Nutritional Management in a 101-Year-Old Woman with Physical Inactivity and General Weakness: A Case Report
Abstract
:1. Introduction
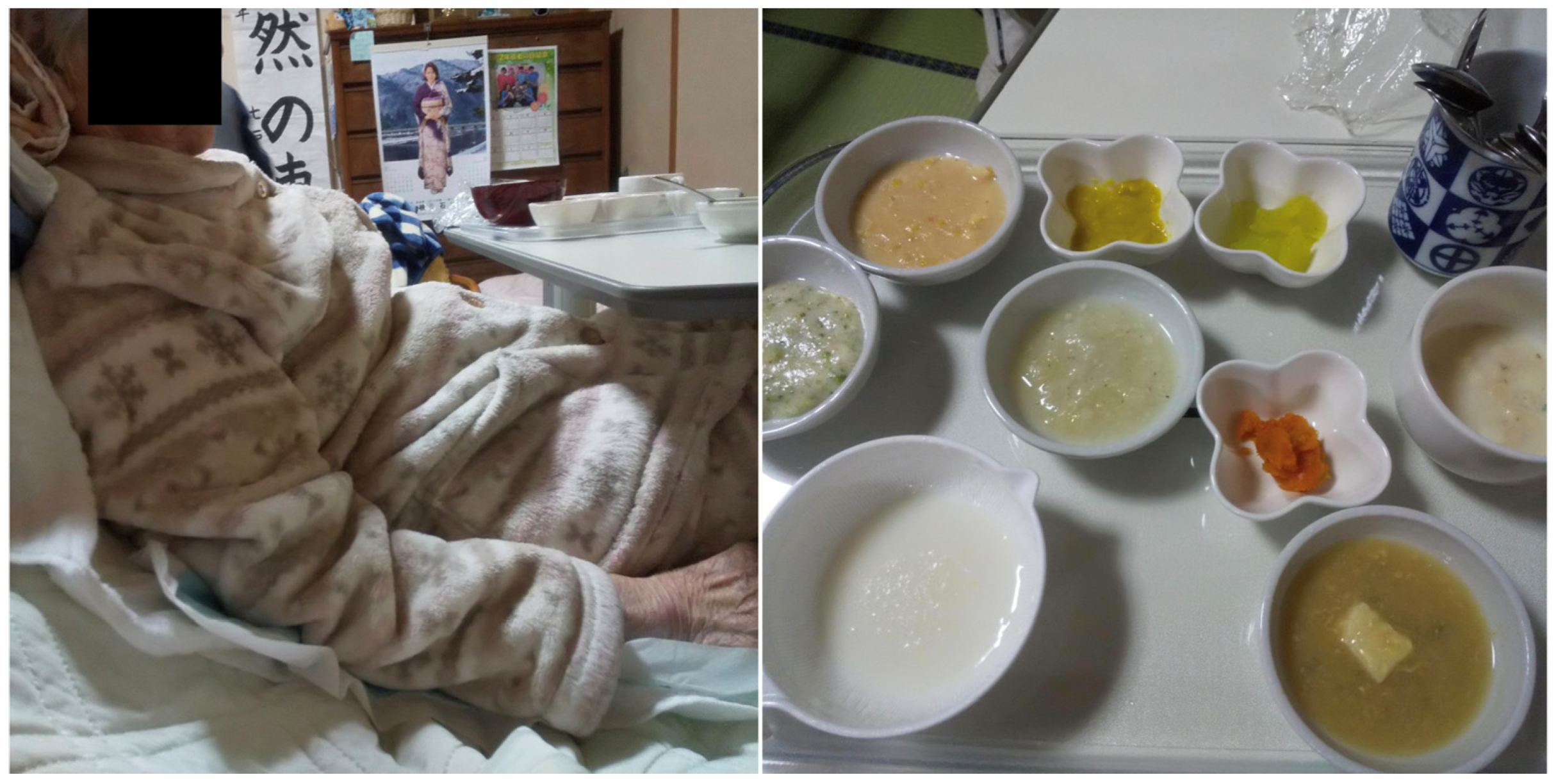
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vollset, S.E.; Goren, E.; Yuan, C.W.; Cao, J.; Smith, A.E.; Hsiao, T.; Bisignano, C.; Azhar, G.S.; Castro, E.; Chalek, J.; et al. Fertility, mortality, migration, and population scenarios for 195 countries and territories from 2017 to 2100: A forecast-ing analysis for the Global Burden of Disease Study. Lancet 2020, 396, 1285–1306. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Niti, M.; Yap, K.B.; Tan, C.T.Y.; Zin, N.M.S.; Feng, L.; Tan, B.Y.; Chan, G.; Khoo, S.A.; Chan, S.M.; et al. Assessment of Sarcopenia Among Community-Dwelling At-Risk Frail Adults Aged 65 Years and Older Who Received Multidomain Lifestyle Interventions: A Secondary Analysis of a Randomized Clinical Trial. JAMA Netw. Open 2019, 2, e1913346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandes, A.L.; Hayashi, A.P.; Jambassi-Filho, J.C.; de Capitani, M.D.; de Santana, D.A.; Gualano, B.; Roschel, H. Different protein and derivatives supplementation strategies combined with resistance training in pre-frail and frail elderly: Rationale and protocol for the “Pro-Elderly” Study. Nutr. Health 2017, 23, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Dedeyne, L.; Dupont, J.; Koppo, K.; Verschueren, S.; Tournoy, J.; Gielen, E. Exercise and Nutrition for Healthy AgeiNg (ENHANce) project—effects and mechanisms of action of combined anabolic interventions to improve physical functioning in sarcopenic older adults: Study protocol of a triple blinded, randomized controlled trial. BMC Geriatr. 2020, 20, 532. [Google Scholar] [CrossRef]
- da Silva Pereira, A.; Matos, A.; Valente, A.; Gil, Â.; Alonso, I.; Ribeiro, R.; Bicho, M.; Gorjão-Clara, J. Body Composition Assessment and Nutritional Status Evaluation in Men and Women Portuguese Centenarians. J. Nutr. Health Aging 2016, 20, 256–266. [Google Scholar] [CrossRef]
- Hernández-Vicente, A.; Marín-Puyalto, J.; Pueyo, E.; Vicente-Rodríguez, G.; Garatachea, N. Physical Activity in Centenarians beyond Cut-Point-Based Accelerometer Metrics. Int. J. Environ. Res. Public Health 2022, 19, 11384. [Google Scholar] [CrossRef]
- Okada, T.; Ikebe, K.; Kagawa, R.; Inomata, C.; Takeshita, H.; Gondo, Y.; Ishioka, Y.; Okubo, H.; Kamide, K.; Masui, Y.; et al. Lower Protein Intake Mediates Association Between Lower Occlusal Force and Slower Walking Speed: From the Septuagenarians, Octogenarians, Nonagenarians Investigation with Centenarians Study. J. Am. Geriatr. Soc. 2015, 63, 2382–2387. [Google Scholar] [CrossRef]
- Tai, P.; Yang, S.; Liu, W.; Wang, S.; Chen, K.; Jia, W.; Han, K.; Liu, M.; He, Y. Association of anthropometric and nutrition status indicators with cognitive functions in centenarians. Clin. Nutr. 2021, 40, 2252–2258. [Google Scholar] [CrossRef]
- Nakanishi, Y.; Tsugihashi, Y.; Akahane, M.; Noda, T.; Nishioka, Y.; Myojin, T.; Kubo, S.; Higashino, T.; Okuda, N.; Robine, J.M.; et al. Comparison of Japanese Centenarians’ and Noncentenarians’ Medical Expenditures in the Last Year of Life. JAMA Netw. Open 2021, 4, e2131884. [Google Scholar] [CrossRef]
- Caruso, C.; Passarino, G.; Puca, A.; Scapagnini, G. “Positive biology”: The centenarian lesson. Immun. Ageing 2012, 9, 5. [Google Scholar] [CrossRef]
- Scarpelli, M.C.; Bergamasco, J.G.A.; Arruda, E.A.B.; Cook, S.B.; Libardi, C.A. Resistance Training With Partial Blood Flow Restriction in a 99-Year-Old Individual: A Case Report. Front. Sports Act. Living 2021, 3, 671764. [Google Scholar] [CrossRef] [PubMed]
- Norman, K.; Haß, U.; Pirlich, M. Malnutrition in Older Adults-Recent Advances and Remaining Challenges. Nutrients 2021, 13, 2764. [Google Scholar] [CrossRef] [PubMed]
- Division of the Heath for the Elderly, Health and Welfare Bureau for the Elderly, Ministry of Health, Labour and Welfare. 2020. Available online: https://www.mhlw.go.jp/content/10900000/000818036.pdf (accessed on 14 December 2022).
- Javed, A.A.; Aljied, R.; Allison, D.J.; Anderson, L.N.; Ma, J.; Raina, P. Body mass index and all-cause mortality in older adults: A scoping review of observational studies. Obes. Rev. 2020, 21, e13035. [Google Scholar] [CrossRef] [PubMed]
- Mahoney, F.I.; Barthel, D.W. Functional Evaluation: The Barthel Index. Md. State Med. J. 1965, 14, 61–65. [Google Scholar] [PubMed]
- Osawa, A.; Maeshima, S.; Tanahashi, N. Water-swallowing test: Screening for aspiration in stroke patients. Cerebrovasc. Dis. 2013, 35, 276–2781. [Google Scholar] [CrossRef]
- Harris, J.A.; Benedict, F.G. A biometric study of human basal metabolism. Proc. Natl. Acad. Sci. USA 1918, 4, 370–373. [Google Scholar] [CrossRef] [Green Version]
- Kojima, C.; Fujishima, I.; Ohkuma, R.; Maeda, H.; Shibamoto, I.; Hojo, K.; Arai, M. Jaw opening and swallow triggering method for bilateral-brain-damaged patients: K-point stimulation. Dysphagia 2002, 17, 273–277. [Google Scholar] [CrossRef]
- Barrado-Martín, Y.; Nair, P.; Anantapong, K.; Aker, N.; Moore, K.J.; Smith, C.H.; Rait, G.; Sampson, E.L.; Manthorpe, J.; Davies, N. Family caregivers’ and professionals’ experiences of supporting people living with dementia’s nutrition and hydration needs towards the end of life. Health Soc. Care Community 2022, 30, 307–318. [Google Scholar] [CrossRef]
- Hayashi, A.P.; de Capitani, M.D.; Dias, S.F.; de Souza Gonçalves, L.; Fernandes, A.L.; Jambassi-Filho, J.C.; de Santana, D.A.; Lixandrão, M.; Tavares Dos Santos Pereira R Riani, L.; Hevia-Larraín, V.; et al. Number of high-protein containing meals correlates with muscle mass in pre-frail and frail elderly. Eur. J. Clin. Nutr. 2020, 74, 1047–1053. [Google Scholar] [CrossRef]
- Moore, D.R.; Churchward-Venne, T.A.; Witard, O.; Breen, L.; Burd, N.A.; Tipton, K.D.; Phillips, S.M. Protein ingestion to stimulate myofibrillar protein synthesis requires greater relative protein intakes in healthy older versus younger men. J. Gerontol. A Biol. Sci. Med. Sci. 2015, 70, 57–62. [Google Scholar] [CrossRef]
- Mertz, K.H.; Reitelseder, S.; Bechshoeft, R.; Bulow, J.; Højfeldt, G.; Jensen, M.; Schacht, S.R.; Lind, M.V.; Rasmussen, M.A.; Mikkelsen, U.R.; et al. The effect of daily protein supplementation, with or without resistance training for 1 year, on muscle size, strength, and function in healthy older adults: A randomized controlled trial. Am. J. Clin. Nutr. 2021, 113, 790–800. [Google Scholar] [CrossRef] [PubMed]
- Roschel, H.; Hayashi, A.P.; Fernandes, A.L.; Jambassi-Filho, J.C.; Hevia-Larraín, V.; de Capitani, M.; Santana, D.A.; Gonçalves, L.S.; de Sá-Pinto, A.L.; Lima, F.R.; et al. Supplement-based nutritional strategies to tackle frailty: A multifactorial, double-blind, randomized placebo-controlled trial. Clin. Nutr. 2021, 40, 4849–4858. [Google Scholar] [CrossRef] [PubMed]
- O’Bryan, K.R.; Doering, T.M.; Morton, R.W.; Coffey, V.G.; Phillips, S.M.; Cox, G.R. Do multi-ingredient protein supplements augment resistance training-induced gains in skeletal muscle mass and strength? A systematic review and meta-analysis of 35 trials. Br. J. Sports Med. 2020, 54, 573–581. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoshihara, A.; Seida, Y.; Kataoka, S.; Hanada, N.; Miyazaki, H. The Relationship between Appetite and Quality of Life in Community—dwelling Older Adults. Dent. Health 2004, 54, 241–248. (In Japanese) [Google Scholar]
- Munoz, N.; Posthauer, M.E. Nutrition strategies for pressure injury management: Implementing the 2019 International Clinical Practice Guideline. Nutr. Clin. Pract. 2022, 37, 567–582. [Google Scholar] [CrossRef] [PubMed]
- Fukami, S.; Shumiya, T.; Yamada, C.; Hasegawa, K.; Sugiyama, K.; Ozaki, T. Improvement of meals for patients with dysphagia. J. Rural. Med. 2008, 57, 83–88. (In Japanese) [Google Scholar] [CrossRef]
- Tanoue, H.; Ohta, K.; Kokubo, A.; Minatani, S.; Kanada, Y. The Influence of a Change of Posture on Swal-lowing Function—Change of Swallowing Function with Posture of Neck, Trunk, and Lower Limbs. Jpn. J. Dysphagia Rehabil. 2008, 12, 207–213. (In Japanese) [Google Scholar]

| Item | Value | Normal Range | Item | Value | Normal Range |
|---|---|---|---|---|---|
| White blood cell | 64 | 35~91 (102/mL) | Creatinine | 0.64 | 0.45~0.82 (mg/dL) |
| Red blood cell | 358 | 376~500 (104/mL) | Blood urea nitrogen (BUN) | 24.4 | 8.0~23.0 (mg/dL) |
| Hemoglobin | 11.8 | 11.3~15.2 (g/dL) | Blood glucose | 130 | 70~109 (mg/dL) |
| Hematocrit | 37.2 | 33.4~44.9 (%) | Hemoglobin A1c | 5.3 | 4.6~6.2 (%) |
| Platelet | 16.3 | 13.0~36.9 (104/mL) | TG | 214 | 35~149 (mg/dL) |
| Total protein | 6.1 | 6.7~8.3 (g/dL) | Total cholesterol | 227 | 120~219 (mg/dL) |
| Albumin | 3.6 | 3.8~5.3 (g/dL) | LDL cholesterol | 143 | 70~139 (mg/dL) |
| Aspartate aminotransferase (AST) | 30 | 10~40 (U/L) | Natrium | 143 | 134~147 (mEq/L) |
| Alanine aminotransferase (ALT) | 24 | 5~45 (U/L) | Potassium | 3.8 | 3.4~5.0 (mEq/L) |
| Lactase dehydrogenase (LDH) | 154 | 120~240 (U/L) | Chloride | 105 | 98~108 (mEq/L) |
| Alkaline phosphatase gene (ALP) | 325 | 104~338 (U/L) | Total bilirubin | 0.39 | 0.20~1.10 (mg/dL) |
| γ-glutamyl transpeptidase (γ-GTP) | 16 | 0~35 (U/L) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mineyama, R.; Tezuka, F.; Takagi, N.; Kokabu, S.; Okubo, M. Nutritional Management in a 101-Year-Old Woman with Physical Inactivity and General Weakness: A Case Report. Geriatrics 2023, 8, 8. https://doi.org/10.3390/geriatrics8010008
Mineyama R, Tezuka F, Takagi N, Kokabu S, Okubo M. Nutritional Management in a 101-Year-Old Woman with Physical Inactivity and General Weakness: A Case Report. Geriatrics. 2023; 8(1):8. https://doi.org/10.3390/geriatrics8010008
Chicago/Turabian StyleMineyama, Ryoko, Fumie Tezuka, Nobuko Takagi, Shoichiro Kokabu, and Masahiko Okubo. 2023. "Nutritional Management in a 101-Year-Old Woman with Physical Inactivity and General Weakness: A Case Report" Geriatrics 8, no. 1: 8. https://doi.org/10.3390/geriatrics8010008
APA StyleMineyama, R., Tezuka, F., Takagi, N., Kokabu, S., & Okubo, M. (2023). Nutritional Management in a 101-Year-Old Woman with Physical Inactivity and General Weakness: A Case Report. Geriatrics, 8(1), 8. https://doi.org/10.3390/geriatrics8010008






