Epidemiological and Molecular Investigation of Ocular Fungal Infection in Equine from Egypt
Abstract
1. Introduction
2. Material and Methods
2.1. Ethical Consideration
2.2. Study Area, Animals, and Medical Records
2.3. Clinical Examination and Sample Collection
2.4. Isolation and Identification of Fungal Species
2.5. Fungal DNA Extraction
2.6. Polymerase Chain Reaction (PCR)
2.7. Purification of DNA Fragments from the Gel
2.8. DNA Sequencing, Assembling of Nucleotide Sequences, and Phylogenic Analysis
2.9. Statistical Analysis
3. Results
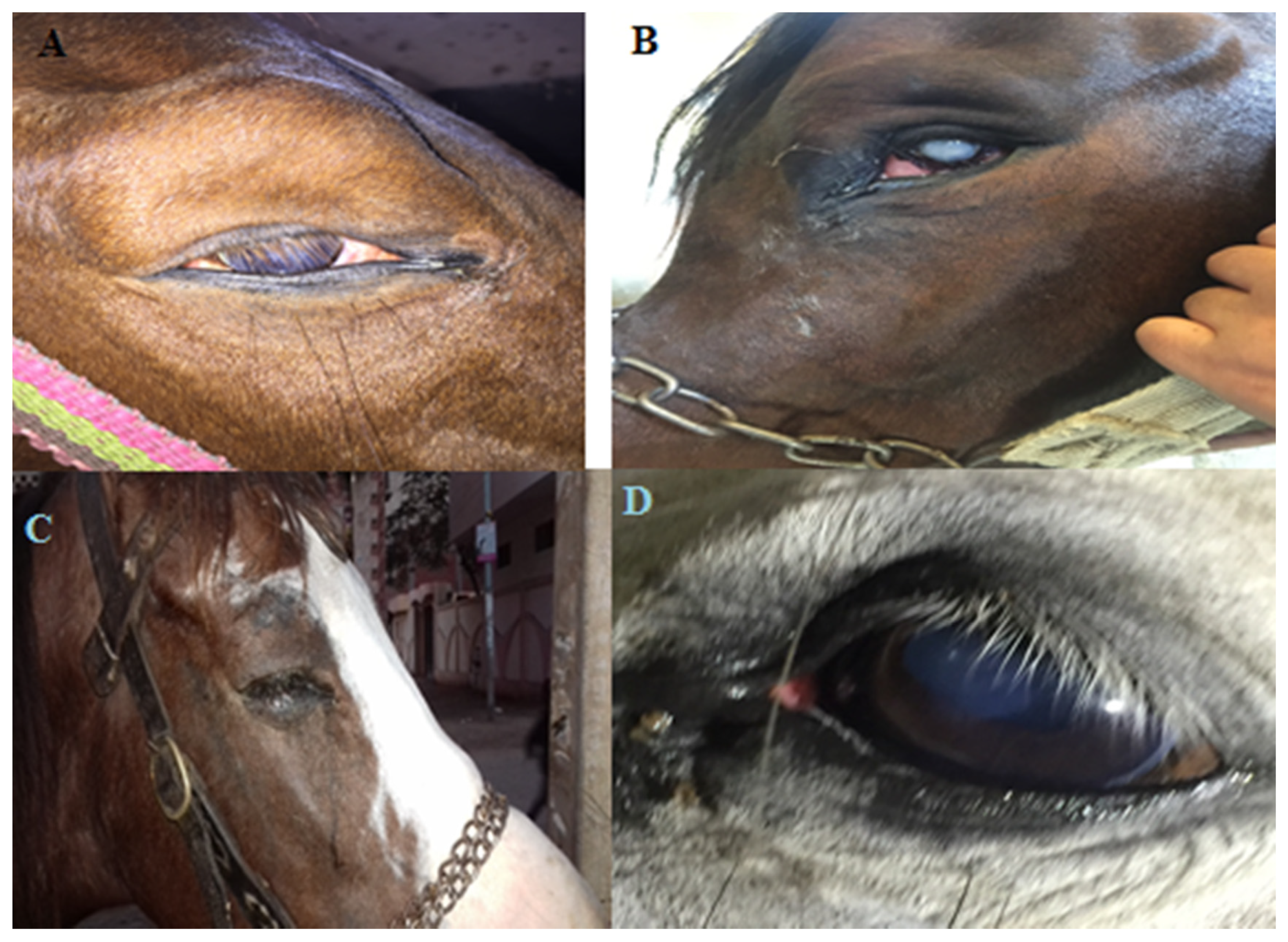
3.1. Clinical Signs
3.2. Morphology of the Isolated Fungal Species
3.3. Molecular Identification, Sequence Analysis, and Phylogeny of Isolated Fungi
3.4. Frequency of Isolated Fungi between Healthy and Diseased Equines’ Eyes and Associated Risk Factors
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Ethical Consideration
References
- Gelatt, K.N.; Gilger, B.C.; Kern, T.J. Veterinary Ophthalmology; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Paschalis-Trela, K.; Cywinska, A. The prevalence of ocular diseases in polish Arabian horses. BMC Vet. Res. 2017, 13, 319. [Google Scholar] [CrossRef] [PubMed]
- Johns, I.C.; Baxter, K.; Booler, H.; Hicks, C.; Menzies-Gow, N. Conjunctival bacterial and fungal flora in healthy horses in the UK. Vet. Ophthalmol. 2011, 14, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Carter, G.R.; Chengappa, M.; Roberts, A.W.; Claus, G.W.; Rikihisa, Y. Essentials of Veterinary Microbiology; Smithsonian Libraries: Washington, DC, USA, 1995; 394p. [Google Scholar]
- Kodikara, D.S.; De Silva, N.; Makuloluwa, C.A.; De Silva, N.; Gunatilake, M. Bacterial and fungal pathogens isolated from corneal ulcerations in domesticated elephants (Elephas maximus maximus) in Sri Lanka. Vet. Ophthalmol. 1999, 2, 191–192. [Google Scholar] [CrossRef] [PubMed]
- Petersen-Jones, S. Quantification of conjunctival sac bacteria in normal dogs and those suffering from keratoconjunctivitis sicca. Vet. Comp. Ophthalmol. 1997, 7, 29–35. [Google Scholar]
- Moore, C.P.; Heller, N.; Majors, L.J.; Whitley, R.D.; Burgess, E.C.; Weber, J. Prevalence of ocular microorganisms in hospitalized and stabled horses. Am. J. Vet. Res. 1988, 49, 773–777. [Google Scholar]
- Samuelson, D.A.; Andresen, T.L.; Gwin, R.M. Conjunctival fungal flora in horses, cattle, dogs, and cats. J. Am. Vet. Med. Assoc. 1984, 184, 1240–1242. [Google Scholar]
- Seyedmousavi, S.; Bosco, S.M.G.; de Hoog, S.; Ebel, F.; Elad, D.; Gomes, R.R.; Jacobsen, I.D.; Jensen, H.E.; Martel, A.; Mignon, B.; et al. Fungal infections in animals: A patchwork of different situations. Med. Mycol. 2018, 56, 165–187. [Google Scholar] [CrossRef]
- Moore, C.P.; Collins, B.K.; Fales, W.H. Antibacterial susceptibility patterns for microbial isolates associated with infectious keratitis in horses: 63 cases (1986-1994). J. Am. Vet. Med. Assoc. 1995, 207, 928–933. [Google Scholar]
- Andrew, S.E.; Brooks, D.E.; Smith, P.J.; Gelatt, K.N.; Chmielewski, N.T.; Whittaker, C.J. Equine ulcerative keratomycosis: Visual outcome and ocular survival in 39 cases (1987-1996). Equine Vet. J. 1998, 30, 109–116. [Google Scholar] [CrossRef]
- Reed, Z.; Thomasy, S.M.; Good, K.L.; Maggs, D.J.; Magdesian, K.G.; Pusterla, N.; Hollingsworth, S.R. Equine keratomycoses in California from 1987 to 2010 (47 cases). Equine Vet. J. 2013, 45, 361–366. [Google Scholar] [CrossRef]
- Beech, J.; Sweeney, C.R.; Irby, N. Keratomycoses in 11 horses. Equine Vet. J. 1983, 15, 39–44. [Google Scholar] [CrossRef]
- Gaarder, J.E.; Rebhun, W.C.; Ball, M.A.; Patten, V.; Shin, S.; Erb, H. Clinical appearances, healing patterns, risk factors, and outcomes of horses with fungal keratitis: 53 cases (1978-1996). J. Am. Vet. Med. Assoc. 1998, 213, 105–112. [Google Scholar] [PubMed]
- Brooks, D.E.; Andrew, S.E.; Dillavou, C.L.; Ellis, G.; Kubilis, P.S. Antimicrobial susceptibility patterns of fungi isolated from horses with ulcerative keratomycosis. Am. J. Vet. Res. 1998, 59, 138–142. [Google Scholar] [PubMed]
- Moore, C.P.; Fales, W.H.; Whittington, P.; Bauer, L. Bacterial and fungal isolates from Equidae with ulcerative keratitis. J. Am. Vet. Med. Assoc. 1983, 182, 600–603. [Google Scholar] [PubMed]
- Sansom, J.; Featherstone, H.; Barnett, K.C. Keratomycosis in six horses in the United Kingdom. Vet. Rec. 2005, 156, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Marolt, J.; Naglić, T.; Hajsig, D. Aspergillus oryzae as a cause of keratomycosis in the horse. Tierarztliche Praxis 1984, 12, 489–492. [Google Scholar]
- Chopin, J.; Sigler, L.; Connole, M.; O’Boyle, D.; Mackay, B.; Goldstein, L. Keratomycosis in a Percheron cross horse caused by Cladorrhinum bulbillosum. J. Med. Vet. Mycol. 1997, 35, 53–55. [Google Scholar] [CrossRef]
- Richter, M.; Hauser, B.; Kaps, S.; Spiess, B.M. Keratitis due to Histoplasma spp. in a horse. Vet. Ophthalmol. 2003, 6, 99–103. [Google Scholar] [CrossRef]
- Rosa, M.; Cardozo, L.M.; da Silva Pereira, J.; Brooks, D.E.; Martins, A.L.; Florido, P.S.; Stussi, J.S. Fungal flora of normal eyes of healthy horses from the State of Rio de Janeiro, Brazil. Vet. Ophthalmol 2003, 6, 51–55. [Google Scholar] [CrossRef]
- Whitley, R.D.; Moore, C.P. Microbiology of the equine eye in health and disease. Vet. Cinics N. Am. 1984, 6, 451–466. [Google Scholar] [CrossRef]
- Ledbetter, E.C.; Patten, V.H.; Scarlett, J.M.; Vermeylen, F.M. In vitro susceptibility patterns of fungi associated with keratomycosis in horses of the northeastern United States: 68 cases (1987-2006). J. Am. Vet. Med. Assoc. 2007, 231, 1086–1091. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.R.d.A.; Santana, A.F.; Almeida, A.C.d.V.R.; Sousa, R.F.; Perecmanis, S.; Galera, P.D. Bacterial culture and antibiotic sensitivity from the ocular conjunctiva of horses. Ciência Rural 2017, 47. [Google Scholar] [CrossRef]
- Mirhendi, H.; Diba, K.; Kordbacheh, P.; Jalalizand, N.; Makimura, K. Identification of pathogenic Aspergillus species by a PCR-restriction enzyme method. J. Med. Microbiol. 2007, 56, 1568–1570. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Sriramajayam, K.; Luo, D.; Liao, J. A Quick, Cost-Free Method of Purification of DNA Fragments from Agarose Gel. J. Cancer 2012, 3, 93–95. [Google Scholar] [CrossRef]
- Sanger, F.; Nicklen, S.; Coulson, A.R. DNA sequencing with chain-terminating inhibitors. Proc. Natl. Acad. Sci. USA 1977, 74, 5463–5467. [Google Scholar] [CrossRef]
- Tabor, S.; Richardson, C.C. A single residue in DNA polymerases of the Escherichia coli DNA polymerase I family is critical for distinguishing between deoxy- and dideoxyribonucleotides. Proc. Natl. Acad. Sci. USA 1995, 92, 6339–6343. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef]
- Brooks, D.E.; Matthews, A. Equine Ophtalmology; Saunders: Philadelphia, PA, USA, 2005; pp. 323–339. [Google Scholar]
- Galera, P.D.; Brooks, D.E. Optimal management of equine keratomycosis. Vet. Med. (Auckl) 2012, 3, 7–17. [Google Scholar] [CrossRef]
- Bharathi, M.J.; Ramakrishnan, R.; Vasu, S.; Meenakshi, R.; Palaniappan, R. Epidemiological characteristics and laboratory diagnosis of fungal keratitis. A three-year study. Indian J. Ophthalmol. 2003, 51, 315–321. [Google Scholar]
- Mann, S.S.; Singh, J.; Kalra, D.; Parihar, J.; Gupta, N.; Kumar, P. Medical and Surgical Management of Keratomycosis. Med. J. Armed Forces India 2008, 64, 40–42. [Google Scholar] [CrossRef]
- Sherman, A.B.; Clode, A.B.; Gilger, B.C. Impact of fungal species cultured on outcome in horses with fungal keratitis. Vet. Ophthalmol. 2017, 20, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Bonelli, F.; Barsotti, G.; Attili, A.R.; Mugnaini, L.; Cuteri, V.; Preziuso, S.; Corazza, M.; Preziuso, G.; Sgorbini, M. Conjunctival bacterial and fungal flora in clinically normal sheep. Vet. Rec. Open. 2014, 1, e000017. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, V.L.; Kinikar, A.G.; Bhalerao, D.S.; Roushani, S. A Case of Keratomycosis Caused by Fusarium Solani at Rural Tertiary Care Center. J. Clin. Diagn. Res. 2017, 11, DD01–DD03. [Google Scholar] [CrossRef] [PubMed]
- Prajna, N.V.; Krishnan, T.; Rajaraman, R.; Patel, S.; Shah, R.; Srinivasan, M.; Das, M.; Ray, K.J.; Oldenburg, C.E.; McLeod, S.D.; et al. Predictors of Corneal Perforation or Need for Therapeutic Keratoplasty in Severe Fungal Keratitis: A Secondary Analysis of the Mycotic Ulcer Treatment Trial II. JAMA Ophthalmol. 2017, 135, 987–991. [Google Scholar] [CrossRef]
- Ahmad, I.; Owais, M.; Shahid, M.; Aqil, F. Combating Fungal Infections: Problems and Remedy; Springer Science & Business Media: New York, NY, USA, 2010. [Google Scholar]
- Galán, A.; Martín-Suárez, E.; Gallardo, J.; Molleda, J. Clinical findings and progression of 10 cases of equine ulcerative keratomycosis (2004–2007). Equine Vet. Educ. 2009, 21, 236–242. [Google Scholar] [CrossRef]
- Blomme, E.; Piero, F.D.; Perle, K.L.; Wilkins, P.A. Aspergillosis in horses: A review. Equine Vet. Educ. 1998, 10, 86–93. [Google Scholar] [CrossRef]
- Seyedmousavi, S. Aspergillosis in Humans and Animals. In Recent Trends in Human and Animal Mycology; Singh, K., Srivastava, N., Eds.; Springer: Singapore, 2019; pp. 81–98. [Google Scholar]
- Wada, S.; Hobo, S.; Ode, H.; Niwa, H.; Moriyama, H. Equine keratomycosis in Japan. Vet. Ophthalmol. 2013, 16, 1–9. [Google Scholar] [CrossRef]
- Scotty, N.C. Equine keratomycosis. Clin. Tech. Equine Pract. 2005, 4, 29–36. [Google Scholar] [CrossRef]
- Teferi, K.; Adem, A. Common Ophthalmic Problems in Equines. Glob. Vet. 2019, 21, 246–254. [Google Scholar]
- Andrew, S.; Willis, M. Disease of the cornea and sclera. In Equine Ophthalmology, 1st ed.; Gilger, B., Ed.; Elsevier Saunders: Philadelphia, PA, USA, 2005; pp. 157–251. [Google Scholar]
- McLaughlin, S.A.; Gilger, B.C.; Whitley, R.D. Infectious keratitisin horses: Evaluation and management. Compend. Contin. Educ. Vet. 1992, 14, 372–379. [Google Scholar]
- Grahn, B.; Wolfer, J.; Keller, C.; Wilcock, B. Equinekeratomycosis: Clinical and laboratory findings in 23 cases. Prog. Vet. Comp. Ophthalmol. 1993, 3, 1–7. [Google Scholar]
- Mustikka, M.P.; Gronthal, T.S.C.; Pietila, E.M. Equine infectious keratitis in Finland: Associated microbial isolates and susceptibility profiles. Vet. Ophthalmol. 2020, 23, 148–159. [Google Scholar] [CrossRef] [PubMed]
- De Linde Henriksen, M.; Andersen, P.H.; Thomsen, P.D.; Plummer, C.E.; Mangan, B.; Heegaard, S.; Toft, N.; Brooks, D.E. Equine deep stromal abscesses (51 cases-2004–2009)—Part 1: The clinical aspects with attention to the duration of the corneal disease, treatment history, clinical appearance, and microbiology results. Vet. Ophthalmol. 2014, 17 (Suppl. 1), 6–13. [Google Scholar] [CrossRef] [PubMed]
- Proietto, L.R.; Plummer, C.E.; Maxwell, K.M.; Lamb, K.E.; Brooks, D.E. A retrospective analysis of environmental risk factors for the diagnosis of deep stromal abscess in 390 horses in North Central Florida from 1991 to 2013. Vet. Ophthalmol. 2016, 19, 291–296. [Google Scholar] [CrossRef]
- De Linde Henriksen, M.; Brooks, D.E. Standing ophthalmic surgeries in horses. Vet. Clin. N. Am. Equine Pract. 2014, 30, 91–110. [Google Scholar] [CrossRef]
- Aho, R.; Tala, M.; Kivalo, M. Mycotic keratitis in a horse caused by Aspergillus fumigatus. The first reported case in Finland. Acta Vet. Scand. 1991, 32, 373–376. [Google Scholar]
- Whitley, R.; Burgess, E.C.; Moore, C. Microbial isolates of the normal equine eye. Equine Vet. J. 1983, 15, 138–140. [Google Scholar] [CrossRef]
- Nikaido, H. Crossing the envelope: How cephalosporins reach their targets. Clin. Microbiol. Infect. 2000, 6 (Suppl. 3), 22–26. [Google Scholar] [CrossRef][Green Version]
- Barsotti, G.; Sgorbini, M.; Nardoni, S.; Corazza, M.; Mancianti, F. Occurrence of fungi from conjunctiva of healthy horses in Tuscany, Italy. Vet. Res. Commun. 2006, 30, 903–906. [Google Scholar] [CrossRef]
- Robinson, E.; Sprayberry, K. Preface. In Robinson’s Current Therapy in Equine Medicine, 7th ed.; Sprayberry, K.A., Robinson, N.E., Eds.; W.B. Saunders: St. Louis, MO, USA, 2015; Section XIV. [Google Scholar]
- Sellon, D.C.; Long, M.T. E Equine Infectious Diseases, 2nd ed.; Saunders: St. Louis, MO, USA, 2014. [Google Scholar]
- Gajjar, D.U.; Pal, A.K.; Ghodadra, B.K.; Vasavada, A.R. Microscopic evaluation, molecular identification, antifungal susceptibility, and clinical outcomes in fusarium, Aspergillus and, dematiaceous keratitis. BioMed Res. Int. 2013, 2013, 605308. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Banos, S.; Lentendu, G.; Kopf, A.; Wubet, T.; Glockner, F.O.; Reich, M. A comprehensive fungi-specific 18S rRNA gene sequence primer toolkit suited for diverse research issues and sequencing platforms. BMC Microbiol. 2018, 18, 190. [Google Scholar] [CrossRef] [PubMed]
| Fungal Species | Appearance on SDA |
|---|---|
| Asp. flavus | Colonies were greenish-yellow to olive and some had a white border. Texture is often floccose, especially near the center and overall can be velvety to woolly. |
| Asp. fumigatus | Colonies appeared blue-green, powdery. |
| Asp. niger | The initial growth of Aspergillus niger is white, becoming black later, giving “salt and pepper appearance,” which results from darkly pigmented conidia borne in large numbers on conidiophores. |
| Mucor species | Colonies appeared cottony to fluffy, white to yellow, becoming dark grey, with the development of sporangia. |
| Penicillium species | Colonies were fast growing, in shades of green, sometimes white, mostly consisting of a dense felt of conidiophores. |
| Alternaria species | Colonies appeared black to olivaceous-black or greyish and are suede-like to floccose. |
| Healthy (n = 72) | Diseased (n = 28) | Odds Ratio | p-Value | CI 95% | |
|---|---|---|---|---|---|
| Aspergillus flavus | 1.327 | 0.344 | 0.545–3.230 | ||
| Positive | 46(63.9%) | 16(57.1%) | |||
| Negative | 26(36.1%) | 12(42.9%) | |||
| Aspergillus fumigatus | 0.812 | 0.420 | 0.315–2.091 | ||
| Positive | 20(27.8%) | 9(32.1%) | |||
| Negative | 52(72.2%) | 19(67.9%) | |||
| Aspergillus niger | 0.661 | 0.322 | 0.218–2.002 | ||
| Positive | 11(15.3%) | 6(21.4%) | |||
| Negative | 61(84.7%) | 22(78.6%) | |||
| Mucor species | 2.864 | 0.143 | 0.603–13.611 | ||
| Positive | 13(18.1%) | 2(7.1%) | |||
| Negative | 59(81.9%) | 26(92.9%) | |||
| Penicillium species | 4.355 | 0.127 | 0.531–35.731 | ||
| Positive | 10(13.9%) | 1(3.6%) | |||
| Negative | 62(86.1%) | 27(96.4%) | |||
| Alternaria species | - | 0.369 | 0.627–0.807 | ||
| Positive | 3(4.2%) | 0(0%) | |||
| Negative | 69(95.8%) | 28(100%) |
| Aspergillus Flavus | Odds Ratio | p-Value | CI 95% | ||
|---|---|---|---|---|---|
| Negative | Positive | ||||
| Age | 1.415 | 0.357 | 0.488–4.105 | ||
| <5 | 32(84.2%) | 49(79%) | |||
| 5–10 | 6(15.8%) | 13(21%) | |||
| Breed | 0.521 | 0.126 | 0.203–1.334 | ||
| Arabian | 30(78.9%) | 41(66.1%) | |||
| Others | 8(21.1%) | 21(33.9%) | |||
| Sex | 0.560 | 0.163 | 0.218–1.440 | ||
| Male | 8(21.1%) | 20(71.4%) | |||
| Female | 30(78.9%) | 42(58.3%) | |||
| Housing | 0.560 | 0.128 | 0.240–1.308 | ||
| Indoor | 12(31.6%) | 28(45.2%) | |||
| Mixed | 26(68.4%) | 34(54.8%) | |||
| Season | 2.000 | 0.095 | 0.823–4.861 | ||
| Winter | 14(36.8%) | 14(22.6%) | |||
| Spring | 24(63.2%) | 48(77.4%) | |||
| Hygiene | 0.924 | 0.591 | 0.252–3.394 | ||
| Good | 4(10.5%) | 7(11.3%) | |||
| Bad | 34(89.5%) | 55(88.7%) | |||
| Ground type | 0.521 | 0.126 | 0.203–1.334 | ||
| Mud | 8(21.1%) | 21(33.9%) | |||
| Concrete | 30(78.9%) | 41(66.1%) | |||
| Stress | 0.645 | 0.219 | 0.271–1.536 | ||
| Individual | 11(28.9%) | 24(38.7%) | |||
| Over-crowding | 27(71.1%) | 38(61.3%) | |||
| Aspergillus Fumigatus | Odds Ratio | p-Value | CI 95% | ||
|---|---|---|---|---|---|
| Negative | Positive | ||||
| Age | 1.564 | 0.284 | 0.546–4.484 | ||
| <5 | 59(83.1%) | 22(75.9%) | |||
| 5–10 | 12(16.9%) | 7(24.1%) | |||
| Breed | 0.358 | 0.025 | 0.143–0.898 | ||
| Arabian | 55(77.5%) | 16(55.2%) | |||
| Others | 16(22.5%) | 13(44.8%) | |||
| Sex | 1.721 | 0.215 | 0.615–4.819 | ||
| Male | 22(31%) | 6(20.7%) | |||
| Female | 49(69%) | 23(79.3%) | |||
| Housing | 0.507 | 0.097 | 0.211–1.218 | ||
| Indoor | 25(35.2%) | 15(51.7%) | |||
| Mixed | 46(64.8%) | 14(48.3%) | |||
| Season | 1.320 | 0.386 | 0.490–3.558 | ||
| Winter | 21(29.6%) | 7(24.1%) | |||
| Spring | 50(70.4%) | 22(75.9%) | |||
| Hygiene | 1.960 | 0.326 | 0.397–9.681 | ||
| Good | 9(12.7%) | 2(6.9%) | |||
| Bad | 62(87.3%) | 27(93.1%) | |||
| Ground type | 0.358 | 0.025 | 0.143–0.898 | ||
| Mud | 16(22.5%) | 13(44.8%) | |||
| Concrete | 55(77.5%) | 16(55.2%) | |||
| Stress | 0.450 | 0.062 | 0.185–1.095 | ||
| Individual | 21(29.6%) | 14(48.3%) | |||
| Over-crowding | 50(70.4%) | 15(51.7%) | |||
| Aspergillus niger | Odd Ratio | p-Value | CI 95% | ||
|---|---|---|---|---|---|
| Negative | Positive | ||||
| Age | 1.395 | 0.410 | 0.399–4.880 | ||
| <5 | 68(81.9%) | 13(76.5%) | |||
| 5–10 | 15(18.1%) | 4(23.5%) | |||
| Breed | 1.401 | 0.411 | 0.416– 4.720 | ||
| Arabian | 58(69.9%) | 13(76.5%) | |||
| Others | 25(30.1%) | 4(23.5%) | |||
| Sex | 0.661 | 0.322 | 0.218–2.002 | ||
| Male | 22(26.5%) | 6(35.3%) | |||
| Female | 61(73.5%) | 11(64.7%) | |||
| Housing | 1.750 | 0.242 | 0.565–5.421 | ||
| Indoor | 35(42.2%) | 5(29.4%) | |||
| Mixed | 48(57.8%) | 12(70.6%) | |||
| Season | 0.920 | 0.549 | 0.292–2.902 | ||
| Winter | 23(27.7%) | 5(29.4%) | |||
| Spring | 60(72.3%) | 12(70.6%) | |||
| Hygiene | 2.192 | 0.406 | 0.262–18.363 | ||
| Good | 10(12%) | 1(5.9%) | |||
| Bad | 73(88%) | 16(94.1%) | |||
| Ground type | 1.401 | 0.411 | 0.416–4.720 | ||
| Mud | 25(30.1%) | 4(23.5%) | |||
| Concrete | 58(69.9%) | 13(76.5%) | |||
| Stress | 1.358 | 0.408 | 0.437–4.228 | ||
| Individual | 30(36.1%) | 5(29.4%) | |||
| Over-crowding | 53(63.9%) | 12(70.6%) | |||
| Mucor Species | Odd Ratio | p-Value | CI 95% | ||
|---|---|---|---|---|---|
| Negative | Positive | ||||
| Age | 1.078 | 0.578 | 0.272–4.273 | ||
| <5 | 69(81.2%) | 12(80%) | |||
| 5–10 | 16(18.8%) | 3(20%) | |||
| Breed | 0.556 | 0.235 | 0.178–1.737 | ||
| Arabian | 62(72.9%) | 9(60%) | |||
| Others | 23(27.1%) | 6(40%) | |||
| Sex | 0.524 | 0.206 | 0.167–1.640 | ||
| Male | 22(25.9%) | 6(40%) | |||
| Female | 63(74.1%) | 9(60%) | |||
| Housing | 0.383 | 0.078 | 0.124–1.177 | ||
| Indoor | 31(36.5%) | 9(60%) | |||
| Mixed | 54(63.5%) | 6(40%) | |||
| Season | 1.667 | 0.342 | 0.433–6.419 | ||
| Winter | 25(29.4%) | 3(20%) | |||
| Spring | 60(70.6%) | 12(80%) | |||
| Hygiene | 0.770 | 0.518 | 0.149–3.973 | ||
| Good | 9(10.6%) | 2(13.3%) | |||
| Bad | 76(89.4%) | 13(86.7%) | |||
| Ground type | 0.556 | 0.235 | 0.178–1.737 | ||
| Mud | 23(27.1%) | 6(40%) | |||
| Concrete | 62(72.9%) | 9(60%) | |||
| Stress | 0.777 | 0.434 | 0.252–2.395 | ||
| Individual | 29(34.1%) | 6(40%) | |||
| Over-crowding | 56(65.9%) | 9(60%) | |||
| Penicillium Species | Odd Ratio | p-Value | CI 95% | ||
|---|---|---|---|---|---|
| Negative | Positive | ||||
| Age | 0.394 | 0.338 | 0.047–3.285 | ||
| <5 | 71(79.8%) | 10(90.9%) | |||
| 5–10 | 18(20.2%) | 1(9.1%) | |||
| Breed | 4.590 | 0.113 | 0.560–37.625 | ||
| Arabian | 61(68.5%) | 10(90.9%) | |||
| Others | 28(31.5%) | 1(9.1%) | |||
| Sex | - | 0.021 | 1.070–1.302 | ||
| Male | 28(31.5%) | 0(0%) | |||
| Female | 61(68.5%) | 11(100%) | |||
| Housing | 7.800 | 0.024 | 0.957–63.559 | ||
| Indoor | 39(43.8%) | 1(9.1%) | |||
| Mixed | 50(56.2%) | 10(90.9%) | |||
| Season | 4.355 | 0.127 | 0.531–35.731 | ||
| Winter | 27(30.3%) | 1(9.1%) | |||
| Spring | 62(69.7%) | 10(90.9%) | |||
| Hygiene | - | 0.258 | 1.055–1.234 | ||
| Good | 11(12.4%) | 0(0%) | |||
| Bad | 78(87.6%) | 11(100%) | |||
| Ground type | 4.590 | 0.113 | 0.560–37.625 | ||
| Mud | 28(31.5%) | 1(9.1%) | |||
| Concrete | 61(68.5%) | 10(90.9%) | |||
| Stress | 6.182 | 0.051 | 0.757–50.463 | ||
| Individual | 34(38.2%) | 1(9.1%) | |||
| Over-crowding | 55(61.8%) | 10(90.9%) | |||
| Alternaria Species | Odd Ratio | p-Value | CI | ||
|---|---|---|---|---|---|
| Negative | Positive | ||||
| Age | - | 0.528 | 0.923–1.005 | ||
| <5 | 78(80.4%) | 3(100%) | |||
| 5–10 | 19(19.6%) | 0(0%) | |||
| Breed | 0.193 | 0.201 | 0.017–2.215 | ||
| Arabian | 70(72.2%) | 1(33.3%) | |||
| Others | 27(27.8%) | 2(66.7%) | |||
| Sex | 0.771 | 0.631 | 0.067–8.861 | ||
| Male | 27(27.8%) | 1(33.3%) | |||
| Female | 70(72.2%) | 2(66.7%) | |||
| Housing | 0.322 | 0.351 | 0.028–3.676 | ||
| Indoor | 38(39.2%) | 2(66.7%) | |||
| Mixed | 59(60.8%) | 1(33.3%) | |||
| Season | - | 0.369 | 0.994–1.095 | ||
| Winter | 28(28.9%) | 0(0%) | |||
| Spring | 69(71.1%) | 3(100%) | |||
| Hygiene | 1.035 | 0.702 | 0.995–1.076 | ||
| Good | 11(11.3%) | 0(0%) | |||
| Bad | 86(88.7%) | 3(100%) | |||
| Ground type | 0.193 | 0.201 | 0.017–2.215 | ||
| Mud | 27(27.8%) | 2(66.7%) | |||
| Concrete | 70(72.2%) | 1(33.3%) | |||
| Stress | 0.258 | 0.280 | 0.023–2.949 | ||
| Individual | 33(34%) | 2(66.7%) | |||
| Over-crowding | 64(66%) | 1(33.3%) | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tahoun, A.; Elnafarawy, H.K.; Elmahallawy, E.K.; Abdelhady, A.; Rizk, A.M.; El-Sharkawy, H.; Youssef, M.A.; El-Khodery, S.; Ibrahim, H.M.M. Epidemiological and Molecular Investigation of Ocular Fungal Infection in Equine from Egypt. Vet. Sci. 2020, 7, 130. https://doi.org/10.3390/vetsci7030130
Tahoun A, Elnafarawy HK, Elmahallawy EK, Abdelhady A, Rizk AM, El-Sharkawy H, Youssef MA, El-Khodery S, Ibrahim HMM. Epidemiological and Molecular Investigation of Ocular Fungal Infection in Equine from Egypt. Veterinary Sciences. 2020; 7(3):130. https://doi.org/10.3390/vetsci7030130
Chicago/Turabian StyleTahoun, Amin, Helmy K. Elnafarawy, Ehab Kotb Elmahallawy, Abdelhamed Abdelhady, Amira M. Rizk, Hanem El-Sharkawy, Mohamed A. Youssef, Sabry El-Khodery, and Hussam M. M. Ibrahim. 2020. "Epidemiological and Molecular Investigation of Ocular Fungal Infection in Equine from Egypt" Veterinary Sciences 7, no. 3: 130. https://doi.org/10.3390/vetsci7030130
APA StyleTahoun, A., Elnafarawy, H. K., Elmahallawy, E. K., Abdelhady, A., Rizk, A. M., El-Sharkawy, H., Youssef, M. A., El-Khodery, S., & Ibrahim, H. M. M. (2020). Epidemiological and Molecular Investigation of Ocular Fungal Infection in Equine from Egypt. Veterinary Sciences, 7(3), 130. https://doi.org/10.3390/vetsci7030130







