Prospective Uncontrolled Interventional Study of Itraconazole and β-Glucans (Euglena gracilis) to Assess Safeness and Clinical Effectiveness in Cats with Cutaneous and Mucosal Sporotrichosis
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Inclusion, Exclusion and Elimination Criteria
2.2. Study Procedures and Techniques
2.3. Treatment
2.4. Monitoring of Adverse Drug Reactions (ADRs)
2.5. Statistical Analysis
3. Results
Treatment Outcome
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rodrigues, A.M.; Gonçalves, S.S.; De Carvalho, J.A.; Borba-Santos, L.P.; Rozental, S.; Camargo, Z.P.D. Current Progress on Epidemiology, Diagnosis, and Treatment of Sporotrichosis and Their Future Trends. J. Fungi 2022, 8, 776. [Google Scholar] [CrossRef]
- Kidd, S.E.; Sandoval-Denis, M.; Malik, R.; Hagen, F.; Rodrigues, A.M. Sporothrix davidellisii: A New Pathogenic Species Belonging to the Sporothrix pallida Complex. Med. Mycol. 2025, 63, myaf034. [Google Scholar] [CrossRef]
- Montenegro, H.; Rodrigues, A.M.; Dias, M.A.G.; Da Silva, E.A.; Bernardi, F.; De Camargo, Z.P. Feline Sporotrichosis Due to Sporothrix brasiliensis: An Emerging Animal Infection in São Paulo, Brazil. BMC Vet. Res. 2014, 10, 269. [Google Scholar] [CrossRef]
- Poester, V.R.; Mattei, A.S.; Madrid, I.M.; Pereira, J.T.B.; Klafke, G.B.; Sanchotene, K.O.; Brandolt, T.M.; Xavier, M.O. Sporotrichosis in Southern Brazil, towards an Epidemic? Zoonoses Public Health 2018, 65, 815–821. [Google Scholar] [CrossRef] [PubMed]
- Cognialli, R.C.R.; Cáceres, D.H.; Bastos, F.D.A.G.D.; Cavassin, F.B.; Lustosa, B.P.R.; Vicente, V.A.; Breda, G.L.; Santos-Weiss, I.; Queiroz-Telles, F. Rising Incidence of Sporothrix brasiliensis Infections, Curitiba, Brazil, 2011–2022. Emerg. Infect. Dis. 2023, 29, 1330–1339. [Google Scholar] [CrossRef] [PubMed]
- Colombo, S.A.; Bicalho, G.C.; de Oliveira, C.S.F.; Soares, D.F.M.; Salvato, L.A.; Keller, K.M.; Bastos, C.V.E.; Morais, M.H.F.; Rodrigues, A.M.; Cunha, J.L.R.; et al. Emergence of zoonotic sporotrichosis due to Sporothrix brasiliensis in Minas Gerais, Brazil: A molecular approach to the current animal disease. Mycoses 2023, 66, 911–922. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, L.A.; De Carvalho, J.A.; De Oliveira E Lima, L.; Da Silva Neto, F.B.; De Oliveira Lima, E.; Da Silva Rocha, W.P.; De Camargo, Z.P.; Rodrigues, A.M.; Dulgheroff, A.C.B.; Guerra, F.Q.S. Analyses of the Genetic Diversity and Population Structures of Sporothrix spp. Clinical Isolates from Paraíba, Brazil. JoF 2024, 10, 848. [Google Scholar] [CrossRef]
- Pinheiro, R.E.E.; Santana, C.C.A.; Martins, L.M.S.; Veloso, D.S.; Luz, J.A.D.S.; Lima, O.B.; Santos, N.C.; Gonsales, F.F.; De Carvalho, J.A.; Rodrigues, A.M. The Emergence of Cat-Transmitted Sporotrichosis Driven by Sporothrix brasiliensis in Piauí, Brazil. Zoonoses Public Health 2025, 72, 127–135. [Google Scholar] [CrossRef]
- Valente, R.M.; Verçosa, J.V.D.M.; Souza, E.R.D.; Gordiano, N.P.; Barroso, L.D.C.; Carvalho, S.M.D.S.; Alves, M.J.; Fonseca, F.R.; Grisolia, M.E.; Almeida, M.E.M.D.; et al. Expansion of Human and Animal Sporotrichosis in Manaus, Amazonas State, Brazil. Cad. Saúde Pública 2025, 41, e00180024. [Google Scholar] [CrossRef]
- De Andrade Galliano Daros Bastos, F.; Raimundo Cognialli, R.C.; Rodrigues De Farias, M.; Dos Santos Monti, F.; Wu, K.; Queiroz-Telles, F. Spread of Sporothrix spp. through Respiratory Droplets from Infected Cats: A Potential Route of Transmission. Med. Mycol. 2022, 60, myac079. [Google Scholar] [CrossRef]
- Siew, H.H. The Current Status of Feline Sporotrichosis in Malaysia. Med. Mycol. J. 2017, 58, E107–E113. [Google Scholar] [CrossRef]
- Han, H.S.; Kano, R. Feline Sporotrichosis in Asia. Braz. J. Microbiol. 2021, 52, 125–134. [Google Scholar] [CrossRef]
- Santos, M.T.; Nascimento, L.F.D.J.; Barbosa, A.A.T.; Martins, M.P.; Tunon, G.I.L.; Santos, P.O.M.; Dantas-Torres, F.; Dolabella, S.S. The Rising Incidence of Feline and Cat-transmitted Sporotrichosis in Latin America. Zoonoses Public Health 2024, 71, 609–619. [Google Scholar] [CrossRef] [PubMed]
- Vizcaychipi, K.A.; López-Joffre, M.C.; Martínez, M.; Cuéllar-Sáenz, J.A.; Ramos, M.; Agüero, C.; Olsina, N.; Grassi, E.; Couto, E.; Mendoza, J.; et al. Emergence of Feline Sporotrichosis near Brazil Border, Argentina, 2023–2024. Emerg. Infect. Dis. 2025, 31, 1045–1048. [Google Scholar] [CrossRef] [PubMed]
- Chieosilapatham, P.; Chuamanochan, M.; Chiewchavit, S.; Saikruatep, R.; Amornrungsun, E.; Preechasuth, K. Sporothrix schenckii Sensu Stricto Related to Zoonotic Transmission in Thailand. Med. Mycol. Case Rep. 2023, 41, 44–47. [Google Scholar] [CrossRef] [PubMed]
- Barnacle, J.R.; Chow, Y.J.; Borman, A.M.; Wyllie, S.; Dominguez, V.; Russell, K.; Roberts, H.; Armstrong-James, D.; Whittington, A.M. The First Three Reported Cases of Sporothrix brasiliensis Cat-Transmitted Sporotrichosis Outside South America. Med. Mycol. Case Rep. 2023, 39, 14–17. [Google Scholar] [CrossRef]
- Hennessee, I.; Barber, E.; Petro, E.; Lindemann, S.; Buss, B.; Santos, A.; Gade, L.; Lockhart, S.R.; Sexton, D.J.; Chiller, T.; et al. Sporotrichosis Cluster in Domestic Cats and Veterinary Technician, Kansas, USA, 2022. Emerg. Infect. Dis. 2024, 30, 1053–1055. [Google Scholar] [CrossRef]
- Miranda, L.; Gillett, S.; Ames, Y.; Krockenberger, M.; Malik, R. Zoonotic Feline Sporotrichosis: A Small Case Cluster in Perth, Western Australia, and a Review of Previous Feline Cases from Australia. Aust. Vet. J. 2024, 102, 638–645. [Google Scholar] [CrossRef]
- Schubach, T.M.P.; Schubach, A.; Okamoto, T.; Barros, M.B.L.; Figueiredo, F.B.; Cuzzi, T.; Fialho-Monteiro, P.C.; Reis, R.S.; Perez, M.A.; Wanke, B. Evaluation of an Epidemic of Sporotrichosis in Cats: 347 Cases (1998–2001). Javma 2004, 224, 1623–1629. [Google Scholar] [CrossRef]
- Reis, E.G.D.; Pereira, S.A.; Miranda, L.H.M.; Oliveira, R.D.V.C.D.; Quintana, M.D.S.B.; Viana, P.G.; Figueiredo, A.B.F.; Honorato, C.C.D.S.; Pereira-Oliveira, G.R.; Silva, J.N.; et al. A Randomized Clinical Trial Comparing Itraconazole and a Combination Therapy with Itraconazole and Potassium Iodide for the Treatment of Feline Sporotrichosis. J. Fungi 2024, 10, 101. [Google Scholar] [CrossRef]
- Gremião, I.D.F.; Martins Da Silva Da Rocha, E.; Montenegro, H.; Carneiro, A.J.B.; Xavier, M.O.; De Farias, M.R.; Monti, F.; Mansho, W.; De Macedo Assunção Pereira, R.H.; Pereira, S.A.; et al. Guideline for the Management of Feline Sporotrichosis Caused by Sporothrix brasiliensis and Literature Revision. Braz. J. Microbiol. 2021, 52, 107–124. [Google Scholar] [CrossRef]
- Miranda, L.H.M.; Silva, J.N.; Gremião, I.D.F.; Menezes, R.C.; Almeida-Paes, R.; Dos Reis, E.G.; De Oliveira, R.D.V.C.; De Araujo, D.S.D.A.; Ferreiro, L.; Pereira, S.A. Monitoring Fungal Burden and Viability of Sporothrix spp. in Skin Lesions of Cats for Predicting Antifungal Treatment Response. J. Fungi 2018, 4, 92. [Google Scholar] [CrossRef]
- Miranda, L.H.M.; Santiago, M.D.A.; Frankenfeld, J.; Reis, E.G.D.; Menezes, R.C.; Pereira, S.A.; Gremião, I.D.F.; Hofmann-Lehmann, R.; Conceição-Silva, F. Neutrophil Oxidative Burst Profile Is Related to a Satisfactory Response to Itraconazole and Clinical Cure in Feline Sporotrichosis. JoF 2024, 10, 422. [Google Scholar] [CrossRef]
- Madrid, I.M.; Mattei, A.; Martins, A.; Nobre, M.; Meireles, M. Feline Sporotrichosis in the Southern Region of Rio Grande Do Sul, Brazil: Clinical, Zoonotic and Therapeutic Aspects. Zoonoses Public Health 2010, 57, 151–154. [Google Scholar] [CrossRef]
- Villalobos, W.R.; Monti, F.; Ferreira, T.; Sato, S.; Telles, F.; Farias, M. Therapeutic Efficacy of Isavuconazole and Potassium Iodide in a Cat with Refractory Sporotrichosis. Vet. Dermatol. 2023, 34, 624–628. [Google Scholar] [CrossRef]
- Rocha, R.F.D.B.; Schubach, T.M.P.; Pereira, S.A.; Dos Reis, E.G.; Carvalho, B.W.; Gremião, I.D.F. Refractory Feline Sporotrichosis Treated with Itraconazole Combined with Potassium Iodide. J. Small Anim. Pract. 2018, 59, 720–721. [Google Scholar] [CrossRef] [PubMed]
- Pereira, S.A.; Passos, S.R.L.; Silva, J.N.; Gremião, I.D.F.; Figueiredo, F.B.; Teixeira, J.L.; Monteiro, P.C.F.; Schubach, T.M.P. Response to Azolic Antifungal Agents for Treating Feline Sporotrichosis. Vet. Rec. 2010, 166, 290–294. [Google Scholar] [CrossRef] [PubMed]
- Crothers, S.L.; White, S.D.; Ihrke, P.J.; Affolter, V.K. Sporotrichosis: A Retrospective Evaluation of 23 Cases Seen in Northern California (1987–2007). Vet. Dermatol. 2009, 20, 249–259. [Google Scholar] [CrossRef]
- Davies, C.; Troy, G. Deep Mycotic Infections in Cats. J. Am. Anim. Hosp. Assoc. 1996, 32, 380–391. [Google Scholar] [CrossRef]
- Nobre, M.O.; Castro, A.P.; Caetano, D.; Souza, L.L.; Meireles, M.C.A.; Ferreiro, L. Recurrence of sporotrichosis in cats with zoonotic involvement. Rev. Iberoam. Micol. 2001, 18, 137–140. [Google Scholar]
- Honse, C.O.; Rodrigues, A.M.; Gremião, I.D.F.; Pereira, S.A.; Schubach, T.M.P. Use of Local Hyperthermia to Treat Sporotrichosis in a Cat. Vet. Rec. 2010, 166, 208–209. [Google Scholar] [CrossRef]
- Reis, E.G.; Gremião, I.D.F.; Kitada, A.A.B.; Rocha, R.F.D.B.; Castro, V.S.P.; Barros, M.B.L.; Menezes, R.C.; Pereira, S.A.; Schubach, T.M.P. Potassium Iodide Capsule Treatment of Feline Sporotrichosis. J. Feline Med. Surg. 2012, 14, 399–404. [Google Scholar] [CrossRef]
- De Souza, C.P.; Lucas, R.; Ramadinha, R.H.; Pires, T.B. Cryosurgery in Association with Itraconazole for the Treatment of Feline Sporotrichosis. J. Feline Med. Surg. 2016, 18, 137–143. [Google Scholar] [CrossRef]
- Thomson, J.; Trott, D.J.; Malik, R.; Galgut, B.; McAllister, M.M.; Nimmo, J.; Renton, D.; Kidd, S.E. An Atypical Cause of Sporotrichosis in a Cat. Med. Mycol. Case Rep. 2019, 23, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Santi, J.P.; Santos, C.R.G.R.; Santos, A.S.D.; Souza, H.J.M. Intranasal Clotrimazole Spray 1% Associated with Oral Itraconazole for Nasal Feline Sporotrichosis: A Case Series. Braz. J. Vet. Med. 2022, 44, e004821. [Google Scholar] [CrossRef]
- Cabral, F.V.; Sellera, F.P.; Ribeiro, M.S. Feline Sporotrichosis Successfully Treated with Methylene Blue-Mediated Antimicrobial Photodynamic Therapy and Low Doses of Itraconazole. Photodiagnosis Photodyn. Ther. 2022, 40, 103154. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, D.S.C.; Machado, L.J.; Pereira, J.G.; Baptista, A.R.D.S.; Rocha, E.M.D.S.D. Laser Therapy in the Treatment of Feline Sporotrichosis: A Case Series. Braz. J. Vet. Med. 2023, 45, e005822. [Google Scholar] [CrossRef] [PubMed]
- Ramos, M.L.M.; Barrinha, A.; Araújo, G.R.D.S.; Alves, V.; Andrade, I.B.D.; Corrêa-Junior, D.; Motta, M.C.M.; Almeida-Paes, R.; Frases, S. Photodynamic Therapy Reduces Viability, Enhances Itraconazole Activity, and Impairs Mitochondrial Physiology of Sporothrix brasiliensis. Microbes Infect. 2025, 27, 105440. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Oliveira, G.R.; Gremião, I.D.F.; Corrêa, M.L.; Honorato, C.C.S.; Viana, P.G.; Figueiredo, A.B.F.; Boechat, J.S.; dos Reis, É.G.; Oliveira, R.D.V.C.; da Silva, A.C.D.A.; et al. Plasma Itraconazole Concentrations during Treatment of Feline Sporotrichosis. Med. Mycol. 2024, 62, myae076. [Google Scholar] [CrossRef]
- Nakasu, C.C.T.; Waller, S.B.; Ripoll, M.K.; Ferreira, M.R.A.; Conceição, F.R.; Gomes, A.D.R.; Osório, L.D.G.; De Faria, R.O.; Cleff, M.B. Feline Sporotrichosis: A Case Series of Itraconazole-Resistant Sporothrix brasiliensis Infection. Braz. J. Microbiol. 2021, 52, 163–171. [Google Scholar] [CrossRef]
- Espinel-Ingroff, A.; Abreu, D.P.B.; Almeida-Paes, R.; Brilhante, R.S.N.; Chakrabarti, A.; Chowdhary, A.; Hagen, F.; Córdoba, S.; Gonzalez, G.M.; Govender, N.P.; et al. Multicenter, International Study of MIC/MEC Distributions for Definition of Epidemiological Cutoff Values for Sporothrix Species Identified by Molecular Methods. Antimicrob. Agents Chemother. 2017, 61, e01057-17. [Google Scholar] [CrossRef]
- Silva, F.S.; Cunha, S.C.S.; Moraes, V.A.; Leite, J.S.; Ferreira, A.M.R. Refractory Feline Sporotrichosis: A Comparative Analysis on the Clinical, Histopathological, and Cytopathological Aspects. Pesq. Vet. Bras. 2022, 42, e06923. [Google Scholar] [CrossRef]
- Miranda, L.H.M.; Santiago, M.D.A.; Schubach, T.M.P.; Morgado, F.N.; Pereira, S.A.; Oliveira, R.D.V.C.D.; Conceição-Silva, F. Severe Feline Sporotrichosis Associated with an Increased Population of CD8low Cells and a Decrease in CD4+ Cells. Med. Myco. 2015, 54, myv079. [Google Scholar] [CrossRef]
- Du, B.; Meenu, M.; Liu, H.; Xu, B. A Concise Review on the Molecular Structure and Function Relationship of β-Glucan. IJMS 2019, 20, 4032. [Google Scholar] [CrossRef] [PubMed]
- Chan, G.C.F.; Chan, W.K.; Sze, D.M.Y. The Effects of β-Glucan on Human Immune and Cancer Cells. J. Hematol. Oncol. 2009, 2, 25. [Google Scholar] [CrossRef] [PubMed]
- Majtan, J.; Jesenak, M. β-Glucans: Multi-Functional Modulator of Wound Healing. Molecules 2018, 23, 806. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.J.; Rezoagli, E.; Major, I.; Rowan, N.J.; Laffey, J.G. β-Glucan Metabolic and Immunomodulatory Properties and Potential for Clinical Application. J. Fungi 2020, 6, 356. [Google Scholar] [CrossRef]
- Barsanti, L.; Vismara, R.; Passarelli, V.; Gualtieri, P. Paramylon (β-1,3-Glucan) Content in Wild Type and WZSL Mutant of Euglena gracilis. Effects of Growth Conditions. J. Appl. Phycol. 2001, 13, 59–65. [Google Scholar] [CrossRef]
- Sugiyama, A.; Hata, S.; Suzuki, K.; Yoshida, E.; Nakano, R.; Mitra, S.; Arashida, R.; Asayama, Y.; Yabuta, Y.; Takeuchi, T. Oral Administration of Paramylon, a β-1,3-D-Glucan Isolated from Euglena gracilis Z Inhibits Development of Atopic Dermatitis-Like Skin Lesions in NC/Nga Mice. J. Vet. Med. Sci. 2010, 72, 755–763. [Google Scholar] [CrossRef]
- Evans, M.; Falcone, P.H.; Crowley, D.C.; Sulley, A.M.; Campbell, M.; Zakaria, N.; Lasrado, J.A.; Fritz, E.P.; Herrlinger, K.A. Effect of a Euglena gracilis Fermentate on Immune Function in Healthy, Active Adults: A Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients 2019, 11, 2926. [Google Scholar] [CrossRef]
- Borosky, J.C.; Costa, M.C.R.; Oliveira, C.A.F.; Vetvicka, V. Euglena gracilis as an Adjuvant for the Treatment of a Dog with Chronic Kidney Disease—Case Report. Arq. Bras. Med. Vet. Zootec. 2023, 75, 1065–1068. [Google Scholar] [CrossRef]
- de Souza Theodoro, S.; Gonçalves Tozato, M.E.; Warde Luis, L.; Goloni, C.; Bassi Scarpim, L.; Bortolo, M.; Cavalieri Carciofi, A. β-Glucans from Euglena gracilis or Saccharomyces Cerevisiae Effects on Immunity and Inflammatory Parameters in Dogs. PLoS ONE 2024, 19, e0304833. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.A.; Jewell, D.E.; Ephraim, E. Feeding Cats with Chronic Kidney Disease Food Supplemented with Betaine and Prebiotics Increases Total Body Mass and Reduces Uremic Toxins. PLoS ONE 2022, 17, e0268624. [Google Scholar] [CrossRef]
- Amaral, A.R.; Risolia, L.W.; Rentas, M.F.; Marchi, P.H.; Balieiro, J.C.D.C.; Vendramini, T.H.A.; Brunetto, M.A. Translating Human and Animal Model Studies to Dogs’ and Cats’ Veterinary Care: Beta-Glucans Application for Skin Disease, Osteoarthritis, and Inflammatory Bowel Disease Management. Microorganisms 2024, 12, 1071. [Google Scholar] [CrossRef]
- Lawrence, Y.A.; Steiner, J.M. Laboratory Evaluation of the Liver. Vet. Clin. N. Am. Small Anim. Pract. 2017, 47, 539–553. [Google Scholar] [CrossRef]
- Rossi, C.N.; Odaguiri, J.; Larsson, C.E. Retrospective assessment of the treatment of sporotrichosis in cats and dogs using itraconazole. Acta Sci. Vet. 2013, 41, 1–5. [Google Scholar]
- Goodridge, H.S.; Wolf, A.J.; Underhill, D.M. β-glucan Recognition by the Innate Immune System. Immunol. Rev. 2009, 230, 38–50. [Google Scholar] [CrossRef]
- Dalonso, N.; Goldman, G.H.; Gern, R.M.M. β-(1→3),(1→6)-Glucans: Medicinal Activities, Characterization, Biosynthesis and New Horizons. Appl. Microbiol. Biotechnol. 2015, 99, 7893–7906. [Google Scholar] [CrossRef]
- Yamamoto, F.Y.; Yin, F.; Rossi, W.; Hume, M.; Gatlin, D.M. β-1,3 Glucan Derived from Euglena gracilis and AlgamuneTM Enhances Innate Immune Responses of Red Drum (Sciaenops ocellatus L.). Fish Shellfish Immunol. 2018, 77, 273–279. [Google Scholar] [CrossRef]
- Ciecierska, A.; Drywień, M.E.; Hamulka, J.; Sadkowski, T. Nutraceutical Functions of Beta-Glucans in Human Nutrition. Rocz. Panstw. Zakl. Hig. 2019, 70, 315–324. [Google Scholar] [CrossRef]
- Beyazit, A.; Inceboz, T.; Over, L. Contribution to one world, one health: A dog with demodicosis. Turk. Parazitoloji Derg. 2010, 34, 68–71. [Google Scholar]
- Guterres, K.A.; De Matos, C.B.; Osório, L.G.; Schuch, I.D.; Cleff, M.B. The Use of (1–3) β-Glucan Along with Itraconazole Against Canine Refractory Sporotrichosis. Mycopathologia 2014, 177, 217–221. [Google Scholar] [CrossRef]
- Kim, Y.-H.; Jeong, Y.; An, J.-H.; Ahn, J.-O.; Chung, J.-Y. Application of a Synbio-Glucan Functional Spray for Canine Atopic Dermatitis. J. Vet. Clin. 2023, 40, 8–15. [Google Scholar] [CrossRef]
- Micháľová, A.; Micháľ, M.; Fialkovičová, M. Combination of Beta Glucan, Honey and Chlorhexidine in the Wound Management in a Cat a Case Report. Folia Vet. 2019, 63, 70–77. [Google Scholar] [CrossRef]
- Chaves, A.R.; De Campos, M.P.; Barros, M.B.L.; Do Carmo, C.N.; Gremião, I.D.F.; Pereira, S.A.; Schubach, T.M.P. Treatment Abandonment in Feline Sporotrichosis—Study of 147 Cases. Zoonoses Public Health 2013, 60, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Bashir, K.M.; Choi, J.-S. Clinical and Physiological Perspectives of β-Glucans: The Past, Present, and Future. Int. J. Mol. Sci. 2017, 18, 1906. [Google Scholar] [CrossRef]
- Chen, S.; Guan, X.; Yong, T.; Gao, X.; Xiao, C.; Xie, Y.; Chen, D.; Hu, H.; Wu, Q. Structural Characterization and Hepatoprotective Activity of an Acidic Polysaccharide from Ganoderma Lucidum. Food Chem. X 2022, 13, 100204. [Google Scholar] [CrossRef]
- Sugiyama, A.; Suzuki, K.; Mitra, S.; Arashida, R.; Yoshida, E.; Nakano, R.; Yabuta, Y.; Takeuchi, T. Hepatoprotective Effects of Paramylon, a BETA-1,3-D-Glucan Isolated from Euglena gracilis Z, on Acute Liver Injury Induced by Carbon Tetrachloride in Rats. J. Vet. Med. Sci. 2009, 71, 885–890. [Google Scholar] [CrossRef]
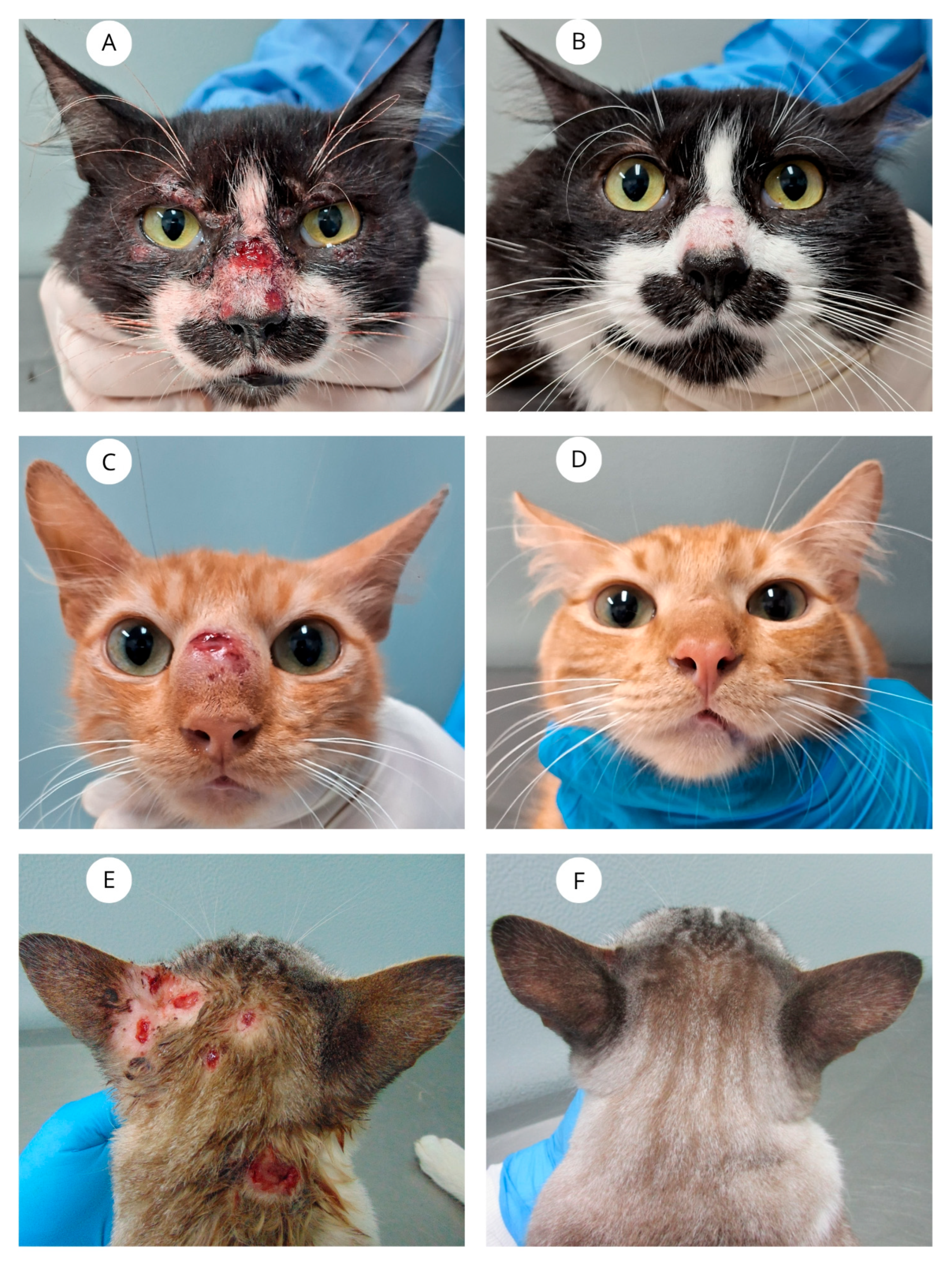
| Cats | Sex | Neutered | Overall Condition | Skin Lesions Distribution | Skin Lesions on the Nasal Region | Mucosal Lesions | Respiratory Signs | Adverse Drug Reactions | Time to Clinical Cure (weeks) | Outcomes |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | M | No | Good | L1 | No | Nasal | Yes | No | 13 | Cure |
| 2 | M | No | Good | L3 | No | Nasal | Yes | No | 9 | Cure |
| 3 | M | No | Fair | L3 | Yes | Nasal, Conjuctival | Yes | No | 16 | Cure |
| 4 | M | No | Good | L2 | Yes | No lesion | No | No | N/A | Abandonment |
| 5 | M | No | Good | L2 | Yes | Nasal | No | No | 10 | Cure |
| 6 | M | No | Fair | L3 | Yes | Nasal | Yes | No | N/A | Failure |
| 7 | M | Yes | Good | L3 | Yes | Nasal, Conjuctival | Yes | No | N/A | Abandonment |
| 8 | M | No | Good | L2 | Yes | Nasal | Yes | No | N/A | Abandonment |
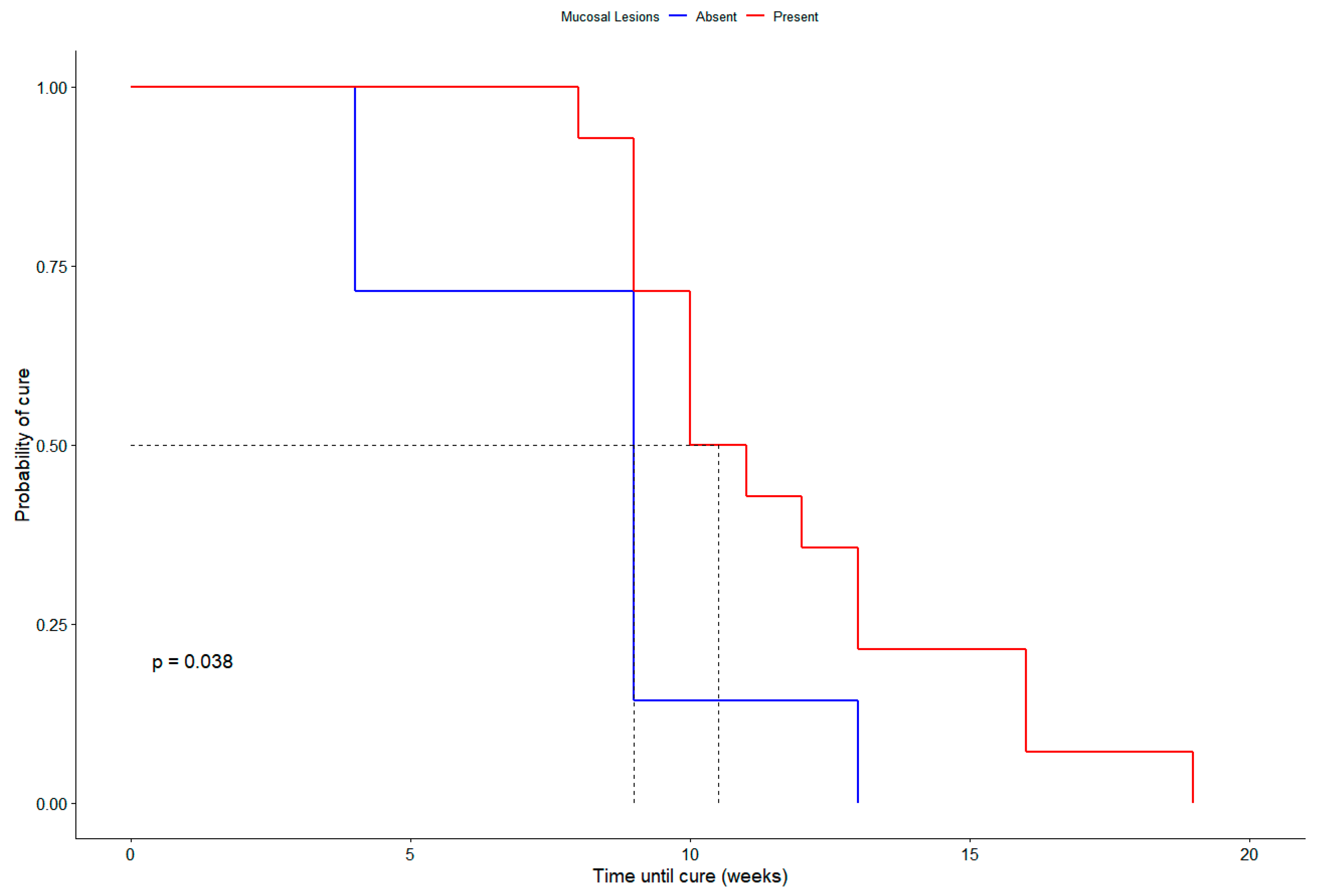
| 9 | M | Yes | Good | L3 | No | Conjuctival | Yes | No | 8 | Cure |
| 10 | M | No | Poor | L3 | Yes | Nasal | Yes | No | 19 | Cure |
| 11 | M | No | Fair | L3 | Yes | Nasal | Yes | No | N/A | Failure |
| 12 | F | No | Good | L2 | No | No lesion | No | No | 4 | Cure |
| 13 | M | Yes | Good | L3 | Yes | Nasal, Conjuctival | Yes | No | 9 | Cure |
| 14 | M | Yes | Good | L3 | No | No lesion | No | No | 9 | Cure |
| 15 | M | Yes | Fair | L1 | No | Nasal | No | No | 9 | Cure |
| 16 | M | Yes | Good | L2 | No | Nasal, Conjuctival | Yes | No | 12 | Cure |
| 17 | M | No | Good | L1 | No | No lesion | No | No | 9 | Cure |
| 18 | M | No | Fair | L3 | Yes | Nasal | Yes | No | N/A | Failure |
| 19 | M | No | Good | L1 | No | No lesion | No | No | 9 | Cure |
| 20 | M | Yes | Good | L3 | Yes | Nasal | Yes | Yes * | 10 | Cure |
| 21 | M | Yes | Good | L1 | No | No lesion | Yes | No | N/A | Failure |
| 22 | M | No | Good | L1 | No | No lesion | No | No | 13 | Cure |
| 23 | F | Yes | Good | L1 | Yes | Nasal | Yes | No | 10 | Cure |
| 24 | M | Yes | Good | L2 | No | No lesion | No | No | N/A | Failure |
| 25 | M | No | Fair | L1 | Yes | Nasal | Yes | No | 13 | Cure |
| 26 | M | No | Fair | L3 | Yes | Nasal | No | No | 16 | Cure |
| 27 | F | Yes | Good | L1 | No | No lesion | No | Yes ** | 9 | Cure |
| 28 | M | Yes | Good | L1 | No | No lesion | No | No | 4 | Cure |
| 29 | F | Yes | Good | L1 | Yes | Nasal | Yes | No | 11 | Cure |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chacon, A.F.P.; Figueiredo, A.B.F.; Boechat, J.S.; Reis, E.G.; Honorato, C.C.d.S.; Corrêa, M.L.; Pereira, S.A.; Gremião, I.D.F. Prospective Uncontrolled Interventional Study of Itraconazole and β-Glucans (Euglena gracilis) to Assess Safeness and Clinical Effectiveness in Cats with Cutaneous and Mucosal Sporotrichosis. Vet. Sci. 2025, 12, 830. https://doi.org/10.3390/vetsci12090830
Chacon AFP, Figueiredo ABF, Boechat JS, Reis EG, Honorato CCdS, Corrêa ML, Pereira SA, Gremião IDF. Prospective Uncontrolled Interventional Study of Itraconazole and β-Glucans (Euglena gracilis) to Assess Safeness and Clinical Effectiveness in Cats with Cutaneous and Mucosal Sporotrichosis. Veterinary Sciences. 2025; 12(9):830. https://doi.org/10.3390/vetsci12090830
Chicago/Turabian StyleChacon, André Felipe Pedrazzi, Anna Barreto Fernandes Figueiredo, Jéssica Sepulveda Boechat, Erica Guerino Reis, Cindy Caroline dos Santos Honorato, Maria Lopes Corrêa, Sandro Antonio Pereira, and Isabella Dib Ferreira Gremião. 2025. "Prospective Uncontrolled Interventional Study of Itraconazole and β-Glucans (Euglena gracilis) to Assess Safeness and Clinical Effectiveness in Cats with Cutaneous and Mucosal Sporotrichosis" Veterinary Sciences 12, no. 9: 830. https://doi.org/10.3390/vetsci12090830
APA StyleChacon, A. F. P., Figueiredo, A. B. F., Boechat, J. S., Reis, E. G., Honorato, C. C. d. S., Corrêa, M. L., Pereira, S. A., & Gremião, I. D. F. (2025). Prospective Uncontrolled Interventional Study of Itraconazole and β-Glucans (Euglena gracilis) to Assess Safeness and Clinical Effectiveness in Cats with Cutaneous and Mucosal Sporotrichosis. Veterinary Sciences, 12(9), 830. https://doi.org/10.3390/vetsci12090830









