Mode of Infection of Metarhizium spp. Fungus and Their Potential as Biological Control Agents
Abstract
:1. Introduction
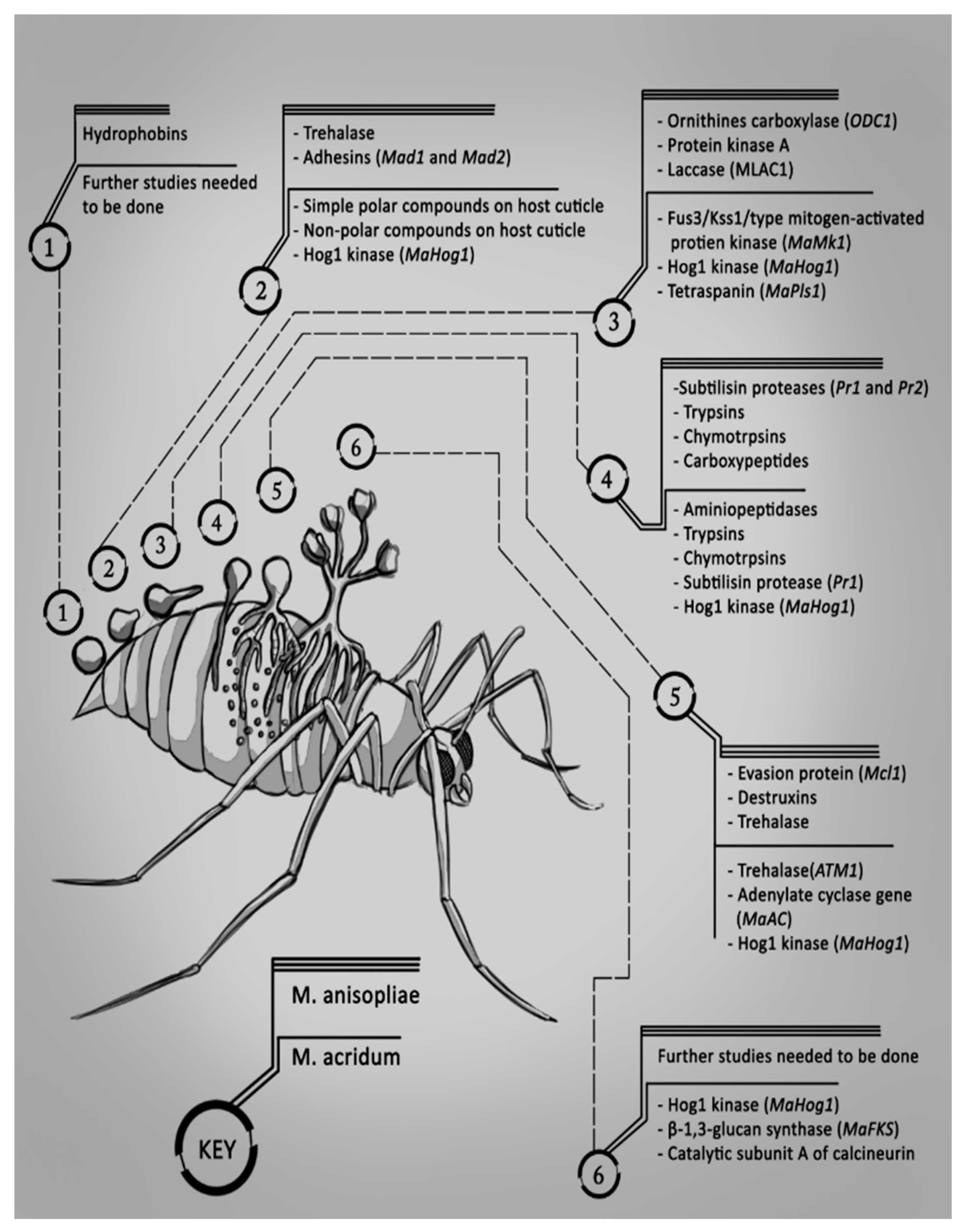
2. Mode of Infection of Metarhizium spp. with Their Hosts
2.1. Adhesion
2.2. Germination
2.3. Appressorium Formation
2.4. Penetration
2.5. Colonization of the Haemolymph
2.6. Extrusion and Sporulation
2.7. Summary
3. Mode of Infection of Metarhizium spp. in Aquatic Insect Larvae
4. Molecular Approaches to Increase the Virulence and Efficacy of Metarhizium spp. as a Biological Control Agent
5. Mass Production of Metarhizium anisopliae and Metarhizium acridum
6. Conidia Formulation for Metarhizium anisopliae and Metarhizium acridum
7. Metarhizium anisopliae as a Biological Control
7.1. Biological Control of Termites
7.2. Biological Control of Mosquitoes
7.2.1. Adult Mosquitoes
7.2.2. Mosquito Larvae and Eggs
7.3. Biological Control of Ticks
8. Metarhizum acridum as a Biological Control of Locusts and Grasshoppers
9. Conclusions
Conflicts of Interest
References
- Long, Z.; Hunter, D.M. Laboratory and Field Trials of Green Guard®; (Metarhizium anisopliae var. acridum) (Deuteromycotina: Hyphomycetes) against the Oriental Migratory Locust (Locusta migratoria manilensis) (Orthoptera: Acrididae) in China. J. Orthoptera Res. 2005, 14, 27–30. [Google Scholar] [CrossRef]
- Yanagawa, A.; Shimizu, S. Resistance of the termite, Coptotermes formosanus Shiraki to Metarhizium anisopliae due to grooming. BioControl 2007, 52, 75–85. [Google Scholar] [CrossRef]
- Leemon, D.M.; Jonsson, N.N. Laboratory studies on Australian isolates of Metarhizium anisopliae as a biopesticide for the cattle tick Boophilus microplus. J. Invertebr. Pathol. 2008, 97, 40–49. [Google Scholar] [CrossRef] [PubMed]
- PikKheng, H.; Bong, C.F.J.; Jugah, K.; Rajan, A. Evaluation of Metarhizium anisopliae var. anisopliae (Deuteromycotina: Hyphomycete) isolates and their effects on subterranean termite Coptotermes curvignathus (Isoptera: Rhinotermitidae). Am. J. Agric. Biol. Sci. 2009, 4, 289–297. [Google Scholar]
- Niassy, S.; Diarra, K.; Ndiaye, S.; Niassy, A. Pathogenicity of local Metarhizium anisopliae var. acridum strains on Locusta migratoria migratorioides Reiche and Farmaire and Zonocerus variegatus Linnaeus in Senegal. Afr. J. Biotechnol. 2011, 10, 28. [Google Scholar]
- Rahimzadeh, A.; Rashid, M.; Sheikhi Garjan, A.; Naseri, B. Laboratory evaluation of Metarhizium anisopliae (Metschnikoff) for controlling Amitermes vilis (Hagen) and Microcerotermes gabrielies (Weidner) (Isoptera: Termitidae). J. Crop Prot. 2012, 1, 27–34. [Google Scholar]
- Guerrero-Guerra, C.; del Roció Reyes-Montes, M.; Toriello, C.; Hernández-Velázquez, V.; Santiago-López, I.; Mora-Palomino, L.; Calderón-Segura, M.E.; Fernández, S.D.; Calderón-Ezquerro, C. Study of the persistence and viability of Metarhizium acridum in Mexico’s agricultural area. Aerobiologia 2013, 29, 249–261. [Google Scholar] [CrossRef]
- Camargo, M.G.; Marciano, A.F.; Sá, F.A.; Perinotto, W.M.S.; Quinelato, S.; Gôlo, P.S.; Angelo, I.C.; Prata, M.C.A.; Bittencourt, V.R.E.P. Commercial formulation of Metarhizium anisopliae for the control of Rhipicephalus microplus in a pen study. Vet. Parasitol. 2014, 205, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Paula, A.R.; Carolino, A.T.; Silva, C.P.; Samuels, R.I. Susceptibility of adult female Aedes aegypti (Diptera: Culicidae) to the entomopathogenic fungus Metarhizium anisopliae is modified following blood feeding. Parasites Vectors 2011, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Greenfield, B.P.; Peace, A.; Evans, H.; Dudley, E.; Ansari, M.A.; Butt, T.M. Identification of Metarhizium strains highly efficacious against Aedes, Anopheles and Culex larvae. Biocontrol Sci. Technol. 2015, 25, 487–502. [Google Scholar] [CrossRef]
- Chouvenc, T.; Su, N.-Y.; Kenneth Grace, J. Fifty years of attempted biological control of termites—Analysis of a failure. Biol. Control 2011, 59, 69–82. [Google Scholar] [CrossRef]
- Peng, G.; Xia, Y. The mechanism of the mycoinsecticide diluent on the efficacy of the oil formulation of insecticidal fungus. BioControl 2011, 56, 893–902. [Google Scholar] [CrossRef]
- Cheraghi, A.; Habibpour, B.; Mossadegh, M.S. Application of bait Treated with the entomopathogenic fungus Metarhizium anisopliae (Metsch.) Sorokin for the control of Microcerotermes diversus Silv. Psyche 2013, 2013, 5. [Google Scholar]
- Atheimine, M.O.; Bashir, M.O.; Ely, S.O.; Kane, C.M.H.; Babah, M.A.O.; Benchekroun, M. Efficacy and persistence of Metarhizium acridum (Hypocreales: Clavicipitaceae) used against desert locust larvae, Schistocerca gregaria (Orthoptera: Acrididae), under different vegetation cover types. Int. J. Trop. Insect Sci. 2014, 34, 106–114. [Google Scholar] [CrossRef]
- Fang, W.; Lu, H.-L.; King, G.F.; Leger, R.J.S. Construction of a hypervirulent and specific mycoinsecticide for locust control. Sci. Rep. 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Kamalakannan, S.; Murugan, K. Laboratory and field evaluation of Metarhizium anisopliae for the control of Dengue vector, Aedes aegypti (Insecta: Diptera: Culicidae). Toxicol. Environ. Chem. 2011, 93, 1195–1201. [Google Scholar] [CrossRef]
- Ment, D.; Churchill, A.C.L.; Gindin, G.; Belausov, E.; Glazer, I.; Rehner, S.A.; Rot, A.; Donzelli, B.G.G.; Samish, M. Resistant ticks inhibit Metarhizium infection prior to haemocoel invasion by reducing fungal viability on the cuticle surface. Environ. Microbiol. 2012, 14, 1570–1583. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Martínez, M.G.; Rodríguez-Meneses, A.; Rodríguez, A.D.; Rodríguez, M.H. Lethal effects of Gliocladium virens, Beauveria bassiana and Metarhizium anisopliae on the malaria vector Anopheles albimanus (Diptera: Culicidae). Biocontrol Sci. Technol. 2013, 23, 1098–1109. [Google Scholar] [CrossRef]
- Bukhari, T.; Middelman, A.; Koenraadt, C.J.; Takken, W.; Knols, B.G. Factors affecting fungus-induced larval mortality in Anopheles gambiae and Anopheles stephensi. Malar. J. 2010, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, J.F.; Rehner, S.A.; Humber, R.A. A multilocus phylogeny of the Metarhizium anisopliae lineage. Mycologia 2009, 101, 512–530. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Jin, K.; Ying, S.H.; Zhang, Y.; Xiao, G.; Shang, Y.; Duan, Z.; Hu, X.; Xie, X.Q.; Zhou, G.; et al. Genome sequencing and comparative transcriptomics of the model entomopathogenic fungi Metarhizium anisopliae and M. acridum. PLoS Genet. 2011, 7, e1001264. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Xiao, G.; Zheng, P.; Shang, Y.; Su, Y.; Zhang, X.; Liu, X.; Zhan, S.; St Leger, R.J.; Wang, C. Trajectory and genomic determinants of fungal-pathogen speciation and host adaptation. Proc. Natl. Acad. Sci. USA 2014, 111, 16796–16801. [Google Scholar] [CrossRef] [PubMed]
- De Faria, M.R.; Wraight, S.P. Mycoinsecticides and Mycoacaricides: A comprehensive list with worldwide coverage and international classification of formulation types. Biol. Control 2007, 43, 237–256. [Google Scholar] [CrossRef]
- Pattemore, J.A.; Hane, J.K.; Williams, A.H.; Wilson, B.A.; Stodart, B.J.; Ash, G.J. The genome sequence of the biocontrol fungus Metarhizium anisopliae and comparative genomics of Metarhizium species. BMC Genom. 2014, 15, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Camargo, M.G.; Golo, P.S.; Angelo, I.C.; Perinotto, W.M.S.; Sá, F.A.; Quinelato, S.; Bittencourt, V.R.E.P. Effect of oil-based formulations of acaripathogenic fungi to control Rhipicephalus microplus ticks under laboratory conditions. Vet. Parasitol. 2012, 188, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, K.; Qiu, D.; Sivaramakrishnan, S. Sporulation characteristics and virulence of Metarhizium anisopliae against subterranean termites (Coptotermes formosanus). Int. J. Microbiol. Res. 2015, 6, 1–4. [Google Scholar]
- Leao, M.P.; Tiago, P.V.; Andreote, F.D.; de Araujo, W.L.; de Oliveira, N.T. Differential expression of the pr1A gene in Metarhizium anisopliae and Metarhizium acridum across different culture conditions and during pathogenesis. Genet. Mol. Biol. 2015, 38, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Ment, D.; Gindin, G.; Rot, A.; Soroker, V.; Glazer, I.; Barel, S.; Samish, M. Novel technique for quantifying adhesion of Metarhizium anisopliae conidia to the tick cuticle. Appl. Environ. Microbiol. 2010, 76, 3521–3528. [Google Scholar] [CrossRef] [PubMed]
- Butt, T.M.; Greenfield, B.P.J.; Greig, C.; Maffeis, T.G.G.; Taylor, J.W.D.; Piasecka, J.; Dudley, E.; Abdulla, A.; Dubovskiy, I.M.; Garrido-Jurado, I.; et al. Metarhizium anisopliae Pathogenesis of mosquito larvae: A verdict of accidental death. PLoS ONE 2013, 8, e81686. [Google Scholar] [CrossRef] [PubMed]
- Tseng, M.-N.; Chung, C.-L.; Tzean, S.-S. Mechanisms Relevant to the Enhanced Virulence of a Dihydroxynaphthalene-Melanin Metabolically Engineered Entomopathogen. PLoS ONE 2014, 9, e90473. [Google Scholar] [CrossRef] [PubMed]
- Santi, L.; Silva, W.O.B.; Pinto, A.F.M.; Schrank, A.; Vainstein, M.H. Metarhizium anisopliae host–pathogen interaction: Differential immunoproteomics reveals proteins involved in the infection process of arthropods. Fungal Biol. 2010, 114, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Greenfield, B.P.; Lord, A.M.; Dudley, E.; Butt, T.M. Conidia of the insect pathogenic fungus, Metarhizium anisopliae, fail to adhere to mosquito larval cuticle. R. Soc. Open Sci. 2014, 1, 140193. [Google Scholar] [CrossRef] [PubMed]
- Ment, D.; Gindin, G.; Soroker, V.; Glazer, I.; Rot, A.; Samish, M. Metarhizium anisopliae conidial responses to lipids from tick cuticle and tick mammalian host surface. J. Invertebr. Pathol. 2010, 103, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Santi, L.; Beys da Silva, W.O.; Berger, M.; Guimarães, J.A.; Schrank, A.; Vainstein, M.H. Conidial surface proteins of Metarhizium anisopliae: Source of activities related with toxic effects, host penetration and pathogenesis. Toxicon 2010, 55, 874–880. [Google Scholar] [CrossRef] [PubMed]
- Blanford, S.; Jenkins, N.E.; Read, A.F.; Thomas, M.B. Evaluating the lethal and pre-lethal effects of a range of fungi against adult Anopheles stephensi mosquitoes. Malar. J. 2012, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lovett, B.; Leger, R.J.S. Stress is the rule rather than the exception for Metarhizium. Curr. Genet. 2015, 61, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; St Leger, R.J. The MAD1 adhesin of Metarhizium anisopliae links adhesion with blastospore production and virulence to insects and the MAD2 adhesin enables attachment to plants. Eukaryot. Cell 2007, 6, 808–816. [Google Scholar] [CrossRef] [PubMed]
- Jarrold, S.L.; Moore, D.; Potter, U.; Charnley, A.K. The contribution of surface waxes to pre-penetration growth of an entomopathogenic fungus on host cuticle. Mycol. Res. 2007, 111, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Jin, K.; Ming, Y.; Xia, Y.X. MaHog1, a Hog1-type mitogen-activated protein kinase gene, contributes to stress tolerance and virulence of the entomopathogenic fungus Metarhizium acridum. Microbiology 2012, 158, 2987–2996. [Google Scholar] [CrossRef] [PubMed]
- Madrigal Pulido, J.; Padilla Guerrero, I.; Magaña Martínez, I.D.J.; Cacho Valadez, B.; Torres Guzman, J.C.; Salazar Solis, E.; Felix Gutierrez Corona, J.; Schrank, A.; Jiménez Bremont, F.; González Hernandez, A. Isolation, characterization and expression analysis of the ornithine decarboxylase gene (ODC1) of the entomopathogenic fungus, Metarhizium anisopliae. Microbiol. Res. 2011, 166, 494–507. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; St Leger, R.J. The Metarhizium anisopliae perilipin homologg MPL1 regulates lipid metabolism, appressorial turgor pressure and virulence. J. Biol. Chem. 2007, 282, 21110–21115. [Google Scholar] [CrossRef] [PubMed]
- Fang, W.; Pava-ripoll, M.; Wang, S.; St. Leger, R. Protein kinase A regulates production of virulence determinants by the entomopathogenic fungus, Metarhizium anisopliae. Fungal Genet. Biol. 2009, 46, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Staats, C.C.; Junges, Â.; Guedes, R.L.M.; Thompson, C.E.; de Morais, G.L.; Boldo, J.T.; de Almeida, L.G.P.; Andreis, F.C.; Gerber, A.L.; Sbaraini, N.; et al. Comparative genome analysis of entomopathogenic fungi reveals a complex set of secreted proteins. BMC Genom. 2014, 15, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Jin, K.; Han, L.; Xia, Y. MaMk1, a FUS3/KSS1-type mitogen-activated protein kinase gene, is required for appressorium formation, and insect cuticle penetration of the entomopathogenic fungus Metarhizium acridum. J. Invertebr. Pathol. 2014, 115, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; He, M.; Cao, Y.; Xia, Y. The tetraspanin gene MaPls1 contributes to virulence by affecting germination, appressorial function and enzymes for cuticle degradation in the entomopathogenic fungus, Metarhizium acridum. Environ. Microbiol. 2013, 15, 2966–2979. [Google Scholar] [PubMed]
- Wang, C.; Duan, Z.; St Leger, R.J. MOS1 osmosensor of Metarhizium anisopliae is required for adaptation to insect host hemolymph. Eukaryot. Cell 2008, 7, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Beys da Silva, W.O.; Santi, L.; Schrank, A.; Vainstein, M.H. Metarhizium anisopliae lipolytic activity plays a pivotal role in Rhipicephalus (Boophilus) microplus infection. Fungal Biol. 2010, 114, 10–15. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Xia, Y. Construction and analysis of a normalized cDNA library from Metarhizium anisopliae var. acridum germinating and differentiating on Locusta migratoria wings. FEMS Microbiol. Lett. 2008, 291, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Boldo, J.T.; Junges, A.; do Amaral, K.B.; Staats, C.C.; Vainstein, M.H.; Schrank, A. Endochitinase CHI2 of the biocontrol fungus Metarhizium anisopliae affects its virulence toward the cotton stainer bug Dysdercus peruvianus. Curr. Genet. 2009, 55, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Kang, Q.; Lu, Y.; Bai, L.; Wang, C. Unveiling the biosynthetic puzzle of destruxins in Metarhizium species. Proc. Natl. Acad. Sci. USA 2012, 109, 1287–1292. [Google Scholar] [CrossRef] [PubMed]
- Peng, G.; Xia, Y. Expression of scorpion toxin LqhIT2 increases the virulence of Metarhizium acridum towards Locusta migratoria manilensis. J. Ind. Microbiol. Biotechnol. 2014, 41, 1659–1666. [Google Scholar] [CrossRef] [PubMed]
- Jin, K.; Peng, G.; Liu, Y.; Xia, Y. The acid trehalase, ATM1, contributes to the in vivo growth and virulence of the entomopathogenic fungus, Metarhizium acridum. Fungal Genet. Biol. 2015, 77, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Peng, G.; Jin, K.; Liu, Y.; Xia, Y. Enhancing the utilization of host trehalose by fungal trehalase improves the virulence of fungal insecticide. Appl. Microbiol. Biotechnol. 2015, 99, 8611–8618. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Peng, G.; Xia, Y. The adenylate cyclase gene MaAC is required for virulence and multi-stress tolerance of Metarhizium acridum. BMC Microbiol. 2012, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ocampo, V.R.; Caoili, B.L. Infection process of entomopathogenic fungi Metarhizium anisopliae in the Tetranychus kanzawai (Kishida) (Tetranynichidae: Acarina). Agrivita 2013, 35, 64. [Google Scholar]
- Gabarty, A.; Salem, H.M.; Fouda, M.A.; Abas, A.A.; Ibrahim, A.A. Pathogencity induced by the entomopathogenic fungi Beauveria bassiana and Metarhizium anisopliae in Agrotis ipsilon (Hufn.). J. Radiat. Res. Appl. Sci. 2014, 7, 95–100. [Google Scholar] [CrossRef]
- Seyoum, E.; Negash, M. Studies on the field performance of Metarhizium anisoliae var. acridum (Green Muscle) against mixed populations of grasshopper in Ethiopia. SINET Ethiop. J. Sci. 2007, 30, 55–64. [Google Scholar] [CrossRef]
- Cao, Y.; Du, M.; Luo, S.; Xia, Y. Calcineurin modulates growth, stress tolerance, and virulence in Metarhizium acridum and its regulatory network. Appl. Microbiol. Biotechnol. 2014, 98, 8253–8265. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Jin, K.; Xia, Y. MaFKS, a β-1,3-glucan synthase, is involved in cell wall integrity, hyperosmotic pressure tolerance and conidiation in Metarhizium acridum. Curr. Genet. 2011, 57, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Sree, K.S.; Padmaja, V. Oxidative stress induced by destruxin from Metarhizium anisopliae (Metch.) involves changes in glutathione and ascorbate metabolism and instigates ultrastructural changes in the salivary glands of Spodoptera litura (Fab.) larvae. Toxicon 2008, 51, 1140–1150. [Google Scholar] [CrossRef] [PubMed]
- Sree, K.S.; Padmaja, V. Destruxin from Metarhizium anisopliae induces oxidative stress effecting larval mortality of the polyphagous pest Spodoptera litura. J. Appl. Entomol. 2008, 132, 68–78. [Google Scholar] [CrossRef]
- Kirubakaran, S.A.; Haripriya, R.; Naveenraj, D.; Thirumalaivasan, P. Virulence of Metarhizium anisopliae soil and commercial isolates against Culex quinquefasciatus Say, a vector of Bancroftian filariasis, and Aedes aegypti L., a vector of dengue fever. Eur. J. Biotechnol. Biosci. 2015, 3, 39–47. [Google Scholar]
- Peng, G.; Xia, Y. Integration of an insecticidal scorpion toxin (BjαIT) gene into Metarhizium acridum enhances fungal virulence towards Locusta migratoria manilensis. Pest Manag. Sci. 2015, 71, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Morales Hernandez, C.E.; Padilla Guerrero, I.E.; Gonzalez Hernandez, G.A.; Salazar Solis, E.; Torres Guzman, J.C. Catalase overexpression reduces the germination time and increases the pathogenicity of the fungus Metarhizium anisopliae. Appl. Microbiol. Biotechnol. 2010, 87, 1033–1044. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; St Leger, R.J. A scorpion neurotoxin increases the potency of a fungal insecticide. Nat. Biotechnol. 2007, 25, 1455–1456. [Google Scholar] [CrossRef] [PubMed]
- Fang, W.; Vega-Rodríguez, J.; Ghosh, A.K.; Jacobs-Lorena, M.; Kang, A.; St Leger, R.J. Development of transgenic fungi that kill human malaria parasites in mosquitoes. Science 2011, 331, 1074–1077. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Ying, H.S.; Feng, M.G. Assessment of oral virulence against Spodoptera litura, acquired by a previously non-pathogenic Metarhizium anisopliae isolate, following integration of a midgut-specific insecticidal toxin. Biol. Control 2014, 79, 8–15. [Google Scholar] [CrossRef]
- Hussain, A.; Tian, M.-Y.; He, Y.-R.; Bland, J.M.; Gu, W.-X. Behavioral and electrophysiological responses of Coptotermes formosanus Shiraki towards entomopathogenic fungal volatiles. Biol. Control 2010, 55, 166–173. [Google Scholar] [CrossRef]
- Maldonado-Blanco, M.G.; Gallegos-Sandoval, J.L.; Fernández-Peña, G.; Sandoval-Coronado, C.F.; Elías-Santos, M. Effect of culture medium on the production and virulence of submerged spores of Metarhizium anisopliae and Beauveria bassiana against larvae and adults of Aedes aegypti (Diptera: Culicidae). Biocontrol Sci. Technol. 2014, 24, 180–189. [Google Scholar] [CrossRef]
- Rangel, D.E.N.; Braga, G.U.L.; Anderson, A.J.; Roberts, D.W. Influence of growth environment on tolerance to UV-B radiation, germination speed, and morphology of Metarhizium anisopliae var. acridum conidia. J. Invertebr. Pathol. 2005, 90, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.; Ahmed, S.; Shahid, M. Laboratory and field evaluation of Metarhizium anisopliae var. anisopliae for controlling subterranean termites. Neotrop. Entomol. 2011, 40, 244–250. [Google Scholar] [PubMed]
- Albernaz, D.A.; Tai, M.H.; Luz, C. Enhanced ovicidal activity of an oil formulation of the fungus Metarhizium anisopliae on the mosquito Aedes aegypti. Med. Vet. Entomol. 2009, 23, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Sousa, N.A.; Lobo, L.S.; Rodrigues, J.; Luz, C. New insights on the effectiveness of Metarhizium anisopliae formulation and application against Aedes aegypti eggs. Lett. Appl. Microbiol. 2013, 57, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Mnyone, L.L.; Kirby, M.J.; Mpingwa, M.W.; Lwetoijera, D.W.; Knols, B.G.; Takken, W.; Koenraadt, C.J.; Russell, T.L. Infection of Anopheles gambiae mosquitoes with entomopathogenic fungi: Effect of host age and blood-feeding status. Parasitol. Res. 2011, 108, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Hedimbi, M.; Kaaya, G.P.; Singh, S.; Chimwamurombe, P.M.; Gindin, G.; Glazer, I.; Samish, M. Protection of Metarhizium anisopliae conidia from ultra-violet radiation and their pathogenicity to Rhipicephalus evertsi evertsi ticks. Exp. Appl. Acarol. 2008, 46, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Kaaya, G.P.; Hedimbi, M. The use of entomopathogenic fungi, Beauveria bassiana and Metarhizium anisopliae, as bio-pesticides for tick control. Int. J. Agric. Sci. 2012, 2, 244–250. [Google Scholar]
- Barreto, L.P.; Luz, C.; Mascarin, G.M.; Roberts, D.W.; Arruda, W.; Fernandes, É.K. Effect of heat stress and oil formulation on conidial germination of Metarhizium anisopliae ss on tick cuticle and artificial medium. J. Invertebr. Pathol. 2016, 138, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Alves, F.M.; Bernardo, C.C.; Paixão, F.R.; Barreto, L.P.; Luz, C.; Humber, R.A.; Fernandes, É.K. Heat-stressed Metarhizium anisopliae: Viability (in vitro) and virulence (in vivo) assessments against the tick Rhipicephalus sanguineus. Parasitol. Res. 2017, 116, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Lobo, L.S.; Rodrigues, J.; Luz, C. Effectiveness of Metarhizium anisopliae formulations against dengue vectors under laboratory and field conditions. Biocontrol Sci. Technol. 2016, 26, 386–401. [Google Scholar] [CrossRef]
- Leemon, D.M.; Turner, L.B.; Jonsson, N.N. Pen studies on the control of cattle tick (Rhipicephalus (Boophilus) microplus) with Metarhizium anisopliae (Sorokin). Vet. Parasitol. 2008, 156, 248–260. [Google Scholar] [CrossRef] [PubMed]
- Khaenje, A.; Gohole, L.; Maniania, N. Evaluation of Metarhizium anisopliae for termite (Isoptera: Termitidae) management in maize under field conditions in Uganda and Kenya. In Proceedings of the 11th African Crop Science Conference on Sowing Innovations for Sustainable Food and Nutrition Security in Africa, Entebbe, Uganda, 14–17 October 2013; pp. 195–198. [Google Scholar]
- Paixao, F.R.S.; Muniz, E.R.; Barreto, L.P.; Bernardo, C.C.; Mascarin, G.M.; Luz, C.; Fernandes, É.K. Increased heat tolerance afforded by oil-based conidial formulations of Metarhizium anisopliae and Metarhizium robertsii. Biocontrol Sci. Technol. 2017, 27, 324–337. [Google Scholar] [CrossRef]
- Diedhiou, P.M.; Badji, K.; Faye, R.; Samb, P.I. Stability of Metarhizium acridum based biopesticide in operational conditions in Senegal. Curr. Agric. Res. J. 2014, 2, 83–88. [Google Scholar] [CrossRef]
- Nyam, V.-T.; Bong, C.-F.J.; King, J.-H.P. Control of subterranean termite Coptotermes curvignathus (Isoptera: Rhinotermitidae) by entomopathogen Metarhizium anisopliae Var. Anisopliae cultured in liquid state fermentation. Am. J. Agric. Biol. Sci. 2015, 10, 35. [Google Scholar]
- Sileshi, A.; Sori, W.; Dawd, M. Laboratory evaluation of entomopathogenic fungi Metarhizium anisopliae and Beauveria bassiana against Termite, Macrotermes (Isoptera: Termitidae). Asian J. Plant Sci. 2013, 12, 1–10. [Google Scholar]
- Wright, M.S.; Cornelius, M.L. Mortality and repellent effects of microbial pathogens on Coptotermes formosanus (Isoptera: Rhinotermitidae). BMC Microbiol. 2012, 12, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.; Tian, M.-Y. Germination pattern and inoculum transfer of entomopathogenic fungi and their role in disease resistance among Coptotermes formosanus (Isoptera: Rhinotermitidae). Int. J. Agric. Biol. 2013, 15, 319–324. [Google Scholar]
- Mburu, D.M.; Ochola, L.; Maniania, N.K.; Njagi, P.G.N.; Gitonga, L.M.; Ndung’u, M.W.; Wanjoya, A.K.; Hassanali, A. Relationship between virulence and repellency of entomopathogenic isolates of Metarhizium anisopliae and Beauveria bassiana to the termite Macrotermes michaelseni. J. Insect Physiol. 2009, 55, 774–780. [Google Scholar] [CrossRef] [PubMed]
- Yanagawa, A.; Fujiwara-Tsujii, N.; Akino, T.; Yoshimura, T.; Yanagawa, T.; Shimizu, S. Odor aversion and pathogen-removal efficiency in grooming behavior of the termite Coptotermes formosanus. PLoS ONE 2012, 7, e47412. [Google Scholar] [CrossRef] [PubMed]
- Singha, D.; Singha, B.; Dutta, B.K. Potential of Metarhizium anisopliae and Beauveria bassiana in the control of tea termite Microtermes obesi Holmgren in vitro and under field conditions. J. Pest Sci. 2011, 84, 69–75. [Google Scholar] [CrossRef]
- Chouvenc, T.; Su, N.Y.; Elliott, M.L. Interaction between the subterranean termite Reticulitermes flavipes (Isoptera: Rhinotermitidae) and the entomopathogenic fungus Metarhizium anisopliae in foraging arenas. J. Econ. Entomol. 2008, 101, 885–893. [Google Scholar] [CrossRef] [PubMed]
- Balachander, M.; Remadevi, O.K.; Sasidharan, T.O. Dissemination of Metarhizium anisopliae infection among the population of Odontotermes obesus (Isoptera: Termitidae) by augmenting the fungal conidia with attractants. J. Asia-Pac. Entomol. 2013, 16, 199–208. [Google Scholar] [CrossRef]
- Blumer, M.S.; Bachelet, I.; Raman, R.; Rosengaus, R.; Sasisekharan, R. Targeting an antimicrobial effector function in insect immunity as a rest control strategy. Proc. Natl. Acad. Sci. USA 2009, 106, 12652–12657. [Google Scholar] [CrossRef] [PubMed]
- Chouvenc, T.; Su, N.-Y.; Robert, A. Inhibition of Metarhizium anisopliae in the alimentary tract of the eastern subterranean termite Reticulitermes flavipes. J. Invertebr. Pathol. 2009, 101, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Villanueva, F.; Garza-Hernandez, J.A.; Garcia-Munguia, A.M.; Tamez-Guerra, P.; Howard, A.F.; Rodriguez-Perez, M.A. Dissemination of Metarhizium anisopliae of low and high virulence by mating behavior in Aedes aegypti. Parasites Vectors 2011, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.H.; Tai, M.H.H.; Rocha, L.F.N.; Silva, H.H.G.; Luz, C. Dependence of Metarhizium anisopliae on high humidity for ovicidal activity on Aedes aegypti. Biol. Control 2009, 50, 37–42. [Google Scholar] [CrossRef]
- Bukhari, T.; Takken, W.; Koenraadt, C.J. Development of Metarhizium anisopliae and Beauveria bassiana formulations for control of malaria mosquito larvae. Parasites Vectors 2011, 4, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Carolino, A.T.; Paula, A.R.; Silva, C.P.; Butt, T.M.; Samuels, R.I. Monitoring persistence of the entomopathogenic fungus Metarhizium anisopliae under simulated field conditions with the aim of controlling adult Aedes aegypti (Diptera: Culicidae). Parasites Vectors 2014, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Luz, C.; Mnyone, L.L.; Russell, T.L. Survival of anopheline eggs and their susceptibility to infection with Metarhizium anisopliae and Beauveria bassiana under laboratory conditions. Parasitol. Res. 2011, 109, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Fang, W.; Azimzadeh, P.; St Leger, R.J. Strain improvement of fungal insecticides for controlling insect pests and vector-borne diseases. Curr. Opin. Microbiol. 2012, 15, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Quinelato, S.; Golo, P.S.; Perinotto, W.M.; Sá, F.A.; Camargo, M.G.; Angelo, I.C.; Moraes, A.M.; Bittencourt, V.R. Virulence potential of Metarhizium anisopliae sl isolates on Rhipicephalus (Boophilus) microplus larvae. Vet. Parasitol. 2012, 190, 556–565. [Google Scholar] [CrossRef] [PubMed]
- Ren, Q.; Sun, M.; Guan, G.; Liu, Z.; Chen, Z.; Liu, A.; Li, Y.; Ma, M.; Yang, J.; Niu, Q.; et al. Susceptibility of the tick Haemaphysalis qinghaiensis to isolates of the fungus Metarhizium anisopliae in China. Exp. Appl. Acarol. 2014, 64, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Ren, Q.; Chen, Z.; Luo, J.; Liu, G.; Guan, G.; Liu, Z.; Liu, A.; Li, Y.; Niu, Q.; Liu, J.; et al. Laboratory evaluation of Beauveria bassiana and Metarhizium anisopliae in the control of Haemaphysalis qinghaiensis in China. Exp. Appl. Acarol. 2016, 69, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Butt, T.M.; Wood, M.; Taylor, J.W.; Bakirci, S.; Hazir, C.; Ulug, D.; Hazir, S. Differential susceptibility of Hyalomma excavatum adults and nymphs to the entomopathogens Metarhizium anisopliae ARSEF 4556 and Steinernema carpocapsae. Int. J. Pest Manag. 2016, 62, 261–266. [Google Scholar] [CrossRef]
- Wassermann, M.; Selzer, P.; Steidle, J.L.; Mackenstedt, U. Biological control of Ixodes ricinus larvae and nymphs with Metarhizium anisopliae blastospores. Ticks Tick-Borne Dis. 2016, 7, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Nana, P.; Nchu, F.; Ekesi, S.; Boga, H.I.; Kamtchouing, P.; Maniania, N.K. Efficacy of spot-spray application of Metarhizium anisopliae formulated in emulsifiable extract of Calpurnia aurea in attracting and infecting adult Rhipicephalus appendiculatus ticks in semi-field experiments. J. Pest Sci. 2015, 88, 613–619. [Google Scholar] [CrossRef]
- Ojeda-Chi, M.M.; Rodriguez-Vivas, R.I.; Galindo-Velasco, E.; Lezama-Gutierrrez, R. Laboratory and field evaluation of Metarhizium anisopliae (Deuteromycotina: Hyphomycetes) for the control of Rhipicephalus microplus (Acari: Ixodidae) in the Mexican tropics. Vet. Parasitol. 2010, 170, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Valizadeh, H.; Abbasipour, H.; Mahmoudvand, M.; Askary, H.; Reza Moniri, V. Laboratory Trials of Metarhizium anisopliae var. acridum (Green Muscle®) against the Saxaul locust, Dericorys albidula Serville (Orthoptera: Dericorythidae). Chil. J. Agric. Res. 2011, 71, 549–553. [Google Scholar]
- Klass, J.I.; Blanford, S.; Thomas, M.B. Development of a model for evaluating the effects of environmental temperature and thermal behaviour on biological control of locusts and grasshoppers using pathogens. Agric. For. Entomol. 2007, 9, 189–199. [Google Scholar] [CrossRef]
- Mullié, W.C.; Guèye, Y. Does bird predation enhance the impact of green muscle® (Metarhizium acridum) used for grasshopper control? J. Orthoptera Res. 2010, 19, 139–155. [Google Scholar] [CrossRef]
- Schneider, L.; Silva, C.; Pamphile, J.; Conte, H. Infection, colonization and extrusion of Metarhizium anisopliae (Metsch) Sorokin (Deuteromycotina: Hyphomycetes) in pupae of Diatraea saccharalis F. (Lepidoptera: Crambidae). J. Entomol. Nematol. 2013, 5, 1–9. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aw, K.M.S.; Hue, S.M. Mode of Infection of Metarhizium spp. Fungus and Their Potential as Biological Control Agents. J. Fungi 2017, 3, 30. https://doi.org/10.3390/jof3020030
Aw KMS, Hue SM. Mode of Infection of Metarhizium spp. Fungus and Their Potential as Biological Control Agents. Journal of Fungi. 2017; 3(2):30. https://doi.org/10.3390/jof3020030
Chicago/Turabian StyleAw, Kimberly Moon San, and Seow Mun Hue. 2017. "Mode of Infection of Metarhizium spp. Fungus and Their Potential as Biological Control Agents" Journal of Fungi 3, no. 2: 30. https://doi.org/10.3390/jof3020030
APA StyleAw, K. M. S., & Hue, S. M. (2017). Mode of Infection of Metarhizium spp. Fungus and Their Potential as Biological Control Agents. Journal of Fungi, 3(2), 30. https://doi.org/10.3390/jof3020030




