Myocarditis in Paediatric Patients: Unveiling the Progression to Dilated Cardiomyopathy and Heart Failure
Abstract
:1. Introduction
2. Aetiology
2.1. Infections
2.2. Hypersensitivity to Drugs and Other Substances
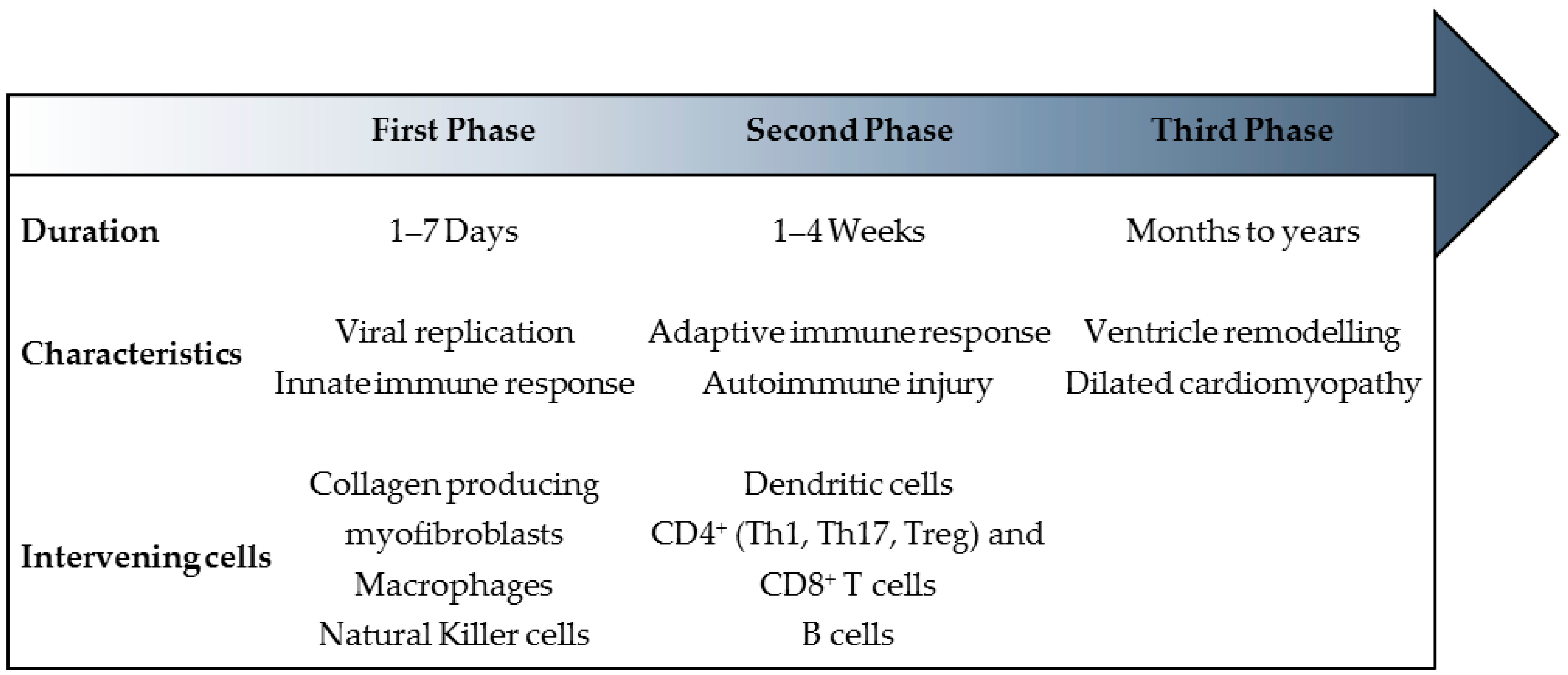
3. Myocarditis: A Triphasic Disease Process
4. Pathogenic Mechanisms
5. Clinical Presentation
6. Differential Diagnoses
7. Diagnosis and Treatment: A Triphasic Approach
7.1. First phase: Viral Replication
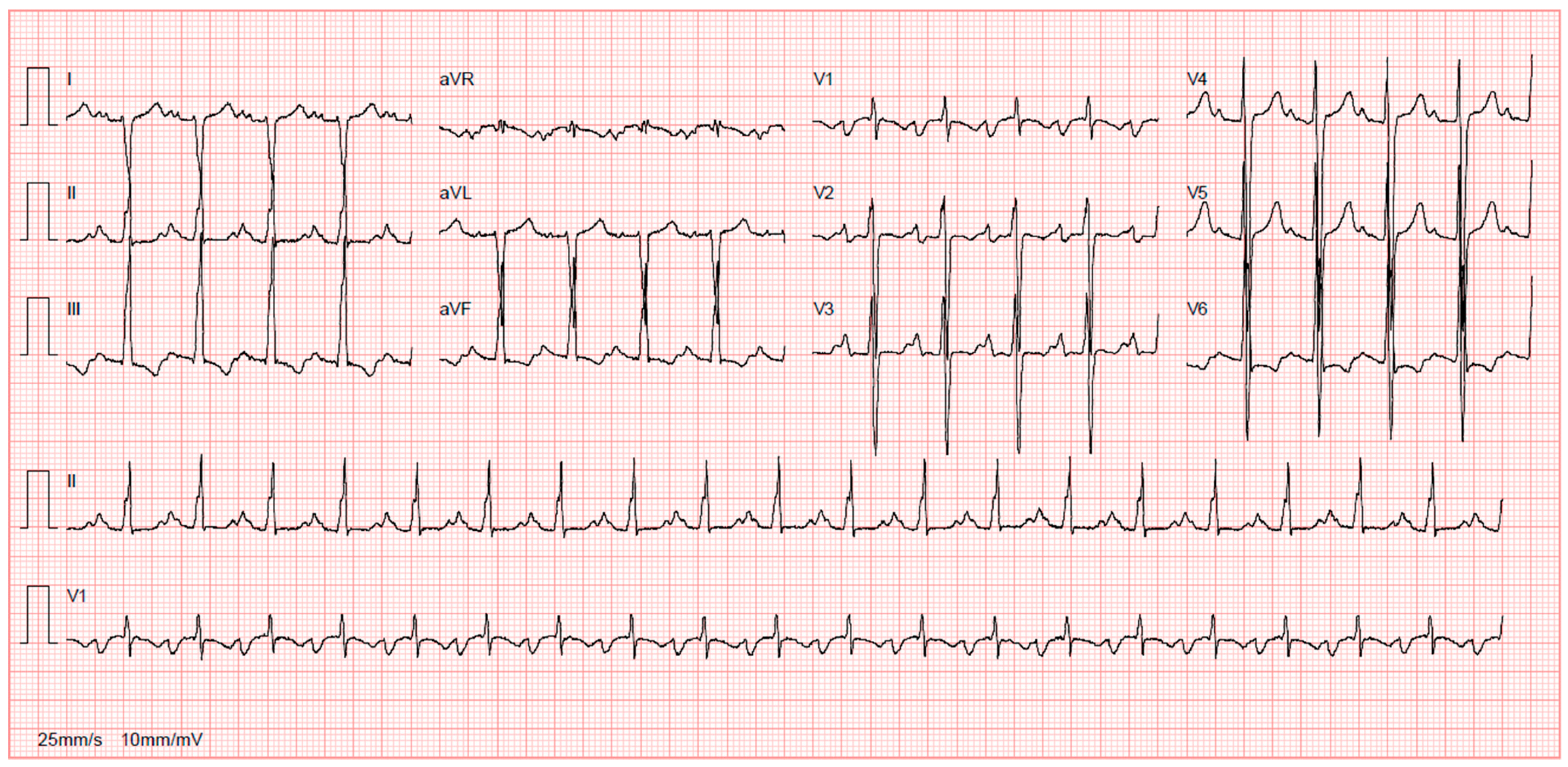
7.1.1. Diagnosis
7.1.2. Treatment
7.2. Second Phase: Autoimmune Activation
7.2.1. Diagnosis
7.2.2. Treatment
7.3. Third Phase: Dilated Cardiomyopathy
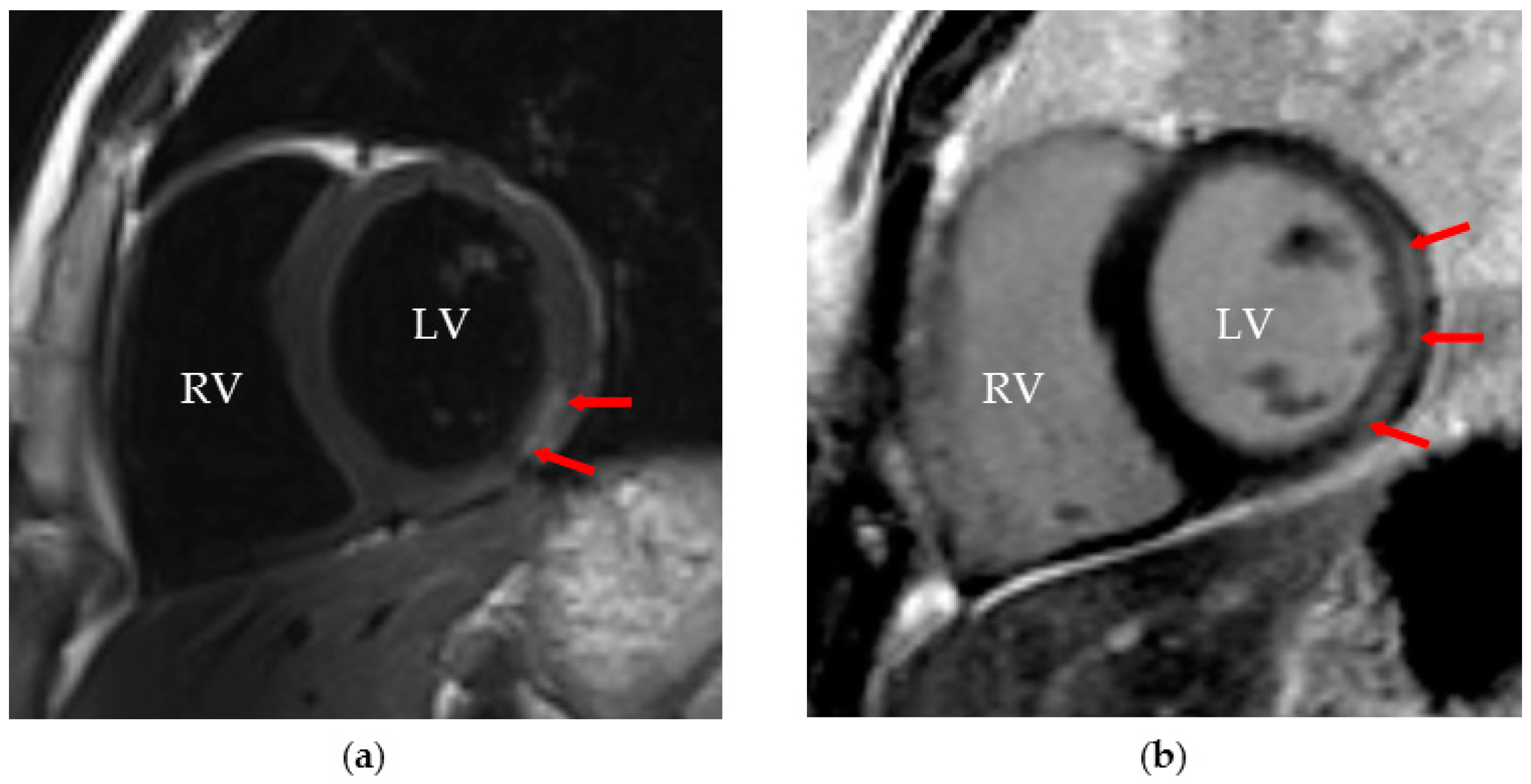
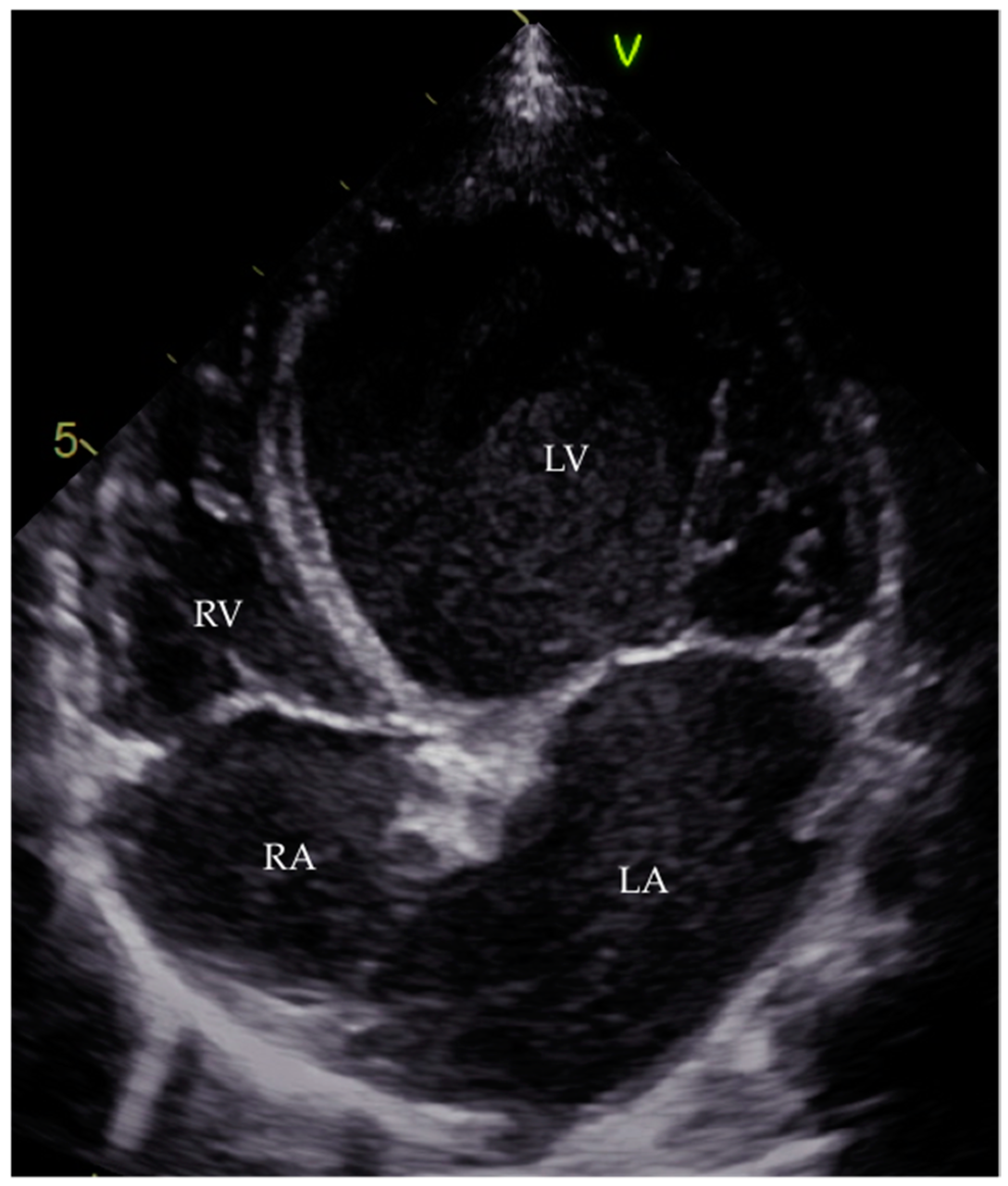
7.3.1. Diagnosis
7.3.2. Treatment
8. Prognosis
9. New Perspectives
10. Conclusions
Conflicts of Interest
References
- Elliott, P.; Andersson, B.; Arbustini, E.; Bilinska, Z.; Cecchi, F.; Charron, P.; Dubourg, O.; Kuhl, U.; Maisch, B.; McKenna, W.J.; et al. Classification of the cardiomyopathies: A position statement from the european society of cardiology working group on myocardial and pericardial diseases. Eur. Heart J. 2008, 29, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Ellis, C.R.; Di Salvo, T. Myocarditis: Basic and clinical aspects. Cardiol. Rev. 2007, 15, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Shauer, A.; Gotsman, I.; Keren, A.; Zwas, D.R.; Hellman, Y.; Durst, R.; Admon, D. Acute viral myocarditis: Current concepts in diagnosis and treatment. Isr. Med. Assoc. J. 2013, 15, 180–185. [Google Scholar] [PubMed]
- Durani, Y.; Giordano, K.; Goudie, B.W. Myocarditis and pericarditis in children. Pediatr. Clin. N. Am. 2010, 57, 1281–1303. [Google Scholar] [CrossRef] [PubMed]
- Richardson, P.; McKenna, W.; Bristow, M.; Maisch, B.; Mautner, B.; O’Connell, J.; Olsen, E.; Thiene, G.; Goodwin, J.; Gyarfas, I.; et al. Report of the 1995 world health organization/international society and federation of cardiology task force on the definition and classification of cardiomyopathies. Circulation 1996, 93, 841–842. [Google Scholar] [PubMed]
- Stiller, B. Management of myocarditis in children: The current situation. Adv. Exp. Med. Biol. 2008, 609, 196–215. [Google Scholar] [PubMed]
- Kuhl, U.; Schultheiss, H.P. Myocarditis in children. Heart Fail. Clin. 2010, 6, 483–496. [Google Scholar] [CrossRef] [PubMed]
- Bowles, N.E.; Bowles, K.R.; Towbin, J.A. Viral genomic detection and outcome in myocarditis. Heart Fail. Clin. 2005, 1, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.B.; Webber, S.; Fricker, F.J.; Jaffe, R.; Demmler, G.; Kearney, D.; Zhang, Y.H.; Bodurtha, J.; Gelb, B.; Ni, J.; et al. Acute myocarditis. Rapid diagnosis by PCR in children. Circulation 1994, 90, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Canter, C.E.; Simpson, K.E. Diagnosis and treatment of myocarditis in children in the current era. Circulation 2014, 129, 115–128. [Google Scholar] [CrossRef] [PubMed]
- Dettmeyer, R.; Baasner, A.; Schlamann, M.; Haag, C.; Madea, B. Coxsackie B3 myocarditis in 4 cases of suspected sudden infant death syndrome: Diagnosis by immunohistochemical and molecular-pathologic investigations. Pathol. Res. Pract. 2002, 198, 689–696. [Google Scholar] [CrossRef] [PubMed]
- Mahrholdt, H.; Wagner, A.; Deluigi, C.C.; Kispert, E.; Hager, S.; Meinhardt, G.; Vogelsberg, H.; Fritz, P.; Dippon, J.; Bock, C.T.; et al. Presentation, patterns of myocardial damage, and clinical course of viral myocarditis. Circulation 2006, 114, 1581–1590. [Google Scholar] [CrossRef] [PubMed]
- Comar, M.; D’Agaro, P.; Campello, C.; Poli, A.; Breinholt, J.P., 3rd; Towbin, J.A.; Vatta, M. Human herpes virus 6 in archival cardiac tissues from children with idiopathic dilated cardiomyopathy or congenital heart disease. J. Clin. Pathol. 2009, 62, 80–83. [Google Scholar] [CrossRef] [PubMed]
- Molina, K.M.; Garcia, X.; Denfield, S.W.; Fan, Y.; Morrow, W.R.; Towbin, J.A.; Frazier, E.A.; Nelson, D.P. Parvovirus B19 myocarditis causes significant morbidity and mortality in children. Pediatr. Cardiol. 2013, 34, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Bowles, N.E.; Ni, J.; Kearney, D.L.; Pauschinger, M.; Schultheiss, H.P.; McCarthy, R.; Hare, J.; Bricker, J.T.; Bowles, K.R.; Towbin, J.A. Detection of viruses in myocardial tissues by polymerase chain reaction. Evidence of adenovirus as a common cause of myocarditis in children and adults. J. Am. Coll. Cardiol. 2003, 42, 466–472. [Google Scholar] [CrossRef]
- Camargo, P.R.; Okay, T.S.; Yamamoto, L.; Del Negro, G.M.; Lopes, A.A. Myocarditis in children and detection of viruses in myocardial tissue: Implications for immunosuppressive therapy. Int. J. Cardiol. 2011, 148, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Matsumori, A.; Shimada, T.; Chapman, N.M.; Tracy, S.M.; Mason, J.W. Myocarditis and heart failure associated with hepatitis c virus infection. J. Card. Fail. 2006, 12, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Savon, C.; Acosta, B.; Valdes, O.; Goyenechea, A.; Gonzalez, G.; Pinon, A.; Mas, P.; Rosario, D.; Capo, V.; Kouri, V.; et al. A myocarditis outbreak with fatal cases associated with adenovirus subgenera c among children from havana city in 2005. J. Clin. Virol. 2008, 43, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Talsma, M.D.; Kroos, M.A.; Visser, G.; Kimpen, J.L.; Niezen, K.E. A rare presentation of childhood pompe disease: Cardiac involvement provoked by epstein-barr virus infection. Pediatrics 2002, 109, e65. [Google Scholar] [CrossRef] [PubMed]
- Bratincsak, A.; El-Said, H.G.; Bradley, J.S.; Shayan, K.; Grossfeld, P.D.; Cannavino, C.R. Fulminant myocarditis associated with pandemic H1N1 influenza a virus in children. J. Am. Coll. Cardiol. 2010, 55, 928–929. [Google Scholar] [CrossRef] [PubMed]
- Blauwet, L.A.; Cooper, L.T. Myocarditis. Prog. Cardiovasc. Dis. 2010, 52, 274–288. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Hemal, A.; Agarwal, S.; Kumar, D. Cardiac manifestations in hiv infected children. Indian J. Pediatr. 2015, 82, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Keesler, M.J.; Fisher, S.D.; Lipshultz, S.E. Cardiac manifestations of hiv infection in infants and children. Ann. N. Y. Acad. Sci. 2001, 946, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Bowles, N.E.; Kearney, D.L.; Ni, J.; Perez-Atayde, A.R.; Kline, M.W.; Bricker, J.T.; Ayres, N.A.; Lipshultz, S.E.; Shearer, W.T.; Towbin, J.A. The detection of viral genomes by polymerase chain reaction in the myocardium of pediatric patients with advanced hiv disease. J. Am. Coll. Cardiol. 1999, 34, 857–865. [Google Scholar] [CrossRef]
- Cooper, L.T., Jr. Myocarditis. N. Engl. J. Med. 2009, 360, 1526–1538. [Google Scholar] [CrossRef] [PubMed]
- Galazka, A. The changing epidemiology of diphtheria in the vaccine era. J. Infect. Dis. 2000, 181 (Suppl. 1), S2–S9. [Google Scholar] [CrossRef] [PubMed]
- Sood, S.K. Lyme disease in children. Infect. Dis. Clin. N. Am. 2015, 29, 281–294. [Google Scholar] [CrossRef] [PubMed]
- Costello, J.M.; Alexander, M.E.; Greco, K.M.; Perez-Atayde, A.R.; Laussen, P.C. Lyme carditis in children: Presentation, predictive factors, and clinical course. Pediatrics 2009, 123, e835–e841. [Google Scholar] [CrossRef] [PubMed]
- Punukollu, G.; Gowda, R.M.; Khan, I.A.; Navarro, V.S.; Vasavada, B.C. Clinical aspects of the chagas’ heart disease. Int. J. Cardiol. 2007, 115, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.M.; Vaz, M.; Kotha, S.; Santosh, N.H. Dapsone hypersensitivity syndrome with myocarditis. J. Assoc. Phys. India 2014, 62, 728–731. [Google Scholar] [PubMed]
- Teo, R.Y.; Tay, Y.K.; Tan, C.H.; Ng, V.; Oh, D.C. Presumed dapsone-induced drug hypersensitivity syndrome causing reversible hypersensitivity myocarditis and thyrotoxicosis. Ann. Acad. Med. Singap. 2006, 35, 833–836. [Google Scholar] [PubMed]
- Park, Y.; Ahn, S.G.; Ko, A.; Ra, S.H.; Cha, J.; Jee, Y.G.; Lee, J.H. Hypersensitivity myocarditis confirmed by cardiac magnetic resonance imaging and endomyocardial biopsy. Korean J. Int. Med. 2014, 29, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Cassimatis, D.C.; Atwood, J.E.; Engler, R.M.; Linz, P.E.; Grabenstein, J.D.; Vernalis, M.N. Smallpox vaccination and myopericarditis: A clinical review. J. Am. Coll. Cardiol. 2004, 43, 1503–1510. [Google Scholar] [CrossRef] [PubMed]
- Dilber, E.; Karagoz, T.; Aytemir, K.; Ozer, S.; Alehan, D.; Oto, A.; Celiker, A. Acute myocarditis associated with tetanus vaccination. Mayo Clin. Proc. 2003, 78, 1431–1433. [Google Scholar] [CrossRef] [PubMed]
- Simmons, A.; Vacek, J.L.; Meyers, D. Anthracycline-induced cardiomyopathy. Postgrad. Med. 2008, 120, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Taliercio, C.P.; Olney, B.A.; Lie, J.T. Myocarditis related to drug hypersensitivity. Mayo Clin. Proc. 1985, 60, 463–468. [Google Scholar] [CrossRef]
- Liu, P.P.; Mason, J.W. Advances in the understanding of myocarditis. Circulation 2001, 104, 1076–1082. [Google Scholar] [CrossRef] [PubMed]
- Mason, J.W. Myocarditis and dilated cardiomyopathy: An inflammatory link. Cardiovasc. Res. 2003, 60, 5–10. [Google Scholar] [CrossRef]
- Miranda, J.O.; Costa, L.; Rodrigues, E.; Teles, E.L.; Baptista, M.J.; Areias, J.C. Paediatric dilated cardiomyopathy: Clinical profile and outcome. The experience of a tertiary centre for paediatric cardiology. Cardiol. Young 2015, 25, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Kawai, C. From myocarditis to cardiomyopathy: Mechanisms of inflammation and cell death: Learning from the past for the future. Circulation 1999, 99, 1091–1100. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, F.; Thiene, G. Myocarditis and inflammatory cardiomyopathy: Microbiological and molecular biological aspects. Cardiovasc. Res. 2003, 60, 11–25. [Google Scholar] [CrossRef]
- Bergelson, J.M. Receptors mediating adenovirus attachment and internalization. Biochem. Pharmacol. 1999, 57, 975–979. [Google Scholar] [CrossRef]
- Bergelson, J.M.; Cunningham, J.A.; Droguett, G.; Kurt-Jones, E.A.; Krithivas, A.; Hong, J.S.; Horwitz, M.S.; Crowell, R.L.; Finberg, R.W. Isolation of a common receptor for coxsackie b viruses and adenoviruses 2 and 5. Science 1997, 275, 1320–1323. [Google Scholar] [CrossRef] [PubMed]
- Bultmann, B.D.; Klingel, K.; Sotlar, K.; Bock, C.T.; Baba, H.A.; Sauter, M.; Kandolf, R. Fatal parvovirus b19-associated myocarditis clinically mimicking ischemic heart disease: An endothelial cell-mediated disease. Hum. Pathol. 2003, 34, 92–95. [Google Scholar] [CrossRef] [PubMed]
- Koehl, B.; Oualha, M.; Lesage, F.; Rambaud, C.; Canioni, D.; Hubert, P.; Leruez-Ville, M. Fatal parvovirus B19 myocarditis in children and possible dysimmune mechanism. Pediatr. Inf. Dis. J. 2012, 31, 418–421. [Google Scholar] [CrossRef] [PubMed]
- Kuhl, U.; Pauschinger, M.; Bock, T.; Klingel, K.; Schwimmbeck, C.P.; Seeberg, B.; Krautwurm, L.; Poller, W.; Schultheiss, H.P.; Kandolf, R. Parvovirus B19 infection mimicking acute myocardial infarction. Circulation 2003, 108, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.P. New concepts in myocarditis: Crossroads in the 1990s. Prog. Pediatr. Cardiol. 1992, 1, 37–47. [Google Scholar] [CrossRef]
- Matsumori, A.; Yamada, T.; Suzuki, H.; Matoba, Y.; Sasayama, S. Increased circulating cytokines in patients with myocarditis and cardiomyopathy. Br. Heart J. 1994, 72, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Binah, O. Cytotoxic lymphocytes and cardiac electrophysiology. J. Mol. Cell. Cardiol. 2002, 34, 1147–1161. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.P.; Opavsky, M.A. Viral myocarditis: Receptors that bridge the cardiovascular with the immune system? Circ. Res. 2000, 86, 253–254. [Google Scholar] [CrossRef] [PubMed]
- Kindermann, I.; Barth, C.; Mahfoud, F.; Ukena, C.; Lenski, M.; Yilmaz, A.; Klingel, K.; Kandolf, R.; Sechtem, U.; Cooper, L.T.; et al. Update on myocarditis. J. Am. Coll. Cardiol. 2012, 59, 779–792. [Google Scholar] [CrossRef] [PubMed]
- Caforio, A.L.; Mahon, N.J.; Tona, F.; McKenna, W.J. Circulating cardiac autoantibodies in dilated cardiomyopathy and myocarditis: Pathogenetic and clinical significance. Eur. J. Heart Fail. 2002, 4, 411–417. [Google Scholar] [CrossRef]
- Kuhl, U.; Pauschinger, M.; Noutsias, M.; Seeberg, B.; Bock, T.; Lassner, D.; Poller, W.; Kandolf, R.; Schultheiss, H.P. High prevalence of viral genomes and multiple viral infections in the myocardium of adults with “idiopathic” left ventricular dysfunction. Circulation 2005, 111, 887–893. [Google Scholar] [CrossRef] [PubMed]
- Sinagra, G.; Anzini, M.; Pereira, N.L.; Bussani, R.; Finocchiaro, G.; Bartunek, J.; Merlo, M. Myocarditis in clinical practice. Mayo Clin. Proc. 2016, 91, 1256–1266. [Google Scholar] [CrossRef] [PubMed]
- Angelini, A.; Calzolari, V.; Calabrese, F.; Boffa, G.M.; Maddalena, F.; Chioin, R.; Thiene, G. Myocarditis mimicking acute myocardial infarction: Role of endomyocardial biopsy in the differential diagnosis. Heart 2000, 84, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, M.; Kuwata, S.; Kurishima, C.; Kim, J.H.; Iwamoto, Y.; Senzaki, H. Cardiac biomarkers in children with congenital heart disease. World J. Pediatr. WJP 2015, 11, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.J.; Chao, H.C.; Hsia, S.H.; Yan, D.C. Myocarditis presenting as gastritis in children. Pediatr. Emerg. Care 2006, 22, 439–440. [Google Scholar] [CrossRef] [PubMed]
- Chavda, K.K.; Dhuper, S.; Madhok, A.; Chowdhury, D. Seizures secondary to a high-grade atrioventricular block as a presentation of acute myocarditis. Pediatr. Emerg. Care 2004, 20, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Durani, Y.; Egan, M.; Baffa, J.; Selbst, S.M.; Nager, A.L. Pediatric myocarditis: Presenting clinical characteristics. Am. J. Emerg. Med. 2009, 27, 942–947. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, M.A.; da Silva, R.P.; de Morais, S.C.; Fragata Filho, A.A.; Correia Ede, B. Clinical aspects and development of dilated cardiomyopathy in infants and children. Arq. Bras. Cardiol. 1991, 56, 213–218. [Google Scholar] [PubMed]
- Amabile, N.; Fraisse, A.; Bouvenot, J.; Chetaille, P.; Ovaert, C. Outcome of acute fulminant myocarditis in children. Heart 2006, 92, 1269–1273. [Google Scholar] [CrossRef] [PubMed]
- Kleinert, S.; Weintraub, R.G.; Wilkinson, J.L.; Chow, C.W. Myocarditis in children with dilated cardiomyopathy: Incidence and outcome after dual therapy immunosuppression. J. Heart Lung Transplant. 1997, 16, 1248–1254. [Google Scholar] [PubMed]
- Dancea, A.B. Myocarditis in infants and children: A review for the paediatrician. Paediatr. Child Health 2001, 6, 543–545. [Google Scholar] [PubMed]
- Cox, G.F.; Sleeper, L.A.; Lowe, A.M.; Towbin, J.A.; Colan, S.D.; Orav, E.J.; Lurie, P.R.; Messere, J.E.; Wilkinson, J.D.; Lipshultz, S.E. Factors associated with establishing a causal diagnosis for children with cardiomyopathy. Pediatrics 2006, 118, 1519–1531. [Google Scholar] [CrossRef] [PubMed]
- Herath, V.C.; Gentles, T.L.; Skinner, J.R. Dilated cardiomyopathy in children: Review of all presentations to a children’s hospital over a 5-year period and the impact of family cardiac screening. J. Paediatr. Child Health 2015, 51, 595–599. [Google Scholar] [CrossRef] [PubMed]
- Towbin, J.A.; Lowe, A.M.; Colan, S.D.; Sleeper, L.A.; Orav, E.J.; Clunie, S.; Messere, J.; Cox, G.F.; Lurie, P.R.; Hsu, D.; et al. Incidence, causes, and outcomes of dilated cardiomyopathy in children. JAMA 2006, 296, 1867–1876. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, J.D.; Westphal, J.A.; Bansal, N.; Czachor, J.D.; Razoky, H.; Lipshultz, S.E. Lessons learned from the pediatric cardiomyopathy registry (PCMR) study group. Cardiology Young 2015, 25 (Suppl. 2), 140–153. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.D.; Hammer, G.B. Cardiomyopathy in childhood. Curr. Opin. Anaesthesiol. 2011, 24, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Franklin, O.; Burch, M. Dilated cardiomyopathy in childhood. Images Paediatr. Cardiol. 2000, 2, 3–10. [Google Scholar] [PubMed]
- Hershberger, R.E.; Morales, A. Dilated cardiomyopathy overview. In GeneReviews(R); Pagon, R.A., Adam, M.P., Ardinger, H.H., Wallace, S.E., Amemiya, A., Bean, L.J.H., Bird, T.D., Fong, C.T., Mefford, H.C., Smith, R.J.H., et al., Eds.; University of Washington: Seattle, WA, USA, 2007. [Google Scholar]
- Kantor, P.F.; Lougheed, J.; Dancea, A.; McGillion, M.; Barbosa, N.; Chan, C.; Dillenburg, R.; Atallah, J.; Buchholz, H.; Chant-Gambacort, C.; et al. Presentation, diagnosis, and medical management of heart failure in children: Canadian cardiovascular society guidelines. Can. J. Cardiol. 2013, 29, 1535–1552. [Google Scholar] [CrossRef] [PubMed]
- Aretz, H.T.; Billingham, M.E.; Edwards, W.D.; Factor, S.M.; Fallon, J.T.; Fenoglio, J.J., Jr.; Olsen, E.G.; Schoen, F.J. Myocarditis. A histopathologic definition and classification. Am. J. Cardiovasc. Pathol. 1987, 1, 3–14. [Google Scholar] [PubMed]
- Freedman, S.B.; Haladyn, J.K.; Floh, A.; Kirsh, J.A.; Taylor, G.; Thull-Freedman, J. Pediatric myocarditis: Emergency department clinical findings and diagnostic evaluation. Pediatrics 2007, 120, 1278–1285. [Google Scholar] [CrossRef] [PubMed]
- Kern, J.; Modi, R.; Atalay, M.K.; Kochilas, L.K. Clinical myocarditis masquerading as acute coronary syndrome. J. Pediatr. 2009, 154, 612–615. [Google Scholar] [CrossRef] [PubMed]
- Monney, P.A.; Sekhri, N.; Burchell, T.; Knight, C.; Davies, C.; Deaner, A.; Sheaf, M.; Baithun, S.; Petersen, S.; Wragg, A.; et al. Acute myocarditis presenting as acute coronary syndrome: Role of early cardiac magnetic resonance in its diagnosis. Heart 2011, 97, 1312–1318. [Google Scholar] [CrossRef] [PubMed]
- Fung, G.; Luo, H.; Qiu, Y.; Yang, D.; McManus, B. Myocarditis. Circ. Res. 2016, 118, 496–514. [Google Scholar] [CrossRef] [PubMed]
- Morgera, T.; Di Lenarda, A.; Dreas, L.; Pinamonti, B.; Humar, F.; Bussani, R.; Silvestri, F.; Chersevani, D.; Camerini, F. Electrocardiography of myocarditis revisited: Clinical and prognostic significance of electrocardiographic changes. Am. Heart J. 1992, 124, 455–467. [Google Scholar] [CrossRef]
- Levine, M.C.; Klugman, D.; Teach, S.J. Update on myocarditis in children. Curr. Opin. Pediatr. 2010, 22, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Lurz, P.; Eitel, I.; Adam, J.; Steiner, J.; Grothoff, M.; Desch, S.; Fuernau, G.; de Waha, S.; Sareban, M.; Luecke, C.; et al. Diagnostic performance of cmr imaging compared with emb in patients with suspected myocarditis. JACC Cardiovasc. Imaging 2012, 5, 513–524. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Aty, H.; Boye, P.; Zagrosek, A.; Wassmuth, R.; Kumar, A.; Messroghli, D.; Bock, P.; Dietz, R.; Friedrich, M.G.; Schulz-Menger, J. Diagnostic performance of cardiovascular magnetic resonance in patients with suspected acute myocarditis: Comparison of different approaches. J. Am. Coll. Cardiol. 2005, 45, 1815–1822. [Google Scholar] [CrossRef] [PubMed]
- Fenster, B.E.; Chan, F.P.; Valentine, H.A.; Yang, E.; McConnell, M.V.; Berry, G.J.; Yang, P.C. Images in cardiovascular medicine. Cardiac magnetic resonance imaging for myocarditis: Effective use in medical decision making. Circulation 2006, 113, e842–e843. [Google Scholar] [CrossRef] [PubMed]
- Etesami, M.; Gilkeson, R.C.; Rajiah, P. Utility of late gadolinium enhancement in pediatric cardiac mri. Pediatr. Radiol. 2016, 46, 1096–1113. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, M.G.; Sechtem, U.; Schulz-Menger, J.; Holmvang, G.; Alakija, P.; Cooper, L.T.; White, J.A.; Abdel-Aty, H.; Gutberlet, M.; Prasad, S.; et al. Cardiovascular magnetic resonance in myocarditis: A jacc white paper. J. Am. Coll. Cardiol. 2009, 53, 1475–1487. [Google Scholar] [CrossRef] [PubMed]
- Mahrholdt, H.; Goedecke, C.; Wagner, A.; Meinhardt, G.; Athanasiadis, A.; Vogelsberg, H.; Fritz, P.; Klingel, K.; Kandolf, R.; Sechtem, U. Cardiovascular magnetic resonance assessment of human myocarditis: A comparison to histology and molecular pathology. Circulation 2004, 109, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Luetkens, J.A.; Homsi, R.; Sprinkart, A.M.; Doerner, J.; Dabir, D.; Kuetting, D.L.; Block, W.; Andrie, R.; Stehning, C.; Fimmers, R.; et al. Incremental value of quantitative cmr including parametric mapping for the diagnosis of acute myocarditis. Eur. Heart J. Cardiovasc. Imaging 2016, 17, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Lurz, P.; Luecke, C.; Eitel, I.; Fohrenbach, F.; Frank, C.; Grothoff, M.; de Waha, S.; Rommel, K.P.; Lurz, J.A.; Klingel, K.; et al. Comprehensive cardiac magnetic resonance imaging in patients with suspected myocarditis: The myoracer-trial. J. Am. Coll. Cardiol. 2016, 67, 1800–1811. [Google Scholar] [CrossRef] [PubMed]
- Goitein, O.; Matetzky, S.; Beinart, R.; di Segni, E.; Hod, H.; Bentancur, A.; Konen, E. Acute myocarditis: Noninvasive evaluation with cardiac mri and transthoracic echocardiography. AJR Am. J. Roentgenol. 2009, 192, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Di Filippo, S. Improving outcomes of acute myocarditis in children. Expert Rev. Cardiovasc. Ther. 2016, 14, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Caforio, A.L.; Pankuweit, S.; Arbustini, E.; Basso, C.; Gimeno-Blanes, J.; Felix, S.B.; Fu, M.; Helio, T.; Heymans, S.; Jahns, R.; et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: A position statement of the european society of cardiology working group on myocardial and pericardial diseases. Eur. Heart J. 2013, 34, 2636–2648. [Google Scholar] [CrossRef] [PubMed]
- Pollack, A.; Kontorovich, A.R.; Fuster, V.; Dec, G.W. Viral myocarditis–diagnosis, treatment options, and current controversies. Nat. Rev. Cardiol. 2015, 12, 670–680. [Google Scholar] [CrossRef] [PubMed]
- Feldman, A.M.; McNamara, D. Myocarditis. N. Engl. J. Med. 2000, 343, 1388–1398. [Google Scholar] [CrossRef] [PubMed]
- Holzmann, M.; Nicko, A.; Kuhl, U.; Noutsias, M.; Poller, W.; Hoffmann, W.; Morguet, A.; Witzenbichler, B.; Tschope, C.; Schultheiss, H.P.; et al. Complication rate of right ventricular endomyocardial biopsy via the femoral approach: A retrospective and prospective study analyzing 3048 diagnostic procedures over an 11-year period. Circulation 2008, 118, 1722–1728. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, A.; Kindermann, I.; Kindermann, M.; Mahfoud, F.; Ukena, C.; Athanasiadis, A.; Hill, S.; Mahrholdt, H.; Voehringer, M.; Schieber, M.; et al. Comparative evaluation of left and right ventricular endomyocardial biopsy: Differences in complication rate and diagnostic performance. Circulation 2010, 122, 900–909. [Google Scholar] [CrossRef] [PubMed]
- Mills, K.I.; Vincent, J.A.; Zuckerman, W.A.; Hoffman, T.M.; Canter, C.E.; Marshall, A.C.; Blume, E.D.; Bergersen, L.; Daly, K.P. Is endomyocardial biopsy a safe and useful procedure in children with suspected cardiomyopathy? Pediatr. Cardiol. 2016, 37, 1200–1210. [Google Scholar] [CrossRef] [PubMed]
- Stiller, B.; Weng, Y.; Hubler, M.; Lemmer, J.; Nagdyman, N.; Redlin, M.; Lange, P.E.; Hetzer, R. Pneumatic pulsatile ventricular assist devices in children under 1 year of age. Eur. J. Cardiothorac. Surg. 2005, 28, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Duncan, B.W.; Bohn, D.J.; Atz, A.M.; French, J.W.; Laussen, P.C.; Wessel, D.L. Mechanical circulatory support for the treatment of children with acute fulminant myocarditis. J. Thorac. Cardiovasc. Surg. 2001, 122, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.J.; McCrindle, B.W.; Bohn, D.J.; Wilson, G.J.; Taylor, G.P.; Freedom, R.M.; Smallhorn, J.F.; Benson, L.N. Clinical outcomes of acute myocarditis in childhood. Heart 1999, 82, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Levi, D.; Alejos, J. Diagnosis and treatment of pediatric viral myocarditis. Curr. Opin. Cardiol. 2001, 16, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Rajagopal, S.K.; Almond, C.S.; Laussen, P.C.; Rycus, P.T.; Wypij, D.; Thiagarajan, R.R. Extracorporeal membrane oxygenation for the support of infants, children, and young adults with acute myocarditis: A review of the extracorporeal life support organization registry. Crit. Care Med. 2010, 38, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.T.; Morris, D.J.; Wilkins, E.G. Successful diagnosis and management of cytomegalovirus carditis. J. Infect. 1997, 34, 243–247. [Google Scholar] [CrossRef]
- Dominguez, F.; Kuhl, U.; Pieske, B.; Garcia-Pavia, P.; Tschope, C. Update on myocarditis and inflammatory cardiomyopathy: Reemergence of endomyocardial biopsy. Rev. Esp. Cardiol. 2016, 69, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Schultheiss, H.P. Dilated cardiomyopathy—A chronic myocarditis? New aspects on diagnosis and therapy. Z. Fur Kardiol. 1993, 82 (Suppl. 4), 25–32. [Google Scholar]
- Escher, F.; Kuhl, U.; Lassner, D.; Stroux, A.; Westermann, D.; Skurk, C.; Tschope, C.; Poller, W.; Schultheiss, H.P. Presence of perforin in endomyocardial biopsies of patients with inflammatory cardiomyopathy predicts poor outcome. Eur. J. Heart Fail. 2014, 16, 1066–1072. [Google Scholar] [CrossRef] [PubMed]
- Kuhl, U.; Schultheiss, H.P. Viral myocarditis. Swiss Med. Wkly. 2014, 144, w14010. [Google Scholar] [CrossRef] [PubMed]
- Magnani, J.W.; Dec, G.W. Myocarditis: Current trends in diagnosis and treatment. Circulation 2006, 113, 876–890. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, K.R.; Kharbanda, A.; Haveman, L. Myocarditis and pericarditis in the pediatric patient: Validated management strategies. Pediatr. Emerg. Med. Pract. 2015, 12, 1–22; quiz 23. [Google Scholar] [PubMed]
- Aziz, K.U.; Patel, N.; Sadullah, T.; Tasneem, H.; Thawerani, H.; Talpur, S. Acute viral myocarditis: Role of immunosuppression: A prospective randomised study. Cardiol. Young 2010, 20, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Gagliardi, M.G.; Bevilacqua, M.; Bassano, C.; Leonardi, B.; Boldrini, R.; Camassei, F.D.; Fierabracci, A.; Ugazio, A.G.; Bottazzo, G.F. Long term follow up of children with myocarditis treated by immunosuppression and of children with dilated cardiomyopathy. Heart 2004, 90, 1167–1171. [Google Scholar] [CrossRef] [PubMed]
- Hia, C.P.; Yip, W.C.; Tai, B.C.; Quek, S.C. Immunosuppressive therapy in acute myocarditis: An 18 year systematic review. Arch. Dis. Child. 2004, 89, 580–584. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.; Hartling, L.; Vandermeer, B.; Klassen, T.P. Intravenous immunoglobulin for presumed viral myocarditis in children and adults. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef]
- Drucker, N.A.; Colan, S.D.; Lewis, A.B.; Beiser, A.S.; Wessel, D.L.; Takahashi, M.; Baker, A.L.; Perez-Atayde, A.R.; Newburger, J.W. Gamma-globulin treatment of acute myocarditis in the pediatric population. Circulation 1994, 89, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Nigro, G.; Bastianon, V.; Colloridi, V.; Ventriglia, F.; Gallo, P.; D’Amati, G.; Koch, W.C.; Adler, S.P. Human parvovirus b19 infection in infancy associated with acute and chronic lymphocytic myocarditis and high cytokine levels: Report of 3 cases and review. Clin. Inf. Dis. 2000, 31, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Haque, A.; Bhatti, S.; Siddiqui, F.J. Intravenous immune globulin for severe acute myocarditis in children. Indian Pediatr. 2009, 46, 810–811. [Google Scholar] [PubMed]
- English, R.F.; Janosky, J.E.; Ettedgui, J.A.; Webber, S.A. Outcomes for children with acute myocarditis. Cardiol. Young 2004, 14, 488–493. [Google Scholar] [CrossRef] [PubMed]
- Klugman, D.; Berger, J.T.; Sable, C.A.; He, J.; Khandelwal, S.G.; Slonim, A.D. Pediatric patients hospitalized with myocarditis: A multi-institutional analysis. Pediatr. Cardiol. 2010, 31, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Wallukat, G.; Muller, J.; Hetzer, R. Specific removal of β1-adrenergic autoantibodies from patients with idiopathic dilated cardiomyopathy. N. Engl. J. Med. 2002, 347, 1806. [Google Scholar] [CrossRef] [PubMed]
- Wallukat, G.; Reinke, P.; Dorffel, W.V.; Luther, H.P.; Bestvater, K.; Felix, S.B.; Baumann, G. Removal of autoantibodies in dilated cardiomyopathy by immunoadsorption. Int. J. Cardiol. 1996, 54, 191–195. [Google Scholar] [CrossRef]
- Jensen, L.D.; Marchant, D.J. Emerging pharmacologic targets and treatments for myocarditis. Pharmacol. Ther. 2016, 161, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Maisch, B.; Pankuweit, S. Current treatment options in (peri)myocarditis and inflammatory cardiomyopathy. Herz 2012, 37, 644–656. [Google Scholar] [CrossRef] [PubMed]
- Muller, J.; Wallukat, G.; Dandel, M.; Bieda, H.; Brandes, K.; Spiegelsberger, S.; Nissen, E.; Kunze, R.; Hetzer, R. Immunoglobulin adsorption in patients with idiopathic dilated cardiomyopathy. Circulation 2000, 101, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Burch, M. Heart failure in the young. Heart 2002, 88, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Jahns, R.; Boivin, V.; Hein, L.; Triebel, S.; Angermann, C.E.; Ertl, G.; Lohse, M.J. Direct evidence for a β1-adrenergic receptor-directed autoimmune attack as a cause of idiopathic dilated cardiomyopathy. J. Clin. Investig. 2004, 113, 1419–1429. [Google Scholar] [CrossRef] [PubMed]
- Jahns, R.; Boivin, V.; Siegmund, C.; Inselmann, G.; Lohse, M.J.; Boege, F. Autoantibodies activating human β1-adrenergic receptors are associated with reduced cardiac function in chronic heart failure. Circulation 1999, 99, 649–654. [Google Scholar] [CrossRef] [PubMed]
- Pinamonti, B.; Alberti, E.; Cigalotto, A.; Dreas, L.; Salvi, A.; Silvestri, F.; Camerini, F. Echocardiographic findings in myocarditis. Am. J. Cardiol. 1988, 62, 285–291. [Google Scholar] [CrossRef]
- Bruns, L.A.; Chrisant, M.K.; Lamour, J.M.; Shaddy, R.E.; Pahl, E.; Blume, E.D.; Hallowell, S.; Addonizio, L.J.; Canter, C.E. Carvedilol as therapy in pediatric heart failure: An initial multicenter experience. J. Pediatr. 2001, 138, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Burch, M. Immune suppressive treatment in paediatric myocarditis: Still awaiting the evidence. Heart 2004, 90, 1103–1104. [Google Scholar] [CrossRef] [PubMed]
- Levi, D.; Alejos, J. An approach to the treatment of pediatric myocarditis. Paediatr. Drugs 2002, 4, 637–647. [Google Scholar] [CrossRef] [PubMed]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.; Coats, A.J.; Falk, V.; Gonzalez-Juanatey, J.R.; Harjola, V.P.; Jankowska, E.A.; et al. 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: The task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC)developed with the special contribution of the heart failure association (HFA) of the ESC. Eur. Heart J. 2016, 37, 2129–2200. [Google Scholar] [PubMed]
- Chen, K.; Williams, S.; Chan, A.K.C.; Mondal, T.K. Thrombosis and embolism in pediatric cardiomyopathy. Blood Coagul. Fibrinolysis 2013, 24, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Vashist, S.; Singh, G.K. Acute myocarditis in children: Current concepts and management. Curr. Treat. Options Cardiovasc. Med. 2009, 11, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Alexander, P.M.; Daubeney, P.E.; Nugent, A.W.; Lee, K.J.; Turner, C.; Colan, S.D.; Robertson, T.; Davis, A.M.; Ramsay, J.; Justo, R.; et al. Long-term outcomes of dilated cardiomyopathy diagnosed during childhood: Results from a national population-based study of childhood cardiomyopathy. Circulation 2013, 128, 2039–2046. [Google Scholar] [CrossRef] [PubMed]
- Foerster, S.R.; Canter, C.E.; Cinar, A.; Sleeper, L.A.; Webber, S.A.; Pahl, E.; Kantor, P.F.; Alvarez, J.A.; Colan, S.D.; Jefferies, J.L.; et al. Ventricular remodeling and survival are more favorable for myocarditis than for idiopathic dilated cardiomyopathy in childhood: An outcomes study from the pediatric cardiomyopathy registry. Circ. Heart Fail. 2010, 3, 689–697. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farinha, I.T.; Miranda, J.O. Myocarditis in Paediatric Patients: Unveiling the Progression to Dilated Cardiomyopathy and Heart Failure. J. Cardiovasc. Dev. Dis. 2016, 3, 31. https://doi.org/10.3390/jcdd3040031
Farinha IT, Miranda JO. Myocarditis in Paediatric Patients: Unveiling the Progression to Dilated Cardiomyopathy and Heart Failure. Journal of Cardiovascular Development and Disease. 2016; 3(4):31. https://doi.org/10.3390/jcdd3040031
Chicago/Turabian StyleFarinha, Inês Teixeira, and Joana Oliveira Miranda. 2016. "Myocarditis in Paediatric Patients: Unveiling the Progression to Dilated Cardiomyopathy and Heart Failure" Journal of Cardiovascular Development and Disease 3, no. 4: 31. https://doi.org/10.3390/jcdd3040031
APA StyleFarinha, I. T., & Miranda, J. O. (2016). Myocarditis in Paediatric Patients: Unveiling the Progression to Dilated Cardiomyopathy and Heart Failure. Journal of Cardiovascular Development and Disease, 3(4), 31. https://doi.org/10.3390/jcdd3040031





