Azole-Resistant COVID-19-Associated Pulmonary Aspergillosis in an Immunocompetent Host: A Case Report
Abstract
1. Introduction
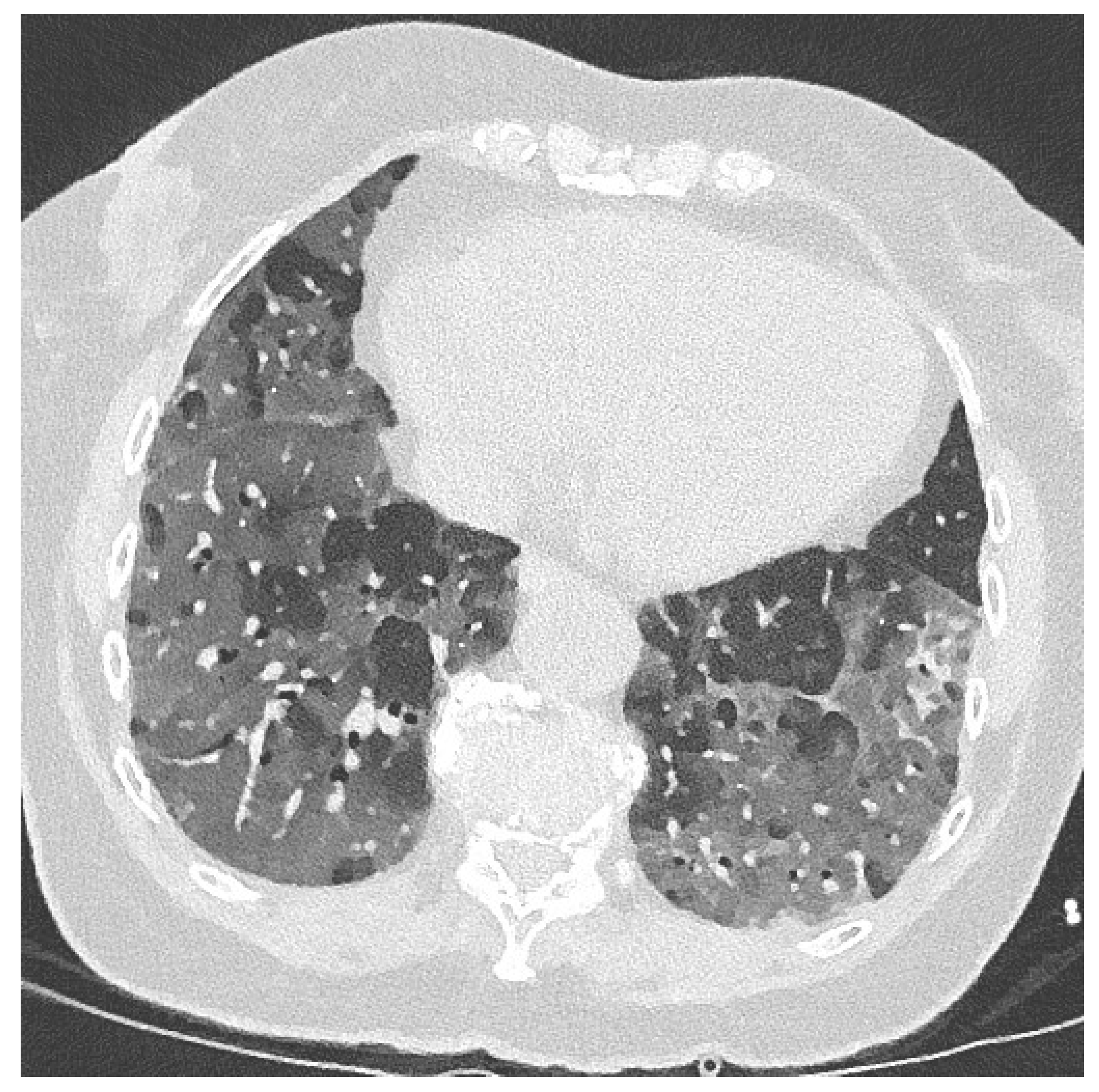
2. Case Report and Results
3. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Clancy, C.J.; Nguyen, M.H. COVID-19, superinfections and antimicrobial development: What can we expect? Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Verweij, P.E.; Gangneux, J.-P.; Bassetti, M.; Brüggemann, R.J.M.; Cornely, O.A.; Koehler, P.; Lass-Flörl, C.; van de Veerdonk, F.L.; Chakrabarti, A.; Hoenigl, M. Diagnosing COVID-19-associated pulmonary aspergillosis. Lancet Microbe 2020. [Google Scholar] [CrossRef]
- van Arkel, A.L.E.; Rijpstra, T.A.; Belderbos, H.N.A.; van Wijngaarden, P.; Verweij, P.E.; Bentvelsen, R.G. COVID-19 associated pulmonary aspergillosis. Am. J. Respir. Crit. Care Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Alanio, A.; Dellière, S.; Fodil, S.; Bretagne, S.; Mégarbane, B. High prevalence of putative invasive pulmonary aspergillosis in critically Ill COVID-19 patients. Lancet Respir. Med. 2020. [Google Scholar] [CrossRef]
- Koehler, P.; Cornely, O.A.; Bottiger, B.W.; Dusse, F.; Eichenauer, D.A.; Fuchs, F.; Hallek, M.; Jung, N.; Klein, F.; Persigehl, T.; et al. COVID-19 associated pulmonary aspergillosis. Mycoses 2020, 63, 528–534. [Google Scholar] [CrossRef]
- Marion, B.; Julien, M.; Cécile, N.; Alexandre, L.; Anne-Geneviève, M.; Marc, T.; Renaud, P.; Alexandre, D.; Arnaud, F. Fatal invasive aspergillosis and coronavirus disease in an immunocompetent patient. Emerg. Infect. Dis. 2020, 26. [Google Scholar] [CrossRef]
- Prattes, J.; Valentin, T.; Hoenigl, M.; Talakic, E.; Reisinger, A.C.; Eller, P. Invasive pulmonary aspergillosis complicating COVID-19 in the ICU - A case report. Med. Mycol. Case Rep. 2020. [Google Scholar] [CrossRef]
- Schauwvlieghe, A.; Rijnders, B.J.A.; Philips, N.; Verwijs, R.; Vanderbeke, L.; Van Tienen, C.; Lagrou, K.; Verweij, P.E.; Van de Veerdonk, F.L.; Gommers, D.; et al. Invasive aspergillosis in patients admitted to the intensive care unit with severe influenza: A retrospective cohort study. Lancet Respir. Med. 2018, 6, 782–792. [Google Scholar] [CrossRef]
- Buil, J.B.; Meijer, E.F.J.; Denning, D.W.; Verweij, P.E.; Meis, J.F. Burden of serious fungal infections in the Netherlands. Mycoses 2020, 63, 625–631. [Google Scholar] [CrossRef]
- Meis, J.F.; Chowdhary, A.; Rhodes, J.L.; Fisher, M.C.; Verweij, P.E. Clinical implications of globally emerging azole resistance in Aspergillus fumigatus. Philos. Trans. R Soc. Lond. B Biol. Sci. 2016, 371. [Google Scholar] [CrossRef]
- Prokop, M.; van Everdingen, W.; van Rees Vellinga, T.; Quarles van Ufford, J.; Stoger, L.; Beenen, L.; Geurts, B.; Gietema, H.; Krdzalic, J.; Schaefer-Prokop, C.; et al. CO-RADS - A categorical CT assessment scheme for patients with suspected COVID-19: Definition and evaluation. Radiology 2020, 201473. [Google Scholar] [CrossRef] [PubMed]
- Corman, V.M.; Landt, O.; Kaiser, M.; Molenkamp, R.; Meijer, A.; Chu, D.K.; Bleicker, T.; Brunink, S.; Schneider, J.; Schmidt, M.L.; et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill. 2020, 25. [Google Scholar] [CrossRef] [PubMed]
- Oostdijk, E.A.; De Jonge, E.; Kullberg, B.J.; Natsch, S.; De Smet, A.M.; Vandenbroucke-Grauls, C.M.J.E.; van der Vorm, E.; Bonten, M.J.M. SWAB-Richtlijn: Selectieve Decontaminatie bij Patiënten op de Intensive Care. 2018. Available online: https://swab.nl/nl/selectieve-decontaminatie-sdd (accessed on 22 May 2020).
- Vergidis, P.; Moore, C.B.; Novak-Frazer, L.; Rautemaa-Richardson, R.; Walker, A.; Denning, D.W.; Richardson, M.D. High-volume culture and quantitative real-time PCR for the detection of Aspergillus in sputum. Clin. Microbiol. Infect. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Ullmann, A.J.; Aguado, J.M.; Arikan-Akdagli, S.; Denning, D.W.; Groll, A.H.; Lagrou, K.; Lass-Flörl, C.; Lewis, R.E.; Munoz, P.; Verweij, P.E.; et al. Diagnosis and management of Aspergillus diseases: Executive summary of the 2017 ESCMID-ECMM-ERS guideline. Clin. Microbiol. Infect. 2018, 24 (Suppl. 1), e1–e38. [Google Scholar] [CrossRef] [PubMed]
- Chowdhary, A.; Kathuria, S.; Xu, J.P.; Meis, J.F. Emergence of azole-resistant Aspergillus fumigatus strains due to agricultural azole use creates an increasing threat to human health. PLoS Pathog. 2013, 9, e1003633. [Google Scholar] [CrossRef]
- Talento, A.F.; Dunne, K.; Murphy, N.; O’Connell, B.; Chan, G.; Joyce, E.A.; Hagen, F.; Meis, J.F.; Fahy, R.; Bacon, L.; et al. Post-influenzal triazole-resistant aspergillosis following allogeneic stem cell transplantation. Mycoses 2018, 61, 570–575. [Google Scholar] [CrossRef]
- Chowdhary, A.; Sharma, C.; Meis, J.F. Azole-resistant aspergillosis: Epidemiology, molecular mechanisms, and treatment. J. Infect. Dis. 2017, 216, S436–S444. [Google Scholar] [CrossRef]
- Lestrade, P.P.; Meis, J.F.; Melchers, W.J.; Verweij, P.E. Triazole resistance in Aspergillus fumigatus: Recent insights and challenges for patient management. Clin. Microbiol. Infect. 2019, 25, 799–806. [Google Scholar] [CrossRef]
- Ahmad, S.; Joseph, L.; Hagen, F.; Meis, J.F.; Khan, Z. Concomitant occurrence of itraconazole-resistant and -susceptible strains of Aspergillus fumigatus in routine cultures. J. Antimicrob. Chemother. 2015, 70, 412–415. [Google Scholar] [CrossRef]
- Singh, A.; Sharma, B.; Kumar Mahto, K.; Meis, J.F.; Chowdhary, A. High-frequency direct detection of triazole resistance in Aspergillus fumigatus from patients with chronic pulmonary fungal diseases in India. J. Fungi 2020, 6, 67. [Google Scholar] [CrossRef]
- Lestrade, P.P.; Buil, J.B.; van der Beek, M.T.; Kuijper, E.J.; van Dijk, K.; Kampinga, G.A.; Rijnders, B.J.A.; Vonk, A.G.; de Greeff, S.C.; Schoffelen, A.F.; et al. Paradoxal trends in azole-resistant Aspergillus fumigatus in a national multicenter surveillance program, 2013–2018. Emerg. Infect. Dis. 2020, 26. [Google Scholar] [CrossRef]
- Blot, S.; Rello, J.; Koulenti, D. Diagnosing invasive pulmonary aspergillosis in ICU patients: Putting the puzzle together. Curr. Opin. Crit. Care 2019, 25, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, J.P.; Chen, S.C.; Kauffman, C.A.; Steinbach, W.J.; Baddley, J.W.; Verweij, P.E.; Clancy, C.J.; Wingard, J.R.; Lockhart, S.R.; Groll, A.H.; et al. Revision and update of the consensus definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin. Infect. Dis. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Blot, S.I.; Taccone, F.S.; Van den Abeele, A.M.; Bulpa, P.; Meersseman, W.; Brusselaers, N.; Dimopoulos, G.; Paiva, J.A.; Misset, B.; Rello, J.; et al. A clinical algorithm to diagnose invasive pulmonary aspergillosis in critically ill patients. Am. J. Respir. Crit. Care Med. 2012, 186, 56–64. [Google Scholar] [CrossRef]
- Wahidi, M.M.; Lamb, C.; Murgu, S.; Musani, A.; Shojaee, S.; Sachdeva, A.; Maldonado, F.; Mahmood, K.; Kinsey, M.; Sethi, S.; et al. American Association for Bronchology and Interventional Pulmonology (AABIP) statement on the use of bronchoscopy and respiratory specimen collection in patients with suspected or confirmed COVID-19 infection. J. Bronchol. Interv. Pulmonol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Meersseman, W.; Lagrou, K.; Maertens, J.; Wilmer, A.; Hermans, G.; Vanderschueren, S.; Spriet, I.; Verbeken, E.; Van Wijngaerden, E. Galactomannan in bronchoalveolar lavage fluid: A tool for diagnosing aspergillosis in intensive care unit patients. Am. J. Respir. Crit. Care Med. 2008, 177, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Verweij, P.E.; Weemaes, C.M.; Curfs, J.H.; Bretagne, S.; Meis, J.F. Failure to detect circulating Aspergillus markers in a patient with chronic granulomatous disease and invasive aspergillosis. J. Clin. Microbiol. 2000, 38, 3900–3901. [Google Scholar] [CrossRef]
- Talento, A.F.; Dunne, K.; Joyce, E.A.; Palmer, M.; Johnson, E.; White, P.L.; Springer, J.; Loeffler, J.; Ryan, T.; Collins, D.; et al. A prospective study of fungal biomarkers to improve management of invasive fungal diseases in a mixed specialty critical care unit. J. Crit. Care 2017, 40, 119–127. [Google Scholar] [CrossRef]
- Acosta, J.; Catalan, M.; del Palacio-Peréz-Medel, A.; Lora, D.; Montejo, J.C.; Cuetara, M.S.; Moragues, M.D.; Ponton, J.; del Palacio, A. A prospective comparison of galactomannan in bronchoalveolar lavage fluid for the diagnosis of pulmonary invasive aspergillosis in medical patients under intensive care: Comparison with the diagnostic performance of galactomannan and of (1→3)-β-d-glucan chromogenic assay in serum samples. Clin. Microbiol. Infect. 2011, 17, 1053–1060. [Google Scholar] [CrossRef]
- Acosta, J.; Catalan, M.; Palacio-Pérez-Medel, A.; Montejo, J.; De-La-Cruz-Bértolo, J.; Moragues, M.; Pontón, J.; Finkelman, M.; Palacio, A. Prospective study in critically ill non-neutropenic patients: Diagnostic potential of (1,3)-β-D-glucan assay and circulating galactomannan for the diagnosis of invasive fungal disease. Eur. J. Clin. Microbiol. Infect. Dis. 2011, 31, 721–731. [Google Scholar] [CrossRef]
- Boch, T.; Reinwald, M.; Spiess, B.; Liebregts, T.; Schellongowski, P.; Meybohm, P.; Rath, P.M.; Steinmann, J.; Trinkmann, F.; Britsch, S.; et al. Detection of invasive pulmonary aspergillosis in critically ill patients by combined use of conventional culture, galactomannan, 1,3-β-D-glucan and Aspergillus specific nested polymerase chain reaction in a prospective pilot study. J. Crit. Care 2018, 47, 198–203. [Google Scholar] [CrossRef] [PubMed]
- De Vlieger, G.; Lagrou, K.; Maertens, J.; Verbeken, E.; Meersseman, W.; Van Wijngaerden, E. Beta-D-glucan detection as a diagnostic test for invasive aspergillosis in immunocompromised critically ill patients with symptoms of respiratory infection: An autopsy-based study. J. Clin. Microbiol. 2011, 49, 3783–3787. [Google Scholar] [CrossRef] [PubMed]
- Lahmer, T.; Neuenhahn, M.; Held, J.; Rasch, S.; Schmid, R.M.; Huber, W. Comparison of 1,3-β-d-glucan with galactomannan in serum and bronchoalveolar fluid for the detection of Aspergillus species in immunosuppressed mechanical ventilated critically ill patients. J. Crit. Care 2016, 36, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Vanderbeke, L.; Van Wijngaerden, E.; Maertens, J.; Wauters, J.; Lagrou, K. Diagnosis of invasive aspergillosis in intensive care unit patients. Curr. Fungal Infect. Rep. 2020. [Google Scholar] [CrossRef]
- Johnson, G.L.; Sarker, S.-J.; Nannini, F.; Ferrini, A.; Taylor, E.; Lass-Flörl, C.; Mutschlechner, W.; Bustin, S.A.; Agrawal, S.G. Aspergillus-specific lateral-flow device and real-time PCR testing of bronchoalveolar lavage fluid: A combination biomarker approach for clinical diagnosis of invasive pulmonary aspergillosis. J. Clin. Microbiol. 2015, 53, 2103–2108. [Google Scholar] [CrossRef] [PubMed]
- Hoenigl, M.; Prattes, J.; Spiess, B.; Wagner, J.; Prueller, F.; Raggam, R.B.; Posch, V.; Duettmann, W.; Hoenigl, K.; Wölfler, A.; et al. Performance of galactomannan, 1,3-β-d-glucan, Aspergillus lateral-flow device, conventional culture, and PCR tests with bronchoalveolar lavage fluid for diagnosis of invasive pulmonary aspergillosis. J. Clin. Microbiol. 2014, 52, 2039–2045. [Google Scholar] [CrossRef]
- Jenks, J.D.; Mehta, S.R.; Taplitz, R.; Aslam, S.; Reed, S.L.; Hoenigl, M. Point-of-care diagnosis of invasive aspergillosis in non-neutropenic patients: Aspergillus galactomannan lateral flow assay versus Aspergillus-specific lateral flow device test in bronchoalveolar lavage. Mycoses 2019, 62, 230–236. [Google Scholar] [CrossRef]
- Eigl, S.; Prattes, J.; Lackner, M.; Willinger, B.; Spiess, B.; Reinwald, M.; Selitsch, B.; Meilinger, M.; Neumeister, P.; Reischies, F.; et al. Multicenter evaluation of a lateral-flow device test for diagnosing invasive pulmonary aspergillosis in ICU patients. Crit. Care 2015, 19, 178. [Google Scholar] [CrossRef]

| Gender | Female | |
| Age (years) | 74 | |
| Medical history | Reflux, polyarthrosis, stopped smoking 20 years ago | |
| Medication | Pantoprazol (PPI) and Etoricoxib (NSAID) | |
| Underlying immuno-compromising condition | None | |
| Initial symptoms | Fever, dry cough, dyspneic, diarrhea | |
| ARDS | Prone positioning | Yes |
| vvECMO | No | |
| Acute renal failure | Yes, continuous venovenous hemofiltration (CVVH) | |
| IPA definition | EORTC/MSG criteria | N/A |
| (modified) AspICU algorithm | N/A | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meijer, E.F.J.; Dofferhoff, A.S.M.; Hoiting, O.; Buil, J.B.; Meis, J.F. Azole-Resistant COVID-19-Associated Pulmonary Aspergillosis in an Immunocompetent Host: A Case Report. J. Fungi 2020, 6, 79. https://doi.org/10.3390/jof6020079
Meijer EFJ, Dofferhoff ASM, Hoiting O, Buil JB, Meis JF. Azole-Resistant COVID-19-Associated Pulmonary Aspergillosis in an Immunocompetent Host: A Case Report. Journal of Fungi. 2020; 6(2):79. https://doi.org/10.3390/jof6020079
Chicago/Turabian StyleMeijer, Eelco F. J., Anton S. M. Dofferhoff, Oscar Hoiting, Jochem B. Buil, and Jacques F. Meis. 2020. "Azole-Resistant COVID-19-Associated Pulmonary Aspergillosis in an Immunocompetent Host: A Case Report" Journal of Fungi 6, no. 2: 79. https://doi.org/10.3390/jof6020079
APA StyleMeijer, E. F. J., Dofferhoff, A. S. M., Hoiting, O., Buil, J. B., & Meis, J. F. (2020). Azole-Resistant COVID-19-Associated Pulmonary Aspergillosis in an Immunocompetent Host: A Case Report. Journal of Fungi, 6(2), 79. https://doi.org/10.3390/jof6020079






