Histoplasmosis and Tuberculosis Co-Occurrence in People with Advanced HIV
Abstract
1. Introduction
2. Materials and Methods
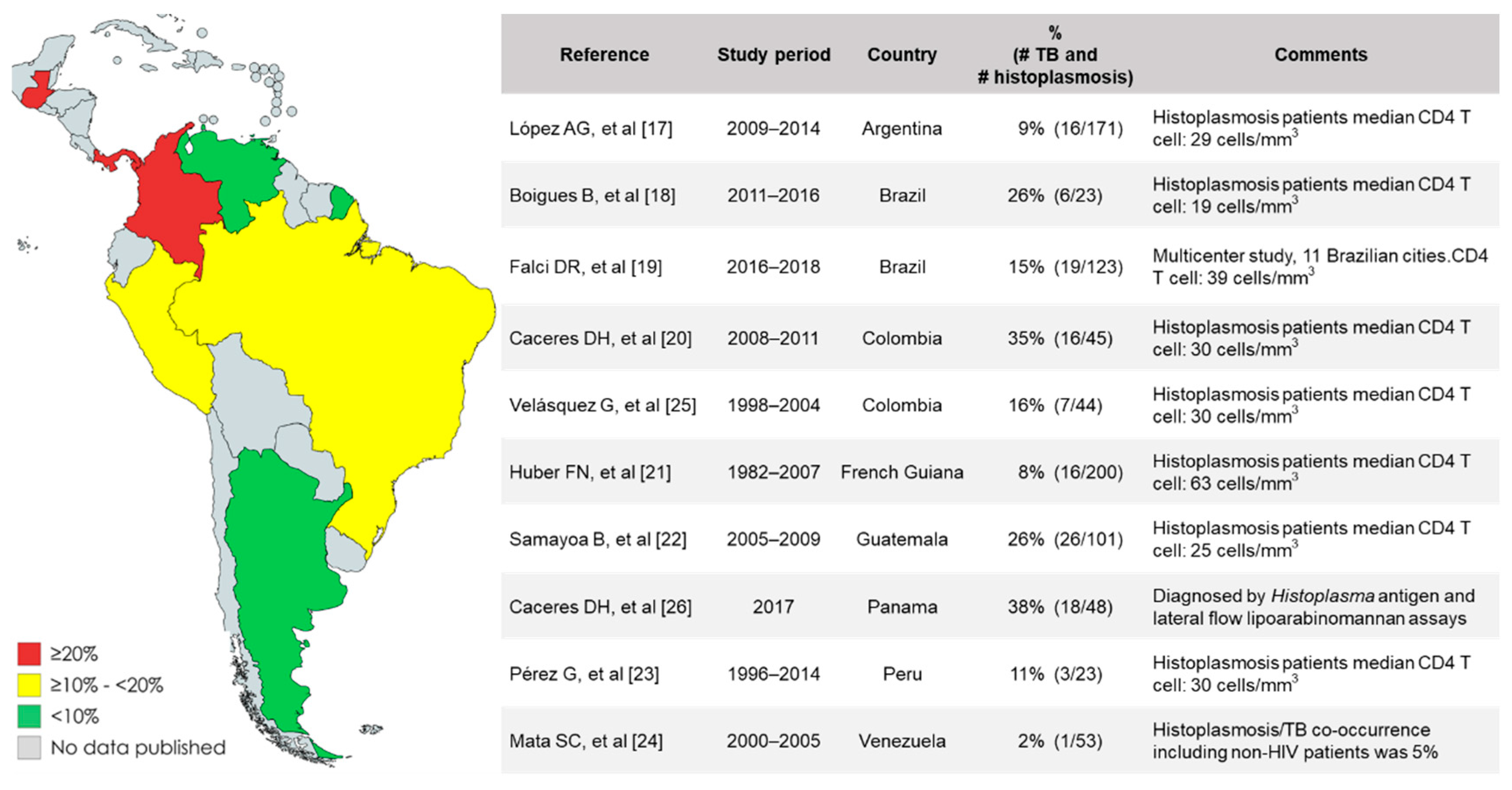
3. Epidemiology of Histoplasmosis/TB Co-Occurrence in People with Advanced HIV
4. Clinical Characteristics
5. Treatment of Histoplasmosis/TB Co-Occurrence
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). Global Tuberculosis Report 2018. Available online: https://www.who.int/tb/publications/global_report/en/ (accessed on 14 July 2019).
- Deepe, G.S. Histoplasma capsulatum (histoplasmosis). In Principles and Practice of Infectious Diseases, 8th ed.; Bennett, J.E., Dolin, R., Blaser, M.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 2949–2962. [Google Scholar]
- Edwards, P.Q.; Klaer, J.H.; Principe, A.; Convit, J.; Pifano, F.; Dodge, H.J. Worldwide geographic distribution of histoplasmosis and histoplasmin sensitivity. Am. J. Trop. Med. Hyg. 1956, 5, 235–257. [Google Scholar] [CrossRef] [PubMed]
- Oladele, R.O.; Ayanlowo, O.O.; Richardson, M.D.; Denning, D.W. Histoplasmosis in Africa: An emerging or a neglected disease? PLoS Negl. Trop. Dis. 2018, 12, e0006046. [Google Scholar]
- Bahr, N.C.; Sarosi, G.A.; Meya, D.B.; Bohjanen, P.R.; Richer, S.M.; Swartzentruber, S.; Halupnick, R.; Jarrett, D.; Wheat, L.J.; Boulware, D.R. Seroprevalence of histoplasmosis in Kampala, Uganda. Med. Mycol. 2016, 54, 295–300. [Google Scholar]
- Dodge, H.J.; Payne, M.W.; Whitehouse, W.M.; Bauman, K.A. Estimates of the Prevalence of Tuberculous and Histoplasmal Infections in a Sample of the Population of Tecumseh, Michigan, 1960. Am. Rev. Respir Dis. 1965, 92, 459–469. [Google Scholar] [PubMed]
- Macedo-Vinas, M.; Denning, D.W. Estimating the Burden of Serious Fungal Infections in Uruguay. J. Fungi 2018, 4, 37. [Google Scholar] [CrossRef] [PubMed]
- Medina, N.; Samayoa, B.; Lau-Bonilla, D.; Denning, D.W.; Herrera, R.; Mercado, D.; Guzmán, B.; Pérez, J.C.; Arathoon, E. Burden of serious fungal infections in Guatemala. Eur. J. Clin. Microbiol. Infect. Dis. 2017, 36, 965–969. [Google Scholar]
- Mandengue, C.E.; Ngandjio, A.; Atangana, P.J. Histoplasmosis in HIV-Infected Persons, Yaounde, Cameroon. Emerg. Infect. Dis. 2015, 21, 2094–2096. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.H.; Lin, H.H. Cases of histoplasmosis reported in Taiwan. J. Formos. Med. Assoc. 2006, 105, 527–528. [Google Scholar] [CrossRef]
- Adenis, A.A.; Valdes, A.; Cropet, C.; McCotter, O.Z.; Derado, G.; Couppie, P.; Chiller, T.; Nacher, M. Burden of HIV-associated histoplasmosis compared with tuberculosis in Latin America: A modelling study. Lancet Infect. Dis. 2018, 18, 1150–1159. [Google Scholar] [CrossRef]
- Souza, S.L.; Feitoza, P.V.; Araujo, J.R.; Andrade, R.V.; Ferreira, L.C. Causes of death among patients with acquired immunodeficiency syndrome autopsied at the Tropical Medicine Foundation of Amazonas. Rev. Soc. Bras. Med. Trop. 2008, 41, 247–251. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dominique, E.; Cerrillo, G.; Castro, C.; Ticona, E.; Morales, D.; Herrera, P.M.; Alfaro, A.; Cabanillas, J.; Barrantes, F.; Benavides, A.; et al. Resultados post mórtem e infecciones oportunistas en pacientes VIH-positivos de un hospital público del Perú. Rev. Peru. Med. Exp. Salud Publica 2006, 23, 270–274. [Google Scholar]
- Cáceres, D.H.; Gómez, B.L.; Restrepo, Á.; Tobón, Á.M. Histoplasmosis y sida: Factores de riesgo clínicos y de laboratorio asociados al pronóstico de la enfermedad. Infectio 2012, 16, 44–50. [Google Scholar]
- Gopalakrishnan, R.; Nambi, P.S.; Ramasubramanian, V.; Abdul Ghafur, K.; Parameswaran, A. Histoplasmosis in India: Truly uncommon or uncommonly recognised? J. Assoc. Physicians India 2012, 60, 25–28. [Google Scholar] [PubMed]
- Bulmer, A.C.; Bulmer, G.S. Incidence of histoplasmin hypersensitivity in the Philippines. Mycopathologia 2001, 149, 69–71. [Google Scholar] [CrossRef] [PubMed]
- López Daneri, A.G.; Arechavala, A.; Iovannitti, C.A.; Mujica, M.T. Histoplasmosis diseminada en pacientes HIV/SIDA: Buenos Aires, 2009–2014. Medicina 2016, 76, 332–337. [Google Scholar] [PubMed]
- Boigues, B.C.S.; Paniago, A.M.M.; Lima, G.M.E.; Nunes, M.O.; Uehara, S.N.O. Clinical outcomes and risk factors for death from disseminated histoplasmosis in patients with AIDS who visited a high-complexity hospital in Campo Grande, MS, Brazil. Rev. Soc. Bras. Med. Trop. 2018, 51, 155–161. [Google Scholar] [CrossRef]
- Falci, D.R.; Monteiro, A.A.; Braz Caurio, C.F.; Magalhaes, T.C.O.; Xavier, M.O.; Basso, R.P.; Melo, M.; Schwarzbold, A.V.; Ferreira, P.R.A.; Vidal, J.E.; et al. Histoplasmosis, An Underdiagnosed Disease Affecting People Living with HIV/AIDS in Brazil: Results of a Multicenter Prospective Cohort Study Using Both Classical Mycology Tests and Histoplasma Urine Antigen Detection. In Open Forum Infectious Diseases; Oxford University Press: Oxford, UK, 2019; Volume 6, p. ofz073. [Google Scholar]
- Caceres, D.H.; Tobon, A.M.; Cleveland, A.A.; Scheel, C.M.; Berbesi, D.Y.; Ochoa, J.; Restrepo, A.; Brandt, M.E.; Chiller, T.; Gómez, B.L.; et al. Clinical and Laboratory Profile of Persons Living with Human Immunodeficiency Virus/Acquired Immune Deficiency Syndrome and Histoplasmosis from a Colombian Hospital. Am. J. Trop. Med. Hyg. 2016, 95, 918–924. [Google Scholar] [CrossRef] [PubMed]
- Huber, F.; Nacher, M.; Aznar, C.; Pierre-Demar, M.; El Guedj, M.; Vaz, T.; Vincent, V.; Abba, M.; Christian, M.; Elodie, C.; et al. AIDS-related Histoplasma capsulatum var. capsulatum infection: 25 years experience of French Guiana. Aids 2008, 22, 1047–1053. [Google Scholar] [CrossRef] [PubMed]
- Samayoa, B.; Roy, M.; Cleveland, A.A.; Medina, N.; Lau-Bonilla, D.; Scheel, C.M.; Gomez, B.L.; Chiller, T.; Arathoon, E. High Mortality and Coinfection in a Prospective Cohort of Human Immunodeficiency Virus/Acquired Immune Deficiency Syndrome Patients with Histoplasmosis in Guatemala. Am. J. Trop. Med. Hyg. 2017, 97, 42–48. [Google Scholar] [CrossRef]
- Perez-Lazo, G.; Maquera-Afaray, J.; Mejia, C.R.; Castillo, R. Disseminated histoplasmosis and HIV infection: Case series in a Peruvian hospital. Rev. Chilena Infectol. 2017, 34, 365–369. [Google Scholar]
- Mata-Essayag, S.; Colella, M.T.; Rosello, A.; de Capriles, C.H.; Landaeta, M.E.; de Salazar, C.P.; Sylvia, M.; Carolina, O.; Maria, C.I.; Liseloth, G. Histoplasmosis: A study of 158 cases in Venezuela, 2000–2005. Medicine 2008, 87, 193–202. [Google Scholar] [CrossRef]
- Uribe, G.V.; Rueda, Z.V.; Vélez, L.A.; Aguirre, D.C.; Gómez-Arias, R.D. Histoplasmosis en pacientes con sida. Un estudio de cohorte en Medellín, Colombia. Infectio 2010, 14, 99–106. [Google Scholar]
- Caceres, D.H.; Arauz, A.B.; Flores, C.; Santiago, E.; Gutiérrez, E.; Borace, J.; Abdo, J.; Lindsley, M.; Flores, R.; Stolz-Sobalvarro, E.; et al. Tuberculosis and histoplasmosis diagnosis among people living with HIV/AIDS: The impact of using rapid diagnostics tests in Panama. In Proceedings of the 20th Congress of the International Society for Human and Animal Mycology, Amsterdam, The Netherlands, 4 June 2018. [Google Scholar]
- Kauffman, C.A. Diagnosis of histoplasmosis in immunosuppressed patients. Curr. Opin. Infect. Dis. 2008, 21, 421–425. [Google Scholar] [CrossRef]
- Sacarlal, J.; Denning, D.W. Estimated Burden of Serious Fungal Infections in Mozambique. J. Fungi 2018, 4, 75. [Google Scholar] [CrossRef] [PubMed]
- Salahuddin, M.; Karanth, S.; Ocazionez, D.; Estrada, Y.M.R.M.; Cherian, S.V. Clinical Characteristics and Etiologies of Miliary Nodules in the US: A Single-Center Study. Am. J. Med. 2019, 132, 767–769. [Google Scholar] [CrossRef] [PubMed]
- Aaron, L.; Saadoun, D.; Calatroni, I.; Launay, O.; Memain, N.; Vincent, V.; Marchal, G.; Dupont, B.; Bouchaud, O.; Valeyre, D.; et al. Tuberculosis in HIV-infected patients: A comprehensive review. Clin. Microbiol. Infect. 2004, 10, 388–398. [Google Scholar] [CrossRef] [PubMed]
- Swaminathan, S.; Padmapriyadarsini, C.; Narendran, G. HIV-associated tuberculosis: Clinical update. Clin. Infect. Dis. 2010, 50, 1377–1386. [Google Scholar] [CrossRef] [PubMed]
- Garzona-Schmidt, R.; Solano-Chinchilla, A. Úlceras Colónicas por TB e Histoplasmosis en un paciente portador de VIH/Sida. Acta Méd. Costarric. 2001, 43, 138–140. [Google Scholar]
- Greene, L.; Peters, B.; Lucas, S.B.; Pozniak, A.L. Extrapulmonary tuberculosis masking disseminated histoplasmosis in AIDS. Sex. Transm. Infect. 2000, 76, 54–56. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.W.; Sohn, J.W.; Kim, M.J.; Choi, J.W.; Kim, C.H.; Choi, S.H.; Kim, J.; Cho, Y. Disseminated histoplasmosis and tuberculosis in a patient with HIV infection. Yonsei Med. J. 2007, 48, 531–534. [Google Scholar] [CrossRef] [PubMed]
- Piscoya Rivera, A.; Cedron Cheng, H.; Huerta-Mercado Tenorio, J.; Pinto Valdivia, J.L.; Ferrufino Llach, J.C.; Bussalleu Rivera, A. GI tuberculosis and histoplasmosis in an HIV (+) patient presenting with lower GI bleeding. Am. J. Gastroenterol. 2005, 100, 1896–1897. [Google Scholar] [PubMed]
- Rezende, R.E.; Brunaldi, M.O.; Girao, M.S.; Zucoloto, S.; Garcia, S.B.; Machado, A.A.; Módena, J.L.P. Case report: Esophageal histoplasmosis associated with disseminated tuberculosis in acquired immunodeficiency syndrome. Am. J. Trop. Med. Hyg. 2009, 80, 347–350. [Google Scholar] [CrossRef] [PubMed]
- Salzman, S.H.; Smith, R.L.; Aranda, C.P. Histoplasmosis in patients at risk for the acquired immunodeficiency syndrome in a nonendemic setting. Chest 1988, 93, 916–921. [Google Scholar] [CrossRef]
- Adenis, A.; Nacher, M.; Hanf, M.; Basurko, C.; Dufour, J.; Huber, F.; Aznar, C.; Carme, B.; Couppie, P. Tuberculosis and histoplasmosis among human immunodeficiency virus-infected patients: A comparative study. Am. J. Trop. Med. Hyg. 2014, 90, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, D.W.; Sterling, T.R.; Haas, D.W. Mycobacterium tuberculosis. In Principles and Practice of Infectious Diseases, 8th ed.; Bennett, J.E., Dolin, R., Blaser, M.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 2234–2236. [Google Scholar]
- Furin, J.; Cox, H.; Pai, M. Tuberculosis. Lancet 2019, 393, 1642–1656. [Google Scholar] [CrossRef]
- Hage, C.A.; Azar, M.M.; Bahr, N.; Loyd, J.; Wheat, L.J. Histoplasmosis: Up-to-Date Evidence-Based Approach to Diagnosis and Management. Semin. Respir Crit. Care Med. 2015, 36, 729–745. [Google Scholar] [PubMed]
- De Pauw, B.; Walsh, T.J.; Donnelly, J.P.; Stevens, D.A.; Edwards, J.E.; Calandra, T.; Pappas, P.G.; Maertens, J.; Lortholary, O.; Kauffman, C.A.; et al. Revised definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) Consensus Group. Clin. Infect. Dis. 2008, 46, 1813–1821. [Google Scholar] [PubMed]
- Messina, F.A.; Corti, M.; Negroni, R.; Arechavala, A.; Bianchi, M.; Santiso, G. Histoplasmosis in AIDS patients without tegumentary manifestations. Rev. Chilena Infectol. 2018, 35, 560–565. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, M.; Robles, A.M.; Vitale, R.; Helou, S.; Arechavala, A.; Negroni, R. The usefulness of blood culture in diagnosing HIV-related systemic mycoses: Evaluation of a manual lysis centrifugation method. Med. Mycol. 2000, 38, 77–80. [Google Scholar]
- Crump, J.A.; Reller, L.B. Two decades of disseminated tuberculosis at a university medical center: The expanding role of mycobacterial blood culture. Clin. Infect. Dis. 2003, 37, 1037–1043. [Google Scholar]
- Ker, C.C.; Hung, C.C.; Huang, S.Y.; Chen, M.Y.; Hsieh, S.M.; Lin, C.C.; Chang, C.C.; Luh, K.T. Comparison of bone marrow studies with blood culture for etiological diagnosis of disseminated mycobacterial and fungal infection in patients with acquired immunodeficiency syndrome. J. Microbiol. Immunol. Infect. 2002, 35, 89–93. [Google Scholar] [PubMed]
- Caceres, D.H.; Scheel, C.M.; Tobon, A.M.; Ahlquist Cleveland, A.; Restrepo, A.; Brandt, M.E.; Chiller, T.; Gómez, B.L. Validation of an enzyme-linked immunosorbent assay that detects Histoplasma capsulatum antigenuria in Colombian patients with AIDS for diagnosis and follow-up during therapy. Clin. Vaccine Immunol. 2014, 21, 1364–1368. [Google Scholar] [PubMed]
- Rubio-Carrasquilla, M.; Santa, C.D.; Rendon, J.P.; Botero-Garces, J.; Guimaraes, A.J.; Moreno, E.; Cano, L.E. An interferon gamma release assay specific for Histoplasma capsulatum to detect asymptomatic infected individuals. Med. Mycol. 2019. Epub ahead of print. [Google Scholar]
- Fandino-Devia, E.; Rodriguez-Echeverri, C.; Cardona-Arias, J.; Gonzalez, A. Antigen Detection in the Diagnosis of Histoplasmosis: A Meta-analysis of Diagnostic Performance. Mycopathologia 2016, 181, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Caceres, D.H.; Samayoa, B.E.; Medina, N.G.; Tobon, A.M.; Guzman, B.J.; Mercado, D.; Restrepo, A.; Chiller, T.; Arathoon, E.E.; Gómez, B.L. Multicenter Validation of Commercial Antigenuria Reagents To Diagnose Progressive Disseminated Histoplasmosis in People Living with HIV/AIDS in Two Latin American Countries. J. Clin. Microbiol. 2018, 56, e01959. [Google Scholar]
- Caceres, D.H.; Minderman, M.C.; Chaney, L.; Gomez, B.L.; Wheat, L.J.; Chiller, T.M.; Lindsley, M.D. Evaluation of a Histoplasma antigen Lateral Flow Assay for the rapid diagnosis of progressive disseminated histoplasmosis in AIDS patie. In Proceedings of the 20th Congress of the International Society for Human and Animal Mycology, Amsterdam, The Netherlands, 4 June 2018. [Google Scholar]
- Caceres, D.H.; Zuluaga, A.; Arango-Bustamante, K.; de Bedout, C.; Tobon, A.M.; Restrepo, A.; Gómez, B.L.; Cano, L.E.; González, Á. Implementation of a Training Course Increased the Diagnosis of Histoplasmosis in Colombia. Am. J. Trop. Med. Hyg. 2015, 93, 662–667. [Google Scholar] [CrossRef] [PubMed]
- Bansal, N.; Sethuraman, N.; Gopalakrishnan, R.; Ramasubramanian, V.; Kumar, D.S.; Nambi, P.S.; Chakrabarti, A. Can urinary histoplasma antigen test improve the diagnosis of histoplasmosis in a tuberculosis endemic region? Mycoses 2019, 62, 502–507. [Google Scholar] [CrossRef] [PubMed]
- Whitworth, H.S.; Badhan, A.; Boakye, A.A.; Takwoingi, Y.; Rees-Roberts, M.; Partlett, C.; Lambie, H.; JohnInnes, F.R.C.P.; Cooke, G.; Lipman, M.M.D. Clinical utility of existing and second-generation interferon-gamma release assays for diagnostic evaluation of tuberculosis: An observational cohort study. Lancet Infect. Dis. 2019, 19, 193–202. [Google Scholar] [CrossRef]
- WHO. The Use of Lateral Flow Urine Lipoarabinomannan Assay (LF-LAM) for the Diagnosis and Screening of Active Tuberculosis in People Living with HIV 2015. Available online: https://www.who.int/tb/publications/use-of-lf-lam-tb-hiv/en/ (accessed on 14 July 2019).
- Buitrago, M.J.; Canteros, C.E.; Frias De Leon, G.; Gonzalez, A.; Marques-Evangelista De Oliveira, M.; Munoz, C.O.; Ramirez, J.A.; Toranzo, A.I.; Zancope-Oliveira, R.; Cuenca-Estrella, M. Comparison of PCR protocols for detecting Histoplasma capsulatum DNA through a multicenter study. Rev. Iberoam Micol. 2013, 30, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Munoz, C.; Gomez, B.L.; Tobon, A.; Arango, K.; Restrepo, A.; Correa, M.M.; Muskus, C.; Cano, L.E.; González, A. Validation and clinical application of a molecular method for identification of Histoplasma capsulatum in human specimens in Colombia, South America. Clin. Vaccine Immunol. 2010, 17, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Buitrago, M.J.; Gomez-Lopez, A.; Monzon, A.; Rodriguez-Tudela, J.L.; Cuenca-Estrella, M. Assessment of a quantitative PCR method for clinical diagnosis of imported histoplasmosis. Enferm. Infecc. Microbiol. Clin. 2007, 25, 16–22. [Google Scholar] [PubMed]
- Buitrago, M.J.; Berenguer, J.; Mellado, E.; Rodriguez-Tudela, J.L.; Cuenca-Estrella, M. Detection of imported histoplasmosis in serum of HIV-infected patients using a real-time PCR-based assay. Eur. J. Clin. Microbiol. Infect. Dis. 2006, 25, 665–668. [Google Scholar] [CrossRef] [PubMed]
- Maubon, D.; Simon, S.; Aznar, C. Histoplasmosis diagnosis using a polymerase chain reaction method. Application on human samples in French Guiana, South America. Diagn. Microbiol. Infect. Dis. 2007, 58, 441–444. [Google Scholar] [CrossRef] [PubMed]
- Toranzo, A.I.; Tiraboschi, I.N.; Fernandez, N.; Ibarra-Camou, B.; Rivas, M.C.; Lee, W.; Davel, G.; Canteros, C.E. Molecular diagnosis of human histoplasmosis in whole blood samples. Rev. Argent. Microbiol. 2009, 41, 20–26. [Google Scholar] [PubMed]
- Gago, S.; Esteban, C.; Valero, C.; Zaragoza, O.; Puig de la Bellacasa, J.; Buitrago, M.J. A multiplex real-time PCR assay for identification of Pneumocystis jirovecii, Histoplasma capsulatum, and Cryptococcus neoformans/Cryptococcus gattii in samples from AIDS patients with opportunistic pneumonia. J. Clin. Microbiol. 2014, 52, 1168–1176. [Google Scholar] [CrossRef] [PubMed]
- Kolia-Diafouka, P.; Carrere-Kremer, S.; Lounnas, M.; Bourdin, A.; Kremer, L.; Van de Perre, P.; Godreuil, S.; Tuaillon, E. Detection of Mycobacterium tuberculosis in paucibacillary sputum: Performances of the Xpert MTB/RIF ultra compared to the Xpert MTB/RIF, and IS6110 PCR. Diagn. Microbiol. Infect. Dis. 2019, 94, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Opota, O.; Mazza-Stalder, J.; Greub, G.; Jaton, K. The rapid molecular test Xpert MTB/RIF ultra: Towards improved tuberculosis diagnosis and rifampicin resistance detection. Clin. Microbiol. Infect. 2019. [Google Scholar] [CrossRef] [PubMed]
- WHO. World Health Organization Model List of Essential In Vitro Diagnostics First edition 2018. Available online: https://www.who.int/medical_devices/diagnostics/WHO_EDL_2018.pdf (accessed on 14 July 2019).
- WHO. Second WHO Model List of Essential In Vitro Diagnostics Second edition 2019. Available online: https://www.who.int/medical_devices/publications/Standalone_document_v8.pdf?ua=1 (accessed on 14 July 2019).
- WHO. Guidelines for Managing Advanced HIV Disease and Rapid Initiation of Antiretroviral Therapy 2017. Available online: https://www.who.int/hiv/pub/guidelines/advanced-HIV-disease/en/ (accessed on 14 July 2019).
- Wheat, L.J.; Freifeld, A.G.; Kleiman, M.B.; Baddley, J.W.; McKinsey, D.S.; Loyd, J.E.; Kauffman, C.A.; Infectious Diseases Society of America. Clinical practice guidelines for the management of patients with histoplasmosis: 2007 update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2007, 45, 807–825. [Google Scholar] [PubMed]
- Agudelo, C.A.; Restrepo, C.A.; Molina, D.A.; Tobon, A.M.; Kauffman, C.A.; Murillo, C.; Restrepo, A. Tuberculosis and histoplasmosis co-infection in AIDS patients. Am. J. Trop. Med. Hyg. 2012, 87, 1094–1098. [Google Scholar]
- Melzani, A.; De Reynal De Saint Michel, R.; Ntab, B.; Djossou, F.; Epelboin, L.; Nacher, M.; Blanchet, D.; Demar, M.; Couppie, P.; Adenis, A. Incidence and trends in immune reconstitution inflammatory syndrome associated with Histoplasma capsulatum among people living with HIV: A 20-year case series and literature review. Clin. Infect. Dis. 2019. [Google Scholar] [CrossRef] [PubMed]
| Itraconazole drug interactions | Antibacterial for TB treatment: • Rifampicin and rifabutin may decrease itraconazole blood levels • Itraconazole may increase blood levels of rifabutin Antiretroviral: • Efavirenz (EFV) and nevirapine (NVP) reduce itraconazole blood levels • Lopinavir/ritonavir (LPV/r) and atazanavir/ritonavir (ATV/r) increase itraconazole blood levels Monitoring itraconazole blood levels. |
| Recommendations for treatment of progressive disseminated histoplasmosis (Infectious Diseases Society of America (71) guidelines) | Moderately severe to severe disease: • Liposomal amphotericin B (3.0 mg/kg daily, 1–2 weeks), followed by oral itraconazole (200 mg 3 times daily for 3 days and then 200 mg twice daily for a total of at least 12 months in disseminated form) [68]. For mild to moderate disease: • Itraconazole (200 mg 3 times daily for 3 days and then twice daily for at least 12 months) [68]. Blood levels of itraconazole and Histoplasma antigen concentrations (sera or urine) should be measured during therapy. Monitor Histoplasma antigen concentrations for 12 months after end of therapy with the aim of early identification of histoplasmosis relapse [68]. |
| Treatment of histoplasmosis/TB co-occurrence | At the time of this review, there were no guidelines for the treatment of histoplasmosis/TB co-occurrence. A report from Colombia used fluoroquinolone as an alternative agent in place of rifampicin for tuberculosis [69]. This was a descriptive study with evidence of a very low certainty. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caceres, D.H.; Valdes, A. Histoplasmosis and Tuberculosis Co-Occurrence in People with Advanced HIV. J. Fungi 2019, 5, 73. https://doi.org/10.3390/jof5030073
Caceres DH, Valdes A. Histoplasmosis and Tuberculosis Co-Occurrence in People with Advanced HIV. Journal of Fungi. 2019; 5(3):73. https://doi.org/10.3390/jof5030073
Chicago/Turabian StyleCaceres, Diego H., and Audrey Valdes. 2019. "Histoplasmosis and Tuberculosis Co-Occurrence in People with Advanced HIV" Journal of Fungi 5, no. 3: 73. https://doi.org/10.3390/jof5030073
APA StyleCaceres, D. H., & Valdes, A. (2019). Histoplasmosis and Tuberculosis Co-Occurrence in People with Advanced HIV. Journal of Fungi, 5(3), 73. https://doi.org/10.3390/jof5030073





