The Merits, Limitations, and Future Directions of Cost-Effectiveness Analysis in Cardiac MRI with a Focus on Coronary Artery Disease: A Literature Review
Abstract
1. Introduction
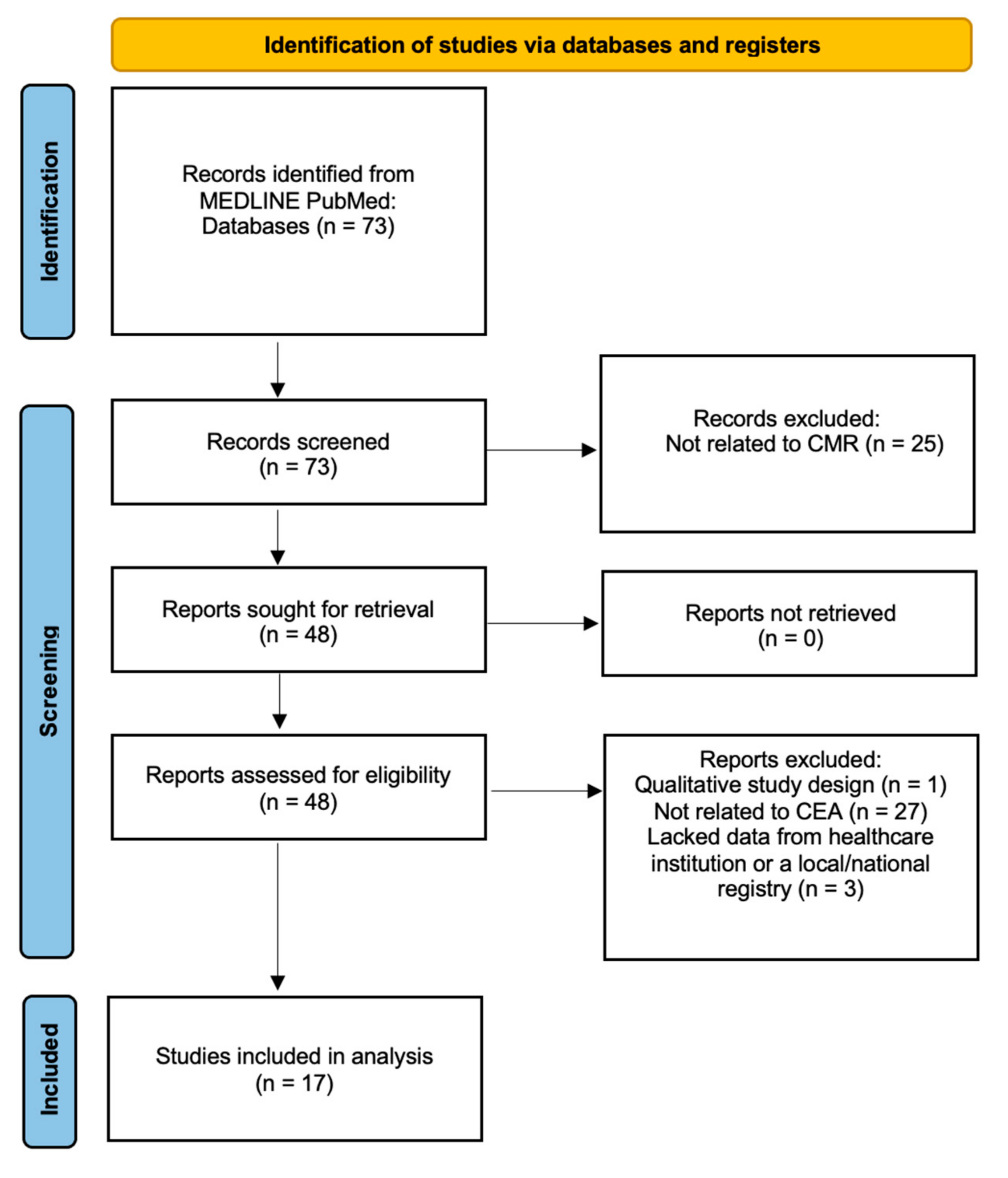
2. Materials and Methods
3. Cost Analysis Models
4. Cost-Effectiveness of Cardiac MRI
4.1. Cost-Effectiveness in Combination with Other Imaging Modalities
4.2. The Role of Diagnostic Accuracy
4.3. Cost-Effectiveness of CMR in Conjunction with Guideline Recommendations
4.4. Stepwise Testing with Other Imaging Modalities
4.5. Regional Applicability of CMR
4.6. The Role of Pretest Probability
4.7. Evidence against Cost-Effectiveness of CMR
4.8. Pretest Probability in the United States
4.9. Relationship with Morise Scores
5. Limitations of Cost-Effectiveness Analysis in CMR
6. Use of Artificial Intelligence in Cost-Effectiveness Analysis of CMR
7. Future Directions and Protocol Recommendations
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Daly, C.; Kwong, R.Y. Cardiac MRI for Myocardial Ischemia. Methodist DeBakey Cardiovasc. J. 2013, 9, 123–131. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dickstein, K. Clinical utilities of cardiac MRI. e-J. Cardiol. Pract. 2008, 6. Available online: https://www.escardio.org/Journals/E-Journal-of-Cardiology-Practice/Volume-6/Clinical-Utilities-of-cardiac-MRI (accessed on 15 September 2022).
- Earls, J.P.; Ho, V.B.; Foo, T.K.; Castillo, E.; Flamm, S.D. Cardiac MRI: Recent Progress and Continued Challenges. J. Magn. Reson. Imaging 2002, 16, 111–127. [Google Scholar] [CrossRef] [PubMed]
- Axel, L.; Otazo, R. Accelerated MRI for the Assessment of Cardiac Function. Br. J. Radiol. 2016, 89, 20150655. [Google Scholar] [CrossRef]
- Hamirani, Y.S.; Kramer, C.M. Advances in Stress Cardiac MRI and Computed Tomography. Futur. Cardiol. 2013, 9, 681–695. [Google Scholar] [CrossRef] [PubMed]
- Busse, A.; Rajagopal, R.; Yücel, S.; Beller, E.; Öner, A.; Streckenbach, F.; Cantré, D.; Ince, H.; Weber, M.-A.; Meinel, F.G. Cardiac MRI—Update 2020. Radiologe 2020, 60, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Gannon, M.P.; Schaub, E.; Grines, C.L.; Saba, S.G. State of the Art: Evaluation and Prognostication of Myocarditis Using Cardiac MRI. J. Magn. Reason. Imaging 2019, 49, e122–e131. [Google Scholar] [CrossRef] [PubMed]
- Seraphim, A.; Knott, K.D.; Augusto, J.; Bhuva, A.N.; Manisty, C.; Moon, J.C. Quantitative Cardiac MRI. J. Magn. Reason. Imaging 2020, 51, 693–711. [Google Scholar] [CrossRef] [PubMed]
- Captur, G.; Manisty, C.; Moon, J.C. Cardiac MRI Evaluation of Myocardial Disease. Heart 2016, 102, 1429–1435. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.C.; Zerhouni, E.A.; Judd, R.M.; Lugo-Olivieri, C.H.; Barouch, L.A.; Schulman, S.P.; Blumenthal, R.S.; Lima, J.A.C. Prognostic Significance of Microvascular Obstruction by Magnetic Resonance Imaging in Patients With Acute Myocardial Infarction. Circulation 1998, 97, 765–772. [Google Scholar] [CrossRef]
- Abou Hassan, O.K.; Kwon, D. Update on MRI Techniques for Evaluation of Pericardial Disease. Curr. Cardiol. Rep. 2020, 22, 147. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, D.H.; Abbara, S.; Chaithiraphan, V.; Yared, K.; Killeen, R.P.; Cury, R.C.; Dodd, J.D. Cardiac Tumors: Optimal Cardiac MR Sequences and Spectrum of Imaging Appearances. AJR Am. J. Roentgenol. 2009, 193, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Hegde, V.A.; Biederman, R.W.; Mikolich, J.R. Cardiovascular Magnetic Resonance Imaging—Incremental Value in a Series of 361 Patients Demonstrating Cost Savings and Clinical Benefits: An Outcome-Based Study. Clin. Med. Insights Cardiol. 2017, 11, 117954681771002. [Google Scholar] [CrossRef]
- Schwitter, J.; Wacker, C.M.; van Rossum, A.C.; Lombardi, M.; Al-Saadi, N.; Ahlstrom, H.; Dill, T.; Larsson, H.B.W.; Flamm, S.D.; Marquardt, M.; et al. MR-IMPACT: Comparison of Perfusion-Cardiac Magnetic Resonance with Single-Photon Emission Computed Tomography for the Detection of Coronary Artery Disease in a Multicentre, Multivendor, Randomized Trial. Eur. Heart J. 2008, 29, 480–489. [Google Scholar] [CrossRef]
- Ibrahim, E.-S.H.; Frank, L.; Baruah, D.; Arpinar, V.E.; Nencka, A.S.; Koch, K.M.; Muftuler, L.T.; Unal, O.; Stojanovska, J.; Rubenstein, J.C.; et al. Value CMR: Towards a Comprehensive, Rapid, Cost-Effective Cardiovascular Magnetic Resonance Imaging. Int. J. Biomed. Imaging 2021, 2021, 8851958. [Google Scholar] [CrossRef]
- Attili, A.K.; Schuster, A.; Nagel, E.; Reiber, J.H.C.; van der Geest, R.J. Quantification in Cardiac MRI: Advances in Image Acquisition and Processing. Int. J. Cardiovasc. Imaging 2010, 26, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Vermersch, M.; Longère, B.; Coisne, A.; Schmidt, M.; Forman, C.; Monnet, A.; Pagniez, J.; Silvestri, V.; Simeone, A.; Cheasty, E.; et al. Compressed Sensing Real-Time Cine Imaging for Assessment of Ventricular Function, Volumes and Mass in Clinical Practice. Eur. Radiol. 2020, 30, 609–619. [Google Scholar] [CrossRef]
- Picano, E. Economic and Biological Costs of Cardiac Imaging. Cardiovasc. Ultrasound 2005, 3, 13. [Google Scholar] [CrossRef]
- Bertoldi, E.G.; Stella, S.F.; Rohde, L.E.P.; Polanczyk, C.A. Cost-Effectiveness of Anatomical and Functional Test Strategies for Stable Chest Pain: Public Health Perspective from a Middle-Income Country. BMJ Open 2017, 7, e012652. [Google Scholar] [CrossRef]
- Francis, S.A.; Daly, C.; Heydari, B.; Abbasi, S.; Shah, R.V.; Kwong, R.Y. Cost-Effectiveness Analysis for Imaging Techniques with a Focus on Cardiovascular Magnetic Resonance. J. Cardiovasc. Magn. Reson. 2013, 15, 52. [Google Scholar] [CrossRef]
- Moschetti, K.; Favre, D.; Pinget, C.; Pilz, G.; Petersen, S.E.; Wagner, A.; Wasserfallen, J.-B.; Schwitter, J. Comparative Cost-Effectiveness Analyses of Cardiovascular Magnetic Resonance and Coronary Angiography Combined with Fractional Flow Reserve for the Diagnosis of Coronary Artery Disease. J. Cardiovasc. Magn. Reason. 2014, 16, 13. [Google Scholar] [CrossRef] [PubMed]
- Boldt, J.; Leber, A.W.; Bonaventura, K.; Sohns, C.; Stula, M.; Huppertz, A.; Haverkamp, W.; Dorenkamp, M. Cost-Effectiveness of Cardiovascular Magnetic Resonance and Single-Photon Emission Computed Tomography for Diagnosis of Coronary Artery Disease in Germany. J. Cardiovasc. Magn. Reson. 2013, 15, 30. [Google Scholar] [CrossRef]
- Moschetti, K.; Petersen, S.E.; Pilz, G.; Kwong, R.Y.; Wasserfallen, J.-B.; Lombardi, M.; Korosoglou, G.; Van Rossum, A.C.; Bruder, O.; Mahrholdt, H.; et al. Cost-Minimization Analysis of Three Decision Strategies for Cardiac Revascularization: Results of the “Suspected CAD” Cohort of the European Cardiovascular Magnetic Resonance Registry. J. Cardiovasc. Magn. Reson. 2015, 18, 3. [Google Scholar] [CrossRef]
- Murphy, T.; Jones, D.A.; Friebel, R.; Uchegbu, I.; Mohiddin, S.A.; Petersen, S.E. A Cost Analysis of Cardiac Magnetic Resonance Imaging in the Diagnostic Pathway of Patients Presenting with Unexplained Acute Myocardial Injury and Culprit-Free Coronary Angiography. Front. Cardiovasc. Med. 2021, 8, 749668. [Google Scholar] [CrossRef] [PubMed]
- Stokes, E.A.; Doble, B.; Pufulete, M.; Reeves, B.C.; Bucciarelli-Ducci, C.; Dorman, S.; Greenwood, J.P.; Anderson, R.A.; Wordsworth, S. Cardiovascular Magnetic Resonance in Emergency Patients with Multivessel Disease or Unobstructed Coronary Arteries: A Cost-Effectiveness Analysis in the UK. BMJ Open 2019, 9, e025700. [Google Scholar] [CrossRef]
- Petrov, G.; Kelle, S.; Fleck, E.; Wellnhofer, E. Incremental Cost-Effectiveness of Dobutamine Stress Cardiac Magnetic Resonance Imaging in Patients at Intermediate Risk for Coronary Artery Disease. Clin. Res. Cardiol. 2015, 104, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Pontone, G.; Andreini, D.; Guaricci, A.I.; Rota, C.; Guglielmo, M.; Mushtaq, S.; Baggiano, A.; Beltrama, V.; Fusini, L.; Solbiati, A.; et al. The STRATEGY Study (Stress Cardiac Magnetic Resonance Versus Computed Tomography Coronary Angiography for the Management of Symptomatic Revascularized Patients): Resources and Outcomes Impact. Circ. Cardiovasc. Imaging 2016, 9, e005171. [Google Scholar] [CrossRef]
- Pletscher, M.; Walker, S.; Moschetti, K.; Pinget, C.; Wasserfallen, J.-B.; Greenwood, J.P.; Schwitter, J.; Girardin, F.R. Cost-Effectiveness of Functional Cardiac Imaging in the Diagnostic Work-up of Coronary Heart Disease. Eur. Heart J. Qual. Care Clin. Outcomes 2016, 2, 201–207. [Google Scholar] [CrossRef]
- Walker, S.; Girardin, F.; McKenna, C.; Ball, S.G.; Nixon, J.; Plein, S.; Greenwood, J.P.; Sculpher, M. Cost-Effectiveness of Cardiovascular Magnetic Resonance in the Diagnosis of Coronary Heart Disease: An Economic Evaluation Using Data from the CE-MARC Study. Heart 2013, 99, 873–881. [Google Scholar] [CrossRef] [PubMed]
- Kozor, R.; Walker, S.; Parkinson, B.; Younger, J.; Hamilton-Craig, C.; Selvanayagam, J.B.; Greenwood, J.P.; Taylor, A.J. Cost-Effectiveness of Cardiovascular Magnetic Resonance in Diagnosing Coronary Artery Disease in the Australian Health Care System. Heart Lung Circ. 2021, 30, 380–387. [Google Scholar] [CrossRef]
- Moschetti, K.; Muzzarelli, S.; Pinget, C.; Wagner, A.; Pilz, G.; Wasserfallen, J.-B.; Schulz-Menger, J.; Nothnagel, D.; Dill, T.; Frank, H.; et al. Cost Evaluation of Cardiovascular Magnetic Resonance versus Coronary Angiography for the Diagnostic Work-up of Coronary Artery Disease: Application of the European Cardiovascular Magnetic Resonance Registry Data to the German, United Kingdom, Swiss, and United States Health Care Systems. J. Cardiovasc. Magn. Reson. 2012, 14, 35. [Google Scholar] [CrossRef]
- Genders, T.S.S.; Petersen, S.E.; Pugliese, F.; Dastidar, A.G.; Fleischmann, K.E.; Nieman, K.; Hunink, M.G.M. The Optimal Imaging Strategy for Patients With Stable Chest Pain: A Cost-Effectiveness Analysis. Ann. Intern. Med. 2015, 162, 474–484. [Google Scholar] [CrossRef]
- Lorenzoni, V.; Bellelli, S.; Caselli, C.; Knuuti, J.; Underwood, S.R.; Neglia, D.; Turchetti, G. Cost-Effectiveness Analysis of Stand-Alone or Combined Non-Invasive Imaging Tests for the Diagnosis of Stable Coronary Artery Disease: Results from the EVINCI Study. Eur. J. Health Econ. 2019, 20, 1437–1449. [Google Scholar] [CrossRef]
- Ge, Y.; Pandya, A.; Steel, K.; Bingham, S.; Jerosch-Herold, M.; Chen, Y.-Y.; Mikolich, J.R.; Arai, A.E.; Bandettini, W.P.; Patel, A.R.; et al. Cost-Effectiveness Analysis of Stress Cardiovascular Magnetic Resonance Imaging for Stable Chest Pain Syndromes. JACC Cardiovasc. Imaging 2020, 13, 1505–1517. [Google Scholar] [CrossRef]
- Pilz, G.; Patel, P.A.; Fell, U.; Ladapo, J.A.; Rizzo, J.A.; Fang, H.; Gunnarsson, C.; Heer, T.; Hoefling, B. Adenosine-Stress Cardiac Magnetic Resonance Imaging in Suspected Coronary Artery Disease: A Net Cost Analysis and Reimbursement Implications. Int. J. Cardiovasc. Imaging 2011, 27, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Farzaneh-Far, A.; Schwitter, J.; Kwong, R.Y. Cost-Effectiveness Analysis for Cardiovascular Magnetic Resonance Imaging. In Cardiovascular Magnetic Resonance; Elsevier: Amsterdam, The Netherlands, 2019; pp. 568–573.e1. [Google Scholar] [CrossRef]
- Centonze, M.; Steidler, S.; Casagranda, G.; Alfonsi, U.; Spagnolli, F.; Rozzanigo, U.; Palumbo, D.; Faletti, R.; De Cobelli, F. Cardiac-CT and Cardiac-MR Cost-Effectiveness: A Literature Review. Radiol. Med. 2020, 125, 1200–1207. [Google Scholar] [CrossRef]
- Vergel, Y.B.; Sculpher, M. Quality-Adjusted Life Years. Pract. Neurol. 2008, 8, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Butler, C.R.; Thompson, R.; Haykowsky, M.; Toma, M.; Paterson, I. Cardiovascular Magnetic Resonance in the Diagnosis of Acute Heart Transplant Rejection: A Review. J. Cardiovasc. Magn. Reson. 2009, 11, 7. [Google Scholar] [CrossRef] [PubMed]
- Gulati, M.; Levy, P.D.; Mukherjee, D.; Amsterdam, E.; Bhatt, D.L.; Birtcher, K.K.; Blankstein, R.; Boyd, J.; Bullock-Palmer, R.P.; Conejo, T.; et al. 2021 AHA/ACC/ASE/CHEST/SAEM/SCCT/SCMR Guideline for the Evaluation and Diagnosis of Chest Pain: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2021, 144, e336–e359. [Google Scholar] [CrossRef]
- Leiner, T.; Bogaert, J.; Friedrich, M.G.; Mohiaddin, R.; Muthurangu, V.; Myerson, S.; Powell, A.J.; Raman, S.V.; Pennell, D.J. SCMR Position Paper (2020) on Clinical Indications for Cardiovascular Magnetic Resonance. J. Cardiovasc. Magn. Reson. 2020, 22, 76. [Google Scholar] [CrossRef]
- von Knobelsdorff-Brenkenhoff, F.; Pilz, G.; Schulz-Menger, J. Representation of Cardiovascular Magnetic Resonance in the AHA/ACC Guidelines. J. Cardiovasc. Magn. Reson. 2017, 19, 70. [Google Scholar] [CrossRef] [PubMed]
- Lucas, F.L.; Siewers, A.E.; Malenka, D.J.; Wennberg, D.E. Diagnostic-Therapeutic Cascade Revisited: Coronary Angiography, Coronary Artery Bypass Graft Surgery, and Percutaneous Coronary Intervention in the Modern Era. Circulation 2008, 118, 2797–2802. [Google Scholar] [CrossRef] [PubMed]
- Kwong, R.Y.; Ge, Y.; Steel, K.; Bingham, S.; Abdullah, S.; Fujikura, K.; Wang, W.; Pandya, A.; Chen, Y.-Y.; Mikolich, J.R.; et al. Cardiac Magnetic Resonance Stress Perfusion Imaging for Evaluation of Patients with Chest Pain. J. Am. Coll. Cardiol. 2019, 74, 1741–1755. [Google Scholar] [CrossRef] [PubMed]
- Morise, A.P.; Haddad, W.J.; Beckner, D. Development and Validation of a Clinical Score to Estimate the Probability of Coronary Artery Disease in Men and Women Presenting with Suspected Coronary Disease. Am. J. Med. 1997, 102, 350–356. [Google Scholar] [CrossRef]
- Petersen, S.E.; Friebel, R.; Ferrari, V.; Han, Y.; Aung, N.; Kenawy, A.; Albert, T.S.E.; Naci, H. Recent Trends and Potential Drivers of Non-Invasive Cardiovascular Imaging Use in the United States of America and England. Front. Cardiovasc. Med. 2021, 7, 617771. [Google Scholar] [CrossRef] [PubMed]
- Goldfarb, J.W.; Weber, J. Trends in Cardiovascular MRI and CT in the U.S. Medicare Population from 2012 to 2017. Radiol. Cardiothorac. Imaging 2021, 3, e200112. [Google Scholar] [CrossRef] [PubMed]
- Leiner, T.; Rueckert, D.; Suinesiaputra, A.; Baeßler, B.; Nezafat, R.; Išgum, I.; Young, A.A. Machine Learning in Cardiovascular Magnetic Resonance: Basic Concepts and Applications. J. Cardiovasc. Magn. Reson. 2019, 21, 61. [Google Scholar] [CrossRef] [PubMed]
- Dilsizian, S.E.; Siegel, E.L. Artificial Intelligence in Medicine and Cardiac Imaging: Harnessing Big Data and Advanced Computing to Provide Personalized Medical Diagnosis and Treatment. Curr. Cardiol. Rep. 2014, 16, 441. [Google Scholar] [CrossRef]
- Loncaric, F.; Camara, O.; Piella, G.; Bijnens, B. Integration of Artificial Intelligence into Clinical Patient Management: Focus on Cardiac Imaging. Rev. Esp. Cardiol. (Engl. Ed.) 2021, 74, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Massalha, S.; Clarkin, O.; Thornhill, R.; Wells, G.; Chow, B.J.W. Decision Support Tools, Systems, and Artificial Intelligence in Cardiac Imaging. Can. J. Cardiol. 2018, 34, 827–838. [Google Scholar] [CrossRef] [PubMed]
- Dey, D.; Slomka, P.J.; Leeson, P.; Comaniciu, D.; Shrestha, S.; Sengupta, P.P.; Marwick, T.H. Artificial Intelligence in Cardiovascular Imaging. J. Am. Coll. Cardiol. 2019, 73, 1317–1335. [Google Scholar] [CrossRef] [PubMed]
| Author | n | Study Population | Study Conclusion |
|---|---|---|---|
| Hegde et al., 2017 [13] | 361 | Cohort that met the ACC and AHA criteria for CMR imaging | CMR is cost-effective when used in conjunction with AHA and ACC, with overall net savings of $833,037 and per patient cost saving of $2308 |
| Francis et al., 2013 [20] | 11,040 | Cohort of cardiology referrals in a European multicenter registry | Two-thirds of the patients experienced a change in therapeutic management and a minority (16%) had uncovered a new diagnosis |
| Moschetti et al., 2014 [21] | N/A | A cohort of CAD-positive patients | CMR followed by coronary angiography was the most cost-effective strategy across most countries, with a lower prevalence of CAD associated with a higher cost-effectiveness * |
| Boldt et al., 2013 [22] | N/A | Patient cohort with suspected CAD | CMR is more cost-effective at low-to-intermediate pretest probability (<60%) for CAD, and coronary angiography is more cost-effective at higher pretest probabilities (>60%) |
| Moschetti et al., 2015 [23] | 3647 | Patient cohort with suspected CAD from the European CMR registry | In patients with typical angina, CMR followed by coronary angiography strategy resulted in 2.3%, 11.6%, 12.8%, and 18.9% cost savings in Germany, United States, Switzerland, and United Kingdom, respectively * |
| Murphy et al., 2021 [24] | 2000 | Modeled population of patients with acute STEMI | Within one-year, routine CMR use increased spending by 14% per patient. Within seven years, CMR-guided management reduced costs by 3% |
| Stokes et al., 2019 [25] | N/A | Two models of multivessel CAD and unobstructive coronary arteries undergoing percutaneous coronary intervention | With CMR being introduced for all patient subgroups that utilize CMR, diagnostic accuracy may be a driver of its cost-effectiveness |
| Petrov et al., 2015 [26] | 1158 | Cohort of stable CAD patients | Patients undergoing CMR had a cost-savings of 12,466€ in hospital costs per life year |
| Pontone et al., 2016 [27] | 600 | Symptomatic CAD patients with a history of revascularization | Stress-CMR had a higher cost-effectiveness compared to CCTA |
| Pletscher et al., 2016 [28] | N/A | Patient cohort with stable angina from Switzerland with a prevalence rate of CAD of 39% and a base-case scenario of 60-year-old male patients | The most cost-effective strategy was exercise stress test, followed by CMR, then coronary angiography * |
| Walker et al., 2013 [29] | N/A | Cohort from the CE-MARC trial referred to cardiologists due to suspicion of angina pectoris | At a lower cost-effectiveness threshold of £20,000, the best diagnostic strategy was exercise stress test, then CMR, then coronary angiography. At a higher cost-effectiveness threshold of £30,000, the best strategy was CMR then coronary angiography * |
| Kozor et al., 2021 [30] | N/A | Cohort from the CE-MARC trial applied to the Austrian healthcare system | On a cost-effectiveness threshold of $45,000 to $75,000 QALYs, the most effective strategy was electrocardiogram stress testing, then CMR, then coronary angiography * |
| Moschetti et al., 2012 [31] | 2717 | A cohort of CAD-positive patients | When using inpatient coronary angiography, CMR strategy had 53.5% lower costs. All tests conducted outpatient in Germany, United Kingdom, and Switzerland were associated with a cost reduction of 50%, 25%, and 23% using the CMR-driven strategy, respectively * |
| Genders et al., 2015 [32] | N/A | 60-year-old patients with low-to-intermediate pretest probability for CAD using a microsimulation model | At low-to-intermediate probability of CAD, stress CMR and SPECT had less efficacy and were more expensive than ECHO 1 |
| Lorenzoni et al., 2019 [33] | 350 | European cohort with low-to-intermediate probability of CAD from the EVINCI study | ICER calculated using the CCTA-ECHO strategy yielded −3776€/correct diagnosis, in comparison to −969€/correct diagnosis *1 |
| Ge et al., 2020 [34] | N/A | Patient cohort from the multicenter cohort SPINS registry | CMR-based assessment of CAD is the most cost-effective approach based on $100,000/QALY applicable to patients with a pretest probability of <60% |
| Pilz et al., 2011 [35] | 218 | German cohort with suspected CAD | Patients with lower pretest probability and Morise scores for CAD had higher catheterization avoidance rates and cost-savings with the CMR-driven strategy |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siddiqui, T.A.; Chamarti, K.S.; Tou, L.C.; Demirjian, G.A.; Noorani, S.; Zink, S.; Umair, M. The Merits, Limitations, and Future Directions of Cost-Effectiveness Analysis in Cardiac MRI with a Focus on Coronary Artery Disease: A Literature Review. J. Cardiovasc. Dev. Dis. 2022, 9, 357. https://doi.org/10.3390/jcdd9100357
Siddiqui TA, Chamarti KS, Tou LC, Demirjian GA, Noorani S, Zink S, Umair M. The Merits, Limitations, and Future Directions of Cost-Effectiveness Analysis in Cardiac MRI with a Focus on Coronary Artery Disease: A Literature Review. Journal of Cardiovascular Development and Disease. 2022; 9(10):357. https://doi.org/10.3390/jcdd9100357
Chicago/Turabian StyleSiddiqui, Taha A., Kiran S. Chamarti, Leila C. Tou, Gregory A. Demirjian, Sarah Noorani, Sydney Zink, and Muhammad Umair. 2022. "The Merits, Limitations, and Future Directions of Cost-Effectiveness Analysis in Cardiac MRI with a Focus on Coronary Artery Disease: A Literature Review" Journal of Cardiovascular Development and Disease 9, no. 10: 357. https://doi.org/10.3390/jcdd9100357
APA StyleSiddiqui, T. A., Chamarti, K. S., Tou, L. C., Demirjian, G. A., Noorani, S., Zink, S., & Umair, M. (2022). The Merits, Limitations, and Future Directions of Cost-Effectiveness Analysis in Cardiac MRI with a Focus on Coronary Artery Disease: A Literature Review. Journal of Cardiovascular Development and Disease, 9(10), 357. https://doi.org/10.3390/jcdd9100357





