European Mistletoe (Viscum album) Extract Is Cytotoxic to Canine High-Grade Astrocytoma Cells In Vitro and Has Additive Effects with Mebendazole
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Viscum album and Mebendazole Preparations
2.3. MTT Cell Viability Assays
2.4. Statistical Analysis
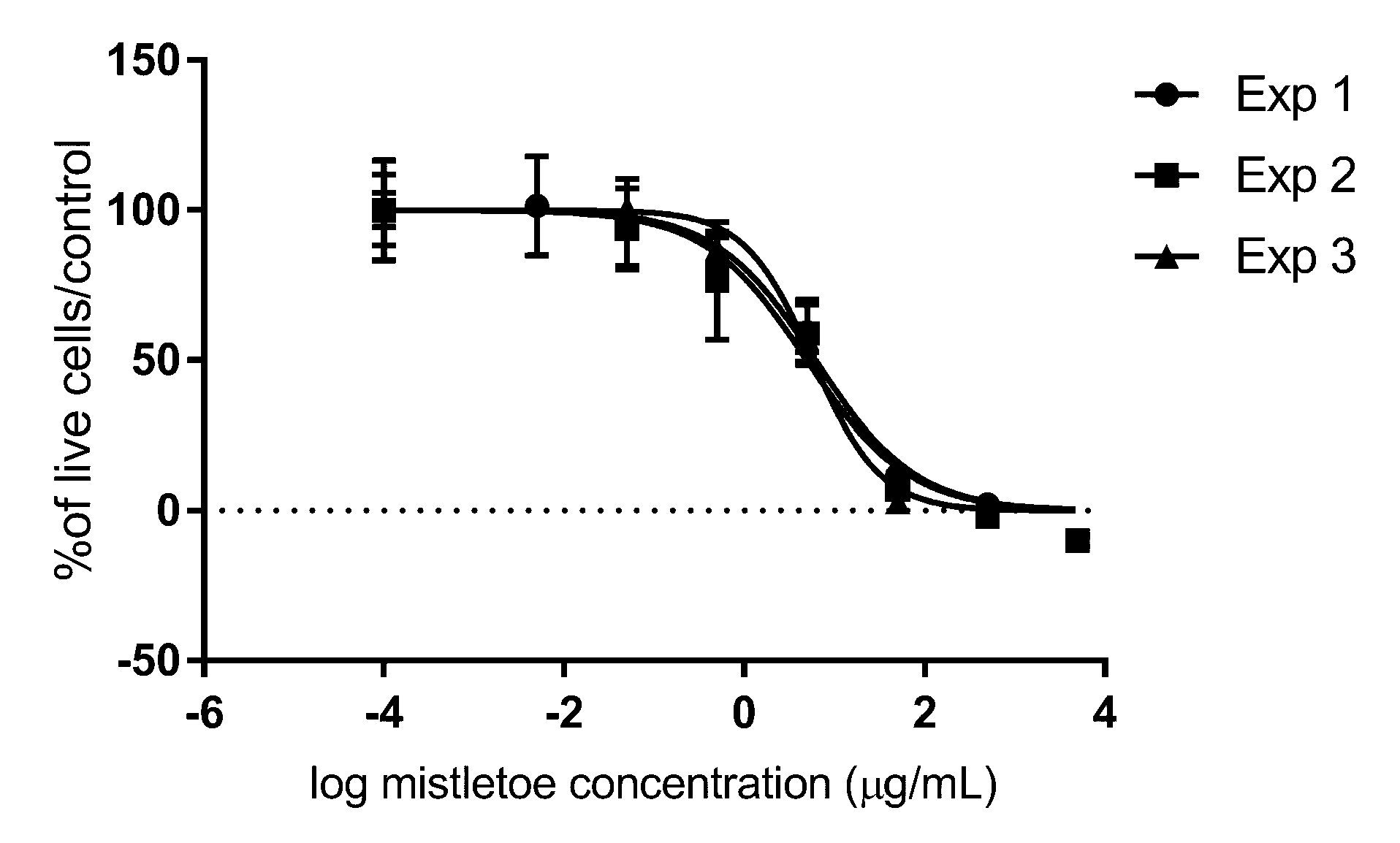
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ostrom, Q.T.; Gittleman, H.; Xu, J.; Kromer, C.; Wolinsky, Y.; Kruchko, C.; Barnholtz-Sloan, J.S. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2009–2013. Neuro-Oncology 2016, 18 (Suppl. 5), v1–v75. [Google Scholar] [CrossRef] [Green Version]
- Van Meir, E.G.; Hadjipanayis, C.G.; Norden, A.D.; Shu, H.K.; Wen, P.Y.; Olson, J.J. Exciting new advances in neuro-oncology: The avenue to a cure for malignant glioma. CA A Cancer J. Clin. 2010, 60, 166–193. [Google Scholar] [CrossRef]
- Boudreau, C.E.; York, D.; Higgins, R.J.; LeCouteur, R.A.; Dickinson, P.J. Molecular signalling pathways in canine gliomas. Vet. Comp. Oncol. 2017, 15, 133–150. [Google Scholar] [CrossRef] [Green Version]
- Heidner, G.L.; Kornegay, J.N.; Page, R.L.; Dodge, R.K.; Thrall, D.E. Analysis of survival in a retrospective study of 86 dogs with brain tumors. J. Vet. Intern. Med. 1991, 5, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Biegel, U.; Stratmann, N.; Knauf, Y.; Ruess, K.; Reif, M.; Wehrend, A. Postoperative adjuvante Therapie mit einem Mistelextrakt (Viscum album ssp. album) bei Hundinnen mit Mammatumoren. Complement. Med. Res. 2017, 24, 349–357. [Google Scholar] [CrossRef] [Green Version]
- von Bodungen, U.; Ruess, K.; Reif, M.; Biegel, U. Kombinierte Anwendung von Strahlentherapie und adjuvanter Therapie mit einem Mistelextrakt (Viscum album L.) zur Behandlung des oralen malignen Melanoms beim Hund: Eine retrospektive Studie. Complement. Med. Res. 2017, 24, 358–363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heddleston, J.M.; Li, Z.; McLendon, R.E.; Hjelmeland, A.B.; Rich, J.N. The hypoxic microenvironment maintains glioblastoma stem cells and promotes reprogramming towards a cancer stem cell phenotype. Cell Cycle 2009, 8, 3274–3284. [Google Scholar] [CrossRef] [Green Version]
- Peng, W.X.; Pan, F.Y.; Liu, X.J.; Ning, S.; Xu, N.; Meng, F.L.; Wang, Y.Q.; Li, C.J. Hypoxia stabilizes microtubule networks and decreases tumor cell chemosensitivity to anticancer drugs through Egr-1. Anat. Rec. 2010, 293, 414–420. [Google Scholar] [CrossRef]
- Drapeau, A.; Fortin, D. Chemotherapy Delivery Strategies to the Central Nervous System: Neither Optional nor Superfluous. Curr. Cancer Drug Targets 2015, 15, 752–768. [Google Scholar] [CrossRef]
- Hicks, J.; Platt, S.; Kent, M.; Haley, A. Canine brain tumours: A model for the human disease? Vet. Comp. Oncol. 2017, 15, 252–272. [Google Scholar] [CrossRef] [PubMed]
- Stoica, G.; Levine, J.; Wolff, J.; Murphy, K. Canine astrocytic tumors: A comparative review. Vet. Pathol. 2011, 48, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Paoloni, M.; Khanna, C. Translation of new cancer treatments from pet dogs to humans. Nat. Rev. Cancer 2008, 8, 147–156. [Google Scholar] [CrossRef]
- Schiffman, J.D.; Breen, M. Comparative oncology: What dogs and other species can teach us about humans with cancer. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370. [Google Scholar] [CrossRef] [PubMed]
- Bentley, R.T.; Ahmed, A.U.; Yanke, A.B.; Cohen-Gadol, A.A.; Dey, M. Dogs are man’s best friend: In sickness and in health. Neuro Oncol. 2017, 19, 312–322. [Google Scholar] [CrossRef] [Green Version]
- LeBlanc, A.K.; Mazcko, C.; Brown, D.E.; Koehler, J.W.; Miller, A.D.; Miller, C.R.; Bentley, R.T.; Packer, R.A.; Breen, M.; Boudreau, C.E.; et al. Creation of an NCI comparative brain tumor consortium: Informing the translation of new knowledge from canine to human brain tumor patients. Neuro Oncol. 2016, 18, 1209–1218. [Google Scholar] [CrossRef]
- Koehler, J.W.; Miller, A.D.; Miller, C.R.; Porter, B.; Aldape, K.; Beck, J.; Brat, D.; Cornax, I.; Corps, K.; Frank, C.; et al. A Revised Diagnostic Classification of Canine Glioma: Towards Validation of the Canine Glioma Patient as a Naturally Occurring Preclinical Model for Human Glioma. J. Neuropathol. Exp. Neurol. 2018, 77, 1039–1054. [Google Scholar] [CrossRef] [PubMed]
- Krex, D.; Klink, B.; Hartmann, C.; von Deimling, A.; Pietsch, T.; Simon, M.; Sabel, M.; Steinbach, J.P.; Heese, O.; Reifenberger, G.; et al. Long-term survival with glioblastoma multiforme. Brain 2007, 130, 2596–2606. [Google Scholar] [CrossRef]
- Hegi, M.E.; Liu, L.; Herman, J.G.; Stupp, R.; Wick, W.; Weller, M.; Mehta, M.P.; Gilbert, M.R. Correlation of O6-methylguanine methyltransferase (MGMT) promoter methylation with clinical outcomes in glioblastoma and clinical strategies to modulate MGMT activity. J. Clin. Oncol. 2008, 26, 4189–4199. [Google Scholar] [CrossRef] [Green Version]
- Zhang, K.; Wang, X.Q.; Zhou, B.; Zhang, L. The prognostic value of MGMT promoter methylation in Glioblastoma multiforme: A meta-analysis. Fam Cancer 2013, 12, 449–458. [Google Scholar] [CrossRef]
- Zhang, J.; Stevens, M.F.; Bradshaw, T.D. Temozolomide: Mechanisms of action, repair and resistance. Curr. Mol. Pharmacol. 2012, 5, 102–114. [Google Scholar] [CrossRef]
- Dolera, M.; Malfassi, L.; Bianchi, C.; Carrara, N.; Finesso, S.; Marcarini, S.; Mazza, G.; Pavesi, S.; Sala, M.; Urso, G. Frameless stereotactic radiotherapy alone and combined with temozolomide for presumed canine gliomas. Vet. Comp. Oncol. 2018, 16, 90–101. [Google Scholar] [CrossRef]
- Denies, S.; Cicchelero, L.; de Rooster, H.; Daminet, S.; Polis, I.; Van de Maele, I.; Sanders, N.N. Immunological and angiogenic markers during metronomic temozolomide and cyclophosphamide in canine cancer patients. Vet. Comp. Oncol. 2017, 15, 594–605. [Google Scholar] [CrossRef] [PubMed]
- Hermisson, M.; Klumpp, A.; Wick, W.; Wischhusen, J.; Nagel, G.; Roos, W.; Kaina, B.; Weller, M. O6-methylguanine DNA methyltransferase and p53 status predict temozolomide sensitivity in human malignant glioma cells. J. Neurochem. 2006, 96, 766–776. [Google Scholar] [CrossRef]
- Marconato, L.; Rohrer Bley, C.; Leone, V.F.; Finotello, R. An open-label dose escalation study evaluating tolerability and safety of a single 5-days course of temozolomide in dogs with advanced cancer. Vet. Comp. Oncol. 2020, 18, 838–842. [Google Scholar] [CrossRef] [PubMed]
- Laclette, J.P.; Guerra, G.; Zetina, C. Inhibition of tubulin polymerization by mebendazole. Biochem. Biophys. Res. Commun. 1980, 92, 417–423. [Google Scholar] [CrossRef]
- Mukhopadhyay, T.; Sasaki, J.; Ramesh, R.; Roth, J.A. Mebendazole elicits a potent antitumor effect on human cancer cell lines both in vitro and in vivo. Clin. Cancer Res. 2002, 8, 2963–2969. [Google Scholar] [PubMed]
- Bai, R.Y.; Staedtke, V.; Aprhys, C.M.; Gallia, G.L.; Riggins, G.J. Antiparasitic mebendazole shows survival benefit in 2 preclinical models of glioblastoma multiforme. Neuro Oncol. 2011, 13, 974–982. [Google Scholar] [CrossRef] [PubMed]
- Bai, R.Y.; Staedtke, V.; Wanjiku, T.; Rudek, M.A.; Joshi, A.; Gallia, G.L.; Riggins, G.J. Brain Penetration and Efficacy of Different Mebendazole Polymorphs in a Mouse Brain Tumor Model. Clin. Cancer Res. 2015, 21, 3462–3470. [Google Scholar] [CrossRef] [Green Version]
- Dayan, A.D. Albendazole, mebendazole and praziquantel. Review of non-clinical toxicity and pharmacokinetics. Acta Trop. 2003, 86, 141–159. [Google Scholar] [CrossRef]
- Lai, S.R.; Castello, S.A.; Robinson, A.C.; Koehler, J.W. In vitro anti-tubulin effects of mebendazole and fenbendazole on canine glioma cells. Vet. Comp. Oncol. 2017, 15, 1445–1454. [Google Scholar] [CrossRef] [PubMed]
- Scheffler, A.; Fiebig, H.H.; Kabelitz, D.; Metelmann, H.R. Zur direkten zytotoxizitat von mistelpraparaten. Erfahrungsheilkunde 1993, 42, 338–346. [Google Scholar]
- Schumacher, U.; Stamouli, A.; Adam, E.; Peddie, M.; Pfuller, U. Biochemical, histochemical and cell biological investigations on the actions of mistletoe lectins I, II and III with human breast cancer cell lines. Glycoconj. J. 1995, 12, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Ribereau-Gayon, G.; Jung, M.L.; Frantz, M.; Anton, R. Modulation of cytotoxicity and enhancement of cytokine release induced by Viscum album L. extracts or mistletoe lectins. Anti-Cancer Drugs 1997, 8 (Suppl. 1), S3–S8. [Google Scholar] [CrossRef] [PubMed]
- Burger, A.M.; Mengs, U.; Schuler, J.B.; Fiebig, H.H. Antiproliferative activity of an aqueous mistletoe extract in human tumor cell lines and xenografts in vitro. Arzneimittelforschung 2001, 51, 748–757. [Google Scholar] [CrossRef] [PubMed]
- Lenartz, D.; Andermahr, J.; Plum, G.; Menzel, J.; Beuth, J. Efficiency of treatment with galactoside-specific lectin from mistletoe against rat glioma. Anticancer. Res. 1998, 18, 1011–1014. [Google Scholar]
- Lenartz, D.; Dott, U.; Menzel, J.; Schierholz, J.M.; Beuth, J. Survival of glioma patients after complementary treatment with galactoside-specific lectin from mistletoe. Anticancer. Res. 2000, 20, 2073–2076. [Google Scholar]
- Ribereau-Gayon, G.; Jung, M.L.; Beck, J.P. Die proteine, alkaloide und polysaccharide der Mistel (Viscum album L.). Therapeutikon 1989, 3, 22–26. [Google Scholar]
- Bantel, H.; Engels, I.H.; Voelter, W.; Schulze-Osthoff, K.; Wesselborg, S. Mistletoe lectin activates caspase-8/FLICE independently of death receptor signaling and enhances anticancer drug-induced apoptosis. Cancer Res. 1999, 59, 2083–2090. [Google Scholar]
- Endo, Y.; Tsurugi, K.; Franz, H. The site of action of the A-chain of mistletoe lectin I on eukaryotic ribosomes The RNA N-glycosidase activity of the protein. FEBS Lett. 1988, 231, 378–380. [Google Scholar] [CrossRef] [Green Version]
- Harmsma, M.; Gromme, M.; Ummelen, M.; Dignef, W.; Tusenius, K.J.; Ramaekers, F.C. Differential effects of Viscum album extract IscadorQu on cell cycle progression and apoptosis in cancer cells. Int. J. Oncol. 2004, 25, 1521–1529. [Google Scholar]
- Mockel, B.; Schwarz, T.; Zinke, H.; Eck, J.; Langer, M.; Lentzen, H. Effects of mistletoe lectin I on human blood cell lines and peripheral blood cells. Cytotoxicity, apoptosis and induction of cytokines. Arzneimittelforschung 1997, 47, 1145–1151. [Google Scholar] [PubMed]
- Beuth, J.; Ko, H.L.; Tunggal, L.; Pulverer, G. Das lektin der Mistel als immunodulator in der adjuvanten tumortherapie. Onkologie 1993, 25, 73–76. [Google Scholar]
- Ribéreau-Gayon, G.; Dumont, S.; Muller, C.; Jung, M.L.; Poindron, P.; Anton, R. Mistletoe lectins I, II and III induce the production of cytokines by cultured human monocytes. Cancer Lett. 1996, 109, 33–38. [Google Scholar] [CrossRef]
- Elsasser-Beile, U.; Voss, M.; Schuhle, R.; Wetterauer, U. Biological effects of natural and recombinant mistletoe lectin and an aqueous mistletoe extract on human monocytes and lymphocytes in vitro. J. Clin. Lab. Anal. 2000, 14, 255–259. [Google Scholar] [CrossRef]
- Hajto, T.; Hostanska, K.; Weber, K.; Zinke, H.; Fischer, J.; Mengs, U.; Lentzen, H.; Saller, R. Effect of a Recombinant Lectin, Viscum album Agglutinin on the Secretion of Interleukin-12 in Cultured Human Peripheral Blood Mononuclear Cells and on NK-Cell-Mediated Cytotoxicity of Rat Splenocytes in vitro and in vivo. Nat. Immun. 1998, 16, 34–46. [Google Scholar] [CrossRef]
- Baxevanis, C.N.; Papamichail, M. Characterization of the anti-tumor immune response in human cancers and strategies for immunotherapy. Crit. Rev. Oncol. Hematol. 1994, 16, 157–179. [Google Scholar] [CrossRef]
- Mengs, U.; Gothel, D.; Leng-Peschlow, E. Mistletoe extracts standardized to mistletoe lectins in oncology: Review on current status of preclinical research. Anticancer. Res. 2002, 22, 1399–1407. [Google Scholar] [PubMed]
- Elluru, S.R.; Duong Van Huyen, J.P.; Delignat, S.; Prost, F.; Heudes, D.; Kazatchkine, M.D.; Friboulet, A.; Kaveri, S.V. Antiangiogenic properties of viscum album extracts are associated with endothelial cytotoxicity. Anticancer. Res. 2009, 29, 2945–2950. [Google Scholar]
- Folkman, J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat. Med. 1995, 1, 27–31. [Google Scholar] [CrossRef]
- Podlech, O.; Harter, P.N.; Mittelbronn, M.; Poschel, S.; Naumann, U. Fermented mistletoe extract as a multimodal antitumoral agent in gliomas. Evid. Based Complement. Alternat Med. 2012, 2012, 501796. [Google Scholar] [CrossRef] [Green Version]
- Mistletoe Extracts (PDQ(R)): Health Professional Version. In PDQ Cancer Information Summaries; Bethesda, MD, USA, 2002. Available online: https://www.cancer.gov/publications/pdq/information-summaries (accessed on 1 December 2021).
- Zuzak, T.J.; Rist, L.; Eggenschwiler, J.; Grotzer, M.A.; Viviani, A. Paediatric medulloblastoma cells are susceptible to Viscum album (Mistletoe) preparations. Anticancer. Res. 2006, 26, 3485–3492. [Google Scholar] [PubMed]
- Lefebvre, G.B.L.; Oliveira, C.M. Tratamento de tumor venéreo transmissivel (tvt) canino utilizando Viscum album em associação à quimioterapia [Treatment of transmissible venereal tumor (tvt) in dogs with Viscum album (mistletoe) associated with chemotherapy]. Clin. Vet. 2007, 70, 78–86. [Google Scholar]
- Dickinson, P.J.; York, D.; Higgins, R.J.; LeCouteur, R.A.; Joshi, N.; Bannasch, D. Chromosomal Aberrations in Canine Gliomas Define Candidate Genes and Common Pathways in Dogs and Humans. J. Neuropathol. Exp. Neurol. 2016, 75, 700–710. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Markossian, S.; Grossman, A.; Brimacombe, K.; Arkin, M.; Auld, D.; Austin, C.P.; Baell, J.; Chung, T.D.Y.; Coussens, N.P.; Dahlin, J.L.; et al. (Eds.) Assay Guidance Manual [Internet]; Eli Lilly & Company and the National Center for Advancing Translational Sciences: Bethesda, MD, USA, 2004. Available online: https://www.ncbi.nlm.nih.gov/books/NBK53196/ (accessed on 1 December 2021).
- Huber, R.; Eisenbraun, J.; Miletzki, B.; Adler, M.; Scheer, R.; Klein, R.; Gleiter, C.H. Pharmacokinetics of natural mistletoe lectins after subcutaneous injection. Eur. J. Clin. Pharmacol. 2010, 66, 889–897. [Google Scholar] [CrossRef] [Green Version]
- Amabeoku, G.J.; Leng, M.J.; Syce, J.A. Antimicrobial and anticonvulsant activities of Viscum capense. J. Ethnopharmacol. 1998, 61, 237–241. [Google Scholar] [CrossRef]
- Jordan, E.; Wagner, H. Structure and properties of polysaccharides from Viscum album (L.). Oncology 1986, 43 (Suppl. 1), 8–15. [Google Scholar] [CrossRef]
- Carballo-Molina, O.A.; Velasco, I. Hydrogels as scaffolds and delivery systems to enhance axonal regeneration after injuries. Front. Cell. Neurosci. 2015, 9, 13. [Google Scholar] [CrossRef] [Green Version]
- Schoffski, P.; Riggert, S.; Fumoleau, P.; Campone, M.; Bolte, O.; Marreaud, S.; Lacombe, D.; Baron, B.; Herold, M.; Zwierzina, H.; et al. Phase I trial of intravenous aviscumine (rViscumin) in patients with solid tumors: A study of the European Organization for Research and Treatment of Cancer New Drug Development Group. Ann. Oncol. 2004, 15, 1816–1824. [Google Scholar] [CrossRef] [PubMed]
- Zarkovic, N.; Vukovic, T.; Loncaric, I.; Miletic, M.; Zarkovic, K.; Borovic, S.; Cipak, A.; Sabolovic, S.; Konitzer, M.; Mang, S. An overview on anticancer activities of the Viscum album extract Isorel. Cancer Biother. Radiopharm. 2001, 16, 55–62. [Google Scholar] [CrossRef]
- Kaegi, E. Unconventional therapies for cancer: 3. Iscador. Task Force on Alternative Therapies of the Canadian Breast Cancer Research Initiative. CMAJ 1998, 158, 1157–1159. [Google Scholar]
- Hutt, N.; Kopferschmitt-Kubler, M.; Cabalion, J.; Purohit, A.; Alt, M.; Pauli, G. Anaphylactic reactions after therapeutic injection of mistletoe (Viscum album L.). Allergol. Immunopathol. 2001, 29, 201–203. [Google Scholar] [CrossRef]
- Stauder, H.; Kreuser, E.D. Mistletoe extracts standardised in terms of mistletoe lectins (ML I) in oncology: Current state of clinical research. Onkologie 2002, 25, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Ernst, E.; Schmidt, K.; Steuer-Vogt, M.K. Mistletoe for cancer? A systematic review of randomised clinical trials. Int. J. Cancer 2003, 107, 262–267. [Google Scholar] [CrossRef]
- Lenartz, D.; Stoffel, B.; Menzel, J.; Beuth, J. Immunoprotective activity of the galactoside-specific lectin from mistletoe after tumor destructive therapy in glioma patients. Anticancer. Res. 1996, 16, 3799–3802. [Google Scholar]
- Piao, B.K.; Wang, Y.X.; Xie, G.R.; Mansmann, U.; Matthes, H.; Beuth, J.; Lin, H.S. Impact of complementary mistletoe extract treatment on quality of life in breast, ovarian and non-small cell lung cancer patients. A prospective randomized controlled clinical trial. Anticancer. Res. 2004, 24, 303–309. [Google Scholar] [PubMed]
- Kienle, G.S.; Kiene, H. Review article: Influence of Viscum album L. (European mistletoe) extracts on quality of life in cancer patients: A systematic review of controlled clinical studies. Integr. Cancer Ther. 2010, 9, 142–157. [Google Scholar] [CrossRef]
- Heiny, B.M.; Albrecht, V.; Beuth, J. Correlation of immune cell activities and beta-endorphin release in breast carcinoma patients treated with galactose-specific lectin standardized mistletoe extract. Anticancer. Res. 1998, 18, 583–586. [Google Scholar]
- Hartwig, A.C. Peripheral beta-endorphin and pain modulation. Anesth. Prog. 1991, 38, 75–78. [Google Scholar] [PubMed]
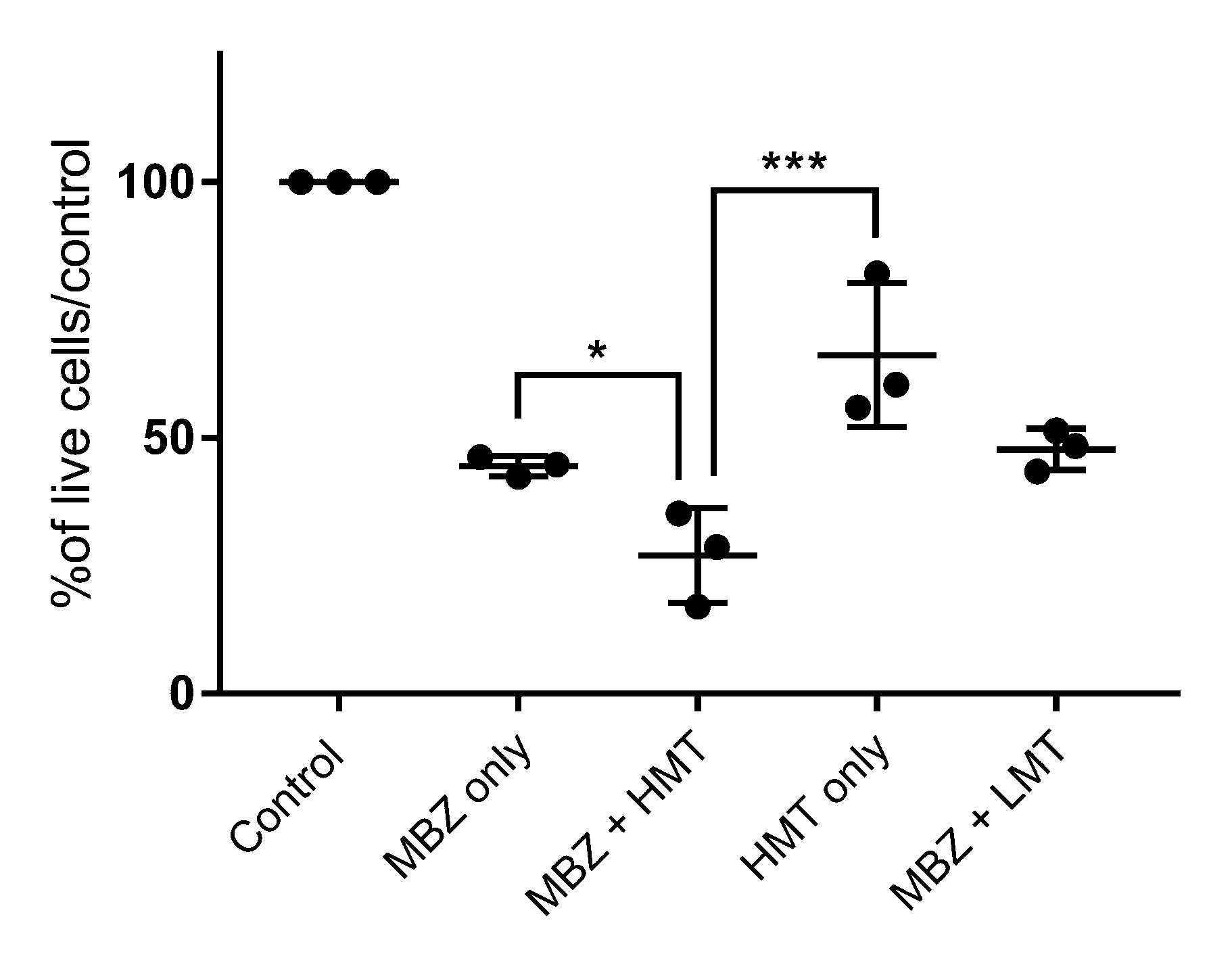
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wright, A.; Watanabe, R.; Koehler, J.W. European Mistletoe (Viscum album) Extract Is Cytotoxic to Canine High-Grade Astrocytoma Cells In Vitro and Has Additive Effects with Mebendazole. Vet. Sci. 2022, 9, 31. https://doi.org/10.3390/vetsci9010031
Wright A, Watanabe R, Koehler JW. European Mistletoe (Viscum album) Extract Is Cytotoxic to Canine High-Grade Astrocytoma Cells In Vitro and Has Additive Effects with Mebendazole. Veterinary Sciences. 2022; 9(1):31. https://doi.org/10.3390/vetsci9010031
Chicago/Turabian StyleWright, Anna, Rie Watanabe, and Jey W. Koehler. 2022. "European Mistletoe (Viscum album) Extract Is Cytotoxic to Canine High-Grade Astrocytoma Cells In Vitro and Has Additive Effects with Mebendazole" Veterinary Sciences 9, no. 1: 31. https://doi.org/10.3390/vetsci9010031
APA StyleWright, A., Watanabe, R., & Koehler, J. W. (2022). European Mistletoe (Viscum album) Extract Is Cytotoxic to Canine High-Grade Astrocytoma Cells In Vitro and Has Additive Effects with Mebendazole. Veterinary Sciences, 9(1), 31. https://doi.org/10.3390/vetsci9010031




