Pre-Eclampsia and Eclampsia: An Update on the Pharmacological Treatment Applied in Portugal †
Abstract
:1. Introduction
2. Methods
3. Pre-Eclampsia and Eclampsia
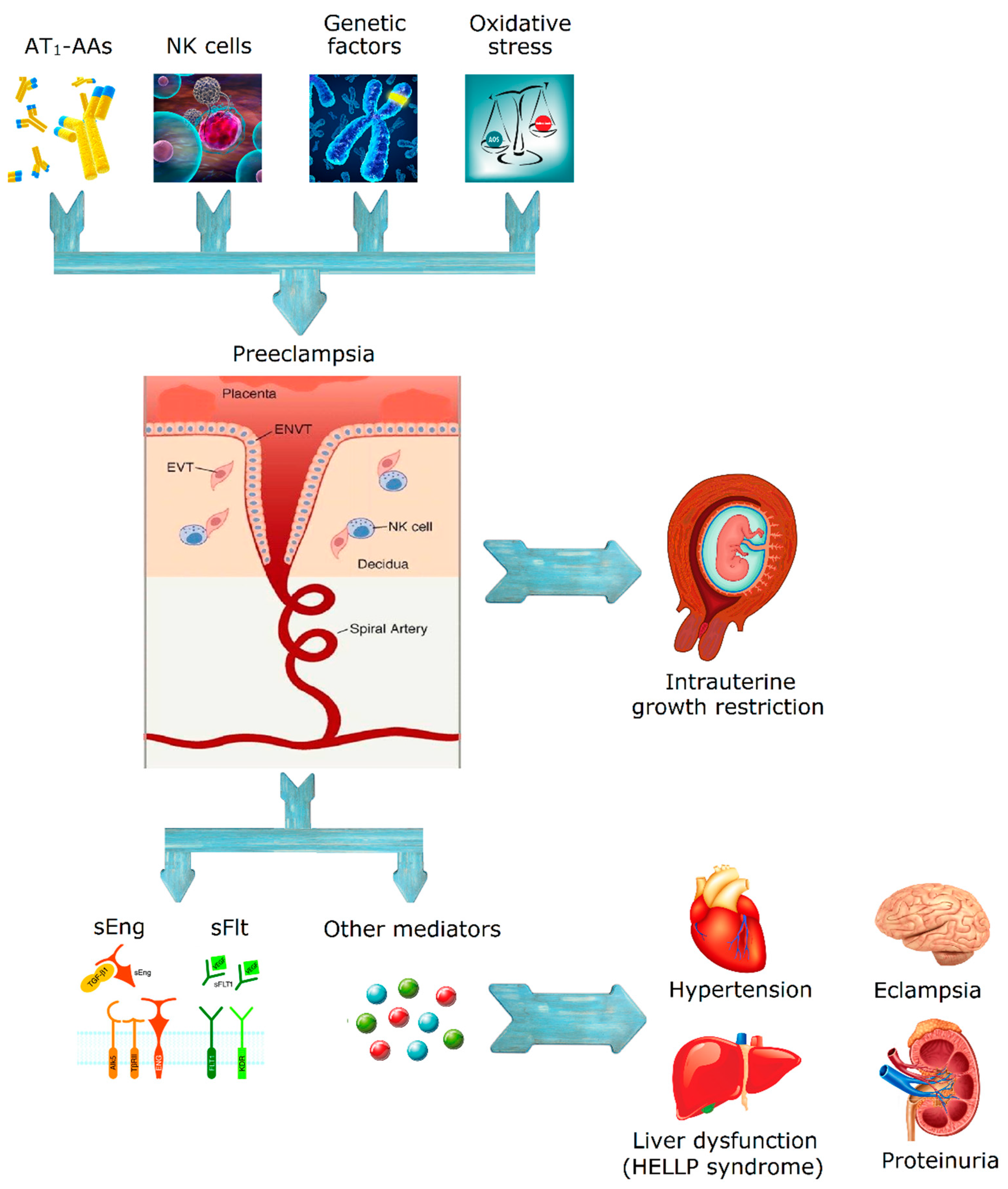
4. Pathophysiology
5. Pharmacological Therapy
6. Mild Pre-Eclampsia
7. Severe Pre-Eclampsia
8. Eclampsia
9. Corticosteroids
10. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gongora, M.C.; Wenger, N.K. Cardiovascular complications of pregnancy. Int. J. Mol. Sci. 2015, 16, 23905–23928. [Google Scholar] [CrossRef] [PubMed]
- Flack, J.M.; Peters, R.; Mehra, V.C.; Nasser, S.A. Hypertension in special populations. Cardiol. Clin. 2002, 20, 303–319. [Google Scholar] [CrossRef]
- Mustafa, R.; Ahmed, S.; Gupta, A.; Venuto, R.C. A comprehensive review of hypertension in pregnancy. J. Pregnancy 2012, 2012, 105918. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.S.; Samuelsen, S.O. Hypertensive disorders in pregnancy and fetal death at different gestational lengths: A population study of 2 121 371 pregnancies. BJOG 2012, 119, 1521–1528. [Google Scholar] [CrossRef] [PubMed]
- Lindheimer, M.D.; Taler, S.J.; Cunningham, F.G. Hypertension in pregnancy. J. Am. Soc. Hypertens. 2010, 4, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Say, L.; Chou, D.; Gemmill, A.; Tuncalp, O.; Moller, A.B.; Daniels, J.; Gulmezoglu, A.M.; Temmerman, M.; Alkema, L. Global causes of maternal death: A who systematic analysis. Lancet Glob. Health 2014, 2, e323–e333. [Google Scholar] [CrossRef]
- Folic, M.; Folic, N.; Varjacic, M.; Jakovljevic, M.; Jankovic, S. Antihypertensive drug therapy for hypertensive disorders in pregnancy. Acta Med. Median. 2008, 47, 65–72. [Google Scholar]
- Kuklina, E.V.; Ayala, C.; Callaghan, W.M. Hypertensive disorders and severe obstetric morbidity in the united states. Obstet. Gynecol. 2009, 113, 1299–1306. [Google Scholar] [CrossRef] [PubMed]
- Moussa, H.N.; Arian, S.E.; Sibai, B.M. Management of hypertensive disorders in pregnancy. Womens Health 2014, 10, 385–404. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, F.; Moura, T.R.; Silva, S.S.; Peraçoli, J.C. Medicamentos anti-hipertensivos na gestação e puerpério. Complementos Ciências Saúde 2011, 22, 55–68. [Google Scholar]
- Chaiworapongsa, T.; Chaemsaithong, P.; Yeo, L.; Romero, R. Pre-eclampsia part 1: Current understanding of its pathophysiology. Nat. Rev. Nephrol. 2014, 10, 466–480. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.; Palmira, J. Distúrbios hipertensivos. In CHTV, EPE—Hospital são Teotónio, Viseu. Departamento de Obstetrícia e Ginecologia: Normas de Orientação Clínica, 1st ed.; Martins, N.N., Veríssimo, R., Eds.; Richter, G.: Viseu, Portugal, 2014; pp. 257–278. [Google Scholar]
- Dhariwal, N.K.; Lynde, G.C. Update in the management of patients with preeclampsia. Anesthesiol. Clin. 2016, 35, 95–106. [Google Scholar] [CrossRef] [PubMed]
- ACOG Committee on Obstetric Practice. ACOG practice bulletin. Diagnosis and management of preeclampsia and eclampsia. Number 33, January 2002. American college of obstetricians and gynecologists. Int. J. Gynaecol. Obstet. 2002, 77, 67–75. [Google Scholar]
- Grand’Maison, S.; Pilote, L.; Okano, M.; Landry, T.; Dayan, N. Markers of vascular dysfunction after hypertensive disorders of pregnancy: A systematic review and meta-analysis. Hypertension 2016, 68, 1447–1458. [Google Scholar] [CrossRef] [PubMed]
- English, F.A.; Kenny, L.C.; McCarthy, F.P. Risk factors and effective management of preeclampsia. Integr. Blood Press Control 2015, 8, 7–12. [Google Scholar] [PubMed]
- Xiong, X.; Wang, F.L.; Davidge, S.T.; Demianczuk, N.N.; Mayes, D.C.; Olson, D.M.; Saunders, L.D. Maternal smoking and preeclampsia. J. Reprod. Med. 2000, 45, 727–732. [Google Scholar] [PubMed]
- Zdravkovic, T.; Genbacev, O.; McMaster, M.T.; Fisher, S.J. The adverse effects of maternal smoking on the human placenta: A review. Placenta 2005, 26 (Suppl. A), S81–S86. [Google Scholar] [CrossRef] [PubMed]
- Stamilio, D.M.; Sehdev, H.M.; Morgan, M.A.; Propert, K.; Macones, G.A. Can antenatal clinical and biochemical markers predict the development of severe preeclampsia? Am. J. Obstet. Gynecol. 2000, 182, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Bellamy, L.; Casas, J.P.; Hingorani, A.D.; Williams, D.J. Pre-eclampsia and risk of cardiovascular disease and cancer in later life: Systematic review and meta-analysis. BMJ 2007, 335, 974. [Google Scholar] [CrossRef] [PubMed]
- Skjaerven, R.; Wilcox, A.J.; Lie, R.T. The interval between pregnancies and the risk of preeclampsia. N. Engl. J. Med. 2002, 346, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Ayansina, D.; Black, C.; Hall, S.J.; Marks, A.; Millar, C.; Prescott, G.J.; Wilde, K.; Bhattacharya, S. Long term effects of gestational hypertension and pre-eclampsia on kidney function: Record linkage study. Pregnancy Hypertens. 2016, 6, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Povoa, A.M.; Costa, F.; Rodrigues, T.; Patricio, B.; Cardoso, F. Prevalence of hypertension during pregnancy in portugal. Hypertens. Pregnancy 2008, 27, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Campos, D.A.; Silva, I.S.; Costa, F.J. Eclâmpsia. In Emergências Obstétricas, 1st ed.; LIDEL, Ed.; Elsevier: Amsterdam, The Netherlands, 2011; pp. 77–87. [Google Scholar]
- Société française d’anesthésie et de réanimation (Sfar); Collège national des gynécologues et obstétriciens français (CNGOF); Société française de médecine périnatale (SFMP); Société française de néonatalogie (SFNN). [multidisciplinary management of severe pre-eclampsia (PE). Experts’ guidelines 2008. Societe francaise d’anesthesie et de reanimation. College national des gynecologues et obstetriciens francais. Societe francaise de medecine perinatale. Societe francaise de neonatalogie]. Ann. Fr. Anesth. Reanim. 2009, 28, 275–281. [Google Scholar]
- Stekkinger, E.; Zandstra, M.; Peeters, L.L.; Spaanderman, M.E. Early-onset preeclampsia and the prevalence of postpartum metabolic syndrome. Obstet. Gynecol. 2009, 114, 1076–1084. [Google Scholar] [CrossRef] [PubMed]
- Ray, J.G.; Vermeulen, M.J.; Schull, M.J.; Redelmeier, D.A. Cardiovascular health after maternal placental syndromes (champs): Population-based retrospective cohort study. Lancet 2005, 366, 1797–1803. [Google Scholar] [CrossRef]
- Enkhmaa, D.; Wall, D.; Mehta, P.K.; Stuart, J.J.; Rich-Edwards, J.W.; Merz, C.N.; Shufelt, C. Preeclampsia and vascular function: A window to future cardiovascular disease risk. J. Womens Health 2016, 25, 284–291. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.S.; Tang, C.H.; Yang, C.Y.; Wu, L.S.; Hung, S.T.; Hwa, H.L.; Chu, P.H. Effect of pre-eclampsia-eclampsia on major cardiovascular events among peripartum women in taiwan. Am. J. Cardiol. 2011, 107, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Kestenbaum, B.; Seliger, S.L.; Easterling, T.R.; Gillen, D.L.; Critchlow, C.W.; Stehman-Breen, C.O.; Schwartz, S.M. Cardiovascular and thromboembolic events following hypertensive pregnancy. Am. J. Kidney Dis. 2003, 42, 982–989. [Google Scholar] [CrossRef] [PubMed]
- Kajantie, E.; Eriksson, J.G.; Osmond, C.; Thornburg, K.; Barker, D.J. Pre-eclampsia is associated with increased risk of stroke in the adult offspring: The helsinki birth cohort study. Stroke 2009, 40, 1176–1180. [Google Scholar] [CrossRef] [PubMed]
- Davis, E.F.; Lazdam, M.; Lewandowski, A.J.; Worton, S.A.; Kelly, B.; Kenworthy, Y.; Adwani, S.; Wilkinson, A.R.; McCormick, K.; Sargent, I.; et al. Cardiovascular risk factors in children and young adults born to preeclamptic pregnancies: A systematic review. Pediatrics 2012, 129, e1552–e1561. [Google Scholar] [CrossRef] [PubMed]
- Fisher, S.J. Why is placentation abnormal in preeclampsia? Am. J. Obstet. Gynecol. 2015, 213, S115–S122. [Google Scholar] [CrossRef] [PubMed]
- Gathiram, P.; Moodley, J. Pre-eclampsia: Its pathogenesis and pathophysiolgy. Cardiovasc. J. Afr. 2016, 27, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Hanna, J.; Goldman-Wohl, D.; Hamani, Y.; Avraham, I.; Greenfield, C.; Natanson-Yaron, S.; Prus, D.; Cohen-Daniel, L.; Arnon, T.I.; Manaster, I.; et al. Decidual nk cells regulate key developmental processes at the human fetal-maternal interface. Nat. Med. 2006, 12, 1065–1074. [Google Scholar] [CrossRef] [PubMed]
- Fukui, A.; Yokota, M.; Funamizu, A.; Nakamua, R.; Fukuhara, R.; Yamada, K.; Kimura, H.; Fukuyama, A.; Kamoi, M.; Tanaka, K.; et al. Changes of nk cells in preeclampsia. Am. J. Reprod. Immunol. 2012, 67, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Tessier, D.R.; Yockell-Lelievre, J.; Gruslin, A. Uterine spiral artery remodeling: The role of uterine natural killer cells and extravillous trophoblasts in normal and high-risk human pregnancies. Am. J. Reprod. Immunol. 2015, 74, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Burton, G.J.; Woods, A.W.; Jauniaux, E.; Kingdom, J.C. Rheological and physiological consequences of conversion of the maternal spiral arteries for uteroplacental blood flow during human pregnancy. Placenta 2009, 30, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Hung, T.H.; Skepper, J.N.; Burton, G.J. In vitro ischemia-reperfusion injury in term human placenta as a model for oxidative stress in pathological pregnancies. Am. J. Pathol. 2001, 159, 1031–1043. [Google Scholar] [CrossRef]
- Malik, R.; Kumar, V. Hypertension in pregnancy. Adv. Exp. Med. Biol. 2017, 956, 375–393. [Google Scholar] [PubMed]
- Hayman, R.; Warren, A.; Brockelsby, J.; Johnson, I.; Baker, P. Plasma from women with pre-eclampsia induces an in vitro alteration in the endothelium-dependent behaviour of myometrial resistance arteries. BJOG 2000, 107, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Krupp, J.; Boeldt, D.S.; Yi, F.X.; Grummer, M.A.; Bankowski Anaya, H.A.; Shah, D.M.; Bird, I.M. The loss of sustained Ca2+ signaling underlies suppressed endothelial nitric oxide production in preeclamptic pregnancies: Implications for new therapy. Am. J. Physiol. Heart Circ. Physiol. 2013, 305, H969–H979. [Google Scholar] [CrossRef] [PubMed]
- Goulopoulou, S. Maternal vascular physiology in preeclampsia. Hypertension 2017, 70, 1066–1073. [Google Scholar] [CrossRef] [PubMed]
- Akar, F.; Ark, M.; Uydes, B.S.; Soysal, M.E.; Saracoglu, F.; Abacioglu, N.; Van de Voorde, J.; Kanzik, I. Nitric oxide production by human umbilical vessels in severe pre-eclampsia. J. Hypertens. 1994, 12, 1235–1241. [Google Scholar] [CrossRef] [PubMed]
- Steinert, J.R.; Wyatt, A.W.; Poston, L.; Jacob, R.; Mann, G.E. Preeclampsia is associated with altered Ca2+ regulation and no production in human fetal venous endothelial cells. FASEB J. 2002, 16, 721–723. [Google Scholar] [CrossRef] [PubMed]
- Mahdy, Z.; Otun, H.A.; Dunlop, W.; Gillespie, J.I. The responsiveness of isolated human hand vein endothelial cells in normal pregnancy and in pre-eclampsia. J. Physiol. 1998, 508 Pt 2, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Boeldt, D.S.; Bird, I.M. Vascular adaptation in pregnancy and endothelial dysfunction in preeclampsia. J. Endocrinol. 2017, 232, R27–R44. [Google Scholar] [CrossRef] [PubMed]
- Davidge, S.T.; Everson, W.V.; Parisi, V.M.; McLaughlin, M.K. Pregnancy and lipid peroxide-induced alterations of eicosanoid-metabolizing enzymes in the aorta of the rat. Am. J. Obstet. Gynecol. 1993, 169, 1338–1344. [Google Scholar] [CrossRef]
- Luksha, L.; Agewall, S.; Kublickiene, K. Endothelium-derived hyperpolarizing factor in vascular physiology and cardiovascular disease. Atherosclerosis 2009, 202, 330–344. [Google Scholar] [CrossRef] [PubMed]
- Luksha, L.; Nisell, H.; Luksha, N.; Kublickas, M.; Hultenby, K.; Kublickiene, K. Endothelium-derived hyperpolarizing factor in preeclampsia: Heterogeneous contribution, mechanisms, and morphological prerequisites. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R510–R519. [Google Scholar] [CrossRef] [PubMed]
- Wallukat, G.; Homuth, V.; Fischer, T.; Lindschau, C.; Horstkamp, B.; Jupner, A.; Baur, E.; Nissen, E.; Vetter, K.; Neichel, D.; et al. Patients with preeclampsia develop agonistic autoantibodies against the angiotensin at1 receptor. J. Clin. Investig. 1999, 103, 945–952. [Google Scholar] [CrossRef] [PubMed]
- Harmon, A.C.; Cornelius, D.C.; Amaral, L.M.; Faulkner, J.L.; Cunningham, M.W., Jr.; Wallace, K.; LaMarca, B. The role of inflammation in the pathology of preeclampsia. Clin. Sci. 2016, 130, 409–419. [Google Scholar] [CrossRef] [PubMed]
- Parrish, M.R.; Murphy, S.R.; Rutland, S.; Wallace, K.; Wenzel, K.; Wallukat, G.; Keiser, S.; Ray, L.F.; Dechend, R.; Martin, J.N.; et al. The effect of immune factors, tumor necrosis factor-alpha, and agonistic autoantibodies to the angiotensin ii type i receptor on soluble fms-like tyrosine-1 and soluble endoglin production in response to hypertension during pregnancy. Am. J. Hypertens. 2010, 23, 911–916. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Kellems, R.E. Angiotensin receptor agonistic autoantibodies and hypertension: Preeclampsia and beyond. Circ. Res. 2013, 113, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Yan, M.; Malinowski, A.K.; Shehata, N. Thrombocytopenic syndromes in pregnancy. Obstet. Med. 2016, 9, 15–20. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Exellence (NICE). Severe Hypertension, Severe Pre-Eclampsia and Eclampsia in Critical Care—Nice Clinical Guideline; Royal College of Obstetricians and Gynaecologists: London, UK, 2015. [Google Scholar]
- Kenny, L.C.; Black, M.A.; Poston, L.; Taylor, R.; Myers, J.E.; Baker, P.N.; McCowan, L.M.; Simpson, N.A.; Dekker, G.A.; Roberts, C.T.; et al. Early pregnancy prediction of preeclampsia in nulliparous women, combining clinical risk and biomarkers: The screening for pregnancy endpoints (scope) international cohort study. Hypertension 2014, 64, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Pare, E.; Parry, S.; McElrath, T.F.; Pucci, D.; Newton, A.; Lim, K.H. Clinical risk factors for preeclampsia in the 21st century. Obstet. Gynecol. 2014, 124, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Spradley, F.T. Metabolic abnormalities and obesity’s impact on the risk for developing preeclampsia. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2016, 312, R5–R12. [Google Scholar] [CrossRef] [PubMed]
- WHO. Who Recommendations for Prevention and Treatment of Pre-Eclampsia and Eclampsia, 2013/06/07 ed.; World Health Organization, Ed.; World Health Organization: Geneva, Switzerland, 2011; pp. 8–27. [Google Scholar]
- Roberge, S.; Villa, P.; Nicolaides, K.; Giguere, Y.; Vainio, M.; Bakthi, A.; Ebrashy, A.; Bujold, E. Early administration of low-dose aspirin for the prevention of preterm and term preeclampsia: A systematic review and meta-analysis. Fetal Diagn. Ther. 2012, 31, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Mol, B.W.; Roberts, C.T.; Thangaratinam, S.; Magee, L.A.; de Groot, C.J.; Hofmeyr, G.J. Pre-eclampsia. Lancet 2015, 387, 999–1011. [Google Scholar] [CrossRef]
- Tong, S.; Mol, B.W.; Walker, S.P. Preventing preeclampsia with aspirin: Does dose or timing matter? Am. J. Obstet. Gynecol. 2017, 216, 95–97. [Google Scholar] [CrossRef] [PubMed]
- Meher, S.; Duley, L.; Hunter, K.; Askie, L. Antiplatelet therapy before or after 16 weeks’ gestation for preventing preeclampsia: An individual participant data meta-analysis. Am. J. Obstet. Gynecol. 2017, 216, 121–128.e2. [Google Scholar] [CrossRef] [PubMed]
- National Collaborating Centre for Women’s and Children’s Health. Hypertension in Pregnancy: The Management of Hypertensive Disorders during Pregnancy—Nice Clinical Guideline, 2012/01/06 ed.; Royal College of Obstetricians and Gynaecologists: London, UK, 2010. [Google Scholar]
- Hofmeyr, G.J.; Lawrie, T.A.; Atallah, A.N.; Duley, L.; Torloni, M.R. Calcium supplementation during pregnancy for preventing hypertensive disorders and related problems. Cochrane Database Syst. Rev. 2014, CD001059. [Google Scholar] [CrossRef]
- WHO. Guideline: Calcium Supplementation in Pregnant Women, 2013/09/06 ed.; World Health Organization, Ed.; World Health Organization: Geneva, Switzerland, 2013; pp. 2–3. [Google Scholar]
- Inversetti, A.; Smid, M.; Candiani, M.; Ferrari, M.; Galbiati, S. Predictive biomarkers of pre-eclampsia and effectiveness of preventative interventions for the disease. Expert Opin. Biol. Ther. 2014, 14, 1161–1173. [Google Scholar] [CrossRef] [PubMed]
- Montenegro, N.; Campos, D.A.; Rodrigues, T.; Ramalho, C.; Silva, J.L.; Machado, A.P. Pré-eclâmpsia: Vigilância e tratamento. In Protocolos de Medicina Materno-Fetal, 3rd ed.; LIDEL, Ed.; LIDEL: Lisboa, Portugal, 2014; pp. 122–129. [Google Scholar]
- Júlio, C.; Francisco, C.; Dias, E.; Campos, A. Pré-eclâmpsia. In Protocolos de Atuação da Maternidade dr. Alfredo da Costa, 2nd ed.; LIDEL, Ed.; LIDEL: Lisboa, Portugal, 2011; pp. 39–48. [Google Scholar]
- Podymow, T.; August, P. Antihypertensive drugs in pregnancy. Semin. Nephrol. 2011, 31, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Bouet, P.E.; Brun, S.; Madar, H.; Baisson, A.L.; Courtay, V.; Gascoin-Lachambre, G.; Lasocki, S.; Sentilhes, L. Implementation of an antenatal magnesium sulfate protocol for fetal neuroprotection in preterm infants. Sci. Rep. 2015, 5, 14732. [Google Scholar] [CrossRef] [PubMed]
| Systems | Signs/Symptoms |
|---|---|
| Central Nervous system | Headaches |
| Visual disturbances | |
| Seizures (eclampsia) | |
| Renal system | Proteinuria |
| Oliguria | |
| Abnormal kidney tests | |
| Hypertension | |
| Vascular system | Severe hypertension |
| Cardiorespiratory system | Chest pain |
| Dyspnea | |
| Low oxygen saturation | |
| Pulmonary edema | |
| Hepatic system | Abnormal liver function |
| Epigastric pain | |
| Nausea | |
| Hematologic system | Hemorrhage |
| Coagulation impairment | |
| Intravascular disseminated coagulation | |
| Shock |
| Risk Factors for Pre-Eclampsia | Mean Relative Risk (95% Confidence Interval) | References |
|---|---|---|
| Antiphospholipid syndrome | 9.72 (4.34–21.75) | [16] |
| Relative risk of preeclampsia | 7.19 (5.85–8.83) | |
| Previous pre-eclampsia | 7.19 (5.85–8.83) | |
| Diabetes mellitus (type I or II) | 3.56 (2.54–4.99) | |
| Multiple pregnancy | 2.93 (2.04–4.21) | |
| First pregnancy | 2.91 (1.28–6.61) | |
| Familiar history of pre-eclampsia | 2.90 (1.70–4.93) | |
| BMI ≥ 35 Kg/m2 | 2.47 (1.66–3.67) | |
| Maternal age <20 or >40 years old | 1.96 (1.34–2.87) | |
| Chronic hypertension | 1.38 (1.01–1.87) | |
| Chronic autoimmune disease | 6.9 (1.1–42.3) | [19] |
| Venous thromboembolism (VTE) | 2.2 (1.3–3.7) | [20] |
| Intergestational interval ≥10 years | Similar to multiple pregnancy | [21] |
| Chronic kidney disease | 1.70 (1.30–2.23) * | [22] |
| Mild Pre-Eclampsia | ||
|---|---|---|
| Blood Pressure <150/100 mmHg | Blood Pressure ≥150/100 and <160/110 mmHg | |
Expectant management. The pregnant woman should maintain:
| First line | Second line |
|
| |
| Severe Pre-Eclampsia | ||
|---|---|---|
| First Line | Second Line | |
| Labetalol | Nifedipine | Hydralazine |
|
|
|
| Eclampsia | ||
|---|---|---|
| Magnesium Sulphate | ||
| Loading Dose | Maintenance Dose | “Booster” Dose (If Necessary) |
|
|
|
| If magnesium sulphate is contraindicated or if the patient is refractory to this treatment: Diazepam, 5 mg IV (5 min), repeat until max dose (20 mg). | ||
| Corticosteroids for Fetal Lung Maturation | |
|---|---|
| Corticotherapy should only be recommended if: Gestational age between 24 and 36 weeks Birth is planned or likely to happen in 7 days (limit) | |
| Betamethasone | Dexamethasone |
| 12 g IM, 2 doses with a 24 h interval. | 10 mg IV, 2 doses with a 24 h interval. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peres, G.M.; Mariana, M.; Cairrão, E. Pre-Eclampsia and Eclampsia: An Update on the Pharmacological Treatment Applied in Portugal. J. Cardiovasc. Dev. Dis. 2018, 5, 3. https://doi.org/10.3390/jcdd5010003
Peres GM, Mariana M, Cairrão E. Pre-Eclampsia and Eclampsia: An Update on the Pharmacological Treatment Applied in Portugal. Journal of Cardiovascular Development and Disease. 2018; 5(1):3. https://doi.org/10.3390/jcdd5010003
Chicago/Turabian StylePeres, Gonçalo Miguel, Melissa Mariana, and Elisa Cairrão. 2018. "Pre-Eclampsia and Eclampsia: An Update on the Pharmacological Treatment Applied in Portugal" Journal of Cardiovascular Development and Disease 5, no. 1: 3. https://doi.org/10.3390/jcdd5010003
APA StylePeres, G. M., Mariana, M., & Cairrão, E. (2018). Pre-Eclampsia and Eclampsia: An Update on the Pharmacological Treatment Applied in Portugal. Journal of Cardiovascular Development and Disease, 5(1), 3. https://doi.org/10.3390/jcdd5010003






