Shifting Paradigms in Antifungal Prophylaxis and Their Effects on Candidemia Outcomes in Hematological Malignancies: A 14-Year Experience from a Single Tertiary Center
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Data Collection
2.2. Definitions
2.3. Microbiology and Susceptibility Tests
2.4. Statistical Analysis
3. Results
3.1. Characteristics of Patients with Candidemia
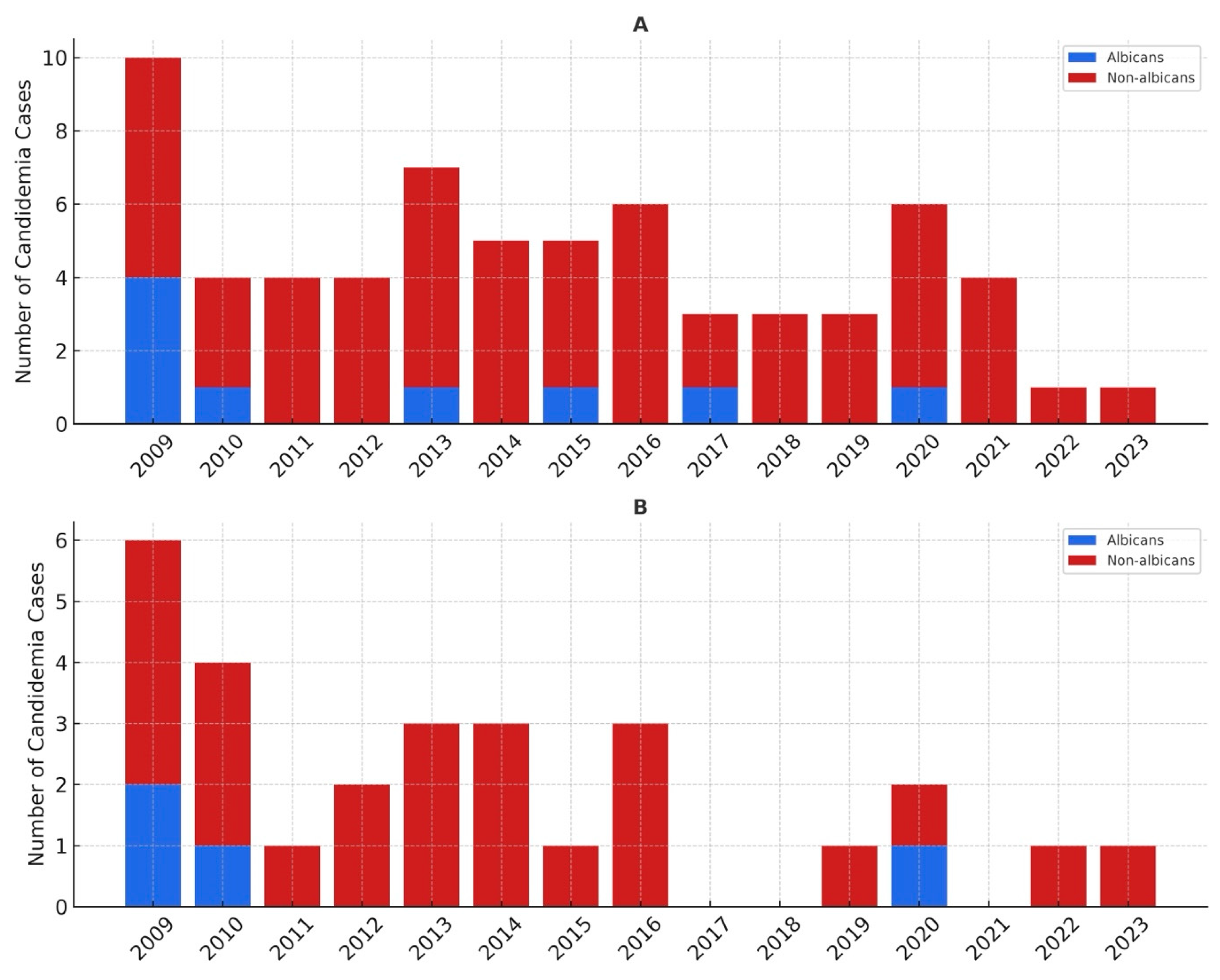
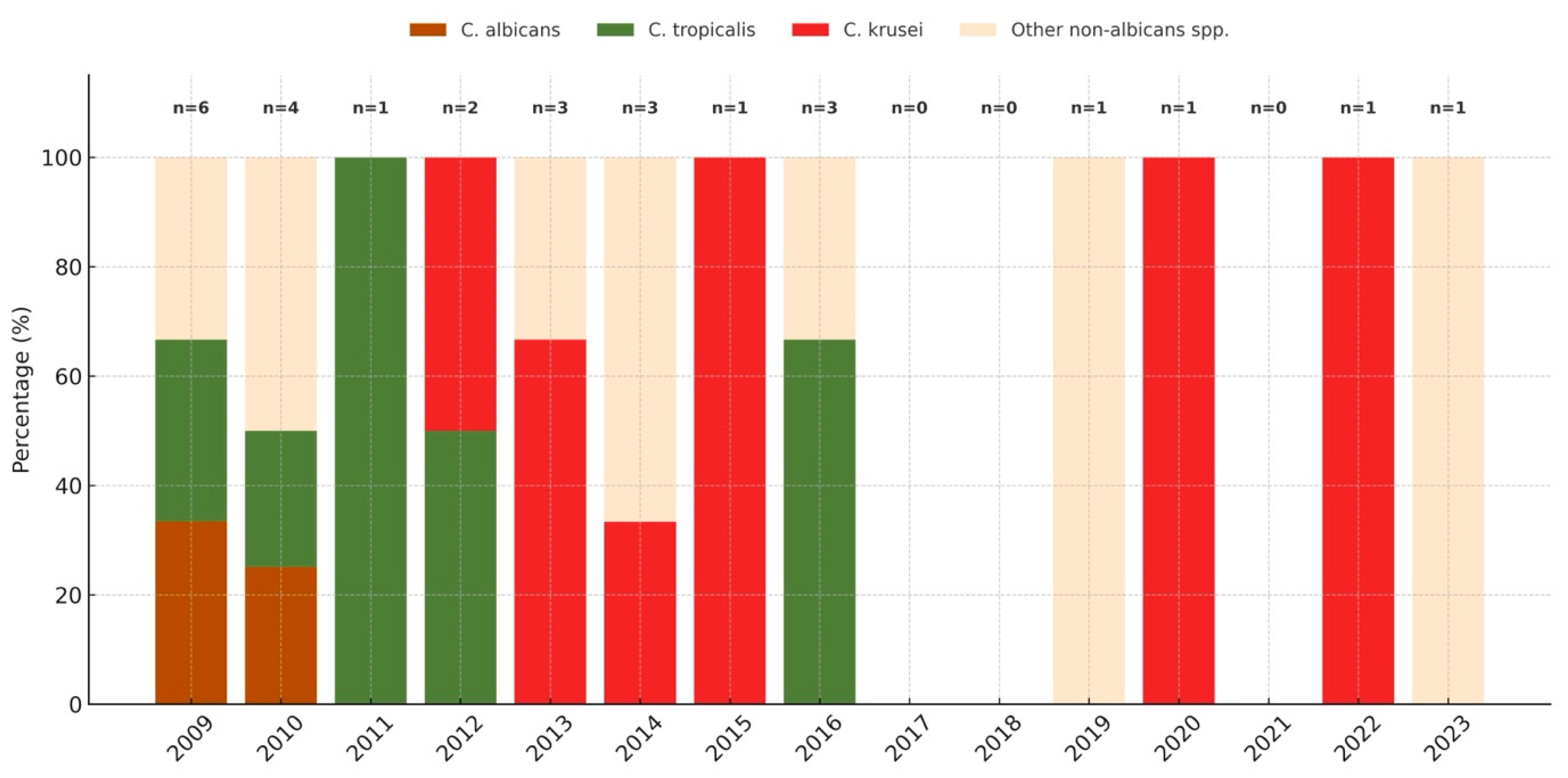
3.2. Distribution and Temporal Trends of Candida Species
3.3. Antifungal Therapy and Drug Susceptibility
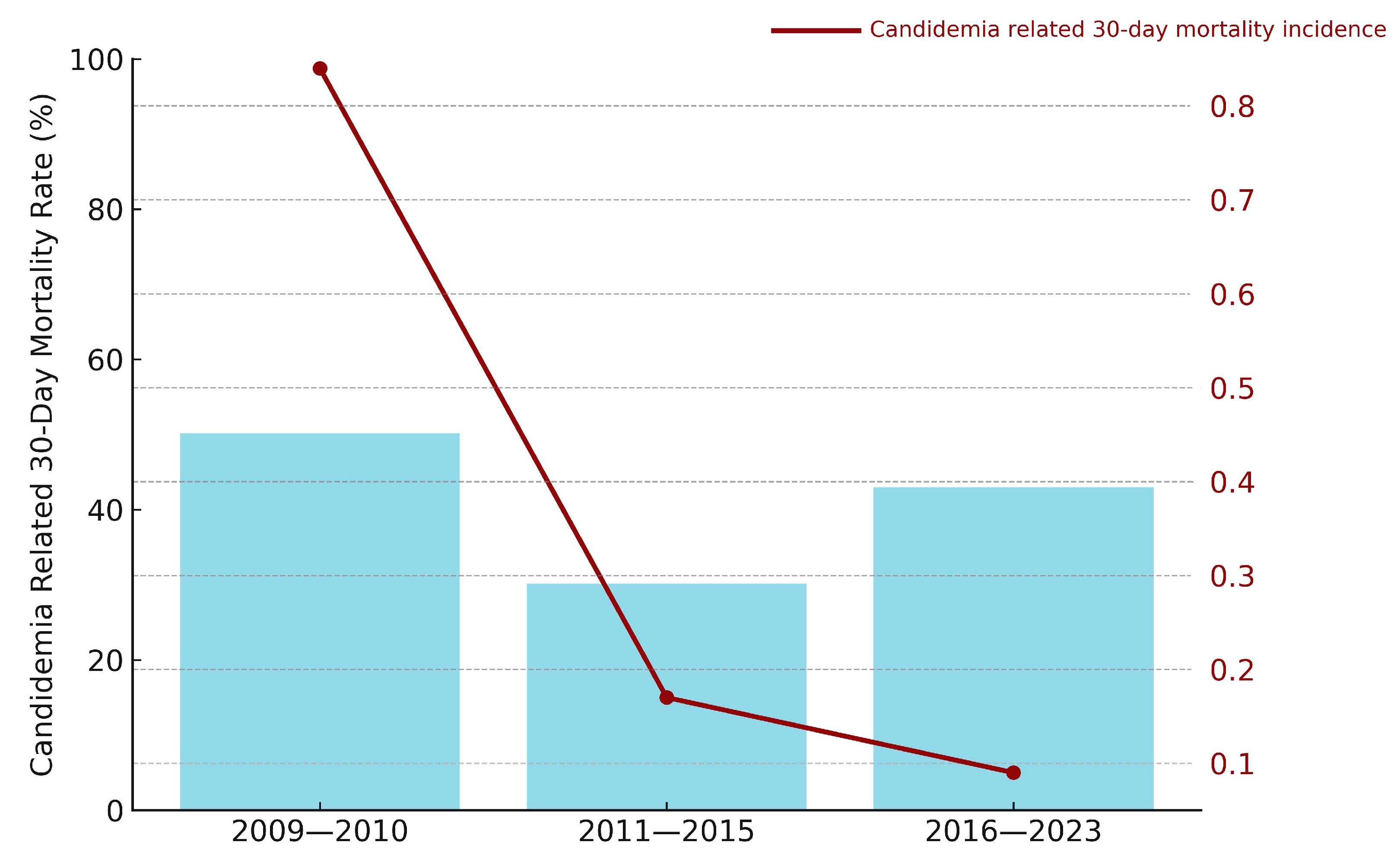
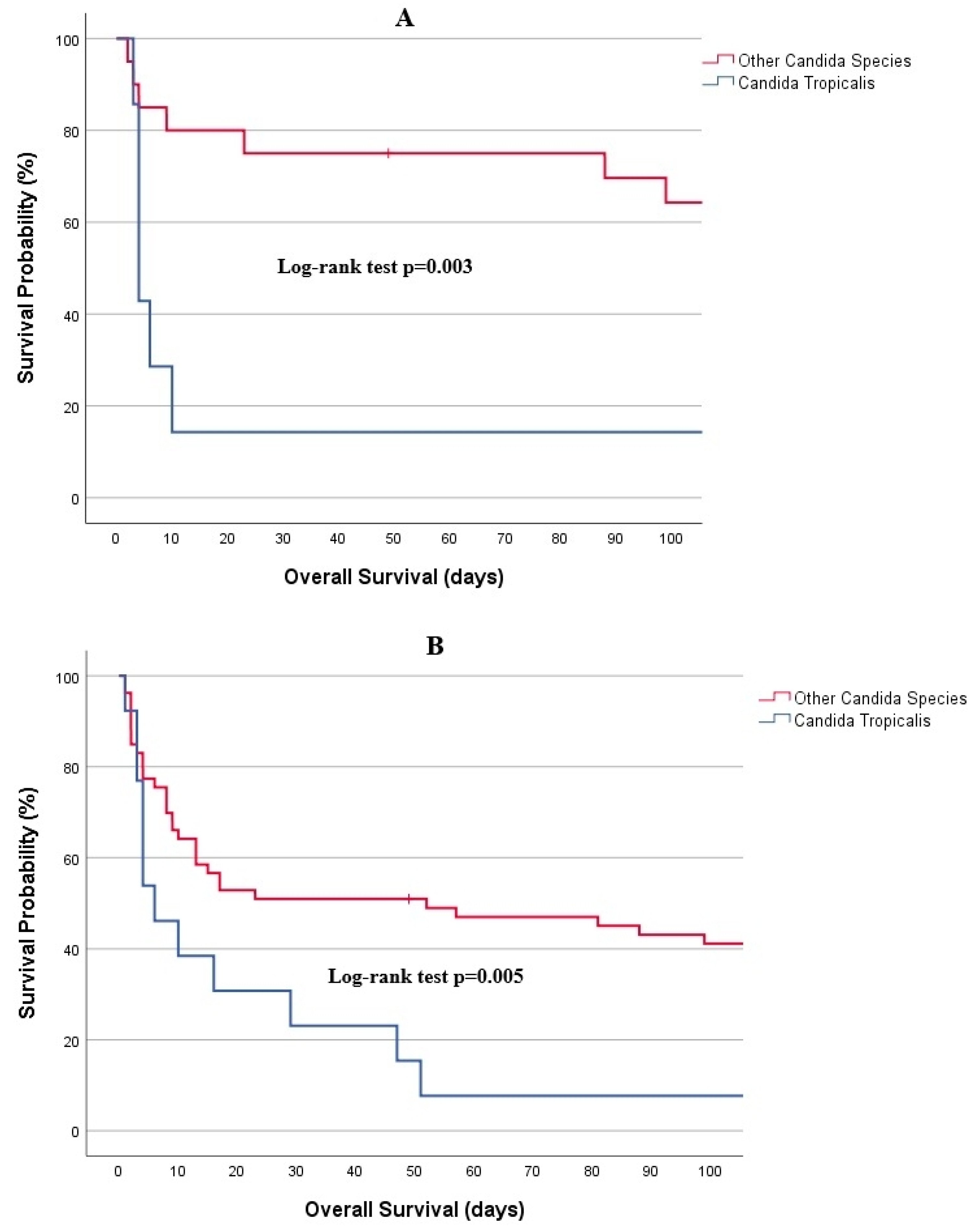
3.4. Survival Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Denning, D.W. Global Incidence and Mortality of Severe Fungal Disease. Lancet Infect. Dis. 2024, 24, e428–e438. [Google Scholar] [CrossRef]
- Cornely, O.A.; Gachot, B.; Akan, H.; Bassetti, M.; Uzun, O.; Kibbler, C.; Marchetti, O.; De Burghgraeve, P.; Ramadan, S.; Pylkkanen, L.; et al. Epidemiology and Outcome of Fungemia in a Cancer Cohort of the Infectious Diseases Group (IDG) of the European Organization for Research and Treatment of Cancer (EORTC 65031). Clin. Infect. Dis. 2015, 61, 324–331. [Google Scholar] [CrossRef]
- Şanlı, K.; Arslantaş, E.; Ceylan, A.N.; Öncel, B.; Özkorucu, D.; Özkan Karagenç, A. Candidemia in Pediatric-Clinic: Frequency of Occurrence, Candida Species, Antifungal Susceptibilities, and Effects on Mortality (2020–2024). Diagnostics 2024, 14, 2343. [Google Scholar] [CrossRef]
- Bongomin, F.; Gago, S.; Oladele, R.O.; Denning, D.W. Global and Multi-National Prevalence of Fungal Diseases-Estimate Precision. J. Fungi 2017, 3, 57. [Google Scholar] [CrossRef] [PubMed]
- Tragiannidis, A.; Fegeler, W.; Rellensmann, G.; Debus, V.; Müller, V.; Hoernig-Franz, I.; Siam, K.; Pana, Z.D.; Jürgens, H.; Groll, A.H. Candidaemia in a European Paediatric University Hospital: A 10-Year Observational Study. Clin. Microbiol. Infect. 2012, 18, E27–E30. [Google Scholar] [CrossRef] [PubMed]
- Arendrup, M.C.; Dzajic, E.; Jensen, R.H.; Johansen, H.K.; Kjældgaard, P.; Knudsen, J.D.; Kristensen, L.; Leitz, C.; Lemming, L.E.; Nielsen, L.; et al. Epidemiological Changes with Potential Implication for Antifungal Prescription Recommendations for Fungaemia: Data from a Nationwide Fungaemia Surveillance Programme. Clin. Microbiol. Infect. 2013, 19, E343–E353. [Google Scholar] [CrossRef] [PubMed]
- Nucci, M.; Queiroz-Telles, F.; Alvarado-Matute, T.; Tiraboschi, I.N.; Cortes, J.; Zurita, J.; Guzman-Blanco, M.; Santolaya, M.E.; Thompson, L.; Sifuentes-Osornio, J.; et al. Epidemiology of Candidemia in Latin America: A Laboratory-Based Survey. PLoS ONE 2013, 8, e59373. [Google Scholar] [CrossRef]
- Pappas, P.G.; Kauffman, C.A.; Andes, D.R.; Clancy, C.J.; Marr, K.A.; Ostrosky-Zeichner, L.; Reboli, A.C.; Schuster, M.G.; Vazquez, J.A.; Walsh, T.J.; et al. Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2016, 62, e1–e50. [Google Scholar] [CrossRef]
- Posteraro, B.; De Carolis, E.; Criscuolo, M.; Ballanti, S.; De Angelis, G.; Del Principe, M.I.; Delia, M.; Fracchiolla, N.; Marchesi, F.; Nadali, G.; et al. Candidaemia in Haematological Malignancy Patients from a SEIFEM Study: Epidemiological Patterns According to Antifungal Prophylaxis. Mycoses 2020, 63, 900–910. [Google Scholar] [CrossRef]
- Kothavade, R.J.; Kura, M.M.; Valand, A.G.; Panthaki, M.H. Candida Tropicalis: Its Prevalence, Pathogenicity and Increasing Resistance to Fluconazole. J. Med. Microbiol. 2010, 59, 873–880. [Google Scholar] [CrossRef]
- Malinovská, Z.; Čonková, E.; Váczi, P. Biofilm Formation in Medically Important Candida Species. J. Fungi 2023, 9, 955. [Google Scholar] [CrossRef]
- Sanitá, P.V.; Zago, C.E.; De Oliveira Mima, E.G.; Pavarina, A.C.; Jorge, J.H.; MacHado, A.L.; Vergani, C.E. In Vitro Evaluation of the Enzymatic Activity Profile of Non-Albicans Candida Species Isolated from Patients with Oral Candidiasis with or without Diabetes. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2014, 118, 84–91. [Google Scholar] [CrossRef]
- Sipsas, N.V.; Lewis, R.E.; Tarrand, J.; Hachem, R.; Rolston, K.V.; Raad, I.I.; Kontoyiannis, D.P. Candidemia in Patients with Hematologic Malignancies in the Era of New Antifungal Agents (2001–2007): Stable Incidence but Changing Epidemiology of a Still Frequently Lethal Infection. Cancer 2009, 115, 4745–4752. [Google Scholar] [CrossRef]
- Cornely, O.A.; Maertens, J.; Winston, D.J.; Perfect, J.; Ullmann, A.J.; Walsh, T.J.; Helfgott, D.; Holowiecki, J.; Stockelberg, D.; Goh, Y.-T.; et al. Posaconazole vs. Fluconazole or Itraconazole Prophylaxis in Patients with Neutropenia. N. Engl. J. Med. 2007, 356, 348–359. [Google Scholar] [CrossRef] [PubMed]
- Pagano, L.; Maschmeyer, G.; Lamoth, F.; Blennow, O.; Xhaard, A.; Spadea, M.; Busca, A.; Cordonnier, C.; Maertens, J.; Guisado, M.A.; et al. Primary Antifungal Prophylaxis in Hematological Malignancies. Updated Clinical Practice Guidelines by the European Conference on Infections in Leukemia (ECIL). Leukemia 2025, 39, 1547–1557. [Google Scholar] [CrossRef]
- Chau, A.S.; Mendrick, C.A.; Sabatelli, F.J.; Loebenberg, D.; McNicholas, P.M. Application of Real-Time Quantitative PCR to Molecular Analysis of Candida Albicans Strains Exhibiting Reduced Susceptibility to Azoles. Antimicrob. Agents Chemother. 2004, 48, 2124–2131. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, C.F.; Alves, D.F.; Henriques, M. Combination of Posaconazole and Amphotericin B in the Treatment of Candida Glabrata Biofilms. Microorganisms 2018, 6, 123. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Hagihara, M.; Shibata, Y.; Asai, N.; Yamagishi, Y.; Iwamoto, T.; Mikamo, H. Comparison of Mortality between Echinocandins and Polyenes for an Initial Treatment of Candidemia: A Systematic Review and Meta-Analysis. J. Infect. Chemother. 2021, 27, 1562–1570. [Google Scholar] [CrossRef]
- Antoniadou, A.; Torres, H.A.; Lewis, R.E.; Thornby, J.; Bodey, G.P.; Tarrand, J.J.; Han, X.Y.; Rolston, K.V.I.; Safdar, A.; Raad, I.I.; et al. Candidemia in a Tertiary Care Cancer Center: In Vitro Susceptibility and Its Association with Outcome of Initial Antifungal Therapy. Medicine 2003, 82, 309–321. [Google Scholar] [CrossRef]
- Freifeld, A.G.; Bow, E.J.; Sepkowitz, K.A.; Boeckh, M.J.; Ito, J.I.; Mullen, C.A.; Raad, I.I.; Rolston, K.V.; Young, J.A.H.; Wingard, J.R. Clinical Practice Guideline for the Use of Antimicrobial Agents in Neutropenic Patients with Cancer: 2010 Update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2011, 52, 427–431. [Google Scholar] [CrossRef]
- Hughes, W.T.; Armstrong, D.; Bodey, G.P.; Bow, E.J.; Brown, A.E.; Calandra, T.; Feld, R.; Pizzo, P.A.; Rolston, K.V.I.; Shenep, J.L.; et al. 2002 Guidelines for the Use of Antimicrobial Agents in Neutropenic Patients with Cancer. Clin. Infect. Dis. 2002, 34, 730–751. [Google Scholar] [CrossRef]
- CLSI M27-ED4; Reference Method for Broth Dilution Antifungalsusceptibility Testing of Yeasts, 4th Edition. Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2017.
- CLSI M60-ED2; Reference Method for Broth Dilution Antifungalsusceptibility Testing of Yeasts, 2nd Edition. Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2020.
- CLSI M59-ED3; Reference Method for Broth Dilution Antifungalsusceptibility Testing of Yeasts, 3rd Edition. Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2020.
- Pasqualotto, A.C.; Rosa, D.D.; Medeiros, L.R.; Severo, L.C. Candidaemia and Cancer: Patients Are Not All the Same. BMC Infect. Dis. 2006, 6, 50. [Google Scholar] [CrossRef] [PubMed]
- Gamaletsou, M.N.; Walsh, T.J.; Zaoutis, T.; Pagoni, M.; Kotsopoulou, M.; Voulgarelis, M.; Panayiotidis, P.; Vassilakopoulos, T.; Angelopoulou, M.K.; Marangos, M.; et al. A Prospective, Cohort, Multicentre Study of Candidaemia in Hospitalized Adult Patients with Haematological Malignancies. Clin. Microbiol. Infect. 2014, 20, 12312. [Google Scholar] [CrossRef]
- McCort, M.E.; Tsai, H. Epidemiology of Invasive Candidiasis in Patients with Hematologic Malignancy on Antifungal Prophylaxis. Mycopathologia 2023, 188, 885–892. [Google Scholar] [CrossRef] [PubMed]
- Pristov, K.E.; Ghannoum, M.A. Resistance of Candida to Azoles and Echinocandins Worldwide. Clin. Microbiol. Infect. 2019, 25, 792–798. [Google Scholar] [CrossRef]
- Pagano, L.; Dragonetti, G.; Cattaneo, C.; Marchesi, F.; Veggia, B.; Busca, A.; Candoni, A.; Prezioso, L.; Criscuolo, M.; Cesaro, S.; et al. Changes in the Incidence of Candidemia and Related Mortality in Patients with Hematologic Malignancies in the Last Ten Years. A SEIFEM 2015-B Report. Haematologica 2017, 102, e407–e410. [Google Scholar] [CrossRef][Green Version]
- Mesini, A.; Mikulska, M.; Giacobbe, D.R.; Del Puente, F.; Gandolfo, N.; Codda, G.; Orsi, A.; Tassinari, F.; Beltramini, S.; Marchese, A.; et al. Changing Epidemiology of Candidaemia: Increase in Fluconazole-Resistant Candida Parapsilosis. Mycoses 2020, 63, 361–368. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Diekema, D.J.; Turnidge, J.D.; Castanheira, M.; Jones, R.N. Twenty Years of the SENTRY Antifungal Surveillance Program: Results for Candida Species From 1997–2016. Open Forum Infect. Dis. 2019, 6, S79–S94. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, E.; Hammarström, H.; Ataollahy, N.; Kondori, N. Species Distribution and Antifungal Drug Susceptibilities of Yeasts Isolated from the Blood Samples of Patients with Candidemia. Sci. Rep. 2019, 9, 3838. [Google Scholar] [CrossRef]
- Maldonado, N.A.; Cano, L.E.; De Bedout, C.; Arbeláez, C.A.; Roncancio, G.; Tabares, Á.M.; Robledo, C.G.; Robledo, J. Association of Clinical and Demographic Factors in Invasive Candidiasis Caused by Fluconazole-Resistant Candida Species: A Study in 15 Hospitals, Medellín, Colombia 2010–2011. Diagn. Microbiol. Infect. Dis. 2014, 79, 280–286. [Google Scholar] [CrossRef]
- Desplanques, P.Y.; Burlacu, R.; Poinsignon, V.; Boussion, H.; Borget, I.; Wyplosz, B.; De Botton, S.; Billaud, E.; Chachaty, E.; Gachot, B.; et al. Factors Influencing Posaconazole Plasmatic Concentrations in Patients Presenting with Acute Myeloid Leukemia. Médecine Mal. Infect. 2014, 44, 174–179. [Google Scholar] [CrossRef]
- Patel, T.S.; Carver, P.L.; Eschenauer, G.A. Are In Vitro Susceptibilities to Azole Antifungals Predictive of Clinical Outcome in the Treatment of Candidemia? J. Clin. Microbiol. 2018, 56, e01072-18. [Google Scholar] [CrossRef] [PubMed]
- Ye, N.; Liu, Z.; Tang, W.; Li, X.; Chu, W.; Zhou, Q. Systematic Characterization of Epidemiology, Antifungal Susceptibility, Risk Factors and Outcomes of Candidaemia: A Six-Year Chinese Study. Infect. Drug Resist. 2022, 15, 4887–4898. [Google Scholar] [CrossRef]
- Yakut, N.; Kepenekli, E.; Ergenc, Z.; Baran, E.; Cerikcioglu, N. Antifungal Susceptibility, Species Distribution and Risk Factors Associated with Mortality of Invasive Candidiasis in Children in Turkey: A Six-Year Retrospective, Single-Centre Study. J. Med. Mycol. 2021, 31, 101082. [Google Scholar] [CrossRef]
- Aydin, S.; Derin, O.; Sahin, M.; Dinleyici, R.; Yilmaz, M.; Ceylan, B.; Tosun, A.I.; Ozturk, R.; Mert, A. Epidemiology of Nosocomial Candidemia, Mortality, and Antifungal Resistance: 7-Year Experience in Turkey. Jpn. J. Infect. Dis. 2022, 75, 597–603. [Google Scholar] [CrossRef]
- Hachem, R.; Hanna, H.; Kontoyiannis, D.; Jiang, Y.; Raad, I. The Changing Epidemiology of Invasive Candidiasis: Candida Glabrata and Candida Krusei as the Leading Causes of Candidemia in Hematologic Malignancy. Cancer 2008, 112, 2493–2499. [Google Scholar] [CrossRef]
- Chen, X.C.; Xu, J.; Wu, D.P. Clinical Characteristics and Outcomes of Breakthrough Candidemia in 71 Hematologic Malignancy Patients and/or Allogeneic Hematopoietic Stem Cell Transplant Recipients: A Single-Center Retrospective Study From China, 2011–2018. Clin. Infect. Dis. 2020, 71, S394–S399. [Google Scholar] [CrossRef] [PubMed]
- Pongrácz, J.; Szabó, T.; Juhász, E.; Iván, M.; Kristóf, K. Epidemiology, Risk Factors and Mortality of Fungal Bloodstream Infection: 14 Years of Experience at a Teaching Hospital. Mycopathologia 2025, 190, 55. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Andes, D.R.; Diekema, D.J.; Horn, D.L.; Reboli, A.C.; Rotstein, C.; Franks, B.; Azie, N.E. Epidemiology and Outcomes of Invasive Candidiasis Due to Non-Albicans Species of Candida in 2,496 Patients: Data from the Prospective Antifungal Therapy (PATH) Registry 2004–2008. PLoS ONE 2014, 9, e101510. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.H.; Chakrabarti, A.; Li, R.Y.; Patel, A.K.; Watcharananan, S.P.; Liu, Z.; Chindamporn, A.; Tan, A.L.; Sun, P.L.; Wu, U.I.; et al. Incidence and Species Distribution of Candidaemia in Asia: A Laboratory-Based Surveillance Study. Clin. Microbiol. Infect. 2015, 21, 946–953. [Google Scholar] [CrossRef]
- Puig-Asensio, M.; Ruiz-Camps, I.; Fernández-Ruiz, M.; Aguado, J.M.; Muñoz, P.; Valerio, M.; Delgado-Iribarren, A.; Merino, P.; Bereciartua, E.; Fortún, J.; et al. Epidemiology and Outcome of Candidaemia in Patients with Oncological and Haematological Malignancies: Results from a Population-Based Surveillance in Spain. Clin. Microbiol. Infect. 2015, 21, 491.e1–491.e10. [Google Scholar] [CrossRef] [PubMed]
- Kullberg, B.J.; Arendrup, M.C. Invasive Candidiasis. N. Engl. J. Med. 2015, 373, 1445–1456. [Google Scholar] [CrossRef]
- Abdullah, N.M.; Cheah, S.K.; Abdul Rahman, R.; Nor, N.M.; Maaya, M.; Musthafa, Q.A. External Validation of Risk Prediction Score for Candidemia in Critically Ill Patients: A Retrospective Observational Study. J. Fungi 2025, 11, 204. [Google Scholar] [CrossRef]
- Schroeder, M.; Weber, T.; Denker, T.; Winterland, S.; Wichmann, D.; Rohde, H.; Ozga, A.K.; Fischer, M.; Kluge, S. Epidemiology, Clinical Characteristics, and Outcome of Candidemia in Critically Ill Patients in Germany: A Single-Center Retrospective 10-Year Analysis. Ann. Intensive Care 2020, 10, 142. [Google Scholar] [CrossRef]
- Kimura, S.I.; Kameda, K.; Harada, K.; Saburi, M.; Okinaka, K.; Shinohara, A.; Uchida, N.; Nishijima, A.; Ozawa, Y.; Tanaka, M.; et al. Risk and Predictive Factors for Candidemia After Allogeneic Hematopoietic Cell Transplantation: JSTCT Transplant Complications Working Group. Transplant. Cell. Ther. 2022, 28, 209.e1–209.e9. [Google Scholar] [CrossRef]
- Bretagne, S.; Desnos-Ollivier, M.; Sitbon, K.; Lortholary, O.; Che, D.; Dromer, F. No Impact of Fluconazole to Echinocandins Replacement as First-Line Therapy on the Epidemiology of Yeast Fungemia (Hospital-Driven Active Surveillance, 2004–2017, Paris, France). Front. Med. 2021, 8, 641965. [Google Scholar] [CrossRef]
- You, L.; Yao, C.; Yang, F.; Yang, Q.; Lan, J.; Song, X.; Shen, J.; Sheng, X.; Chen, X.; Tang, H.; et al. Echinocandins versus Amphotericin B against Candida Tropicalis Fungemia in Adult Hematological Patients with Neutropenia: A Multicenter Retrospective Cohort Study. Infect. Drug Resist. 2020, 13, 2229–2235. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Jones, R.N.; Castanheira, M. Regional Data Analysis of Candida Non-Albicans Strains Collected in United States Medical Sites over a 6-Year Period, 2006–2011. Mycoses 2014, 57, 602–611. [Google Scholar] [CrossRef]
- Semet, C.; Kazak, E.; Ener, B.; Ak, S.; Özkaya, G.; Ağca, H.; Heper, Y.; Yılmaz, E.; Akalın, H. Risk Factors and Outcome for Bloodstream Infections Due to Fluconazole-Resistant Candida Parapsilosis: A 22-Year Single-Center Retrospective Study. Antimicrob. Resist. Infect. Control 2025, 14, 49. [Google Scholar] [CrossRef] [PubMed]
- Fuller, J.; Dingle, T.C.; Bull, A.; Shokoples, S.; Laverdière, M.; Baxter, M.R.; Adam, H.J.; Karlowsky, J.A.; Zhanel, G.G. Species Distribution and Antifungal Susceptibility of Invasive Candida Isolates from Canadian Hospitals: Results of the CANWARD 2011–16 Study. J. Antimicrob. Chemother. 2019, 74, IV48–IV54. [Google Scholar] [CrossRef] [PubMed]
- Arikan-Akdagli, S.; Gülmez, D.; Doğan, Ö.; Çerikçioğlu, N.; Doluca Dereli, M.; Birinci, A.; Yıldıran, Ş.T.; Ener, B.; Öz, Y.; Metin, D.Y.; et al. First Multicentre Report of in Vitro Resistance Rates in Candidaemia Isolates in Turkey. J. Glob. Antimicrob. Resist. 2019, 18, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Arendrup, M.C.; Arikan-Akdagli, S.; Jørgensen, K.M.; Barac, A.; Steinmann, J.; Toscano, C.; Arsenijevic, V.A.; Sartor, A.; Lass-Flörl, C.; Hamprecht, A.; et al. European Candidaemia Is Characterised by Notable Differential Epidemiology and Susceptibility Pattern: Results from the ECMM Candida III Study. J. Infect. 2023, 87, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Kazak, E.; Akın, H.; Ener, B.; Sığırlı, D.; Özkan, Ö.; Gürcüoğlu, E.; Yılmaz, E.; Çelebi, S.; Akçağlar, S.; Akalın, H. An Investigation of Candida Species Isolated from Blood Cultures during 17 Years in a University Hospital. Mycoses 2014, 57, 623–629. [Google Scholar] [CrossRef]
- Gürcüoğlu, E.; Ener, B.; Akalin, H.; Sinirtaş, M.; Evci, C.; Akçağlar, S.; Yilmaz, E.; Heper, Y. Epidemiology of Nosocomial Candidaemia in a University Hospital: A 12-Year Study. Epidemiol. Infect. 2010, 138, 1328–1335. [Google Scholar] [CrossRef] [PubMed]

| Variables | n: 66 |
|---|---|
| Median Age, years (IQR) | 48.5 (32–58) |
| Sex, male (%) | 38 (57.6) |
| Underlying Disease | |
| Acute Myeloid Leukemia, (%) | 27 (40.9) |
| Acute Lymphoblastic Leukemia, (%) | 20 (30.3) |
| Non-Hodgkin Lymphoma, (%) | 14 (21.2) |
| Multiple Myeloma, (%) | 4 (6.1) |
| Hodgkin Lymphoma, (%) | 1 (1.5) |
| Disease Status | |
| Newly Diagnosed, (%) | 20 (30.3) |
| Consolidation Phase, (%) | 8 (12.1) |
| Relapsed/Refractory, (%) | 38 (57.6) |
| Predisposing Factors | |
| Diabetes Mellitus, (%) | 8 (13.6) |
| Systemic Corticosteroids, (%) | 43 (65.2) |
| Central Venous Catheter, (%) | 34 (51.5) |
| Total Parenteral Nutrition, (%) | 24 (36.4) |
| Broad-spectrum Antimicrobial Agents, (%) | 63 (95.4) |
| Hospital Stay (previous 90 days), (%) | 30 (45.5) |
| Neutropenia/ Severe Neutropenia, (%) | 55/49 (83.3/74.2) |
| Candidemia Type | |
| Breakthrough Candidemia, (%) | 54 (81.8) |
| De Novo Infection, (%) | 12 (18.2) |
| Death Within 30 Days After Candidemia Onset, (%) | 35 (53) |
| AML Patients n: 27 | Non-AML Patients n: 39 | p-Value | |
|---|---|---|---|
| Candida albicans, (%) | 3 (11.2) | 6 (15.4) | 0.946 b |
| Non-albicans Candida, (%) | 24 (88.8) | 33 (84.6) | |
| Candida krusei, (%) | 7 (25.9) | 14 (42.4) | |
| Candida tropicalis, (%) | 7 (25.9) | 6 (18.1) | |
| Candida parapsilosis, (%) | 5 (18.5) | 5 (15.1) | |
| Candida norvegensis/Candida inconspicua, (%) | 2 (7.4) | 2 (6.1) | |
| Candida glabrata, (%) | 1 (3.7) | 2 (6.1) | |
| Candida kefyr, (%) | 1 (3.7) | 2 (6.1) | |
| Other Candida Species a, (%) | 1 (3.7) | 2 (6.1) |
| Period | Total Patient-Days in Hospital | Candidemia Incidence (Median, IQR) | p-Value |
|---|---|---|---|
| 2009–2010 | 5924 | 1.67 (1.52–1.81) | 0.022 a |
| 2011–2015 | 17,336 | 0.48 (0.29–0.87) | |
| 2016–2023 | 33,074 | 0.25 (0–0.35) |
| Clinical Factors | AML Patients n: 27 | Non-AML Patients n: 39 | p-Value |
|---|---|---|---|
| Antifungal Prophylaxis, (%) | 27 (100) | 27 (69.2) | <0.001 a |
| Fluconazole, (%) | 18 (66.7) | 22 (81.5) | 0.002 a |
| Posaconazole, (%) | 8 (29.6) | - | |
| Itraconazole, (%) | - | 3 (11.1) | |
| Liposomal Amphotericin B, (%) | 1 (3.7) | 2 (7.4) | |
| Initial Antifungal Treatment, (%) | |||
| Fluconazole, (%) | 8 (29.6) | 10 (25.6) | 0.529 a |
| Liposomal Amphotericin B, (%) | 14 (51.9) | 20 (51.3) | |
| Echinocandin, (%) | 5 (18.5) | 7 (17.9) | |
| Voriconazole, (%) | - | 2 (5.2) | |
| Modification of Initial Antifungal Treatment, (%) | 15 (55.6) | 17 (43.6) | >0.999 a |
| Due to Lack of Clinical Response | 12 (80) | 14 (82.4) | |
| Due to Adverse Event | 3 (20) | 3 (17.6) | |
| Crude 30-day Mortality | 11 (40.7) | 24 (61.5) | 0.096 a |
| Species (No. of Tested) | Antifungal Agent | Resistant n (%) | SDD/I n (%) | Non-Wild-Type n (%) |
|---|---|---|---|---|
| C. krusei (20) | Amphotericin B | - | - | 1 (5) |
| Itraconazole | - | - | - | |
| Posaconazole | - | - | 1 (5) | |
| Voriconazole | - | - | - | |
| Anidulafungin | - | - | - | |
| C. tropicalis (8) | Amphotericin B | - | - | - |
| Fluconazole | - | - | - | |
| Itraconazole | - | - | - | |
| Posaconazole | - | - | 1 (12.5) | |
| Voriconazole | - | - | - | |
| Anidulafungin | - | - | - | |
| C. parapsilosis (8) | Amphotericin B | - | - | 1 (12.5) |
| Fluconazole | - | - | 2 (25) | |
| Itraconazole | - | - | - | |
| Posaconazole | - | - | - | |
| Voriconazole | - | 1 (12.5) | - | |
| Anidulafungin | - | - | - | |
| C. glabrata (4) | Amphotericin B | - | - | - |
| Fluconazole | 1 (25) | 3 (75) | - | |
| Itraconazole | - | - | - | |
| Posaconazole | - | - | - | |
| Voriconazole | - | - | 1 (25) | |
| Anidulafungin | - | - | - | |
| C. albicans (3) | Amphotericin B | - | - | - |
| Fluconazole | - | - | - | |
| Itraconazole | - | - | - | |
| Posaconazole | - | - | - | |
| Voriconazole | - | - | - | |
| Anidulafungin | - | - | - |
| Variables | AML Patients Univariate Analysis (OR, 95% CI), p-Value | AML Patients Multivariate Analysis (OR, 95% CI), p-Value | Overall Cohort Univariate Analysis (OR, 95% CI), p-Value | Overall Cohort Multivariate Analysis (OR, 95% CI), p-Value | |
|---|---|---|---|---|---|
| Age, (years) | 1.006 (0.948–1.068) 0.845 | 1.003 (0.970–1.037) 0.873 | |||
| Gender | Male (R) vs. Female | 0.625 (0.118–3.316) 0.581 | 1.038 (0.390–2.762) 0.940 | ||
| Infection Period | <A (R) vs. ≥B,C | 0.823 (0.308–2.204) 0.699 | 1.386 (0.731–2.630) 0.318 | ||
| Disease Status | Active (R) vs. Remission | 0.301 (0.072–1.256) 0.100 | 0.660 (0.300–1.452) 0.302 | ||
| Diabetes Mellitus | Absent (R) vs. Presence | 5.625 (0.500–63.282) 0.162 | 1.931 (0.440–8.483) 0.383 | ||
| Systemic Corticosteroids | Presence (R) vs. Absent | 0.500 (0.105–2.379) 0.384 | 0.614 (0.219–1.717) 0.352 | ||
| Central Venous Catheter | Presence (R) vs. Absent | 0.343 (0.070–1.684) 0.187 | 0.778 (0.295–2.051) 0.611 | ||
| Total Parenteral Nutrition | Absent (R) vs. Presence | 2.500 (0.485–12.886) 0.273 | 4.412 (1.453–13.394) 0.009 | 4.615 (1.342–15.869) 0.015 | |
| Hospital Stay (previous 90 days) | Absent (R) vs. Presence | 0.370 (0.059–2.323) 0.289 | 1.023 (0.387–2.700) 0.964 | ||
| Severe Neutropenia | Absent (R) vs. Presence | - | 1.381 (0.457–4.174) 0.568 | ||
| Candidemia Type | Denovo (R) vs. BrC | - | 0.073 (0.009–0.604) 0.015 | 0.070 (0.008–0.628) 0.017 | |
| Antifungal Prophylaxis | Posa (R) vs. Flu | 2.095 (0.356–12.322) 0.413 | 2.222 (0.436–11.320) 0.336 | ||
| Candida Species | Others (R) vs. Tropicalis | 18 (1.723–188.082) 0.016 | 18 (1.723–188.082) 0.016 | 2.337 (0.640–8.531) 0.199 | 4.301 (1.030–17.955) 0.045 |
| First Line Antifungal Therapy | Others (R) vs. ECH | 0.300 (0.029–3.135) 0.315 | 0.371 (0.100–1.383) 0.140 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hunutlu, F.Ç.; Özkalemkaş, F.; Ener, B.; Demirayak, D.; Çalışır, B.; Öztop, H.; Pınar, İ.E.; Gürsoy, V.; Ersal, T.; Koca, T.G.; et al. Shifting Paradigms in Antifungal Prophylaxis and Their Effects on Candidemia Outcomes in Hematological Malignancies: A 14-Year Experience from a Single Tertiary Center. J. Fungi 2025, 11, 630. https://doi.org/10.3390/jof11090630
Hunutlu FÇ, Özkalemkaş F, Ener B, Demirayak D, Çalışır B, Öztop H, Pınar İE, Gürsoy V, Ersal T, Koca TG, et al. Shifting Paradigms in Antifungal Prophylaxis and Their Effects on Candidemia Outcomes in Hematological Malignancies: A 14-Year Experience from a Single Tertiary Center. Journal of Fungi. 2025; 11(9):630. https://doi.org/10.3390/jof11090630
Chicago/Turabian StyleHunutlu, Fazıl Çağrı, Fahir Özkalemkaş, Beyza Ener, Dilay Demirayak, Büşra Çalışır, Hikmet Öztop, İbrahim Ethem Pınar, Vildan Gürsoy, Tuba Ersal, Tuba Güllü Koca, and et al. 2025. "Shifting Paradigms in Antifungal Prophylaxis and Their Effects on Candidemia Outcomes in Hematological Malignancies: A 14-Year Experience from a Single Tertiary Center" Journal of Fungi 11, no. 9: 630. https://doi.org/10.3390/jof11090630
APA StyleHunutlu, F. Ç., Özkalemkaş, F., Ener, B., Demirayak, D., Çalışır, B., Öztop, H., Pınar, İ. E., Gürsoy, V., Ersal, T., Koca, T. G., Akalın, E. H., & Özkocaman, V. (2025). Shifting Paradigms in Antifungal Prophylaxis and Their Effects on Candidemia Outcomes in Hematological Malignancies: A 14-Year Experience from a Single Tertiary Center. Journal of Fungi, 11(9), 630. https://doi.org/10.3390/jof11090630






