The Effects of Polyhexamethylene Biguanide (PHMB) and TLR Agonists Alone or as Polyplex Nanoparticles against Leishmania infantum Promastigotes and Amastigotes
Abstract
1. Introduction
2. Materials and Methods
2.1. Leishmania infantum Parasite Maintenance
2.2. Macrophages Maintenance
2.3. Drugs and TLR Agonists
2.4. Preparation and Analyses of Polyplex Nanoparticles
2.5. Half Inhibitory Concentration (IC50) Promastigote Susceptibility
2.6. Cytotoxicity for DH-82 Cell Line
2.7. Intracellular L. infantum Amastigotes Susceptibility Assay
2.8. Percentage of L. infantum-Infected Macrophages, Intensity of Infection and Infection Index
2.9. Sandwich ELISA for the Determination of TNF-α and IL-6
2.10. Statistical Analysis
3. Results
3.1. Polyplex Nanoparticles
3.2. Promastigotes and Macrophage Drugs Susceptibility
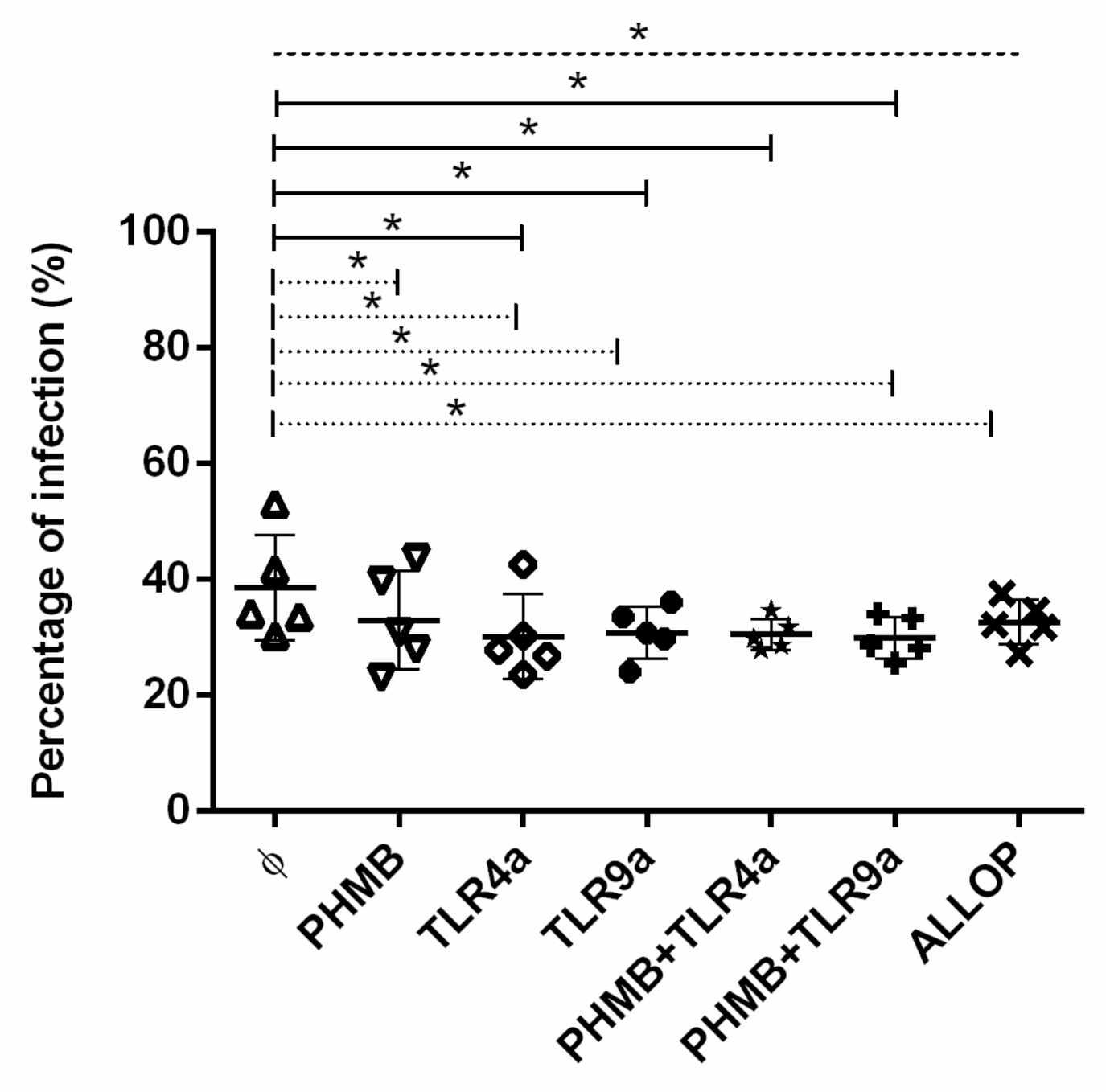
3.3. Percentages of Infected Macrophages
3.4. Intensity of Infection
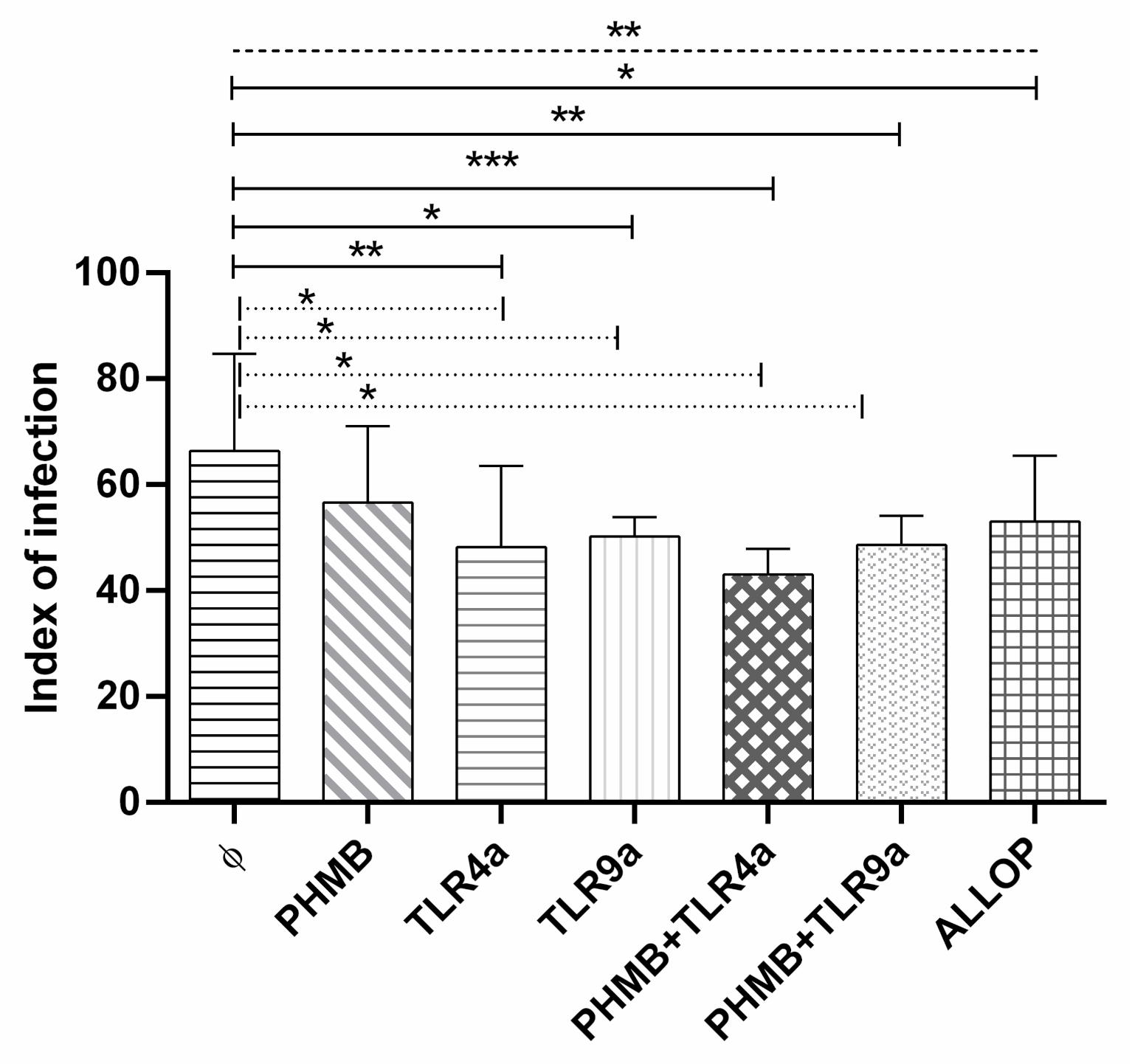
3.5. Infectivity Index
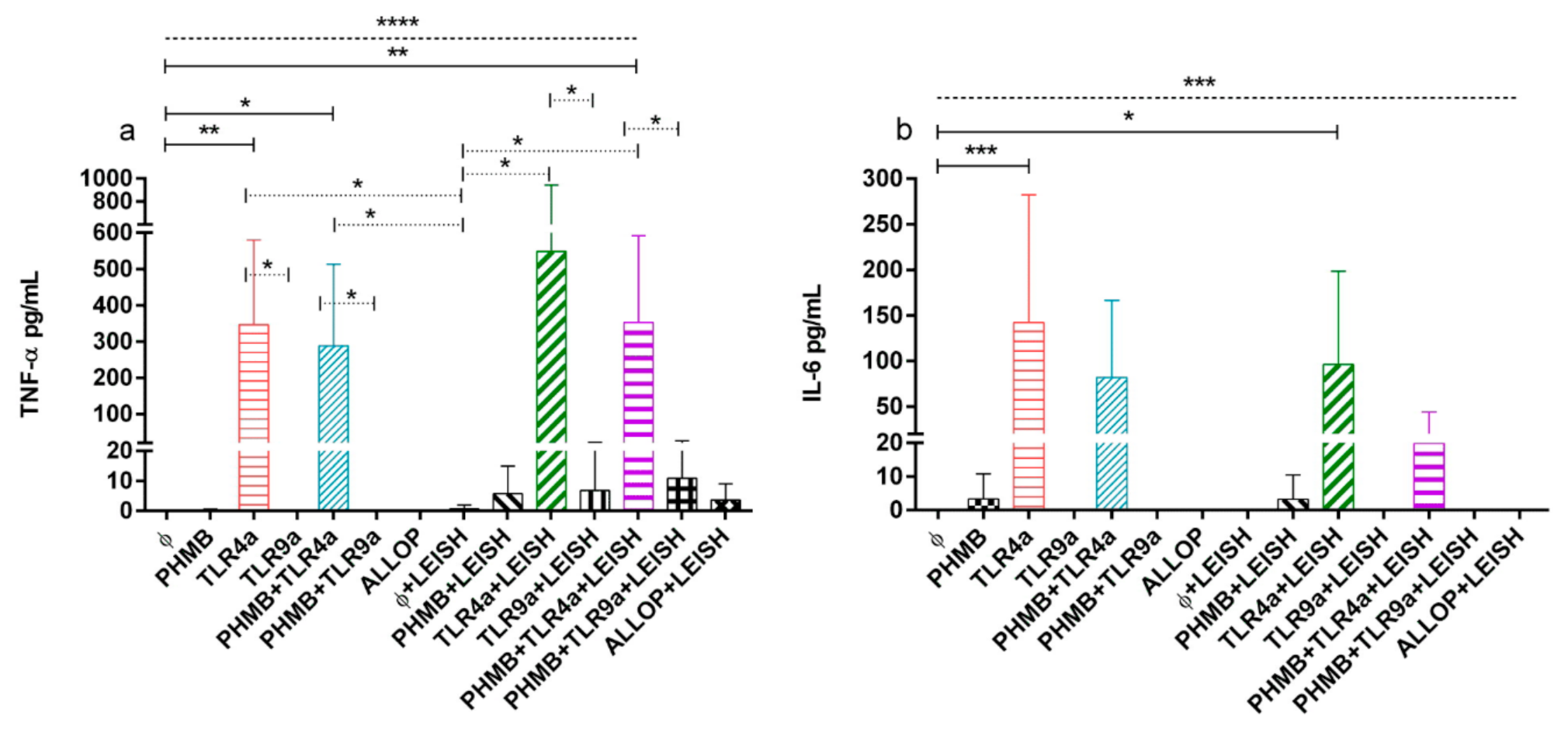
3.6. TNF-α and IL-6 Concentrations on Supernatant of DH-82 Treated Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gramiccia, M.; Gradoni, L. The current status of zoonotic leishmaniases and approaches to disease control. Int. J. Parasitol. 2005, 35, 1169–1180. [Google Scholar] [PubMed]
- Alvar, J.; Vélez, I.D.; Bern, C.; Herrero, M.; Desjeux, P.; Cano, J.; Jannin, J.; den Boer, M. Leishmaniasis worldwide and global estimates of its incidence. PLoS ONE 2012, 7, e35671. [Google Scholar]
- Ready, P.D. Biology of Phlebotomine sand flies as vectors of disease agents. Annu. Rev. Entomol. 2013, 58, 227–250. [Google Scholar] [PubMed]
- Olivier, M.; Gregory, D.J.; Forget, G. Subversion mechanisms by which Leishmania parasites can escape the host immune response: A signaling point of view. Clin. Microbiol. Rev. 2005, 18, 293–305. [Google Scholar] [PubMed]
- Baneth, G.; Koutinas, A.F.; Solano-Gallego, L.; Bourdeau, P.; Ferrer, L. Canine leishmaniosis–new concepts and insights on an expanding zoonosis: Part one. Trends Parasitol. 2008, 24, 324–330. [Google Scholar] [PubMed]
- Solano-Gallego, L.; Koutinas, A.F.; Miró, G.; Cardoso, L.; Pennisi, M.G.; Ferrer, L.; Bourdeau, P.; Oliva, G.; Baneth, G. Directions for the diagnosis, clinical staging, treatment and prevention of canine leishmaniosis. Vet. Parasitol. 2009, 165, 1–18. [Google Scholar]
- Solano-Gallego, L.; Cardoso, L.; Pennisi, M.G.; Petersen, C.; Bourdeau, P.; Oliva, G.; Miró, G.; Ferrer, L.; Baneth, G. Diagnostic challenges in the era of canine Leishmania infantum vaccines. Trends Parasitol. 2017, 33, 706–717. [Google Scholar]
- Koutinas, A.F.; Koutinas, C.K. Pathologic mechanisms underlying the clinical findings in canine leishmaniosis due to Leishmania infantum/chagasi. Vet. Pathol. 2014, 51, 527–538. [Google Scholar]
- Solano-Gallego, L.; Miró, G.; Koutinas, A.; Cardoso, L.; Pennisi, M.G.; Ferrer, L.; Bourdeau, P.; Oliva, G.; Baneth, G. LeishVet guidelines for the practical management of canine leishmaniosis. Parasites Vectors 2011, 4, 86. [Google Scholar]
- Ikeda-Garcia, F.A.; Lopes, R.S.; Marques, F.J.; de Lima, V.M.F.; Morinishi, C.K.; Bonello, F.L.; Zanette, M.F.; Perri, S.H.V.; Feitosa, M.M. Clinical and parasitological evaluation of dogs naturally infected by Leishmania (Leishmania) chagasi submitted to treatment with meglumine antimoniate. Vet. Parasitol. 2007, 143, 254–259. [Google Scholar]
- Miró, G.; Oliva, G.; Cruz, I.; Cañavate, C.; Mortarino, M.; Vischer, C.; Bianciardi, P. Multicentric, controlled clinical study to evaluate effectiveness and safety of miltefosine and allopurinol for canine leishmaniosis. Vet. Dermatol. 2009, 20, 397–404. [Google Scholar] [PubMed]
- Koutinas, A.F.; Saridomichelakis, M.N.; Mylonakis, M.E.; Leontides, L.; Polizopoulou, Z.; Billinis, C.; Argyriadis, D.; Diakou, N.; Papadopoulos, O. A randomised, blinded, placebo-controlled clinical trial with allopurinol in canine leishmaniosis. Vet. Parasitol. 2001, 98, 247–261. [Google Scholar] [PubMed]
- Ponte-Sucre, A.; Gamarro, F.; Dujardin, J.C.; Barrett, M.P.; López-Vélez, R.; García-Hernández, R.; Pountain, A.W.; Mwenechanya, R.; Papadopoulou, B. Drug resistance and treatment failure in leishmaniasis: A 21st century challenge. PLoS Negl. Trop. Dis. 2017, 11, 1–24. [Google Scholar]
- Vanaerschot, M.; Dumetz, F.; Roy, S.; Ponte-Sucre, A.; Arevalo, J.; Dujardin, J.C. Treatment failure in leishmaniasis: Drug-resistance or another (epi-) phenotype? Expert Rev. Anti. Infect. Ther. 2014, 12, 937–946. [Google Scholar] [PubMed]
- Murray, H.W.; Nathan, C.F. Macrophage microbicidal mechanisms in vivo: Reactive nitrogen versus oxygen intermediates in the killing of intracellular visceral Leishmania donovani. J. Exp. Med. 1999, 189, 741–746. [Google Scholar]
- Pinelli, E.; Gebhard, D.; Mommaas, A.M.; Van Hoeij, M.; Langermans, J.A.M.; Ruitenberg, E.J.; Rutten, V.P.M.G. Infection of a canine macrophage cell line with Leishmania infantum: Determination of nitric oxide production and anti-leishmanial activity. Vet. Parasitol. 2000, 92, 181–189. [Google Scholar]
- Holzmuller, P.; Hide, M.; Sereno, D.; Lemesre, J.L. Leishmania infantum amastigotes resistant to nitric oxide cytotoxicity: Impact on in vitro parasite developmental cycle and metabolic enzyme activities. Infect. Genet. Evol. 2006, 6, 187–197. [Google Scholar]
- Tuon, F.F.; Amato, V.S.; Bacha, H.A.; AlMusawi, T.; Duarte, M.I.; Neto, V.A. Toll-like receptors and leishmaniasis. Infect. Immun. 2008, 76, 866–872. [Google Scholar]
- Sacks, D.; Sher, A. Evasion of innate immunity by parasitic protozoa. Nat. Immunol. 2002, 3, 1041–1047. [Google Scholar]
- Mosser, D.M.; Rosenthal, L.A. Leishmania-macrophage interactions: Multiple receptors, multiple ligands and diverse cellular responses. Semin. Cell Biol. 1993, 4, 315–322. [Google Scholar]
- Saldarriaga, O.A.; Velásquez, J.I.; Ossa, J.E.; Rugeles, M.T. Standardization of bovine macrophage monolayers and isolation and culture of Trypanosomes. Memórias Inst. Oswaldo Cruz 2003, 98, 269–271. [Google Scholar]
- Graham, M.L.; Prescott, M.J. The multifactorial role of the 3Rs in shifting the harm-bene fi t analysis in animal models of disease. Eur. J. Pharmacol. 2015, 759, 19–29. [Google Scholar] [PubMed]
- Carpenter, S.; O’Neill, L.A.J. How important are Toll-like receptors for antimicrobial responses? Cell. Microbiol. 2007, 9, 1891–1901. [Google Scholar] [PubMed]
- Janssens, S.; Beyaert, R. Role of Toll-Like receptors in pathogen recognition. Clin. Microbiol. Rev. 2003, 16, 637–646. [Google Scholar] [PubMed]
- Zeytun, A.; Chaudhary, A.; Pardington, P.; Cary, R.B.; Gupta, G. Induction of cytokines and chemokines by Toll-like receptor signaling: Strategies for control of inflammation. Crit. Rev. Immunol. 2010, 30, 53–67. [Google Scholar] [PubMed]
- Akira, S.; Sato, S. Toll-like receptors and their signaling mechanisms. Scand. J. Infect. Dis. 2003, 35, 555–562. [Google Scholar] [PubMed]
- de Veer, M.J.; Curtis, J.M.; Baldwin, T.M.; DiDonato, J.A.; Sexton, A.; McConville, M.J.; Handman, E.; Schofield, L. MyD88 is essential for clearance of Leishmania major: Possible role for lipophosphoglycan and Toll-like receptor 2 signaling. Eur. J. Immunol. 2003, 33, 2822–2831. [Google Scholar]
- Flandin, J.F.; Chano, F.; Descoteaux, A. RNA interference reveals a role for TLR2 and TLR3 in the recognition of Leishmania donovani promastigotes by interferon-γ-primed macrophages. Eur. J. Immunol. 2006, 36, 411–420. [Google Scholar]
- Kropf, P.; Freudenberg, M.A.; Modolell, M.; Price, H.P.; Herath, S.; Antoniazi, S.; Galanos, C.; Smith, D.F.; Müller, I. Toll-like receptor 4 contributes to efficient control of infection with the protozoan parasite Leishmania major. Infect. Immun. 2004, 72, 1920–1928. [Google Scholar]
- Sacramento, L.; Trevelin, S.C.; Nascimento, M.S.; Lima-Jùnior, D.S.; Costa, D.L.; Almeida, R.P.; Cunha, F.Q.; Silva, J.S.; Carregaro, V. Toll-like receptor 9 signaling in dendritic cells regulates neutrophil recruitment to inflammatory foci following Leishmania infantum infection. Infect. Immun. 2015, 83, 4604–4616. [Google Scholar]
- Viana, K.F.; Aguiar-Soares, R.D.O.; Roatt, B.M.; Resende, L.A.; Silveira-Lemos, D.; Corrêa-Oliveira, R.; Martins-Filho, O.A.; Moura, S.L.; Zanini, M.S.; Araújo, M.S.S.; et al. Analysis using canine peripheral blood for establishing in vitro conditions for monocyte differentiation into macrophages for Leishmania chagasi infection and T-cell subset purification. Vet. Parasitol. 2013, 198, 62–71. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chindera, K.; Mahato, M.; Kumar Sharma, A.; Horsley, H.; Kloc-Muniak, K.; Kamaruzzaman, N.F.; Kumar, S.; McFarlane, A.; Stach, J.; Bentin, T.; et al. The antimicrobial polymer PHMB enters cells and selectively condenses bacterial chromosomes. Sci. Rep. 2016, 6, 23121. [Google Scholar] [CrossRef] [PubMed]
- Worsley, A.; Vassileva, K.; Tsui, J.; Song, W.; Good, L. Polyhexamethylene biguanide: Polyurethane blend nanofibrous membranes for wound infection control. Polymers 2019, 11, 915. [Google Scholar]
- Firdessa, R.; Good, L.; Amstalden, M.C.; Chindera, K.; Kamaruzzaman, N.F.; Schultheis, M.; Röger, B.; Hecht, N.; Oelschlaeger, T.A.; Meinel, L.; et al. Pathogen- and host-directed antileishmanial effects mediated by polyhexanide (PHMB). PLoS Negl. Trop. Dis. 2015, 9, 1–22. [Google Scholar] [CrossRef]
- Vanaerschot, M.; Maes, I.; Ouakad, M.; Adaui, V.; Maes, L.; de Doncker, S.; Rijal, S.; Chappuis, F.; Dujardin, J.C.; Decuypere, S. Linking in vitro and in vivo survival of clinical Leishmania donovani strains. PLoS ONE 2010, 5, e12211. [Google Scholar] [CrossRef]
- Maia, C.; Nunes, M.; Marques, M.; Henriques, S.; Rolão, N.; Campino, L. In vitro drug susceptibility of Leishmania infantum isolated from humans and dogs. Exp. Parasitol. 2013, 135, 36–41. [Google Scholar]
- Yasur-Landau, D.; Jaffe, C.L.; Doron-Faigenboim, A.; David, L.; Baneth, G. Induction of allopurinol resistance in Leishmania infantum isolated from dogs. PLoS Negl. Trop. Dis. 2017, 11, e0005910. [Google Scholar] [CrossRef] [PubMed]
- Yasur-Landau, D.; Jaffe, C.L.; David, L.; Baneth, G. Allopurinol resistance in Leishmania infantum from dogs with disease relapse. PLoS Negl. Trop. Dis. 2016, 10, 1–13. [Google Scholar] [CrossRef]
- Yasur-Landau, D.; Jaffe, C.L.; David, L.; Doron-Faigenboim, A.; Baneth, G. Resistance of Leishmania infantum to allopurinol is associated with chromosome and gene copy number variations including decrease in the S-adenosylmethionine synthetase (METK) gene copy number. Int. J. Parasitol. Drugs Drug Resist. 2018, 8, 403–410. [Google Scholar]
- Campino, L.; Maia, C. The role of reservoirs: Canine leishmaniasis. In Drug Resistance in Leishmania Parasites–Consequences, Molecular Mechanism and Possible Treatments; Ponte-Sucre, A., Padrón-Nieves, M., Eds.; Springer: Vienna, Austria, 2018; pp. 45–64. [Google Scholar]
- Cojean, S.; Houzé, S.; Haouchine, D.; Huteau, F.; Sylvie Lariven, V.H.; Michard, F.; Bories, C.; Pratlong, F.; Le Bras, J.; Loiseau, P.M.; et al. Leishmania resistance to miltefosine associated with genetic marker. Emerg. Infect. Dis. 2012, 18, 704–706. [Google Scholar]
- Mondelaers, A.; Sanchez-Cañete, M.P.; Hendrickx, S.; Eberhardt, E.; Garcia-Hernandez, R.; Lachaud, L.; Cotton, J.; Sanders, M.; Cuypers, B.; Imamura, H.; et al. Genomic and molecular characterization of miltefosine resistance in Leishmania infantum strains with either natural or acquired resistance through experimental selection of intracellular amastigotes. PLoS ONE 2016, 11, e0154101. [Google Scholar] [CrossRef] [PubMed]
- Gómez Pérez, V.; García-Hernandez, R.; Corpas-López, V.; Tomás, A.M.; Martín-Sanchez, J.; Castanys, S.; Gamarro, F. Decreased antimony uptake and overexpression of genes of thiol metabolism are associated with drug resistance in a canine isolate of Leishmania infantum. Int. J. Parasitol. Drugs Drug Resist. 2016, 6, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, T.; Tazuke, S.; Watanabe, M. Interaction of biologically active molecules with phospholipid membranes. Biochim. Biophys. Acta Biomembr. 1983, 735, 380–386. [Google Scholar] [CrossRef]
- Moon, E.; Choi, H.; Kong, H.; Quan, F. Experimental parasitology polyhexamethylene biguanide and chloroquine induce programmed cell death in Acanthamoeba castellanii. Exp. Parasitol. 2018, 191, 31–35. [Google Scholar] [PubMed]
- Maia, C.; Rolão, N.; Nunes, M.; Gonçalves, L.; Campino, L. Infectivity of five different types of macrophages by Leishmania infantum. Acta Trop. 2007, 103, 150–155. [Google Scholar] [CrossRef]
- Terreros, M.J.S.; de Luna, L.A.V.; Giorgio, S. Evaluation of antileishmanial drugs activities in an ex vivo model of leishmaniasis. Parasitol. Int. 2019, 71, 163–166. [Google Scholar] [CrossRef]
- da Luz, R.I.; Vermeersch, M.; Dujardin, J.C.; Cos, P.; Maes, L. In vitro sensitivity testing of Leishmania clinical field isolates: Preconditioning of promastigotes enhances infectivity for macrophage host cells. Antimicrob. Agents Chemother. 2009, 53, 5197–5203. [Google Scholar]
- Vermeersch, M.; da Luz, R.I.; Tote, K.; Timmermans, J.P.; Cos, P.; Maes, L. In vitro susceptibilities of Leishmania donovani promastigote and amastigote stages to antileishmanial reference drugs: Practical relevance of stage-specific differences. Antimicrob. Agents Chemother. 2009, 53, 3855–3859. [Google Scholar] [CrossRef]
- Rezaei Riabi, T.; Sharifi, I.; Miramin Mohammadi, A.; Khamesipour, A.; Hakimi Parizi, M. Evaluation of a possible synergistic effect of meglumine antimoniate with paromomycin, miltefosine or allopurinol on in vitro susceptibility of Leishmania tropica resistant isolate. Iran. J. Parasitol. 2013, 8, 396–401. [Google Scholar]
- Das, S.; Rani, M.; Pandey, K.; Sahoo, G.C.; Rabidas, V.N.; Singh, D.; Das, P. Combination of paromomycin and miltefosine promotes TLR4-dependent induction of antileishmanial immune response in vitro. J. Antimicrob. Chemother. 2012, 67, 2373–2378. [Google Scholar] [CrossRef]
- Martínez-Orellana, P.; Montserrat-Sangrà, S.; Quirola-Amores, P.; González, N.; Solano-Gallego, L. Cytokine effect of TLR3, TLR4, and TLR7 agonists alone or associated with Leishmania infantum antigen on blood from dogs. Biomed Res. Int. 2018, 2018, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Galdino, H.; Saar Gomes, R.; dos Santos, J.C.; Pessoni, L.L.; Maldaner, A.E.; Marques, S.M.; Gomes, C.M.; Dorta, M.L.; de Oliveira, M.A.P.; Joosten, L.A.B.; et al. Leishmania (Viannia) braziliensis amastigotes induces the expression of TNFα and IL-10 by human peripheral blood mononuclear cells in vitro in a TLR4-dependent manner. Cytokine 2016, 88, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Hosein, S.; Rodríguez-Cortés, A.; Blake, D.P.; Allenspach, K.; Alberola, J.; Solano-Gallego, L. Transcription of toll-like receptors 2, 3, 4 and 9, FoxP3 and Th17 cytokines in a susceptible experimental model of canine Leishmania infantum infection. PLoS ONE 2015, 10, e0140325. [Google Scholar] [CrossRef] [PubMed]
- Turchetti, A.P.; da Costa, L.F.; de Romão, E.L.; Fujiwara, R.T.; da Paixão, T.A.; Santos, R.L. Transcription of innate immunity genes and cytokine secretion by canine macrophages resistant or susceptible to intracellular survival of Leishmania infantum. Vet. Immunol. Immunopathol. 2015, 163, 67–76. [Google Scholar] [CrossRef]
- Quixabeira, V.B.L.; de Pereira, L.I.A.; Veras, P.R.V.; da Costa, A.C.V.; Fonseca, L.G.; Galdino, H., Jr.; da Silva, I.A., Jr.; Morato, C.I.; Pinto, S.A.; Pereira, A.J.C.S.; et al. Alterations in monocyte subsets and cytokine production after TLR activation in american cutaneous leishmaniasis. Parasite Immunol. 2019, 41, e12623. [Google Scholar] [CrossRef]
- Ramírez-Pineda, J.R.; Fröhlich, A.; Berberich, C.; Moll, H. Dendritic cells (DC) activated by CpG DNA ex vivo are potent inducers of host resistance to an intracellular pathogen that is independent of IL-12 derived from the immunizing DC. J. Immunol. 2004, 172, 6281–6289. [Google Scholar] [CrossRef]
- Roatt, B.M.; de Aguiar-Soares, R.D.O.; Coura-Vital, W.; Ker, H.G.; das Moreira, N.D.; Vitoriano-Souza, J.; Giunchetti, R.C.; Carneiro, C.M.; Reis, A.B. Immunotherapy and immunochemotherapy in visceral leishmaniasis: Promising treatments for this neglected disease. Front. Immunol. 2014, 5, 1–12. [Google Scholar] [CrossRef]

| TLR | Ligand | Ligand mg/mL | Polymer mg/mL | DLS Values ± Standard Deviation | ||
|---|---|---|---|---|---|---|
| Size [d.nm] | PDI | Count Rate [kcps] | ||||
| TLR4 | MPLA-SM | 0.092 | 0.69 | 166.7 ± 2.49 | 0.157 ± 0.01 | 190.1 ± 1.90 |
| TLR9 | 5′tccatgacgttcctgatgct | 0.092 | 1 | 75.9 ± 0.83 | 0.101 ± 0.01 | 216.8 ± 1.00 |
| Parasites and Cells | Allopurinol µM | Miltefosine µM | PHMB µM |
|---|---|---|---|
| L. infantum promastigotes | 0.124 | 9.455 | 1.495 |
| DH-82 macrophages | >18.3 | >122.678 | 6.133 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Orellana, P.; Baxarias, M.; Good, L.; Solano-Gallego, L. The Effects of Polyhexamethylene Biguanide (PHMB) and TLR Agonists Alone or as Polyplex Nanoparticles against Leishmania infantum Promastigotes and Amastigotes. Vet. Sci. 2020, 7, 179. https://doi.org/10.3390/vetsci7040179
Martínez-Orellana P, Baxarias M, Good L, Solano-Gallego L. The Effects of Polyhexamethylene Biguanide (PHMB) and TLR Agonists Alone or as Polyplex Nanoparticles against Leishmania infantum Promastigotes and Amastigotes. Veterinary Sciences. 2020; 7(4):179. https://doi.org/10.3390/vetsci7040179
Chicago/Turabian StyleMartínez-Orellana, Pamela, Marta Baxarias, Liam Good, and Laia Solano-Gallego. 2020. "The Effects of Polyhexamethylene Biguanide (PHMB) and TLR Agonists Alone or as Polyplex Nanoparticles against Leishmania infantum Promastigotes and Amastigotes" Veterinary Sciences 7, no. 4: 179. https://doi.org/10.3390/vetsci7040179
APA StyleMartínez-Orellana, P., Baxarias, M., Good, L., & Solano-Gallego, L. (2020). The Effects of Polyhexamethylene Biguanide (PHMB) and TLR Agonists Alone or as Polyplex Nanoparticles against Leishmania infantum Promastigotes and Amastigotes. Veterinary Sciences, 7(4), 179. https://doi.org/10.3390/vetsci7040179





