Microsporidiosis in Vertebrate Companion Exotic Animals
Abstract
:1. Introduction
2. Ornamental Fish Microsporidiosis
2.1. Agents and Disease in Fish
2.2. Diagnosis of Fish Microsporidiosis
2.3. Treatment of Piscine Microsporidiosis
3. Rabbit and Rodents Encephalitozoonosis
3.1. Agent and Disease in Rabbits and Rodents
3.2. Diagnostic Challenge in Rabbits
3.2.1. Antibody Detection, Electrophoresis and Acute Phase Proteins
3.2.2. Antemortem Antigenic Tests
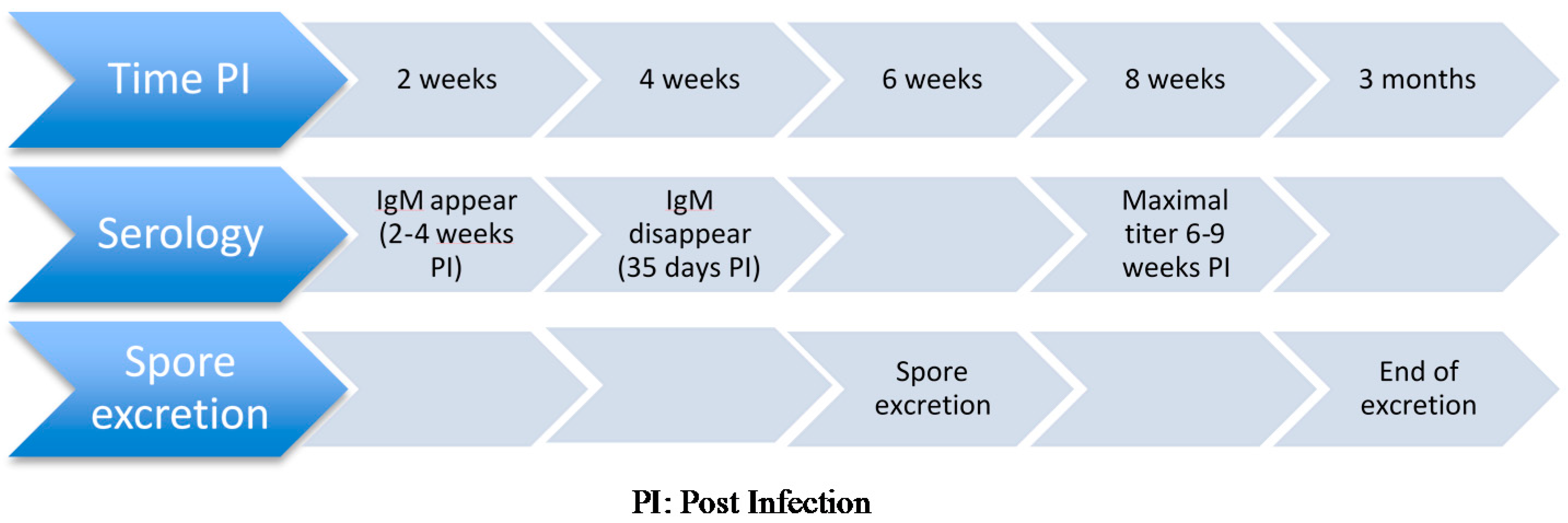
3.2.3. PostMortem Diagnosis
3.3. Therapeutic Challenges
4. Avian, Reptile and Amphibian Microsporidiosis
4.1. Microsporidia in Amphibians
4.2. Microsporidia in Reptiles
4.3. Microsporidia in Birds
4.3.1. Agents and Disease in Birds
| Avian Host Species | Host Latin Name | Microsporidia Species | Characterization Technique Used | Reference |
|---|---|---|---|---|
| Anna’s hummingbird | Calypte anna | Encephalitozoon hellem | PCR | [26] |
| Budgerigars | Melopsittacus undulatus | Encephalitozoon hellem and Encephalitozoon cuniculi | PCR | [142,148] |
| Double yellow-headed amazon | Amazona ochrocephala orathrix | Encephalitozoon sp. | TEM (morphologic) | [150] |
| Eclectus parrots | Eclectus roratus | Encephalitozoon hellem | PCR | [149] |
| Ostrich | Struthio camelus | Encephalitozoon hellem | PCR | [25] |
| Peach-faced lovebird | Agapornis roseicollis | Encephalitozoon sp. | TEM (morphologic) | [145,147] |
| Umbrella cockatoo | Cacatua alba | Encephalitozoon hellem | PCR | [62] |
| Yellow-streaked lori | Chalcopsitta scintillata | Encephalitozoon hellem | PCR | [152] |
4.3.2. Diagnosis of Avian Microsporidiosis
4.3.3. Treatment of Avian Microsporidiosis
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Campbell, S.E.; Williams, T.A.; Yousuf, A.; Soanes, D.M.; Paszkiewicz, K.H.; Williams, B.A. The genome of Spraguea lophii and the basis of host-microsporidian interactions. PLoS Genet. 2013, 9. [Google Scholar] [CrossRef] [PubMed]
- Thomarat, F.; Vivarès, C.P.; Gouy, M. Phylogenetic analysis of the complete genome sequence of Encephalitozoon cuniculi supports the fungal origin of microsporidia and reveals a high frequency of fast-evolving genes. J. Mol. Evol. 2004, 59, 780–791. [Google Scholar] [CrossRef] [PubMed]
- Gill, E.E.; Fast, N.M. Assessing the microsporidia-fungi relationship: Combined phylogenetic analysis of eight genes. Gene 2006, 375, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Heinz, E.; Williams, T.A.; Nakjang, S.; Noël, C.J.; Swan, D.C.; Goldberg, A.V.; Harris, S.R.; Weinmaier, T.; Markert, S.; Becher, D.; et al. The genome of the obligate intracellular parasite Trachipleistophora hominis: New insights into microsporidian genome dynamics and reductive evolution. PLoS Pathog. 2012, 8, e1002979. [Google Scholar] [CrossRef] [PubMed]
- Hibbett, D.S.; Binder, M.; Bischoff, J.F.; Blackwell, M.; Cannon, P.F.; Eriksson, O.E.; Huhndorf, S.; James, T.; Kirk, P.M.; Lucking, R.; et al. A higher-level phylogenetic classification of the fungi. Mycol. Res. 2007, 111, 509–547. [Google Scholar] [CrossRef] [PubMed]
- Xiang, H.; Zhang, R.; de Koeyer, D.; Pan, G.; Li, T.; Liu, T.; Zhou, Z. New evidence on the relationship between microsporidia and fungi: A genome-wide analysis by darkhorse software. Can. J. Microbiol. 2014, 60, 557–568. [Google Scholar] [CrossRef] [PubMed]
- Mathis, A.; Weber, R.; Deplazes, P. Zoonotic potential of the microsporidia. Clin. Microbiol. Rev. 2005, 18, 423–445. [Google Scholar] [CrossRef] [PubMed]
- Richter, B.; Graner, I.; Csokai, J. Heterosporis anguillarum infection in a garter snake (Thamnophis sirtalis). J. Comp. Pathol. 2014, 150, 332–335. [Google Scholar] [CrossRef] [PubMed]
- Pasteur, L. Etudes sur la Maladie des vers a Soie, Moyen Pratique Assuré de la Combattre et d’en Prévenir le Retour; Gauthier-Villars: Paris, France, 1870; p. 434. (In France) [Google Scholar]
- Didier, E.S. Microsporidiosis: An emerging and opportunistic infection in humans and animals. Acta Trop. 2005, 94, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.C.; Nai, Y.S.; Wang, C.Y.; Solter, L.F.; Hsu, H.C.; Wang, C.H.; Lo, C.F. A new microsporidium, Triwangia caridinae gen. nov., sp. nov. Parasitizing fresh water shrimp, Caridina formosae (decapoda: Atyidae) in Taiwan. J. Invertebr. Pathol. 2013, 112, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Noga, E. Problem 70: Microsporidian infection. In Fish Disease Diagnosis and Treatment; Wiley-Blackwell: Ames, IA, USA, 2006; pp. 247–253. [Google Scholar]
- Jacobson, E.R. Chapter 12: Parasites and parasitic diseases of reptiles. In Infectious Diseases and Pathology of Reptiles; CRC Press: Boca Raton, FL, USA, 2007; pp. 571–665. [Google Scholar]
- Bruno, D.W.; Nowak, B.; Elliott, D.G. Guide to the identification of fish protozoan and metazoan parasites in stained tissue sections. Dis. Aquat. Organ. 2006, 70, 1–36. [Google Scholar] [CrossRef] [PubMed]
- Künzel, F.; Joachim, A. Encephalitozoonosis in rabbits. Parasitol. Res. 2010, 106, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Graczyk, T.K.; Cranfield, M.R. Cryptosporidium serpentis oocysts and microsporidian spores in feces of captive snakes. J. Parasitol. 2000, 86, 413–414. [Google Scholar] [CrossRef]
- Ferguson, J.A.; Watral, V.; Schwindt, A.R.; Kent, M.L. Spores of two fish microsporidia (Pseudoloma neurophilia and Glugea anomala) are highly resistant to chlorine. Dis. Aquat. Organ. 2007, 76, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Didier, E.S.; Didier, P.J.; Snowden, K.F.; Shadduck, J.A. Microsporidiosis in mammals. Microbes Infect. 2000, 2, 709–720. [Google Scholar] [CrossRef]
- Sanders, J.L.; Lawrence, C.; Nichols, D.K.; Brubaker, J.F.; Peterson, T.S.; Murray, K.N.; Kent, M.L. Pleistophora hyphessobryconis (microsporidia) infecting zebrafish Danio rerio in research facilities. Dis. Aquat. Organ. 2010, 91, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Sak, B.; Kváč, M.; Kučerová, Z.; Květoňová, D.; Saková, K. Latent microsporidial infection in immunocompetent individuals—A longitudinal study. PLoS Negl. Trop. Dis. 2011, 5, e1162. [Google Scholar] [CrossRef] [PubMed]
- Garcia, L. Laboratory identification of the microsporidia. J. Clin. Microbiol. 2002, 40, 1892–1901. [Google Scholar] [CrossRef] [PubMed]
- Luna, V.A.; Stewart, B.K.; Bergeron, D.L.; Clausen, C.R.; Plorde, J.J.; Fritsche, T.R. Use of the fluorochrome calcofluor white in the screening of stool specimens for spores of microsporidia. Am. J. Clin. Pathol. 1995, 103, 656–659. [Google Scholar] [PubMed]
- Peterson, T.S.; Spitsbergen, J.M.; Feist, S.W.; Kent, M.L. Luna stain, an improved selective stain for detection of microsporidian spores in histologic sections. Dis. Aquat. Organ. 2011, 95, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Mumcuoglu, I.; Cetin, F.; AL Dogruman, F.; Oguz, I.; Aksu, N. Prevalence of microsporidia in healthy individuals and immunocompetent patients with acute and chronic diarrhea. Infect. Dis. 2016, 48, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Snowden, K.; Logan, K. Molecular identification of Encephalitozoon hellem in an ostrich. Avian Dis. 1999, 43, 779–782. [Google Scholar] [CrossRef] [PubMed]
- Snowden, K.; Daft, B.; Nordhausen, R.W. Morphological and molecular characterization of Encephalitozoon hellem in hummingbirds. Avian Pathol. 2001, 30, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Clarridge, J.R.; Karkhanis, S.; Rabeneck, L.; Marino, B.; Foote, L.W. Quantitative light microscopic detection of Enterocytozoon bieneusi in stool specimens: A longitudinal study of human immunodeficiency virus-infected microsporidiosis patients. J. Clin. Microbiol. 1996, 34, 520–523. [Google Scholar] [PubMed]
- Anane, S.; Attouchi, H. Microsporidiosis: Epidemiology, clinical data and therapy. Gastroenterol. Clin. Biol. 2010, 34, 450–464. [Google Scholar] [CrossRef] [PubMed]
- Didier, E.S.; Weiss, L.M. Microsporidiosis: Current status. Curr. Opin. Infect. Dis. 2006, 19, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Song, M.; Wan, Q.; Li, Y.; Lu, Y.; Jiang, Y.; Tao, W.; Li, W. Enterocytozoon bieneusi genotypes in children in Northeast China and assessment of risk of zoonotic transmission. J. Clin. Microbiol. 2014, 52, 4363–4367. [Google Scholar] [CrossRef] [PubMed]
- Ghoshal, U.; Khanduja, S.; Pant, P.; Prasad, K.N.; Dhole, T.N.; Sharma, R.K.; Ghoshal, U.C. Intestinal microsporidiosis in renal transplant recipients: Prevalence, predictors of occurrence and genetic characterization. Indian J. Med. Microbiol. 2015, 33, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Lobo, M.L.; Xiao, L.; Antunes, F.; Matos, O. Microsporidia as emerging pathogens and the implication for public health: A 10-year study on HIV-positive and -negative patients. Int. J. Parasitol. 2012, 42, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Bern, C.; Kawai, V.; Vargas, D.; Rabke-Verani, J.; Williamson, J.; Chavez-Valdez, R.; Xiao, L.; Sulaiman, I.; Vivar, A.; Ticona, E.; et al. The epidemiology of intestinal microsporidiosis in patients with HIV/AIDS in Lima, Peru. J. Infect. Dis. 2005, 191, 1658–1664. [Google Scholar] [CrossRef] [PubMed]
- Hocevar, S.N.; Paddock, C.D.; Spak, C.W.; Rosenblatt, R.; Diaz-Luna, H.; Castillo, I.; Luna, S.; Friedman, G.C.; Antony, S.; Stoddard, R.A.; et al. Microsporidiosis acquired through solid organ transplantation: A public health investigation. Ann. Intern. Med. 2014, 160, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Theng, J.; Chang, C.; Ling, M.L.; Tan, D. Microsporidial keratoconjunctivitis in a healthy contact lens wearer without human immunodeficiency virus infection. Ophthalmology 2001, 108, 976–978. [Google Scholar] [CrossRef]
- Matos, O.; Lobo, M.; Xiao, L. Epidemiology of Enterocytozoon bieneusi infection in humans. J. Parasitol. Res. 2012, 2012, 981424. [Google Scholar] [CrossRef] [PubMed]
- Dascomb, K.; Frazer, T.; Clark, R.A.; Kissinger, P.; Didier, E. Microsporidiosis and HIV. J. Acquir. Immune Defic. Syndr. 2000, 24, 290–292. [Google Scholar] [CrossRef] [PubMed]
- Horváth, M.; Leng, L.; Stefkovic, M.; Révajová, V.; Halanová, M. Lethal encephalitozoonosis in cyclophosphamide-treated rabbits. Acta Vet. Hung. 1999, 47, 85–93. [Google Scholar] [PubMed]
- Jeklova, E.; Leva, L.; Kovarcik, K.; Matiasovic, J.; Kummer, V.; Maskova, J.; Skoric, M.; Faldyna, M. Experimental oral and ocular Encephalitozoon cuniculi infection in rabbits. Parasitology 2010, 137, 1749–1757. [Google Scholar] [CrossRef] [PubMed]
- Didier, E.; Vossbrinck, C.R.; Baker, M.D.; Rogers, L.B.; Bertucci, D.C.; Shadduck, J.A. Identification and characterization of three Encephalitozoon cuniculi strains. Parasitology 1995, 111, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Rabinowitz, P.; Conti, L. Human-animal Medicine: Clinical Approachto Zoonoses, Toxicants and Other Shared Health Risks; Saunders Elsevier: Maryland Heights, MO, USA, 2010. [Google Scholar]
- Słodkowicz-Kowalska, A. Animal reservoirs of human virulent microsporidian species. Wiad. Parazytol. 2009, 55, 63–65. [Google Scholar] [PubMed]
- Zhao, W.; Zhang, W.; Wang, R.; Liu, W.; Liu, A.; Yang, D.; Yang, F.; Karim, M.R.; Zhang, L. Enterocytozoon bieneusi in sika deer (Cervus nippon) and red deer (Cervus elaphus): Deer specificity and zoonotic potential of its genotypes. Parasitol. Res. 2014, 113, 4243–4250. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Huang, J.; Karim, M.R.; Zhao, J.; Dong, H.; Ai, W.; Li, F.; Zhang, L.; Wang, R. Zoonotic Enterocytozoon bieneusi genotypes in Pere David’s deer (Elaphurus davidianus) in Henan, C-China. Exp. Parasitol. 2015, 155, 46–48. [Google Scholar] [CrossRef] [PubMed]
- Karim, M.R.; Wang, R.; Dong, H.; Zhang, L.; Li, J.; Zhang, S.; Rume, F.I.; Qi, M.; Jian, F.; Sun, M.; et al. Genetic polymorphism and zoonotic potential of Enterocytozoon bieneusi from nonhuman primates in China. Appl. Environ. Microbiol. 2014, 80, 1893–1898. [Google Scholar] [CrossRef] [PubMed]
- Cama, V.A.; Pearson, J.; Cabrera, L.; Pacheco, L.; Gilman, R.; Meyer, S.; Ortega, Y.; Xiao, L. Transmission of Enterocytozoon bieneusi between a child and guinea pigs. J. Clin. Microbiol. 2007, 45, 2708–2710. [Google Scholar] [CrossRef] [PubMed]
- Lom, J. A catalogue of described genera and species of microsporidians parasitic in fish. Syst. Parasitol. 2002, 53, 81–99. [Google Scholar] [CrossRef] [PubMed]
- Garner, M.M. A retrospective study of disease in elasmobranchs. Vet. Pathol. 2013, 50, 377–389. [Google Scholar] [CrossRef] [PubMed]
- Diamant, A.; Goren, M.; Yokes, M.B.; Galil, B.S.; Klopman, Y.; Huchon, D.; Szitenberg, A.; Karhan, S.U. Dasyatispora levantinae gen. et sp. nov., a new microsporidian parasite from the common stingray Dasyatis pastinaca in the eastern mediterranean. Dis. Aquat. Organ. 2010, 91, 137–150. [Google Scholar] [CrossRef] [PubMed]
- Lom, J.; Dykova, I. Microsporidian xenomas in fish seen in wider perspective. Folia Parasitol. 2005, 52, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Weber, R.; Bryan, R.T.; Schwartz, D.A.; Owen, R.L. Human microsporidial infections. Clin. Microbiol. Rev. 1994, 7, 426–461. [Google Scholar] [PubMed]
- Post, G. Chapter VII: Animal parasites of fishes. In Textbook of Fish Health, 2nd ed.; Post, G., Ed.; T.F.H. Publications, Inc.: Neptune City, NJ, USA, 1987; pp. 172–174. [Google Scholar]
- Sanders, J.L.; Peterson, T.S.; Kent, M.L. Early development and tissue distribution of Pseudoloma neurophilia in the zebrafish, Danio rerio. J. Eukaryot. Microbiol. 2014, 61, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Sanders, J.L.; Watral, V.; Kent, M.L. Microsporidiosis in zebrafish research facilities. ILAR J. 2012, 53, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Murray, K.; Dreska, M.; Nasiadka, A.; Rinne, M.; Matthews, J.L.; Carmichael, C.; Bauer, J.; Varga, Z.M.; Westerfield, M. Comparative medicine, transmission, diagnosis, and recommendations for control of Pseudoloma neurophilia infections in laboratory zebrafish (Danio rerio) facilities. Comp. Med. 2011, 61, 322–329. [Google Scholar] [PubMed]
- Kent, M.L.; Speare, D.J. Review of the sequential development of Loma salmonae (microsporidia) based on experimental infections of rainbow trout (Oncorhynchus mykiss) and chinook salmon (O. tshawytscha). Folia Parasitol. 2005, 52, 63–68. [Google Scholar] [CrossRef] [PubMed]
- McConnachie, S.; Sheppard, J.; Wright, G.M.; Speare, D.J. Development of the microsporidian parasite, Loma salmonae, in a rainbow trout gill epithelial cell line (RTG-1): Evidence of xenoma development in vitro. Parasitology 2015, 142, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Becker, J.; Speare, D. Ultraviolet light control of horizontal transmission of Loma salmonae. J. Fish Dis. 2004, 27, 177–180. [Google Scholar] [CrossRef] [PubMed]
- Leiro, J.M.; Piazzon, C.; Dominguez, B.; Mallo, N.; Lamas, J. Evaluation of some physical and chemical treatments for inactivating microsporidian spores isolated from fish. Int. J. Food Microbiol. 2012, 156, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Speare, D.J.; Athanassopoulou, F.; Daley, J.; Sanchez, J.G. A preliminary investigation of alternatives to fumagillin for the treatment of Loma salmonae infection in rainbow trout. J. Comp. Pathol. 1999, 121, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Lauren, D.J.; Wishkovsky, A.; Groff, J.M.; Hedrick, R.P.; Hinton, D.E. Toxicity and pharmacokinetics of the antibiotic fumagillin in yearling rainbow trout (Salmo gairdneri). Toxicol. Appl. Pharmacol. 1989, 98, 444–453. [Google Scholar] [CrossRef]
- Phalen, D.N.; Logan, K.S.; Snowden, K.F. Encephalitozoon hellem infection as the cause of a unilateral chronic keratoconjunctivitis in an umbrella cockatoo (Cacatua alba). Vet. Ophthalmol. 2006, 9, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Speare, D.J.; Ritter, G.; Schmidt, H. Quinine hydrochloride treatment delays xenoma formation and dissolution in rainbow trout challenged with Loma salmonae. J. Comp. Pathol. 1998, 119, 459–465. [Google Scholar] [CrossRef]
- Becker, J.A.; Speare, D.J.; Daley, J.; Dick, P. Effects of monensin dose and treatment time on xenoma reduction in microsporidial gill disease in rainbow trout, Oncorhynchus mykiss (walbaum). J. Fish Dis. 2002, 25, 673–680. [Google Scholar] [CrossRef]
- Food Animal Residue Avoidance Databank, Veterinarian’s Guide to Residue Avoidance Management. 2015. Available online: http://www.farad.org/ (accessed on 5 December 2015).
- Daniel, P. Available chemotherapy in mediterranean fish farming: Use and needs. In The Use of Veterinary Drugs and Vaccines in Mediterranean Aquaculture; Rogers, C., Basurco, B., Eds.; CIHEAM: Zaragoza, Spain, 2009; Volume 86, pp. 197–205. [Google Scholar]
- Health Canada. List of Veterinary Drugs That are Authorized for Sale by Health Canada for Use in Food-producing Aquatic Animals. 2010. Available online: http://www.hc-sc.gc.ca/dhp-mps/vet/legislation/pol/aquaculture_anim-eng.php (accessed on 5 July 2015).
- Rodríguez-Tovar, L.; Becker, J.A.; Markham, R.J.; Speare, D. Induction time for resistance to microsporidial gill disease caused by loma salmonae following vaccination of rainbow trout (Oncorhynchus mykiss) with a spore-based vaccine. Fish Shellfish Immunol. 2006, 21, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Speare, D.; Markham, R.; Guselle, N. Development of an effective whole-spore vaccine to protect against microsporidial gill disease in rainbow trout (Oncorhynchus mykiss) by using a low-virulence strain of Loma salmonae. Clin. Vaccine Immunol. 2007, 14, 1652–1654. [Google Scholar] [CrossRef] [PubMed]
- Snowden, K.; Logan, K.; Didier, E.S. Encephalitozoon cuniculi strain III is a cause of encephalitozoonosis in both humans and dogs. J. Infect. Dis. 1999, 180, 2086–2088. [Google Scholar] [CrossRef] [PubMed]
- Reetz, J.; Wiedemann, M.; Aue, A.; Wittstatt, U.; Ochs, A.; Thomschke, A.; Manke, H.; Schwebs, M.; Rinder, H. Disseminated lethal Encephalitozoon cuniculi (genotype III) infections in cotton-top tamarins (Oedipomidas oedipus)-a case report. Parasitol. Int. 2004, 53, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Didier, E.S.; Visvesvara, G.S.; Baker, M.D.; Rogers, L.B.; Bertucci, D.C.; de Groote, M.A.; Vossbrinck, C.R. A microsporidian isolated from an aids patient corresponds to Encephalitozoon cuniculi III, originally isolated from domestic dogs. J. Clin. Microbiol. 1996, 34, 2835–2837. [Google Scholar] [PubMed]
- Sokolova, O.I.; Demyanov, A.V.; Bowers, L.C.; Didier, E.S.; Yakovlev, A.V.; Skarlato, S.O.; Sokolova, Y.Y. Emerging microsporidian infections in russian HIV-infected patients. J. Clin. Microbiol. 2011, 49, 2102–2108. [Google Scholar] [CrossRef] [PubMed]
- Talabani, H.; Sarfati, C.; Pillebout, E.; van Gool, T.; Derouin, F.; Menotti, J. Disseminated infection with a new genovar of Encephalitozoon cuniculi in a renal transplant recipient. J. Clin. Microbiol. 2010, 48, 2651–2653. [Google Scholar] [CrossRef] [PubMed]
- Benz, P.; Maass, G.; Csokai, J.; Fuchs-Baumgartinger, A.; Schwendenwein, I.; Tichy, A.; Nell, B. Detection of Encephalitozoon cuniculi in the feline cataractous lens. Vet. Ophthalmol. 2011, 14, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Richter, B.; Csokai, J.; Graner, I.; Eisenberg, T.; Pantchev, N.; Eskens, H.U.; Nedorost, N. Encephalitozoonosis in two inland bearded dragons (Pogona vitticeps). J. Comp. Pathol. 2013, 148, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Percy, D.; Barthold, S. Chapter 6: Rabbit, parasitic diseases. In Pathology of Laboratory Rodents and Rabbits, 3rd ed.; Percy, D., Barthold, S., Eds.; Blackwell Publishing: Ames, IA, USA, 2007; pp. 287–294. [Google Scholar]
- Künzel, F.; Gruber, A.; Tichy, A.; Edelhofer, R.; Nell, B.; Hassan, J.; Leschnik, M.; Thalhammer, J.G.; Joachim, A. Clinical symptoms and diagnosis of encephalitozoonosis in pet rabbits. Vet. Parasitol. 2008, 151, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Berger Baldotto, S.; Cray, C.; Turner Giannico, A.; Reifer, L.; Montiani-Ferreira, F. Seroprevalence of Encephalitozoon cuniculi infection in pet rabbits in Brazil. J. Exot. Pet Med. 2015, 24, 435–440. [Google Scholar] [CrossRef]
- Anete Lallo, M.; Porta Miche Hirschfeld, M. Encephalitozoonosis in pharmacologically immunosuppressed mice. Exp. Parasitol. 2012, 131, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Kunstyr, I.; Lev, L.; Naumann, S. Humoral antibody response of rabbits to experimental infection with Encephalitozoon cuniculi. Vet. Parasitol. 1986, 21, 223–232. [Google Scholar] [CrossRef]
- Grüber, A.; Pakozdy, A.; Weissenböck, H.; Csokai, J.; Künzel, F. A retrospective study of neurological disease in 118 rabbits. J. Comp. Pathol. 2009, 140, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Leipig, M.; Matiasek, K.; Rinder, H.; Janik, D.; Emrich, D.; Baiker, K.; Hermanns, W. Value of histopathology, immunohistochemistry, and real-time polymerase chain reaction in the confirmatory diagnosis of Encephalitozoon cuniculi infection in rabbits. J. Vet. Diagn. Investig. 2012, 25, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Reusch, B.; Murray, J.K.; Papasouliotis, K.; Redrobe, S.P. Urinary protein:creatinine ratio in rabbits in relation to their serological status to Encephalitozoon cuniculi. Vet. Rec. 2009, 164, 293–295. [Google Scholar] [CrossRef] [PubMed]
- Percy, D.; Barthold, S. Chapter 1: Mouse. In Pathology of Laboratory Rodents and Rabbits, 3rd ed.; Percy, D., Barthold, S., Eds.; Blackwell Publishing: Ames, IA, USA, 2007; pp. 3–124. [Google Scholar]
- Jeklova, E.; Jekl, V.; Kovarcik, K.; Hauptman, K.; Koudela, B.; Neumayerova, H.; Knotek, Z.; Faldyna, M. Usefulness of detection of specific IgM and IgG antibodies for diagnosis of clinical encephalitozoonosis in pet rabbits. Vet. Parasitol. 2010, 170, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Csokai, J.; Gruber, A.; Kunzel, F.; Tichy, A.; Joachim, A. Encephalitozoonosis in pet rabbits (Oryctolagus cuniculus): Pathohistological findings in animals with latent infection versus clinical manifestation. Parasitol. Res. 2009, 104, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Waller, T.; Morein, B.; Fabiansson, E. Humoral immune response to infection with Encephalitozoon cuniculi in rabbits. Lab. Anim. 1978, 12, 145–148. [Google Scholar] [CrossRef] [PubMed]
- Cray, C.; Rodriguez, M.; Fernandez, Y. Acute phase protein levels in rabbits with suspected Encephalitozoon cuniculi infection. J. Exot. Pet Med. 2013, 22, 280–286. [Google Scholar] [CrossRef]
- Sobottka, I.; Iglauer, F.; Schuler, T.; Schmetz, C.; Visvesvara, G.S.; Albrecht, H.; Schwartz, D.A.; Pieniazek, N.J.; Bartscht, K.; Laufs, R.; et al. Acute and long-term humoral immunity following active immunization of rabbits with inactivated spores of various Encephalitozoon species. Parasitol. Res. 2001, 87, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Cox, J.C.; Gallichio, H.A. Serological and histological studies on adult rabbits with recent, naturally acquired encephalitozoonosis. Res. Vet. Sci. 1978, 24, 260–261. [Google Scholar] [PubMed]
- Lyngset, A. A survey of serum antibodies to Encephalitozoon cuniculi in breeding rabbits and their young. Lab. Anim. Sci. 1980, 30, 558–561. [Google Scholar] [PubMed]
- Harcourt-Brown, F.M.; Holloway, H.K. Encephalitozoon cuniculi in pet rabbits. Vet. Rec. 2003, 152, 427–431. [Google Scholar] [CrossRef] [PubMed]
- Csokai, J.; Joachim, A.; Gruber, A.; Tichy, A.; Pakozdy, A.; Kunzel, F. Diagnostic markers for encephalitozoonosis in pet rabbits. Vet. Parasitol. 2009, 163, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, M.; Oohashi, E.; Dautu, G.; Ueno, A.; Kariya, T.; Furuya, K. High seroprevalence of Encephalitozoon cuniculi in pet rabbits in Japan. J. Vet. Med. Sci. 2008, 70, 1301–1304. [Google Scholar] [CrossRef] [PubMed]
- Cray, C.; Arcia, G.; Schneider, R.; Kelleher, S.A.; Arheart, K.L. Evaluation of the usefulness of an ELISA and protein electrophoresis in the diagnosis of Encephalitozoon cuniculi infection in rabbits. Am. J. Vet. Res. 2009, 70, 478–482. [Google Scholar] [CrossRef] [PubMed]
- Cray, C.; McKenny, S.; Perritt, E.; Arheart, K.L. Utility of IgM titers with IgG and C-reactive protein quantification in the diagnosis of suspected Encephalitozoon cuniculi infection in rabbits. J. Exot. Pet Med. 2015, 24, 356–360. [Google Scholar] [CrossRef]
- Furuya, K.; Asakura, T.; Igarashi, M.; Morita, T. Microsporidian Encephalitozoon cuniculi antibodies in rabbit urine samples. Vet. Rec. 2009, 165, 85–86. [Google Scholar] [CrossRef] [PubMed]
- Jass, A.; Matiasek, K.; Henke, J.; Küchenhoff, H.; Hartmann, K.; Fischer, A. Analysis of cerebrospinal fluid in healthy rabbits and rabbits with clinically suspected encephalitozoonosis. Vet. Rec. 2008, 162, 618–622. [Google Scholar] [CrossRef] [PubMed]
- Sieg, J.; Hein, J.; Jass, A.; Sauter-Louis, C.; Hartmann, K.; Fischer, A. Clinical evaluation of therapeutic success in rabbits with suspected encephalitozoonosis. Vet. Parasitol. 2012, 187, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Lallo, M.A.; Dos Santos, M.J.; Bondan, E.F. Experiental Encephalitozoon cuniculi infection in dexamethasone-immunosuppressed mice. Rev. Saude Publica 2006, 36, 621–626. [Google Scholar]
- Herich, R.; Levkutova, M.; Kokincakova, T.; Reiterova, K.; Hipikova, V.; Levkut, M. Diagnosis and manifestation of encephalitozoonosis in mice after experimental infection with different species and application of dexamethasone. J. Vet. Med. Ser. A 2009, 53, 340–345. [Google Scholar] [CrossRef] [PubMed]
- Felchle, L.M.; Sigler, R.L. Phacoemulsification for the management of Encephalitozoon cuniculi-induced phacoclastic uveitis in a rabbit. Vet. Ophthalmol. 2002, 5, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Habenbacher, B.; Klang, A.; Fragner, K.; Dinhopl, N.; Kunzel, F.; Weissenbock, H. Comparative evaluation of specific methods for labeling of Encephalitozoon cuniculi in paraffin wax-embedded tissue samples. J. Vet. Diagn. Investig. 2012, 24, 370–375. [Google Scholar] [CrossRef] [PubMed]
- Suter, C.; Müller-Doblies, U.U.; Hatt, J.M.; Deplazes, P. Prevention and treatment of Encephalitozoon cuniculi infection in rabbits with fenbendazole. Vet. Rec. 2001, 148, 478–480. [Google Scholar] [CrossRef] [PubMed]
- Franssen, F.F.; Lumeij, J.T.; van Knapen, F. Susceptibility of Encephalitozoon cuniculi to several drugs in vitro. Antimicrob. Agents Chemother. 1995, 39, 1265–1268. [Google Scholar] [CrossRef] [PubMed]
- Didier, E.S.; Bowers, L.; Stovall, M.E.; Kuebler, D.; Mittleider, D.; Brindley, P.J.; Didier, P.J. Antimicrosporidial activity of (fluoro)quinolones in vitro and in vivo. Folia Parasitol. 2005, 52, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Marangos, M.N.; Zhu, Z.; Nicolau, D.P.; Klepser, M.E.; Nightingale, C.H. Disposition of ofloxacin in female New Zealand white rabbits. J. Vet. Pharmacol. Ther. 1997, 20, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Short, C.R.; Barker, S.A.; Hsieh, L.C.; Ou, S.P.; McDowell, T. Disposition of fenbendazole in the rabbit. Res. Vet. Sci. 1988, 44, 215–219. [Google Scholar] [PubMed]
- Short, C.R.; Flory, W.; Hsieh, L.C.; Barker, S.A. The oxidative metabolism of fenbendazole: A comparative study. J. Vet. Pharmacol. Ther. 1988, 11, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Graham, J.; Garner, M.; Reavill, D. Benzimidazole toxicosis in rabbits: 13 cases (2003 to 2011). J. Exot. Pet Med. 2014, 23, 188–195. [Google Scholar] [CrossRef]
- Fischer, P.; Carpenter, J.W. Chapter 19: Neurologic and musculoskeletal diseases. In Ferrets, Rabbits and Rodents Medicine and Surgery, 3rd ed.; Elsevier: St. Louis, MO, USA, 2013; pp. 245–256. [Google Scholar]
- Kotkova, M.; Sak, B.; Kvetonova, D.; Kvac, M. Latent microsporidiosis caused by Encephalitozoon cuniculi in immunocompetent hosts: A murine model demonstrating the ineffectiveness of the immune system and treatment with albendazole. PLoS ONE 2013, 8, e60941. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.; Hurtado, M.; Medina, M.T.; Sanchez, M.; Sotelo, J. Dexamethasone increases plasma levels of albendazole. J. Neurol. 1990, 237, 279–280. [Google Scholar] [CrossRef] [PubMed]
- Takayanagui, O.M.; Lanchote, V.L.; Marques, M.P.; Bonato, P.S. Therapy for neurocysticercosis: Pharmacokinetic interaction of albendazole sulfoxide with dexamethasone. Ther. Drug Monit. 1997, 19, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Daniel, B.J.; Brumlik, M.J.; Burow, M.E.; Zou, W.; Khan, I.A.; Wadsworth, S.; Siekierka, J.; Curiel, T.J. Drugs designed to inhibit human p38 mitogen-activated protein kinase activation treat Toxoplasma gondii and Encephalitozoon cuniculi infection. Antimicrob. Agents Chemother. 2007, 51, 4324–4328. [Google Scholar] [CrossRef] [PubMed]
- Shadduck, J.A. Effect of fumagillin on in vitro multiplication of Encephalitozoon cuniculi. J. Protozool. 1980, 27, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, M. Therapeutic review: Ponazuril. J. Exot. Pet Med. 2008, 17, 228–229. [Google Scholar] [CrossRef]
- Beauvais, B.; Sarfati, C.; Challier, S.; Derouin, F. In vitro model to assess effect of antimicrobial agents on Encephalitozoon cuniculi. Antimicrob. Agents Chemother. 1994, 38, 2440–2448. [Google Scholar] [CrossRef] [PubMed]
- Waller, T. Sensitivity of Encephalitozoon cuniculi to various temperatures, disinfectants and drugs. Lab. Anim. 1979, 13, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Jordan, C.N.; Dicristina, J.A.; Lindsay, D.S. Activity of bleach, ethanol and two commercial disinfectants against spores of Encephalitozoon cuniculi. Vet. Parasitol. 2006, 136, 343–346. [Google Scholar] [CrossRef] [PubMed]
- Poynton, S.; Whitaker, B.R. Protozoa and metazoa infecting amphibians. In Amphibian Medicine and Captive Husbandry; Wright, G.M., Whitaker, B.R., Eds.; Krieger Publishing Co: Malabar, FL, USA, 2001; pp. 401–495. [Google Scholar]
- Canning, E.U.; Elkan, E.; Trigg, P.I. Plistophora myotrophica spec. nov., causing high mortality in the common toad Bufo bufo L., with notes on the maintenance of Bufo and Xenopus in the laboratory. J. Protozool. 1964, 11, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Paperna, I.; Lainson, R. Alloglugea bufonis nov. gen., nov. sp. (microsporidia: Glugeidae), a microsporidian of Bufo marinus tadpoles and metamorphosing toads (amphibia: Anura) from Amazon Brazil. Dis. Aquat. Organ. 1995, 23, 7–16. [Google Scholar] [CrossRef]
- Graczyk, T.K.; Cranfield, M.R.; Bicknese, E.J.; Wisnieski, A.P. Progressive ulcerative dermatitis in captive, wild-caught, south-american giant tree frog (Phyllomedusa bicolor) with microsporidial septicemia. J. Zoo Wildl. Med. 1996, 27, 522–527. [Google Scholar]
- Gamble, K.; Garner, M.M.; West, E.S.; West, G.; Didier, E.S.; Cali, A.; Alvarado, T.P. Kyphosis associated with microsporidial myositis in San Marcos salamanders. J. Herpetol. Med. Surg. 2005, 15, 14–17. [Google Scholar]
- Pessier, A.P. Infectious diseases of amphibians: It isn’t just redleg anymore. In Current Therapy in Reptile Medicine and Surgery; Divers, S., Mader, D.R., Eds.; Elsevier: St. Louis, MO, USA, 2014; pp. 247–254. [Google Scholar]
- Liu, S.K.; King, F.W. Microsporidiosis in the tuatara. J. Am. Vet. Med. Assoc. 1971, 159, 1578–1582. [Google Scholar] [PubMed]
- Jacobson, E.R.; Green, D.E.; Undeen, A.H.; Cranfield, M.; Vaughn, K.L. Systemic microsporidiosis in inland bearded dragons (Pogona vitticeps). J. Zoo Wildl. Med. 1998, 29, 315–323. [Google Scholar] [PubMed]
- Guyénot, E.; Naville, A. Recherches sur le parasitisme et l’evolution d’une microsporidie Glugea danilewskyi l. Pfr. Revue Suisse Zool. 1922, 30, 1–61. [Google Scholar]
- Narasimhamurti, C.; Kalavati, C.; Ahamed, S. A new microsporidian, Plistophora atretii sp. n. From the freshwater snake Atretium schistosum Gunther. Acta Protozool. 1982, 21, 127–137. [Google Scholar]
- Canning, E.U. Encephalitozoon lacertae n. sp., a microsporidian parasite of the lizard Podarcis muralis. In Society of Protozoologists Special Publication, No 1; Canning, E.U., Ed.; Society of Protozoologists: Utica, NY, USA, 1981; pp. 57–64. [Google Scholar]
- Koudela, B.; Didier, E.S.; Rogers, L.B.; Modry, D.; Kucerova, S. Intestinal microsporidiosis in african skink Mabuya perrotetii. Folia Parasitol. 1998, 45, 149–155. [Google Scholar] [PubMed]
- Malcekova, B.; Valencakova, A.; Molnar, L.; Kocisova, A. First detection and genotyping of human-associated microsporidia in wild waterfowl of Slovakia. Acta Parasitol. 2013, 58, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Pirestani, M.; Sadraei, J.; Forouzandeh, M. Molecular characterization and genotyping of human related microsporidia in free-ranging and captive pigeons of Tehran, Iran. Infect. Genet. Evol. 2013, 20, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Kasickova, D.; Sak, B.; Kvac, M.; Ditrich, O. Detection of Encephalitozoon cuniculi in a new host—Cockateel (Nymphicus hollandicus) using molecular methods. Parasitol. Res. 2007, 101, 1685–1688. [Google Scholar] [CrossRef] [PubMed]
- Kasickova, D.; Sak, B.; Kvac, M.; Ditrich, O. Sources of potentially infectious human microsporidia: Molecular characterisation of microsporidia isolates from exotic birds in the Czech republic, prevalence study and importance of birds in epidemiology of the human microsporidial infections. Vet. Parasitol. 2009, 165, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Lee, S.S.; Lyoo, Y.S.; Park, H.M. DNA detection and genotypic identification of potentially human-pathogenic microsporidia from asymptomatic pet parrots in South Korea as a risk factor for zoonotic emergence. Appl. Environ. Microbiol. 2011, 77, 8442–8444. [Google Scholar] [CrossRef] [PubMed]
- Slodkowicz-Kowalska, A.; Graczyk, T.K.; Nowosad, A.; Majewska, A.C. First detection of microsporidia in raised pigeons in poland. Ann. Agric. Environ. Med. 2013, 20, 13–15. [Google Scholar] [PubMed]
- Lallo, M.A.; Calabria, P.; Milanelo, L. Encephalitozoon and Enterocytozoon (microsporidia) spores in stool from pigeons and exotic birds: Microsporidia spores in birds. Vet. Parasitol. 2012, 190, 418–422. [Google Scholar] [CrossRef] [PubMed]
- Haro, M.; Izquierdo, F.; Henriques-Gil, N.; Andres, I.; Alonso, F.; Fenoy, S.; del Aguila, C. First detection and genotyping of human-associated microsporidia in pigeons from urban parks. Appl. Environ. Microbiol. 2005, 71, 3153–3157. [Google Scholar] [CrossRef] [PubMed]
- Sak, B.; Kasickova, D.; Kvac, M.; Kvetonova, D.; Ditrich, O. Microsporidia in exotic birds: Intermittent spore excretion of Encephalitozoon spp. In naturally infected budgerigars (Melopsittacus undulatus). Vet. Parasitol. 2010, 168, 196–200. [Google Scholar] [CrossRef] [PubMed]
- Reetz, J.; Rinder, H.; Thomschke, A.; Manke, H.; Schwebs, M.; Bruderek, A. First detection of the microsporidium Enterocytozoon bieneusi in non-mammalian hosts (chickens). Int. J. Parasitol. 2002, 32, 785–787. [Google Scholar] [CrossRef]
- Kemp, R.L.; Kluge, J.P. Encephalitozoon sp. in the blue-masked lovebird, Agapornis personata (Reichenow): First confirmed report of microsporidan infection in birds. J. Protozool. 1975, 22, 489–491. [Google Scholar] [CrossRef] [PubMed]
- Novilla, M.N. Microsporidian infection in the pied peach-faced lovebird (Agapornis roseicollis). Avian Dis. 1978, 22, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Randall, C.J.; Lees, S.; Higgins, R.J.; Harcourt-Brown, N.H. Microsporidian infection in lovebirds (Agapornis spp.). Avian Pathol. 1986, 15, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Norton, J.H.; Prior, H.C. Microsporidiosis in a peach-faced lovebird (Agapornis roseicollis). Aust. Vet. J. 1994, 71, 23–24. [Google Scholar] [CrossRef] [PubMed]
- Black, S.S.; Steinohrt, L.A.; Bertucci, D.C.; Rogers, L.B.; Didier, E.S. Encephalitozoon hellem in budgerigars (Melopsittacus undulatus). Vet. Pathol. 1997, 34, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Pulparampil, N.; Graham, D.; Phalen, D.; Snowden, K. Encephalitozoon hellem in two eclectus parrots (Eclectus roratus): Identification from archival tissues. J. Eukaryot. Microbiol. 1998, 45, 651–655. [Google Scholar] [CrossRef] [PubMed]
- Poonacha, K.B.; William, P.D.; Stamper, R.D. Encephalitozoonosis in a parrot. J. Am. Vet. Med. Assoc. 1985, 186, 700–702. [Google Scholar] [PubMed]
- Canny, C.; Ward, D.; Patton, S. Microsporidian keratoconjunctivitis in a double yellow-headed amazon parrot (Amazona ochrocephala orathrix). J. Avian Med. Surg. 1999, 13, 279–286. [Google Scholar]
- Suter, C.; Mathis, A.; Hoop, R.; Deplazes, P. Encephalitozoon hellem infection in a yellow-streaked lory (Chalcopsitta scintillata) imported from indonesia. Vet. Rec. 1998, 143, 694–695. [Google Scholar] [PubMed]
- Gray, M.L.; Puette, M.; Latimer, K.S. Microsporidiosis in a young ostrich (Struthio camelus). Avian Dis. 1998, 42, 832–836. [Google Scholar] [CrossRef] [PubMed]
- Malcekova, B.; Valencakova, A.; Luptakova, L.; Molnar, L.; Ravaszova, P.; Novotny, F. First detection and genotyping of Encephalitozoon cuniculi in a new host species, gyrfalcon (Falco rusticolus). Parasitol. Res. 2011, 108, 1479–1482. [Google Scholar] [CrossRef] [PubMed]
- Slodkowicz-Kowalska, A.; Graczyk, T.K.; Tamang, L.; Jedrzejewski, S.; Nowosad, A.; Zduniak, P.; Solarczyk, P.; Girouard, A.S.; Majewska, A.C. Microsporidian species known to infect humans are present in aquatic birds: Implications for transmission via water? Appl. Environ. Microbiol. 2006, 72, 4540–4544. [Google Scholar] [CrossRef] [PubMed]
- Lightfoot, T.; Yeager, J. Pet bird toxicity and related environmental concerns. Vet. Clin. N. Am. 2008, 11, 229–259. [Google Scholar] [CrossRef] [PubMed]
- Howard, L.L.; Papendick, R.; Stalis, I.H.; Allen, J.L.; Sutherland-Smith, M.; Zuba, J.R.; Ward, D.L.; Rideout, B.A. Fenbendazole and albendazole toxicity in pigeons and doves. J. Avian Med. Surg. 2002, 16, 203–210. [Google Scholar] [CrossRef]
- Gozalo, A.S.; Schwiebert, R.S.; Lawson, G.W. Mortality associated with fenbendazole administration in pigeons (Columba livia). J. Am. Assoc. Lab. Anim. Sci. 2006, 45, 63–66. [Google Scholar] [PubMed]
- Bonar, C.; Lewandowski, A.; Schaul, J. Suspected fenbendazole toxicosis in 2 vulture species (Gyps africanus, Torgos tracheliotus) and marabou storks (Leptoptilos crumeniferus). J. Avian Med. Surg. 2003, 17, 16–19. [Google Scholar] [CrossRef]
- Weber, M.A.; Terrell, S.P.; Neiffer, D.L.; Miller, M.A.; Mangold, B.J. Bone marrow hypoplasia and intestinal crypt cell necrosis associated with fenbendazole administration in five painted storks. J. Am. Vet. Med. Assoc. 2002, 221, 417–419, 369. [Google Scholar] [CrossRef] [PubMed]
- Weber, M. Chapter 13: Pelicaniformes. In Zoo and Wildlife Medicine, 5th ed.; Fowler, M.E., Miller, R., Eds.; Sauders: St. Louis, MO, USA, 2003; pp. 118–122. [Google Scholar]
- Wiley, J.L.; Whittington, J.K.; Wilmes, C.M.; Messick, J.B. Chronic myelogenous leukemia in a great horned owl (Bubo virginianus). J. Avian Med. Surg. 2009, 23, 36–43. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vergneau-Grosset, C.; Larrat, S. Microsporidiosis in Vertebrate Companion Exotic Animals. J. Fungi 2016, 2, 3. https://doi.org/10.3390/jof2010003
Vergneau-Grosset C, Larrat S. Microsporidiosis in Vertebrate Companion Exotic Animals. Journal of Fungi. 2016; 2(1):3. https://doi.org/10.3390/jof2010003
Chicago/Turabian StyleVergneau-Grosset, Claire, and Sylvain Larrat. 2016. "Microsporidiosis in Vertebrate Companion Exotic Animals" Journal of Fungi 2, no. 1: 3. https://doi.org/10.3390/jof2010003
APA StyleVergneau-Grosset, C., & Larrat, S. (2016). Microsporidiosis in Vertebrate Companion Exotic Animals. Journal of Fungi, 2(1), 3. https://doi.org/10.3390/jof2010003





