertebr. Pathol. 2018, 159, 87–94. [Google Scholar] [CrossRef]
- Ansari, M.; Shah, F.; Butt, T. Combined use of entomopathogenic nematodes and Metarhizium anisopliae as a new approach for black vine weevil, Otiorhynchus sulcatus (Coleoptera: Curculionidae) control. Entomo. Exp. Appl. 2008, 129, 340–347. [Google Scholar] [CrossRef]
- Navarro, P.; Ii, J.; Stock, S.P. Interactions between the entomopathogenic nematode Heterorhabditis sonorensis (Nematoda: Heterorhabditidae) and the saprobic fungus Fusarium oxysporum (Ascomycota: Hypocreales). J. Invertebr. Pathol. 2013, 115, 41–47. [Google Scholar] [CrossRef]
- Carneiro, F.; Ramalho, M.; Pereira, M. Fusarium oxysporum f. sp. phaseoli and Meloidogyne incognita interaction in common bean. Crop. Breed. Appl. Biotechnol. 2010, 10, 271–274. [Google Scholar] [CrossRef]
- El-Shennawy, M.Z.; Khalifa, E.Z.; Ammar, M.M.; Mousa, E.M.; Hafez, S.L. Biological control of the disease complex on potato caused by root-knot nematode and Fusarium wilt fungus. Nematol. Medit. 2012, 40, 169–172. [Google Scholar]
- Alhazmi, A.; Al-Nadary, S.N. Interaction between Meloidogyne incognita and Rhizoctonia solani on green beans. Saudi J. Biol. Sci. 2015, 27, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Parkunan, V.; Timper, P.; Ji, P. Lack of influence of Meloidogyne incognita on resistance of bell pepper cultivars to Phytophthora capsici. Can. J. Plant Pathol. 2016, 38, 1–7. [Google Scholar] [CrossRef]
- Wanjohi, W.J.; Wafula, G.O.; Macharia, C.M. Integrated management of Fusarium wilt-root knot nematode complex on tomato in central highlands of Kenya. Sustain. Agric. Res. 2018, 7, 8. [Google Scholar] [CrossRef]
- Hajji-Hedfi, L.; M’Hamdi-Boughalleb, N.; Horrigue-Raouani, N. Fungal diversity in rhizosphere of root-knot nematode infected tomatoes in Tunisia. Symbiosis 2019, 79, 171–181. [Google Scholar] [CrossRef]
- Beyan, A. Response of tomato genotypes to Meloidogyne javanica and Fusarium oxysporum f.sp. lycopersici co-infestation under glasshouse conditions. Pak. J. Nematol. 2019, 37, 63–82. [Google Scholar] [CrossRef]
- Alfadhl, F. Efficacy of some control agents in controlling seedling decline in figs caused by disease complex of Meloidogyne incognita and Fusarium solani. Plant Soil 2019, 17, 323–330. [Google Scholar]
- Keinath, A.P.; Wechter, W.P.; Rutter, W.B.; Agudelo, P.A. Cucurbit Rootstocks Resistant to Fusarium oxysporum f. sp. niveum Remain Resistant When Coinfected by Meloidogyne incognita in the Field. Plant Dis. 2019, 103, 1383–1390. [Google Scholar] [CrossRef]
- Scherlach, K.; Hertweck, C. Mediators of mutualistic microbe—Microbe interactions. Nat. Prod. Rep. 2018, 35, 303–308. [Google Scholar] [CrossRef]
- Khan, M.; Siddiqui, Z.A. Interactions of Meloidogyne incognita, Ralstonia solanacearum and Phomopsis vexans on eggplant in sand mix and fly ash mix soils. Sci. Hortic. 2017, 225, 177–184. [Google Scholar] [CrossRef]
- Ahmad, L.; Siddiqui, Z.A.; Abd_Allah, E.F. Effects of interaction of Meloidogyne incognita, Alternaria dauci and Rhizoctonia solani on the growth, chlorophyll, carotenoid and proline contents of carrot in three types of soil. Acta Agric. Scand. Sect. B Plant Soil Sci. 2019, 69, 324–331. [Google Scholar] [CrossRef]
- Björsell, P.; Edin, E.; Viketoft, M. Interactions between some plant-parasitic nematodes and Rhizoctonia solani in potato fields. Appl. Soil Ecol. 2017, 113, 151–154. [Google Scholar] [CrossRef]
- Memari, Z.; Karimi, J.; Kamali, S.; Goldansaz, S.; Hosseini, M. Are entomopathogenic nematodes effective biological control agents against the carob moth, Ectomyelois ceratoniae? J. Nematol. 2016, 48, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.-Y.; Duncan, L. Recruitment of an insect and its nematode natural enemy by olfactory cues from a saprophytic fungus. Soil Biol. Biochem. 2020, 144, 107781. [Google Scholar] [CrossRef]
- Back, M.; Haydock, P.; Jenkinson, P. Disease complexes involving plant parasitic nematodes and soilborne pathogens. Plant Pathol. 2002, 51, 683–697. [Google Scholar] [CrossRef]
- Ibrahim, S.A.; Salem, H.H. Initial fungal infection reduce the penetration and reproduction rate of Steinernema riobravae in Galleria mellonella. Egypt. Acad. J. Biol. Sci. A, Entomol. 2019, 12, 101–109. [Google Scholar] [CrossRef]
- Martinuz, A.; Zewdu, G.; Ludwig, N.; Grundler, F.; Sikora, R.; Schouten, A. The application of Arabidopsis thaliana in studying tripartite interactions among plants, beneficial fungal endophytes and biotrophic plant-parasitic nematodes. Planta 2014, 241, 1015–1025. [Google Scholar] [CrossRef]
- Martinez-Medina, A.; Fernandez, I.; Lok, G.B.; Pozo, M.J.; Pieterse, C.M.; Van Wees, S.C. Shifting from priming of salicylic acid- to jasmonic acid-regulated defences by Trichoderma protects tomato against the root knot nematode Meloidogyne incognita. New Phytol. 2017, 213, 1363–1377. [Google Scholar] [CrossRef]
- Schouten, A. Mechanisms involved in nematode control by endophytic fungi. Annu. Rev. Phytopathol. 2016, 54, 121–142. [Google Scholar] [CrossRef]
- Khan, A.; Williams, K.; Nevalainen, H. Effects of Paecilomyces lilacinus protease and chitinase on the eggshell structures and hatching of Meloidogyne javanica juveniles. Biol. Control 2004, 31, 346–352. [Google Scholar] [CrossRef]
- Zhou, W.; Wheeler, T.A.; Starr, J.L.; Valencia, C.U.; Sword, G.A. A fungal endophyte defensive symbiosis affects plant-nematode interactions in cotton. Plant Soil 2016, 422, 251–266. [Google Scholar] [CrossRef]
- Singh, U.; Sahu, P.; Singh, S.; Malviya, D.; Chaurasia, R.; Sharma, S.; Saxena, A. Drechslerella dactyloides and Dactylaria brochopaga mediated induction of defense related mediator molecules in tomato plants pre-challenged with Meloidogyne incognita. Indian Phytopathol. 2019, 72, 309–320. [Google Scholar] [CrossRef]
- Larriba, E.; Jaime, M.; Nislow, C.; Martín-Nieto, J.; Lopez-Llorca, L. Endophytic colonization of barley (Hordeum vulgare) roots by the nematophagous fungus Pochonia chlamydosporia reveals plant growth promotion and a general defense and stress transcriptomic response. J. Plant Res. 2015, 128, 665–678. [Google Scholar] [CrossRef] [PubMed]
- Terhonen, E.; Sipari, N.; Asiegbu, F. Inhibition of phytopathogens by fungal root endophytes of Norway spruce. Biol. Control 2016, 99, 53–63. [Google Scholar] [CrossRef]
- Escudero, N.; Lopez-Moya, F.; Ghahremani, Z.; Zavala-González, E.; Alaguero-Cordovilla, A.; Ros Ibáñez, C.; Lacasa, A.; Sorribas, F.; Lopez-Llorca, L. Chitosan increases tomato root colonization by Pochonia chlamydosporia and their combination reduces root-knot nematode damage. Front. Plant Sci. 2017, 8, 1415. [Google Scholar] [CrossRef]
- Strom, N.; Hu, W.; Haarith, D.; Chen, S.; Bushley, K. Corn and soybean host root endophytic fungi with toxicity toward the soybean cyst nematode. Phytopathology 2019, 110, 603–614. [Google Scholar] [CrossRef]
- Torto, B.; Cortada, L.; Murungi, L.; Haukeland, S.; Coyne, D. Management of cyst and root knot nematodes: A chemical ecology perspective. J. Agric. Food Chem. 2018, 66, 8672–8678. [Google Scholar] [CrossRef]
- Hallmann, J.; Sikoraand, R.A. Endophytic fungi. In Biological Control of Plant-Parasitic Nematodes; Spiegel, K.D.A.Y., Ed.; Springer: Dordrecht, The Netherlands, 2011; pp. 227–258. [Google Scholar]
- Vos, C.; Tesfahun, A.; Panis, B.; De Waele, D.; Elsen, A. Arbuscular mycorrhizal fungi induce systemic resistance in tomato against the sedentary nematode Meloidogyne incognita and the migratory nematode Pratylenchus penetrans. Appl. Soil Ecol. 2012, 61, 1–6. [Google Scholar] [CrossRef]
- Grundler, F.; Schnibbe, L.; Wyss, U. In vitro studies on the behaviour of second-stage juveniles of Heterodera schachtii (Nematoda: Heteroderidae) in response to host plant root exudates. Parasitology 1991, 103, 149–155. [Google Scholar] [CrossRef]
- Kirwa, H.; Murungi, L.; Beck, J.; Torto, B. Elicitation of differential responses in the root-knot nematode Meloidogyne incognita to tomato root exudate cytokinin, flavonoids, and alkaloids. J. Agric. Food Chem. 2018, 66, 11291–11300. [Google Scholar] [CrossRef]
- Boller, T.; Felix, G. A renaissance of elicitors: Perception of microbe-associated molecular patterns and danger signals by pattern-recognition receptors. Annu. Rev. Plant Biol. 2009, 60, 379–406. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, J.; Kaplan, F.; Ajredini, R.; Zachariah, C.; Alborn, H.; Teal, P.; Malik, R.; Edison, A.; Sternberg, P.; Schroeder, F. A blend of small molecules regulates both mating and development in Caenorhabditis elegans. Nature 2008, 454, 1115–1118. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, J.; von Reuss, S.; Bose, N.; Zaslaver, A.; Mahanti, P.; Ho, M.; O’Doherty, O.; Edison, A.; Sternberg, P.; Schroeder, F. A modular library of small molecule signals regulates social behaviors in Caenorhabditis elegans. PLoS Biol. 2012, 10, e1001237. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Ahmad, F.; Lu, M.; Zhang, W.; Wickham, J.; Sun, J. Ascarosides promote the prevalence of ophiostomatoid fungi and an invasive pathogenic nematode, Bursaphelenchus xylophilus. J. Chem. Ecol. 2018, 44, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.X.; Tan, J.L.; Wei, L.X.; Wang, Y.L.; Zhang, C.P.; Wu, D.K.; Zhu, C.Y.; Zhang, Y.; Zhang, K.Q.; Niu, X.M. Morphology regulatory metabolites from Arthrobotrys oligospora. J. Nat. Prod. 2012, 75, 1419–1423. [Google Scholar] [CrossRef]
- Su, H.N.; Xu, Y.Y.; Wang, X.; Zhang, K.Q.; Li, G.H. Induction of trap formation in nematode-trapping fungi by bacteria-released ammonia. Lett. Appl. Microbiol. 2016, 62, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Willett, D.; Alborn, H.; Duncan, L.; Stelinski, L. Social networks of educated nematodes. Sci. Rep. 2015, 5, 14388. [Google Scholar] [CrossRef]
- Willett, D.; Alborn, H.; Stelinski, L. Multitrophic effects of belowground parasitoid learning. Sci. Rep. 2017, 7, 2067. [Google Scholar] [CrossRef]
- Dirksen, P.; Marsh, S.; Braker, I.; Heitland, N.; Wagner, S.; Nakad, R.; Mader, S.; Petersen, C.; Kowallik, V.; Rosenstiel, P.; et al. The native microbiome of the nematode Caenorhabditis elegans: Gateway to a new host-microbiome model. BMC Biol. 2016, 14, 38. [Google Scholar] [CrossRef]
- Toju, H.; Tanaka, Y. Consortia of anti-nematode fungi and bacteria in the rhizosphere of soybean plants attacked by root-knot nematodes. R. Soc. Open Sci. 2019, 6, 181693. [Google Scholar] [CrossRef]
- Hamid, M.I.; Hussain, M.; Yunpeng, W.; Xiaoling, Z.; Xiang, M.; Liu, X. Successive soybean-monoculture cropping assembles rhizosphere microbial communities for the soil suppression of soybean cyst nematode. FEMS Microbiol. Ecol. 2017, 93, fiw222. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Cao, Y.; Zhang, K.-Q. Metagenomic insights into communities, functions of endophytes, and their associates with infection by root-knot nematode, Meloidogyne incognita, in tomato roots. Sci. Rep. 2015, 5, 17087. [Google Scholar] [CrossRef] [PubMed]
- Xiao, G.; Ying, S.-h.; Peng, Z.; Wang, Z.-L.; Zhang, S.; Xie, X.-Q.; Shang, Y.; Stleger, R.; Zhao, G.-P.; Wang, C.; et al. Genomic perspectives on the evolution of fungal entomopathogenicity in Beauveria bassiana. Sci. Rep. 2012, 2, 483. [Google Scholar] [CrossRef]
- Chanthala, L.; Kurrey, N.; Appaiah, A.; Rao, R. Probiotic yeasts inhibit virulence of non-albicans Candida Species. mBio 2019, 10, e02307–e02319. [Google Scholar]
- Haarith, D.; Kim, D.-G.; Strom, N.; Chen, S.; Bushley, K. In vitro screening of a culturable soybean cyst nematode cyst mycobiome for potential biological control agents and biopesticides. Phytopathology 2020, 110, 1388–1397. [Google Scholar] [CrossRef]
- Jun, O.-K.; Kim, Y.H. Aphelenchus avenae and antagonistic fungi as biological control agents of pythium spp. Plant Pathol. J. 2004, 20, 271–276. [Google Scholar] [CrossRef]
- Griffin, G.; Eisenback, J.; Yancey, M.; Templeton, J. Aphelenchoides hylurgi as a carrier of white, Hypovirulent Cryphonectria parasitica and its possible role in hypovirulence spread on blight-controlled American Chestnut trees. J. Nematol. 2009, 41, 267–273. [Google Scholar]
- Nickle, W.; McIntosh, P. Studies on the feeding and reproduction of seven mycophagous nematodes on Rhizoctonia, Fusarium, and Verticillium. Nematologica 1968, 14, 11–12. [Google Scholar]
- De la Cruz, R.G.; Knudsen, G.R.; Carta, L.K.; Newcombe, G. Either low inoculum or a multi-trophic interaction can reduce the ability of Sclerotinia sclerotiorum to kill an invasive plant. Rhizosphere 2018, 5, 76–80. [Google Scholar] [CrossRef]
- De la Cruz, R.G.; Knudsen, G.R.; Dandurand, L.-M.C. Colonisation of sclerotia of Sclerotinia sclerotiorum by a fungivorous nematode. Biocontrol Sci. Technol. 2016, 26, 1166–1170. [Google Scholar] [CrossRef]
- Barnes, G. Aphelenchus avenae, a Potential biological control agent for root rot fungi. Plant Dis. 1981, 65, 423. [Google Scholar] [CrossRef]
- Knudsen, G.R.; Kim, T.G.; Bae, Y.-S.; Dandurand, L.M.C. Use of quantitative real-time pcr to unravel ecological complexity in a biological control system. Adv. Biosci. Biotechnol. 2015, 6, 237–244. [Google Scholar] [CrossRef][Green Version]
- Forghani, F.; Hajihassani, A. Recent advances in the development of environmentally benign treatments to control root-knot nematodes. Front. Plant Sci. 2020, 11, 1125. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Hussain, M.; Zhang, W.; Stadler, M.; Liu, X.; Xiang, M. Current insights into fungal species diversity and perspective on naming the environmental DNA sequences of fungi. Mycology 2019, 10, 127–140. [Google Scholar] [CrossRef] [PubMed]
- Hawksworth, D.; Lücking, R. Fungal diversity revisited: 2.2 to 3.8 million species. Microbiol. Spectr. 2017, 5, 79–95. [Google Scholar]
- Xu, J. Fungal species concepts in the genomics era. Genome 2020, 63, 459–468. [Google Scholar] [CrossRef]
- Toju, H.; Peay, K.; Yamamichi, M.; Narisawa, K.; Hiruma, K.; Naito, K.; Fukuda, S.; Ushio, M.; Nakaoka, S.; Onoda, Y.; et al. Core microbiomes for sustainable agroecosystems. Nat. Plants 2018, 4, 247–257. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, E.; Xiang, M.; Liu, X.; Chen, S. Population dynamics and biocontrol efficacy of the nematophagous fungus Hirsutella rhossiliensis as affected by stage of the soybean cyst nematode. Biol. Control 2008, 47, 244–249. [Google Scholar] [CrossRef]
- Liang, L.M.; Zou, C.G.; Xu, J.; Zhang, K.Q. Signal pathways involved in microbe—Nematode interactions provide new insights into the biocontrol of plant-parasitic nematodes. Philos. Trans. R. Soc. B Biol. Sci. 2019, 374, 20180317. [Google Scholar] [CrossRef]
- Escudero, N.; Lopez-Llorca, L. Effects on plant growth and root-knot nematode infection of an endophytic GFP transformant of the nematophagous fungus Pochonia chlamydosporia. Symbiosis 2012, 57, 33–42. [Google Scholar] [CrossRef]
- Luns, F.; Assis, R.; Silva, L.; Ferraz, C.; Braga, F.; Araújo, J. Coadministration of Nematophagous Fungi for Biological Control over Nematodes in Bovine in the South-Eastern Brazil. BioMed Res. Int. 2018, 2018, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Baron Cozentino, N.; Souza-Pollo, A.; Rigobelo, E. Purpureocillium lilacinum and Metarhizium marquandii as plant growth-promoting fungi. PeerJ 2020, 8, e9005. [Google Scholar] [CrossRef] [PubMed]
| Group | Representative Species | Host Recognition | Adhesion | Penetration | Digestion |
|---|---|---|---|---|---|
| Nematode-trapping fungi | A. oligospora | Mediated by lectin, proteins on the fungal surface interacting with sugar molecules on the nematode cuticle (GalNAc [57], AOL [58], AofleA [59], AoMad1 [60]), a nematode specific pheromone ascaroside [24], or olfactory mimicry that attracts nematode prey [29]. | A physical contact between the trap cells and the nematodes, cell-to-cell communication [61], a group of volatile organic compounds furanone, pyrone, and maltol [62], nitrate [63] and autophagy [64] are required for switching from the saprophytic to the pathogenic stage during trap formation and adhesion. | Fungi pierce the cuticle by forming a penetration tube, with a combination of mechanical pressure and extracellular hydrolytic enzymes, such as serine proteases (PII [65], Aoz1 [55], Ac1 [66], Ds1 [67] Dv1 [68], Mlx [69], Mc1 [70]), collagenase, and chitinase [55]. | Nematode content is converted to lipid droplets, these fungi obtain nutrients from the nematodes for their growth and reproduction. |
| Endoparasitic fungi | Drechmeria coniospora | Obligate parasites, using conidia that are ingested by their host, or by spores that adhere to the cuticle of the host [71]. | Adhesive conidia that adhere to the nematode cuticle will form an appressorium that presses firmly against the nematode cuticle. Motile zoospores encyst on the nematode’s surface and germinate to produce the injection tube, to infect nematodes by injecting a sporidium [37]. | A combination of enzymatic action and mechanical force, followed by vigorous growth of the trophic hyphae, to invade nematodes [72,73,74]. | New conidiophores develop from bulbs at the tips of trophic hyphae inside the cadaver, tightly pressed to the internal surface of the cuticle, preventing leakage of host nutrients, perturbing nematode metabolism, and causing nematode death [75]. |
| Egg- and cyst-parasitic fungi | Pochonia chlamydosporia | Aurovertin D showed strong toxicity and recognition of host [76]. | Glycoproteins and appressoria responsible for the adhesion of conidia and hyphae to the eggshell [77]. | Proteases and chitinases, e.g., PrC from Clonostachys rosea and Ver112 from Lecanicillium psalliotae [78]. | Colonizes the host tissues to obtain nutrients and uses available sugars in the egg as a carbon source [46]. |
| Toxin-producing fungi | Pleurotus ostreatus | Induces paralysis via the cilia of nematode sensory neurons [79]. | All developmental stages of C. elegans are sensitive to P. ostreatus. Nematodes become paralyzed upon contacting the P. ostreatus hyphae. | Excess calcium influx and hypercontraction of the head and pharyngeal muscle cells in nematodes. | Toxins cause rapid and systemic necrosis in multiple tissues throughout the organism. |
| Fungi producers of special nematode-attacking devices | Coprinus comatus; Stropharia rugosoannulata | Sharp projections of the special attack devices, mechanically damage the cuticle of the nematode [41,42]. | A penetration peg is formed and penetrates the nematode cuticle via mechanical forces and enzymatic activities. | Hyphae colonize the interior of the nematode and project themselves from the infected nematode. | Need toxin assistance to be successful in their nematicidal role (spiny balls). |
| Nematode | Pathogen | Crop/Insect | Reference |
|---|---|---|---|
| Steinernema diaprepesi | Fusarium solani | Wax moth, Weevil | [103] |
| Heterorhabditis bacteriophora, Steinernema feltiae, S. kraussei, | Metarhizium anisopliae | Black vine weevil | [104] |
| H. sonorensis | F. oxysporum | Corn earworm | [105] |
| Meloidogyne incognita | F. oxysporium f. sp. phaseoli | Bean | [106] |
| M. incognita | F. oxysporium f. sp. | Potato | [107] |
| M. incognita | Rhizoctonia solani | Green bean | [108] |
| M. incognita | Phytophthora capsici | Pepper | [109] |
| M. spp. | F. oxysporum f. sp. lycopersici | Tomato | [110] |
| M. spp. | F. oxysporum, F. solani | Tomato | [111] |
| M. javanica | F. oxysporum f. sp. lycopersici | Tomato | [112] |
| M. incognita | F. solani | Fig | [113] |
| M. incognita | F. oxysporum f. sp. niveum | Watermelon | [114,115] |
| M. incognita | Ralstonia solanacearum, Phomopsis vexans | Eggplant | [116] |
| M. incognita | Alternaria dauci, Rhizoctonia solani | Carrot | [117] |
| Pratylenchus spp., Trichodoridae, Heteroderidae | Rhizoctonia solani | Potato | [118] |
| S. feltiae, S. carpocapsae, H. bacteriophora, | Aspergillus spp., Penicillium spp. | Carob moth | [119] |
| S. diaprepesi | F. solani | Weevil | [120] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Li, S.; Li, H.; Wang, R.; Zhang, K.-Q.; Xu, J. Fungi–Nematode Interactions: Diversity, Ecology, and Biocontrol Prospects in Agriculture. J. Fungi 2020, 6, 206. https://doi.org/10.3390/jof6040206
Zhang Y, Li S, Li H, Wang R, Zhang K-Q, Xu J. Fungi–Nematode Interactions: Diversity, Ecology, and Biocontrol Prospects in Agriculture. Journal of Fungi. 2020; 6(4):206. https://doi.org/10.3390/jof6040206
Chicago/Turabian StyleZhang, Ying, Shuoshuo Li, Haixia Li, Ruirui Wang, Ke-Qin Zhang, and Jianping Xu. 2020. "Fungi–Nematode Interactions: Diversity, Ecology, and Biocontrol Prospects in Agriculture" Journal of Fungi 6, no. 4: 206. https://doi.org/10.3390/jof6040206
APA StyleZhang, Y., Li, S., Li, H., Wang, R., Zhang, K.-Q., & Xu, J. (2020). Fungi–Nematode Interactions: Diversity, Ecology, and Biocontrol Prospects in Agriculture. Journal of Fungi, 6(4), 206. https://doi.org/10.3390/jof6040206








