The Multifaceted Role of T-Helper Responses in Host Defense against Aspergillus fumigatus
Abstract
1. Introduction
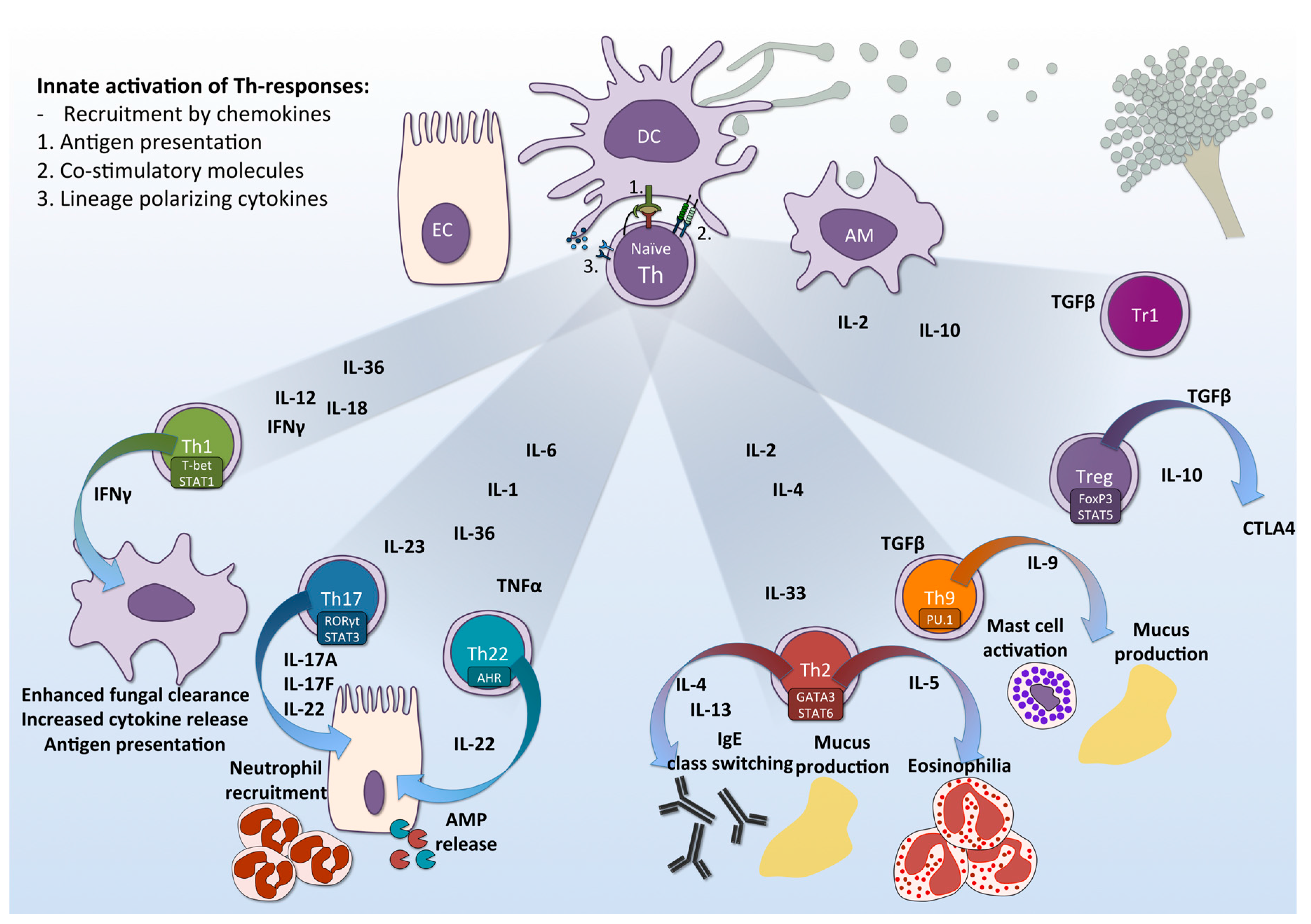
2. Induction of T-Helper Responses by the Innate Immune System
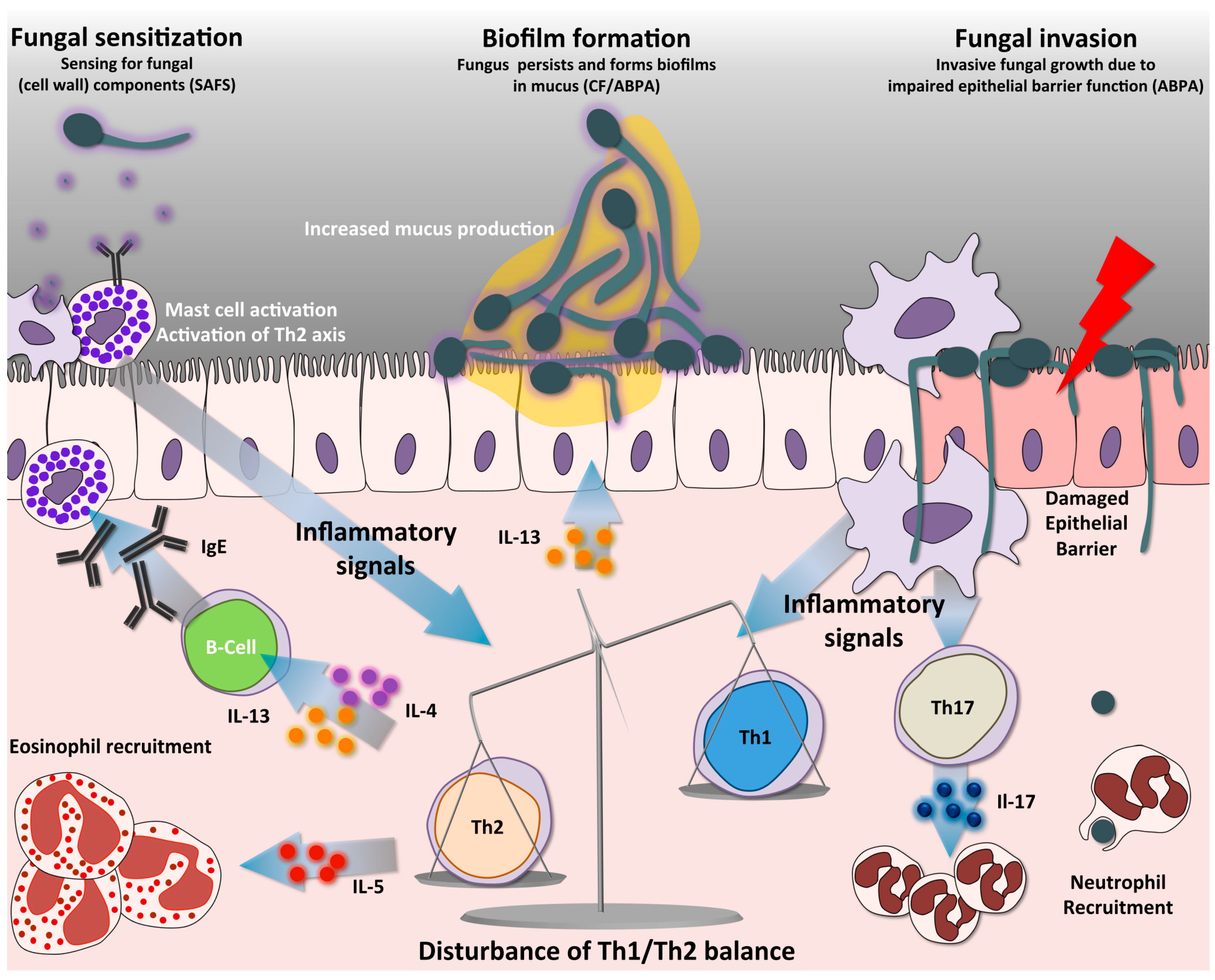
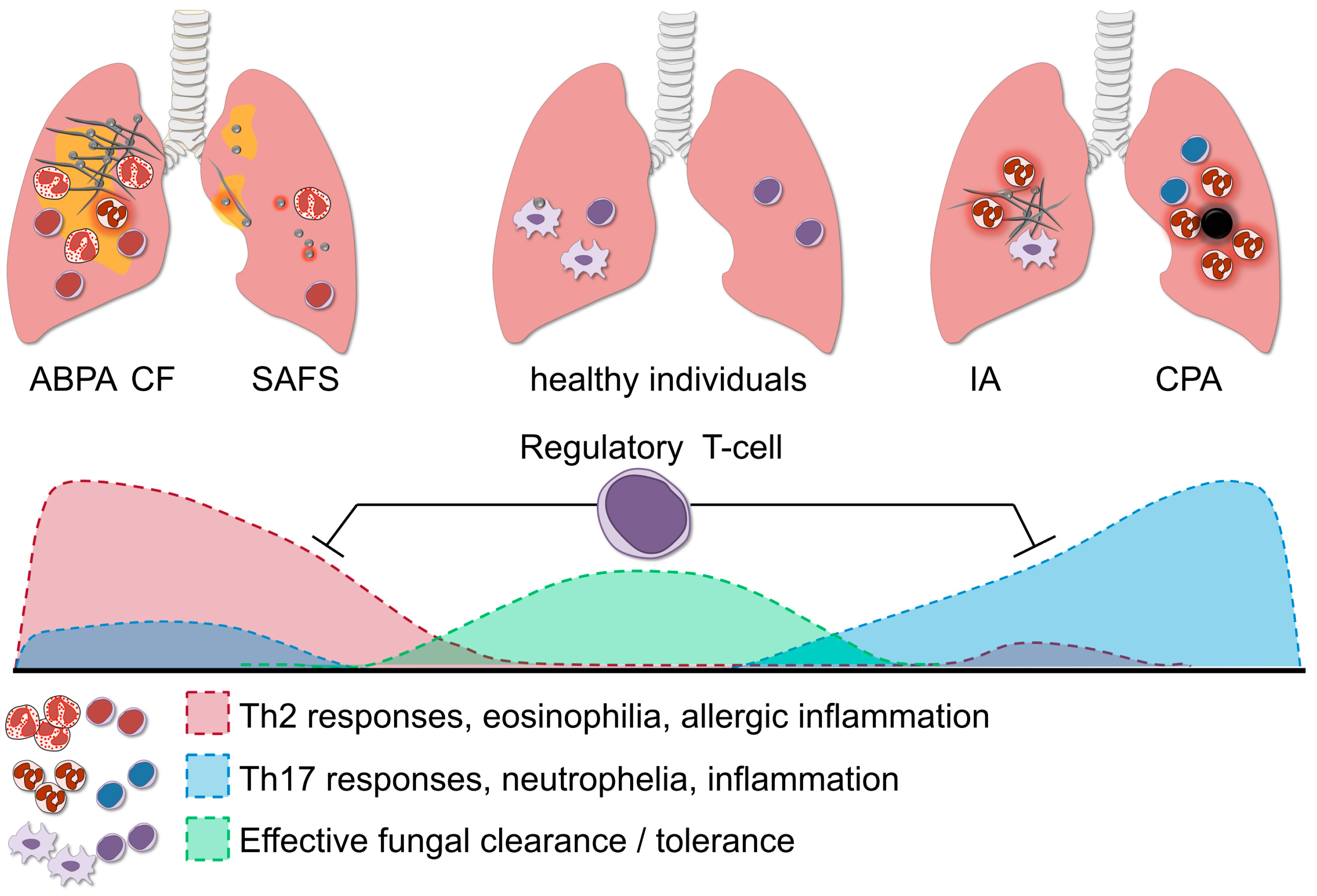
3. T-Helper Responses in Aspergillosis
4. The Two Faces of the Th17 Response
5. Th2 Response Drives Allergic Hypersensitivity Reactions to Aspergillus
6. Regulatory T-Cells
7. Immunotherapy: Optimizing the Potential of T-Helper Responses
8. Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gavalda, J.; Len, O.; San Juan, R.; Aguado, J.M.; Fortun, J.; Lumbreras, C.; Moreno, A.; Munoz, P.; Blanes, M.; Ramos, A.; et al. Risk factors for invasive aspergillosis in solid-organ transplant recipients: A case-control study. Clin. Infect. Dis. 2005, 41, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Fishman, J.A. Infection in solid-organ transplant recipients. N. Engl. J. Med. 2007, 357, 2601–2614. [Google Scholar] [CrossRef] [PubMed]
- Stuehler, C.; Kuenzli, E.; Jaeger, V.K.; Baettig, V.; Ferracin, F.; Rajacic, Z.; Kaiser, D.; Bernardini, C.; Forrer, P.; Weisser, M.; et al. Immune reconstitution after allogeneic hematopoietic stem cell transplantation and association with occurrence and outcome of invasive aspergillosis. J. Infect. Dis. 2015, 212, 959–967. [Google Scholar] [CrossRef] [PubMed]
- Lionakis, M.S.; Kontoyiannis, D.P. Glucocorticoids and invasive fungal infections. Lancet 2003, 362, 1828–1838. [Google Scholar] [CrossRef]
- Todeschini, G.; Murari, C.; Bonesi, R.; Pizzolo, G.; Verlato, G.; Tecchio, C.; Meneghini, V.; Franchini, M.; Giuffrida, C.; Perona, G.; et al. Invasive aspergillosis in neutropenic patients: Rapid neutrophil recovery is a risk factor for severe pulmonary complications. Eur. J. Clin. Investig. 1999, 29, 453–457. [Google Scholar] [CrossRef]
- Marr, K.A.; Carter, R.A.; Boeckh, M.; Martin, P.; Corey, L. Invasive aspergillosis in allogeneic stem cell transplant recipients: Changes in epidemiology and risk factors. Blood 2002, 100, 4358–4366. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, S.; Dulery, R.; Regany, K.; Bouketouche, M.; Magro, L.; Coiteux, V.; Alfandari, S.; Berthon, C.; Quesnel, B.; Yakoub-Agha, I. Long-term follow up of invasive aspergillosis in allogeneic stem cell transplantation recipients and leukemia patients: Differences in risk factors and outcomes. Curr. Res. Transl. Med. 2017, 65, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Gresnigt, M.S.; Netea, M.G.; van de Veerdonk, F.L. Pattern recognition receptors and their role in invasive aspergillosis. Ann. N. Y. Acad. Sci. 2012, 1273, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Camargo, J.F.; Husain, S. Immune correlates of protection in human invasive aspergillosis. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2014, 59, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, V.; Rivera, A. First line of defense: Innate cell-mediated control of pulmonary aspergillosis. Front. Microbiol. 2016, 7, 272. [Google Scholar] [CrossRef] [PubMed]
- Heinekamp, T.; Schmidt, H.; Lapp, K.; Pahtz, V.; Shopova, I.; Koster-Eiserfunke, N.; Kruger, T.; Kniemeyer, O.; Brakhage, A.A. Interference of Aspergillus fumigatus with the immune response. Semin. Immunopathol. 2015, 37, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Hohl, T.M.; Feldmesser, M. Aspergillus fumigatus: Principles of pathogenesis and host defense. Eukaryot. Cell 2007, 6, 1953–1963. [Google Scholar] [CrossRef] [PubMed]
- Morton, C.O.; Bouzani, M.; Loeffler, J.; Rogers, T.R. Direct interaction studies between Aspergillus fumigatus and human immune cells; what have we learned about pathogenicity and host immunity? Front. Microbiol. 2012, 3, 413. [Google Scholar] [CrossRef] [PubMed]
- Garth, J.M.; Steele, C. Innate lung defense during invasive aspergillosis: New mechanisms. J. Innate Immun. 2017, 9, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Kolls, J.K. T cell-mediated host immune defenses in the lung. Annu. Rev. Immunol. 2013, 31, 605–633. [Google Scholar] [CrossRef] [PubMed]
- Romani, L. Immunity to fungal infections. Nat. Rev. Immunol. 2004, 4, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Osherov, N. Interaction of the pathogenic mold Aspergillus fumigatus with lung epithelial cells. Front. Microbiol. 2012, 3, 346. [Google Scholar] [CrossRef] [PubMed]
- Saenz, S.A.; Taylor, B.C.; Artis, D. Welcome to the neighborhood: Epithelial cell-derived cytokines license innate and adaptive immune responses at mucosal sites. Immunol. Rev. 2008, 226, 172–190. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, J.K.; Mulligan, R.M.; Atkinson, C.; Schlosser, R.J. Human sinonasal epithelial cells direct dendritic function and T-cell T helper 1/T helper 2 skewing following aspergillus exposure. Int. Forum. Allergy Rhinol. 2011, 1, 268–274. [Google Scholar] [CrossRef] [PubMed]
- De Luca, A.; Bozza, S.; Zelante, T.; Zagarella, S.; D’Angelo, C.; Perruccio, K.; Vacca, C.; Carvalho, A.; Cunha, C.; Aversa, F.; et al. Non-hematopoietic cells contribute to protective tolerance to Aspergillus fumigatus via a trif pathway converging on ido. Cell. Mol. Immunol. 2010, 7, 459–470. [Google Scholar] [CrossRef] [PubMed]
- Steele, C.; Rapaka, R.R.; Metz, A.; Pop, S.M.; Williams, D.L.; Gordon, S.; Kolls, J.K.; Brown, G.D. The beta-glucan receptor dectin-1 recognizes specific morphologies of Aspergillus fumigatus. PLoS Pathog. 2005, 1, e42. [Google Scholar] [CrossRef] [PubMed]
- Hohl, T.M.; Rivera, A.; Lipuma, L.; Gallegos, A.; Shi, C.; Mack, M.; Pamer, E.G. Inflammatory monocytes facilitate adaptive CD4 T cell responses during respiratory fungal infection. Cell Host Microbe 2009, 6, 470–481. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Ortiz, Z.G.; Means, T.K. The role of dendritic cells in the innate recognition of pathogenic fungi (A. fumigatus, C. neoformans and C. albicans). Virulence 2012, 3, 635–646. [Google Scholar] [CrossRef] [PubMed]
- Bozza, S.; Gaziano, R.; Spreca, A.; Bacci, A.; Montagnoli, C.; di Francesco, P.; Romani, L. Dendritic cells transport conidia and hyphae of Aspergillus fumigatus from the airways to the draining lymph nodes and initiate disparate th responses to the fungus. J. Immunol. 2002, 168, 1362–1371. [Google Scholar] [CrossRef] [PubMed]
- Gafa, V.; Remoli, M.E.; Giacomini, E.; Gagliardi, M.C.; Lande, R.; Severa, M.; Grillot, R.; Coccia, E.M. In vitro infection of human dendritic cells by Aspergillus fumigatus conidia triggers the secretion of chemokines for neutrophil and Th1 lymphocyte recruitment. Microbes Infect. Inst. Pasteur 2007, 9, 971–980. [Google Scholar] [CrossRef] [PubMed]
- Barrios, C.S.; Johnson, B.D.; Henderson, J.D.; Fink, J.N.; Kelly, K.J.; Kurup, V.P. The costimulatory molecules CD80, CD86 and OX40l are up-regulated in Aspergillus fumigatus sensitized mice. Clin. Exp. Immunol. 2005, 142, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Gafa, V.; Lande, R.; Gagliardi, M.C.; Severa, M.; Giacomini, E.; Remoli, M.E.; Nisini, R.; Ramoni, C.; Di Francesco, P.; Aldebert, D. Human dendritic cells following Aspergillus fumigatus infection express the CCR7 receptor and a differential pattern of interleukin-12 (IL-12), IL-23, and IL-27 cytokines, which lead to a th1 response. Infect. Immun. 2006, 74, 1480–1489. [Google Scholar] [CrossRef] [PubMed]
- Lother, J.; Breitschopf, T.; Krappmann, S.; Morton, C.O.; Bouzani, M.; Kurzai, O.; Gunzer, M.; Hasenberg, M.; Einsele, H.; Loeffler, J. Human dendritic cell subsets display distinct interactions with the pathogenic mould Aspergillus fumigatus. Int. J. Med. Microbiol. 2014, 304, 1160–1168. [Google Scholar] [CrossRef] [PubMed]
- Bozza, S.; Clavaud, C.; Giovannini, G.; Fontaine, T.; Beauvais, A.; Sarfati, J.; D’Angelo, C.; Perruccio, K.; Bonifazi, P.; Zagarella, S.; et al. Immune sensing of Aspergillus fumigatus proteins, glycolipids, and polysaccharides and the impact on th immunity and vaccination. J. Immunol. 2009, 183, 2407–2414. [Google Scholar] [CrossRef] [PubMed]
- Fei, M.; Bhatia, S.; Oriss, T.B.; Yarlagadda, M.; Khare, A.; Akira, S.; Saijo, S.; Iwakura, Y.; Fallert Junecko, B.A.; Reinhart, T.A.; et al. TNF-α from inflammatory dendritic cells (DCS) regulates lung IL-17a/IL-5 levels and neutrophilia versus eosinophilia during persistent fungal infection. Proc. Natl. Acad. Sci. USA 2011, 108, 5360–5365. [Google Scholar] [CrossRef] [PubMed]
- Chamilos, G.; Ganguly, D.; Lande, R.; Gregorio, J.; Meller, S.; Goldman, W.E.; Gilliet, M.; Kontoyiannis, D.P. Generation of IL-23 producing dendritic cells (DCS) by airborne fungi regulates fungal pathogenicity via the induction of Th-17 responses. PLoS ONE 2010, 5, e12955. [Google Scholar] [CrossRef] [PubMed]
- Bacher, P.; Kniemeyer, O.; Schonbrunn, A.; Sawitzki, B.; Assenmacher, M.; Rietschel, E.; Steinbach, A.; Cornely, O.A.; Brakhage, A.A.; Thiel, A.; et al. Antigen-specific expansion of human regulatory T cells as a major tolerance mechanism against mucosal fungi. Mucosal Immunol. 2014, 7, 916–928. [Google Scholar] [CrossRef] [PubMed]
- Jolink, H.; Hagedoorn, R.S.; Lagendijk, E.L.; Drijfhout, J.W.; van Dissel, J.T.; Falkenburg, J.H.F.; Heemskerk, M.H.M. Induction of a. Fumigatus-specific CD4-positive T cells in patients recovering from invasive aspergillosis. Haematologica 2014, 99, 1255–1263. [Google Scholar] [CrossRef] [PubMed]
- Bedke, T.; Iannitti, R.G.; De Luca, A.; Giovannini, G.; Fallarino, F.; Berges, C.; Latge, J.P.; Einsele, H.; Romani, L.; Topp, M.S. Distinct and complementary roles for Aspergillus fumigatus-specific TR1 and FOXP3+ regulatory t cells in humans and mice. Immunol. Cell Biol. 2014, 92, 659–670. [Google Scholar] [CrossRef] [PubMed]
- Cenci, E.; Mencacci, A.; Bacci, A.; Bistoni, F.; Kurup, V.P.; Romani, L. T cell vaccination in mice with invasive pulmonary aspergillosis. J. Immunol. (Baltimore, MD) 2000, 165, 381–388. [Google Scholar] [CrossRef]
- Cenci, E.; Mencacci, A.; Fè d’Ostiani, C.; Del Sero, G.; Mosci, P.; Montagnoli, C.; Bacci, A.; Romani, L. Cytokine- and T helper-dependent lung mucosal immunity in mice with invasive pulmonary aspergillosis. J. Infect. Dis. 1998, 178, 1750–1760. [Google Scholar] [CrossRef] [PubMed]
- Cenci, E.; Mencacci, A.; DelSero, G.; Bistoni, F.; Romani, L. Induction of protective th1 responses to candida albicans by antifungal therapy alone or in combination with an interleukin-4 antagonist. J. Infect. Dis. 1997, 176, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Cenci, E.; Perito, S.; Enssle, K.H.; Mosci, P.; Latge, J.P.; Romani, L.; Bistoni, F. Th1 and th2 cytokines in mice with invasive aspergillosis. Infect. Immun. 1997, 65, 564–570. [Google Scholar] [PubMed]
- Romani, L.; Bistoni, F.; Gaziano, R.; Bozza, S.; Montagnoli, C.; Perruccio, K.; Pitzurra, L.; Bellocchio, S.; Velardi, A.; Rasi, G.; et al. Thymosin alpha 1 activates dendritic cells for antifungal Th1 resistance through toll-like receptor signaling. Blood 2004, 103, 4232–4239. [Google Scholar] [CrossRef] [PubMed]
- Brieland, J.K.; Jackson, C.; Menzel, F.; Loebenberg, D.; Cacciapuoti, A.; Halpern, J.; Hurst, S.; Muchamuel, T.; Debets, R.; Kastelein, R.; et al. Cytokine networking in lungs of immunocompetent mice in response to inhaled Aspergillus fumigatus. Infect. Immun. 2001, 69, 1554–1560. [Google Scholar] [CrossRef] [PubMed]
- Damsker, J.M.; Hansen, A.M.; Caspi, R.R. Th1 and th17 cells: Adversaries and collaborators. Ann. N. Y. Acad. Sci. 2010, 1183, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Antachopoulos, C.; Roilides, E. Cytokines and fungal infections. Br. J. Haematol. 2005, 129, 583–596. [Google Scholar] [CrossRef] [PubMed]
- Chai, L.Y.; Van De Veerdonk, F.; Marijnissen, R.J.; Cheng, S.C.; Khoo, A.L.; Hectors, M.; Lagrou, K.; Vonk, A.G.; Maertens, J.; Joosten, L.A. Anti-aspergillus human host defence relies on type 1 T helper (th1), rather than type 17 T helper (Th17), cellular immunity. Immunology 2010, 130, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Nagai, H.; Guo, J.; Choi, H.; Kurup, V. Interferon-γ and tumor necrosis factor-α protect mice from invasive aspergillosis. J. Infect. Dis. 1995, 172, 1554–1560. [Google Scholar] [CrossRef] [PubMed]
- Moss, R.B. Pathophysiology and immunology of allergic bronchopulmonary aspergillosis. Med. Mycol. 2005, 43, 203–206. [Google Scholar] [CrossRef]
- Cenci, E.; Mencacci, A.; Del Sero, G.; Bacci, A.; Montagnoli, C.; Fè d’Ostiani, C.; Mosci, P.; Bachmann, M.; Bistoni, F.; Kopf, M.; et al. Interleukin-4 causes susceptibility to invasive pulmonary aspergillosis through suppression of protective type i responses. J. Infect. Dis. 1999, 180, 1957–1968. [Google Scholar] [CrossRef] [PubMed]
- Del Sero, G.; Mencacci, A.; Cenci, E.; d’Ostiani, C.F.; Montagnoli, C.; Bacci, A.; Mosci, P.; Kopf, M.; Romani, L. Antifungal type 1 responses are upregulated in IL-10-deficient mice. Microbes Infect. Inst. Pasteur 1999, 1, 1169–1180. [Google Scholar] [CrossRef]
- Homma, T.; Kato, A.; Bhushan, B.; Norton, J.E.; Suh, L.A.; Carter, R.G.; Gupta, D.S.; Schleimer, R.P. Role of Aspergillus fumigatus in triggering protease-activated receptor-2 in airway epithelial cells and skewing the cells toward a T-helper 2 bias. Am. J. Respir. Cell Mol. Biol. 2016, 54, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Croston, T.L.; Nayak, A.P.; Lemons, A.R.; Goldsmith, W.T.; Gu, J.K.; Germolec, D.R.; Beezhold, D.H.; Green, B.J. Influence of Aspergillus fumigatus conidia viability on murine pulmonary microrna and mRNA expression following subchronic inhalation exposure. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2016, 46, 1315–1327. [Google Scholar] [CrossRef] [PubMed]
- Castanhinha, S.; Sherburn, R.; Walker, S.; Gupta, A.; Bossley, C.J.; Buckley, J.; Ullmann, N.; Grychtol, R.; Campbell, G.; Maglione, M.; et al. Pediatric severe asthma with fungal sensitization is mediated by steroid-resistant il-33. J. Allergy Clin. Immunol. 2015, 136, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Li, Z.; Yang, X.O.; Chang, S.H.; Nurieva, R.; Wang, Y.H.; Wang, Y.; Hood, L.; Zhu, Z.; Tian, Q.; et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat. Immunol. 2005, 6, 1133–1141. [Google Scholar] [CrossRef] [PubMed]
- Ghoreschi, K.; Laurence, A.; Yang, X.P.; Tato, C.M.; McGeachy, M.J.; Konkel, J.E.; Ramos, H.L.; Wei, L.; Davidson, T.S.; Bouladoux, N.; et al. Generation of pathogenic Th17 cells in the absence of TGF-beta signalling. Nature 2010, 467, 967–971. [Google Scholar] [CrossRef] [PubMed]
- Gresnigt, M.S.; Rosler, B.; Jacobs, C.W.; Becker, K.L.; Joosten, L.A.; van der Meer, J.W.; Netea, M.G.; Dinarello, C.A.; van de Veerdonk, F.L. The IL-36 receptor pathway regulates Aspergillus fumigatus-induced Th1 and Th17 responses. Eur. J. Immunol. 2013, 43, 416–426. [Google Scholar] [CrossRef] [PubMed]
- van de Veerdonk, F.L.; Gresnigt, M.S.; Kullberg, B.J.; van der Meer, J.W.; Joosten, L.A.; Netea, M.G. Th17 responses and host defense against microorganisms: An overview. BMB Rep. 2009, 42, 776–787. [Google Scholar] [CrossRef] [PubMed]
- Way, E.E.; Chen, K.; Kolls, J.K. Dysregulation in lung immunity—The protective and pathologic Th17 response in infection. Eur. J. Immunol. 2013, 43, 3116–3124. [Google Scholar] [CrossRef] [PubMed]
- Werner, J.L.; Metz, A.E.; Horn, D.; Schoeb, T.R.; Hewitt, M.M.; Schwiebert, L.M.; Faro-Trindade, I.; Brown, G.D.; Steele, C. Requisite role for the dectin-1 β-glucan receptor in pulmonary defense against Aspergillus fumigatus. J. Immunol. 2009, 182, 4938–4946. [Google Scholar] [CrossRef] [PubMed]
- Dudakov, J.A.; Hanash, A.M.; van den Brink, M.R.M. Interleukin-22: Immunobiology and pathology. Annu. Rev. Immunol. 2015, 33, 747–785. [Google Scholar] [CrossRef] [PubMed]
- Rivera, A.; Hohl, T.M.; Collins, N.; Leiner, I.; Gallegos, A.; Saijo, S.; Coward, J.W.; Iwakura, Y.; Pamer, E.G. Dectin-1 diversifies Aspergillus fumigatus–specific t cell responses by inhibiting T helper type 1 CD4 T cell differentiation. J. Exp. Med. 2011, 208, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Gessner, M.A.; Werner, J.L.; Lilly, L.M.; Nelson, M.P.; Metz, A.E.; Dunaway, C.W.; Chan, Y.R.; Ouyang, W.; Brown, G.D.; Weaver, C.T.; et al. Dectin-1-dependent interleukin-22 contributes to early innate lung defense against Aspergillus fumigatus. Infect. Immun. 2012, 80, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Gresnigt, M.S.; Bozza, S.; Becker, K.L.; Joosten, L.A.; Abdollahi-Roodsaz, S.; van der Berg, W.B.; Dinarello, C.A.; Netea, M.G.; Fontaine, T.; De Luca, A.; et al. A polysaccharide virulence factor from Aspergillus fumigatus elicits anti-inflammatory effects through induction of interleukin-1 receptor antagonist. PLoS Pathog. 2014, 10, e1003936. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, T.; Delangle, A.; Simenel, C.; Coddeville, B.; van Vliet, S.J.; van Kooyk, Y.; Bozza, S.; Moretti, S.; Schwarz, F.; Trichot, C.; et al. Galactosaminogalactan, a new immunosuppressive polysaccharide of Aspergillus fumigatus. PLoS Pathog. 2011, 7, e1002372. [Google Scholar] [CrossRef] [PubMed]
- Delsing, C.E.; Becker, K.L.; Simon, A.; Kullberg, B.J.; Bleeker-Rovers, C.P.; van de Veerdonk, F.L.; Netea, M.G. Th17 cytokine deficiency in patients with aspergillus skull base osteomyelitis. BMC Infect. Dis. 2015, 15, 140. [Google Scholar] [CrossRef] [PubMed]
- Smeekens, S.P.; Henriet, S.S.; Gresnigt, M.S.; Joosten, L.A.; Hermans, P.W.; Netea, M.G.; Warris, A.; van de Veerdonk, F.L. Low interleukin-17A production in response to fungal pathogens in patients with chronic granulomatous disease. J. Interferon Cytokine Res. 2012, 32, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Zelante, T.; De Luca, A.; D’Angelo, C.; Moretti, S.; Romani, L. IL-17/Th17 in anti-fungal immunity: What’s new? Eur. J. Immunol. 2009, 39, 645–648. [Google Scholar] [CrossRef] [PubMed]
- Zelante, T.; Bozza, S.; De Luca, A.; D’Angelo, C.; Bonifazi, P.; Moretti, S.; Giovannini, G.; Bistoni, F.; Romani, L. Th17 cells in the setting of aspergillus infection and pathology. Med. Mycol. 2009, 47, S162–S169. [Google Scholar] [CrossRef] [PubMed]
- Zelante, T.; De Luca, A.; Bonifazi, P.; Montagnoli, C.; Bozza, S.; Moretti, S.; Belladonna, M.L.; Vacca, C.; Conte, C.; Mosci, P.; et al. IL-23 and the Th17 pathway promote inflammation and impair antifungal immune resistance. Eur. J. Immunol. 2007, 37, 2695–2706. [Google Scholar] [CrossRef] [PubMed]
- Armstrong-James, D.P.H.; Turnbull, S.A.; Teo, I.; Stark, J.; Rogers, N.J.; Rogers, T.R.F.; Bignell, E.; Haynes, K. Impaired interferon-γ responses, increased interleukin-17 expression, and a tumor necrosis factor—α transcriptional program in invasive aspergillosis. J. Infect. Dis. 2009, 200, 1341–1351. [Google Scholar] [CrossRef] [PubMed]
- Bozza, S.; Zelante, T.; Moretti, S.; Bonifazi, P.; DeLuca, A.; D’Angelo, C.; Giovannini, G.; Garlanda, C.; Boon, L.; Bistoni, F.; et al. Lack of toll IL-1R8 exacerbates Th17 cell responses in fungal infection. J. Immunol. 2008, 180, 4022–4031. [Google Scholar] [CrossRef] [PubMed]
- Jolink, H.; de Boer, R.; Hombrink, P.; Jonkers, R.E.; van Dissel, J.T.; Falkenburg, J.H.; Heemskerk, M.H. Pulmonary immune responses against Aspergillus fumigatus are characterized by high frequencies of IL-17 producing T-cells. J. Infect. 2017, 74, 81–88. [Google Scholar] [CrossRef] [PubMed]
- McAleer, J.P.; Nguyen, N.L.; Chen, K.; Kumar, P.; Ricks, D.M.; Binnie, M.; Armentrout, R.A.; Pociask, D.A.; Hein, A.; Yu, A.; et al. Pulmonary Th17 antifungal immunity is regulated by the gut microbiome. J. Immunol. 2016, 197, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Denning, D.W.; O’Driscoll, B.R.; Hogaboam, C.M.; Bowyer, P.; Niven, R.M. The link between fungi and severe asthma: A summary of the evidence. Eur. Respir. J. Off. J. Eur. Soc. Clin. Respir. Physiol. 2006, 27, 615–626. [Google Scholar] [CrossRef] [PubMed]
- Radauer, C.; Nandy, A.; Ferreira, F.; Goodman, R.E.; Larsen, J.N.; Lidholm, J.; Pomes, A.; Raulf-Heimsoth, M.; Rozynek, P.; Thomas, W.R.; et al. Update of the who/IUIS allergen nomenclature database based on analysis of allergen sequences. Allergy 2014, 69, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Crameri, R.; Garbani, M.; Rhyner, C.; Huitema, C. Fungi: The neglected allergenic sources. Allergy 2014, 69, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Simon-Nobbe, B.; Denk, U.; Poll, V.; Rid, R.; Breitenbach, M. The spectrum of fungal allergy. Int. Arch. Allergy Immunol. 2008, 145, 58–86. [Google Scholar] [CrossRef] [PubMed]
- Crameri, R. Structural aspects of fungal allergens. Semin. Immunopathol. 2015, 37, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Sehgal, I.S.; Agarwal, R. Specific ige is better than skin testing for detecting aspergillus sensitization and allergic bronchopulmonary aspergillosis in asthma. Chest 2015, 147, e194. [Google Scholar] [CrossRef] [PubMed]
- De Barros Bezerra, G.F.; Haidar, D.M.C.; da Silva, M.A.C.N.; Filho, W.E.M.; dos Santos, R.M.; Rosa, I.G.; de Castro Viana, G.M.; Zaror, L.; Soares Brandão Nascimento, M.d.D. Ige serum concentration against airborne fungi in children with respiratory allergies. Allergy Asthma Clin. Immunol. Off. J. Can. Soc. Allergy Clin. Immunol. 2016, 12, 18. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, R.; Chakrabarti, A.; Shah, A.; Gupta, D.; Meis, J.F.; Guleria, R.; Moss, R.; Denning, D.W. Allergic bronchopulmonary aspergillosis: Review of literature and proposal of new diagnostic and classification criteria. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2013, 43, 850–873. [Google Scholar] [CrossRef] [PubMed]
- Wark, P.A.B.; Gibson, P.G. Allergic bronchopulmonary aspergillosis: New concepts of pathogenesis and treatment. Respirology 2001, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Becker, K.L.; Gresnigt, M.S.; Smeekens, S.P.; Jacobs, C.W.; Magis-Escurra, C.; Jaeger, M.; Wang, X.; Lubbers, R.; Oosting, M.; Joosten, L.A.B.; et al. Pattern recognition pathways leading to a Th2 cytokine bias in allergic bronchopulmonary aspergillosis patients. Clin. Exp. Allergy 2015, 45, 423–437. [Google Scholar] [CrossRef] [PubMed]
- Murdock, B.J.; Shreiner, A.B.; McDonald, R.A.; Osterholzer, J.J.; White, E.S.; Toews, G.B.; Huffnagle, G.B. Coevolution of Th1, Th2, and Th17 responses during repeated pulmonary exposure to Aspergillus fumigatus conidia. Infect. Immun. 2011, 79, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Kurup, V.P.; Choi, H.; Murali, P.S.; Coffman, R.L. IgE and eosinophil regulation in a murine model of allergic aspergillosis. J. Leukoc. Biol. 1994, 56, 593–598. [Google Scholar] [PubMed]
- Grünig, G.; Corry, D.B.; Leach, M.W.; Seymour, B.W.P.; Kurup, V.P.; Rennick, D.M. Interleukin-10 is a natural suppressor of cytokine production and inflammation in a murine model of allergic bronchopulmonary aspergillosis. J. Exp. Med. 1997, 185, 1089–1100. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.M.; Denis, M.; Fournfer, M.; Laviolette, M. Experimental allergic bronchopulmonary aspergillosis in the mouse: Immunological and histological features. Scand. J. Immunol. 1994, 39, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Murdock, B.J.; Falkowski, N.R.; Shreiner, A.B.; Sadighi Akha, A.A.; McDonald, R.A.; White, E.S.; Toews, G.B.; Huffnagle, G.B. Interleukin-17 drives pulmonary eosinophilia following repeated exposure to Aspergillus fumigatus conidia. Infect. Immun. 2012, 80, 1424–1436. [Google Scholar] [CrossRef] [PubMed]
- De Luca, A.; Pariano, M.; Cellini, B.; Costantini, C.; Villella, V.R.; Jose, S.S.; Palmieri, M.; Borghi, M.; Galosi, C.; Paolicelli, G.; et al. The IL-17F/IL-17RC axis promotes respiratory allergy in the proximal airways. Cell Rep. 2017, 20, 1667–1680. [Google Scholar] [CrossRef] [PubMed]
- Fukahori, S.; Matsuse, H.; Tsuchida, T.; Kawano, T.; Nishino, T.; Fukushima, C.; Kohno, S. Clearance of Aspergillus fumigatus is impaired in the airway in allergic inflammation. Ann. Allergy Asthma Immunol. Off. Publ. Am. Coll. Allergy Asthma Immunol. 2014, 113, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Lathrop, M.J.; Brooks, E.M.; Bonenfant, N.R.; Sokocevic, D.; Borg, Z.D.; Goodwin, M.; Loi, R.; Cruz, F.; Dunaway, C.W.; Steele, C.; et al. Mesenchymal stromal cells mediate aspergillus hyphal extract-induced allergic airway inflammation by inhibition of the Th17 signaling pathway. Stem Cells Transl. Med. 2014, 3, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Borghi, M.; Renga, G.; Puccetti, M.; Oikonomou, V.; Palmieri, M.; Galosi, C.; Bartoli, A.; Romani, L. Antifungal th immunity: Growing up in family. Front. Immunol. 2014, 5, 506. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, M.H.; Hufford, M.M.; Olson, M.R. The development and in vivo function of T helper 9 cells. Nat. Rev. Immunol. 2015, 15, 295–307. [Google Scholar] [CrossRef] [PubMed]
- Becker, K.L.; Rösler, B.; Wang, X.; Lachmandas, E.; Kamsteeg, M.; Jacobs, C.W.; Joosten, L.A.; Netea, M.G.; van de Veerdonk, F.L. Th2 and Th9 responses in patients with chronic mucocutaneous candidiasis and hyper-ige syndrome. Clin. Exp. Allergy 2016, 46, 1564–1574. [Google Scholar] [CrossRef] [PubMed]
- Moretti, S.; Renga, R.; Oikonomou, V.; Galosi, C.; Pariano, M.; Napolioni, V.; Romani, L. A mast cell-ILC2-Th9 pathway promotes lung inflammation in cystic fibrosis. Nat. Commun. 2017, 8, 14017. [Google Scholar] [CrossRef] [PubMed]
- Vignali, D.A.A.; Collison, L.W.; Workman, C.J. How regulatory t cells work. Nat. Rev. Immunol. 2008, 8, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Montagnoli, C.; Fallarino, F.; Gaziano, R.; Bozza, S.; Bellocchio, S.; Zelante, T.; Kurup, W.P.; Pitzurra, L.; Puccetti, P.; Romani, L. Immunity and tolerance to aspergillus involve functionally distinct regulatory t cells and tryptophan catabolism. J. Immunol. 2006, 176, 1712–1723. [Google Scholar] [CrossRef] [PubMed]
- Ostroukhova, M.; Seguin-Devaux, C.; Oriss, T.B.; Dixon-McCarthy, B.; Yang, L.; Ameredes, B.T.; Corcoran, T.E.; Ray, A. Tolerance induced by inhaled antigen involves CD4(+) T cells expressing membrane-bound TGF-beta and FOXP3. J. Clin. Investig. 2004, 114, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Kreindler, J.L.; Steele, C.; Nguyen, N.; Chan, Y.R.; Pilewski, J.M.; Alcorn, J.F.; Vyas, Y.M.; Aujla, S.J.; Finelli, P.; Blanchard, M.; et al. Vitamin d3 attenuates Th2 responses to Aspergillus fumigatus mounted by CD4+ T cells from cystic fibrosis patients with allergic bronchopulmonary aspergillosis. J. Clin. Investig. 2010, 120, 3242–3254. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.L.; Pilewski, J.M.; Celedon, J.C.; Mandalapu, S.; Blanchard, M.L.; DeRicco, A.; Hartigan, E.; Alcorn, J.F.; Kolls, J.K. Vitamin D supplementation decreases Aspergillus fumigatus specific Th2 responses in CF patients with aspergillus sensitization: A phase one open-label study. Asthma Res. Pract. 2015, 1. [Google Scholar] [CrossRef] [PubMed]
- Segal, B.H.; Romani, L.R. Invasive aspergillosis in chronic granulomatous disease. Med. Mycol. 2009, 47, S282–S290. [Google Scholar] [CrossRef] [PubMed]
- Romani, L.; Fallarino, F.; De Luca, A.; Montagnoli, C.; D’Angelo, C.; Zelante, T.; Vacca, C.; Bistoni, F.; Fioretti, M.C.; Grohmann, U.; et al. Defective tryptophan catabolism underlies inflammation in mouse chronic granulomatous disease. Nature 2008, 451, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Porpon, R.; Chen, Y.C.; Chakrabarti, A.; Li, R.Y.; Shivaprakash, R.M.; Yu, J.; Kung, H.C.; Watcharananan, S.; Tan, A.L.; Saffari, S.E.; et al. Epidemiology and clinical characteristics of invasive mould infections: A multicenter, retrospective analysis in five asian countries. Med. Mycol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.D.; Denning, D.W.; Gow, N.A.; Levitz, S.M.; Netea, M.G.; White, T.C. Hidden killers: Human fungal infections. Sci. Transl. Med. 2012, 4, 165rv113. [Google Scholar] [CrossRef] [PubMed]
- Armstrong-James, D.; Brown, G.D.; Netea, M.G.; Zelante, T.; Gresnigt, M.S.; van de Veerdonk, F.L.; Levitz, S.M. Immunotherapeutic approaches to treatment of fungal diseases. Lancet Infect. Dis. 2017. [Google Scholar] [CrossRef]
- Carvalho, A.; Cunha, C.; Bistoni, F.; Romani, L. Immunotherapy of aspergillosis. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2012, 18, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou, A.; Kaloyannidis, P.; Yannaki, E.; Cruz, C.R. Adoptive transfer of aspergillus-specific t cells as a novel anti-fungal therapy for hematopoietic stem cell transplant recipients: Progress and challenges. Crit. Rev. Oncol. Hematol. 2016, 98, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Safdar, A.; Rodriguez, G.; Zuniga, J.; Al Akhrass, F.; Georgescu, G.; Pande, A. Granulocyte macrophage colony-stimulating factor in 66 patients with myeloid or lymphoid neoplasms and recipients of hematopoietic stem cell transplantation with invasive fungal disease. Acta Haematol. 2013, 129, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Armstrong-James, D.; Teo, I.A.; Shrivastava, S.; Petrou, M.A.; Taube, D.; Dorling, A.; Shaunak, S. Exogenous interferon-gamma immunotherapy for invasive fungal infections in kidney transplant patients. Am. J. Transpl. 2010, 10, 1796–1803. [Google Scholar] [CrossRef] [PubMed]
- Delsing, C.E.; Gresnigt, M.S.; Leentjens, J.; Preijers, F.; Frager, F.A.; Kox, M.; Monneret, G.; Venet, F.; Bleeker-Rovers, C.P.; van de Veerdonk, F.L.; et al. Interferon-gamma as adjunctive immunotherapy for invasive fungal infections: A case series. BMC Infect. Dis. 2014, 14, 166. [Google Scholar] [CrossRef] [PubMed]
- Tramsen, L.; Koehl, U.; Tonn, T.; Latge, J.P.; Schuster, F.R.; Borkhardt, A.; Uharek, L.; Quaritsch, R.; Beck, O.; Seifried, E.; et al. Clinical-scale generation of human anti-aspergillus T cells for adoptive immunotherapy. Bone Marrow Transplant. 2009, 43, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Marciano, B.E.; Wesley, R.; De Carlo, E.S.; Anderson, V.L.; Barnhart, L.A.; Darnell, D.; Malech, H.L.; Gallin, J.I.; Holland, S.M. Long-term interferon-γ therapy for patients with chronic granulomatous disease. Clin. Infect. Dis. 2004, 39, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Wan, L.; Zhang, Y.; Lai, Y.; Jiang, M.; Song, Y.; Zhou, J.; Zhang, Z.; Duan, X.; Fu, Y.; Liao, L.; et al. Effect of granulocyte-macrophage colony-stimulating factor on prevention and treatment of invasive fungal disease in recipients of allogeneic stem-cell transplantation: A prospective multicenter randomized phase iv trial. J. Clin. Oncol. 2015, 33, 3999–4006. [Google Scholar] [CrossRef] [PubMed]
- Bandera, A.; Trabattoni, D.; Ferrario, G.; Cesari, M.; Franzetti, F.; Clerici, M.; Gori, A. Interferon-γ and granulocyte-macrophage colony stimulating factor therapy in three patients with pulmonary aspergillosis. Infection 2008, 36, 368–373. [Google Scholar] [CrossRef] [PubMed]
- Iannitti, R.G.; Napolioni, V.; Oikonomou, V.; De Luca, A.; Galosi, C.; Pariano, M.; Massi-Benedetti, C.; Borghi, M.; Puccetti, M.; Lucidi, V.; et al. IL-1 receptor antagonist ameliorates inflammasome-dependent inflammation in murine and human cystic fibrosis. Nat. Commun. 2016, 7, 10791. [Google Scholar] [CrossRef] [PubMed]
- de Luca, A.; Smeekens, S.P.; Casagrande, A.; Iannitti, R.; Conway, K.L.; Gresnigt, M.S.; Begun, J.; Plantinga, T.S.; Joosten, L.A.; van der Meer, J.W.; et al. IL-1 receptor blockade restores autophagy and reduces inflammation in chronic granulomatous disease in mice and in humans. Proc. Natl. Acad. Sci. USA 2014, 111, 3526–3531. [Google Scholar] [CrossRef] [PubMed]
- Gresnigt, M.S.; Rekiki, A.; Rasid, O.; Savers, A.; Jouvion, G.; Dannaoui, E.; Parlato, M.; Fitting, C.; Brock, M.; Cavaillon, J.M.; et al. Reducing hypoxia and inflammation during invasive pulmonary aspergillosis by targeting the interleukin-1 receptor. Sci. Rep. 2016, 6, 26490. [Google Scholar] [CrossRef] [PubMed]
- Geddes, M.; Storek, J. Immune reconstitution following hematopoietic stem-cell transplantation. Best Pract. Res. Clin. Haematol. 2007, 20, 329–348. [Google Scholar] [CrossRef] [PubMed]
- Perruccio, K.; Tosti, A.; Burchielli, E.; Topini, F.; Ruggeri, L.; Carotti, A.; Capanni, M.; Urbani, E.; Mancusi, A.; Aversa, F. Transferring functional immune responses to pathogens after haploidentical hematopoietic transplantation. Blood 2005, 106, 4397–4406. [Google Scholar] [CrossRef] [PubMed]
- Lehrnbecher, T.; Schmidt, S.; Tramsen, L.; Klingebiel, T. Immunotherapy of invasive fungal infection in hematopoietic stem cell transplant recipients. Front. Oncol. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Kumaresan, P.R.; Manuri, P.R.; Albert, N.D.; Maiti, S.; Singh, H.; Mi, T.; Roszik, J.; Rabinovich, B.; Olivares, S.; Krishnamurthy, J.; et al. Bioengineering t cells to target carbohydrate to treat opportunistic fungal infection. Proc. Natl. Acad. Sci. USA 2014, 111, 10660–10665. [Google Scholar] [CrossRef] [PubMed]
- Koenen, H.J.; Smeets, R.L.; Vink, P.M.; van Rijssen, E.; Boots, A.M.; Joosten, I. Human cd25highfoxp3pos regulatory t cells differentiate into il-17-producing cells. Blood 2008, 112, 2340–2352. [Google Scholar] [CrossRef] [PubMed]
- Raijmakers, R.P.H.; Sprenkeler, E.G.G.; Aleva, F.E.; Jacobs, C.W.M.; Kanneganti, T.D.; Joosten, L.A.B.; van de Veerdonk, F.L.; Gresnigt, M.S. Toll-like receptor 2 induced cytotoxic t-lymphocyte-associated protein 4 regulates aspergillus-induced regulatory T-cells with pro-inflammatory characteristics. Sci. Rep. 2017, 7, 11500. [Google Scholar] [CrossRef] [PubMed]
- Zielinski, C.E.; Mele, F.; Aschenbrenner, D.; Jarrossay, D.; Ronchi, F.; Gattorno, M.; Monticelli, S.; Lanzavecchia, A.; Sallusto, F. Pathogen-induced human Th17 cells produce IFN-gamma or IL-10 and are regulated by IL-1β. Nature 2012, 484, 514–518. [Google Scholar] [CrossRef] [PubMed]
- Gresnigt, M.S.; Becker, K.L.; Smeekens, S.P.; Jacobs, C.W.; Joosten, L.A.; van der Meer, J.W.; Netea, M.G.; van de Veerdonk, F.L. Aspergillus fumigatus-induced IL-22 is not restricted to a specific th cell subset and is dependent on complement receptor 3. J. Immunol. 2013, 190, 5629–5639. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dewi, I.M.W.; Van de Veerdonk, F.L.; Gresnigt, M.S. The Multifaceted Role of T-Helper Responses in Host Defense against Aspergillus fumigatus. J. Fungi 2017, 3, 55. https://doi.org/10.3390/jof3040055
Dewi IMW, Van de Veerdonk FL, Gresnigt MS. The Multifaceted Role of T-Helper Responses in Host Defense against Aspergillus fumigatus. Journal of Fungi. 2017; 3(4):55. https://doi.org/10.3390/jof3040055
Chicago/Turabian StyleDewi, Intan M. W., Frank L. Van de Veerdonk, and Mark S. Gresnigt. 2017. "The Multifaceted Role of T-Helper Responses in Host Defense against Aspergillus fumigatus" Journal of Fungi 3, no. 4: 55. https://doi.org/10.3390/jof3040055
APA StyleDewi, I. M. W., Van de Veerdonk, F. L., & Gresnigt, M. S. (2017). The Multifaceted Role of T-Helper Responses in Host Defense against Aspergillus fumigatus. Journal of Fungi, 3(4), 55. https://doi.org/10.3390/jof3040055




