Gene Cloning, Tissue Expression Profiles and Antiviral Activities of Interferon-β from Two Chinese Miniature Pig Breeds
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cells and Viruses
2.2. Animal and Ethical Approval
2.3. Tissue Sampling
2.4. Gene Cloning and Sequence Analysis
2.5. Sequence Analysis of PoIFN-β Regulatory Elements
2.6. Real-Time Quantitative RT-PCR
2.7. Expression and Purification of rPoIFN-β
2.8. Antiviral Assay
2.9. Statistical Analysis
3. Results
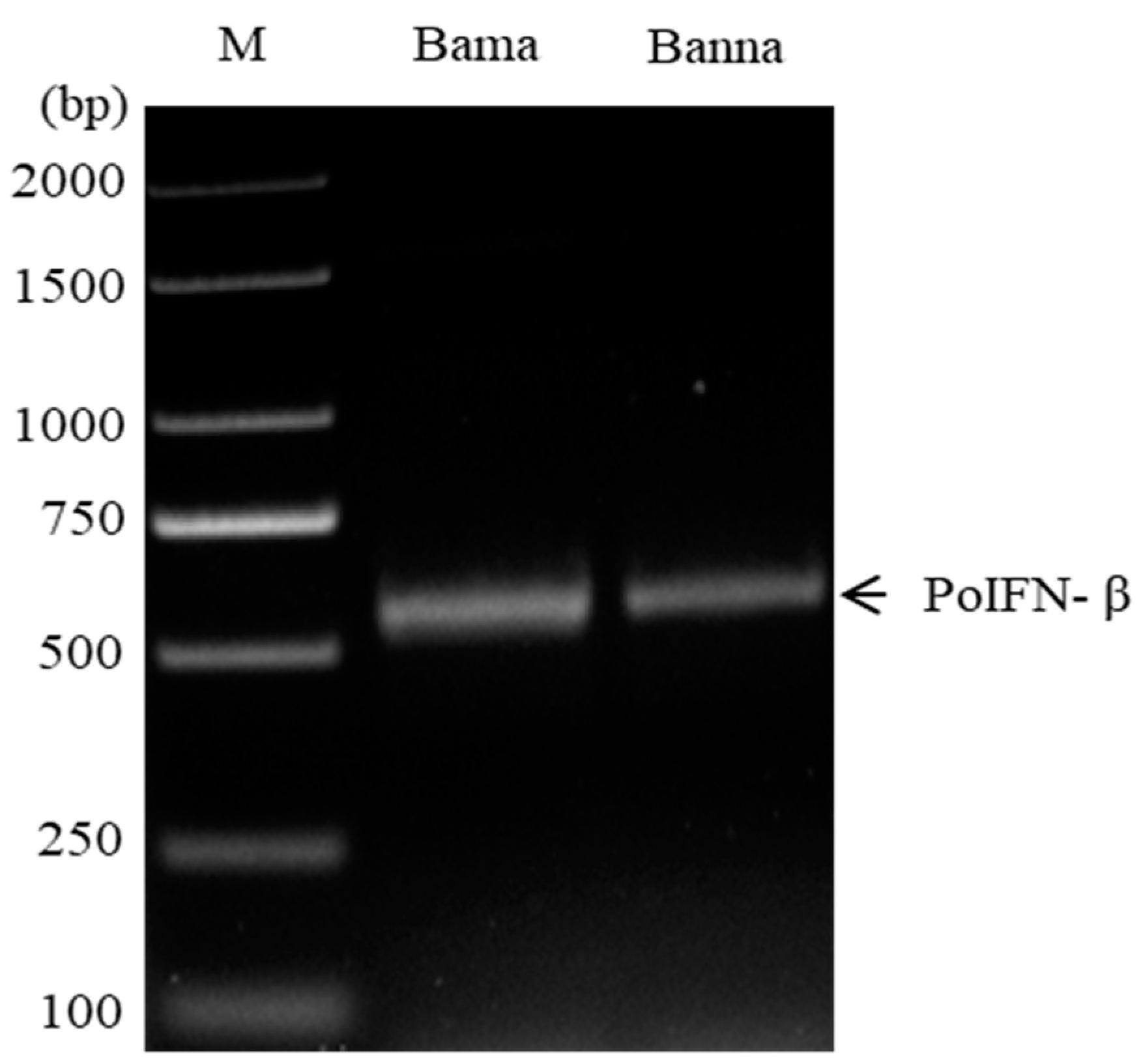
3.1. Cloning and Sequence Analysis of PoIFN-β Genes
3.2. Sequence Analysis of PoIFN-β Regulatory Elements
3.3. Detection of PoIFN-β Tissue Expression Profiles
3.4. Expression and Identification rPoIFN-β
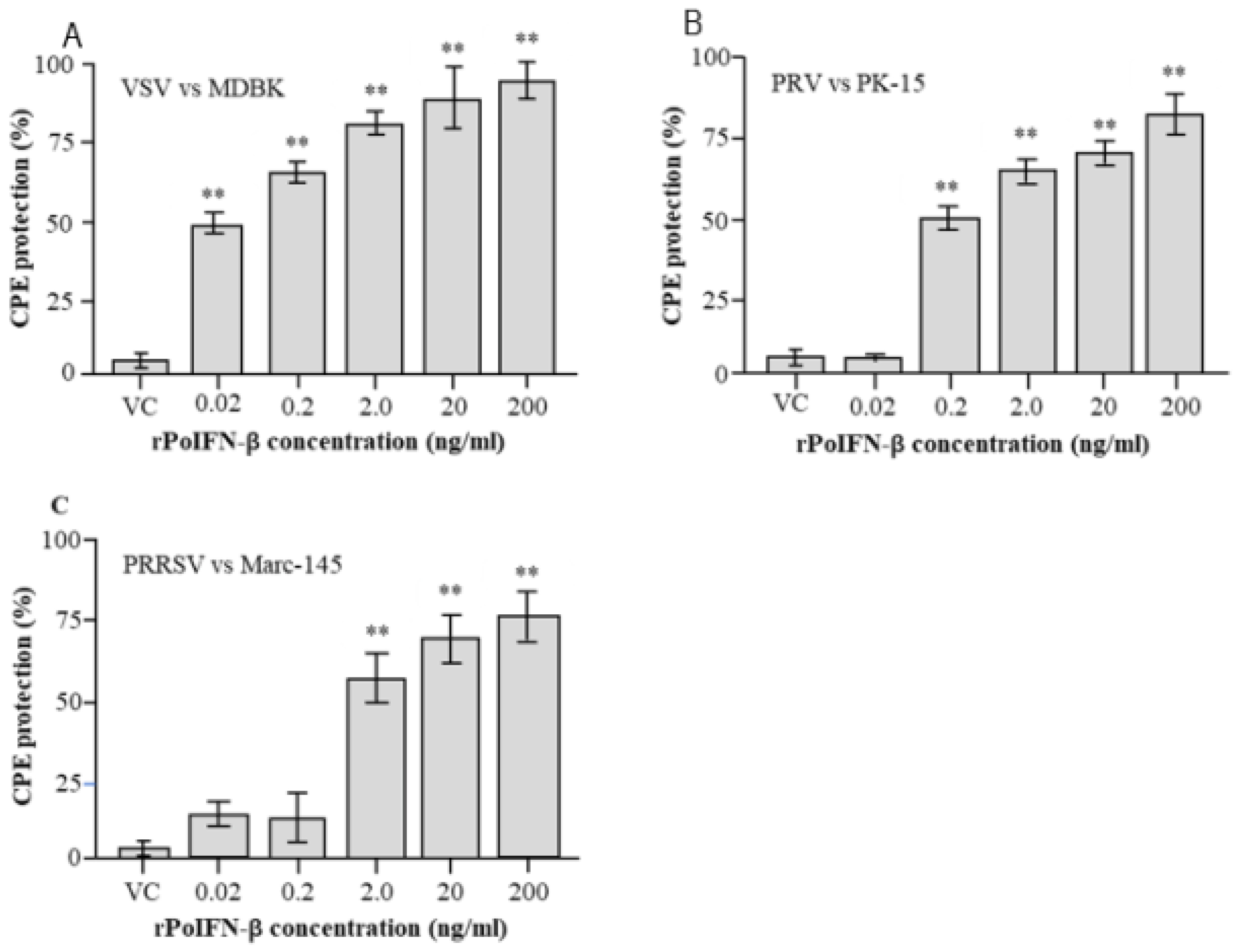
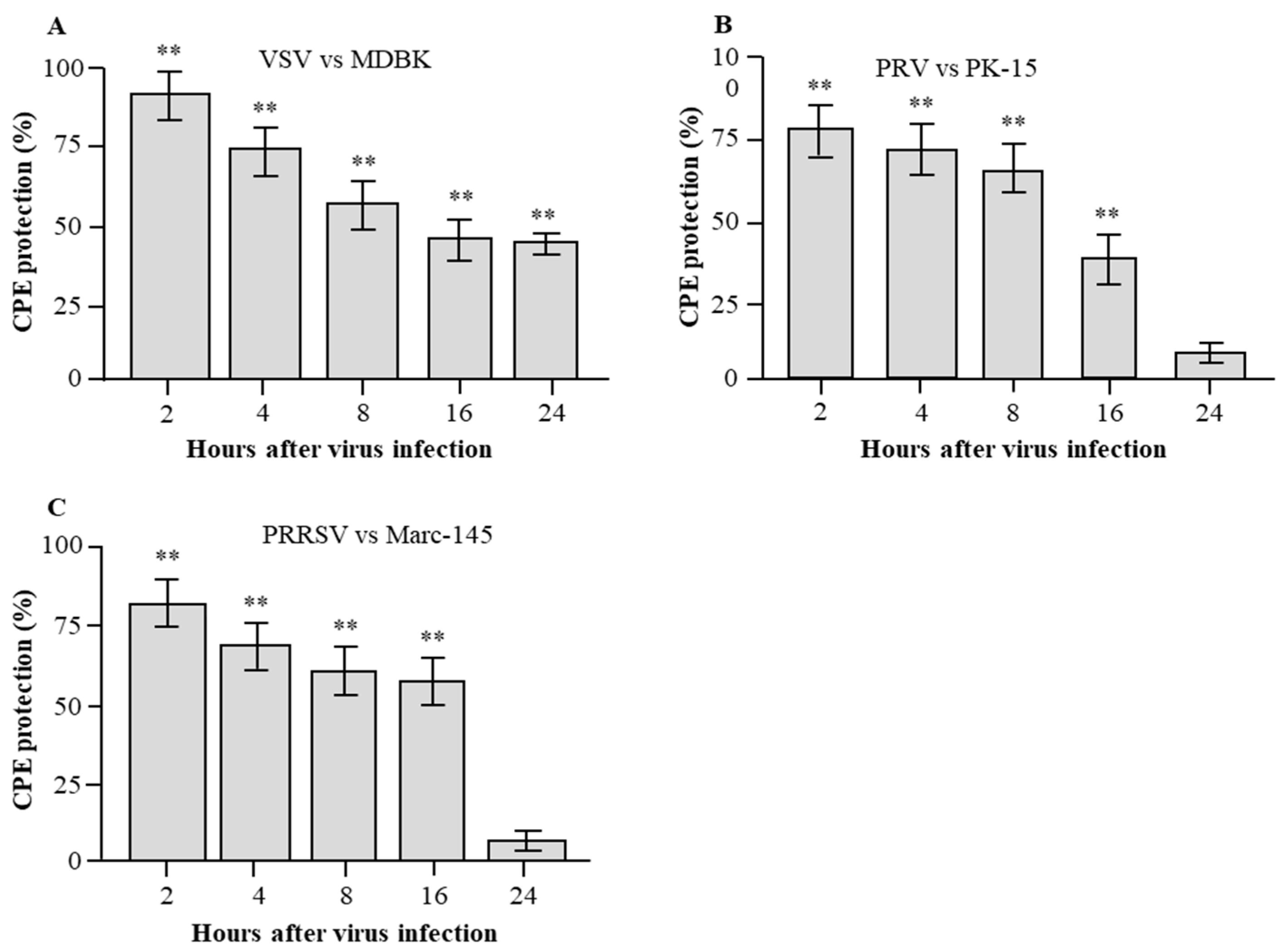
3.5. Detection of Antiviral Activities of rPoIFN-β
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jennings, J.; Sang, Y. Porcine interferon complex and co-evolution with increasing viral pressure after domestication. Viruses 2019, 11, 555. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sang, Y.; Rowland, R.R.; Hesse, R.A.; Blecha, F. Differential expression and activity of the porcine type I interferon family. Physiol. Genom. 2010, 42, 248–258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morra, M.E.; Van Thanh, L.; Kamel, M.G.; Ghazy, A.A.; Altibi, A.M.; Dat, L.M.; Thy, T.N.X.; Vuong, N.L.; Mostafa, M.R.; Ahmed, S.I. Clinical outcomes of current medical approaches for Middle East respiratory syndrome: A systematic review and meta-analysis. Rev. Med. Virol. 2018, 28, e1977. [Google Scholar] [CrossRef] [PubMed]
- Abdolvahab, M.H.; Mofrad, M.; Schellekens, H. Interferon beta: From molecular level to therapeutic effects. Int. Rev. Cell Mol. Biol. 2016, 326, 343–372. [Google Scholar]
- Schneider, W.M.; Chevillotte, M.D.; Rice, C.M. Interferon-stimulated genes: A complex web of host defenses. Annu. Rev. Immuno. 2014, 32, 513–545. [Google Scholar] [CrossRef] [Green Version]
- Jalkanen, J.; Pettilä, V.; Huttunen, T.; Hollmén, M.; Jalkanen, S. Glucocorticoids inhibit type I IFN beta signaling and the upregulation of CD73 in human lung. Intensive Care Med. 2020, 46, 1937–1940. [Google Scholar] [CrossRef]
- Fu, R.; Fang, M.; Xu, K.; Ren, J.; Zou, J.; Su, L.; Chen, X.; An, P.; Yu, D.; Ka, M. Generation of GGTA1−/− β2M−/− CIITA−/− Pigs Using CRISPR/Cas9 Technology to Alleviate Xenogeneic Immune Reactions. Transplantation 2020, 104, 1566–1573. [Google Scholar] [CrossRef]
- Wang, P.; Zhang, X.; Huo, J.; Huo, H.; Wang, S.; Pan, W.; Teng, X.; Cheng, X. Cloning, sequence analysis and expression of GOT1 gene in Banna mini-pig inbred line. J. Yunnan Univ. Nat. Sci. Ed. 2019, 34, 29–36. [Google Scholar]
- Ren, J.; Yu, D.; Fu, R.; An, P.; Sun, R.; Wang, Z.; Guo, R.; Li, H.; Zhang, Y.; Li, Z. IL2RG-deficient minipigs generated via CRISPR/Cas9 technology support the growth of human melanoma-derived tumours. Cell Prolif. 2020, 53, e12863. [Google Scholar] [CrossRef]
- Yu, P.; Zhang, L.; Li, S.; Li, Y.; Cheng, J.; Lu, Y.; Zeng, Y.; Bu, H. Screening and analysis of porcine endogenous retrovirus in Chinese Banna minipig inbred line. Transplant. Proc. 2004, 36, 2485–2487. [Google Scholar] [CrossRef]
- Zeng, R.; Zeng, Y.Z. Molecular cloning and characterization of SLA-DR genes in the 133-family of the Banna mini-pig inbred line. Anim. Genet. 2005, 36, 267–269. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Jiang, Q.; Tian, D.Y.; Lin, H.; Li, H.; Han, Q.Y.; Han, W.; Si, C.D.; Hu, S.P.; Zhang, Z. Experimental infection of Bama miniature pigs with a highly virulent classical swine fever virus. Virol. J. 2011, 8, 452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, W.-J.; Su, W.-W.; Li, P.-B.; Rao, H.-Y.; Lin, Q.-W.; Zeng, X.; Chen, T.-B.; Yan, Z.-H.; Liu, H.; Yao, H.-L. Naoxintong Capsule inhibits the development of cardiovascular pathological changes in Bama Minipig through improving gut microbiota. Front. Pharmacol. 2019, 10, 1128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Groenen, M.A.; Archibald, A.L.; Uenishi, H.; Tuggle, C.K.; Takeuchi, Y.; Rothschild, M.F.; Rogel-Gaillard, C.; Park, C.; Milan, D.; Megens, H.J. Analyses of pig genomes provide insight into porcine demography and evolution. Nature 2012, 491, 393–398. [Google Scholar] [CrossRef]
- Noor, A.U.; Du, Z.; Lu, H.; Zhou, X.; Liu, X.; Xiao, J.; Zhang, X.; Sun, H. Gene cloning, prokaryotic expression and antiviral activities of interferon-αω from Chinese Bama miniature pigs. Vet. Res. Commun. 2022, 46, 59–66. [Google Scholar] [CrossRef]
- Liu, H.B.; Lv, P.R.; He, R.G.; Yang, X.G.; Qin, X.E.; Pan, T.B.; Huang, G.Y.; Huang, M.R.; Lu, Y.Q.; Lu, S.S. Cloned Guangxi Bama minipig (Sus scrofa) and its offspring have normal reproductive performance. Cloning Stem Cells 2010, 12, 543–550. [Google Scholar] [CrossRef]
- Zhang, L.; Huang, Y.; Si, J.; Wu, Y.; Wang, M.; Jiang, Q.; Guo, Y.; Liang, J.; Lan, G. Comprehensive inbred variation discovery in Bama pigs using de novo assemblies. Gene 2018, 679, 81–89. [Google Scholar] [CrossRef]
- Liu, X.; Xia, W.; Zhang, X.; Xia, X.; Sun, H. Fusion expression of the two soluble viral receptors of porcine reproductive and respiratory syndrome virus with a single adeno-associated virus vector. Res. Vet. 2021, 135, 78–84. [Google Scholar] [CrossRef]
- Abdelmageed, A.A.; Ferran, M.C. The Propagation, Quantification, and Storage of Vesicular Stomatitis Virus. Curr. Protoc. Microbiol. 2020, 58, e110. [Google Scholar] [CrossRef]
- Babic, N.; Klupp, B.; Brack, A.; Mettenleiter, T.C.; Ugolini, G.; Flamand, A. Deletion of glycoprotein gE reduces the propagation of pseudorabies virus in the nervous system of mice after intranasal inoculation. Virology 1996, 219, 279–284. [Google Scholar] [CrossRef] [Green Version]
- Jiang, K.; Chen, J.; Liu, Y.; Liu, J.; Liu, A.; Dong, J.; Huang, Z. Heat-irrigate effect’of radiofrequency ablation on relevant regional hepatocyte in living swine liver-initial study on pathology. Cell Biochem. Biophys. 2015, 72, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.Y.; Gong, T.F.; He, J.L.; Li, F.; Li, W.C.; Xie, L.X.; Xie, X.R.; Liu, Y.S.; Zhou, Y.F.; Liu, W. Molecular Characterization and Phylogenetic Analysis of Spirometra Tapeworms from Snakes in Hunan Province. J. Vet. Sci. 2022, 9, 62. [Google Scholar] [CrossRef] [PubMed]
- Korbie, D.J.; Mattick, J.S. Touchdown PCR for increased specificity and sensitivity in PCR amplification. Nat. Protoc. 2008, 3, 1452–1456. [Google Scholar] [CrossRef] [PubMed]
- Sang, Y.; Patil, A.A.; Zhang, G.; Ross, C.R.; Blecha, F. Bioinformatic and expression analysis of novel porcine β-defensins. Mamm. Genome. 2006, 17, 332–339. [Google Scholar] [CrossRef]
- Artursson, K.; Gobl, A.; Lindersson, M.; Johansson, M.; Alm, G. Molecular cloning of a gene encoding porcine interferon-β. J. Interferon Cytokine Res. 1992, 12, 153–160. [Google Scholar] [CrossRef]
- Cheng, G.; Chen, W.; Li, Z.; Yan, W.; Zhao, X.; Xie, J.; Liu, M.; Zhang, H.; Zhong, Y.; Zheng, Z. Characterization of the porcine alpha interferon multigene family. Gene 2006, 382, 28–38. [Google Scholar] [CrossRef]
- Lu, H.; Zhang, X.; Wang, Y.; Zong, Y.; Wang, Y.; Zhang, X.; Xia, X.; Sun, H. Superior adjuvanticity of the genetically fused D1 domain of Neisseria meningitides Ag473 lipoprotein among three Toll-like receptor ligands. Biosci. Rep. 2020, 40, BSR20193675. [Google Scholar] [CrossRef] [Green Version]
- Li, S.F.; Gong, M.J.; Xie, Y.L.; Shao, J.J.; Zhao, F.R.; Zhang, Y.G.; Chang, H.Y. A novel type I interferon, interferon alphaomega, shows antiviral activity against foot-and-mouth disease virus in vitro. Microb. Pathog. 2019, 127, 79–84. [Google Scholar] [CrossRef]
- Usharani, J.; Park, S.Y.; Cho, E.J.; Kim, C.; Ko, Y.J.; Tark, D.; Kim, S.M.; Park, J.H.; Lee, K.N.; Lee, M.H. Antiviral activity of ovine interferon tau 4 against foot-and-mouth disease virus. Antivir. Res. 2017, 143, 134–141. [Google Scholar] [CrossRef]
- Javed, A.; Reder, A.T. Therapeutic role of beta-interferons in multiple sclerosis. Pharmacol. Ther. 2006, 110, 35–56. [Google Scholar] [CrossRef] [PubMed]



| Gene | Primer (5′-3′) | Amplicon | Reference | |
|---|---|---|---|---|
| PoIFN-β a | Sense | ATGGCTAACAAGTGCATCC | 561 | This study |
| Anti-sense | TCAGTTCCGGAGGTAATCT | |||
| PoIFN-β b | Sense | AAATCGCTCTCCTGATGTGTT | 539 | This study |
| Anti-sense | TCAGTGGTGGTGGTGGTGGTGGTTCCGGAGGTAATCTGTAA | |||
| PoIFN-β c | Sense | ATGTCAGAAGCTCCTGGGACAGTT | 246 | Sang et al. |
| Anti-sense | AGGTCATCCATCTGCCCATCAAGT | |||
| PoIFN-β R | Sense | AGTGTTGGATGAATGCTAACAA | 259 | This study |
| Anti-sense | TGGTGGAGAAACACATCAGG | |||
| GAPDH | Sense | TGGYATCGTGGAAGGRCTCAT | 370 | Sang et al. |
| Anti-sense | RTGGGWGTYGCTGTTGAAGTC | |||
| Species | Homology (Clustal W Score) | ||
|---|---|---|---|
| ORF% | Protein% | GenBank Accession No | |
| Bama minipig | Reference | Reference | OL446997 |
| Banna minipig | 100 | 100 | OL446998 |
| Congjiang Xiang minipig | 100 | 100 | MH538100 |
| Guizhou Baixiang minipig | 99.0 | 98.4 | JF906509 |
| African minipig | 99.0 | 99.5 | JN391525 |
| Domestic pig | 100 | 99.5 | GQ415073 |
| 99.8 | 100 | NM_0010039231 | |
| 98.8 | 97.9 | KF4147411 | |
| 99.8 | 99.4 | EU744562 | |
| 99.8 | 99.5 | M86762 | |
| Phacochoerus africanus | 99.0 | 99.5 | JN391529 |
| Potamochoerus porcus | 99.0 | 99.5 | JN391527 |
| Potamochoerus larvatus | 99.0 | 99.5 | JN391526 |
| Minipig | Skin | Uterus | Heart | Lung | Intestine | Liver | Spleen | Lymph Node | Kidney | Brain |
|---|---|---|---|---|---|---|---|---|---|---|
| Banna | - | 0.375 | - | 1.142 | 0.488 | 1.784 | 0.551 | 0.474 | - | - |
| Bama | - | 0.044 | 0.204 | 1.398 | 0.465 | 0.329 | 0.135 | 0.008 | 0.239 | 0.041 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noor, A.U.; Du, Z.; Song, C.; Lu, H.; Zhou, X.; Liu, X.; Zhang, X.; Sun, H. Gene Cloning, Tissue Expression Profiles and Antiviral Activities of Interferon-β from Two Chinese Miniature Pig Breeds. Vet. Sci. 2022, 9, 190. https://doi.org/10.3390/vetsci9040190
Noor AU, Du Z, Song C, Lu H, Zhou X, Liu X, Zhang X, Sun H. Gene Cloning, Tissue Expression Profiles and Antiviral Activities of Interferon-β from Two Chinese Miniature Pig Breeds. Veterinary Sciences. 2022; 9(4):190. https://doi.org/10.3390/vetsci9040190
Chicago/Turabian StyleNoor, Aziz Ullah, Zhanyu Du, Chengyi Song, Huipeng Lu, Xiaohui Zhou, Xiaoming Liu, Xinyu Zhang, and Huaichang Sun. 2022. "Gene Cloning, Tissue Expression Profiles and Antiviral Activities of Interferon-β from Two Chinese Miniature Pig Breeds" Veterinary Sciences 9, no. 4: 190. https://doi.org/10.3390/vetsci9040190
APA StyleNoor, A. U., Du, Z., Song, C., Lu, H., Zhou, X., Liu, X., Zhang, X., & Sun, H. (2022). Gene Cloning, Tissue Expression Profiles and Antiviral Activities of Interferon-β from Two Chinese Miniature Pig Breeds. Veterinary Sciences, 9(4), 190. https://doi.org/10.3390/vetsci9040190







